Abstract
Dietary restriction (DR) and mutations in nutrient signalling pathways can extend healthy lifespan in diverse organisms. Studying the interaction between these interventions should reveal mechanisms of ageing, but has yielded some apparently contradictory results. A multidimensional representation of nutrition, called the geometric framework, can better describe the responses of lifespan and other traits including metabolism, and can reconcile these apparent contradictions. We provide examples showing that it is more informative to analyse of DR in terms of dietary balance and that dietary optimisation for lifespan is critical for studies examining the biology of ageing and other traits.
Keywords: dietary restriction, calorie restriction, dietary balance, geometric framework, nutrition, ageing, lifespan
Graphical Abstract
Introduction
In 1935, it was first reported that limiting the food intake of rats extended their lifespan (McCay et al., 1935). There has since been a substantial body of research on calorie (CR) or dietary (DR) restriction. DR is a moderate reduction of food intake that increases healthy lifespan and is thus distinct from severe reductions that shorten lifespan through malnutrition. DR can extend the lifespan of already healthy animals, implying that it does not merely rescue illness caused by over-consumption of an inappropriate diet. DR increases lifespan in diverse organisms, from yeast to primates (Weindruch and Walford, 1988; Jiang et al., 2000; Mair and Dillin, 2008; Colman et al., 2009) and may thus act through evolutionarily conserved mechanisms meaning that studies of short-lived model organisms may point to interventions that ameliorate the negative effects of human ageing.
Recently, this traditional view of DR has been challenged. First, studies in insects and rodents have shown that altering the balance of dietary components an organism ingests, without reducing its overall food intake, can increase lifespan (Grandison et al., 2009a; Lee et al., 2008; Skorupa et al., 2008; Orentreich et al., 1993; Richie, Jr. et al., 1994; Zimmerman et al., 2003; Miller et al., 2005; Maklakov et al., 2008; Fanson et al., 2009; Simpson and Raubenheimer, 2009), implying that specific nutrients, or their ratios, may be important in determining longevity. Second, the genotype of the organism can affect the response to DR, in some cases apparently with complete elimination of lifespan-extension (Greer and Brunet, 2009; Grandison et al., 2009a; Bartke et al., 2001; Bonkowski et al., 2006; Liao et al., 2010).
These new findings clearly challenge the simple idea that moderately reduced intake of a fixed type of food predictably extends lifespan. However, the new observations could have a general explanation, if examined and modeled in a quantitative framework that captures these complexities.
The Geometric Framework for nutrition (Simpson and Raubenheimer, 2009) enables different nutritional implementations of DR and the responses of different genotypes to them to be described and compared.
Capturing nutritional complexity
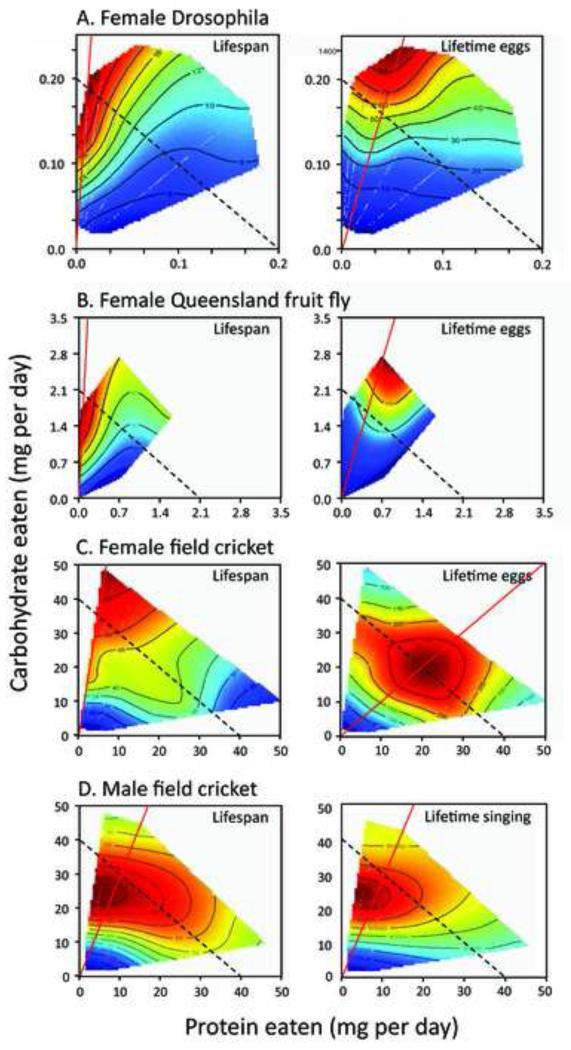
The Geometric Framework (GF) (Simpson and Raubenheimer, 1993; Raubenheimer and Simpson, 1993; Simpson and Raubenheimer, 2007) considers nutrition as an n-dimensional space in which the n components of any diet are represented by separate axes. Responses of individuals are superimposed on this n-dimensional nutritional space, by plotting response surfaces. These responses could include changes in any quantifiable phenotypic trait such as lifespan, metabolic parameters, egg laying or gene expression. Figure 1 shows several GF plots for how nutrient balance affects lifespan and reproductive output for different insects. These pairs of plots allow us to compare the different traits and demonstrate that the nutrient intake balance that is best for female lifespan is not the same as that for egg laying. Diets can be represented on the GF plot as lines (‘rails’) radiating from the origin, such as the red lines in Figure 1 or the blue diagonal line in Figure 2A. The distance along the rail from the origin reflects the amount of the particular diet eaten, and the distance along the respective axes, the amount of each nutrient that has been eaten. Because all possible levels of intake of any combination of nutrients can be represented in this intake space, the approach makes it possible to combine data on dietary intake from different experiments, to describe how a given phenotypic response variable is affected (eg see (Lee et al., 2008)).
