Abstract
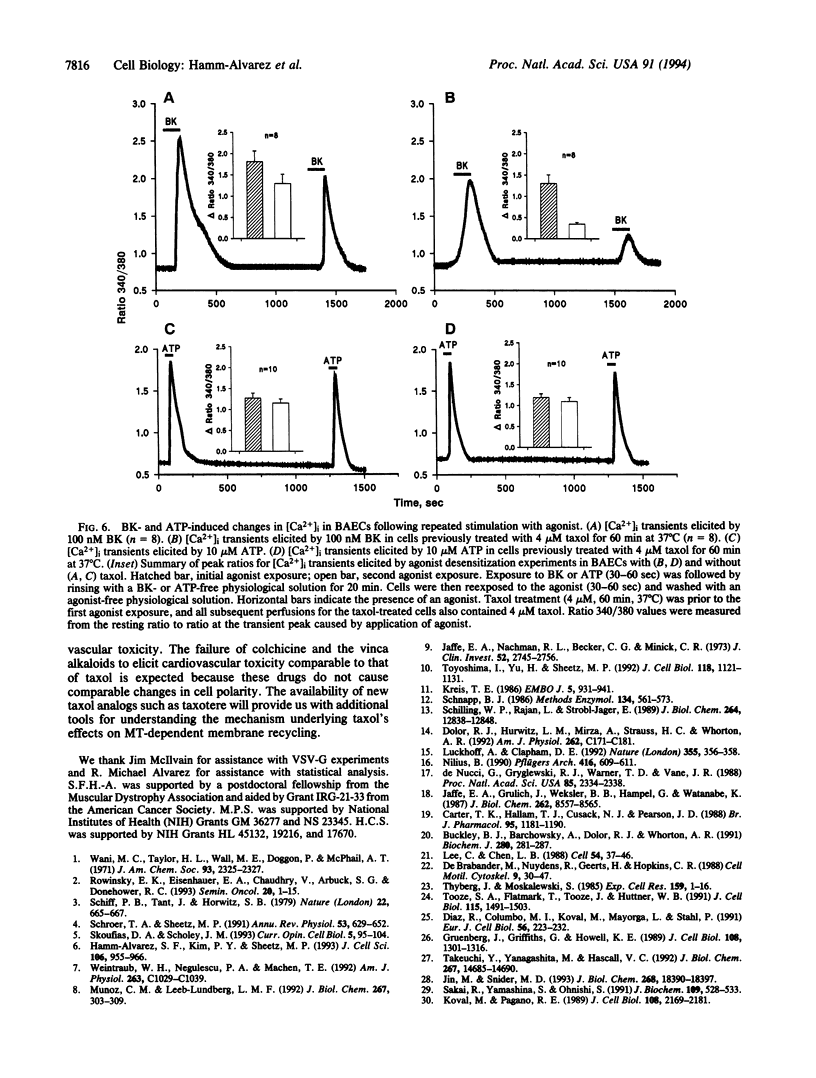
Significant cardiovascular side effects have limited the use of taxol as an anticancer drug. A link between decreased plasma membrane dynamics and taxol has been implied because taxol can inhibit intracellular vesicle movements. Reduced membrane recycling caused by taxol could inhibit agonist-evoked Ca2+ signaling within endothelial cells, resulting in endothelium-dependent vasodilation. Bradykinin and ATP are two agonists that evoke Ca2+ transients in endothelial cells. Since the bradykinin receptor-agonist complex is internalized and recycled whereas the ATP agonist-receptor complex is not, we expected that a taxol inhibition of recycling would decrease bradykinin but not ATP receptor activity. We found that taxol depresses (i) the frequency (to 41% of control) and velocity (to 55% of control) of microtubule-dependent vesicle transport and (ii) bradykinin-evoked cytosolic Ca2+ transients (to 76% of control) in bovine aortic endothelial cells. In studying bradykinin receptor desensitization, which reflects receptor recycling, we demonstrate that taxol inhibits bradykinin-evoked Ca2+ transients by 50%. Taxol did not significantly alter ATP-evoked Ca2+ transients in either single-exposure or desensitization experiments. We suggest that taxol's reduction of bradykinin-evoked Ca2+ transients is due to altered microtubule-dependent membrane recycling. This report describes taxol's ability to alter plasma membrane composition through effects on vesicle transport and membrane trafficking pathways. This finding provides a possible mechanism by which taxol can substantially alter cardiovascular function.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Buckley B. J., Barchowsky A., Dolor R. J., Whorton A. R. Regulation of arachidonic acid release in vascular endothelium. Ca(2+)-dependent and -independent pathways. Biochem J. 1991 Dec 1;280(Pt 2):281–287. doi: 10.1042/bj2800281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter T. D., Hallam T. J., Cusack N. J., Pearson J. D. Regulation of P2y-purinoceptor-mediated prostacyclin release from human endothelial cells by cytoplasmic calcium concentration. Br J Pharmacol. 1988 Dec;95(4):1181–1190. doi: 10.1111/j.1476-5381.1988.tb11754.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Brabander M., Nuydens R., Geerts H., Hopkins C. R. Dynamic behavior of the transferrin receptor followed in living epidermoid carcinoma (A431) cells with nanovid microscopy. Cell Motil Cytoskeleton. 1988;9(1):30–47. doi: 10.1002/cm.970090105. [DOI] [PubMed] [Google Scholar]
- Diaz R., Colombo M. I., Koval M., Mayorga L., Stahl P. Endosomal density shift is related to a decrease in fusion capacity. Eur J Cell Biol. 1991 Dec;56(2):223–232. [PubMed] [Google Scholar]
- Dolor R. J., Hurwitz L. M., Mirza Z., Strauss H. C., Whorton A. R. Regulation of extracellular calcium entry in endothelial cells: role of intracellular calcium pool. Am J Physiol. 1992 Jan;262(1 Pt 1):C171–C181. doi: 10.1152/ajpcell.1992.262.1.C171. [DOI] [PubMed] [Google Scholar]
