Abstract
Background. To combat the 2014–2015 Ebola virus disease (EVD) epidemic in West Africa, the World Health Organization urged the rapid evaluation of convalescent whole blood (CWB) and plasma (CP) transfusion therapy. However, the feasibility and likely impacts of broad implementation of transfusions are yet unknown.
Methods. We extended an Ebola virus transmission model published by the Centers for Disease Control and Prevention to include hospital-based convalescent donations and transfusions. Using recent epidemiological estimates for EVD in Liberia and assuming that convalescent transfusions reduce the case-fatality rate to 12.5% (range, 7.5%–17.5%), we projected the impacts of a countrywide ramp-up of transfusion therapy.
Results. Under the 10% case-hospitalization rate estimated for Liberia in September 2014, large-scale CP therapy is expected to save 3586 lives by October 2015 (3.1% mortality reduction; 95% confidence interval [CI], .52%–4.5%). Under a higher 30% hospitalization rate, CP transfusions are expected to save 151 lives (0.9% of the total; 95% CI, .21%–11%).
Conclusions. Transfusion therapy for EVD is a low-cost measure that can potentially save many lives in West Africa but will not measurably influence the prevalence. Under all scenarios considered, CP transfusions are predicted to achieve greater reductions in mortality than CWB.
Keywords: Ebola virus disease, dynamic model, transfusion, polyclonal antibody therapy, convalescent blood
(See the editorial commentary by Colebunders and Cannon on pages 1208–10.)
The ongoing Ebola virus disease (EVD) epidemic in West Africa is the largest ever recorded and has overwhelmed the healthcare systems in Sierra Leone, Liberia, and Guinea [1–4]. There are several promising vaccines and therapeutics under accelerated testing and production [3, 5], but they are not expected to become widely available for many months [6]. One of the only therapeutic methods immediately available and prioritized for evaluation by the World Health Organization (WHO) [7, 8] is blood transfusions from EVD survivors [9–12]. Convalescent patients have Ebola virus–specific antibodies in their serum that potentially facilitate recovery in acutely ill patients. This form of passive polyclonal antibody therapy has been used successfully to treat other infectious diseases [13], including cytomegalovirus infection, hepatitis B, rabies, respiratory syncytial virus infection, vaccinia, and varicella-zoster virus infection [14].
Convalescent transfusions are relatively simple, low cost, and scalable in low-income countries [7]. The evidence for their efficacy is limited but positive, including an 8-patient human study during a 1995 EVD outbreak in the Democratic Republic of the Congo [9], which reported a case-fatality rate of 12.5% under transfusion therapy and supportive care, and multiple animal studies [15]. Even minimally equipped medical settings can perform whole-blood transfusions, which require blood collection, blood grouping, testing for Ebola virus and several transfusion-transmittable diseases (human immunodeficiency virus infection, hepatitis B, and hepatitis C), and administration to critically acute patients [16]. Convalescent patients can donate blood starting 28 days following recovery as either convalescent plasma (CP) or convalescent whole blood (CWB) [16]. While CWB collection requires minimal equipment, CWB donation is physiologically demanding, and donors must wait 12–16 weeks between consecutive donations [16]. CP donation requires medical facilities that can separate the plasma and return the red blood cells [8], but the wait between CP donations is just 2 weeks, which increases the availability of treatments [16]. These facilities are not yet common in Africa [17], but international efforts are underway to bring the technology to hospitals in the affected countries.
The WHO has advocated transfusion of convalescent specimens as the most promising of the available treatments for the ongoing West African epidemic [8, 16, 18]. Although mathematical models have been used extensively to investigate and design intervention measures for EVD [2, 19–25], none have addressed the challenges and impact of broad use of transfusion therapy. Here, we extend an established mathematical modeling framework [2] to assess scalability of transfusions and likely impacts on EVD incidence and mortality. Both the supply (convalescent survivors) and demand (hospitalized patients) are limited by Ebola hospitalization rates, which are perilously low in some regions and have become a major focus of international public health efforts.