Figure 1.
The effects of protein and carbohydrate intake on response surfaces for lifespan and reproductive output of three insect species.
Phenotypic response surfaces for longevity and reproductive output (eggs laid or in the case of male crickets, time spent singing to attract mates) are plotted for three insect species: the fruitfly Drosophila melanogaster, the Queensland fruitfly Bactrocera tryoni and the field cricket Teleogryllus commodus. Insects were given ad libitum access to one of 28 (Drosophila and Bactrocera) or 24 (Teleogryllus) diets varying protein to carbohydrate (P:C) ratio. Surface values increase from dark blue to dark red. Unbroken red lines show the dietary P:C that maximized the response variable; dotted lines indicate isocaloric intakes. Data re-plotted from (Lee et al., 2008) (Drosophila), (Maklakov et al., 2008) (field crickets), and (Fanson et al., 2009) (Queensland fruit fly). Figure from (Simpson and Raubenheimer, 2012).
Figure 2.
Effect of nutritional balance on lifespan.
Response surfaces for median lifespans of wild-type (A) and mutant flies (E) mapped onto intake estimates for carbohydrate and protein. Diets can be represented on the surface as lines (‘rails’) and consumption represented by distance from the origin. Thus, as an animal consumes a diet, its position in nutrient space moves along a dietary rail away from the origin.
(A) Fitted lifespan response surface for wild-type Drosophila. Data from 22 diets in 5 published studies (adapted from (Lee et al., 2008)). The surface for the mutant (E) was created by transforming the data in (A) such that the lifespan peak is right-shifted to higher protein concentrations. The transformation applied was as follows:
LM = LW - 3(LW.R/100) where:
LM = median lifespan of mutant; LW = median lifespan of wild-type (Figure 1A); R = ratio of carbohydrate to protein in the diet.
The surface was then normalised to the same range of lifespans as in wild-type flies and food intake left unchanged. The effect is that for a given level of carbohydrate intake there is a lifespan shortening cost for high dietary carbohydrate: protein ratios. This could be, for example, due to increased deposition of body fat. In addition to this cost, the mutants also gain a lifespan advantage with lower dietary carbohydrate: protein ratios. Together, these cause the lifespan peak to be right-shifted along the protein axis. Variation in the propensity to lay down body fat on high carbohydrate: protein diets is known to occur in insects (see (Warbrick-Smith et al., 2006)). Drosophila genotypes too vary in body fat; fatness confers advantages in some environments through resistance to starvation (e.g. (Ballard et al., 2008), and refs therein) and when energy is limited on low carbohydrate: protein diets.
(B-D; F-H) Standard representation of median lifespan responses of flies to three different dietary restriction (DR) protocols. These plots are generated from the data in the GF plots. Each of the three differently coloured straight lines transecting the GF plots (Figures 2A & E) represents a range of nutrient intake levels for animals treated with one of three different DR interventions. Each of these nutrient intake lines is used as a different x-axis for a traditional DR plot. The lifespan responses to each DR treatment are the heat map values that lie along each of the coloured lines. Plotted lifespan values are colour matched to the nutrient intake lines for each DR intervention on the GF plot. (B-D; F-G) By restricting access to protein and/or carbohydrate, lifespan shows at least part of a typical ‘tent’-shaped response to DR: as nutrient(s) are restricted, lifespan increases to peak at intermediate concentrations, whereupon it decreases again with further restriction due to malnutrition.
(H) For the same nutrient intakes as wild types in Figure 2D, the mutation modifies the lifespan response such that no variation is detected.
Using the GF to represent different implementations of DR
Often more than one implementations of DR can yield an increase in lifespan (Weindruch and Walford, 1988; Piper and Bartke, 2008; Greer and Brunet, 2009). Traditional DR experiments usually either restrict access to a diet of constant composition, or provide diets whose composition varies, with or without maintenance of a constant intake of calories.
