Abstract
Background:
We aim to report the prevalence of irritable bowel syndrome (IBS) and elucidate the influence of IBS on the incidence of colorectal neoplasm through a community-screening-based, longitudinal follow-up study.
Methods:
We enroled 39 384 community residents aged 40 years or older who had participated in a community-based colorectal cancer-screening programme with an immunochemical faecal occult test since 1999. We followed a cohort that was free of colorectal neoplasm (excluding colorectal neoplasm at baseline) to ascertain the incident colorectal neoplasm through each round of screening and used a nationwide cancer registry. Information on IBS was obtained by linking this screened cohort with population-based health insurance claim data. Other confounding factors were also collected via questionnaire or biochemical tests.
Results:
The overall period prevalence of IBS was 23%, increasing from 14.7% for subjects aged 40–49 years to 43.7% for those aged 70 years and more. After controlling for age, gender and family history of colorectal cancer, screenees who had been diagnosed as having IBS exhibited a significantly elevated level (21% adjusted hazard ratio (HR)=1.21 (95% CI: 1.02–1.42)) of incident colorectal adenoma compared with those who had not been diagnosed with IBS. A similar finding was noted for invasive carcinoma; however, the size of the effect was of borderline statistical significance (adjusted HR=1.20 (95% CI: 0.94–1.53)).
Conclusions:
IBS led to an increased risk for incident colorectal neoplasm.
Keywords: irritable bowel syndrome, colorectal neoplasm, risk factor
Irritable bowel syndrome (IBS) is a prevalent functional disorder; however, the epidemiological profile is still short of empirical data. The previous, population-based studies in Western countries have reported the prevalence of IBS in adults within a wide range between 3 and 22% (Oberndorff-Klein Woolthuis et al, 2004), in contrast to the range of 6.5–10.1% that was reported in a recent Asian study (Chang et al, 2010). The established risk factors include age, gender, education, social economic status, race, drug and psychological factors. Currently, few population-based studies have investigated the relationship between biological markers and IBS from a clinical viewpoint.
As IBS shares symptoms with other major gastrointestinal diseases, for example, colorectal neoplasia, it is worthwhile to evaluate the relationship between IBS and colorectal neoplasia in addition to the predisposing risk factors. Two studies from the United Kingdom and Denmark revealed a lack of relationship between IBS and colorectal cancer (CRC) after excluding CRCs within the first year of follow-up (Garcia Rodriguez et al, 2000; Nørgaard et al, 2011). However, little is known about the association of long-term IBS with the potential risk of developing colorectal neoplasia. To the best of our knowledge, few reports on the risk for colorectal neoplasia in large cohorts of IBS subjects with a long follow-up time exist. The aims of this study were to estimate the prevalence of IBS and to assess the risk factors associated with IBS using a population-based, integrated screening programme and to assess the risk of developing colorectal neoplasia in cohorts of subjects with IBS.
Materials and methods
Setting and study population
To build a prospective cohort, we recruited 39 384 community residents aged over 40 who participated in a Keelung community-based, integrated screening programme between 1999 and 2004. The details about the protocol of this multiple screening programme have been described in full elsewhere (Chen et al, 2004). To sum up, the programme was initially designed to screen five cancers (cervical, breast, oral, colorectal and liver cancer) and three chronic diseases (diabetes, hypertension and hyperlipidaemia). For CRC screening, the immunochemical faecal occult blood test (FIT) was applied for the early detection of colorectal neoplasia. Details of the protocol and screening results have been published previously (Chen et al, 2011). The 39 384 participants were invited to undergo CRC screening with FIT in a staggered entry with numbers of participants being 1967, 8370, 10 683, 7560, 5872 and 4932 in years 1999, 2000, 2001, 2002, 2003 and 2004, respectively. Those with positive FIT results were referred to undergo colonoscopy as the standard, confirmatory procedure for the detection of colorectal neoplasia. Note that, although the cohort for the following analysis is based on these screeness, they should be representative of the underlying population in Keelung as they were invited from the underlying population registry with such a large size. Even some selective factors may affect the uptake of CRC, but they would not affect the association between IBS and colorectal neoplasia as the participants who were invited were not aware of this study before. Moreover, the demographic features in the Keelung city are also representative of those of Taiwanese people because the incidence rates of several chronic diseases and cancers in Keelung city were close to those of Taiwanese people.
