Abstract
Background:
Circulating tumour DNA (ctDNA) is an emerging candidate biomarker for malignancies and may be useful for monitoring the disease status of gastric cancer.
Methods:
We performed targeted deep sequencing of plasma cell-free DNA (cfDNA) by massively parallel sequencing in patients with tumours harbouring TP53 mutations. The quantitative values of TP53-ctDNA during the clinical course were compared with the tumour status.
Results:
Three out of ten patients with TP53 mutations in primary tumours showed detectable TP53 mutation levels in preoperative cfDNA. Although the cfDNA concentrations were not always reflective of the disease course, the ctDNA fraction correlated with the disease status.
Conclusions:
ctDNA may serve as a useful biomarker to monitor gastric cancer progression and residual disease.
Keywords: gastric cancer, circulating tumour DNA, next-generation sequencing, deep sequencing
Computed tomography (CT) is the major diagnostic tool for clinical management of gastric cancer. However, minimal residual disease and peritoneal dissemination, the most frequent recurrent patterns in gastric cancer, are typically difficult to detect with CT. Circulating tumour DNA (ctDNA)—fragmented DNA released from cancer cells into the bloodstream—is an emerging candidate marker to follow disease progression (Schwarzenbach et al, 2011). Previous studies have indicated that ctDNA levels correlate with tumour burden in colon cancer (Diehl et al, 2008) and breast cancer (Dawson et al, 2013). However, no comparative studies have been conducted in gastric cancer, and it is necessary to examine the possibility that ctDNA can be used for monitoring disease status, especially minimal residual disease and peritoneal dissemination. We previously developed a system to detect EGFR mutations in the plasma of lung cancer patients using a massively parallel sequencer (Kukita et al, 2013) and applied it to monitor advanced gastric cancer.
Materials and methods
Sample collection
Gastric cancer tissue and blood were prospectively collected from 42 patients who underwent gastrectomy for advanced gastric cancer at the Osaka University Hospital from April 2011 to November 2012. Tumour tissue was obtained from the primary lesion of the resected specimen immediately after surgery and stored at −80 °C as a fresh-frozen sample. Peripheral blood (5 ml) was drawn using an EDTA-2Na tube at admission (before surgery), before discharge (after surgery), and, in three patients, after recurrence. After removal of cellular debris and platelets by centrifugation, the plasma samples were stored at −80 °C until DNA extraction. Written informed consent for this study was obtained from all patients. The study was approved by the Ethics Committees of the Osaka University and the Osaka Medical Center for Cancer and Cardiovascular Diseases.
Mutation detection in the primary tumours
DNA was extracted using a DNA mini kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions, using serial 40-nm sections. TP53 exons 4–10 were amplified by PCR (Supplementary Table 1), and direct sequencing was performed with an ABI3730xl/ABI3100 using BD v3.1, following the EXO-1/SAP procedure.
Mutations in other genes were searched using Ion AmpliSeq Cancer Panel, version 1 (Life Technologies, Carlsbad, CA, USA), targeted for 739 hot-spot mutations in 604 loci in the 46 cancer-related genes, following the supplier's protocol.
Deep sequencing of the amplified target regions
Cell-free DNA was extracted from 1.2 ml of plasma using a Circulating Nucleic Acids Kit (Qiagen). DNA was eluted in a final volume of 40 μl. A volume of 10 μl was used for a single sequencing reaction. DNA concentrations were measured using a method based on LINE-1 copy number (Rago et al, 2007). The PCR primers were designed to generate final products ∼100 base pairs in length (Supplementary Table 2).
TP53 target regions were amplified from patient cell-free DNA or genomic DNA purified from the white blood cells obtained from normal individuals. They were then sequenced using a semiconductor sequencer as previously described (Kukita et al, 2013), maintaining the number of reads as >100 000. Data analysis was performed as described.
Results
Selection of patients for the assessment of gastric cancer progression
Given that gastric cancer somatic mutations occur most frequently in TP53, we chose TP53 as the target gene. Direct sequencing of the TP53 gene revealed that 10 (24%) of the 42 tumours analysed harboured mutations (Supplementary Table 3). Because cell-free DNA was available before surgical resection in 6 of these 10 cases, we performed cell-free DNA deep sequencing to evaluate the mutations identified in each primary tumour (Supplementary Figure 1, Supplementary Table 4). Given that one plasma sample corresponding to 1 ml of whole blood typically includes cell-free DNA corresponding to ∼5000 genomes deduced from the mean concentration in our lung cancer study (Kukita et al, 2013), base changes of <0.02% are considered background. Mutations in three of the six cases fulfilled this criterion. It may be noted that the number of reads with base changes of excluded cases did not exceed that corresponding to a single molecule estimated from the amount of DNA used for sequencing.