The most common implementation of DR is to restrict the intake of a diet of fixed composition, either by providing limited food (McCay et al., 1935; Yu et al., 1982; Weindruch and Walford, 1982; Weindruch and Walford, 1988) or diluting a diet with water or another bulking agent (Iwasaki et al., 1988; Chapman and Partridge, 1996), to an extent that any compensatory feeding fails to maintain nutrient intake. This type of DR is represented by the line radiating from the origin in Figure 2A, and the animals in the DR group are maintained at a point closer to the origin than are the ad libitum-fed controls.
DR can also be implemented by varying dietary composition. For instance, the protein component of the diet can be varied at a fixed level of carbohydrate, by replacing protein with water or another bulking agent, thereby also modifying the carbohydrate: protein ratio of the diets (Bass et al., 2007; Grandison et al., 2009b; Chippindale et al., 1993; Tatar, 2007). If animals consumed the same volume of these diets, then the carbohydrate intake between treatments would be constant but the protein intake would vary, represented by a horizontal line in Figure 2A (two such lines are shown – red at a relatively high level of carbohydrate intake, and ochre at a relatively low level). Animals maintained on diets with different nutrient balances could also eat different volumes of food, in which case the line joining points of intake will not be horizontal. Whatever the lines may be, the GF enables different DR interventions to be described and compared in a single model.
Traditionally, the responses of lifespan and other traits to DR have been analysed representing nutrition as a single variable which is graphically depicted on an x-axis. The coloured lines transecting the GF plot in Figure 2A would thus become the single x-axis for each DR intervention: the blue line represents restricted access to a diet containing a fixed ratio of nutrients (Figure 2B), and the horizontal lines reflect fixed ingestion of diets varying in protein concentration at either a high (red line; Figure 2C) or low (ochre; Figure 2D) level of carbohydrate intake. The lifespan values in Figures 2B-2D are read from the heat-map values in Figure 2A that fall on these lines. The lifespan of wild-type animals exhibits a ‘tent’-shaped DR response to varying intake of a diet of fixed ratio of carbohydrate to protein (Figure 2B). Reduction in intake of protein at fixed levels of carbohydrate intake (Figures 2C & 2D) also results in increased lifespan. Thus all these dietary interventions can yield lifespan responses that meet the generally accepted criteria for DR.
However, other transects of the surface in Figure 2A give outcomes that are not consistent with DR. For example, a vertical section, in which protein intake is fixed while carbohydrate intake is varied, produces a monotonic increase in lifespan with increasing carbohydrate intake. In addition, it can be concluded from Figure 2A and Figure 1 that calorie intake per se does not predict lifespan. Since protein and carbohydrate yield similar amounts of energy per gram ingested, isocaloric intakes fall along diagonal lines with slope -1 (dashed lines in Figure 1). Rather than lifespan being similar for a given calorie intake, independent of the protein to carbohydrate ratio, isocaloric intake lines yield substantial changes in lifespan: it is not calories that matter, but rather the ratio of protein to carbohydrate ingested.
It can therefore be concluded that experimentation and representation of alterations in diet using a single nutritional axis in the nutritional space represented in Figure 2A can yield a variety of conclusions about the efficacy of DR in extending lifespan. The reason is that the response to alterations in intake of one nutrient depends upon the level of intake of the other, a situation that cannot be visualised using a single nutritional axis.
Comparing the response of lifespan to nutrient balance in different genotypes
Usually the effect of genotype on the response of lifespan to DR has been assessed using single nutritional axes of the type illustrated in Figure 2B-D. For instance, using this approach, single gene mutations in the insulin/insulin-like growth factor, target of rapamycin and growth hormone pathways have been shown to alter the response of lifespan to DR in worms, flies and mice (Clancy et al., 2002; Johnson et al., 1990; Panowski et al., 2007; Bishop and Guarente, 2007; Gronke et al., 2010; Bartke et al., 2001; Bonkowski et al., 2006; Grandison et al., 2009a). Although these studies have pointed to mechanisms mediating phenotypic responses to diet, the use of a single nutritional axis can yield a less than full description. The Geometric Framework can capture the phenotypic responses of different genotypes in full nutritional space. To illustrate this, the data in Figure 2A can be transformed by introducing a hypothetical mutant genotype. Maximum attainable lifespan is no different for mutant and control animals, but the relationship of lifespan to nutrient intake is altered. For a given carbohydrate intake, the mutant animal suffers lifespan-shortening obesity as protein intake declines from the optimum for the wild type, and a lifespan-extending metabolic advantage as protein intake increases from the wild type optimum (Figure 2E). (Details of the data transformation used to derive the hypothetical mutant’s response surface are provided in the caption to Figure 2). The lifespan response surface of the mutant is therefore right-shifted along the protein axis (Figure 2E), for reasons that are unrelated to the process of aging.
