Abstract
Neuroprotection, recovery of function, and gene expression were evaluated in an animal model of traumatic brain injury (TBI) after a combination treatment of nicotinamide (NAM) and progesterone (Prog). Animals received a cortical contusion injury over the sensorimotor cortex, and were treated with either Vehicle, NAM, Prog, or a NAM/Prog combination for 72 h and compared with a craniotomy only (Sham) group. Animals were assessed in a battery of behavioral, sensory, and both fine and gross motor tasks, and given histological assessments at 24 h post-injury to determine lesion cavity size, degenerating neurons, and reactive astrocytes. Microarray-based transcriptional profiling was used to determine treatment-specific changes on gene expression. Our results confirm the beneficial effects of treatment with either NAM or Prog, demonstrating significant improvements in recovery of function and a reduction in lesion cavitation, degenerating neurons, and reactive astrocytes 24 h post-injury. The combination treatment of NAM and Prog led to a significant improvement in both neuroprotection at 24 h post-injury and recovery of function in sensorimotor related tasks when compared with individual treatments. The NAM/Prog-treated group was the only treatment group to show a significant reduction of cortical loss 24 h post-injury. The combination appears to affect inflammatory and immune processes, reducing expression of a significant number of genes in both pathways. Further preclinical trials using NAM and Prog as a combination treatment should be conducted to identify the window of opportunity, determine the optimal duration of treatment, and evaluate the combination in other pre-clinical models of TBI.
Key words: : controlled cortical impact, nicotinamide, polytherapy, progesterone, recovery of function, traumatic brain injury
Introduction
Traumatic brain injury (TBI) is a serious health issue creating widespread disability to survivors, including residual physical, cognitive, emotional, and behavioral impairment. The primary injury causes direct damage to the brain and only injury prevention will reduce the consequences of the primary injury. The secondary brain injury is indirect and progressive and may include a host of likely interrelated processes, including mitochondrial energy failure, excessive generation of reactive oxygen species, activation of destructive enzymes (such as poly [ADP-ribose] polymerase [PARP] and caspase family of proteases), membrane disruption, neuronal death, thrombosis due to intravascular coagulation in small vessels, increased synaptic concentrations of excitatory amino acids, and activation of innate inflammatory responses.1
Preclinical trials of neuroprotective compounds are many, but successes are few. To date, no single pharmacological agent has been shown to improve the outcome of TBI in humans. It has been suggested that pharmacological strategies of neuroprotection should include drugs that target multiple mechanisms of the secondary injury. The concept of using a combination of drugs to target multiple pathways involved in secondary injury led to a 2008 National Institutes of Health (NIH) workshop on combination therapies in TBI and several reviews have suggested the need for pharmacological treatments that target multiple secondary factors or combination treatment strategies.1,2 Results from animal models are the major determinants of whether these neuroprotective drugs reach clinical trials in humans. Preclinical animal research with nicotinamide (NAM) and progesterone (Prog) has demonstrated beneficial effects.
Nicotinamide
NAM is the amide form of vitamin B3. It is a broad-spectrum neuroprotectant and a soluble B-group vitamin that has shown encouraging results when administered following TBI. The Hoane laboratory has spent the past decade establishing the window of opportunity, dosing parameters, and mode of administration of NAM needed to provide neuroprotection in multiple animal models of TBI. NAM treatment has been shown to significantly reduce injury volume, decrease glial fibrillary acidic protein (GFAP)+ activation, reduce the blood brain barrier (BBB) breach, reduce acute edema formation, reduce behavioral impairments, and improve outcomes following cortical contusion injury (CCI) and fluid percussive injury (FPI).3–13 A bolus dose of either 50 or 500 mg/kg following TBI provides improved behavioral outcomes and attenuation of cortical loss and this effectiveness is heightened one week following treatment.6 To establish and maintain a steady-state and provide a more clinically relevant model of neuroprotection, we also demonstrated beneficial effects of a continuous infusion of NAM on both sensorimotor and cognitive tasks with both a low dose (150 mg/kg/d)11 and a clinically achievable dose (288 mg/kg/d) following bilateral frontal and unilateral parietal injury.9,12
NAM is a precursor to nicotinamide adenine dinucleotide (NAD+), a coenzyme found in all living cells, and is commonly known for its role in the electron transport chain allowing for the production of adenosine triphosphate (ATP).14,15 NAM indirectly supplements the bioavailability of ATP to cells, which provides additional energy when normal intake and metabolism are impaired following neural insult. This supplementation can lead to reductions in apoptotic and necrotic death.16,17 NAD +'s involvement in redox reactions also reduces free radicals, which are increased following injury. NAM both directly and indirectly inhibits PARP-1,14,18 which can be taxing on ATP stores (by reducing NAD+) and can lead to apoptosis15 after TBI. Inhibitors of PARP-1, like NAM, have shown beneficial effects on tissue and behavior following injury,19,20 while activation of the PARP-1 pathway has been shown to be detrimental.21,22
NAM had significant effects on gene expression for functional pathways14 involving cellular movement, cell-to-cell signaling, antigen presentation and cell death.23 Signaling pathways involved in inflammatory processes were the primary canonical pathways identified. In general, NAM treatment counteracted gene expression changes due to the TBI by down-regulating genes that were significantly up-regulated in the injured, vehicle-treated animals, compared with sham.
Progesterone
The beneficial effects of Prog administration have been seen in several brain injury models, including diffuse injury models,24,25 penetrating brain injury,26,27 CCI,9,27–30 and aspiration.31 After recognizing that there is a gender difference in response to TBI,32 it was suggested that Prog treatment may have similar functions after the brain sustains an injury.32 As early as 1980, Fass and colleagues33 reported that female rats recovered better than males following a calibrated contusion to the medial frontal cortex, and in 1993, Roof and colleagues29 showed that with increased levels of circulating Prog, only mild edema develops. Since these preliminary findings, several hundred studies using animal models have shown that Prog treatment following TBI provides sustained improvement in morphological, cytological, and functional outcomes.34 When two doses of Prog (10 mg/kg or 20 mg/kg 12 h) were compared with a NAM infusion for 72 h following unilateral CCI, both low dose Prog and NAM improved reference memory acquisition and reversal learning in the Morris water maze and reduced tissue loss in the injured cortex and ipsilateral hippocampus, compared with vehicle. The beneficial effects of Prog were dose dependent, with the lower (10 mg/kg) dose producing significant effects that were not observed at the higher dose. Direct comparison between NAM and low-dose Prog suggested that both were equally effective.
The proposed mechanism of neuroprotection provided by Prog is due to a reduction in cerebral edema, inflammation, apoptosis, oxidative stress, and the glial cell response.35 Prog reduces both vasogenic and cytotoxic edema after TBI,36 and although it does not appear that Prog repairs the BBB following frontal CCI, it does prevent the influx of water into the central nervous system.30 This is probably mediated through the aquaporin-4 water channel on astrocyte feet37,38 and 25-Dx, a Prog membrane binding protein thought to be important for CSF production.39 Prog treatment has multiple global and local anti-inflammatory effects. Locally, Prog can regulate microglial activation, reducing excitotoxicity, as well as cytokine release.40,41 Study results showed that Prog had significant effects on gene expression of genes involved in regulating inflammatory response and apoptosis.41 In contrast to NAM, Prog treatment primarily affected genes that were not differently expressed in the TBI injured animals.
Therefore, given the substantial preclinical literature on both NAM and Prog following TBI and the different biochemical pathways affected by NAM and Prog, they are excellent candidates for a combination treatment. Thus, our main objective was to directly compare the preclinical effectiveness of NAM or Prog with a combination treatment of both following TBI.
Methods
Male Sprague Dawley (Harlan, Indianapolis, IN) rats approximately 3.5 months of age at the time of the injury (mean body weight, 364 g; standard error of the mean [SEM]=2) were used for all of the studies. All animal and surgical procedures adhered to the NIH Guide for the Care and Use of Laboratory Animals. The Southern Illinois University Institutional Animal Care and Use Committee approved all experimental procedures. Before and after injury, animals were housed in a university-maintained, Association for Assessment and Accreditation of Laboratory Animal Care–accredited vivarium, with a 12 h light/dark schedule and a controlled environmental temperature of 22°C in standard housing cages with food and water available ad libitum.
Surgery
All surgeries were performed under aseptic conditions. The CCI model utilized in the present study was based on previous studies and was intended to produce a moderately severe injury.8,9,12,41 Animals were anesthetized using a mixture of isoflurane (2–4%) and oxygen (0.8 L/m). When the animal became unresponsive (no ocular or pedal reflexes) the head was shaved and scrubbed with 70% alcohol followed by betadine and placed into a stereotaxic device. A midline incision was made in the skin as well as through the underlying fascia. A trephine was used to create a circular craniotomy (5.0 mm) centered 0.5 mm anterior to and 3.5 mm lateral (left) to bregma. The unilateral contusion injury was created with a sterile stainless steel impactor tip (4.0 mm in diameter) attached to the Benchmark™ stereotaxic impactor (Leica, St. Louis, MO) at a ten-degree angle towards the midline.
The injury was induced with an impact speed of 3.0 m/sec and an impact depth of 2.5 mm. The impact tip maintained contact with the dural surface for 0.5 sec before retraction. To maintain normal body temperature (37°C) during surgery and recovery, the rats were placed on a warm water recycling bed and pump system (EZ Anesthesia, Palmer, PA). Rats with sham surgeries underwent identical surgical preparation as the injured animals, received craniotomies, were sutured, and were then transferred to recovery. All animals in every group underwent the same anesthesia, regardless of the therapy applied.
The mini-pump (Alzet #2ML1, Alzet, Cuperzino, CA) subcutaneous implantation procedure was performed 4 h post-CCI. Gas anesthesia was administered, the surgical site was sterilized and a midline incision was made in the back halfway between the forelimb and hindlimbs. After the incision was made, the underlying fascia and connective tissue were separated from the skin and the osmotic mini-pump was placed within the “pocket.” Afterwards, the incision was closed with skin staples. Under anesthesia, pumps were explanted 3 d later and the incision was sutured closed. Regardless of treatment, all groups were anesthetized in the same manner.
Drug administration
Prog and NAM doses were selected by targeting clinically significant concentrations as described previously.23,41 Prog (Sigma #P3972, St. Louis, MO) was administered intraperitoneally at a dose of 10 mg/kg. NAM (Sigma #N5535, St. Louis, MO) was administered with osmotic pumps, which allow for continuous infusion (12 mg/kg/h; 240 μL/d) plus an intraperitoneal 75-mg/kg loading dose. To control for effects following injections and/or pump implantation, all animals were injected, anesthetized, and implanted with osmotic pumps. Prog was diluted in fresh peanut oil (LouAna, Brea, CA). All animals received an injection 4 h post-injury and an additional injection every 12 h for 72 h post-injury or until sacrifice. Following the 75 mg/kg loading dose (diluted in saline) of NAM 4 h post-injury, NAM-administered animals received injections of peanut oil for the remaining injection time-points. The combination therapy group received both the NAM and Prog treatments. Both the Sham and Vehicle groups were administered intraperitoneal injections of peanut oil and osmotic pumps filled with 0.9% sterile saline (Sigma #14190, St. Louis, MO) for all injection time-points. The behavioral study group comparisons included a NAM/Prog-injured group (n=9), a NAM-injured group (n=9), a Prog-injured group (n=9), a Vehicle-injured group (n=9), and a Sham (n=9) group. The anatomical study group comparisons included a NAM/Prog-injured group (n=6), a NAM-injured group (n=6), a Prog-injured group (n=6), a Vehicle-injured group (n=6), and a Sham (n=6) group.
