Abstract
Background
The hepatitis B surface antigen (HBsAg) has been administered over the last 20 years as a parenteral vaccine against the hepatitis B virus (HBV). Despite high seroconversion rates, chronic infection rates are still high worldwide. Orally delivered vaccines provide a practical alternative to injected vaccines, potentially helping poorly responding populations and providing a viable alternative for populations in remote locations. Anamnestic responses are vital to establishing the efficacy of a given vaccine and have been assessed in this study using a plant-based oral delivery platform expressing the hepatitis B surface antigen (HBsAg).
Methods
Long-term immunological memory was assessed in mice injected with a primary dose of Recombivax® and boosted with orally-delivered HBsAg wafers, control wafers, or parenterally-delivered commercial vaccine (Recombivax®).
Results
Mice boosted with HBsAg orally-administered wafers displayed sharp increases in mucosal IgA titers in fecal material and steep increases in serum IgA, whereas mice boosted with Recombivax® showed no detectable levels of IgA in either fecal or serum samples following four boosting treatments. Long-term memory in the orally-treated mice was evidenced by sustained fecal IgA, and serum IgA, IgG, and mIU/mL over one year, while Recombivax®-treated mice displayed sustained serum IgG and mIU/mL. Furthermore, sharp increases in these same antibodies were induced after re-boosting at 47 and 50 weeks post-primary injection.
Conclusions
Orally-delivered vaccines can provide long-term immune responses mucosally and systemically. For sexually-transmitted diseases that can be acquired at mucosal surfaces, such as HBV, an oral delivery platform may provide added protection over a conventional parenterally administered vaccine.
Keywords: Subunit vaccine, mucosal, maize oral vaccine, plant vaccine, bioencapsulation, immunogenicity, supercritical fluid extraction, long-term immune memory, anamnestic response
Introduction
The hepatitis B virus (HBV) remains an important pathogen worldwide, despite the production and administration of effective parenteral vaccines for over 20 years. An estimated 240 million individuals harbor chronic infections around the world, 15-25% of whom will progress to cirrhosis of the liver or liver cancer [1, 2].
The persistence of chronic infections is unsettling considering seroconversion occurs in > 90% of the general population following a three dose treatment [3]. Many individuals forego one or more of the doses, most notably among healthcare workers [4], and there are many poor-responding populations whose seroconversion rates range from 30-64%, such as the elderly, the obese, hemodialysis patients, and chronic kidney disease patients [3]. In addition, it is difficult to reach many parts of the world due to cost and logistics of maintaining a cold chain.
Thermostable oral vaccines encompass characteristics greatly needed for the next generation of vaccines. They provide easy administration at service locations with unreliable refrigeration. They can also induce immunologic responses at mucosal sites, tissues which are points of access for 90% of pathogens. Plant-produced oral vaccine candidates have been displaying these attributes in proof-of-concept studies for over 10 years, but have only recently overcome some important practical limitations, such as high expression of the vaccine in a bioencapsulated form. As an example, the HBsAg subunit candidate was expressed in another plant system, potato, which did induce antibody responses in human volunteers [5]. While providing hope for this overall approach, the immune response was relatively weak and there were other practical limitations such as thermal stability and palatability of using raw potatoes.
In contrast to the potato system, the maize-based expression system, in particular, has shown high expression of antigen [6], strong immunologic responses following oral delivery in mice, sheep, chickens and pigs [6-9], and thermostability across a broad range of temperatures [8, 10]. It is also a very cost-effective production system, with an estimated cost of less than 1 cent per dose for raw materials [11], an attribute which is most relevant in low-income countries where the highest disease burden exists.
A maize-based oral vaccine candidate with all of the above attributes has been developed against HBV using the hepatitis B surface antigen (HBsAg) as the target subunit. This was used to demonstrate the key attributes, above, using the same subunit used in the majority of commercial parenteral HBV vaccines. Commercially available HBV vaccines provide exceptionally long-term immunological memory, lasting 20 or more years in most individuals [12-14]. Although there is some evidence that a potato-based oral primary vaccination can provide a small increase in antibody titers after injected boosting at 32 weeks [15] it is still to be determined whether plant-based oral vaccines can provide a strong and long-lasting anamnestic response. In the present study, we sought to determine whether long-term immunologic memory is established following oral delivery of HBsAg boosting doses in mice over a one year period of time.
Materials and Methods
Maize material
Maize grain containing the HBG construct was generated by backcrossing the initial transgenic line to two inbred parental lines from previous studies [6, 10]. T1 plants were backcrossed independently to two elite inbred lines, to line 16038 over two generations and to line MBS5411 once. These introgressed lines were then crossed to each other to produce hybrid seed, and hybrid seeds were grown to produce hybrid grain. Backcrosses were effectuated in either the field or the greenhouse and all grain was produced under field conditions. Control germ (G909) was obtained from the Grain Processing Corporation (Muscatine, IA).