The modified responses of a mutant to DR interventions (Figures 2B-D) can be visualised using the dashed coloured lines transecting the GF plot (Figure 2E) as single x-axes (Figures 2F-2H). In contrast to the lifespan responses for the wild-type, we now see a typical tent-shaped curve both when varying intake of a diet of fixed carbohydrate: protein ratio (Figure 2F) as well as when protein intake alone is varied at a high level of carbohydrate intake (Figure 2G). In contrast, varying protein intake at a fixed, low, carbohydrate intake yields no lifespan response for the mutant (Figure 2H), unlike the wild type (Figure 2D).
Interpreting interactions between diet and longevity mutations
To understand the molecular mechanisms of DR, it has been common practice to use single nutritional axes to determine how candidate mutations modify the shape of the responses to DR (illustrated in Figure 3A-C; redrawn from (Mair and Dillin, 2008)), assigning the mutation into one of three categories.
Figure 3.
Mechanistic interpretations of how a gene mutation may interact with traditional DR interventions (redrawn from (Mair and Dillin, 2008)).
(A) Independent: a mutant that is longer lived than the wild type for all levels of nutrient intake, including at the lifespan peak, is thought to extend life by a mechanism independent of DR. This is because the processes required to extend life by DR are assumed to be maximized in wild types at the point of the lifespan peak, meaning any further extension caused by the mutation must be due to additional, unrelated, processes.
(B) DR Mimetic: a mutation that mimics DR is expected to right-shift all lifespan values to higher levels of food intake. Thus, the organism is more sensitive to malnutrition at low food concentrations and less sensitive to the lifespan shortening effects of high food. Importantly, because the mutation interacts with the pathways employed by DR, combining both interventions does not extend lifespan beyond the peak level achievable with DR alone.
(C) Master Regulator: this mutation does not extend life beyond the maximum achievable by DR. However, mutating this gene completely blocks any lifespan response to DR, fixing lifespan either at a constitutively high (depicted) or low level.
(D-F) Comparison of wild type and mutant flies to each of the three DR interventions illustrated in Figure 2. According to the definitions above, these modifications can be interpreted as the mutation being independent of (F), a mimetic of (G) or a master regular of (H) the mechanisms for lifespan extension by DR (see similarities to Figure 3A-C). An alternative explanation, gained from the GF perspective (Figure 2A & E), is that the mutation alters the ability of flies to cope with changes in nutrient balance. Because the maximum attainable lifespan across all nutrient combinations is unchanged between the genotypes, ageing may not be altered.
First, the mutation can extend life through mechanisms apparently independent of those mediating the response to DR (Independent; Figure 3A). At the intake point where wild-type lifespan peaks, the mechanisms of DR are assumed to be fully activated. The greater lifespan increase in the mutant must therefore employ additional pathway(s). Moreover, the slope of lifespan decline with increasing food intake is similar, suggesting that the mechanisms for the DR response are unaffected. An example is the response of age1 mutant worms to bacterial dilution (Johnson et al., 1990).
Second, the mutation could alter mechanisms mediating the responses to DR, causing the mutant to appear already partially diet-restricted (DR mimetic; Figure 3B). The lifespan response curve of mutant animals is right-shifted on the x-axis compared with wild types. Thus, the mutants are shorter-lived at lower food intakes and are longer lived at higher food intakes, with similar peak lifespans. DR and the mutation could therefore extend lifespan by the same mechanisms, and once they are maximised by DR, the mutant cannot further increase lifespan. An example of this response profile has been observed for the chico1 mutation in Drosophila (Clancy et al., 2002).
Third, the mutation could extend life by the same mechanisms as DR and hence completely block the response of lifespan (Master Regulator; Figure 3C; DR (Mair and Dillin, 2008)). In this example, the mutant achieves the same longevity at all food intakes as the wild-type at its peak, and could therefore activate the same lifespan extension mechanisms as those for DR. An example has been reported for flies with the insulin-producing cells (median neurosecretory cells) ablated (Broughton et al., 2010). The important feature of a master regulator is that it blocks the response to DR and could thus also shorten lifespan to a constant level for all food intakes. Examples of such master regulators are pha-4 and skn-1 in worms (Panowski et al., 2007; Bishop and Guarente, 2007) .
Apparently undermining this analysis, depending upon exactly how DR is applied, the mutant in Figure 2E could be interpreted as belonging to all three categories. However the Geometric Framework can resolve these three interpretations into a single consistent interpretation as explained below.
Independent of DR by restricting dietary access?
The heatmap values along the blue diagonal line in Figures 2A and 2E represent the responses of lifespan of wild-type and mutant to varying intake of a diet of fixed composition. These lifespan values can be re-plotted and compared on a common x-axis representing carbohydrate and protein intake (Figure 3D). This traditional DR plot shows that the mutant flies are longer lived than wild types on all non-starvation foods, even where lifespan peaks under DR, similar to Figure 3A, and suggesting that the mutation extends lifespan through mechanisms independent of those for DR.