Detection threshold values were based on deep sequencing data from 24 normal samples as described previously (Kukita et al, 2013). We used DNA purified from white blood cells, because the error profile of white blood cells was not substantially different from that of cell-free DNA in our previous study. The threshold values obtained for base changes in 100 000 reads were as follows: 603 for c.103delT (patient 4), 11 for c.747G>C (patient 5), and 39 for c.166G>T (patient 8; Supplementary Table 5). Because the initial levels of all three cases exceeded the threshold, these cases were used for the follow-up study.
Comparison of cell-free DNA and ctDNA as indicators of disease progression
The median concentration of extracted cell-free DNA of all samples from the three cases was 43.1 ng ml−1 plasma (range 9.5–1338 ng ml−1). The mutant fraction range was 0.001–77.8% (median 0.90%).
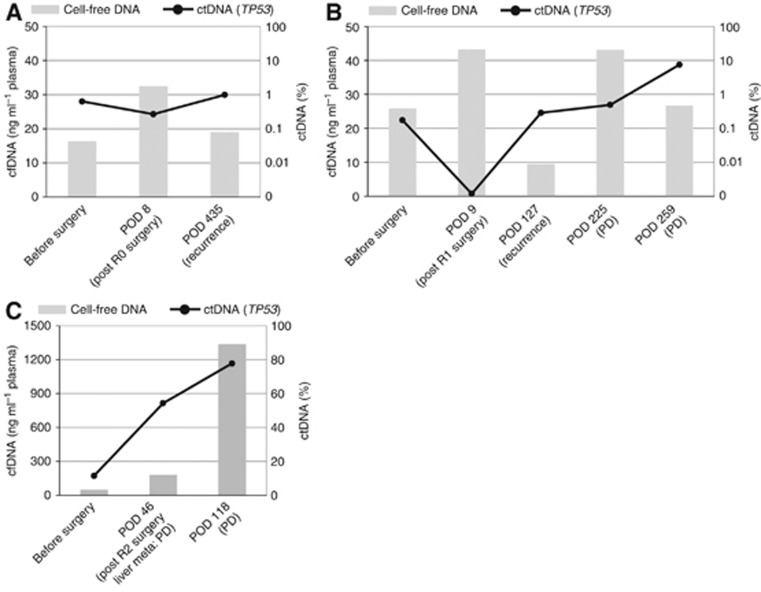
First, we compared cell-free DNA and ctDNA dynamics for the ability to monitor the treatment response (Figure 1). The cell-free DNA values fluctuated in all but one of the patients, regardless of the disease course. In one patient (patient 8), cell-free DNA increased along with the rapid progression of multiple liver metastases. In this patient, the majority of the cell-free DNA was derived from the tumour. By contrast, ctDNA dynamics were concordant with disease progression.
Figure 1.
Comparison of cell-free DNA and ctDNA with regard to treatment response. The levels of cell-free DNA (cfDNA; grey bar) and ctDNA (line graph) at various time points in patients 4 (A), 5 (B), and 8 (C) are displayed. The vertical axis on the left represents the concentration of cfDNA, and the vertical axis on the right represents the TP53-mutant fraction in cfDNA. The horizontal axis labels represent the time points of plasma sampling and tumour status (in parenthesis). The presence or absence of tumour after surgery is described by the R status (R0: no residual tumour, R1: microscopic residual tumour, R2: macroscopic residual tumour). Abbreviations: POD: postoperative day; PD: progressive disease.