Seed processing
HBsAg-expressing germ was removed from whole grain by hand degerming, as previously described [6]. Supercritical fluid CO2 extraction (SFE-CO2) was used to remove lipids from HBsAg germ flour at 350 bar, 35-43°C vessel temperature with a peak temperature of 53°C, and from control germ flour at 350 bar, 35-54°C vessel temperature with a peak temperature of 62°C. CO2 flow rate was between 10 and 40 SCFH. All extractions were done in a 5L vessel housed within an SFT-250 (Supercritical Fluid Technologies, Newark, DE) until >90% of lipids extractable by SFE were removed.
Wafer processing
Control wafers used in boosts 1 and 2 contained 2.5g of defatted germ, 0.625g sugar, and 0.75g water while HBsAg wafers contained 2.5g of defatted germ, 0.625g sugar, and 0.625g water. Control wafers used in boosts 3 and 4 contained 2.5g of defatted germ, 1.25g sugar, and 0.9 to 1.25g water while HBsAg wafers contained 2.5g of defatted germ, 1.25g sugar, and 0.50g water. All wafers were formed by hand using circular plastic molds (cat#40115, Decagon/Aqualab, Pullman, WA) and vacuum dried in a VWR 1430 oven (VWR Scientific, Radnor, PA) at 58-62°C, 23.5-24.5” Hg of vacuum pressure and dried until > 90% of the added water was removed, as determined by weight change.
Antigen detection
HBsAg levels in wafers were assayed as described previously [6]. A minimum of 2 wafers per production batch were assayed.
Mouse study
BALB/c inbred mice (Harlan) were randomly assigned to treatments 1 through 3 and housed individually. Ten mice were assigned to each treatment. All treatments were injected intramuscularly with 0.25 μg of Recombivax® (Lot #J001183, Merck, Whitehouse Station, NJ) on day zero, and were boosted with SFE-defatted wafers, Recombivax®, or control wafers (treatments 1-3, respectively), with boosting initiated on 13, 15, 47, and 50 weeks post-primary injection. For each boost, two wafers (each containing 2.5g of germ) were offered per day for three consecutive days or a single 0.25 μg intra-muscular Recombivax® injection was administered on the first day of boosting. HBsAg concentration in wafers is detailed in Table 1. Consumption of wafers was assessed for individual mice 24 hours after wafers were placed in cages for consumption. Mice were individually monitored for consumption of wafersMice were fasted the night before oral boosting to improve ingestion of germ material on day 1 of boosting.
Table 1.
Concentration of HBsAg in wafers, as determined by ELISA
| Sample | Mean [HBsAg] (μg/g wafer) | Std. dev. |
|---|---|---|
| Wafers used for boosts 1 and 2 | 181 | 26 |
| Wafers used for boosts 3 and 4 | 567 | 65 |
| Control wafers | None detected* |
Limit of detection is 15μg/g
Anti-HBsAg antibody detection in mice
Blood samples were collected by submandibular venous puncture every 2 to 4 weeks until 3 weeks following boost #2, then every 10 weeks until boost #3, and finally every 2-4 weeks until the terminal bleed, collected via heart puncture. Serum was collected from blood and stored as previously described [6]. On boosting days, serum was collected a few hours prior to boosting. Fecal material was collected from cages that were cleaned 24 hours prior to sampling, and samples were stored at –20°C. Fecal samples for individual mice were collected prior to the primary injection (pre-immune), twice a week for 6 weeks starting one day prior to boost 1, and twice a week for 5 weeks starting one day prior to boost 3.
Serum anti-HBsAg IgG and IgA were detected using sandwich ELISAs. Plates were coated with rHBsAg (Meridian, cat#R86872-BU), serum samples diluted 1:250, and AP-conjugated anti-mouse IgG (Jackson Immunoresearch Laboratories, cat# 115-055-008) or AP-conjugated anti-mouse IgA (Abcam, cat# 134422) were used to detect IgG and IgA, respectively. Serum samples were further diluted when ODs were beyond the linear range of the assay and titers were transformed using a log-log regression so that all samples contained representative ODs at a 1:250 dilution. The limit of detection was determined to be 1.5-fold above the background (pre-immune) level. Total serum Ig titers (mIU/mL) were assessed using the ETI-AB-AUK PLUS assay kit (Diasorin, Saluggia, Italy) and samples were diluted 1:50 or greater so that titers fell on the linear part of the standard curve (contains the WHO 2nd International Standard). An ELISA was used to assess IgA in 100mg fecal samples resuspended in 1mL of PBS + 1% BSA and further diluted 1:50. Plates were coated with rHBsAg, as above, and sample anti-HBsAg IgA detected using AP-conjugated anti-mouse IgA (Abcam, cat#97232).