- Gruenberg J., Griffiths G., Howell K. E. Characterization of the early endosome and putative endocytic carrier vesicles in vivo and with an assay of vesicle fusion in vitro. J Cell Biol. 1989 Apr;108(4):1301–1316. doi: 10.1083/jcb.108.4.1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamm-Alvarez S. F., Kim P. Y., Sheetz M. P. Regulation of vesicle transport in CV-1 cells and extracts. J Cell Sci. 1993 Nov;106(Pt 3):955–966. doi: 10.1242/jcs.106.3.955. [DOI] [PubMed] [Google Scholar]
- Jaffe E. A., Grulich J., Weksler B. B., Hampel G., Watanabe K. Correlation between thrombin-induced prostacyclin production and inositol trisphosphate and cytosolic free calcium levels in cultured human endothelial cells. J Biol Chem. 1987 Jun 25;262(18):8557–8565. [PubMed] [Google Scholar]
- Jaffe E. A., Nachman R. L., Becker C. G., Minick C. R. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Invest. 1973 Nov;52(11):2745–2756. doi: 10.1172/JCI107470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin M., Snider M. D. Role of microtubules in transferrin receptor transport from the cell surface to endosomes and the Golgi complex. J Biol Chem. 1993 Aug 25;268(24):18390–18397. [PubMed] [Google Scholar]
- Koval M., Pagano R. E. Lipid recycling between the plasma membrane and intracellular compartments: transport and metabolism of fluorescent sphingomyelin analogues in cultured fibroblasts. J Cell Biol. 1989 Jun;108(6):2169–2181. doi: 10.1083/jcb.108.6.2169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreis T. E. Microinjected antibodies against the cytoplasmic domain of vesicular stomatitis virus glycoprotein block its transport to the cell surface. EMBO J. 1986 May;5(5):931–941. doi: 10.1002/j.1460-2075.1986.tb04306.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee C., Chen L. B. Dynamic behavior of endoplasmic reticulum in living cells. Cell. 1988 Jul 1;54(1):37–46. doi: 10.1016/0092-8674(88)90177-8. [DOI] [PubMed] [Google Scholar]
- Lückhoff A., Clapham D. E. Inositol 1,3,4,5-tetrakisphosphate activates an endothelial Ca(2+)-permeable channel. Nature. 1992 Jan 23;355(6358):356–358. doi: 10.1038/355356a0. [DOI] [PubMed] [Google Scholar]
- Munoz C. M., Leeb-Lundberg L. M. Receptor-mediated internalization of bradykinin. DDT1 MF-2 smooth muscle cells process internalized bradykinin via multiple degradative pathways. J Biol Chem. 1992 Jan 5;267(1):303–309. [PubMed] [Google Scholar]
- Nilius B. Permeation properties of a non-selective cation channel in human vascular endothelial cells. Pflugers Arch. 1990 Jul;416(5):609–611. doi: 10.1007/BF00382697. [DOI] [PubMed] [Google Scholar]
- Rowinsky E. K., Eisenhauer E. A., Chaudhry V., Arbuck S. G., Donehower R. C. Clinical toxicities encountered with paclitaxel (Taxol). Semin Oncol. 1993 Aug;20(4 Suppl 3):1–15. [PubMed] [Google Scholar]