METHODS
A dynamic model of Ebola virus transmission was recently used by the Centers for Disease Control and Prevention to project the current epidemic in West Africa [2]. We extended this model to include hospital-based collection and transfusion of CWB and CP. It is a deterministic, compartmental susceptible-exposed-infectious-recovered model that tracks the numbers of susceptible individuals, exposed and incubating cases, symptomatic and infectious patients, convalescent survivors of EVD, and fatalities due to EVD. Infectious patients with EVD are segregated into one of 4 groups: hospitalized in EVD treatment units but not receiving transfusion therapy, hospitalized and receiving transfusion therapy, nonhospitalized but isolated and quarantined in community care centers, and nonhospitalized and nonisolated. Based on data from Liberia, we assumed that isolation rates steadily increased from 8% of all EVD cases on 1 September 2014 [2] to an average of 40% by 1 December 2014. However, unlike the isolation rate, the hospitalization rate remained low throughout this period, owing to shortages of local staff and other logistical challenges in scaling up EVD treatment units. Our baseline scenario assumes that 10% of cases were hospitalized. We also model a United Nations/WHO target in which the isolation and hospitalization rate steadily increase to 70% by 1 December 2014, with 30% assumed to be hospitalized and 40% isolated and quarantined [26].
The model tracks the number of courses of treatment available from CWB and CP donations from EVD survivors and dispenses them immediately to treat hospitalized patients. Convalescent patients are recruited for donation when they are discharged from hospitals. We assume no compensation for donations and that only 50% enter the donation system and <70% return for repeat donation, based on empirical observations [16]. An estimated 43% of patients are eligible to donate whole blood, with the remainder disqualified because of transfusion-transmittable infections, blood type mismatches, and low hemoglobin levels, while 86% of patients are eligible for plasma donation (Supplementary Materials). Following WHO guidelines [16], donations can be made 1 month following discharge and can be repeated multiple times after a delay (2 weeks for CP and approximately 14 weeks for CWB). The per-treatment cost is $75, including supplies for both donation and transfusion [27–29], spot testing for transfusion-transmittable diseases (eg, human immunodeficiency virus infection and malaria) [9], and blood group typing but not including supportive care, training of staff, and equipment for preparation of plasma.
In the absence of transfusions, our model correctly predicts cases and fatalities (Supplementary Figure 2). The model equations, the parameter ranges, and a validation of model projections from data are available in the Supplementary Materials. We ran the model on 120 parameter combinations, selected by Latin hypercube sampling [30] and report the 95% confidence intervals (CIs) across the 120 analyses. This study uses published aggregated anonymized data and thus meets legal and institutional requirements for protection of human subjects.
RESULTS
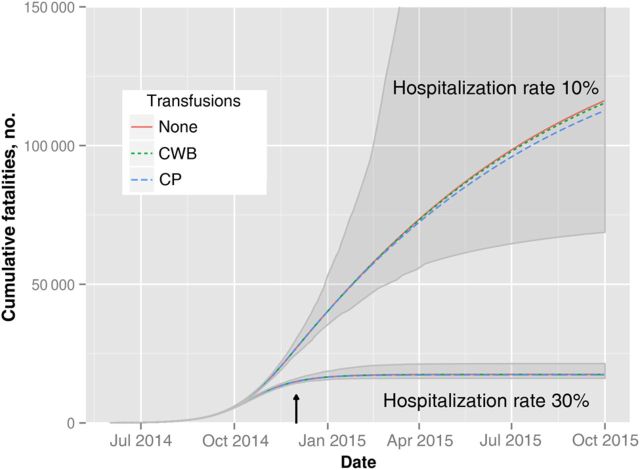
On the assumptions that the case-hospitalization rate remains at 10% and that convalescent transfusion therapy is maximally scaled up in Liberia starting 1 December 2014, we estimate that the CWB- and CP-based interventions will avert a total of 851 (0.73%; 95% CI, .19%–1.1%) and 3586 (3.1%; 95% CI, .52%–4.5%) EVD fatalities, respectively, but that neither will measurably influence the overall incidence (Figure 1). However, the available data are consistent with a hospitalization rate of 30% in early December 2014. The model suggests that a 30% hospitalization rate should significantly mitigate the course of the epidemic to the extent that the epidemic in Liberia would be controlled by January 2015, consistent with the recent leveling of the EVD incidence in Liberia reported by the WHO [31]. In this scenario, CWB and CP transfusions are estimated to reduce the number of deaths by 65 (0.37%; 95% CI, .07%–2.6%) and 151 (0.9%; 95% CI, .21%–11%), respectively.
Figure 1.