Statistical analysis
Data were analyzed using a series of one-way ANOVAs versus treatment for response variables representing fecal IgA, serum IgA, serum IgG, and total Ig (mIU/mL) at the terminal time point. In addition, normalized serum total Ig (a ratio of mIU/mL at week n versus mIU/mL at week 7) was analyzed by one-way ANOVA versus treatment for three different time points. For each ANOVA differences were assessed at a 1% overall significance level using Tukey's HSD procedure. Treatments sharing a group letter for a given assay show insufficient evidence of statistically significant differences. Fecal IgA responses were compared using data collected 51.3 weeks post-primary injection. Serum IgA and IgG titers were compared at terminal bleed, as was the total Ig. Arithmetic means of fecal IgA and serum IgA titers were calculated for each treatment and compared across treatments. Due to somewhat variable serum IgG titers between mice following the primary injection, geometric means were compared between each treatment at the terminal bleed. Total Ig (mIU/mL) values were normalized to pre-boost values due to highly variable responses to the primary injection and geometric means of treatments were compared for the terminal bleed.
Because the study spanned an extended period of time in terms of mouse lifespan, data were excluded from statistical analysis for mice that died prior to the terminal bleed. A single mouse died in the oral HBsAg treatment, 3 mice died in the Recombivax® treatment, and 2 mice died in the oral control treatment.
Results
HBsAg in maize material
In order to produce a strong immunologic response to oral vaccination, it has been shown that milligram levels of antigen are highly effective [6-9]. Therefore, the production of highly expressing HBsAg lines was undertaken by backcrossing transgenic lines into two inbred parent lines and crossing the transgenic lines to produce hybrid grain (see Materials and Methods). In the HBG line, recombinant HBsAg is primarily produced in the embryo (germ) fraction of the seed. In order to increase the concentration of the HBsAg in the final product (wafers), the grain was fractionated and ground into germ flour suitable for oil extraction.
It has also been shown that maize material in which oil has been removed is much more thermostable and immunogenic than full fat maize material [6, 10]. Oil was removed from the germ material by supercritical fluid extraction (SFE) using CO2 and packaged into wafers for administration to mice. Maize material was generated for the mouse trial over two field seasons, the first season generating material for boosts 1 and 2 expressing at a level of 110 μg HBsAg/g grain, and the second season generating material for boosts 3 and 4 at a level of 149 μg/g. Improvements in maize material storage, fractionation, and oil extraction resulted in a 3-fold improvement in final HBsAg wafer concentration, as depicted in Table 1.
Under optimized processing conditions, the material generated from the second season demonstrated expression levels of 149 μg/g in whole seed, 754 μg/g in the full fat germ, 854 μg/g in the SFE-treated germ, and 567 μg/g in the wafer. These numbers are consistent with a 5-fold increase in concentration following fractionation, an additional 13% increase following oil removal, and no loss during wafer formulation (sugar comprises 1/3 of the total wafer weight). Formation of monomers, dimers, and higher order oligomers, as assessed by western blot, were typical in both seasons of previously reported data (data not shown; [6]).
Long-term mucosal immune response using different vaccination routes
The anamnestic response is a key determining factor for the efficacy of vaccines, especially one such as the hepatitis B vaccine which can impart protective antibody titers for decades [12-14]. Previous studies have shown that mice administered a primary injected dose of Recombivax® and orally fed HBsAg maize wafers could elicit a strong immune response following two boosting doses [6, 16]. In the present study, two boosting doses were injected or orally administered at weeks 13 and 15 post-primary injection (ppi), followed by two additional boosting doses at weeks 47 and 50 ppi, providing immunologic data covering more than one half of the mouse model's lifespan.
During oral boosting, mice consumed approximately 70% of the offered wafers. As a result, HBsAg wafer-treated mice consumed an average of 2.4 mg HBsAg during each of the first two boosts, and 8.9 mg HBsAg during the final two boosts. A second treatment consisted of mice injected with 0.25 μg Recombivax® for all four boosts, and a third treatment consisted of mice orally fed control wafers made with commercial germ.