The screening procedures consisted of registration, fasting blood drawing, anthropometric measurement, questionnaire, physical examination and health condition. The contents of the questionnaire, including demographic characteristics, family history, dietary behaviour and lifestyle, were surveyed in each screening activity. A series of biochemical variables, including haemoglobin, platelets, white blood cells and fasting glucose, were collected after blood collection.
Study design
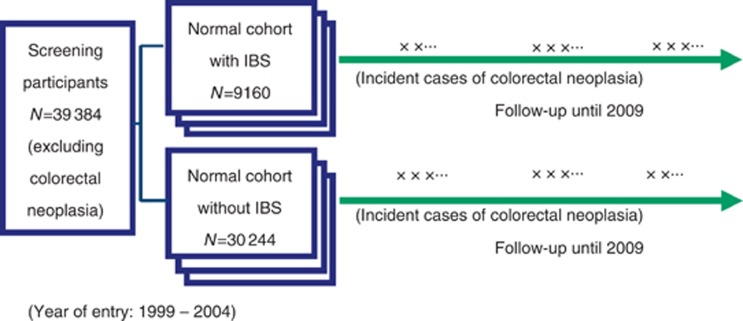
The prevalence of IBS is defined as period prevalence as IBS is rarely an acute disease and point prevalence may not be adequate. A prospective cohort study with a staggered entry design was adopted to ensure the temporal relationship between IBS and incident cases of colorectal neoplasia in our present study, and 1008 cases of colorectal adenomas and invasive carcinomas of the colon and rectum were excluded (Figure 1). To establish the temporal sequence that IBS occurs before CRC neoplasia, for those subjects with colorectal neoplasia, if the date of IBS diagnosis was later the confirmatory date of colorectal neoplasia, they were excluded from incident analysis.
Figure 1.
The cohort study design for assessing the relationship between IBS and colorectal neoplasia.
Data collection
The colorectal neoplasia cases were obtained from colonoscopy after repeated FIT screening with follow-up until the end of 2009. The symptomatic CRCs were also ascertained based on linkage with national cancer registry data until the end of 2009. Among these participants, information on IBS cases was obtained from the Bureau of National Health Insurance claim database, which covered the medical services of ∼95% of the entire population from 1 January 1999 to 31 December 2004 (Wen et al, 2008). During this period, participants with at least one outpatient visit with ICD-9-codes 5640 and 5641 were identified as having IBS, and the record of the first visit, with the date of the visit, was collected for each individual. To enhance the accuracy of identifying patients with IBS, we further validated prescription data from pharmaceutical claims for each prescription of these patients that contained at least one of the drugs listed below: laxative, antidiarrhoea and antispasmotic/anticholinergic drugs.
In the present study, anthropometric measurements (including blood pressure, height, weight and waist circumference), information on demographic characteristics (including age, gender and education) and lifestyle, family history and a series of biochemical variables were also collected and used at the first entry of screening. When classifying the years of education, we classified education into three levels. Six years or less of education was classified as low education level. Between 6 and 12 years of education was classified as medium education level. Twelve years of education or above was classified as high education level. Current or former smokers were placed in the smoking group. Similarly, current or former drinkers were placed in the alcoholic-drinking group. Exercise frequency was categorised into three groups (never, low and high). Exercise time less than 100 min per week was defined as the low-frequency group, whereas exercise time over 100 min per week was defined as the high-frequency group.
Patients with blood pressure ⩾140 mm Hg/90 mm Hg or those with a history of hypertension were placed in the hypertension group. Patients with fasting plasma glucose ⩾126 mg dl−1 or those with a history of diabetes mellitus were placed in the diabetes mellitus group. Metabolic syndrome was defined according to NCEP and was modified using the ATP III criteria with the adjustment of waist circumference for Asians. Three or more of the following five criteria must have been met to be placed in the metabolic syndrome group: (1) obesity – men ⩾90 cm and women ⩾80 cm; (2) hypertriglyceridaemia – ⩾150 mg dl−1; (3) low HDL – cholesterol concentration <40 mg dl−1 for men and <50 mg dl−1 for women; (4) high blood pressure – ⩾130 mmHg/85 mm Hg or hypertension; and (5) high fasting plasma glucose – ⩾100 mg dl−1 or diabetes mellitus.