A mimetic of DR by protein restriction?
The horizontal red lines in Figures 2A & 2E represent the lifespans of wild type and mutant animals in which the protein source only is varied. When these are compared on a single x-axis representing protein consumption, the lifespan response curve of the mutant is right-shifted to higher protein intake (Figure 3E), similar to Figure 3B and suggesting that the mutation partially imposes DR on the mutant and so acts as a DR mimetic. This conclusion is supported by other data (Figure 1A), showing that lifespan declines at very low protein to carbohydrate ratios; hence wild type lifespan in Figure 3E is approaching a maximum at its far left end.
A Master regulator of DR by protein restriction?
Finally, a subtle modification to the nutritional context in which single nutrient DR is implemented can result in altogether different lifespan responses. The ochre lines transecting the GF plots (Figures 2A &2E) represent animals subject to DR by protein dilution, but with an approximately 25% lower carbohydrate concentration than those represented by the red lines. Plotting the lifespan values for the two genotypes on a common x-axis (Figure 3F) reveals that the mutant completely blocks lifespan variation in response to DR, and achieves the same longevity as the wild-type at its peak, suggesting that the mutation constitutively activates the same lifespan extension mechanism as that for DR. It would thus be categorised as a ‘master regulator’ of DR (see similarity to Figure 3C).
These three scenarios demonstrate how, when DR/mutant interactions are interpreted without reference to the precise nutritional manipulation applied, opposing interpretations can be reached of the effect of the mutant on the response of lifespan to DR (both independent and master regulator). Even though the three above interventions are classified as DR because they restrict nutrient intake and extend lifespan, each present a different range of nutrient mixtures to the animals, and their effects on lifespan are different. Applying the GF, it becomes clear that the single dimensions of diet (DR) or energy (CR) do not provide an adequate description of diet for the phenotype under study. In our hypothetical example the patterns in lifespan differences between the mutant and wild type animals are explicable primarily in terms of changes in the costs of different macronutrient ratios such that changes in dietary balance represent an environmental risk factor to alter lifespan, without altering the intrinsic rate of ageing. We are thus faced with a situation whereby to understand fully the way in which diet affects longevity, we must properly represent nutritional complexity.
Taming the multidimensionality of nutrition
The scenarios we modelled above considered just two nutritional dimensions, protein and carbohydrate. The gain in explanatory power in moving from one dimension (energy or food) to two (protein and carbohydrate) is substantial. The smoothness of the response surfaces indicates that these two nutritional dimensions, or close correlates of them, are major nutritional determinants of longevity in flies and crickets (Figure 1). Surfaces such as those in Figure 2A, which is based on real data, provide an adequate description of the phenotype, against which the effects of putative molecular mechanisms can then be measured and the life-extending properties of other nutrients and dietary supplements can be understood (Simpson and Raubenheimer, 2009).
This is not to say that other nutrients or food components are not important. For example, protein is a compound axis comprised of twenty amino acid dimensions. Having highlighted a key role for protein in relation to carbohydrate, it then becomes possible to begin to dissect the single and interactive roles of individual amino acids within protein (e.g. (Grandison et al., 2009a)). In a similar way, the effects of modifying other nutrients can also be tested having established optima for dietary carbohydrate and protein. It may, for instance, be sensible in other contexts to include a third nutrient dimension for the other major energy-yielding nutrient, fat (Simpson and Raubenheimer, 2007). For Drosophila, the two main macronutrients in the natural diet are carbohydrates and proteins, with lipids only present in small amounts; but for other species, such as rodents, lipids and their component fatty acids are a significant part of the diet and warrant inclusion in experimental designs. It is possible that the fat content of the diet could alter the effect of carbohydrate and protein or their ratio on lifespan, and testing this would require exploration of a 3-dimensional nutritional environment.
Having established the interactive effects of the major macronutrients on longevity, these can be collapsed at particular ratios into a single dimension before studying the effects of other nutrients. These are illustrated within the GF as nutritional rails (see Figure 1A & E and (Lee et al., 2008)). In this way, the GF can help tame the rampant multidimensionality of nutrition, and provide a methodical basis for undertaking further nutrient interaction studies that will together develop a detailed understanding of the common pathways underlying longevity and ageing. The alternative approach of picking a nutrient or dietary supplement a priori and studying its effects independently of its interactions with other nutrients risks misinterpretation and forgoes the opportunity to study important effects of nutrient balance on longevity and health.