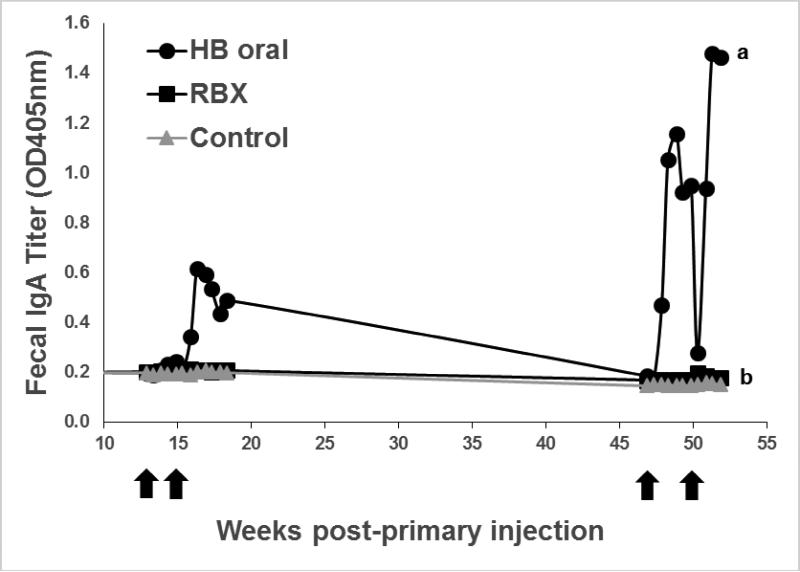
The mucosal immune response was measured by analyzing IgA in fecal pellets. IgA responses in fecal samples displayed a marginal increase after boost 1 (0.04 OD over background), and a 10-fold increase in antibody response over the background-subtracted initial response following boost 2 (0.42 OD; Figure 1). Upon initiation of the third and fourth boosts, an even greater response was observed in the HBsAg wafer-treated mice, increasing in amplitude by >30-fold over the initial response (1.28 OD over background). A single mouse that was orally administered HBsAg showed detectable levels of IgA in samples at 47 weeks ppi, a collection directly preceding boost 3, indicating that persistent antibody production can occur at mucosal sites. These sustained titers can only be attributed to the oral vaccine candidate, as the parenteral and control treatments did not produce detectable IgA titers. In addition, 8 out of 9 mice demonstrated a classic anamnestic response following boost 3, showing a sharp increase in titers. Strikingly, no fecal antibody response was detectable in the Recombivax® treatment over the course of the study.
Figure 1.
Fecal IgA response to oral and injected boosting doses of HBsAg. Mice were injected with 0.25μg of Recombivax® on day 0 and boosted by HBsAg wafer (HB oral), control wafer (Control), or by Recombivax® injection (RBX). A 500-fold dilution of fecal pellets was used and arithmetic mean O.D. for each treatment group was plotted relative to weeks post-primary injection (ppi). Black arrows indicate boosting on weeks 13, 15, 47, and 50 ppi. Different lower-case letters represent significantly different responses (p<0.0001).
Long-term systemic immune response using different vaccination routes
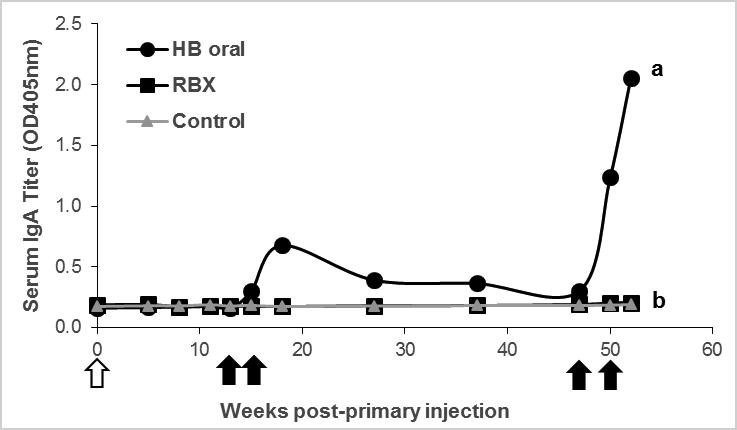
Mice were then tested for their long-term IgA, IgG, and total Ig response in serum in order to analyze systemic immunologic memory as a result of oral versus parenteral vaccine boosting. As can be seen in Figure 2, serum IgA titers paralleled fecal IgA titers for all treatments. After two boosts, 9 out of 10 mice treated with HBsAg wafers showed titers above the limit of detection (LOD), and after four boosts, all mice produced high titers of IgA. Titers seemed to be longer-lived in serum than in fecal such that the number of mice showing titers above the LOD at 27 weeks ppi was 8 out of 10, at 37 weeks ppi was 7 out of 10, and at 47 weeks ppi (pre-boost 3) was 4 out of 10. This is in contrast to a single mouse at 47 weeks ppi showing IgA levels above the LOD in fecal samples.