Statistical analysis
Continuous variables such as age and follow-up time are expressed as the mean with s.d., and categorical variables are presented as the number and proportion. Student's t-test was used to compare continuous variables. Categorical variables, such as gender and education levels, were analysed using the χ2-test. All P-values were two-tailed, and a P-value less than 0.05 was considered statistically significant. The Cox proportional hazards regression model was applied to assess the relationship between IBS and the development of adenoma or CRC with adjustments for possible confounding factors. The proportional hazard assumption was tested for all the possible associations between IBS and the developing adenoma or CRC. All the results tested with proportional hazard assumption were not statistical significant (P=0.8250 for adenoma, P=0.2182 for CRC and P=0.4670 for colorectal neoplasia).
Results
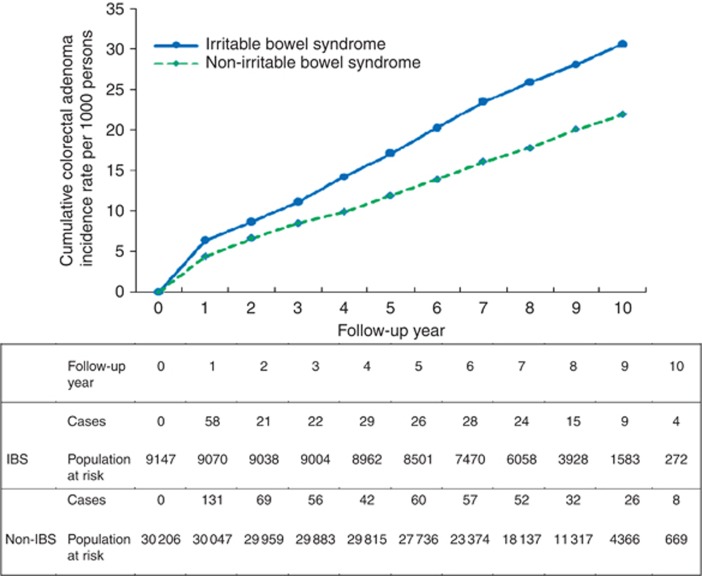
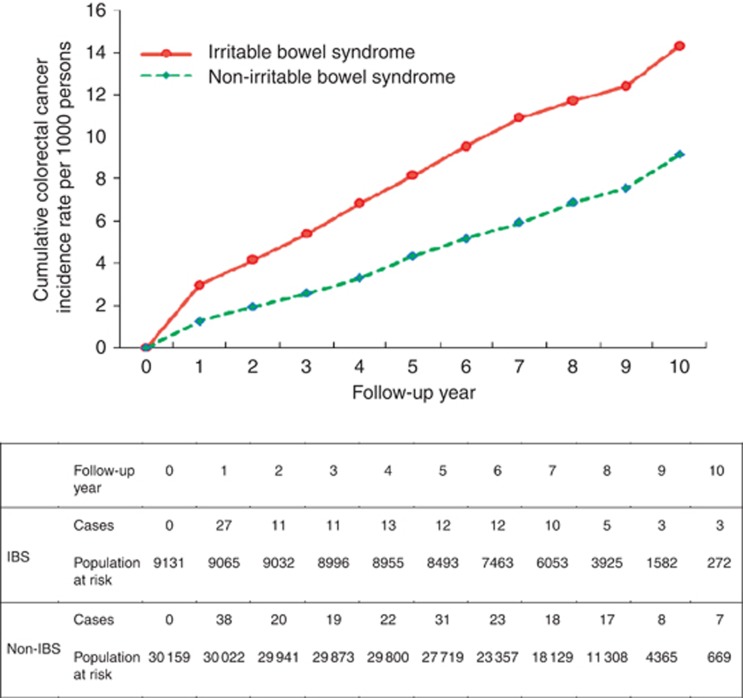
Of the 39 384 subjects, 9160 had IBS. The prevalence of IBS was 23.3% (95% CI: 22.8–23.7%). Subjects with IBS were older than those without (60.9±12.2 vs 55.9±11.1). The difference was statistically significant. By dividing age into four groups, those with IBS in the eldest group almost the triple of that with IBS in the youngest group. Age-specific prevalence rates of IBS increased with age, ranging from 14.7% for ages 40–49 years to 43.7% for ages over 70 years. Those with diabetes had higher chance of having IBS than those without (29.8% vs 22.6%). The similar finding was observed for higher odds of having IBS in subjects with hypertension compared with those without (24.6% vs 22.5%). It is very interesting to note that those without family history, lacking the habit of smoking and alcohol drinking, and frequent exercise had higher proportion of IBS than those with the opposite group. The prevalence was 32.0% for those suffering from colorectal neoplasia and 34.5% for those suffering from CRC (Table 1). The estimated results on the associations between each factor and the risk for adenoma, CRC and colorectal neoplasia are given in Table 2. As the sample size is sufficiently large, all the variables except BMI and family history of CRC in association with IBS would be statistically significant, even