Abstract
Background:
Tumour-infiltrating lymphocyte (TIL) therapy is showing great promise in the treatment of patients with advanced malignant melanoma. However, the translation of TIL therapy to non-melanoma tumours such as renal cell carcinoma has been less successful with a major constraint being the inability to reproducibly generate TILs from primary and metastatic tumour tissue.
Methods:
Primary and metastatic renal cell carcinoma biopsies were subjected to differential tumour disaggregation methods and procedures that stimulate the specific expansion of TILs tested to determine which reliably generated TIL maintained antitumour specificity.
Results:
Enzymatic or combined enzymatic/mechanical disaggregation resulted in equivalent numbers of TILs being liberated from renal cell carcinoma biopsies. Following mitogenic activation of the isolated TILs with anti-CD3/anti-CD28-coated paramagnetic beads, successful TIL expansion was achieved in 90% of initiated cultures. The frequency of T-cell recognition of autologous tumours was enhanced when tumours were disaggregated using the GentleMACS enzymatic/mechanical system.
Conclusion:
TILs can be consistently produced from renal cell carcinoma biopsies maintaining autologous tumour recognition after expansion in vitro. While the method of disaggregation has little impact on the success of TIL growth, methods that preserve the cell surface architecture facilitate TIL recognition of an autologous tumour, which is important in terms of characterising the functionality of the expanded TIL population.
Keywords: renal cell carcinoma; T-cell; adoptive cell therapy; lymphocyte, culture; tumour; disaggregation; mitogen; TIL
Reports of durable and complete tumour regressions in malignant melanoma patients treated with tumour-infiltrating lymphocyte (TIL) therapy (Dudley et al, 2008; Besser et al, 2010; Rosenberg et al, 2011; Besser et al, 2013) have stimulated a strong interest in the development of this approach for other cancer indications. Both malignant melanoma and renal cell carcinoma (RCC) are sensitive to immunotherapies including cytokines such as interferon-α and interleukin-2, implying that RCC may also be suitable for cellular immune therapies. However, while tumour-specific melanoma TIL can be readily expanded from source tumour tissue by culture in IL-2 alone (Dudley et al, 2003), a consistently similar level of successful in vitro tumour-specific TIL expansion in RCC has not been observed (Yannelli et al, 1996; Bouet-Toussaint et al, 2000; Malone et al, 2001; Markel et al, 2009).
Clinical trials involving the adoptive transfer of RCC TILs have shown limited clinical efficacy. Most encouraging was a single-centre study involving 36 patients who received TIL and low-dose IL-2 who achieved an overall response rate (complete+partial response) of 34.6% (Figlin et al, 1997). Even more encouragingly, a further 23 patients who received CD8+-selected TIL with low-dose IL-2 achieved a 43.5% overall response rate (Figlin et al, 1997). However, a phase-III multi-centre study based upon these data investigating CD8+ TIL+low-dose IL-2 was terminated early after 80 patients were recruited due to a lack of efficacy as compared with placebo+low-dose IL-2 (Figlin et al, 1999). The key issues arising from this trial were the high TIL production failure rate (41%), which adversely impacted on the intention-to-treat response rate, and the excessive variability in TIL phenotype, which together highlight the need to develop robust cellular bioprocesses that ensure consistent cell production between multiple sites. Further small-scale clinical trials testing RCC TILs given with IL-2 support have also shown limited clinical responses (Goedegebuure et al, 1995; Thompson et al, 2003; Dillman et al, 2004), which together dampened enthusiasm for TIL therapy in RCC.
Technological advances that have supported the worldwide development of melanoma TIL therapy based upon the initial work reported at the Surgery Branch, NIH (Dudley et al, 2008; Tran et al, 2008; Dudley et al, 2010; Rosenberg et al, 2011) suggest it is timely to revisit the generation of RCC TIL for therapy. Indeed, the successful generation of TIL from 60% of metastatic RCC tumours using current melanoma-based culture technology has been recently reported (Markel et al, 2009). In this study, variable tumour-specific activity was reported questioning whether tumour-specific TIL can be generated from RCC tumour biopsy, or whether other tumour-based factors inhibit RCC TIL effector function, which will be likely to impact on the potential efficacy of the therapy. In this report, we have investigated the impact of tissue disaggregation and mitogenic activation on TIL expansion and maintenance of antitumour activity to develop a protocol that reproducibly generates large numbers of tumour-specific TILs from primary RCC biopsies suitable for clinical application.
Patients and Methods
Patients and samples
All tissue and blood samples were collected with informed patient consent from three hospitals in the South Manchester region (Christie Hospital, University Hospital of South Manchester and Salford Royal Hospital) under local Ethics committee approval (LREC 09/H1003/75) or through the Manchester Cancer Research Centre Biobank. Blood was collected directly into a 50-ml polypropylene tube containing 100 IU of heparin or into vacutainers (BD Biosciences, Oxford, UK). Tumour samples were collected into serum-free RPMI-1640 media and all samples transported directly to the laboratory for processing.
Cell lines
The renal cell line 2220R was kindly donated by Dr J Yang (Surgery Branch NIH, Bethesda, MD, USA) as previously described (Griffiths et al, 2005); the gastric carcinoma cell line MKN45K was previously described (Gilham et al, 2002); the mel624 melanoma cell line was a kind gift from Professor Alan Melcher (The University of Leeds, Leeds, UK); neuroblastoma cell line LA15S was a kind gift from Dr Guy Makin (University of Manchester, Manchester, UK) and SK-N-SH (ATCC HTB11) was purchased from ATCC (Manassas, VA, USA).
Isolation and expansion of RCC TILs
Tumour biopsies were subjected to either an overnight digest or to a commercial mechanical/enzymatic dissociation system (GentleMACS, Miltenyi Biotec, Bergish Gladbach, Germany). The overnight digest was based upon methodology previously established for the generation of melanoma TILs (Dudley et al, 2003, 2008). In brief, tumour biopsies were cut into small fragments ∼2–3 mm in length and put in an enzyme digest mix consisting of 3000 U ml−1 DNAse, 10 mg ml−1 collagenase and 10 mg ml−1 hyaluronidase (all from Sigma-Aldrich, Gillingham, UK) and incubated overnight at room temperature under continuous rotation. GentleMACS dissociation was performed according to the manufacturer's protocol. Briefly, the tumour was cut into small fragments about 2–3 mm in length and put in a C-tube (Miltenyi Biotech) with RPMI 1640 (Lonza, Slough, UK) and solutions 1, 2 and 3 (all from Miltenyi Biotec) according to the manufacturer's recommendation; the digest mix containing the tumour was then subjected to three 36-second mechanical disaggregation steps (programs h_tumor_01.01, 02.01 and 03.01) in the GentleMACS dissociator interspersed by two 30-min incubations at 37 °C performed after the first and the second disaggregation steps, respectively.