Figure 2.
Serum IgA mean titers in mice boosted with oral versus injected vaccine. A 250-fold dilution of samples was used and the arithmetic mean O.D. for each treatment group was plotted relative to weeks post-primary injection (ppi). The white arrow indicates primary injection at week 0, and black arrows indicate boosting on weeks 13, 15, 47, and 50 ppi. Different lower-case letters represent significantly different responses (p<0.0001). Abbreviations are as in Figure 1.
Serum IgA levels were not detectable in any of the control or Recombivax®-treated mice, even after four boosting doses, indicating that serum IgA is not efficiently induced upon injection of the vaccine. These results parallel the fecal IgA results in Figure 1.
Serum IgG levels were also assayed in all three treatment groups after diluting the samples 250-fold or greater so that values fell in the linear range of the assay (Figure 3). As expected, the Recombivax® treatment produced a robust IgG response over four boosting doses (4.56 OD) while the control treatment declined after the primary injection (0.46 OD). The HBsAg wafers also produced significant IgG titers after four boosting doses (1.84 OD), and at the terminal bleed geometric mean titers were not significantly different than Recombivax® titers and both were higher than control titers, as expected (Figure 3).
Figure 3.
Serum IgG geometric mean titers as determined by ELISA. A white arrow indicates primary injection of 0.25μg Recombivax® on day 0. Black arrows indicate initiation of oral HBsAg, Recombivax®, or control boosting. Different lower-case letters represent significantly different responses (p<0.0001). Abbreviations are as in Figure 1.
IgG titers were sustained between boosts 2 and 3, and were higher at 47 weeks ppi (pre-boost 3) than directly preceding the first boost in 6 out of 10 mice receiving the HBsAg wafers, in 8 out of 9 mice receiving the Recombivax® injection, and in 1 out of 9 mice receiving the control wafers. The single control mouse showing an increase in titer between boosts 2 and 3 is presumed to have had a delayed antibody response to the primary injection. Therefore, both oral and injected routes of administration were successful in boosting IgG titers.
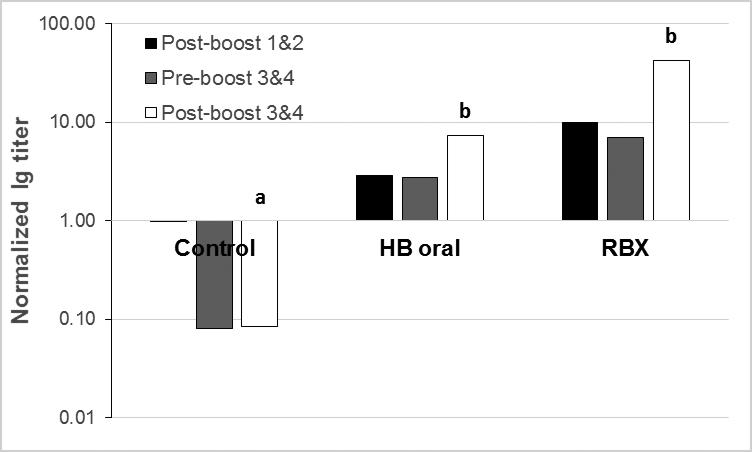
The standard method of demonstrating protective antibodies for hepatitis B is with the WHO International Standard where a titer of ≥10mIU/mL reflects protection [1]. Using this method, samples were diluted 50-fold, or greater, so that values fell on the linear part of the standard curve. Total serum Ig (mIU/mL) showed clear evidence of immunologic memory over one year of oral and parenteral administration. As seen in Figure 4, Ig normalized titers were elevated in HBsAg oral and injected treatments following all boosting doses, with a slight decline in titer between boosts 2 and 3, as expected. Final titers were not significantly different between HBsAg wafer-treated and Recombivax®-treated mice, and both were significantly higher than control mouse titers, which were drastically reduced at 47 weeks ppi (pre-boost 3). The large jump in titers following boosts 3 and 4 is consistent with immunologic memory being present in the HBsAg orally and parenterally treated mice.
Figure 4.
Total serum Ig response, as determined by the WHO 2nd International Standard. Titers of mice boosted with control germ, HBsAg germ (HB oral), or Recombivax (RBX) were normalized to pre-boost titers (mIU/mL at week n/ mIU/mL at week 13) and geometric means calculated for each treatment group. Different lower-case letters represent significantly different responses at the terminal bleed (p=0.0005).