though the difference was small. These relationships were not of great interest because we are interested in how the relationship between IBS and colorectal neoplasia was affected by these variables (see below). As far as demographic features are concerned, an increase in 1 year of age led to a 5% (OR=1.05, 95%% CI: 1.04–1.06) elevated risk for CRC. Male patients were at greater risk than female patients by 53% (OR=1.53, 95%% CI: 1.22–1.91). Similar findings were also observed for the risks of adenoma and colorectal neoplasia with age and gender. The higher education levels were at lower risk than the low education levels. Lifestyle including smoking, alcohol drinking and exercise were not associated with the risk for CRC (OR=1.15, 95% CI: 0.81–1.61); however, the elevated risks for adenoma and colorectal neoplasia were noted in those who has the habbit of smoking or drinking. Obesity (BMI over 30) was positively associated with the risk of developing colorectal neoplasia. Table 2 also shows that, among the study subjects, metabolic syndrome, diabetes and hypertension were positively associated with the risk of developing CRC and colorectal neoplasia. Table 2 shows that the presence of IBS was positively associated with the risk for developing adenoma and/or invasive carcinoma of the colon. Figure 2 shows the comparison of two cumulative incidences of colorectal adenoma according to the presence of IBS. Subjects with IBS had higher cumulative incidence of colorectal adenoma at 10 years of follow-up (30.6‰ vs 21.9‰). Figure 3 shows the higher incidence of CRC in subjects with IBS compared with those without IBS after a 10-year follow-up (14.3‰ vs 9.2‰). Table 2 shows the adjusted hazard ratios (HRs) of IBS for the risk of adenoma, CRC and colorectal neoplasia. Subjects with IBS had a 21% increased risk (95% CI: 2–42%) of developing adenoma after adjusting for specifying the factors instead of age, gender, BMI, CRC family history, alcohol drinking, education and hypertension. A similar finding, albeit not statistically significant (HR=1.20; 95% CI: 0.94–1.53), was noted for the risk of CRC after the adjustment for other confounding factors, such as family history of CRC, diabetes and BMI.
Table 1. Selected characteristics of the Keelung population w/o irritable bowel syndrome.