After disaggregation, TILs from the overnight digest and the GentleMACS dissociation were passed through 100-μm strainers and then cultured in complete media (RPMI 1640 (Lonza) supplemented with 10% foetal calf serum (FCS), 1% glutamine, 100 IU ml−1 penicillin, 100 μg ml−1 streptomycin (all from Life Technologies, Paisley, UK), 25 mM HEPES and 50 μM β-mercaptoethanol (both from Sigma-Aldrich) in 24-well plates at a concentration of 0.5 × 106 ml−1 with 6000 or 3000 IU ml−1 recombinant IL-2 (Novartis, Camberley, UK). Every second day, half of the media was removed and replaced with fresh media supplemented with fresh IL-2. After day 7, cultures were expanded by the addition of media supplemented with IL-2 to maintain a cell density of 106 cells ml−1.
Mitogenic stimulation of TIL post disaggregation
Isolated RCC TILs were mitogenically stimulated in wells of a 24-well plate previously coated with 30ng ml−1 anti-hCD3ɛ (Orthoclone OKT3, Janssen-Cilag, High Wycombe, UK) and 30 ng ml−1 anti-hCD28 (R&D systems, Abingdon, UK) at a cell concentration of 106 cells ml−1. Alternatively, RCC TILs were mitogenically activated using two different paramagnetic bead systems. MACSi bead particles (Miltenyi Biotec) consisting of anti-human CD2, anti-human CD3 and anti-human CD28 mabs bound to paramagnetic beads were used at a bead:T-cell ratio of 1 : 1. Dynabeads Human T Activator CD3/CD28 (Life Technologies) were also used at a bead:T-cell ratio of 1 : 1. Dynabeads were removed 7 days after culture initiation.
IFNγ secretion assay
The co-culture was established with a 1 : 1 ratio of expanded TILs and autologous tumour cells (105 TILs and 105 autologous tumour cells per well) derived from the initial tumour digest that was cryopreserved in 10% DMSO (Sigma-Aldrich) and 90% FCS (Life Technologies). Where class I or class II blocking experiments were performed, anti-HLA-ABC (W6/32, Biolegend, London, UK) or the anti-HLA DR DP DQ (Tu39, BD Biosciences) were added to the tumour cells at 50 μg ml−1, incubated for 45 min at 4 °C and the excess washed off before setting up the co-culture. The limited quantities of tumour tissue prevented the use of suitable isotype controls. To control for this, antibody clones W6/32 and Tu39 were sourced from companies for which extensive testing had been previously performed, showing that the isotype control for these antibodies had no significant effect upon the T-cell cultures (McLoughlin et al, 2008; Takahara et al, 2008; Takeuchi et al, 2010; Danilova et al, 2015). Where the tumour cell lines 2220R, MKN45K, Mel624, LA15S and SK-N-SH were utilised as targets, they were trypsined, washed and resuspended in T-cell media before being added to 105 rapidly expanded TILs at a 1 : 1 ratio. The co-culture was incubated in a humidified incubator for 24 h at 37 °C before the supernatant was harvested and analysed for interferon gamma (IFNγ) production in an ELISA assay. The ELISA assays were carried out using the ELI-pair kit (GenProbe, San Diego, CA, USA) according to the manufacturer's instructions.
Flow cytometry
Analyses of TILs from RCC samples were performed on cells either directly following isolation or expanded for 15 days in high-dose IL-2 and complete media. The following anti-human antibodies were used: PE anti-CD4, clone RPA-T4, FITC anti-CD8, clone HIT8a, PE anti-CD27, clone M-T271, PE anti-CD28, clone CD28.2, PE anti-CD45RA, clone HI100 (all from BD Biosciences), PE-Cy7 anti-CD62L, clone DREG-56 (eBioscience, Hatfield, UK) and APC anti-CCR7, clone REA108 (Miltenyi Biotec) for the phenotype panel; PE anti-CD107a, clone H4A3 (BD Biosciences) was used in the CD107a assay; PerCP Cy5.5 anti-CD4, clone SK3 and V500 anti-CD8, clone RPA-T8 (both from BD Biosciences), PE-Cy7 anti-HLA A,B,C, clone W6/32 and PE anti-EpCAM, clone 9C4 (both from Biolegend), FITC anti-HLA DR DP DQ, clone CR3/43 (Dako, Ely, UK), PE-AF647 anti-CD3, clone MEM-57 and APC anti-CD45, clone MEM-28 (both from Immunotools, Friesoythe, Germany), LIVE/DEAD fixable near-IR dead cell stain, 633 nm (Life Technologies), FcR block (Miltenyi Biotec) for the staining directly after isolation. Cells were stained in PBS containing 1% FCS for 30 min at 4 °C in the dark. Acquisition was performed on a FACSCalibur flow cytometer (BD Biosciences) for the phenotype panel and the CD107a assay; LSR II flow cytometer (BD Biosciences) was used to acquire cells stained directly after TIL isolation. FlowJo v. 7.6.2 software (Tree Star Inc., Ashland, OR, USA) was used to analyse the data.
Rapid expansion protocol (REP)
TIL cultures were incubated with feeder cells consisting of irradiated (50 Gy) allogeneic PBMCs from three different healthy donors at a 200 : 1 feeder cell:TILs ratio. Anti-CD3 antibody (OKT3, OrthoBiotech, High Wycombe, UK) was added at a concentration of 30 ng ml−1 and cells were kept in culture at a concentration of 1 × 106 ml−1 for further 14 days with IL-2 (1000 IU ml−1) that was refreshed every other day.
Immunohistochemistry
Tumour samples were snap-frozen in liquid nitrogen after collection and sections were stained with anti-human CD68 (DAKO (Cat no: IR613) ready to use), anti-human HLA A-B-C (clone W6/32, Biolegend; 1 : 400) and the corresponding isotype controls. IHC was carried out using the Leica Bond automated platform using the Leica Bond Refine detection kit (Leica Biosystems, Milton Keynes, UK) as per the manufacturer's instructions. Prior to staining, sections were fixed in acetone/ethanol (1 : 1) for 15 min.
Results
Mitogenic activation by antibody-coated paramagnetic beads increases the frequency of successful TIL generation from primary RCC tumour biopsies
With the intention of working up towards a clinical investigation of the potency of RCC TIL, we undertook a preliminary analysis to determine the frequency of success of TIL expansion from 17 RCC patient tumour biopsies (R1–R17, Table 1). Using a protocol based upon high-dose IL-2 (6000 IU ml−1) as employed to routinely expand TILs from melanoma (Yannelli et al, 1996; Dudley et al, 2003), TIL were successfully cultured and expanded in only 2 out of 17 samples (11.7% Table 1). The majority of the patient biopsies (15 out of 17) were from primary tumour sites and these results reflected the experiences reported by others when attempting to expand TIL from primary RCC tumour biopsies (Yannelli et al, 1996). In order to assess whether mitogenic activation of T cells within the tumour digest could expand TIL, parallel cultures were initiated where a disaggregated tumour was cultured on plates pre-coated with anti-CD3ɛ and CD-28 monoclonal antibodies or cultured with paramagnetic beads loaded with anti-CD3ɛ/CD28/CD2 antibodies (Miltenyi MACSi bead particles). Both culture conditions resulted in an increased frequency of TIL expansion; although the paramagnetic bead approach was superior in terms of frequency (5 out of 13 (38.4%) vs 5 out of 9 (55.6%)), the variation in final fold expansion was much greater (25.6±9.1-fold expansion for antibody vs 101±141 for MACSi beads; Table 1).