ctDNA validation using additional mutant genes
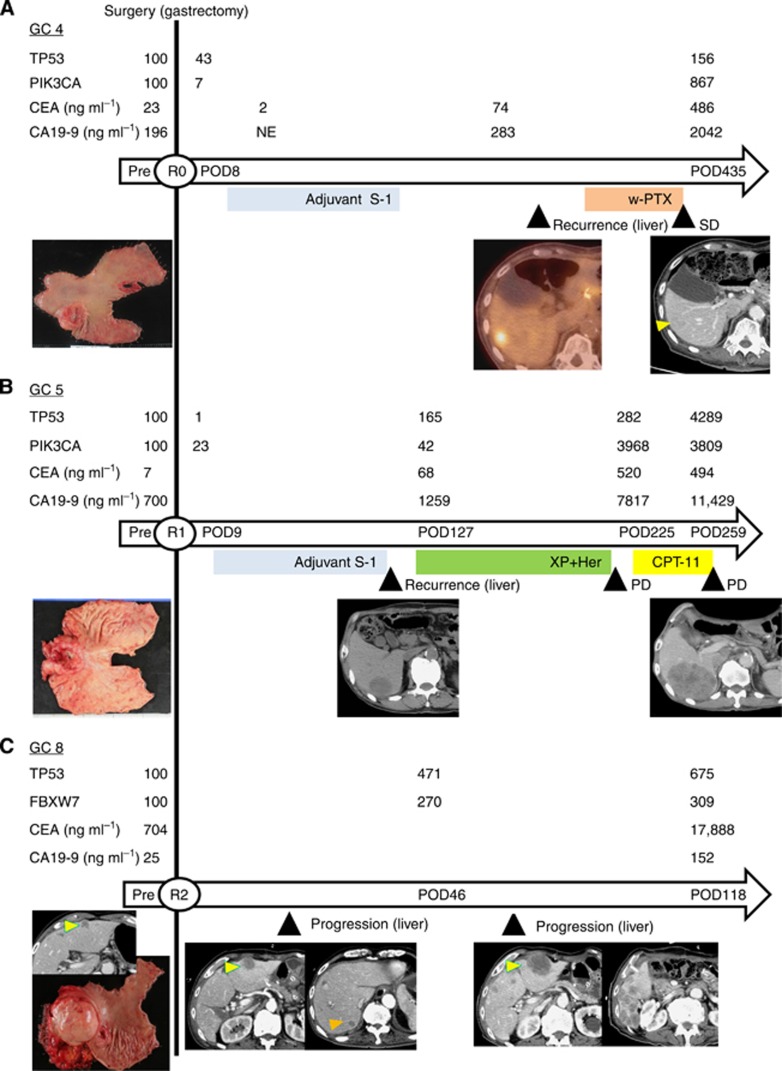
For the three cases, mutations in genes other than TP53 were surveyed with the Ion AmpliSeq Cancer Panel. The results are shown in Supplementary Table 6. We focused on PIK3CA c.1633G>A (found in patients 4 and 5) and FBXW7 c.1177C>T (found in patient 8) and confirmed these mutations by direct sequencing (Supplementary Table 1). We analysed the same plasma samples, which were subjected to TP53 analysis, by deep sequencing of the new genes. ctDNA fraction values estimated from the assay results of two different genes (TP53 and PIK3CA/FBXW7) were within one order of magnitude in 8 (73%) of the 11 samples, suggesting a good dynamic concordance of the different genes (Figure 2).
Figure 2.
The relationship between ctDNA and clinical observations during the treatment course. ctDNA values during the clinical course are depicted in comparison with serum tumour markers, treatment, response, and tumour burden (specimen photos and radiological images). The horizontal arrows indicate the time scale for each case. The day of gastrectomy is represented by the vertical line. The presence or absence of tumour after surgery is described by the R status (R0: no residual tumour, R1: microscopic residual tumour, R2: macroscopic residual tumour). The intervals from surgery are indicated inside the arrows. Chemotherapy is depicted by colour bars under the time scale. The specimen photos depict the resected stomach, representing the tumour burden before surgery. CT (A, B, and C) and positron emission tomography (PET)-CT (A) images display the status of metastatic liver tumours at each assessment point, indicated by triangle markers. In each case, ctDNA is expressed as the percent relative to the ctDNA values before surgery. POD: postoperative day, PD: progressive disease, XP: capecitabine+cisplatin, Her: trastuzumab, CPT-11: irinotecan, w-PTX: weekly administration of paclitaxel. (A) The ctDNA fraction was measured for both TP53 (c.103delT) and PIK3CA (c.1633G>A). (B) The ctDNA fraction was measured for both TP53 (c.747G>C) and PIK3CA (c.1633G>A). (C) The ctDNA fraction was measured for both TP53 (c.155G>T) and FBXW7 (c.1177C>T).
The relationship between ctDNA and clinical findings during the clinical course of gastric cancer
The clinical and genetic characteristics of the three cases are summarised in Table 1. CT images were evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST; Eisenhauer et al, 2009). The alterations in the ctDNA levels and clinical disease course are presented in Figure 2. Details of individual cases are provided below.