Serum analyses
Serum samples were collected and analyzed to measure the amounts of circulating NAM and Prog in each group. Blood samples (1 mL) were collected from the tail vein 1 h after the final injection. Blood serum was separated by microcentrifuge and stored at −80°C. NAM concentrations were analyzed in serum using high-performance liquid chromatography (HPLC) with ultraviolet detection at 254 nm on a Varian Pro Star 210 HPLC system (Varian Medical Systems, Inc., Palo Alto, CA) using a previously-developed assay.5 Prog concentrations were analyzed using an enzyme linked immunoassays (R&D Systems Inc, Minneapolis, MN, and VWR International, West Chester, PA).
Sensory assessment: Bilateral tactile adhesive removal task
This task has been shown to be an effective assessment of somatosensory deficits following injuries to the rodent frontal lobe.13,42–44 To assess sensory deficits, this test was administered on Days 4, 6, 8, 11, 17, 24, and 28 post-injury. Rats were pre-tested for baseline levels on the two days prior to surgery. A small circular sticker approximately 113 mm2 (Avery Dennison Corporation, Brea, CA) was applied to the radial aspect of each forelimb. The rat was then returned to its home cage and the latency to initially contact the sticker was used as the dependent variable. The trial was terminated when the right and left stickers were both removed or 2 min had elapsed. Each animal received two trials each day with a 15 min inter-trial interval. Researchers who were blind to the treatment condition performed all testing.
Motor assessment: Forelimb asymmetry task
To assess the amount of reliance on the “uninjured limb” (ipsilateral to injury), this test was administered on Days 4, 6, 8, 11, 17, 24, and 28 post-injury following methods outlined in previous studies.42,45 Rats were pre-tested for baseline levels on the two days prior to surgery. On each test day, the rats were placed in an empty fish tank (50 cm ×25 cm ×30 cm) and allowed to explore for 120 sec. Rats were administered one trial per test day, which was recorded with a video camera. Later, the number of right and left forelimb contacts on the vertical surface was recorded as the primary dependent variable of interest. The following formula was used to calculate the reliance on the uninjured limb: Right forelimb contacts/(Right forelimb contacts+Left forelimb contacts) ×100. Researchers who were blind to the treatment condition performed all testing. Inter-rater reliability scores were calculated for approximately 25% of the data collected. Data were chosen randomly and the criterion for acceptance was a correlation coefficient of r>0.90.
Motor assessment: Locomotor placing task
To assess recovery of coordinated, locomotor limb movement, this test was administered on Days 4, 6, 8, 11, 17, 24, and 28 post-injury following methods outlined in previous studies.11–13 Rats were pre-tested for baseline levels on the two days prior to surgery. On each test day, the rat was placed on an elevated grid floor (56.0 cm ×54.0 cm) with openings measuring 3.2 cm ×3.2 cm in size and allowed to freely explore for 120 sec. A “foot-fault” was defined as an inaccurate limb placement through one of these openings. Rats were administered one trial per test day, which was recorded with a video camera. Later, total movement on the grid, as well as foot-faults, were recorded. The total number of foot-faults was the primary dependent variable of interest. The following formula was used to calculate the foot faults as a function of total movement on the grid: (Right forelimb faults-Left forelimb faults)/ lines crossed.12 Researchers who were blind to the treatment condition performed all testing. Inter-rater reliability scores were calculated for approximately 25% of the data collected. Data were chosen randomly and the criterion for acceptance was a correlation coefficient of r>0.90.
Behavioral data analysis
Behavioral data were analyzed using a mixed model factorial analysis of variance (ANOVA) or one-way between-subjects ANOVA (SPSS v. 15 for Windows). The between factor was Treatment (NAM/Prog-injured, NAM-injured, Prog-injured, Vehicle-injured, and Sham) and the within-group factor was day of testing. Both the main effects and the interaction effects were considered. Huynh-Feldt corrections and Tukey's Honestly Significant Different test (Tukey's HSD) were used to control for type I error in the repeated measures and post hoc means comparison, respectively. Planned comparisons using a t-test were performed when the interaction effects were not significant to examine differences in performance on each test. A significant level of p≤0.05 was used for all statistical analyses.
Lesion analysis
At 29 d post-injury, the rats that were assessed on functional recovery were euthanized with Euthasol (Virbac Animal Health; 0.3 mL intraperitoneally), and transcardially perfused with 0.9% phosphate-buffered saline (PBS), followed by 10% phosphate buffered formalin (PBF). Brains were post-fixed in PBF following removal from the cranium for 24 h and then transferred into a 30% sucrose solution to cryopreserve the brains 4 d prior to frozen sectioning. Serial coronal sections (40-μm thick) were sliced using a sliding microtome on a frozen stage and collected into a cryopreservative solution and stored at −20°C. Animals in the behavioral portion of the experiment also underwent a cresyl violet lesion analysis. At 24 h post-CCI, the rats in the histological portion of this study were euthanized and underwent identical procedures and were examined for neuron degeneration, as well as astrocyte activation.
A series of sections were brush mounted on gelatin-subbed microscope slides, stained with cresyl violet, dehydrated, and cover slipped. The extent of the lesion was analyzed with an Olympus microscope (BX-51) and an Olympus 13.5 megapixel digital camera (DP-70). Images of sections throughout the extent of the injury coordinates were captured using the digital capturing system and area measures of the lesioned tissue were determined using the ImageJ software package (1.43u, NIH). The Calvalieri method was used to calculate the volumes of the ipsilateral cortex and the contralateral cortex.46 Four stereotaxic coordinates throughout the lesion, at approximately+1.7, +0.9, +0.1, and −0.7 mm relative to bregma, were selected for lesion analysis. The number of sections and the section thickness (40 μm) were multiplied by the mean area of the remaining cortex. The extent of cortical injury was measured by calculating the percent reduction in the injured ipsilateral cortex, compared with the contralateral cortex at each level using the following formula: 1 − (ipsi/contra) ×100). We have reliably shown that this technique is sensitive enough to detect treatment-induced reductions in injury size.6,9,13,47 Inter-rater reliability scores were calculated for approximately 25% of the data collected. Data were chosen randomly and the criterion for acceptance was a correlation coefficient of r>0.90.
A one-way ANOVA was completed where the between-subject factor of Treatment (NAM/Prog-injured, NAM-injured, Prog-injured, Vehicle-injured, and Sham) was used to analyze the lesion analysis data. Tukey's HSD were used to control for type I error and a significance level of p≤0.05 was used for all statistical analyses. Inter-rater reliability measures were evaluated using the bivariate correlation of scores obtained between the two raters. Post hoc analyses were conducted using Tukey's HSD for comparison of means whenever appropriate. A £ value of<0.05 was considered significant. All data are shown as mean scores±SEM.
Neuronal degeneration and astrocyte reactivity
Two series of frozen sections throughout the injury cavity (+1.2 and −0.2 mm, relative to bregma) were stained for degenerating neurons using Fluoro-Jade (FJ) B stain (Chemicon Int. Billerica, MA)48 following a standard protocol.6,7 The tissue went through a rehydration procedure prior to a 0.06% potassium permanganate solution for 10 min under agitation. Following the 0.06% potassium permanganate solution, there was another 1 min rinse before being transferred into the 0.004% FJ staining solution for 10 min. The slides were then rinsed with dH20 (3×1 min), allowed to dry for 15 min at 50°C, cleared in xylene for 1 min, and cover-slipped. Tissue sections were visualized with an Olympus fluorescent microscope (BX-51) system using a green fluorescent protein filter. Each section was viewed at 4× magnification, and then increased to 40×, before the image was captured with a 13 megapixel Olympus (DP-70) digital camera. The region of interest was set to be 300 μm ×300 μm. The number of FJ+cells within the captured field of view (90 mm2) were counted using ImageTool software (imagetool.software.informer.com).
Another series of frozen sections throughout the injury cavity (+1.5 and −0.1 mm, relative to bregma) were examined with a protocol for GFAP immunoreactivity to determine astrocyte expression following injury and standard protocol was followed.6,7 Tissue sections were mounted on subbed slides and dehydrated over several steps and placed in a citrate buffer at 90°C for 10 min. After cooling, the slides were placed in normal goat serum (Vector Laboratory Inc., Burlingame, CA, diluted 1:50) for 24 h, then incubated with a GFAP goat anti-rabbit (Dako, Carpinteria, CA, diluted 1:2000) for 48 h, rinsed in PBS+0.2% Triton-X (TX; 6×5 min) and incubated using goat anti-rabbit immunoglobulin G (Vector Laboratory Inc., Burlingame, CA, diluted 1:200) for 90 min. The slides were rinsed again (PBS+0.2% TX, 6×5 min), placed in an avidin biotin complex (Vector Laboratory Inc., Burlingame, CA) for 90 min and then rinsed with PBS+0.2 % TX (3×5 min) followed by a 0.1 PB (3×5 min) rinse. They were then reacted in a nickel-intensified chromagen solution containing acetate-imidizole buffer, 2.5 % nickel ammonium sulfate, 0.05% diaminobenzidine (DAB), and 0.01% H2O2. The slides were rinsed with 0.1 PB (3×1 min), allowed to dry for 15 min at 50° C., cleared in xylene for 1 min and then cover-slipped.
Tissue sections were visualized with an Olympus microscope (BX-51) system 4× magnification, and then increased to 40×, before the image was captured with a 13 megapixel Olympus (DP-70) digital camera. The regions of interest were set to be 300 μm ×300 μm. The number of GFAP+cells within the captured field of view (90 mm2) were counted using ImageTool software. There were six regions of interest examined; four in the ipsilateral cortex, surrounding the injury and two regions in the contralateral cortex. Cell counts for both the FJ+and GFAP+stained cells used selective process but without the use of unbiased stereology. Cell criterion included distinct cell type morphology and focus clarity of cell bodies of which the entire cell body was within the area of interest. Researchers who were blind to the treatment condition performed all counting. Inter-rater reliability scores were calculated for approximately 25% of the data collected. Data were chosen randomly and the criterion for acceptance was a correlation coefficient of r>0.90.