Table 1. Patients characteristics and preliminary TIL expansion data obtained from 17 renal cell carcinoma patients between January 2006 and September 2007.
|
Fold expansion |
||||||
|---|---|---|---|---|---|---|
| Patient | Sex | Age at the time of surgery (years) | Tumour | IL-2 alone | Immobilised αCD3/αCD28 | Paramagnetic beads |
| R1 | M | 70 | Primary | No growth | ND | ND |
| R2 | F | 53 | Primary | 2 | ND | ND |
| R3 | F | 67 | Primary | No growth | 30 | ND |
| R4 | F | 73 | Primary | No growth | 10 | ND |
| R5 | M | 47 | Primary | No growth | 32 | ND |
| R6 | M | 74 | Primary | No growth | 25 | ND |
| R7 | M | 50 | Primary | No growth | No growth | ND |
| R8 | M | 49 | Primary | No growth | No growth | 40 |
| R9 | M | 75 | Primary | No growth | No growth | 3 |
| R10 | M | 64 | Primary | 32 | Infected | 106 |
| R11 | F | 46 | Primary | No growth | ND | 344 |
| R12 | F | 67 | Primary | No growth | No growth | No growth |
| R13 | M | 64 | Primary | No growth | No growth | No growth |
| R14 | M | 47 | Renal bed | No growth | No growth | ND |
| R15 | F | 54 | Primary | No growth | 31 | 12 |
| R16 | M | 48 | Lung Met | No growth | No growth | No growth |
| R17 | M | 61 | Primary | No growth | No growth | No growth |
| 2/17 | 5/13 | 5/9 | ||||
Abbreviations: F=female; M=male; ND=normal donor.
‘Renal bed' indicates that the samples analysed were recurrences at the site of the previous radical nephrectomies.
TIL isolation from RCC biopsies: overnight enzymatic disaggregation compared with short-term mechanical/enzymatic disaggregation
On the basis of these initial observations, a further study was performed involving 19 patient samples collected between January 2012 and July 2013 (V1–V9 and V11–V20; patient characteristics detailed in Table 2). Eleven patients were male aged between 46 and 79 years (median age 63.5 years) and eight patients were female aged between 50 and 73 years (median age 68.5 years). The majority of the samples consisted of conventional clear cell RCC (15 out of 19); the remaining four were reported as being papillary RCC (two samples) and chromophobe RCC (two samples). Among the 19 samples, 17 consisted of primary RCC with no previous treatment before the surgery. Sample V8 was a metastatic peritoneal nodule that developed after nephrectomy for primary RCC and enrolment on the phase-III SORCE trial (NCT00492258) where sorafenib or placebo was given after surgery. Sample V20 was from a metastatic site on the adrenal gland that developed after previous radical nephrectomy and was treated with sunitinib 2 months before surgery.
Table 2. Tumour characteristics and patient demographics for 25 renal cell carcinoma biopsies collected between January 2012 and July 2013.
| Patient | Sex | Age at the time of surgery (years) | RCC histology | Stage | Previous treatments |
|---|---|---|---|---|---|
| V1 | M | 46 | Primary, conventional clear cell | pT3a Nx Mx | None |
| V2 | M | 69 | Primary, conventional clear cell | pT1a N0 Mx | None |
| V3 | M | 76 | Primary, conventional clear cell | T3b N2 | None |
| V4 | M | 55 | Primary, conventional clear cell | pT3 N0 Mx | None |
| V5 | F | 57 | Primary, conventional clear cell | pT1a N0 Mx | None |
| V6 | M | 55 | Primary, conventional clear cell | pT1a N0 Mx | None |
| V7 | F | 50 | Primary, conventional clear cell | T1b | —2 Cycles of IL-2 —Left nephrectomy |
| V8 | F | 68 | Metastatic, peritoneal nodule (conventional clear cell) | —Left nephrectomy —SORCE trial (sorafenib vs placebo) | |
| V9 | F | 69 | Primary, papillary | pT1a Nx Mx | None |
| V11 | M | 73 | Primary, papillary | pT1a Nx Mx | None |
| V12 | M | 61 | Primary, conventional clear cell | pT3a N0 Mx | None |
| V13 | M | 79 | Primary, conventional clear cell | pT3b Nx Mx | None |
| V14 | F | 71 | Primary, chromophobe | pT2b N0 Mx | None |
| V15 | F | 73 | Primary, conventional clear cell | pT1a Nx Mx | None |
| V16 | F | 51 | Primary, conventional clear cell | T1a Nx Mx | None |
| V17 | M | 67 | Primary, chromophobe | T2b N1 Mx | None |
| V18 | M | 52 | Primary, conventional clear cell | T3a N0 Mx | None |
| V19 | F | 73 | Primary, conventional clear cell | T3b N0 R0 | None |
| V20 | M | 64 | Metastatic, right adrenal gland (conventional clear cell) | —Left nephrectomy —Sunitinib | |
| V29 | M | 48 | Metastatic, right 4th rib (conventional clear cell) | —Left nephrectomy —2 Cycles of IL-2 | |
| V30 | M | 65 | Primary, conventional clear cell | T2a N0 Mx | None |
| V31 | M | 45 | Metastatic, brain (conventional clear cell) | —Left nephrectomy —Pazopanib —Axitinib | |
| V37 | F | 65 | Metastatic, lung (conventional clear cell) | —Left nephrectomy —2 Cycles of IL-2 | |
| V39 | M | 48 | Primary, chromophobe | T1b Nx Mx | None |
| V40 | M | 73 | Primary, papillary | T3a N0 Mx | None |
Abbreviations: F=female; IL-2=interleukin-2; M=male; RCC=renal cell carcinoma.