Projected impact of large-scale transfusion therapy in Liberia, beginning 1 December 2014, on cumulative fatalities. The gray ribbon denotes the 95% confidence interval (CI) for expected fatalities, assuming no transfusion intervention. The arrow denotes the start of the transfusion intervention. Under a 10% hospitalization rate, convalescent plasma (CP) transfusions are expected to reduce cumulative fatalities by 3.1% (95% CI, .52%–4.5%) by 1 October 2015. Increasing the hospitalization rate to 30% by 1 December 2014 is projected to contain the spread; in this case, CP transfusion is expected to achieve a 0.9% reduction in cumulative fatalities (95% CI, .21%–11%). Given the more limited supply of convalescent whole blood (CWB) transfusions, they are projected to lower expected cumulative fatalities by only 0.73% (95% CI, .19%–1.1%) and 0.37% (95% CI, .069%–2.6%) under 10% and 30% hospitalization rates, respectively.
Under the dire scenario of a 10% hospitalization rate, the model predicts that mass CWB-based transfusions starting in December 2014 would result in 1594 of 14 883 hospitalized cases receiving transfusion therapy (Figure 2). Among the cases hospitalized after the intervention rollout, 14% would be treated (95% CI, 8.6%–23%). With CP transfusions, the model predicts a larger supply, resulting in 6683 patients treated (60% of those hospitalized after the rollout; 95% CI, 33%–70%). At a cost of $75 per transfusion, the total costs of the intervention are estimated to be $0.120 M and $0.503 M for CWB and CP transfusions, respectively, excluding transfusion infrastructure and labor.
Figure 2.
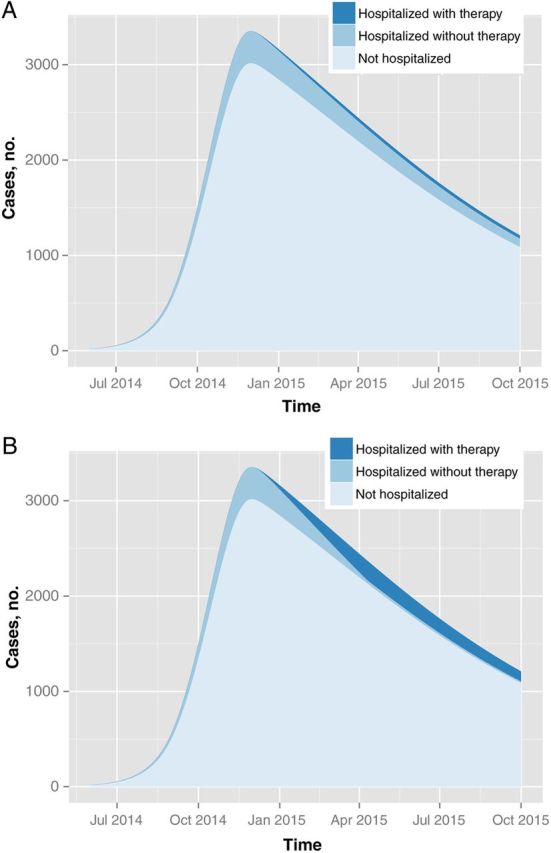
Projected numbers of patients with Ebola virus disease who are not hospitalized, are hospitalized without transfusion therapy, and are hospitalized with transfusion therapy, assuming a 10% hospitalization rate in Liberia, using convalescent-whole blood (CWB) transfusions (A), and convalescent plasma (CP) transfusions (B). Even if equipment and staff are available, it would not be possible to treat all hospitalized patients, owing to shortages of convalescent donors. With a CWB-based intervention, we estimate that 14% of patients hospitalized after the December 2014 implementation would receive therapy; with CP, the estimated fraction treated increases to 60%.
The epidemiological impact of a large-scale transfusion therapy intervention is limited by several factors. While CWB and CP transfusions lower case-fatality rates in treated patients, they have only a minimal effect on transmission, primarily through prevention of postmortem exposure during traditional burials. Since transfusions are undertaken only in the hospital setting, the number of treated patients is constrained by the hospitalization rate. If we denote the hospitalization rate as pH and the difference in case-fatality rates for untreated and treated hospital patients as δH–δT, then the relative reduction in mortality is at most pH[δH–δT]. Thus, at hospitalization rates of 10% and 30%, a case-fatality rate (δH) of 67% for patients hospitalized without transfusion treatment [1], and a case-fatality rate (δT) of 12.5% for patients hospitalized with transfusion treatment [9], transfusion therapy can at most reduce overall mortality by 5.4% and 16%, respectively, during the intervention period. When hospitalized patients receive supportive intravenous fluids, case-fatality rate is estimated to decrease to 43% [32]. In this case, transfusion therapy is expected to reduce overall mortality by 3.1% and 9.2%, based on hospitalization rates of 10% and 30%, respectively. The supply of CWB and CP donations is expected to further limit the number of transfusions until late in the epidemic (Figure 2), when supply starts to exceed the number of treatable patients. Consequently, the estimated reduction in mortality is lower than these theoretical upper bounds during the intervention period.