Table 1. Clinical and genetic characteristics of three cases assessed by follow-up study.
| Patient number. | Age | Gender | Histological type | p-Stage | Residual tumour after surgery | Gene mutation 1 | Gene mutation 2 | Postoperative chemotherapy | Metastasis site, number, timing | Treatment for metastasis | Responsea |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 |
86 |
Male |
Differentiated |
IIIC |
R0 |
TP53 c.103delT |
PIK3CA c.1633G>A |
Yes |
Liver, single, POD275 |
Chemotherapy |
SD |
| 5 |
73 |
Male |
Differentiated |
IVb |
R1 |
TP53 c.747G>C |
PIK3CA c.1633G>A |
Yes |
Liver, multiple, POD93 |
Chemotherapy molecular-target drug |
PD |
| 8 | 80 | Male | Differentiated | IVc | R2 | TP53 c.166G>T | FBXW7 c.1177C>T | No | Liver, multiple, (residue) | None | PD |
Abbreviations: PD=progressive disease; POD=postoperative day; R1=microscopic residual tumour; R2=macroscopic residual tumour; R0=no residual tumour (curative resection); SD=stable disease.
According to the Response Evaluation Criteria in Solid Tumors (RECIST).
Peritoneal cytology positive for carcinoma cells, and
Hepatic metastasis.
Case 1 (patient 4)
Curative distal gastrectomy was performed for a 55-mm tumour with multiple lymph node metastases, but the patient suffered from a single liver metastasis. ctDNA of TP53 c.103delT that was 0.63% before surgery, decreased to 0.27% (below the background noise level) after surgery, and increased to 0.98% after recurrence. Similar alterations were observed in the PIK3CA c.1633G>A ctDNA.
Case 2 (patient 5)
Distal gastrectomy was performed for a 65-mm tumour with multiple lymph node metastases. Intraoperative peritoneal cytology was positive. Despite postoperative S-1 chemotherapy, the patient developed multiple liver metastases. Recurrent tumours were treated with chemotherapy, but the disease progressed. ctDNA levels of TP53 c.747G>C and PIK3CA c.1633G>A were 0.17% and 0.84%, respectively, before surgery. ctDNA levels decreased after resection and increased after recurrence and tumour progression in both the genes. However, there was some discrepancy between the two genes after the use of trastuzumab.
Case 3 (patient 8)
A Stage IV gastric cancer with a 100-mm lymph node metastasis was removed by palliative gastrectomy. A 17-mm liver metastasis was left unresected. The liver tumours grew rapidly in size and number after surgery. The ctDNA levels of TP53 c.166G>T and FBXW7 c.1177C>T were 11% before surgery and continually increased with tumour progression, reaching 77.8%.
Discussion
In this report, we demonstrated that concentrations of ctDNA, not total plasma DNA, exhibited good correlation with the disease status of gastric cancer. We observed a decrease in ctDNA levels after surgical resection, indicating its potential application for the detection of residual disease. Tie et al (2014) demonstrated the possibility that recurrence of stage II colorectal cancer after surgical resection may be predicted by ctDNA. Similar applications may be possible for gastric cancer cases. Serological biomarkers such as CA19-9 and CEA are used to assist in the diagnosis of CT, but they have limitations owing to their low sensitivity (Shimada et al, 2013). In our study, ctDNA levels were parallel with these markers, suggesting a comparable ability to monitor the disease progression. However, from our analysis, it is still unknown whether ctDNA is advantageous over serological biomarkers because of our small number of patients.
There have been several studies on cell-free DNA obtained from the blood of gastric cancer patients, using concentration (Park et al, 2012), MYC/GAPDH ratio (Park et al, 2009), and promoter methylation status of MGMT, p15, and hMLH1 (Kolesnikova et al, 2008). These markers are not necessarily specific to cancer cells and therefore lack theoretical backgrounds for their validity. By contrast, cancer-related somatic mutations such as those in TP53 are specific to malignancies, and the DNA with these mutations is indicative for the presence of malignancies. Our results also agree with this reasoning; levels of the somatic mutations were correlated with surgical resection and disease progression, but concentrations of cell-free DNA were not.
The recent attention to ctDNA is due mainly to technical developments, especially that of digital PCR pioneered by BEAMing (Dressman et al, 2003). It is important to note that massively parallel sequencers may be regarded as digital PCR because they employ digital PCR in the template preparation step after global amplification of target DNA. Because of the much higher throughput of data production than with other digital PCR technologies and the considerable investment, the massively parallel sequencers should be the method of choice if several technical problems, such as read error (Junemann et al, 2013), are resolved.