Cell counts (FJ+, GFAP+) from the six selected areas of interest for each slice (at two different coordinates) in the ipsilateral and contralateral cortex were used to determine the number of cells in each area. If no population of each cell type were found in an area of interest, tissue images were still captured and used in analysis. Cell counts were performed with ImageTool software.6,7 Counts were averaged across the slices from each coordinate for each hemisphere. One-way ANOVA tests were performed on all of the data. Independent variables were treatment (NAM/Prog-injured, NAM-injured, Prog-injured, Vehicle-injured, and Sham). Post hoc analyses were conducted using Fischer's Least Significant Difference for comparison of means whenever appropriate. A p value<0.05 was considered significant. All data are shown as mean scores±SEM. Although some of the tasks required subjective scoring, each scoring procedure was well defined with operational definitions. Additionally, 25% of the data were scored by a second, blind experimenter. These data were analyzed with a Pearson's correlation to assess the covariance between the two independent datasets.
Gene expression studies
Rats were randomly assigned to five groups and dosed as described above: a NAM-injured group (n=15), a Prog–injured group (n=15), a NAM/Prog-injured group, a Vehicle-injured group (n=15) and a Sham group (n=5). Animals were sacrificed and tissues collected at 24 h, 72 h, and 7 d after injury according to previously published protocols.23,41 At each time-point, five animals from each treatment group were sacrificed. Sham animals were sacrificed at 72 h so that they received an equivalent amount of experience with vehicle administration in order to account for any effects related to the stress response.
Animals were put under deep anesthesia with a mixture of CO2 (80%) and O2 (20%), a cardiac blood sample taken, and then decapitated. Brains were then rapidly extracted, sliced into a 4-mm coronal slab and placed on a cold plate, and a 5 mm tissue punch was taken, which included the injured cortex and small amount of peri-injury cortex. Tissue samples were then placed in centrifuge tubes, snap frozen, and stored at −80°C. Cardiac blood samples were collected at the time of sacrifice and from the tail vein at 72 h in the 7 d group to verify serum drug levels. Blood serum was separated by microcentrifuge, snap frozen and stored at −80°C. All samples were shipped by overnight carrier to the University of Washington on dry ice. NAM and Prog concentrations were assayed as described above.
The processing and analysis of the ribonucleic acid samples, the microarray analysis using the AffymetrixGeneChip® 3000 scanner (Affymetrix, Inc., Santa Clara, CA) and Bioconductor (Bioconductor, Inc., Seattle, WA) package p.adjust were performed as previously described.41 We carried out Gene Ontology (GO) category analysis via the cumulative hypergeometric distribution method to determine enhanced GO categories49 using the Bioconductor package GOstats.50 GO analysis shifts the emphasis from evaluation of single genes to evaluation of pathways, networks, and functions. We used differentially-expressed genes (more than 1.5-fold up or down regulated; p<0.05) for this analysis to identify GO categories by evidence of over-representation of significant genes. The validation of the data obtained with the microarrays was performed using fluorogenic 5′-nuclease–based assay and quantitative reverse transcription polymerase chain reaction (RT-PCR) as previously described.23,41 The RT-PCR data were normalized to the housekeeping genes, β-actin, and glyceraldehyde 3-phosphate dehydrogenase.
Results
NAM and Prog serum concentrations were 74 μg/mL (SEM=7) and 64 ng/mL (SEM=10), respectively, when sampled 72 h post-CCI in the monotherapy treatment groups. The NAM/Prog combination treatment resulted in a mean NAM serum concentration of 74 μg/mL (SEM=6) and a mean Prog serum concentration of 54 ng/mL (SEM=10) when sampled 72 h post-CCI. Serum concentration in the Vehicle-treated and Sham groups had normative circulating NAM and Prog concentrations (i.e., less than 3 μg/mL or 3 ng/mL, respectively). Based on clinical studies in patients and/or healthy subjects, Prog and NAM serum concentration were in the range that would result from an infusion of 0.125 mg/kg/h of Prog51 or a 3 mg/d oral dose of NAM, respectively.52–54
Sensory assessment: Tactile adhesive removal task
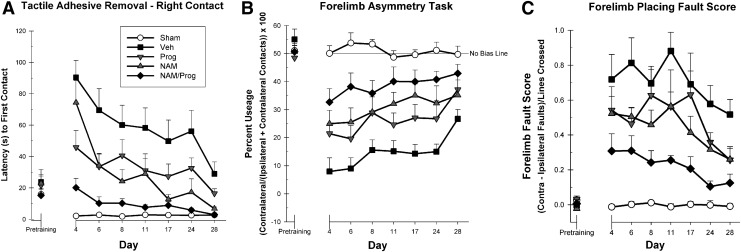
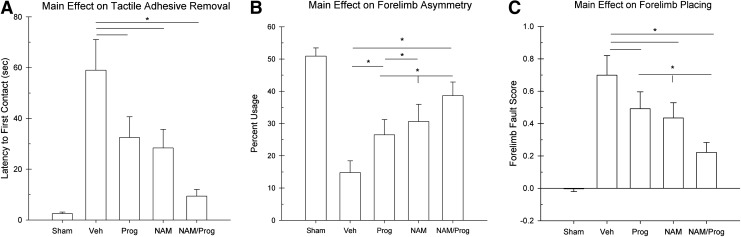
Using a repeated measures ANOVA, both the main effect of the within-subjects factor of Day (F [5.119, 209.861]=14.785; p<0.001) and Treatment (F [4, 41]=28.660; p<0.001) were significant. In addition, the Group×Day interaction also was significant, suggesting that the recovery rates for the Vehicle (F [6, 54]=4.263; p<0.001), the Prog (F [6, 48]=2.428; p<0.05), the NAM (F [6, 48]=8.170; p<0.001), and the NAM/Prog-treated (F [6, 48]=7.415; p<0.001) groups changed across days, whereas the recovery rates for the Sham group (F [6, 48]=0.607; p=0.724) did not change across days. The simple main effects revealed significant (p<0.05) differences between the Vehicle-treated and Sham groups on all days of testing. Additionally, the simple main effects revealed significant (p<0.05) differences between the NAM-treated and the Vehicle-treated animals on post-injury Days 6, 8, 11, 17, 24, and 28 and significant (p<0.05) differences between the Prog-treated and the Vehicle-treated animals on post-injury Days 4 and 6. More importantly, simple main effects revealed no significant differences between the NAM/Prog-treated group and the Sham animals on all days of testing. Performance on this task is shown in Figures 1A and 2A.
FIG. 1.
Functional recovery assessments. (A) Treatment improves sensorimotor function evidenced by shorter latencies (±standard error of the mean [SEM]) to remove the adhesive from the impaired forelimb in the tactile adhesive removal task. All groups were significantly different (p<0.05) than the Vehicle group. The nicotinamide (NAM)/progesterone (Prog)-treated group was significantly different than both individual treatment groups and not significantly different than the Sham group. (B) Treatment improves fine motor performance indicated by the percent (±SEM) usage bias for the unimpaired forelimb in the forelimb asymmetry task using the following formula: Contralateral contacts/(Contralateral contacts+ipsilateral contacts) ×100. All groups were significantly different (p<0.05) than the Vehicle group. The NAM/Prog-treated group was significantly different than the Prog-treated group. (C) Treatment improves fine motor performance evidenced by a reduction in fault scores for the impaired forelimb (±SEM) in the locomotor placing task using the following formula: (Contralateral faults – ipsilateral faults)/lines crossed. All groups were significantly different (p<0.05) than the Vehicle group. The NAM/Prog-treated group was significantly different than both of the individual treatment groups.
FIG. 2.
Functional recovery assessments: main effect graphs. Treatment improves sensorimotor function evidenced by shorter latencies (±standard error of the mean [SEM]) to remove the adhesive from the impaired forelimb in the tactile adhesive removal task as shown collapsed across all days. All groups were significantly different (*p<0.05) than the Vehicle group. The nicotinamide (NAM)/ progesterone (Prog)-treated group was significantly different than both individual treatment groups and not significantly different than the Sham group. (B) Treatment improves fine motor performance indicated by the percent (±SEM) usage bias in the forelimb asymmetry task as shown collapsed across days. All groups were significantly different (*p<0.05) than the Vehicle group. The NAM/Prog-treated group was significantly different than the Prog-treated group. (C) Treatment improves fine motor performance as evidenced by a reduction in fault scores collapsed across days. All groups were significantly different (*p<0.05) than the Vehicle group. The NAM/Prog-treated group was significantly different than both of the individual treatment groups.
Motor assessment: Forelimb asymmetry task
A bias score was used to determine the amount of reliance on the “uninjured limb” (ipsilateral to injury). Using a repeated measures ANOVA, both the main effect of the within-subjects factor of Day (F [5.818, 238.558]=5.132; p<0.001) and Treatment (F [4, 41]=31.105; p<0.001) were significant. The interaction of Day×Treatment was not significant (F [23.274, 238.558]=0.937; p=0.549). Post hoc analysis for the main effect of treatment was done to determine specific treatment effects. The Vehicle-treated animals performed significantly worse than the Sham-treated (HSD [17]=36.077; p<0.001), the Prog-treated (HSD [17]=11.694; p<0.05), NAM-treated (HSD [17]=15.796; p<0.001), and NAM/Prog-treated (HSD [17]=28.823; p<0.001) animals on all testing days.
Further analysis indicated that the NAM/Prog-treated group performed significantly better than the Prog-treated group (HSD [16]=12.129; p<0.05) on all days of testing. Although significant improvements were seen in the NAM and the Prog-treated groups, it is evident that the NAM/Prog group performed significantly better than the Prog-treated animals in this task. Performance on this task is shown in Figure 1B and Figure 2B.
Motor assessment: Locomotor placing task
The animal's right and left forelimb faults and total steps were counted and evaluated as a percent of total steps that were faults on the grid surface. Using a repeated measures ANOVA, both the main effect of the within-subjects factor of Day (F [5.448, 223.365]=5.665; p<0.001) and Treatment (F [4, 41]=29.055; p<0.001) were significant. The interaction of Day×Treatment was not significant (F [21.792, 223.365]=0.798; p=0.726). Post hoc analysis for the main effect of treatment was done to determine specific treatment effects. The Vehicle-treated animals performed significantly worse than the Sham-treated (HSD [17]=0.7024; p<0.001), Prog-treated (HSD [17]=0.208; p<0.05), NAM-treated (HSD [17]=0.265; p<0.01), and NAM/Prog-treated (HSD [17]=0.478; p<0.001) animals on all testing days. Further analysis indicated that the NAM/Prog-treated group performed significantly better than the NAM-treated (HSD [16]=0.213; p<0.05) and Prog-treated (HSD [16]=0.270; p<0.01) groups on all days of testing. Although significant improvements were seen in the NAM-treated and the Prog-treated groups, it is evident that the NAM/Prog group performed significantly better than both the NAM-treated and the Prog-treated animals in this task. Performance on this task is shown in Figure 1C and Figure 2C.