Perspective
The GF offers a conceptual nutritionally explicit framework (Raubenheimer et al., 2009) within which to study the molecular and physiological responses of an organism to its diet, providing a platform for future integrative modelling to capture the full complexity of an organism’s molecular and physiological responses to its environment. The key point is that the network of interactions from genes to metabolic and life-history outcomes needs to be considered in relation to an adequate description of the nutritional phenotype, and that this is not provided when too few dietary treatments are included in an experiment. Here we have illustrated the pitfalls of not employing an adequate description of the nutritional phenotype with an hypothetical study aiming to explore the role of a mutation in longevity. The GF does not resolve the mechanistic complexities underlying gene-by-environment interactions in ageing, but it does offer a tool for handling and exploring these mechanisms.
With respect to lifespan, GF has provided a perspective that can explain some of the controversies in the DR literature. For example, it captures how a mutation or genetic background that alters the responses of lifespan to dietary balance might extend lifespan beyond wild-type levels in one laboratory, but have no effect or even shorten lifespan in another, if slightly different foods are used at the two sites. The GF also explains how small differences in the way DR is implemented in different laboratories can yield dramatically different, and even opposing, interpretations about how the mutation interacts with the mechanisms of DR. Moreover, the GF offers a perspective within which to test the anti-ageing effects of diet, mutations or drugs. If, as in our example, a mutant is shown not to extend life beyond the maximum level achievable by the wild-type, it is reasonable to conclude that the gene or mutation functions not to slow a central mechanism of ageing, but instead modifies an organism’s ability to cope with nutrient imbalance. A true anti-ageing intervention would be expected to extend the life of an organism beyond its healthy maximum, found only when that individual is maintained under nutritional conditions ideal for longevity. Finally, for some species it has been argued that DR does not exist as no degree of food dilution is associated with longer life (eg tephritid flies (Carey et al., 2002), house flies (Cooper et al., 2004) and some species of rotifers (Kirk, 2001)). In these cases, a wider exploration of nutrient space may well find nutrient dilutions that alter dietary balance and increase lifespan (e.g. see Fanson et al., 2009 for reconciliation of results for tephritid flies).
Aside from the discussion about the effects of diet on ageing, examining the mechanisms by which dietary balance affects adult health and lifespan has the potential to improve health in the elderly. Towards this goal, accurate and detailed descriptions of nutritional treatments and their effects on lifespan and health are required.
Highlights.
Diet modifications alter lifespan
The Geometric Framework (GF) can quantify lifespan and other responses in complex nutrient space
Change in dietary balance, not dietary or calorie restriction, alters longevity
The GF can be used to reconcile apparently contradictory longevity data
Acknowledgements
We gratefully acknowledge the following funding: MDWP is a University Research Fellow at UCL; LP is a director of the Max Planck Institute for Biology of Ageing; DR is part-funded by the National Research Centre for Growth and Development, New Zealand; SJS is in receipt of an Australian Research Council Laureate Fellowship.
Reference List
- Ballard JW, Melvin RG, Simpson SJ. Starvation resistance is positively correlated with body lipid proportion in five wild caught Drosophila simulans populations. J Insect Physiol. 2008;54:1371–1376. doi: 10.1016/j.jinsphys.2008.07.009. [DOI] [PubMed] [Google Scholar]
- Bartke A, Wright JC, Mattison JA, Ingram DK, Miller RA, Roth GS. Extending the lifespan of long-lived mice. Nature. 2001;414:412. doi: 10.1038/35106646. [DOI] [PubMed] [Google Scholar]
- Bass TM, Grandison RC, Wong R, Martinez P, Partridge L, Piper MD. Optimization of dietary restriction protocols in Drosophila. J. Gerontol. A Biol. Sci. Med. Sci. 2007;6:1071–1081. doi: 10.1093/gerona/62.10.1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop NA, Guarente L. Two neurons mediate diet-restriction-induced longevity in C. elegans. Nature. 2007;447:545–549. doi: 10.1038/nature05904. [DOI] [PubMed] [Google Scholar]