| Characteristics | Total (N=39 384) | Irritable bowel syndrome (N=9160) | Non-irritable bowel syndrome (N=30 224) | P-value |
|---|---|---|---|---|
| Mean age±s.d. | 55.9±11.6 | 60.9±12.2 | 54.4±11.1 | <0.0001 |
|
Age at entry | ||||
| 40–49 | 14 624 | 2155 (14.7%) | 12 469 (85.3%) | <0.0001 |
| 50–59 | 9945 | 2026 (20.4%) | 7919 (79.6%) | |
| 60–69 | 8746 | 2328 (26.6%) | 6418 (73.4%) | |
| 70+ | 6069 | 2651 (43.7%) | 3418 (56.3%) | |
|
Gender | ||||
| Female | 23 847 | 5648 (23.7%) | 18 199 (76.3%) | 0.013 |
| Male | 15 537 | 3512 (22.6%) | 12 025 (77.4%) | |
|
Education level | ||||
| Low | 19 352 | 5504 (28.4%) | 13 848 (71.6%) | <0.0001 |
| Medium | 15 208 | 2855 (18.8%) | 12 353 (81.2%) | |
| High | 4571 | 749 (16.4%) | 3811 (83.6%) | |
| Body mass index (BMI)±s.d. | 24.9±3.7 | 24.9±3.7 | 24.9±3.6 | 0.8031 |
|
Diabetes | ||||
| Yes | 3153 | 939 (29.8%) | 2214 (70.2%) | <0.0001 |
| No | 35 988 | 8150 (22.6%) | 27 848 (77.4%) | |
|
Hypertension | ||||
| Yes | 13 705 | 3371 (24.6%) | 10 334 (75.4%) | <0.0001 |
| No | 25 487 | 5742 (22.5%) | 19 745 (77.5%) | |
|
CRC family history | ||||
| Yes | 877 | 181 (20.6%) | 696 (79.4%) | 0.0633 |
| No | 38 507 | 8979 (23.3%) | 29 528 (76.7%) | |
|
Smoking | ||||
| Current or former | 10 118 | 2229 (22%) | 7889 (78%) | 0.0003 |
| No | 28 816 | 6863 (23.8%) | 21 953 (76.2%) | |
|
Drinking | ||||
| Current or former | 8912 | 1784 (20%) | 7100 (80%) | <0.0001 |
| No | 29 833 | 7281 (24.4%) | 22 466 (75.6%) | |
|
Exercise frequency | ||||
| Never | 11 102 | 2458 (22.1%) | 8644 (77.9%) | <0.0001 |
| Low | 17 297 | 3820 (22.1%) | 13 477 (77.9%) | |
| High | 10 207 | 2752 (27%) | 7455 (73%) | |
| Colorectal Neoplasia | 1073 | 343 | 730 | |
| CRC | 310 | 107 | 203 | |
| Follow-up time | 7.78 | 7.92 | 7.73 | |
Table 2. Associations between colorectal adenoma and colorectal cancer or colorectal neoplasia (both combined) and IBS with adjustment for relevant risk factors using proportion hazards regression models.
|
Adenoma |
CRC |
Colorectal neoplasia |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | HR | 95% CI | aHR | 95% CI | HR | 95% CI | aHR | 95% CI | HR | 95% CI | aHR | 95% CI |
| IBS (Y/N) | 1.43 | (1.23–1.67)*** | 1.21 | (1.02–1.42)* | 1.70 | (1.35–2.15)*** | 1.20 | (0.94–1.53) | 1.52 | (1.34–1.73)*** | 1.22 | (1.07–1.40)** |
| Age | 1.03 | (1.02–1.04)*** | 1.02 | (1.01–1.02)*** | 1.05 | (1.04–1.06)*** | 1.05 | (1.04–1.06)*** | 1.04 | (1.03–1.04)*** | 1.03 | (1.02–1.03)*** |
| Gender (M/F) | 2.34 | (2.02–2.71)*** | 2.14 | (1.80–2.55)*** | 1.53 | (1.22–1.91)** | 1.31 | (1.04–1.64)* | 2.07 | (1.84–2.34)*** | 1.87 | (1.62–2.15)*** |
| BMI (⩾30/<30) | 1.31 | (1.04–1.66)* | 1.19 | (0.93–1.54) | 1.38 | (0.96–1.98) | 1.27 | (0.88–1.84) | 1.33 | (1.09–1.61)** | 1.33 | (1.09–1.62)** |
| CRC family history (Y/N) | 1.27 | (0.83–1.97) | 1.59 | (1.03–2.46)* | 1.05 | (0.50–2.22) | 1.54 | (0.72–3.27) | 1.13 | (0.76–1.66) | 1.47 | (0.99–2.17) |
| Smoking (Y/N) | 1.93 | (1.67–2.23)*** | — | — | 1.06 | (0.82–1.37) | — | — | 1.65 | (1.45–1.87)*** | — | — |
| Drinking (Y/N) | 1.83 | (1.57–2.12)*** | 1.36 | (1.15–1.61)** | 1.05 | (0.81–1.37) | — | — | 1.57 | (1.38–1.79)*** | 1.25 | (1.08–1.44)** |
|
Exercise frequency | ||||||||||||
| Medium/few | 1.07 | (0.90–1.28) | — | — | 0.86 | (0.65–1.13) | — | — | 1.00 | (0.86–1.16) | 0.85 | (0.73–0.98)* |
| High/few | 1.20 | (0.99–1.46) | — | — | 1.25 | (0.93–1.68) | — | — | 1.22 | (1.04–1.44)* | 0.61 | (0.48–0.79)*** |
|
Education level | ||||||||||||
| Medium/low | 0.75 | (0.64–0.88)** | 0.79 | (0.67–0.94) ** | 0.61 | (0.48–0.79)** | — | — | 0.71 | (0.62–0.81)*** | — | — |
| High/low | 0.59 | (0.45–0.78)** | 0.58 | (0.43–0.78)** | 0.43 | (0.27–0.69)** | — | — | 0.54 | (0.43–0.68)*** | — | — |
| Mets (Y/N) | 1.38 | (1.18–1.60)*** | — | — | 1.33 | (1.04–1.69)* | — | — | 1.34 | (1.18–1.53)*** | — | — |
| Diabetes (Y/N) | 1.01 | (0.78–1.31) | — | — | 1.87 | (1.36–2.59)** | 1.43 | (1.03–1.99)* | 1.24 | (1.02–1.52)* | — | — |