Histological analyses
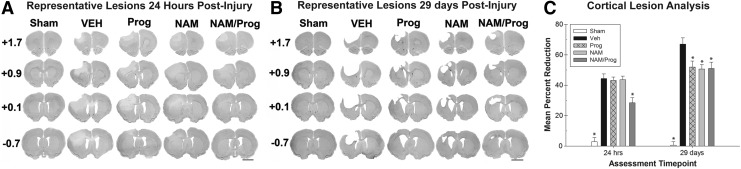
A lesion analysis was completed with the histological portion of the experiment 24 h post-injury, as well as following the behavioral assessment 29 d post-injury. Representative images of cresyl-stained coronal slices demonstrating damage 24 h post-injury are displayed in Figure 3A. For the ratio of lesion volume in the cortices 24 h post-injury, a one-way ANOVA indicated a significant difference between the treatments (F [4, 25]=29.912; p<0.001). More specifically, the Vehicle-treated animals were significantly different than the Sham-treated (HSD [10]=41.515, p<0.001) and the NAM/Prog-treated (HSD [10]=15.912; p<0.05) groups. Neither the NAM-treated (HSD [10]=0.789; p=1.000) or the Prog-treated (HSD [10]=1.209; p=0.999) groups were significantly different than the Vehicle-treated group. The remaining comparisons did not yield p values of less than 0.05 (Fig. 3C).
FIG. 3.
Lesion analysis. (A) Representative images of cresyl violet–stained (24 h post- cortical contusion injury [CCI]) tissue throughout the injury coordinates: +1.7 mm, +0.9 mm, +0.1 mm, and −0.7 mm, coordinates (anterior/posterior) relative to bregma; scale bar=3.0 mm. (B) Representative images of cresyl violet–stained (24 h post-CCI) tissue throughout the same coordinates. (C) Treatment significantly decreases the percent reduction of cortical volume (±standard error of the mean) between the ipsilateral and contralateral sides to the injury following 24 h and 29 d post-injury using the following formula: 1 - (ipsilateral/contralateral) ×100). At 24 h post-injury, the Sham-treated and the nicotinamide (NAM)/ progesterone (Prog)-treated groups were significantly different (p<0.05) than the Vehicle-treated group. At 29 d post-injury, all groups were significantly different (p<0.05) than the Vehicle group.
Representative images of cresyl-stained coronal slices demonstrating damage 29 d post-injury are displayed in Figure 3B. A one-way ANOVA indicated a significant difference between the treatments for the ratio of lesion volume in the cortices 29 d post-injury (F [4, 41]=61.667; p<0.001). Post hoc analysis indicated that the Sham (HSD [10]=66.437; p<0.001), the NAM (HSD [10]=16.378; p<0.01), the Prog (HSD [10]=15.098; p<0.05), and the NAM/Prog-treated groups (HSD [10]=15.978; p<0.01) had a significantly smaller mean percent reduction when compared with the Vehicle-treated group (Fig. 3C).
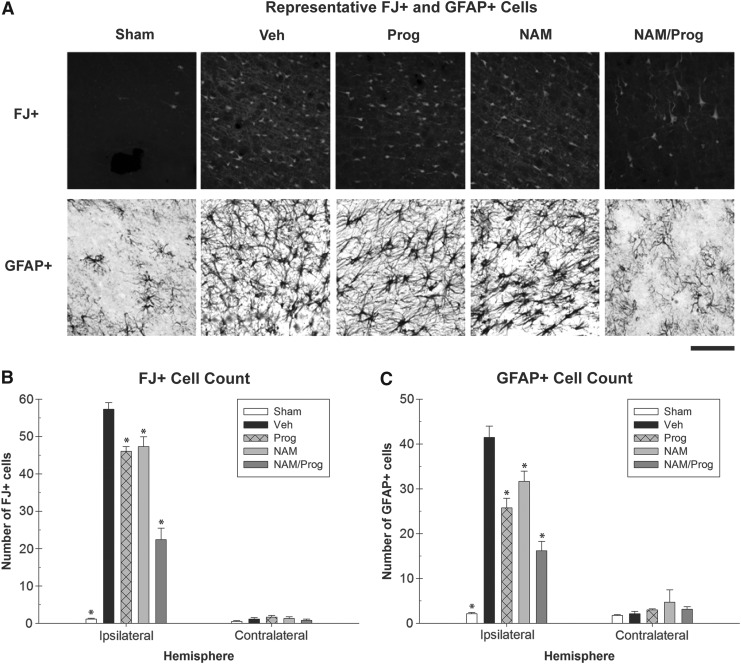
Degenerating neurons in the cortices both ipsilateral and contralateral to the injury were measured by counting the number of FJ+cells within specific regions of the cortices (Fig. 4A). A one-way ANOVA of the ipsilateral cortices indicated that there were significant differences in the number of FJ+ cells between treatment groups (F [4, 26]=101.688; p<0.001). Post hoc analysis indicated that the Sham-treated (HSD [10]=56.188; p<0.001), NAM-treated (HSD [10]=9.979; p<0.05), Prog-treated (HSD [10]=11.250; p<0.05), and NAM/Prog-treated (HSD [11]=34.902; p<0.001) groups had significantly fewer FJ+ cells when compared with the Vehicle-treated group. Further analysis indicated that the NAM/Prog-treated group had significantly fewer FJ+cells than the Prog-treated group (HSD [11]=23.652; p<0.001) and the NAM-treated group (HSD [11]=24.923; p<0.001; Fig. 4B). Analysis of the contralateral cortex showed that there were not significant differences in the number of FJ+cells between treatment groups (F [4, 26]=1.061; p=0.396).
FIG. 4.
Pathophysiological analyses. (A) Photomicrograph representing Fluoro-Jade (FJ)+and glial fibrillary acidic protein (GFAP)+stained cell bodies for each treatment group 24 h post-injury; scale bar=100 μm. (B) Treatment reduces the number (±standard error of the mean [SEM]) of FJ+degenerating neurons in the cortices both ipsilateral and contralateral to the injury. In the ipsilateral hemisphere, all groups were significantly different (p<0.05) than the Vehicle group. The nicotinamide (NAM)/progesterone (Prog)-treated group was significantly different than both of the individual treatment groups. There were no significant differences in the contralateral hemisphere. (C) Treatment reduces the number (±SEM) of GFAP+reactive astrocytes in the cortices both ipsilateral and contralateral to the injury. In the ipsilateral hemisphere, all groups were significantly different (p<0.05) than the Vehicle group. The NAM/Prog-treated group was significantly different than both of the individual treatment groups. There were no significant differences in the contralateral hemisphere.
Reactive astrocytes in the cortices both ipsilateral and contralateral to the injury were measured by counting the number of GFAP+cells within specific regions of the cortices (Fig. 4A). A one-way ANOVA of the ipsilateral cortices indicated significant differences in the number of GFAP+cells between treatment groups (F [4, 26]=46.675; p<0.001). Post hoc analysis indicated that the Sham (HSD [10]=39.122; p<0.001), the NAM (HSD [10]=9.792; p<0.05), the Prog (HSD [10]=15.708; p<0.001), and the NAM/Prog-treated groups (HSD [11]=25.280; p<0.001) had significantly fewer GFAP+cells when compared with the Vehicle-treated group. Further analysis indicated that the NAM/Prog-treated group had significantly fewer GFAP+cells than the Prog (HSD [11]=9.571; p<0.05) and the NAM-treated group (HSD [11]=15.488; p<0.001; Fig. 4C). Analysis of the contralateral cortex showed that there were not significant differences in the number of GFAP+cells between treatment groups (F [4, 26]=0.688; p=0.607).
Gene expression
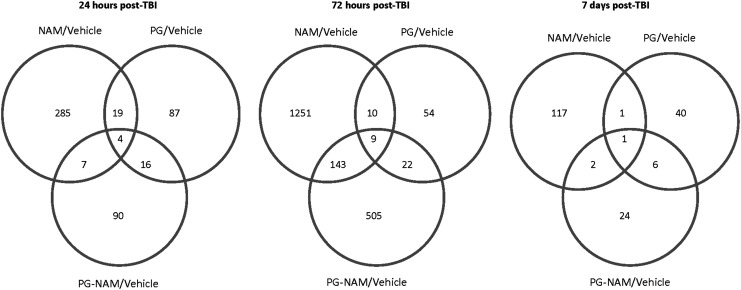
The microarray data passed all the standard and advanced quality control metrics. The number of differentially expressed genes (>1.5-fold change; p<0.05) at 24 h, 72 h, and 7 d are presented in Table 1. The vehicle to sham comparison reflects the effect of the TBI without treatment, relative to sham controls. The comparison of CCI animals that received treatment to those that received vehicle evaluates the effect of treatment on gene expression in the context of TBI. The most significant effect of the NAM/Prog treatment was at the 72 h time-point; the top GO categories are given in Table 2. GO analysis identified a significant effect of NAM monotherapy on signaling and transport pathways and cell-to-cell adhesion while Prog monotherapy primarily affected inflammatory and immune pathways. The combination of NAM and Prog resulted in a significant increase in the number of differentially-expressed genes involved in inflammatory processes, compared with Prog alone; increasing from 4 to 62 significant genes of the 355 genes identified in the inflammatory response pathway (GO:0030595) and from 6 to 64 significant genes of 1193 identified in the immune response pathway (GO:0006955).
Table 1.
The Number of Differentially Expressed Genes Probes: >1.5-Fold Up or Down; p<0.05
| 24 h | 72 h | 7 d | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Down | Up | Total | Down | Up | Total | Down | Up | Total | |
| Vehicle/Sham | 1261 | 1555 | 2816 | 2180 | 2618 | 4798 | 1309 | 2012 | 3321 |
| NAM/Vehicle | 170 | 145 | 315 | 1086 | 327 | 1413 | 52 | 69 | 121 |
| Prog/Vehicle | 85 | 41 | 126 | 77 | 18 | 95 | 19 | 29 | 48 |
| NAM-Prog/Vehicle | 48 | 69 | 117 | 469 | 210 | 679 | 14 | 19 | 33 |
NAM, nicotinamide; Prog, progesterone.
Table 2.