- Bonkowski MS, Rocha JS, Masternak MM, Al Regaiey KA, Bartke A. Targeted disruption of growth hormone receptor interferes with the beneficial actions of calorie restriction. Proc. Natl. Acad. Sci. U. S. A. 2006;103:7901–7905. doi: 10.1073/pnas.0600161103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broughton SJ, Slack C, Alic N, Metaxakis A, Bass TM, Driege Y, Partridge L. DILP-producing median neurosecretory cells in the Drosophila brain mediate the response of lifespan to nutrition. Aging Cell. 2010 doi: 10.1111/j.1474-9726.2010.00558.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carey JR, Liedo P, Harshman L, Zhang Y, Muller HG, Partridge L, Wang JL. Life history response of Mediterranean fruit flies to dietary restriction. Aging Cell. 2002;1:140–148. doi: 10.1046/j.1474-9728.2002.00019.x. [DOI] [PubMed] [Google Scholar]
- Chapman T, Partridge L. Female fitness in Drosophila melanogaster: an interaction between the effect of nutrition and of encounter rate with males. Proc. R. Soc. Lond B Biol. Sci. 1996;263:755–759. doi: 10.1098/rspb.1996.0113. [DOI] [PubMed] [Google Scholar]
- Chippindale AK, Leroi AM, Kim SB, Rose MR. Phenotypic plasticity and selection in Drosophila life-history evolution. I. Nutrition and the cost of reproduction. J. Evol. Biol. 1993;6:171–193. [Google Scholar]
- Clancy DJ, Gems D, Hafen E, Leevers SJ, Partridge L. Dietary restriction in long-lived dwarf flies. Science. 2002;296:319. doi: 10.1126/science.1069366. [DOI] [PubMed] [Google Scholar]
- Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasley TM, Allison DB, Cruzen C, Simmons HA, Kemnitz JW, Weindruch R. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science. 2009;325:201–204. doi: 10.1126/science.1173635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper TM, Mockett RJ, Sohal BH, Sohal RS, Orr WC. Effect of caloric restriction on life span of the housefly, Musca domestica. FASEB J. 2004;18:1591–1593. doi: 10.1096/fj.03-1464fje. [DOI] [PubMed] [Google Scholar]
- Fanson BG, Weldon CW, Perez-Staples D, Simpson SJ, Taylor PW. Nutrients, not caloric restriction, extend lifespan in Queensland fruit flies (Bactrocera tryoni) Aging Cell. 2009;8:514–523. doi: 10.1111/j.1474-9726.2009.00497.x. [DOI] [PubMed] [Google Scholar]
- Grandison RC, Piper MDW, Partridge L. Amino-acid imbalance explains extension of lifespan by dietary restriction in Drosophila. Nature. 2009a;462:1061–1064. doi: 10.1038/nature08619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grandison RC, Wong R, Bass TM, Partridge L, Piper MDW. Effect of a Standardised Dietary Restriction Protocol on Multiple Laboratory Strains of Drosophila melanogaster. PLoS ONE. 2009b;4:e4067. doi: 10.1371/journal.pone.0004067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greer EL, Brunet A. Different dietary restriction regimens extend lifespan by both independent and overlapping genetic pathways in C. elegans. Aging Cell. 2009;8:113–127. doi: 10.1111/j.1474-9726.2009.00459.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gronke S, Clarke DF, Broughton S, Andrews TD, Partridge L. Molecular evolution and functional characterization of Drosophila insulin-like peptides. PLoS Genet. 2010;6:e1000857. doi: 10.1371/journal.pgen.1000857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwasaki K, Gleiser CA, Masoro EJ, McMahan CA, Seo EJ, Yu BP. Influence of the restriction of individual dietary components on longevity and age-related disease of Fischer rats: the fat component and the mineral component. J. Gerontol. 1988;43:B13–B21. doi: 10.1093/geronj/43.1.b13. [DOI] [PubMed] [Google Scholar]
- Jiang JC, Jaruga E, Repnevskaya MV, Jazwinski SM. An intervention resembling caloric restriction prolongs life span and retards aging in yeast. FASEB J. 2000;14:2135–2137. doi: 10.1096/fj.00-0242fje. [DOI] [PubMed] [Google Scholar]
- Johnson TE, Friedman DB, Foltz N, Fitzpatrick PA, Shoemaker JE. Genetic variants and mutations of Caenorhabditis elegans provide tools for dissecting the aging process. In: Harrison D, editor. Genetic Effects of Aging. Telford; Caldwell, NJ: 1990. pp. 101–126. [Google Scholar]
- Kirk KL. Dietary restriction and aging: comparative tests of evolutionary hypotheses. J. Gerontol. A Biol. Sci. Med. Sci. 2001;56:B123–B129. doi: 10.1093/gerona/56.3.b123. [DOI] [PubMed] [Google Scholar]