At the conclusion of the study, geometric mean titers using the WHO Standard were 12,755 mIU/mL in the HBsAg oral treatment, and 55,254 mIU/mL in the Recombivax® treatment, but the means were not statistically significantly different. These numbers are very high but not surprising, considering 4 boosting doses were administered in total.
Discussion
We have developed a practical oral vaccination platform that delivers sufficiently high doses of HBsAg to induce a high titer immunological response. In this paper we sought to assess the long-term immunologic response induced by oral vaccination, compare immunologic memory following oral delivery to that generated following parenteral delivery, and provide some insight into the recalcitrance of the system to oral tolerance.
In order for a hepatitis B oral vaccine to be commercially and practically feasible, high levels of HBsAg accumulation must be achieved to deliver a palatable dose. Early attempts to express HBsAg in plant systems provided evidence of orally-induced immunological responses in a human clinical trial, but a single dose required ingestion of 100 g of raw potato expressing 8μg HBsAg/g tissue, rendering the system impractical without increasing the antigen's concentration [5]. The present study has achieved levels of expression > 100 μg/g in whole seed which can be fractionated to produce germ expressing > 700 μg/g. After additional processing and formulation, this can translate to a concentration of > 500 μg/g in a wafer, demonstrating the clear capacity of the system to deliver milligrams of antigen in a small wafer formulation. These concentrations are the highest reported in a plant system to date and could facilitate development of other non-wafer formulations, such as a pill.
Some variability in HBsAg expression was seen from field season to field season, with seed expressing 110 μg/g in the first field season and seed expressing 149 μg/g in the second field season. This variability does not impact the ability to administer a precise dose, as the antigenic defatted maize germ can be blended with control material to deliver a precise dose. Also, the grain used in this study was derived from material generated early in the breeding program and more advanced genetic material should result in higher and more uniform expression levels from harvest to harvest.
Feeding the highly-expressing HBsAg wafers to mice as four boosting doses over one year resulted in a far superior induction of mucosal and systemic HBsAg-specific IgA responses relative to the parenteral vaccine (Figures 1 and 2). Fecal and serum IgA were not detected above background levels in any mice in the control or Recombivax® boost treatments, indicating that mucosal and systemic IgA responses are not efficiently induced upon injection of the vaccine. Previous fecal IgA responses in mice corroborate these data [6]. Serum IgA antibodies, however, seemed to be induced by injection of Recombivax® in the previous study but it has since been determined that the detection antibody for this earlier study bound to IgG as well as IgA, contrary to the manufacturer's specifications. Therefore, earlier reported results reflect a combination of the serum IgA and IgG responses.
One anomaly that has consistently occurred in the fecal IgA data is a dip in titers immediately following oral boosting. This is most likely a result of undigested HBsAg antigen from the wafer interfering with the anti-HBsAg IgA assay. Any available HBsAg will bind to the anti-HBsAg IgA from mouse secretions, thereby reducing the capture of fecal IgA with HBsAg that is bound to the ELISA plates. Consistent with this hypothesis, drastic reduction in titers was only observed in wafer-colored fecal samples. Once the remnants of HBsAg wafers were visibly cleared from fecal samples, titers were once again detectable at high levels.
Significantly higher fecal and serum IgA responses in the orally-treated mice relative to the injected mice may indicate that a mucosal vaccine could protect more effectively against entry of hepatitis B virus at mucosal surfaces, such as via the uro-genital tract where many hepatitis B infections are acquired. There is a precedent for induction of protection at distal mucosal sites. Intranasal immunization with herpes simplex virus (HSV) antigen protected 90% of mice from lethal vaginal challenge [17] and sublingual vaccination with influenza virus protected 100% of mice against lethal intranasal challenge [18]. In the latter study, transgenic mice with defective epithelial membrane IgA transport were used to show that IgA was critical for full protection.
The impact of an IgA response to provide protection is credible since IgA has demonstrated neutralization activity [19] and is the most abundant immunoglobulin produced in the body [20]. In addition, mucosal vaccination has been shown to be superior to parenteral vaccination following HSV vaginal challenge in terms of pathogenesis and long-term protection [21, 22]. Clearly, mucosal vaccination can provide substantial advantages over parenteral vaccination.
Use of wafers combine both sublingual and oral modes of delivery, modes which have been shown to elicit IgA antibodies at all major mucosal surfaces [23]. Therefore it is hypothesized that oral delivery via a wafer formulation could present significant advantages in terms of inducing protection at mucosal surfaces. There is some evidence that injected booster vaccines could induce mucosal antibodies following oral priming [15, 24], but this has not been extensively studied and requires further experimentation in the case of hepatitis B vaccines.