Gene Ontology (GO) Analysis 72 h Post-Traumatic Brain Injury
| GO ID | GO Term | Annotated | Significant | Expected | p value |
|---|---|---|---|---|---|
| NAMVehicle | |||||
| GO:0035637 | multicellular organismal signaling | 573 | 148 | 39.1 | 5.82e-49 |
| GO:0007268 | synaptic transmission | 452 | 124 | 30.8 | 8.09e-44 |
| GO:0006836 | neurotransmitter transport | 121 | 41 | 8.26 | 1.11e-18 |
| GO:0051969 | regulation of transmission of nerve impulse | 247 | 60 | 16.9 | 1.39e-18 |
| GO:0007215 | glutamate receptor signaling pathway | 47 | 21 | 3.21 | 5.92e-13 |
| GO:0006816 | calcium ion transport | 232 | 44 | 15.8 | 4.27e-10 |
| GO:0048167 | regulation of synaptic plasticity | 115 | 29 | 7.85 | 4.62e-10 |
| GO:0051932 | synaptic transmission, GABAergic | 32 | 13 | 2.18 | 6.54e-08 |
| GO:0016337 | cell-cell adhesion | 296 | 45 | 20.2 | 2.98e-07 |
| GO:0032412 | regulation of ion transmembrane transporter activity | 77 | 19 | 5.25 | 6.68e-07 |
| ProgVehicle | |||||
| GO:0007186 | G-protein coupled receptor signaling pathway | 1629 | 9 | 2.43 | 0.000245 |
| GO:0030595 | leukocyte chemotaxis | 99 | 3 | 0.148 | 0.000407 |
| GO:0006954 | inflammatory response | 355 | 4 | 0.531 | 0.001670 |
| GO:0050729 | positive regulation of inflammatory response | 61 | 2 | 0.0913 | 0.003690 |
| GO:0002376 | immune system process | 1193 | 6 | 1.79 | 0.006320 |
| GO:0006935 | chemotaxis | 305 | 3 | 0.457 | 0.010000 |
| GO:0043547 | positive regulation of GTPase activity | 118 | 2 | 0.1770 | 0.013200 |
| GO:0043087 | regulation of GTPase activity | 164 | 2 | 0.2460 | 0.024600 |
| GO:0030097 | hemopoiesis | 482 | 3 | 0.7220 | 0.033600 |
| GO:0033121 | regulation of purine nucleotide catabolic process | 189 | 2 | 0.283 | 0.031900 |
| NAM/Prog Vehicle | |||||
| GO:0006954 | inflammatory response | 355 | 62 | 11.3 | 5.25e-29 |
| GO:0002376 | immune system process | 1193 | 108 | 37.9 | 8.82e-25 |
| GO:0006955 | immune response | 601 | 64 | 19.1 | 5.65e-18 |
| GO:0033993 | response to lipid | 754 | 67 | 23.9 | 8.92e-15 |
| GO:0007159 | leukocyte cell-cell adhesion | 36 | 15 | 1.14 | 7.88e-14 |
| GO:0051050 | positive regulation of transport | 507 | 49 | 16.1 | 2.57e-12 |
| GO:0050900 | leukocyte migration | 153 | 24 | 4.86 | 8.46e-11 |
| GO:0001816 | cytokine production | 363 | 38 | 11.5 | 8.64e-11 |
| GO:0006935 | chemotaxis | 305 | 34 | 9.69 | 1.66e-10 |
| GO:0001568 | blood vessel development | 430 | 40 | 13.7 | 9.57e-10 |
Prog, progesterone; NAM, nicotinamide.
In contrast, the combination of NAM and Prog decreased the number of significant genes involved in pathways significant in NAM monotherapy. The number of differentially expressed genes involved in the multicellular organismal signaling (GO:0035637 comprised of a total of 573 genes) and synaptic transmission pathway (GO:0007268 comprised of a total of 452), GO categories decreased from 148 to 29 and from 124 to 12, respectively. In general, the combination increased the treatment effects of Prog and decreased the effects found with NAM monotherapy treatment. As shown in the Venn diagram (Fig. 5), there were an additional 500 gene probes differentially-expressed by NAM/Prog treatment that were not significantly altered by either NAM or Prog monotherapy. The effect of the NAM/Prog on differentially expressed genes is presented in Table 3 for those with greater than two-fold changes in expression. For the large majority of the genes identified, NAM/Prog decreased the gene expression of genes up-regulated by TBI (Vehicle/sham) at 72 h post-CCI. Specifically, there was a down-regulation of a number of genes involved in the inflammatory/immune response and blood vessel development.
FIG. 5.
The Venn diagrams show the number of genes whose expression was up- or down-regulated more than 1.5-fold (p<0.05) in the progesterone (Prog)/Vehicle, nicotinamide (NAM)/Vehicle, and Prog-NAM/Vehicle contrasts at the 24 h, 72 h, and 7 d time-points. Venn diagrams were generated with the Bioconductor limma package.
Table 3.
The Effect of Combined NAM and Prog Treatment on Unique Selected Genes of Interest at 72 h Post-TBI (2.0-Fold Change, p<0.05)
| Affymetrix ID | Gene Symbol | Genes | NAM+ProgaVehicle | VehicleaSham | Gene Ontology |
|---|---|---|---|---|---|
| 72 h | |||||
| 17759790 | Il1rn | interleukin 1 receptor antagonist | 0.22 | 24.01 | Inflammatory/Immune response |
| 17803496 | Pla2g2d | phospholipase A2, group IID | 0.28 | 5.49 | Lipid catabolic process |
| 17658217 | Scimp | SLP adaptor and CSK interacting membrane protein | 0.29 | 11.01 | Immunological synapse |
| 17649432 | Ccl7 | chemokine (C-C motif) ligand 7 | 0.31 | 20.01 | Inflammatory/Immune response |
| 17862599 | Trem1 | triggering receptor expressed on myeloid cells 1 | 0.33 | 15.21 | Neutrophil chemotaxis |
| 17786997 | Clec4d | C-type lectin domain family 4, member D | 0.33 | 4.65 | Immune response |
| 17792489 | Reg3g | regenerating islet-derived 3 gamma | 0.34 | 4.35 | Acute phase response |
| 17784380 | Cd8a | CD8a molecule | 0.35 | 8.71 | Immune response |
| 17779186 | Slpi* | secretory leukocyte peptidase inhibitor | 0.35 | 19.46 | Peptidase inhibitor activity |
| 17679492 | Serpinb8 | serpin peptidase inhibitor, clade B | 0.36 | 5.79 | Negative regulation of endopeptidase activity |
| 17776804 | Atp8b4 | ATPase, class I, type 8B, member 4 | 0.37 | 7.08 | ATP binding |
| 17851037 | Slc37a2 | solute carrier family 37 (glycerol-3-phosphate transporter), member 2 | 0.37 | 16.96 | Transmembrane transport |
| 17634203 | Anpep | alanyl (membrane) aminopeptidase | 0.38 | 16.05 | Blood vessel development |
| 17619793 | Calcb | calcitonin-related polypeptide, beta | 0.38 | 10.21 | Hormone activity |
| 17659402 | Ccl3 | chemokine (C-C motif) ligand 3 | 0.38 | 7.29 | Inflammatory response |
| 17787756 | Emp1 | epithelial membrane protein 1 | 0.38 | 12.92 | Cell growth |
| 17756767 | RT1-Bb | RT1 class II, locus Bb | 0.39 | 4.79 | Immune response |
| 17719197 | Mboat1 | membrane bound O-acyltransferase domain containing 1 | 0.41 | 11.66 | Integral component of membrane |
| 17740581 | S100a11 | S100 calcium binding protein A11 | 0.41 | 27.08 | Regulation of cell proliferation |
| 17725716 | Cd14 | CD14 molecule | 0.41 | 9.58 | Inflammatory/Immune response |
| 17770934 | Fcnb | ficolin B | 0.41 | 16.08 | Complement activation |
| 17638084 | Adam8 | ADAM metallopeptidase domain 8 | 0.41 | 7.38 | Inflammatory/Immune response |
| 17622112 | Ifitm1 | interferon induced transmembrane protein 1 | 0.43 | 18.12 | Immune response |
| 17700983 | Plau | plasminogen activator, urokinase | 0.43 | 5.96 | Blood vessel development |
| 17786961 | Clec4a2 | C-type lectin domain family 4, member A2 | 0.43 | 7.43 | Immune response |
| 17865301 | Fn1 | fibronectin 1 | 0.43 | 8.00 | Blood vessel development |
| 17670122 | Kng1l1 | kininogen 1-like 1 | 0.44 | 2.44 | Inflammatory response |
| 17854969 | Il20rb | interleukin 20 receptor beta | 0.44 | 6.10 | Inflammatory/Immune response |
| 17808473 | Plin2 | perilipin 2 | 0.44 | 10.96 | Long chain fatty acid transport |
| 17633019 | Emp3 | epithelial membrane protein 3 | 0.45 | 9.62 | Cell growth |
| 17758158 | Tmem26 | transmembrane protein 26 | 0.45 | 5.62 | Integral component of membrane |
| 17784371 | Cd8b | CD8b molecule | 0.45 | 6.42 | Immune system |
| 17681515 | F5 | coagulation factor V | 0.45 | n.s. | Positive regulation of leukocyte chemotaxis |
| 17728071 | Hmox1 | heme oxygenase (decycling) 1 | 0.45 | 30.39 | Inflammatory/Immune response |
| 17866147 | Htr2b | 5-hydroxytryptamine (serotonin) receptor 2B, G protein-coupled | 0.45 | 7.29 | Positive regulation of transport |
| 17795527 | Ly49si1 | immunoreceptor Ly49si1 | 0.46 | 12.56 | Carbohydrate binding |
| 17855233 | Acpp | acid phosphatase, prostate | 0.46 | 3.95 | Nucleotide metabolic process |
| 17696859 | Hpse | heparanase | 0.46 | 3.78 | Blood vessel development |
| 17735298 | F2rl2 | coagulation factor II (thrombin) receptor-like 2 | 0.46 | 3.48 | Blood coagulation |
| 17846467 | Bcl2a1 | BCL2-related protein A1 | 0.46 | 9.28 | Immune response |
| 17630326 | Apoc1 | apolipoprotein C-I | 0.47 | 4.55 | Lipid transport |
| 17708162 | Msr1 | macrophage scavenger receptor 1 | 0.47 | 17.30 | Lipoprotein transport |
| 17784528 | Capg | capping protein (actin filament), gelsolin-like | 0.47 | 6.96 | Actin filament capping |
| 17657767 | Cd68 | Cd68 molecule | 0.48 | 23.25 | Cellular response to organic substance |
| 17795409 | Olr1 | oxidized low density lipoprotein (lectin-like) receptor 1 | 0.48 | 12.06 | Inflammatory response |
| 17697948 | Lgals3 | lectin, galactoside-binding, soluble, 3 | 0.48 | 61.65 | Immune response |
| 17636814 | Igsf6 | immunoglobulin superfamily, member 6 | 0.49 | 6.28 | Integral component of membrane |
| 17714504 | Hk3 | hexokinase 3 (white cell) | 0.49 | 4.42 | Glucose 6-phosphate metabolic process |
| 17765931 | Dusp2 | dual specificity phosphatase 2 | 0.49 | 3.22 | Regulation of apoptotic process |
| 17610985 | Arg1 | arginase, liver | 0.49 | 2.41 | Response to lipid |
| 17614190 | Plaur | plasminogen activator, urokinase receptor | 0.49 | 5.47 | Blood vessel development |
| 17653377 | Slc16a3 | solute carrier family 16, member 3 (monocarboxylic acid transporter 4) | 0.49 | 2.55 | Transmembrane transporter |
| 17649426 | Ccl2 | chemokine (C-C motif) ligand 2 | 0.49 | 82.63 | Inflammatory/Immune response |
| 17670575 | B3gnt5* | UDP-GlcNAc:betaGal beta-1,3-N-acetylglucosaminyltransferase 5 | 0.50 | 3.10 | Protein glycosylation |
| 17686562 | Fcgr2a | Fc fragment of IgG, low affinity IIa, receptor | 0.50 | 8.06 | Inflammatory/Immune response |
| 17730510 | Necab2 | N-terminal EF-hand calcium binding protein 2 | 2.01 | 0.50 | Calcium mediated signaling |
| 17870022 | C3 | complement component 3 | 2.03 | n.s. | Inflammatory/Immune response |
| 17705165 | Slitrk6 | SLIT and NTRK-like family, member 6 | 2.07 | 0.58 | Axonogenesis |
| 17722829 | Sh3rf2 | SH3 domain containing ring finger 2 | 2.10 | 0.55 | Negative regulation of phosphatase activity |
| 17813816 | Six3 | SIX homeobox 3 | 2.28 | n.s. | Negative regulation of Wnt signaling |
| 17757925 | Adora2a | adenosine A2a receptor | 2.31 | n.s. | Inflammatory/Immune response |
| 17738845 | Mme | membrane metallo-endopeptidase | 2.50 | n.s. | Beta amyloid metabolic process |
The numbers in these columns are ratios of gene expression of the “(Nam+Prog) versus Vehicle,” or “Vehicle versus Sham” experimental groups.