- Lee KP, Simpson SJ, Clissold FJ, Brooks R, Ballard JWO, Taylor PW, Soran N, Raubenheimer D. Ageing and reproduction in Drosophila: new insights from nutritional geometry. Proc. Natl. Acad. Sci. U. S. A. 2008;105:2498–2503. doi: 10.1073/pnas.0710787105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao CY, Rikke BA, Johnson TE, Diaz V, Nelson JF. Genetic variation in the murine lifespan response to dietary restriction: from life extension to life shortening. Aging Cell. 2010;9:92–95. doi: 10.1111/j.1474-9726.2009.00533.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mair W, Dillin A. Aging and survival: the genetics of life span extension by dietary restriction. Annu. Rev. Biochem. 2008;77:727–54. doi: 10.1146/annurev.biochem.77.061206.171059. 727-754. [DOI] [PubMed] [Google Scholar]
- Maklakov AA, Simpson SJ, Zajitschek F, Hall MD, Dessmann J, Clissold F, Raubenheimer D, Bonduriansky R, Brooks RC. Sex-specific fitness effects of nutrient intake on reproduction and lifespan. Curr. Biol. 2008;18:1062–1066. doi: 10.1016/j.cub.2008.06.059. [DOI] [PubMed] [Google Scholar]
- McCay CM, Crowell MF, Maynard LA. The effect of retarded growth upon the length of lifespan and upon the ultimate body size. J. Nutr. 1935;10:63–79. [PubMed] [Google Scholar]
- Miller RA, Buehner G, Chang Y, Harper JM, Sigler R, Smith-Wheelock M. Methionine-deficient diet extends mouse lifespan, slows immune and lens aging, alters glucose, T4, IGF-I and insulin levels, and increases hepatocyte MIF levels and stress resistance. Aging Cell. 2005;4:119–125. doi: 10.1111/j.1474-9726.2005.00152.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orentreich N, Matias JR, DeFelice A, Zimmerman JA. Low methionine ingestion by rats extends life span. J. Nutr. 1993;123:269–274. doi: 10.1093/jn/123.2.269. [DOI] [PubMed] [Google Scholar]
- Panowski SH, Wolff S, Aguilaniu H, Durieux J, Dillin A. PHA-4/Foxa mediates diet-restriction-induced longevity of C. elegans. Nature. 2007 doi: 10.1038/nature05837. [DOI] [PubMed] [Google Scholar]
- Piper MD, Bartke A. Diet and aging. Cell Metab. 2008;8:99–104. doi: 10.1016/j.cmet.2008.06.012. [DOI] [PubMed] [Google Scholar]
- Raubenheimer D, Simpson SJ. The Geometry of Compensatory Feeding in the Locust. Anim. Behav. 1993;45:953–964. [Google Scholar]
- Raubenheimer D, Simpson SJ, Mayntz D. Nutrition, ecology and nutritional ecology: toward an intergrated framework. Functional Ecology. 2009;23:4–16. [Google Scholar]
- Richie JP, Jr., Leutzinger Y, Parthasarathy S, Malloy V, Orentreich N, Zimmerman JA. Methionine restriction increases blood glutathione and longevity in F344 rats. FASEB J. 1994;8:1302–1307. doi: 10.1096/fasebj.8.15.8001743. [DOI] [PubMed] [Google Scholar]
- Simpson SJ, Raubenheimer D. A multi-level analysis of feeding behaviour: the geometry of nutritional decisions. Phil. Trans. R. Soc. Lond. B. 1993;342:381–402. [Google Scholar]
- Simpson SJ, Raubenheimer D. Caloric restriction and aging revisited: the need for a geometric analysis of the nutritional bases of aging. J Gerontol. A Biol Sci. Med. Sci. 2007;62:707–713. doi: 10.1093/gerona/62.7.707. [DOI] [PubMed] [Google Scholar]
- Simpson SJ, Raubenheimer D. Macronutrient balance and lifespan. Aging (Albany. NY) 2009;1:875–880. doi: 10.18632/aging.100098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simpson SJ, Raubenheimer D. The Nature of Nutrition. Princeton University Press; Princeton: 2012. [Google Scholar]
- Skorupa DA, Dervisefendic A, Zwiener J, Pletcher SD. Dietary composition specifies consumption, obesity, and lifespan in Drosophila melanogaster. Aging Cell. 2008;7:478–490. doi: 10.1111/j.1474-9726.2008.00400.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tatar M. Diet restriction in Drosophila melanogaster. Design and analysis. Interdiscip. Top. Gerontol. 2007;35:115–36. doi: 10.1159/000096559. 115-136. [DOI] [PubMed] [Google Scholar]
- Warbrick-Smith J, Behmer ST, Lee KP, Raubenheimer D, Simpson SJ. Evolving resistance to obesity in an insect. Proc. Natl. Acad. Sci. U. S. A. 2006;103:14045–14049. doi: 10.1073/pnas.0605225103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weindruch R, Walford RL. Dietary restriction in mice beginning at 1 year of age: effect on life-span and spontaneous cancer incidence. Science. 1982;215:1415–1418. doi: 10.1126/science.7063854. [DOI] [PubMed] [Google Scholar]
- Weindruch R, Walford RL. The retardation of aging and disease by dietary restriction. Thomas; Springfield, Ill.: 1988. [Google Scholar]
- Yu BP, Masoro EJ, Murata I, Bertrand HA, Lynd FT. Life span study of SPF Fischer 344 male rats fed ad libitum or restricted diets: longevity, growth, lean body mass and disease. J Gerontol. 1982;37:130–141. doi: 10.1093/geronj/37.2.130. [DOI] [PubMed] [Google Scholar]
- Zimmerman JA, Malloy V, Krajcik R, Orentreich N. Nutritional control of aging. Exp. Gerontol. 2003;38:47–52. doi: 10.1016/s0531-5565(02)00149-3. [DOI] [PubMed] [Google Scholar]