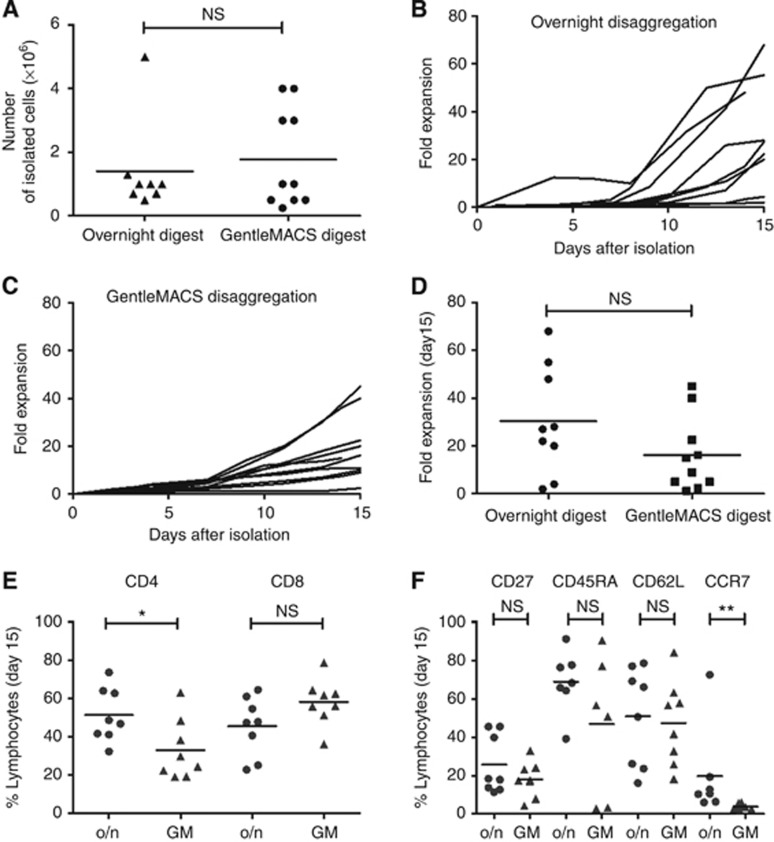
In order to determine whether the disaggregation method impacted on TIL isolation, samples V1–V7 and V9 were subjected to overnight enzyme disaggregation as used in samples R1–R17. At this point, the GentleMACS dissociator, which combines enzymatic disaggregation with mechanical disruption in a three-h process, became available and this was used to obtain single-cell suspensions from samples V11–V20. There was no statistical difference in the overall number of TILs liberated between the GentleMACS-dissociated samples (1.8±1.5 × 106 total cells; median 1.0 × 106; range 0.3–4.0 × 106) and overnight digestion (1.4±1.5 × 106 total cells; median 1.0 × 106; range 0.5–5 × 106; Figure 1A). One additional sample (V8) proved to be much larger than previous samples and liberated 35 × 106 TIL after enzymatic disaggregation (Supplementary Figure 1A). Overall, there appeared to be no significant difference in the number of TILs liberated by either disaggregation method.
Figure 1.
Isolation, expansion and phenotype of TILs from renal biopsies. (A) Number of isolated TILs after overnight (n=8) or GentleMACS (GM) (n=10) digestion from each biopsy sample. No statistically significant difference was observed between the digestion methods. NS, P>0.05, Mann–Whitney test. (B) Growth curves of TILs throughout the 15-day culture period in overnight-digested or (C) GM-processed samples. (D) Fold expansion of TILs after 15 days of culture after overnight or GM digests. (E, F) Phenotypic characterisation of TILs expanded from renal biopsies after 15 days in culture in overnight (o/n) digested and GM-processed samples. NS, P>0.05, *P<0.05, **P<0.01, Mann–Whitney test. Abbreviation: NS, not significant.
RCC TIL cultured with antibody-coated paramagnetic beads display an effector-like phenotype
Equivalent numbers of cells isolated from each sample were cultured using anti-CD3/anti-CD28-coated Dynabeads as the source of antibody-coated paramagnetic bead T-cell activation, since this bead system was also available in a clinical format and thus the results of this work could be rapidly translated to the clinical cell-production environment. Cultures were maintained for a total of 15 days supplemented with IL-2 (3000 IU ml−1 until day 7 after isolation and reduced to 1,000 IU ml−1 after) and with the Dynabeads removed on day 7 of culture. To be deemed positive, a culture had to produce a minimum of 1 × 107 cells within a 15-day culture period. Approximately 90% of the TIL cultures reached this threshold (8 out of 9 for overnight digest and 9 out of 10 for GentleMACS disaggregation; Figure 1B and C) with no statistically significant difference in the average fold expansion of TILs produced from the overnight disaggregation method (30.4±7.4; range 68–2; median 24.5) as compared with the GentleMACS (19.1±4.4; range 45–2.5; median 15.6; Figure 1D).
In terms of cellular phenotype, there was a small but significant reduction in the frequency of CD4+ T cells in the GentleMACS-disaggregated cultures (32.9±5.6%) when compared with overnight disaggregated cultures (51.4±5.0%), although the complimentary difference in CD8+ subset frequency did not reach significance (Figure 1E). There was also no significant difference in the relative frequency of CD27, CD45RA or CD62L expression between cultures, although there was a broad range in relative frequency for the expression of CD45RA and CD62L in particular (Figure 1F). There was found to be a significant reduction of the relative frequency of CCR7+ T cells within GentleMACS-disaggregated cultures (3.7±0.5%) compared with overnight disaggregated cultures (19.7±9.0%), although the overall frequency of CCR7+ cells remained relatively small within all cultures (Figure 1F). These observations together suggested that the expanded TIL cultures displayed a differentiated effector-like phenotype. Indeed, through the analysis of the combination of CD27 and CCR7 expression, irrespective of the disaggregation method, a preponderance of effector-like cells (CD27− CCR7−) was found within cultures with a reduced frequency of effector memory-like cells (CD27+ CCR7−) and negligible numbers of naive/central memory cells (CD27+ CCR7+) within both CD8+ and CD8− TIL populations (Supplementary Figure 1B and C).
In vitro functionality of expanded RCC TILs: conservation of HLA expression by GentleMACS disaggregation preserves TIL antitumour response
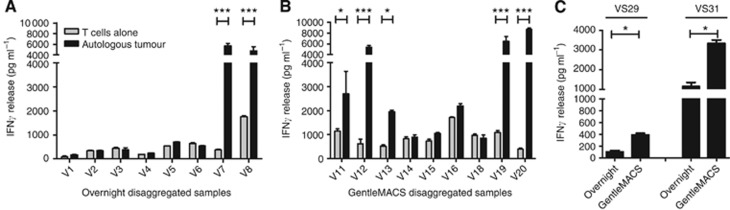
Functional response against an autologous tumour remains the key criteria to assess the tumour specificity of expanded TIL with IFNγ release after co-culture of TIL with a thawed, disaggregated tumour routinely used for this assessment (Dudley et al, 2003). Of the overnight digested cultures, only 25% (2 out of 8) co-cultures demonstrated IFNγ secretion above that of TIL alone (Figure 2A). By contrast, 56% (5 out of 9) of the GentleMACS-digested cultures produced significant levels of IFNγ during co-culture assays (Figure 2B). To confirm that this observation was not simply due to the tumour biopsies themselves, two biopsy samples (V29 and V31) that were large enough for parallel processing were cut into two identically sized pieces and processed by overnight digest or the GentleMACS dissociator in parallel and aliquots of tumour cells in the initial cell suspension were frozen for reactivity assays. IFNγ was only readily observed in both samples when expanded autologous TILs were co-cultured with a GentleMACS-disaggregated tumour (Figure 2C).
Figure 2.