NAM, nicotinamide; Prog, progesterone; TBI, traumatic brain injury; ID, identification; n.s., non-significant.
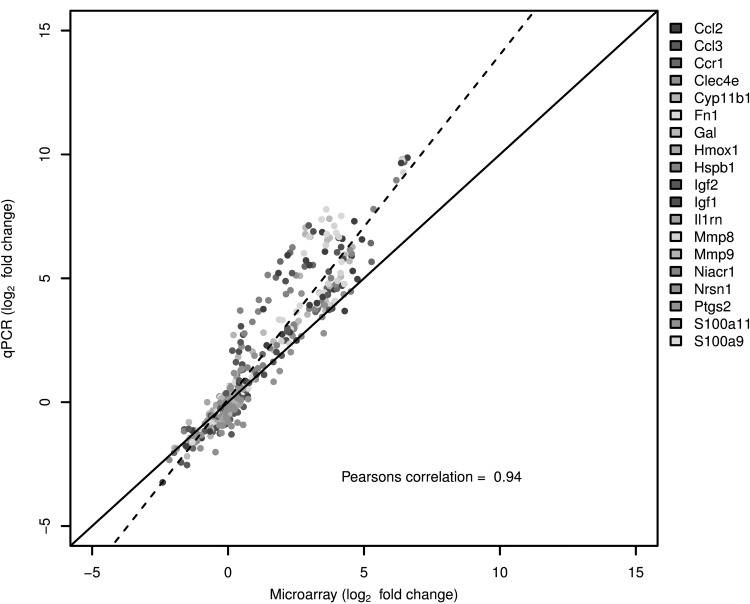
To validate gene expression changes, Figure 6 shows the gene expression of 19 genes selected from specific pathways of interest that are affected by TBI. The data generated via microarray and quantitative-PCR (qPCR) was normalized to GAPDH and β-actin. The qPCR findings were highly correlated with the microarray data; the Pearson correlation was 0.94 when normalized by either GAPDH or β-actin. Although the absolute change in expression was greater for the qPCR data, compared with the microarray data, possibly due to the higher detection sensitivity of qPCR, the direction of change (up-regulation or down-regulation) was consistent.
FIG. 6.
TaqMan based reverse transcription polymerase chain reaction (RT-PCR) validation of the microarray data for the selected genes: Ccl2 (chemokine (C-C motif) ligand 2), Ccl3 (chemokine (C-C motif) ligand 3), Ccr1 (chcmokine (C-C motif) receptor 1), Clec4e (C-type lectin domain family 4, member 3), Cyp11b1 (cytochrome P450 11b1), Fn1 (fibronectin 1), Gal (Galanin), Hmox1 (hemeoxygenase 1), Hspb1 (heat shock protein b1), Igf1 (insulin like growth factor 2), Igf2 (insulin like growth factor 2), Il1b (interleukin 1 beta), Il16 (interleukin 16), Il18 (interleukin 18), IL1rn (interleukin 1 receptor antagonist), Mmp8 (matrix metallopeptidase 8), Mmp9 (matrix metallopeptidase 9), Niacr1 (niacin receptor 1), Ptgs2 (prostaglandin-endoperoxide synthase 2), S100a19 (S100 calcium binding protein A9), and S100a11 (S100 calcium binding protein A11). The RT-PCR data shown in the figure was normalized to the housekeeping gene β-actin.
Discussion
The hypothesis was confirmed that the combination of NAM/Prog was not only beneficial following traumatic brain injury but more beneficial than either of these (NAM or Prog) neuroprotective therapies alone. The NAM treatment in this study either confirmed or was more beneficial than previous preclinical data in animal models of TBI.3–7,9,11–13,55 NAM treatment resulted in significant improvements in both of the fine motor assessments. In the forelimb asymmetry task, the NAM-treated animals performed similar to previous research.12,13 In the locomotor placing task, the NAM-treated animals performed significantly better than the Vehicle-treated animals on all days of testing and confirmed previous data in the locomotor placing task using either delayed13 or continuous NAM administration, with a lower dose in a unilateral parietal injury12 and a similar dose in a frontal injury model.11
Beneficial effects of NAM treatment also were found in the tactile adhesive removal task. In this task, NAM-treated animals showed significant improvements, compared with the Vehicle-treated animals but only after the 6 d post-injury time-point. In contrast, the majority of previous studies demonstrated significant improvements with NAM-treated animals initially (for approximately 7–14 d), but not later.6,12,13 By centering the injury over the sensorimotor cortex, this model leads to substantial and enduring behavioral deficits, which allow us to see the beneficial effects of NAM at later time-points in sensorimotor tasks.
At 24 h post-injury, NAM treatment did not significantly attenuate cortical loss, compared with the Vehicle-treated animals, similar to previous results with treatment of NAM at both the 50 and 500 mg/kg doses7 as well as with treatments at different time-points (15 min, 4 h, 8 h, and 24 h) in a window of opportunity study.13 Significant attenuation of cortical loss following NAM treatment has been shown as early as 7 d7 and as late as 30–40 d.6,9,11,12 Volumetric lesion analysis following behavioral assessment at 29 d post-injury, demonstrated that NAM treatment attenuated cortical loss, when compared with the Vehicle-treated animals, also confirming previous research exhibiting a reduction of lesion size up to six weeks post-injury.6,9,11,12 The acute histological analyses also demonstrated that NAM treatment provided neuroprotection and a reduction of activated astrocytes. FJ+cell counts resulted in a significantly lower number of degenerating neurons in NAM-treated animals, when compared with the Vehicle-treated animals as in previous studies.6,7
We also demonstrated a reduction in the number of GFAP+cells 24 h post-injury after NAM treatment, similar to previous research.7 It appears that although volumetric measurements of tissue do not reveal the neuroprotective effects of NAM at acute time-points, more precise cell counts of both FJ+and GFAP+neurons demonstrate the early stages of this neuroprotection.
There have been many preclinical experiments with Prog treatment demonstrating beneficial effects on recovery of function and neuroprotection over the past thirty years. Although the administration, window of opportunity, and dose varied, the current study produced similar results.9 In the forelimb asymmetry task, the Prog-treated animals performed significantly better than the Vehicle-treated animals on all days of testing. To our knowledge, this is the first time that beneficial effects have been shown on this task with Prog treatment following TBI. Prog-treated animals performed significantly better than the Vehicle-treated animals in the locomotor placing task on all days of testing, similar to previous research with a 16 mg/kg dose of Prog for 7 d, starting at 1 h post-injury.56,57 However, the current study was able to demonstrate these beneficial effects with a lower dose and later administration of Prog (4 h) as well as shorter duration (72 h). The Prog-treated group performed significantly better than the Vehicle-treated group on all days of testing. Previously, Shear and colleagues44 demonstrated significant improvements on a similar sensory task out to 20 d following injury with 4 mg/kg Prog treatment. Beneficial effects on this task also have been found on Days 1 and 7 post-injury with 16 mg/kg dosing.58 However, these beneficial effects only occurred with treatment for 5 d, not a 3-d treatment. The higher (10 mg/kg) dose for 3 d post-injury in the current study appears to be enough to replicate a lower dose (4 mg/kg) for 5 d.
A volumetric lesion analysis at 24 h post-injury showed that Prog treatment did not significantly reduce the injury cavity, compared with Vehicle treatment. The volumetric lesion analysis following the behavioral assessment at 29 d post-injury and demonstrated that Prog treatment attenuated cortical loss when compared with the Vehicle treatment. Previous research has established that Prog treatment leads to a reduction of necrosis, cell loss, and the size of the injury following three weeks post-injury.9,44,58 Results from the histological analysis at 24 h post-injury suggest that Prog does provide neuroprotection and reduced gliosis, compared with the Vehicle. A significant reduction in the number of FJ+stained cells was found following Prog treatment replicating several previous studies.56,59,60 A reduction of GFAP+reactive astrocytes also was found 24 h post-injury with Prog treatment, compared with Vehicle treatment, as was found in several previous studies.56,58–60 In addition, the Prog-treated group treated at had significantly fewer GFAP+stained astrocytes than the NAM-treated group at 24 h post-injury.
Overall, combination treatment with NAM and Prog provided both functional recovery and neuroprotection beyond both of each of the individual treatments. NAM/Prog-treated animals performed better than the individual treatment groups in the majority of the sensorimotor assessments, had reduced tissue loss (24 h post-injury), and a fewer number of FJ+and GFAP+cells in the ipsilateral hemisphere following unilateral sensorimotor CCI. Animals treated with NAM/Prog performed significantly better than Vehicle-treated animals on both fine motor movement tasks, the forelimb asymmetry task and the locomotor placing task, and the sensorimotor task on all assessment days. Further, the NAM/Prog-treated group was not significantly different than the experimental control group of uninjured animals (Sham) in the tactile adhesive removal task. The NAM/Prog-treated group was the only treatment group to show a significant reduction of cortical loss 24 h post-injury.
These results confirm previous studies that demonstrate that NAM and Prog treatment reduces neuronal degeneration and the glial response following injury. However, the current study also extended the findings to show that the NAM/Prog-treated group had significantly fewer FJ+neurons and a lower number of GFAP+stained astrocytes than both the NAM and Prog-treated groups. This suggests that the individual treatments can reduce neuronal degeneration and astrogliosis to a degree but that the combination of NAM/Prog is more effective than either treatment alone. In addition to the beneficial effects demonstrated with the behavioral and histological assessments employed in the current study, no abnormal behavior was noted following this combination treatment.
Previous gene expression studies with NAM treatment in the CCI model also identified a significant effect of NAM on immune and inflammatory response, altering genes associated with signaling pathways.23 In general, NAM treatment primarily counteracted gene expression changes due to the TBI by down-regulating genes that were significantly up-regulated in the injured, vehicle-treated animals, compared with sham animals. In contrast, Prog treatment primarily affected genes that were not differently expressed in the TBI injured animals.41 Prog altered genes involved in DNA damage response, positive regulation of cell proliferation, innate immune response, positive regulation of anti-apoptosis, and blood vessel remodeling. Therefore, the results of the previous gene expression studies suggested that Prog and NAM are excellent candidates for a combination treatment.