Acknowledgments
This work was partly supported by KAKENHI 23591928.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on British Journal of Cancer website (http://www.nature.com/bjc)
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Supplementary Material
References
- Dawson S-J, Tsui DWY, Murtaza M, Biggs H, Rueda OM, Chin S-F, Dunning MJ, Gale D, Forshew T, Mahler-Araujo B, Rajan S, Humphray S, Becq J, Halsall D, Wallis M, Bentley D, Caldas C, Rosenfeld N. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med. 2013;368 (13:1199–1209. doi: 10.1056/NEJMoa1213261. [DOI] [PubMed] [Google Scholar]
- Diehl F, Schmidt K, Choti MA, Romans K, Goodman S, Li M, Thornton K, Agrawal N, Sokoll L, Szabo SA, Kinzler KW, Vogelstein B, Diaz LA., Jr Circulating mutant DNA to assess tumor dynamics. Nat Med. 2008;14 (9:985–990. doi: 10.1038/nm.1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dressman D, Yan H, Traverso G, Kinzler KW, Vogelstein B. Transforming single DNA molecules into fluorescent magnetic particles for detection and enumeration of genetic variations. Proc Natl Acad Sci USA. 2003;100 (15:8817–8822. doi: 10.1073/pnas.1133470100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur J Cancer. 2009;45 (2:228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- Junemann S, Sedlazeck FJ, Prior K, Albersmeier A, John U, Kalinowski J, Mellmann A, Goesmann A, von Haeseler A, Stoye J, Harmsen D. Updating benchtop sequencing performance comparison. Nat Biotechnol. 2013;31 (4:294–296. doi: 10.1038/nbt.2522. [DOI] [PubMed] [Google Scholar]
- Kolesnikova EV, Tamkovich SN, Bryzgunova OE, Shelestyuk PI, Permyakova VI, Vlassov VV, Tuzikov AS, Laktionov PP, Rykova EY. Circulating DNA in the blood of gastric cancer patients. Ann N Y Acad Sci. 2008;1137:226–231. doi: 10.1196/annals.1448.009. [DOI] [PubMed] [Google Scholar]
- Kukita Y, Uchida J, Oba S, Nishino K, Kumagai T, Taniguchi K, Okuyama T, Imamura F, Kato K. Quantitative Identification of mutant alleles derived from lung cancer in plasma cell-free DNA via anomaly detection using deep sequencing data. PloS One. 2013;8 (11:e81468. doi: 10.1371/journal.pone.0081468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JL, Kim HJ, Choi BY, Lee HC, Jang HR, Song KS, Noh SM, Kim SY, Han DS, Kim YS. Quantitative analysis of cell-free DNA in the plasma of gastric cancer patients. Oncol Lett. 2012;3 (4:921–926. doi: 10.3892/ol.2012.592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park KU, Lee HE, Park do J, Jung EJ, Song J, Kim HH, Choe G, Kim WH, Lee HS. MYC quantitation in cell-free plasma DNA by real-time PCR for gastric cancer diagnosis. Clin Chem Lab Med. 2009;47 (5:530–536. doi: 10.1515/CCLM.2009.126. [DOI] [PubMed] [Google Scholar]
- Rago C, Huso DL, Diehl F, Karim B, Liu G, Papadopoulos N, Samuels Y, Velculescu VE, Vogelstein B, Kinzler KW, Diaz Jr., LA Serial assessment of human tumor burdens in mice by the analysis of circulating DNA. Cancer Res. 2007;67 (19:9364–9370. doi: 10.1158/0008-5472.CAN-07-0605. [DOI] [PubMed] [Google Scholar]
- Schwarzenbach H, Hoon DS, Pantel K. Cell-free nucleic acids as biomarkers in cancer patients. Nat Rev Cancer. 2011;11 (6:426–437. doi: 10.1038/nrc3066. [DOI] [PubMed] [Google Scholar]
- Shimada H, Noie T, Ohashi M, Oba K, Takahashi Y. Clinical significance of serum tumor markers for gastric cancer: a systematic review of literature by the Task Force of the Japanese Gastric Cancer Association. Gastric Cancer. 2013;17 (1:26–33. doi: 10.1007/s10120-013-0259-5. [DOI] [PubMed] [Google Scholar]
- Tie J, Kinde I, Wang Y, Vogelstein B, Gibbs P. Circulating tumor DNA (ctDNA) as a marker of recurrence risk in stage II colon cancer (CC) J Clin Oncol. 2014;32 (suppl 5s:abstr 11015. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.