Functional activity of expanded TILs against their autologous tumour cells. (A) Interferon gamma (IFNγ) secreted after 24-h co-culture of TILs with overnight and (B) GentleMACS-disaggregated autologous tumour samples. (C) IFNγ release of TILs after co-culture with an autologous tumour generated with parallel processing of the same tumour with either overnight digestion or GentleMACS disaggregation. Mean±s.e.m. of three experimental replicates are shown. *P<0.05, ***P<0.001. Two-way ANOVA with Bonferroni post-test correction.
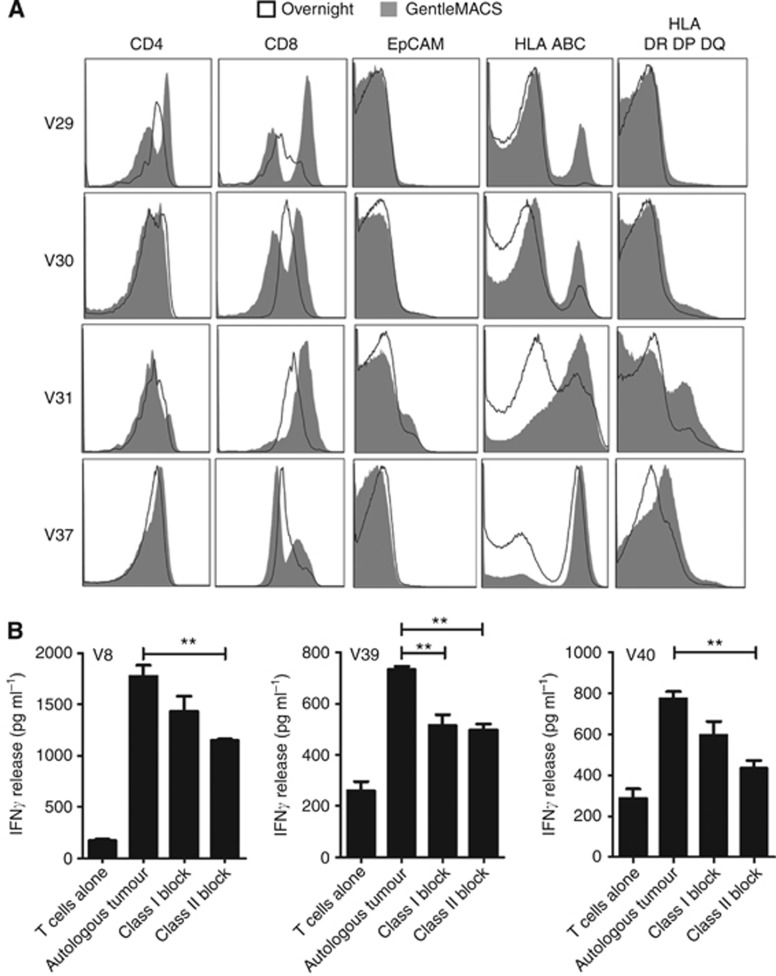
Since there were no obvious differences in the isolation, expansion and phenotype of the TIL populations expanded after the different disaggregation methods; to explain this differential level of antitumour functionality, we investigated what impact the disaggregation methods had on the tumour that was used as the assay target. Four further tumour samples (V29, V30, V31 and V37) were cut into two identically sized pieces and processed by either overnight or GentleMACS disaggregation, washed and analysed for the expression of various cell markers (using the scheme shown in Supplementary Figure 2). Within the lymphocyte gated CD3+CD45+ cell population, there was evidence of higher expression of the CD8 marker in all samples and an increased expression of CD4 in one sample undergoing GentleMACS as compared with overnight disaggregation (Figure 3A). In three samples processed by the GentleMACS, there was evidence of higher HLA class-I expression and in all four samples evidence of improved HLA class-II expression as compared with the matched overnight disaggregated sample (Figure 3A). In comparison, there was no difference in the immune reactivity to EpCAM between all samples (Figure 3A). Interestingly, blockade of HLA class II resulted in a consistent reduction of TIL-specific activity above that of HLA class I-specific blockade (Figure 3B), suggesting that both HLA I and II targeting were contributing to the functional activity of the expanded TIL against a digested autologous tumour. This importance of HLA expression raised the question of whether HLA expression was being induced during the manipulation process or whether the HLA molecules present upon the initial tumour sample were being lost during overnight enzymatic disaggregation. To examine this, 10 of the original tumour samples for which sample was available were immunostained for HLA-A,B,C expression, which clearly demonstrated that a consistently high level of expression of HLA class I was found on all tumours before disaggregation (Supplementary Figure 3). This strongly implies that cell surface HLA molecules are lost during overnight disaggregation but preserved by GentleMACS disaggregation technology. In addition, these tumour samples were immunostained for CD68, which indicated a significant variability in the level of macrophage infiltration into the tumour environment (Supplementary Figure 4). However, there was no obvious correlation between CD68+ macrophage infiltration and numbers or cytokine release potential of the TIL isolated from the specific tumour, albeit the number of tumours available for testing was limited (Supplementary Figure 4).
Figure 3.
Surface expression of markers on TILs after overnight or GentleMACS disaggregation. (A) Flow cytometric analysis of CD4, CD8, EpCAM, HLA A,B,C and HLA DR,DP, DQ expression on isolated, uncultured TILs from four renal cell carcinoma biopsies after overnight (black empty line) or GentleMACS (grey-filled line) disaggregation. (B) Effect of MHC class I and class II blockade on the release of IFNγ after 24-h co-culture of TILs with an autologous tumour in two RCC biopsies. **P<0.01 Unpaired t-test.
Maintenance of RCC TIL functionality after rapid expansion
Current TIL-adoptive transfer studies employ a REP to generate the large numbers of TILs required for clinical application (Dudley et al, 2003; Besser et al, 2010). Consequently, maintenance of tumour-targeting functionality after rapid expansion would be an essential criterion for RCC TILs to be relevant for therapeutic application. Six TIL cultures (two processed with overnight digest and four with GentleMACS dissociation) were subjected to a small-scale REP for 14 days after the initial culture, resulting in a 79.3±33-fold expansion (Figure 4A). Of these expanded TIL's, an autologous tumour was available for four (V6, V12, V14 and V15). Previously, V12, V14 and V15 TILs produced IFNγ in response to challenge with an autologous tumour (Figure 2B) and this was maintained in the REP-expanded cultures (Figure 4B). REP-expanded V6 TIL showed a limited increase in IFNγ response against an autologous tumour that failed to reach statistical significance (Figure 4B), which paralleled with the responses observed with the original expanded V6 TIL culture (Figure 2A). In order to determine whether the REP enhanced allogeneic response, expanded TILs failed to produce IFNγ after co-culture with a variety of tumour cell lines including the renal carcinoma cell line 2220R (Figure 4C), suggesting that increased allogeneic cytokine responses were not induced by REP. These observations strongly suggest that rapid expansion did not adversely impact on the specificity of the autologous tumour response of the individual TIL cultures.
Figure 4.