The significant increase in the number of genes that are down-regulated and are associated with the inflammatory/immune pathways and blood vessel development with the combination of NAM/Prog, compared with Prog and NAM treatment alone, is intriguing and suggests a possible mechanism for the improved neuroprotection. Preventing the inflammatory response is considered a high potential target for neuroprotection after TBI and the improvement in the recovery of function with the combination treatment is consistent with the improved neuroprotection.1,61 The effect of the NAM/Prog combination treatment on the expression of genes invoved in blood vessel development was not found with either NAM or Prog treatment alone.
TBI significantly increased the expression of plasminogen activating a major regulator of the component and coagulation cascade, heparanese, an enzyme that promotes arterial thrombosis in experimental models of vascular injury, fibronectin, a heparin sulfate proteoglycan, recently shown to be involved in the long term changes to brain vasculature in a experimental model of juvenile TBI.62 Jullienne and colleagues63 hypothesized that the long-term changes in cognitive dysfunction after TBI is due to changes in vasulcar dysfunction, along with changes at the blood brain barrier altering amyloid-beta clearance. Therefore, the effect of the combination treatment on blood vessel development may be responsible for the improvement in the recovery of function over the single treatments alone.
It appears that both NAM and Prog individually can reduce the amount of neuronal degeneration and inflammation initially, as well as tissue loss at later time-points. However, when combined, NAM and Prog are beneficial not only to the inflammatory response and degenerating neurons but also in reducing overall volumetric tissue loss at acute time-points. We are not the first to combine relevant combinations of treatments to administer following injury. There have been other instances of combination treatments showing benefit in the past,64–70 including a combination of Prog and vitamin D.64,65,68
In conclusion, we have demonstrated that a combination treatment of NAM and Prog provides improved neuroprotection and recovery of function in a variety of sensorimotor tasks, compared with NAM or Prog treatment alone. The current study provides a good initial assessment of the combination of NAM and Prog in a preclinical trial of controlled cortical impact of the sensorimotor cortex of the rat. However, these positive results require several follow-up studies. Further preclinical trials using NAM and Prog as a combination treatment should be conducted to identify the window of opportunity and to determine the optimal duration of treatment. It also may be the case that additional therapeutic strategies—such as additional drug interventions, environmental enrichment, and/or stem cell therapy— can improve recovery. In addition, the combination should show beneficial effects in multiple injury models (FPI, closed head injury, projectile, etc.), locations (bilateral-frontal, unilateral-parietal), and species. These results suggest that the scientific community should further explore the combination treatment of NAM and Prog following TBI in preclinical trials to advance progress towards reaching clinical trials.
Acknowledgments
The authors would like to thank William Maass and Lynn Moore for their assistance on this project. The research was supported by a grant from the National Institutes of Health/National Institute of Child, Health and Development (HD061944-01) and by the National Institute of Environmental Health Sciences Center for Ecogenetics & Environmental Health (P30ES007033).
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Schouten J.W. (2007). Neuroprotection in traumatic brain injury: a complex struggle against the biology of nature. Curr. Opin. Crit. Care 13, 134–142 [DOI] [PubMed] [Google Scholar]
- 2.Faden A. and Stoica B. (2007). Neuroprotection: Challenges and opportunities. Arch. Neurology 64, 794–800 [DOI] [PubMed] [Google Scholar]
- 3.Hoane M.R., Akstulewicz S.L., and Toppen J. (2003). Treatment with vitamin B3 improves functional recovery and reduces GFAP expression following traumatic brain injury in rats. J. Neurotrauma 20, 1189–1199 [DOI] [PubMed] [Google Scholar]
- 4.Hoane M.R., Gilbert D.R., Holland M.A., and Pierce J.L. (2006). Nicotinamide reduces acute cortical neuronal death and edema in the traumatically injured brain. Neurosci. Lett. 408, 35–39 [DOI] [PubMed] [Google Scholar]
- 5.Hoane M.R., Kaplan S.A., and Ellis A.L. (2006). The effects of nicotinamide on apoptosis and blood-brain barrier breakdown following traumatic brain injury. Brain Res. 1125, 185–193 [DOI] [PubMed] [Google Scholar]
- 6.Hoane M.R., Tan A.A., Pierce J.L., Anderson G.D., and Smith D.C. (2006). Nicotinamide treatment reduces behavioral impairments and provides cortical protection after fluid percussion injury in the rat. J. Neurotrauma 23, 1535–1548 [DOI] [PubMed] [Google Scholar]
- 7.Holland M.A., Tan A.A., Smith D.C., and Hoane M.R. (2008). Nicotinamide treatment provides acute neuroprotection and GFAP regulation following fluid percussion injury. J. Neurotrauma 25, 140–152 [DOI] [PubMed] [Google Scholar]
- 8.Quigley A., Tan A.A., and Hoane M.R. (2009). The effects of hypertonic saline and nicotinamide on sensorimotor and cognitive function following cortical contusion injury in the rat. Brain Res. 1304, 138–148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Peterson T.C., Anderson G.D., Kantor E.D., and Hoane M.R. (2012). A comparison of the effects of nicotinamide and progesterone on functional recovery of cognitive behavior following cortical contusion injury in the rat. J. Neurotrauma 29, 2823–2830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Swan A.A., Chandrashekar R., Beare J., and Hoane M.R. (2011). Preclinical efficacy testing in middle-aged rats: nicotinamide, a novel neuroprotectant, demonstrates diminished preclinical efficacy after controlled cortical impact. J. Neurotrauma 28, 431–440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vonder Haar C., Anderson G.D., and Hoane M.R. (2011). Continuous nicotinamide administration improves behavioral recovery and reduces lesion size following bilateral frontal controlled cortical impact injury. Behav. Brain Res. 224, 311–317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Goffus A.M., Anderson G.D., and Hoane M. (2010). Sustained delivery of nicotinamide limits cortical injury and improves functional recovery following traumatic brain injury. Oxid. Med. Cell. Longev. 3, 145–152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hoane M.R., Pierce J.L., Kauffman N.A., and Beare J. (2008). Variation in chronic nicotinamide treatment after traumatic brain injury can alter components of functional recovery independent of histological damage. Oxid Med Cell Longev 1, 46–53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maiese K. and Chong Z.Z. (2003). Nicotinamide: necessary nutrient emerges as a novel cytoprotectant for the brain. Trends Pharmacol. Sci. 24, 228–232 [DOI] [PubMed] [Google Scholar]
- 15.Ying W. (2008). NAD+/NADH and NADP+/NADPH in cellular functions and cell death: regulation and biological consequences. Antioxid. Redox Signal. 10, 179–206 [DOI] [PubMed] [Google Scholar]
- 16.Choi D.W. (1992). Excitotoxic cell death. J. Neurobiol. 23, 1261–1276 [DOI] [PubMed] [Google Scholar]
- 17.Raghupathi R. (2004). Cell death mechanisms following traumatic brain injury. Brain Pathol. 14, 215–222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kolthur-Seetharam U., Dantzer F., McBurney M.W., de Murcia G., and Sassone-Corsi P. (2006). Control of AIF-mediated cell death by the functional interplay of SIRT1 and PARP-1 in response to DNA damage. Cell Cycle 5, 873–877 [DOI] [PubMed] [Google Scholar]
- 19.Clark R.S., Vagni V.A., Nathaniel P.D., Jenkins L.W., Dixon C.E., and Szabo C. (2007). Local administration of the poly(ADP-ribose) polymerase inhibitor INO-1001 prevents NAD+depletion and improves water maze performance after traumatic brain injury in mice. J. Neurotrauma 24, 1399–1405 [DOI] [PubMed] [Google Scholar]
- 20.LaPlaca M.C., Zhang J., Raghupathi R., Li J.H., Smith F., Bareyre F.M., Snyder S.H., Graham D.I., and McIntosh T.K. (2001). Pharmacologic inhibition of poly(ADP-ribose) polymerase is neuroprotective following traumatic brain injury in rats. J. Neurotrauma 18, 369–376 [DOI] [PubMed] [Google Scholar]
- 21.Besson V.C., Croci N., Boulu R.G., Plotkine M., and Marchand-Verrecchia C. (2003). Deleterious poly(ADP-ribose)polymerase-1 pathway activation in traumatic brain injury in rat. Brain Res. 989, 58–66 [DOI] [PubMed] [Google Scholar]
- 22.Endres M., Wang Z.Q., Namura S., Waeber C., and Moskowitz M.A. (1997). Ischemic brain injury is mediated by the activation of poly(ADP-ribose)polymerase. J. Cereb. Blood Flow Metab. 17, 1143–1151 [DOI] [PubMed] [Google Scholar]
- 23.Anderson G.D., Peterson T.C., Farin F.M., Bammler T.K., Beyer R.P., Kantor E.D., and Hoane M.R. (2013). The effect of nicotinamide on gene expression in a traumatic brain injury model. Front. Neurosci. 7, 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.O'Connor C.A., Cernak I., and Vink R. (2005). Both estrogen and progesterone attenuate edema formation following diffuse traumatic brain injury in rats. Brain Res. 1062, 171–174 [DOI] [PubMed] [Google Scholar]
- 25.Vink R. and Van Den Heuvel C. (2004). Recent advances in the development of multifactorial therapies for the treatment of traumatic brain injury. Expert Opin. Investig. Drugs 13, 1263–1274 [DOI] [PubMed] [Google Scholar]
- 26.Garcia-Estrada J., Luquin S., Fernandez A.M., and Garcia-Segura L.M. (1999). Dehydroepiandrosterone, pregnenolone and sex steroids down-regulate reactive astroglia in the male rat brain after a penetrating brain injury. Int. J. Dev. Neurosci. 17, 145–151 [DOI] [PubMed] [Google Scholar]
- 27.Gibson C.L., Gray L.J., Bath P.M., and Murphy S.P. (2008). Progesterone for the treatment of experimental brain injury; a systematic review. Brain 131, 318–328 [DOI] [PubMed] [Google Scholar]
- 28.Wright D.W., Bauer M.E., Hoffman S.W., and Stein D.G. (2001). Serum progesterone levels correlate with decreased cerebral edema after traumatic brain injury in male rats. J. Neurotrauma 18, 901–909 [DOI] [PubMed] [Google Scholar]