While there is a strong theoretical rationale for believing that a mucosal response may improve protection, the current standard for HBV protection relies on the titer in sera. The evidence that the HBsAg wafers can provide protective titers in serum is suggested by IgG and IgA titers that increased significantly over control mice and were markedly elevated at the termination of the study (Figure 2 and 3). This is further supported using the WHO standard biomarker (Figure 4) in that a titer of > 10mIU/mL is considered protective and the orally-delivered wafers raised these titers to > 12,000 mIU/mL.
This response can be very long-lived, as indicated not only by the sustained Ig titers (measured as mIU/mL), but also by the sustained serum IgA titers in orally-fed mice between boosts 2 and 3, and the sustained fecal IgA titers in one strong-responding mouse. In further support of an anamnestic response, boosts 3 and 4 resulted in sharp increases in antibody production across all assays.
Not only did this study provide evidence for long-term efficacy, but also safety of oral administration of the wafers. The serum and fecal IgA data demonstrated that oral tolerance was not an issue for mice fed with HBsAg wafers over four boosting doses. All mice in this treatment produced increased titers (above the limit of detection) in both fecal and serum samples which would not have been possible if oral tolerance had been an issue. Protective titers, as determined by mIU/mL were also increased in all orally fed mice between the time just preceding the first boost to the time directly following the final boost. No overt signs of allergy following boosting doses were observed in any of the treatments, indicating that the oral maize vaccine system is safe.
Both oral and injected routes of administration were successful in boosting IgG titers and any significant differences between the two routes cannot be established without examining the effect of dosage of each in more detail, which was beyond the scope of this study.
Another pertinent question is whether the oral vaccine could be used as a primary dose and efficiently induce serum IgG, serum IgA, serum Ig, and mucosal IgA antibodies without the need for a parenteral dose. There are reports that plant-produced antigens that are exclusively orally fed in animals can provide 80-100% protection against diseases such as rabies, Newcastle disease virus, and anthrax [7, 9, 25]. The level of protection afforded by priming and boosting with the oral hepatitis B vaccine, however, remains to be determined.
Conclusions
Both oral and injected HBsAg vaccines produced high IgG and mIU/mL titers, while only orally-delivered antigen produced IgA in either fecal or serum samples. The IgG and IgA titers induced by oral delivery were long-lived in a mouse model, persisting more than one half of the mouse's lifespan. These data provide solid support for the continued development of an oral HBV vaccine using the maize production platform.
Highlights.
Mucosal and systemic antibody response in mice when orally fed HBsAg after one year
Mice orally fed HBsAg produce mucosal and systemic IgA whereas injected mice do not
HBsAg accumulation in maize material can reach > 700ug/g
Acknowledgments
This project was supported by NIH grant 2R44AI068239-03A1. The authors would like to thank Erin Fanning and Jenna Kranz for their assistance during the animal studies. They would also like to acknowledge Todd Keener and Erin Miller for their propagation of plant material.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of Interest
The authors declare no conflicts of interest.
References
- 1.CDC A comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the united states. Pediatrics. 2006;118(1):404–404. [PubMed] [Google Scholar]
- 2.Ott JJ, et al. Global epidemiology of hepatitis B virus infection: New estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine. 2012;30(12):2212–2219. doi: 10.1016/j.vaccine.2011.12.116. [DOI] [PubMed] [Google Scholar]
- 3.Hayden CA. An oral vaccine for hepatitis B: challenges, setbacks, and breakthroughs. In: Howard J.A.a.H., E. E., editors. Commercial plant-produced recombinant protein products: Case studies. Springer-Verlab; Heidelberg: 2014. pp. 197–228. [Google Scholar]
- 4.Byrd KK, Lu P-J, Murphy TV. Hepatitis B vaccination coverage among health-care personnel in the United States. Public Health Reports. 2013;128(6):498–509. doi: 10.1177/003335491312800609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thanavala Y, et al. Immunogenicity in humans of an edible vaccine for hepatitis B. Proceedings of the National Academy of Sciences, USA. 2005;102(9):3378–3382. doi: 10.1073/pnas.0409899102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hayden CA, et al. Supercritical fluid extraction provides an enhancement to the immune response for orally-delivered hepatitis B surface antigen. Vaccine. 2014;32(11):1240–1246. doi: 10.1016/j.vaccine.2014.01.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guerrero-Andrade O, et al. Expression of the Newcastle disease virus fusion protein in transgenic maize and immunological studies. Transgenic Research. 2006;15(4):455–463. doi: 10.1007/s11248-006-0017-0. [DOI] [PubMed] [Google Scholar]