Large changes to the donation rates, mortality rates, and efficacy of transfusion have intuitive effects (Table 1). Under a CP intervention with 10% hospitalization, increasing the initial donation rate from 50% to 100% of previously hospitalized survivors is expected to increase the number of donations by 21% and decrease overall fatalities by 0.6%; reducing the case-fatality rates of hospitalized patients from 67% to 40% is expected to decrease the overall fatality rate by 2.1%, because more survivors means more potential convalescent donors. Delaying the implementation of transfusions until January 2015 increases the expected mortality by 0.5%, and an additional 30-day delay would increase it by 1%. CWB is expected to be in much shorter supply than CP, and thus its success is much more sensitive to donor recruitment and retention rates. For example, increasing the probability of a second donation from 70% to 90% would increase the number of CWB transfusion courses by 60%, but the number of CP transfusion course would increase only by 9.6%. In theory, the CWB supply could be significantly augmented (by 331%) by donations from convalescent cases who were never hospitalized.
Table 1.
Projected Impacts of Large-scale Convalescent Whole Blood (CWB) and Convalescent Plasma (CP) Transfusion Campaigns Under a Range of Scenarios
| Scenario | Base Valuea | Alternative Valuea | Percentage Change in Mortality Rateb |
Percentage Change in No. of Patients Treatedc |
||
|---|---|---|---|---|---|---|
| CWB | CP | CWB | CP | |||
| Based | … | … | −0.73 | −3.1 | … | … |
| Alternative | ||||||
| Higher q1 | 50 | 100 | −1.8 | −3.7 | 147 | 20.8 |
| Higher q2 | 70 | 90 | −1.2 | −3.4 | 60.3 | 9.6 |
| Higher σC | 0 | 30 | −3.2 | −3.9 | 331 | 25.0 |
| Lower therapeutic efficacy (1 − δT) | 87.5 | 70 | −0.5 | −2.0 | −6.0 | −4.3 |
| Higher δC | 80 | 90 | 9.4 | 8.3 | 0.0 | 0.0 |
| Higher δH | 67 | 90 | 2.3 | 0.2 | −67.0 | −36.6 |
| Lower δH | 67 | 40 | −3.7 | −5.2 | 65.5 | 14.2 |
| Delayed implementation | ||||||
| By 30 d | 1 Dec 2014 | 31 Dec 2014 | −0.6 | −2.6 | −18.4 | −17.0 |
| By 60 d | 1 Dec 2014 | 30 Jan 2015 | −0.5 | −2.1 | −35.0 | −32.6 |
Abbreviations: q1, donation rate; q2, donor retention rate; δC, case-fatality rate among nonhospitalized patients without transfusion treatment; δH, case-fatality rate among patients hospitalized with transfusion treatment; δT, case-fatality rate among patients hospitalized without transfusion treatment; σC, community donation rate.
a Values are percentages or dates.
b Data are percentage changes from the 116 229 patients expected to die in the absence of a mass transfusion campaign.
c Data are percentage changes from the base scenario, in which 1594 patients would receive CWB transfusion therapy, and 6683 patients would receive CP transfusion therapy.
d The base scenario is calculated using the specified base values and a case hospitalization rate of 10%.