| Hypertension (Y/N) | 1.34 | (1.16–1.55)*** | 1.19 | (1.02–1.40)* | 1.34 | (1.07–1.68)* | — | — | 1.33 | (1.18–1.50)** | — | — |
***P<0.0001, **P<0.01, *P<0.05.
Figure 2.
Cumulative colorectal adenoma incidence rates by IBS status.
Figure 3.
Cumulative colorectal cancer incidence rates by IBS status.
Discussion
The estimated prevalence of IBS was 23.3% in our study, which is similar to the estimated prevalence from a previous questionnaire survey study in Taiwan (Lu et al, 2003). Moreover, there was no significant difference of IBS prevalence among different genders noted by the study by Lu et al (2003) and our present study. Our study demonstrated that the prevalence rates of IBS increased with age from 14.7% for ages 40–49 years to 43.7% for ages over 70 years. In contrast, Lu et al (2003) found that the IBS prevalence decreased significantly with advancing age. In their study, the IBS prevalence was 34.1% for ages 30–39 years, 25% for ages 40–49 years and 18.6% for ages over 70 years. The participants of the previous study, who were enroled from voluntarily health check-up, might represent a more healthy population with high motivation for health behaviour. Therefore, the IBS prevalence of the general population that is inferred from the health check-up population might be underestimated.
Our study population was a community-based screening cohort. In contrast to the diagnosis of IBS using the Rome II questionnaire survey, we used national health insurance data to make the IBS diagnosis. As a portion of IBS patients, especially those with mild symptoms, do not seek medical help, underestimation of the prevalence of IBS is highly suspected, especially among younger people. This could explain why our study showed a lower IBS prevalence among people aged 40–49 years than what was found in the previous study.
In the past, IBS was viewed as a purely functional disorder. However, in recent years, numerous data have suggested that immune activation occurs in IBS patients (Matricon et al, 2012) and is more frequently observed in IBS patients than in healthy controls. Several studies have reported an increase in the number of mast cells (Barbara et al, 2004), lymphocytes (Kim et al, 2010 and Cremon et al, 2009), and intestinal permeabilisation (Piche et al, 2009) of the colon and rectum in IBS patients. The hypothesis of patients with a long-term course of IBS having the potential risk of developing colorectal disease is considered.
In the present study, we found that IBS patients were associated with a significantly higher risk of colorectal adenoma and colon neoplasia compared with those without IBS. Three possible reasons could explain why IBS patients have a higher risk of developing colon neoplasia. First, IBS and colon neoplasia have similar clinical symptoms that result in the higher detection rate of colon neoplasia. Second, IBS and colon neoplasia share common risk factors. Third, IBS has a causal role in the pathogenesis of colon neoplasia.
Two previous studies from the United Kingdom and Denmark revealed a lack of relationship between IBS and CRC after excluding CRCs within the first year of follow-up (Garcia Rodriguez et al, 2000; Nørgaard et al, 2011). The overlapping symptoms among IBS and colon rectal cancer can result in diagnostic confusion and are considered to be the major cause of the increased risk of CRC in the first year after IBS diagnosis. However, our study demonstrated that IBS patients have a higher risk of developing colon neoplasia, especially colon adenoma. IBS is a clinical syndrome characterised by chronic abdominal pain, discomfort and altered bowel habits. The majority of colon adenomas are usually asymptomatic but can present as abdominal pain, change of bowel habits or occult bleeding if the tumour size increases. The clinical symptoms of IBS and colon neoplasia/adenoma are not highly correlated. Therefore, the significant relationship between IBS and the incidence of colon neoplasia is less likely caused by the similar symptoms of IBS and colon neoplasia.