Expansion and functional activity of TILs after undergoing a rapid expansion protocol. (A) Growth curves of TILs subjected to a rapid expansion protocol (REP). (B) IFNγ release of rapid-expanded TILs V6, V12, V14 and V15 after co-culture with an autologous tumour. (C) Alloreactivity of rapid-expanded TILs V6, V12, V14 and V15 as determined by IFNγ release after co-culture of TILs with renal (2220R), gastric (MKN45K), melanoma (Mel624) and neuroblastoma (LA15S and SK-N-SH) cell lines. ***P<0.001, two-way ANOVA with Bonferroni post-test correction.
Discussion
Isolating TILs is technically demanding and difficult to standardise, given the variance in quantity and quality of the starting tissue and the relative lymphocyte infiltration into the tumour. Indeed, the only phase-III multi-centre clinical trial of RCC TIL therapy performed to date was terminated early due to the absence of clinical response in part due to the high TIL production failure rate and variety in the TIL product (Figlin et al, 1999).
TIL production for melanoma has been successfully adopted in different sites worldwide with largely equivalent frequencies of clinical responses reported between different centres, highlighting the success of technology transfer between establishments (Besser et al, 2010; Dudley et al, 2010) while efforts to standardise melanoma TIL production in compliance with good manufacturing process (GMP) are proceeding as a part of large multi-centre trial applications (e.g., NKI-AVL, Amsterdam (John Haanen), Herlev Hospital, Copenhagen, Denmark (Inge-Marie Svane), Christie Hospital, Manchester, UK (REH)).
However, efforts to culture TILs from RCC primary tumour biopsies using IL-2 alone to drive in vitro proliferation, as is the case in melanoma after tumour disaggregation, largely failed. RCC TIL outgrowth required the lymphocytes within the tumour digest to be activated with the most consistent results achieved when paramagnetic beads were added to the culture. Recent studies suggest that RCC TIL demonstrate signs of proliferative exhaustion potentially resulting from CD70 expression within RCC tumours (Wang et al, 2012), and this may explain the lack of proliferative response of RCC TIL to cytokine alone. With mitogenic stimulation, TILs were generated from the majority of primary tumour biopsies tested irrespective of the mode of tumour disaggregation employed. Moreover, the TIL phenotype was also similar between disaggregation methods, with the majority of the expanded cells displaying an effector-memory phenotype. The key differences found between the disaggregation methods were the reduced variability of TIL number and the preservation of cell surface antigen expression when the mechanical/enzymatic Gentlemacs system was used. Reduced variability in cell number is potentially important for standardisation of bioprocessing; however, antigen preservation is important to identify tumour-reactive TILs since the maintenance of MHC protein expression on tumour digests is essential for TIL functionality. Potency assays that consistently assess the activity of natural T-cells (i.e., those not endowed with antigen specificity by genetic modification) such as TILs are lacking. These results suggest that improved tumour disaggregation methods can produce target cells that are more consistently recognised by expanded TILs and this may form the basis for the development of a reliable potency assay.
To date, IFNγ release during co-culture has been the ‘gold standard' assay used to assess TIL reactivity (Dudley et al, 2003). In this work, IFNγ release was performed and identified functionally reactive RCC TILs; however, a detailed assessment of the ability to act as a potency assay that is predictive of RCC TIL function can only be determined within the clinical trial setting. Importantly, the GentleMACS system is not currently available in a GMP compliant format. However, with respect to potency assay methodology, it is feasible to use a strategy in the short term where a part of the tumour, which is prepared by GMP-compliant TIL production, is taken and processed using this system in a research laboratory environment, whereas the majority of the tumour is used to generate TIL under standard production conditions. Consequently, the expanded TIL populations can be tested against the GentleMACS autologous prepared tumour in potency assay testing without the need for validation of the GentleMACS system for clinical TIL production. However, in the long term, selection of tumour-specific TILs using capture of cells expressing activation-related markers such as PD-1 (Gros et al, 2014) or CD137 (Ye et al, 2014) directly from the tumour digest may be more required, and preservation of antigen integrity in the clinical setting may be required.
Outside of the RCC TIL-adoptive cell therapy context, maintenance of cell surface antigen expression will also facilitate studies investigating expression profiles of cells liberated from tumours that would have been lost by standard tissue preparation methods including the analysis of T-cell receptor usage (Gerlinger et al, 2013; Sittig et al, 2013), differentiation status (Wang et al, 2012) and subset frequencies (Igarashi et al, 2002) and, potentially, cytokine secretion profiles of lymphocytes invading into the tumour.
Irrespective of the tumour disaggregation method, the expanded RCC TIL displayed a predominantly effector phenotype reflecting other reports detailing the differentiated status of RCC TILs (Wang et al, 2012). Mitogenic activation to expand the RCC TILs in the presence of IL-2 would also contribute to this phenotype. Various studies investigating T-cell engraftment indicate that less-differentiated T cells possess enhanced in vivo engraftment (Gattinoni et al, 2012). Modulating the phenotype of isolated TIL by modulating culture conditions by the addition of cytokine cocktails such as IL-7 and IL-15 (Alcantar-Orozco et al, 2013; Yang et al, 2013) could modulate the TIL phenotype. Interestingly, antibody blockade experiments identified that tumour-driven RCC IFNγ secretion was driven by MHC I- and -II-restricted T cells, implying a functional role of CD4+ cells within the RCC TIL population. Others have reported CD4+ T-cell responses in RCC patients that recognise shared tumour antigens and also imply that MHC II expression may be upregulated on RCC tumour cells, suggesting the potential for CD4+ T cells to directly target RCC tumour cells (Mautner et al, 2005; Tassi et al, 2006). Increased frequencies of regulatory T cells have been identified in RCC patients (Griffiths et al, 2007). However, the CD4+ RCC TIL population expanded here secrete IFNγ and rapidly proliferate after mitogenic stimulation strongly suggesting that these cells are not regulatory in nature. Unfortunately, the activation process results in upregulation of markers used to identify regulatory T cells in peripheral blood (e.g., FoxP3), making such phenotypic analysis for regulatory T cells impossible in the expanded TIL population. Consequently, when considering developing a clinical trial protocol, the benefits of selecting CD8+ TIL as compared with adoptively transferring the unselected expanded TIL population where the CD4+ compartment may provide antitumour activity needs to be further examined. Moreover, there was no observed correlation between CD68+ macrophage infiltration and TIL number/antigen-specific cytokine secretion in this study. However, larger studies should include deeper investigation of the immune environment and tumour status (such as regulatory T-cell frequency in the initial TIL isolation, and immune checkpoint expression such as PD-L1) to determine whether factors that could predict the success of TIL expansion and function can be identified.
In summary, this work demonstrates that TIL can be reproducibly generated from primary and metastatic RCC biopsies. Importantly, these expanded TIL populations maintain in vitro autologous tumour reactivity that is enhanced by the use of methods that preserve the cell surface architecture of the disaggregated tumour sample. However, the mode of disaggregation did not appear to affect initial TIL production. Thus, as the GentleMACS is currently not certified for clinical use, the likely procedure would involve disaggregating the majority of the tumour biopsies using standard methods in the cleanroom, with a smaller tumour fragment processed using the GentleMACS in the research environment for use in functionality testing of the expanded TIL product. On the basis of this work, phase I/II trial protocols are being developed to assess the clinical potency of TILs against the RCC setting, which includes validation of the TIL production process using GMP-compliant materials. Moreover, these approaches are being tested in other tumour indications to assess the general applicability of this process to generate TILs for therapy.