DISCUSSION
To our knowledge, this is the first quantitative assessment of a large-scale convalescent blood transfusion campaign for the current EVD epidemic. In a best-case scenario, such an intervention may substantially reduce the number of fatalities but will not measurably influence the incidence. Hospitalization improves EVD survival rates and is a prerequisite for becoming either a convalescent blood product donor or a transfusion therapy recipient. Thus, as public health efforts increase hospitalization rates, the overall burden of disease should decrease, and the availability of convalescent donors and patients would increase. However, even under a more optimistic scenario of a 30% hospitalization rate, CWB donations are expected to remain in short supply until very late in the epidemic. This could potentially be remedied by expanding the donor pool outside the hospital setting and recruiting known survivors of EVD. CP therapy is considered safer and more efficient than CWB therapy, and the waiting period between consecutive donations is shorter. However, plasma extraction requires expensive equipment that is not yet available in many Liberian healthcare facilities. If it is assumed that hospitalization rates remain low (10%), broad implementation of CWB therapy (but not CP therapy) on 1 December 2014 is expected to prevent 851 deaths (0.73%) by October 2015. CP therapy is expected to prevent many more deaths, exceeding the number prevented by CWB by factors of 4.2 and 2.3 under the 10% and 30% hospitalization rates, respectively. Delaying the start of CP by 2 months is expected to increase mortality by 1%, pointing to the need for timely implementation of any plasma transfusions. In the 30% hospitalization scenario, in which the epidemic is controlled, a nationwide CP therapy intervention should still save an additional 151 lives (0.9% of the total) by October 2015. At a cost of $75 per CWB transfusion, excluding costs associated with labor, the overall cost of a transfusion campaign in Liberia is estimated be quite modest, costing <1 million US dollars. However, there may also be significant additional costs associated with training staff and treating infections detected in donor blood. A CP system would require additional infrastructure investments but would potentially quadruple the number of lives saved if hospitalization rates remain low.
There are numerous challenges to implementing a large-scale transfusion therapy campaign. In addition to severe resource limitations, including lack of supplies, equipment, space, and personnel for taking, testing, and processing blood donations, the system will be fundamentally constrained by the ratio of convalescent donors to hospitalized patients requiring therapy. This ratio can be particularly low in the early stage of an epidemic, when the increase in the number of new cases outpaces the increase in the number of individuals who have recovered, and it is exacerbated by EVD's high fatality rate and low hospitalization rate in West Africa. Although transfusion therapy coupled with supportive care may have an efficacy as high as 90% [9], these practical challenges of implementation will likely persist throughout resource-limited regions of Africa and severely limit the overall reduction in fatalities. Despite these obstacles, ramping up CWB therapy and, particularly, CP therapy can tangibly reduce mortality in the ongoing West African EVD epidemic.
There are limitations to the assessment presented here. Our analysis relies on a compartmental model that does not account for the complex social and geographic factors that impact Ebola virus transmission and health-seeking behavior. We assume that human behavior, treatment efficacy, and disease transmission and progression rates are static but account for the introduction of greater case isolation and increased hospitalization rates. Although our quantitative findings held up to sensitivity and uncertainty analysis, as well as to comparison with other models and epidemiological data, we place much more stock in our qualitative findings.
The use of blood transfusion therapy for EVD is controversial. While it is supported by some small studies [9, 15, 16], indirectly supported by the positive findings with monoclonal therapies [33–36], and biologically plausible [16], its efficacy has not been rigorously established through published randomized controlled trials [16], but such studies are now underway in West Africa [37, 38]. The beneficial effect was not successfully reproduced in all animal models [39]. Additionally, the possible adverse events associated with passive immunization for Ebola virus include antibody-dependent enhancement [10] and an increased risk of accidental exposure among healthcare workers, which are difficult to estimate. The broad use of transfusion therapy to treat cases in developed countries reflects medical consensus about some beneficial effect of the therapy [40]. CWB and CP transfusion therapy may also indirectly improve outcomes, as they require a higher level of supportive care than commonly provided in Liberia, including intravenous rather than oral rehydration therapy or no hydration therapy at all. The model accounted for the supportive care given to all treated cases [9, 15] and considered the impacts of lower therapeutic efficacy in the sensitivity analysis.
In summary, large-scale use of CWB or CP transfusion therapy is likely to be a relatively inexpensive means to save lives but not reduce transmission in the ongoing West African EVD epidemics. Broad expansion of hospital-based care should reduce the overall EVD mortality and increase the fraction of cases that could potentially receive CWB or CP transfusion therapy. Investment in CP transfusion facilities as an alternative to CWB transfusions should substantially increase the blood supply and thereby increase the lifesaving impacts of a large-scale transfusion intervention in Liberia by a factor of 2–4.
Supplementary Data
Supplementary materials are available at The Journal of Infectious Diseases online (http://jid.oxfordjournals.org). Supplementary materials consist of data provided by the author that are published to benefit the reader. The posted materials are not copyedited. The contents of all supplementary data are the sole responsibility of the authors. Questions or messages regarding errors should be addressed to the author.