IBS is a clinical syndrome diagnosed by clinical presentation after removal of the structural abnormality (Drossman et al, 2002). To rule out the possibility of structural or pathological lesions of the large bowel, colonoscopy is performed in the clinical setting, and more frequent colonoscopy examinations will increase the detection rate of colon neoplasia. However, different from the previous UK study that used the general practitioners' database (Nørgaard et al, 2011), our study cohort was enroled from a community-screening project. Colonoscopy was performed in a screening setting and was not based on clinical symptoms. It is possible that bias in increasing surveillance for colorectal lesion may affect the ascertainment of incident colorectal neoplasia in subjects with IBS compared with those without. However, as the percentage of CRCs diagnosed by colonoscopy in our screening setting was similar between the IBS and non-IBS groups (82% for the IBS group and 81% for the non-IBS group), an increased frequency of colonoscopic examination (in a nonscreening setting) because of the demands of the clinical symptoms of IBS is less likely.
The risk factors of IBS are still unclear; however, several have been identified by epidemiologic studies over the past few decades. However, it is difficult to separate factors promoting health-care seeking from true risk factors for the disease (Rey and Talley, 2009). Genetic background and early life events are proposed to be potential risk factors for IBS, and diet composition may be related to IBS. A recent Taiwanese study showed that smoking, obesity and metabolic syndrome were associated with increased risk of colon neoplasia (Hu et al, 2011). Although IBS and colon neoplasia may share common risk factors, our study found that IBS still had an independent and significant effect of increasing the risk of colon neoplasia after adjusting for smoking, BMI, DM and family history of CRC. However, the possibility of potentially unmeasured confounding factors that results in this association of IBS and colon neoplasia still could not be excluded. Further, more studies to discover the association of IBS and the risk of colon neoplasia and to explore the possibility of the causal role of IBS in the development of colon neoplasia are needed in the future.
Conclusion
Our study finds a significant association between IBS and colon neoplasia, and it provides an important suggestion to clinical physicians: they should pay more attention to IBS and the possibility of colon neoplasia or CRC in patients.
Acknowledgments
We thank the Health Bureau of Keelung City for their contribution and support. This study (KCIS No.36) is one of the studies of Keelung Community-based Integrated Screening (KCIS) programme. We also thank for the financial support of this study from Shin-Kong Wu Ho-Su Memorial Hospital and Taipei Medical University (SKH-TMU-101-06).
Footnotes
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
References
- Barbara G, Stanghellini V, De Giorgio R, Cremon C, Cottrell GS, Santini D, Pasquinelli G, Morselli-Labate AM, Grady EF, Bunnett NW, Collins SM, Corinaldesi R. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology. 2004;126:693–702. doi: 10.1053/j.gastro.2003.11.055. [DOI] [PubMed] [Google Scholar]
- Chang FY, Lu CL, Chen. TS. The current prevalence of irritable bowel syndrome in Asia. J Neurogastroenterol Motil. 2010;16:389–400. doi: 10.5056/jnm.2010.16.4.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen LS, Yen AM, Chiu SY, Liao CS, Chen HH. Baseline faecal occult blood concentration as a predictor of incident colorectal neoplasia: longitudinal follow-up of a Taiwanese population-based colorectal cancer screening cohort. Lancet Oncol. 2011;12:551–558. doi: 10.1016/S1470-2045(11)70101-2. [DOI] [PubMed] [Google Scholar]