- Sakai T., Yamashina S., Ohnishi S. Microtubule-disrupting drugs blocked delivery of endocytosed transferrin to the cytocenter, but did not affect return of transferrin to plasma membrane. J Biochem. 1991 Apr;109(4):528–533. doi: 10.1093/oxfordjournals.jbchem.a123415. [DOI] [PubMed] [Google Scholar]
- Schiff P. B., Fant J., Horwitz S. B. Promotion of microtubule assembly in vitro by taxol. Nature. 1979 Feb 22;277(5698):665–667. doi: 10.1038/277665a0. [DOI] [PubMed] [Google Scholar]
- Schilling W. P., Rajan L., Strobl-Jager E. Characterization of the bradykinin-stimulated calcium influx pathway of cultured vascular endothelial cells. Saturability, selectivity, and kinetics. J Biol Chem. 1989 Aug 5;264(22):12838–12848. [PubMed] [Google Scholar]
- Schnapp B. J. Viewing single microtubules by video light microscopy. Methods Enzymol. 1986;134:561–573. doi: 10.1016/0076-6879(86)34121-1. [DOI] [PubMed] [Google Scholar]
- Schroer T. A., Sheetz M. P. Functions of microtubule-based motors. Annu Rev Physiol. 1991;53:629–652. doi: 10.1146/annurev.ph.53.030191.003213. [DOI] [PubMed] [Google Scholar]
- Skoufias D. A., Scholey J. M. Cytoplasmic microtubule-based motor proteins. Curr Opin Cell Biol. 1993 Feb;5(1):95–104. doi: 10.1016/s0955-0674(05)80014-6. [DOI] [PubMed] [Google Scholar]
- Takeuchi Y., Yanagishita M., Hascall V. C. Recycling of transferrin receptors and heparan sulfate proteoglycans in a rat parathyroid cell line. J Biol Chem. 1992 Jul 25;267(21):14685–14690. [PubMed] [Google Scholar]
- Thyberg J., Moskalewski S. Microtubules and the organization of the Golgi complex. Exp Cell Res. 1985 Jul;159(1):1–16. doi: 10.1016/s0014-4827(85)80032-x. [DOI] [PubMed] [Google Scholar]
- Tooze S. A., Flatmark T., Tooze J., Huttner W. B. Characterization of the immature secretory granule, an intermediate in granule biogenesis. J Cell Biol. 1991 Dec;115(6):1491–1503. doi: 10.1083/jcb.115.6.1491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toyoshima I., Yu H., Steuer E. R., Sheetz M. P. Kinectin, a major kinesin-binding protein on ER. J Cell Biol. 1992 Sep;118(5):1121–1131. doi: 10.1083/jcb.118.5.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wani M. C., Taylor H. L., Wall M. E., Coggon P., McPhail A. T. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc. 1971 May 5;93(9):2325–2327. doi: 10.1021/ja00738a045. [DOI] [PubMed] [Google Scholar]
- Weintraub W. H., Negulescu P. A., Machen T. E. Calcium signaling in endothelia: cellular heterogeneity and receptor internalization. Am J Physiol. 1992 Nov;263(5 Pt 1):C1029–C1039. doi: 10.1152/ajpcell.1992.263.5.C1029. [DOI] [PubMed] [Google Scholar]
- de Nucci G., Gryglewski R. J., Warner T. D., Vane J. R. Receptor-mediated release of endothelium-derived relaxing factor and prostacyclin from bovine aortic endothelial cells is coupled. Proc Natl Acad Sci U S A. 1988 Apr;85(7):2334–2338. doi: 10.1073/pnas.85.7.2334. [DOI] [PMC free article] [PubMed] [Google Scholar]