REH and DEG are both co-founders of Cellular Therapeutics Ltd. All other authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on British Journal of Cancer website (http://www.nature.com/bjc)
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Material
References
- Alcantar-Orozco EM, Gornall H, Baldan V, Hawkins RE, Gilham DE. Potential limitations of the NSG humanized mouse as a model system to optimize engineered human T cell therapy for cancer. Hum Gene Ther Methods. 2013;24:310–320. doi: 10.1089/hgtb.2013.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Besser MJ, Shapira-Frommer R, Itzhaki O, Treves AJ, Zippel DB, Levy D, Kubi A, Shoshani N, Zikich D, Ohayon Y, Ohayon D, Shalmon B, Markel G, Yerushalmi R, Apter S, Ben-Nun A, Ben-Ami E, Shimoni A, Nagler A, Schachter J. Adoptive transfer of tumor-infiltrating lymphocytes in patients with metastatic melanoma: intent-to-treat analysis and efficacy after failure to prior immunotherapies. Clin Cancer Res. 2013;19:4792–4800. doi: 10.1158/1078-0432.CCR-13-0380. [DOI] [PubMed] [Google Scholar]
- Besser MJ, Shapira-Frommer R, Treves AJ, Zippel D, Itzhaki O, Hershkovitz L, Levy D, Kubi A, Hovav E, Chermoshniuk N, Shalmon B, Hardan I, Catane R, Markel G, Apter S, Ben-Nun A, Kuchuk I, Shimoni A, Nagler A, Schachter J. Clinical responses in a phase II study using adoptive transfer of short-term cultured tumor infiltration lymphocytes in metastatic melanoma patients. Clin Cancer Res. 2010;16:2646–2655. doi: 10.1158/1078-0432.CCR-10-0041. [DOI] [PubMed] [Google Scholar]
- Bouet-Toussaint F, Genetel N, Rioux-Leclercq N, Bansard JY, Leveque J, Guille F, Patard JJ, Lesimple T, Catros-Quemener V. Interleukin-2 expanded lymphocytes from lymph node and tumor biopsies of human renal cell carcinoma, breast and ovarian cancer. Eur Cytokine Netw. 2000;11:217–224. [PubMed] [Google Scholar]
- Danilova E, Skrindo I, Gran E, Hales BJ, Smith WA, Jahnsen J, Johansen FE, Jahnsen FL, Baekkevold ES. A role for CCL28-CCR3 in T-cell homing to the human upper airway mucosa. Mucosal Immunol. 2015;8 (1:107–114. doi: 10.1038/mi.2014.46. [DOI] [PubMed] [Google Scholar]
- Dillman R, Schiltz P, DePriest C, Barth N, Beutel L, de Leon C, O'Connor A, Nayak S. Tumor-infiltrating lymphocytes and interleukin-2: dose and schedules of administration in the treatment of metastatic cancer. Cancer Biother Radiopharm. 2004;19:730–737. doi: 10.1089/cbr.2004.19.730. [DOI] [PubMed] [Google Scholar]
- Dudley ME, Gross CA, Langhan MM, Garcia MR, Sherry RM, Yang JC, Phan GQ, Kammula US, Hughes MS, Citrin DE, Restifo NP, Wunderlich JR, Prieto PA, Hong JJ, Langan RC, Zlott DA, Morton KE, White DE, Laurencot CM, Rosenberg SA. CD8+ enriched "young" tumor infiltrating lymphocytes can mediate regression of metastatic melanoma. Clin Cancer Res. 2010;16:6122–6131. doi: 10.1158/1078-0432.CCR-10-1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudley ME, Wunderlich JR, Shelton TE, Even J, Rosenberg SA. Generation of tumor-infiltrating lymphocyte cultures for use in adoptive transfer therapy for melanoma patients. J Immunother. 2003;26:332–342. doi: 10.1097/00002371-200307000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudley ME, Yang JC, Sherry R, Hughes MS, Royal R, Kammula U, Robbins PF, Huang J, Citrin DE, Leitman SF, Wunderlich J, Restifo NP, Thomasian A, Downey SG, Smith FO, Klapper J, Morton K, Laurencot C, White DE, Rosenberg SA. Adoptive cell therapy for patients with metastatic melanoma: evaluation of intensive myeloablative chemoradiation preparative regimens. J Clin Oncol. 2008;26:5233–5239. doi: 10.1200/JCO.2008.16.5449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figlin RA, Pierce WC, Kaboo R, Tso CL, Moldawer N, Gitlitz B, deKernion J, Belldegrun A. Treatment of metastatic renal cell carcinoma with nephrectomy, interleukin-2 and cytokine-primed or CD8(+) selected tumor infiltrating lymphocytes from primary tumor. J Urol. 1997;158:740–745. doi: 10.1097/00005392-199709000-00012. [DOI] [PubMed] [Google Scholar]
- Figlin RA, Thompson JA, Bukowski RM, Vogelzang NJ, Novick AC, Lange P, Steinberg GD, Belldegrun AS. Multicenter, randomized, phase III trial of CD8(+) tumor-infiltrating lymphocytes in combination with recombinant interleukin-2 in metastatic renal cell carcinoma. J Clin Oncol. 1999;17:2521–2529. doi: 10.1200/JCO.1999.17.8.2521. [DOI] [PubMed] [Google Scholar]
- Gattinoni L, Klebanoff CA, Restifo NP. Paths to stemness: building the ultimate antitumour T cell. Nat Rev Cancer. 2012;12:671–684. doi: 10.1038/nrc3322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerlinger M, Quezada SA, Peggs KS, Furness AJ, Fisher R, Marafioti T, Shende VH, McGranahan N, Rowan AJ, Hazell S, Hamm D, Robins HS, Pickering L, Gore M, Nicol DL, Larkin J, Swanton C. Ultra-deep T cell receptor sequencing reveals the complexity and intratumour heterogeneity of T cell clones in renal cell carcinomas. J Pathol. 2013;231:424–432. doi: 10.1002/path.4284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goedegebuure PS, Douville LM, Li H, Richmond GC, Schoof DD, Scavone M, Eberlein TJ. Adoptive immunotherapy with tumor-infiltrating lymphocytes and interleukin-2 in patients with metastatic malignant melanoma and renal cell carcinoma: a pilot study. J Clin Oncol. 1995;13:1939–1949. doi: 10.1200/JCO.1995.13.8.1939. [DOI] [PubMed] [Google Scholar]
- Gilham DE, O'Neil A, Hughes C, Guest RD, Kirillova N, Lehane M, Hawkins RE. Primary polyclonal human T lymphocytes targeted to carcino-embryonic antigens and neural cell adhesion molecule tumor antigens by CD3zeta-based chimeric immune receptors. J Immunother. 2002;25 (2:139–151. doi: 10.1097/00002371-200203000-00002. [DOI] [PubMed] [Google Scholar]