- 8.Lamphear BJ, et al. Delivery of subunit vaccines in maize seed. J Control Release. 2002;85(1-3):169–180. doi: 10.1016/S0168-3659(02)00282-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Loza-Rubio E, et al. Induction of a protective immune response to rabies virus in sheep after oral immunization with transgenic maize. Vaccine. 2012;30(27):5551–5556. doi: 10.1016/j.vaccine.2012.06.039. [DOI] [PubMed] [Google Scholar]
- 10.Hayden CA, et al. Production of highly concentrated, heat-stable hepatitis B surface antigen in maize. Plant Biotechnology Journal. 2012;10(8):979–984. doi: 10.1111/j.1467-7652.2012.00727.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Howard JA, Hood E. Bioindustrial and biopharmaceutical products produced in plants. Advances in Agronomy. 2005;85:91–124. [Google Scholar]
- 12.McMahon BJ, et al. Antibody levels and protection after hepatitis B vaccine: results of a 22-year follow-up study and response to a booster dose. Journal of Infectious Diseases. 2009;200(9):1390–1396. doi: 10.1086/606119. [DOI] [PubMed] [Google Scholar]
- 13.Poovorawan Y, et al. Long-term anti-HBs antibody persistence following infant vaccination against hepatitis B and evaluation of anamnestic response: A 20-year follow-up study in Thailand. Human Vaccines & Immunotherapeutics. 2013;9(8):1679. doi: 10.4161/hv.24844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu Q, et al. Comparison of long-term immunogenicity (23 years) of 10 μg and 20 μg doses of hepatitis B vaccine in healthy children. Human Vaccines and Immunotherapeutics. 2012;8(8):1071–1076. doi: 10.4161/hv.20656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Youm J-W, et al. Oral immunogenicity of potato-derived HBsAg middle protein in BALB/c mice. Vaccine. 2007;25(3):577–584. doi: 10.1016/j.vaccine.2006.05.131. [DOI] [PubMed] [Google Scholar]
- 16.Hayden CA, et al. Bioencapsulation of the hepatitis B surface antigen and its use as an effective oral immunogen. Vaccine. 2012;30(19):2937–2942. doi: 10.1016/j.vaccine.2012.02.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cortesi R, et al. Intranasal immunization in mice with non-ionic surfactants vesicles containing HSV immunogens: a preliminary study as possible vaccine against genital herpes. International Journal of Pharmaceutics. 2013;440(2):229–237. doi: 10.1016/j.ijpharm.2012.06.042. [DOI] [PubMed] [Google Scholar]
- 18.Song J-H, et al. Sublingual vaccination with influenza virus protects mice against lethal viral infection. Proceedings of the National Academy of Sciences. 2008;105(5):1644–1649. doi: 10.1073/pnas.0708684105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Devito C, et al. Mucosal and plasma IgA from HIV-exposed seronegative individuals neutralize a primary HIV-1 isolate. AIDS. 2000;14(13):1917–1920. doi: 10.1097/00002030-200009080-00006. [DOI] [PubMed] [Google Scholar]
- 20.Macpherson A, et al. The immune geography of IgA induction and function. Mucosal Immunology. 2008;1(1):11–22. doi: 10.1038/mi.2007.6. [DOI] [PubMed] [Google Scholar]
- 21.Gallichan WS, Rosenthal KL. Long-term immunity and protection against herpes simplex virus type 2 in the murine female genital tract after mucosal but not systemic immunization. Journal of Infectious Diseases. 1998;177(5):1155–1161. doi: 10.1086/515286. [DOI] [PubMed] [Google Scholar]
- 22.Sato A, et al. Vaginal memory T cells induced by intranasal vaccination are critical for protective T cell recruitment and prevention of genital HSV-2 disease. Journal of Virology. 2014;88(23):13699–13708. doi: 10.1128/JVI.02279-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Czerkinsky C, Holmgren J. Mucosal delivery routes for optimal immunization: targeting immunity to the right tissues. In: Kozlowski P, editor. Mucosal Vaccines: Modern Concepts, Strategies, and Challenges. Springer; 2012. pp. 1–18. [DOI] [PubMed] [Google Scholar]
- 24.Sundararajan A, et al. Robust mucosal-homing antibody-secreting B cell responses induced by intramuscular administration of adjuvanted bivalent human norovirus-like particle vaccine. Vaccine. 2014;33(4):568–576. doi: 10.1016/j.vaccine.2014.09.073. [DOI] [PubMed] [Google Scholar]
- 25.Gorantala J, et al. Generation of protective immune response against anthrax by oral immunization with protective antigen plant-based vaccine. Journal of Biotechnology. 2014;176:1–10. doi: 10.1016/j.jbiotec.2014.01.033. [DOI] [PubMed] [Google Scholar]