Notes
Acknowledgments. We thank Alison Galvani and an anonymous reviewer, for suggestions, and Dr A. Borodyanskiy, MD, for a helpful discussion.
Disclaimer. The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Financial support. This work was supported by the National Institute of General Medical Sciences (Models of Infectious Disease Agent Study grant U01GM087719 to L. A. M.).
Potential conflict of interest. Both authors: No reported conflicts.
Both authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.WHO Ebola Response Team. Ebola virus disease in West Africa - the first 9 months of the epidemic and forward projections. N Engl J Med 2014; 371:1481–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Meltzer MI, Atkins CY, Santibanez S, et al. Estimating the future number of cases in the Ebola epidemic—Liberia and Sierra Leone, 2014–2015. MMWR Surveill Summ 2014; 63(suppl 3):1–14. [PubMed] [Google Scholar]
- 3.Galvani AP, Ndeffo-Mbah ML, Wenzel N, Childs JE. Ebola vaccination: if not now, when? Ann Intern Med 2014; 161:749–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Camacho A, Kucharski AJ, Funk S, Breman J, Piot P, Edmunds WJ. Potential for large outbreaks of Ebola virus disease. Epidemics 2014; 9:70–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ledgerwood JE, DeZure AD, Stanley DA, et al. Chimpanzee adenovirus vector Ebola vaccine—preliminary report. N Engl J Med 2014; doi:10.1056/NEJMoa1410863. [DOI] [PubMed] [Google Scholar]
- 6.Strauss S. Biotech drugs too little, too late for Ebola outbreak. Nat Biotechnol 2014; 32:849–50. [DOI] [PubMed] [Google Scholar]
- 7.World Health Organization. Statement on the WHO Consultation on potential Ebola therapies and vaccines. Geneva: World Health Organization, 2014. [Google Scholar]
- 8.Burnouf T, Emmanuel J, Mbanya D, et al. Ebola: a call for blood transfusion strategy in sub-Saharan Africa. Lancet 2014; 384:1347–8. [DOI] [PubMed] [Google Scholar]
- 9.Mupapa K, Massamba M, Kibadi K, et al. Treatment of Ebola hemorrhagic fever with blood transfusions from convalescent patients. J Infect Dis 1999; 179(suppl 1):S18–23. [DOI] [PubMed] [Google Scholar]
- 10.Takada A. Do therapeutic antibodies hold the key to an effective treatment for Ebola hemorrhagic fever? Immunotherapy 2013; 5:441–3. [DOI] [PubMed] [Google Scholar]
- 11.Saphire EO. An update on the use of antibodies against the filoviruses. Immunotherapy 2013; 5:1221–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Emond R, Evans B, Bowen E, Lloyd G. A case of Ebola virus infection. Br Med J 1977; 2:541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Casadevall A, Dadachova E, Pirofski L. Passive antibody therapy for infectious diseases. Nat Rev Microbiol 2004; 2:695–703. [DOI] [PubMed] [Google Scholar]
- 14.Dimitrov DS, Marks JD. Therapeutic antibodies: current state and future trends–is a paradigm change coming soon? In: Dimitrov AS, ed. Methods in Molecular Biology. Vol 525. New York: Humana Press, 2009:1–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dye JM, Herbert AS, Kuehne AI, et al. Postexposure antibody prophylaxis protects nonhuman primates from filovirus disease. Proc Natl Acad Sci 2012; 109:5034–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.World Health Organization. Use of convalescent whole blood or plasma collected from patients recovered from Ebola virus disease for transfusion, as an empirical treatment during outbreaks. Geneva: World Health Organization, 2014. [Google Scholar]
- 17.Tapko JB, Toure B, Sambo LG. Status of blood safety in the WHO African Region: report of the 2010 survey. Geneva: World Health Organization, 2014. [Google Scholar]
- 18.World Health Organization. E xperimental therapies: growing interest in the use of whole blood or plasma from recovered Ebola patients (convalescent therapies). Geneva: World Health Organization, 2014. [Google Scholar]