- Griffiths RW, Gilham DE, Dangoor A, Ramani V, Clarke NW, Stern PL, Hawkins RE. Expression of the 5T4 oncofoetal antigen in renal cell carcinoma: a potential target for T-cell-based immunotherapy. Br J Cancer. 2005;93 (6:670–677. doi: 10.1038/sj.bjc.6602776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths RW, Elkord E, Gilham DE, Ramani V, Clarke N, Stern PL, Hawkins RE. Frequency of regulatory T cells in renal cell carcinoma patients and investigation of correlation with survival. Cancer Immunol Immunother. 2007;56:1743–1753. doi: 10.1007/s00262-007-0318-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gros A, Robbins PF, Yao X, Li YF, Turcotte S, Tran E, Wunderlich JR, Mixon A, Farid S, Dudley ME, Hanada K, Almeida JR, Darko S, Douek DC, Yang JC, Rosenberg SA. PD-1 identifies the patient-specific CD8(+) tumor-reactive repertoire infiltrating human tumors. J Clin Invest. 2014;124:2246–2259. doi: 10.1172/JCI73639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Igarashi T, Takahashi H, Tobe T, Suzuki H, Mizoguchi K, Nakatsu HO, Ito H. Effect of tumor-infiltrating lymphocyte subsets on prognosis and susceptibility to interferon therapy in patients with renal cell carcinoma. Urol Int. 2002;69:51–56. doi: 10.1159/000064361. [DOI] [PubMed] [Google Scholar]
- Malone CC, Schiltz PM, Mackintosh AD, Beutel LD, Heinemann FS, Dillman RO. Characterization of human tumor-infiltrating lymphocytes expanded in hollow-fiber bioreactors for immunotherapy of cancer. Cancer Biother Radiopharm. 2001;16:381–390. doi: 10.1089/108497801753354285. [DOI] [PubMed] [Google Scholar]
- Markel G, Cohen-Sinai T, Besser MJ, Oved K, Itzhaki O, Seidman R, Fridman E, Treves AJ, Keisari Y, Dotan Z, Ramon J, Schachter J. Preclinical evaluation of adoptive cell therapy for patients with metastatic renal cell carcinoma. Anticancer Res. 2009;29:145–154. [PubMed] [Google Scholar]
- Mautner J, Jaffee EM, Pardoll DM. Tumor-specific CD4+ T cells from a patient with renal cell carcinoma recognize diverse shared antigens. Int J Cancer. 2005;115:752–759. doi: 10.1002/ijc.20927. [DOI] [PubMed] [Google Scholar]
- McLoughlin RM, Lee JC, Kasper DL, Tzianabos AO. IFN-gamma regulated chemokine production determines the outcome of Staphylococcus aureus infection. J Immunol. 2008;181 (2:1323–1332. doi: 10.4049/jimmunol.181.2.1323. [DOI] [PubMed] [Google Scholar]
- Rosenberg SA, Yang JC, Sherry RM, Kammula US, Hughes MS, Phan GQ, Citrin DE, Restifo NP, Robbins PF, Wunderlich JR, Morton KE, Laurencot CM, Steinberg SM, White DE, Dudley ME. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res. 2011;17:4550–4557. doi: 10.1158/1078-0432.CCR-11-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sittig SP, Kollgaard T, Gronbaek K, Idorn M, Hennenlotter J, Stenzl A, Gouttefangeas C, Thor Straten P. Clonal expansion of renal cell carcinoma-infiltrating T lymphocytes. Oncoimmunology. 2013;2:e26014. doi: 10.4161/onci.26014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahara M, Miyai M, Tomiyama M, Mutou M, Nicol AJ, Nieda M. Copulsing tumor antigen-pulsed dendritic cells with zoledronate efficiently enhance the expansion of tumor antigen-specific CD8+ T cells via Vgamma9 gamma delta T cell activation. J Leukoc Biol. 2008;83 (3:742–754. doi: 10.1189/jlb.0307185. [DOI] [PubMed] [Google Scholar]
- Takeuchi M, Inoue T, Otani T, Yamasaki F, Nakamura S, Kibata M. Cell-in-cell structures formed between human cancer cell lines and the cytotoxic regulatory T-cell line HOZOT. J Mol Cell Biol. 2010;2 (3:139–151. doi: 10.1093/jmcb/mjq002. [DOI] [PubMed] [Google Scholar]
- Tassi E, Facchinetti V, Seresini S, Borri A, Dell'antonio G, Garavaglia C, Casorati G, Protti MP. Peptidome from renal cell carcinoma contains antigens recognized by CD4+ T cells and shared among tumors of different histology. Clin Cancer Res. 2006;12:4949–4957. doi: 10.1158/1078-0432.CCR-06-0995. [DOI] [PubMed] [Google Scholar]
- Thompson JA, Figlin RA, Sifri-Steele C, Berenson RJ, Frohlich MW. A phase I trial of CD3/CD28-activated T cells (Xcellerated T cells) and interleukin-2 in patients with metastatic renal cell carcinoma. Clin Cancer Res. 2003;9:3562–3570. [PubMed] [Google Scholar]
- Tran KQ, Zhou J, Durflinger KH, Langhan MM, Shelton TE, Wunderlich JR, Robbins PF, Rosenberg SA, Dudley ME. Minimally cultured tumor-infiltrating lymphocytes display optimal characteristics for adoptive cell therapy. J Immunother. 2008;31:742–751. doi: 10.1097/CJI.0b013e31818403d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang QJ, Hanada K, Robbins PF, Li YF, Yang JC. Distinctive features of the differentiated phenotype and infiltration of tumor-reactive lymphocytes in clear cell renal cell carcinoma. Cancer Res. 2012;72:6119–6129. doi: 10.1158/0008-5472.CAN-12-0588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S, Ji Y, Gattinoni L, Zhang L, Yu Z, Restifo NP, Rosenberg SA, Morgan RA. Modulating the differentiation status of ex vivo-cultured anti-tumor T cells using cytokine cocktails. Cancer Immunol Immunother. 2013;62:727–736. doi: 10.1007/s00262-012-1378-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yannelli JR, Hyatt C, McConnell S, Hines K, Jacknin L, Parker L, Sanders M, Rosenberg SA. Growth of tumor-infiltrating lymphocytes from human solid cancers: summary of a 5-year experience. Int J Cancer. 1996;65:413–421. doi: 10.1002/(SICI)1097-0215(19960208)65:4<413::AID-IJC3>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- Ye Q, Song DG, Poussin M, Yamamoto T, Best A, Li C, Coukos G, Powell DJ., Jr. CD137 accurately identifies and enriches for naturally occurring tumor-reactive T cells in tumor. Clin Cancer Res. 2014;20:44–55. doi: 10.1158/1078-0432.CCR-13-0945. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.