- Chen TH, Chiu YH, Luh DL, Yen MF, Wu HM, Chen LS, Tung TH, Huang CC, Chan CC, Shiu MN, Yeh YP, Liou HH, Liao CS, Lai HC, Chiang CP, Peng HL, Tseng CD, Yen MS, Hsu WC, Chen CH, Taiwan Community-Based Integrated Screening Group Community-based multiple screening model: design, implementation, and analysis of 42,387 participants. Cancer. 2004;100:1734–1743. doi: 10.1002/cncr.20171. [DOI] [PubMed] [Google Scholar]
- Cremon C, Gargano L, Morselli-Labate AM, Santini D, Cogliandro RF, De Giorgio R, Stanghellini V, Corinaldesi R, Barbara G. Mucosal immune activation in irritable bowel syndrome: gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392–400. doi: 10.1038/ajg.2008.94. [DOI] [PubMed] [Google Scholar]
- Drossman DA, Camilleri M, Mayer EA, Whitehead WE. AGA technical review on irritable bowel syndrome. Gastroenterology. 2002;123:2108–2131. doi: 10.1053/gast.2002.37095. [DOI] [PubMed] [Google Scholar]
- Garcia Rodriguez LA, Ruigomez A, Wallander MA, Johansson S, Olbe L. Detection of colorectal tumor and inflammatory bowel disease during follow-up of patients with initial diagnosis of irritable bowel syndrome. Scand J Gastroenterol. 2000;35:306–311. doi: 10.1080/003655200750024191. [DOI] [PubMed] [Google Scholar]
- Hu NC, Chen JD, Lin YM, Chang JY, Chen YH. Stepwise relationship between components of metabolic syndrome and risk of colorectal adenoma in a Taiwanese population receiving screening colonoscopy. J Formos Med Assoc. 2011;110:100–108. doi: 10.1016/S0929-6646(11)60016-8. [DOI] [PubMed] [Google Scholar]
- Kim HS, Lim JH, Park H, Lee SI. Increased immunoendocrine cells in intestinal mucosa of postinfectious irritable bowel syndrome patients 3 years after acute Shigella infection–an observation in a small case control study. Yonsei Med J. 2010;51:45–51. doi: 10.3349/ymj.2010.51.1.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu CL, Chen CY, Lang HC, Luo JC, Wang SS, Chang FY, Lee SD. Current patterns of irritable bowel syndrome in Taiwan: the Rome II questionnaire on a Chinese population. Aliment Pharmacol Ther. 2003;18:1159–1169. doi: 10.1046/j.1365-2036.2003.01711.x. [DOI] [PubMed] [Google Scholar]
- Matricon J, Meleine M, Gelot A, Piche T, Dapoigny M, Muller E, Ardid D. Review article: associations between immune activation, intestinal permeability and the irritable bowel syndrome. Aliment Pharmacol Ther. 2012;36:1009–1031. doi: 10.1111/apt.12080. [DOI] [PubMed] [Google Scholar]
- Nørgaard M, Farkas DK, Pedersen L, Erichsen R, de la Cour ZD, Gregersen H, Sørensen HT. Irritable bowel syndrome and risk of colorectal cancer: a Danish nationwide cohort study. Br J Cancer. 2011;104:1202–1206. doi: 10.1038/bjc.2011.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberndorff-Klein Woolthuis AH, Brummer RJ, de Wit NJ, Muris JW, Stockbrügger RW. Irritable bowel syndrome in general practice: an overview. Scand J Gastroenterol. 2004;241:17–22. doi: 10.1080/00855920410010942. [DOI] [PubMed] [Google Scholar]
- Piche T, Barbara G, Aubert P, Bruley des Varannes S, Dainese R, Nano JL, Cremon C, Stanghellini V, De Giorgio R, Galmiche JP, Neunlist M. Impaired intestinal barrier integrity in the colon of patients with irritable bowel syndrome: involvement of soluble mediators. Gut. 2009;58:196–201. doi: 10.1136/gut.2007.140806. [DOI] [PubMed] [Google Scholar]
- Rey E, Talley NJ. Irritable bowel syndrome: novel views on the epidemiology and potential risk factors. Dig Liver Dis. 2009;41:772–780. doi: 10.1016/j.dld.2009.07.005. [DOI] [PubMed] [Google Scholar]
- Wen CP, Tsai SP, Chung WSI. A 10-year experience with universal health insurance in Taiwan: measuring changes in health and health disparity. Ann Intern Med. 2008;148:258–267. doi: 10.7326/0003-4819-148-4-200802190-00004. [DOI] [PubMed] [Google Scholar]