- 19.Pandey A, Atkins KE, Medlock J, et al. Strategies for containing Ebola in West Africa. Science 2014; 346:991–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rivers CM, Lofgren ET, Marathe M, Eubank S, Lewis BL. Modeling the impact of interventions on an epidemic of Ebola in Sierra Leone and Liberia. PLoS Curr Outbreaks 2014; doi:10.1371/currents.outbreaks.fd38dd85078565450b0be3fcd78f5ccf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Althaus CL. Estimating the reproduction number of Zaire ebolavirus (EBOV) during the 2014 outbreak in West Africa. PLoS Curr Outbreaks 2014; doi:10.1371/currents.outbreaks.91afb5e0f279e7f29e7056095255b288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chowell G, Hengartner NW, Castillo-Chavez C, Fenimore PW, Hyman JM. The basic reproductive number of Ebola and the effects of public health measures: the cases of Congo and Uganda. J Theor Biol 2004; 229:119–26. [DOI] [PubMed] [Google Scholar]
- 23.Nishiura H, Chowell G. Early transmission dynamics of Ebola virus disease (EVD), West Africa, March to August 2014. Rapid Commun 2014; 19:1–6. [DOI] [PubMed] [Google Scholar]
- 24.Lekone PE, Finkenstaedt BF. Statistical inference in a stochastic epidemic SEIR model with control intervention: Ebola as a case study. Biometrics 2006; 62:1170–7. [DOI] [PubMed] [Google Scholar]
- 25.Legrand J, Grais R, Boelle P, Valleron A, Flahault A. Understanding the dynamics of Ebola epidemics. Epidemiol Infect 2007; 135:610–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.World Health Organization. Ebola response roadmap situation report for 29 October 2014. Geneva: World Health Organization, 2014. [Google Scholar]
- 27.van Hulst M, Sibinga CTS, Postma MJ. Health economics of blood transfusion safety - focus on sub-Saharan Africa. Biologicals 2010; 38:53–8. [DOI] [PubMed] [Google Scholar]
- 28.Jacobs B, Mercer A. Feasibility of hospital-based blood banking: a Tanzanian case study. Health Policy Plan 1999; 14:354–62. [DOI] [PubMed] [Google Scholar]
- 29.Toner R, Pizzi L, Leas B, Ballas S, Quigley A, Goldfarb N. Costs to hospitals of acquiring and processing blood in the US. Appl Health Econ Health Policy 2011; 9:29–37. [DOI] [PubMed] [Google Scholar]
- 30.McKay MD. Latin hypercube sampling as a tool in uncertainty analysis of computer models. In: Proceedings of the 24th Conference on Winter Simulation New York: Association for Computing Machinery, 1992:564. [Google Scholar]
- 31.World Health Organization. Ebola response roadmap situation report for 7 January 2015. Geneva: World Health Organization, 2015. [Google Scholar]
- 32.Bah EI, Lamah M-C, Fletcher T, et al. Clinical presentation of patients with Ebola virus disease in Conakry, Guinea. N Engl J Med 2015; 372:40–7. [DOI] [PubMed] [Google Scholar]
- 33.Qiu X, Audet J, Wong G, et al. Sustained protection against Ebola virus infection following treatment of infected nonhuman primates with ZMAb. Sci Rep 2013; 3365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Qiu X, Wong G, Audet J, et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMapp. Nature 2014; 514:47–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wong G, Kobinger GP, Qiu X. Characterization of host immune responses in Ebola virus infections. Expert Rev Clin Immunol 2014; 10:781–90. [DOI] [PubMed] [Google Scholar]
- 36.Olinger GG, Pettitt J, Kim D, et al. Delayed treatment of Ebola virus infection with plant-derived monoclonal antibodies provides protection in rhesus macaques. Proc Natl Acad Sci 2012; 109:18030–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.First trials for Ebola treatments to start at MSF sites in December. Geneva, Switzerland: Médecins Sans Frontières, 2014. Available at: http://www.msf.org/article/first-trials-ebola-treatments-start-msf-sites-december. Accessed 18 November 2014. [Google Scholar]
- 38.Butler D. First trials of blood-based Ebola therapy kick off. Nature 2014; doi:10.1038/nature.2014.16564. [Google Scholar]
- 39.Jahrling PB, Geisbert JB, Swearengen JR, Larsen T, Geisbert TW. Ebola hemorrhagic fever: evaluation of passive immunotherapy in nonhuman primates. J Infect Dis 2007; 196(suppl 2):S400–3. [DOI] [PubMed] [Google Scholar]
- 40.Feldmann H, Geisbert TW. Ebola haemorrhagic fever. Lancet 2011; 377:849–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.