- 29.Roof R.L., Duvdevani R., and Stein D.G. (1993). Gender influences outcome of brain injury: progesterone plays a protective role. Brain Res. 607, 333–336 [DOI] [PubMed] [Google Scholar]
- 30.Duvdevani R., Roof R.L., Fulop Z., Hoffman S.W., and Stein D.G. (1995). Blood-brain barrier breakdown and edema formation following frontal cortical contusion: does hormonal status play a role? J. Neurotrauma 12, 65–75 [DOI] [PubMed] [Google Scholar]
- 31.Asbury E.T., Fritts M.E., Horton J.E., and Isaac W.L. (1998). Progesterone facilitates the acquisition of avoidance learning and protects against subcortical neuronal death following prefrontal cortex ablation in the rat. Behav. Brain Res. 97, 99–106 [DOI] [PubMed] [Google Scholar]
- 32.Attella M.J., Nattinville A., and Stein D.G. (1987). Hormonal state affects recovery from frontal cortex lesions in adult female rats. Behav. Neural Biol. 48, 352–367 [DOI] [PubMed] [Google Scholar]
- 33.Fass B., Wrege K., Greenough W.T., and Stein D.G. (1980). Behavioral symptoms following serial or simultaneous septal-forebrain lesions: similar syndromes. Physiol. Behav. 25, 683–690 [DOI] [PubMed] [Google Scholar]
- 34.Stein D.G. (2011). Is progesterone a worthy candidate as a novel therapy for traumatic brain injury? Dialogues Clin. Neurosci. 13, 352–359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Feeser V.R. and Loria R.M. (2011). Modulation of traumatic brain injury using progesterone and the role of glial cells on its neuroprotective actions. J. Neuroimmunol. 237, 4–12 [DOI] [PubMed] [Google Scholar]
- 36.Roof R.L., Duvdevani R., Heyburn J.W., and Stein D.G. (1996). Progesterone rapidly decreases brain edema: treatment delayed up to 24 hours is still effective. Exp. Neurol. 138, 246–251 [DOI] [PubMed] [Google Scholar]
- 37.Guo Q., Sayeed I., Baronne L.M., Hoffman S.W., Guennoun R., and Stein D.G. (2006). Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp. Neurol. 198, 469–478 [DOI] [PubMed] [Google Scholar]
- 38.Papadopoulos M.C., and Verkman A.S. (2007). Aquaporin-4 and brain edema. Pediatr. Nephrol. 22, 778–784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Meffre D., Delespierre B., Gouezou M., Leclerc P., Vinson G.P., Schumacher M., Stein D.G., and Guennoun R. (2005). The membrane-associated progesterone-binding protein 25-Dx is expressed in brain regions involved in water homeostasis and is up-regulated after traumatic brain injury. J. Neurochem. 93, 1314–1326 [DOI] [PubMed] [Google Scholar]
- 40.Ganter S., Northoff H., Mannel D., and Gebicke-Harter P.J. (1992). Growth control of cultured microglia. J. Neurosci. Res. 33, 218–230 [DOI] [PubMed] [Google Scholar]
- 41.Anderson G.D., Farin F.M., Bammler T.K., Beyer R.P., Swan A.A., Wilkerson H.W., Kantor E.D., and Hoane M.R. (2011). The effect of progesterone dose on gene expression after traumatic brain injury. J. Neurotrauma 28, 1827–1843 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hoane M.R., Pierce J.L., Holland M.A., Birky N.D., Dang T., Vitek M.P., and McKenna S.E. (2007). The novel apolipoprotein E-based peptide COG1410 improves sensorimotor performance and reduces injury magnitude following cortical contusion injury. J. Neurotrauma 24, 1108–1118 [DOI] [PubMed] [Google Scholar]
- 43.Woodlee M.T., Asseo-Garcia A.M., Zhao X., Liu S.J., Jones T.A., and Schallert T. (2005). Testing forelimb placing “across the midline” reveals distinct, lesion-dependent patterns of recovery in rats. Exp. Neurol. 191, 310–317 [DOI] [PubMed] [Google Scholar]
- 44.Shear D.A., Galani R., Hoffman S.W., and Stein D.G. (2002). Progesterone protects against necrotic damage and behavioral abnormalities caused by traumatic brain injury. Exp. Neurol. 178, 59–67 [DOI] [PubMed] [Google Scholar]
- 45.Becerra G.D., Tatko L.M., Pak E.S., Murashov A.K., and Hoane M.R. (2007). Transplantation of GABAergic neurons but not astrocytes induces recovery of sensorimotor function in the traumatically injured brain. Behav. Brain Res. 179, 118–125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Coggeshall R.E. (1992). A consideration of neural counting methods. Trends Neurosci. 15, 9–13 [DOI] [PubMed] [Google Scholar]
- 47.Hoane M.R., Kaufman N., Vitek M.P., and McKenna S.E. (2009). COG1410 improves cognitive performance and reduces cortical neuronal loss in the traumatically injured brain. J. Neurotrauma 26, 1–129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gregersen N., Christensen M.F., Christensen E., and Kolvraa S. (1986). Riboflavin responsive multiple acyl-CoA dehydrogenation deficiency. Assessment of 3 years of riboflavin treatment. Acta Paediatr. Scand. 75, 676–681 [DOI] [PubMed] [Google Scholar]
- 49.Camon E., Magrane M., Barrell D., Lee V., Dimmer E., Maslen J., Binns D., Harte N., Lopez R., and Apweiler R. (2004). The Gene Ontology Annotation (GOA) Database: sharing knowledge in Uniprot with Gene Ontology. Nucleic Acids Res. 32, D262–D266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gentleman R., Carey V., Huber W., Irizarry R., and Dudoit S. (2005). In: Bioinformatics and Computational Biology Solutions Using R and Bioconductor. Gentleman R., Carey V., Huber W., Irizarry R., and Dudoit S. (eds). Springer Science-Business Media, Inc: New York, pps. 397–420 [Google Scholar]
- 51.Wright D.W., Ritchie J.C., Mullins R.E., Kellermann A.L., and Denson D.D. (2005). Steady-state serum concentrations of progesterone following continuous intravenous infusion in patients with acute moderate to severe traumatic brain injury. J. Clin. Pharmacol. 45, 640–648 [DOI] [PubMed] [Google Scholar]
- 52.Bernier J., Stratford M.R., Denekamp J., Dennis M.F., Bieri S., Hagen F., Kocagoncu O., Bolla M., and Rojas A. (1998). Pharmacokinetics of nicotinamide in cancer patients treated with accelerated radiotherapy: the experience of the Co-operative Group of Radiotherapy of the European Organization for Research and Treatment of Cancer. Radiother. Oncol. 48, 123–133 [DOI] [PubMed] [Google Scholar]
- 53.Horsman M.R., Hoyer M., Honess D.J., Dennis I.F., and Overgaard J. (1993). Nicotinamide pharmacokinetics in humans and mice: a comparative assessment and the implications for radiotherapy. Radiother. Oncol. 27, 131–139 [DOI] [PubMed] [Google Scholar]
- 54.Stratford M.R., Dennis M.F., Hoskin P.J., Saunders M.I., Hodgkiss R.J., and Rojas A. (1996). Nicotinamide pharmacokinetics in normal volunteers and patients undergoing palliative radiotherapy. Acta Oncol. 35, 213–219 [DOI] [PubMed] [Google Scholar]
- 55.Hoane M.R., Pierce J.L., Holland M.A., and Anderson G.D. (2008). Nicotinamide treatment induces behavioral recovery when administered up to 4 hours following cortical contusion injury in the rat. Neuroscience 154, 861–868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cutler S.M., Cekic M., Miller D.M., Wali B., VanLandingham J.W., and Stein D.G. (2007). Progesterone improves acute recovery after traumatic brain injury in the aged rat. J. Neurotrauma 24, 1475–1486 [DOI] [PubMed] [Google Scholar]
- 57.Wali B., Sayeed I., and Stein D.G. (2011). Improved behavioral outcomes after progesterone administration in aged male rats with traumatic brain injury. Restor Neurol Neurosci 29, 61–71 [DOI] [PubMed] [Google Scholar]
- 58.Cutler S.M., Vanlandingham J.W., and Stein D.G. (2006). Tapered progesterone withdrawal promotes long-term recovery following brain trauma. Exp. Neurol. 200, 378–385 [DOI] [PubMed] [Google Scholar]
- 59.Cutler S.M., Pettus E.H., Hoffman S.W., and Stein D.G. (2005). Tapered progesterone withdrawal enhances behavioral and molecular recovery after traumatic brain injury. Exp. Neurol. 195, 423–429 [DOI] [PubMed] [Google Scholar]
- 60.Djebaili M., Guo Q., Pettus E.H., Hoffman S.W., and Stein D.G. (2005). The neurosteroids progesterone and allopregnanolone reduce cell death, gliosis, and functional deficits after traumatic brain injury in rats. J. Neurotrauma 22, 106–118 [DOI] [PubMed] [Google Scholar]
- 61.Jennings J.S., Gerber A.M., and Vallano M.L. (2008). Pharmacological strategies for neuroprotection in traumatic brain injury. Mini Rev Med Chem 8, 689–701 [DOI] [PubMed] [Google Scholar]
- 62.Baker A.B., Gibson W.J., Kolachalama V.B., Golomb M., Indolfi L., Spruell C., Zcharia E., Vlodavsky I., and Edelman E.R. (2012). Heparanese regulates thrombosis in vascular injury and stent-induced flow disturbance. J. Am. Coll. Cardiol. 59, 1551–1560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Julienne A., Roberts J.M., Pop V., Murhy P.M., Head E., Bix G.J., and Badaut J. (2014). Juvenile traumatic brain injury induces long-term perivascular matrix changes alongside amyloid-beta accumulation. J. Cereb. Blood Flow Metab. 34, 1637–1645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Aminmansour B., Nikbakht H., Ghorbani A., Rezvani M., Rahmani P., Torkashvand M., Nourian M., and Moradi M. (2012). Comparison of the administration of progesterone versus progesterone and vitamin D in improvement of outcomes in patients with traumatic brain injury: A randomized clinical trial with placebo group. Adv. Biomed. Res. 1, 58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Atif F., Sayeed I., Ishrat T., and Stein D.G. (2009). Progesterone with vitamin D affords better neuroprotection against excitotoxicity in cultured cortical neurons than progesterone alone. Mol. Med. 15, 328–336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Barbre A.B. and Hoane M.R. (2006). Magnesium and riboflavin combination therapy following cortical contusion injury in the rat. Brain Res. Bull. 69, 639–646 [DOI] [PubMed] [Google Scholar]
- 67.Cekic M., Cutler S.M., VanLandingham J.W., and Stein D.G. (2011). Vitamin D deficiency reduces the benefits of progesterone treatment after brain injury in aged rats. Neurobiol. Aging 32, 864–874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Cekic M., Sayeed I., and Stein D.G. (2009). Combination treatment with progesterone and vitamin D hormone may be more effective than monotherapy for nervous system injury and disease. Front. Neuroendocrinol. 30, 158–172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hoane M.R., Raad C., and Barth T.M. (1997). Non-competitive NMDA antagonists and anti-oxidant drugs reduce striatal atrophy and facilitate recovery of function following lesions of the rat cortex. Restor. Neurol. Neurosci. 11, 71–-82 [DOI] [PubMed] [Google Scholar]
- 70.Chauhan N.B. and Gatto R. (2010). Synergistic benefits of erythropoietin and simvastatin after traumatic brain injury. Brain Res. 1360, 177–192 [DOI] [PubMed] [Google Scholar]