Summary
Successful emergence from the soil is essential for plant establishment in natural and farmed systems. It has been assumed that the absence of light in the soil is the preeminent signal perceived during early seedling development, leading to a distinct morphogenic plan (skotomorphogenesis) [1], characterized by traits providing an adaptive advantage until emergence and photomorphogenesis. These traits include suppressed chlorophyll synthesis, promotion of hypocotyl elongation, and formation of a closed apical hook that protects the stem cell niche from damage [2, 3]. However, absence of light by itself is not a sufficient environmental signal for early seedling development [4, 5]. Reduced oxygen levels (hypoxia) can occur in water-logged soils [6–8]. We therefore hypothesized that below-ground hypoxia may be an important, but thus far undiscovered, ecological component regulating seedling development. Here, we show that survival and establishment of seedlings following darkness depend on their ability to sense hypoxia, through enhanced stability of group VII Ethylene Response Factor (ERFVII) transcription factors. Hypoxia is perceived as a positive environmental component in diverse taxa of flowering plants, promoting maintenance of skotomorphogenic traits. Hypoxia greatly enhances survival once light is perceived, while oxygen is necessary for the subsequent effective completion of photomorphogenesis. Together with light perception, oxygen sensing therefore allows an integrated response to the complex and changing physical microenvironment encountered during early seedling growth. We propose that plants monitor the soil’s gaseous environment after germination, using hypoxia as a key external cue to protect the stem cell niche, thus ensuring successful rapid establishment upon emergence above ground.
Highlights
-
•
Low oxygen (hypoxia) enhances seedling survival in the dark
-
•
Hypoxia is sensed by oxygen-mediated degradation of ERFVII transcription factors
-
•
Stable ERFVIIs enhance dark-activated development and repress light-activated development
-
•
Plants monitor the gaseous environment to coordinate photomorphogenesis
Abbas et al. demonstrate that hypoxia sensing is an important component of photomorphogenesis. Hypoxia acts as a positive external positional cue controlling morphogenetic and biochemical adaptations to protect the stem cell niche. Sensing hypoxia in the dark coordinates responses that maximize survival and establishment in the light.
Results and Discussion
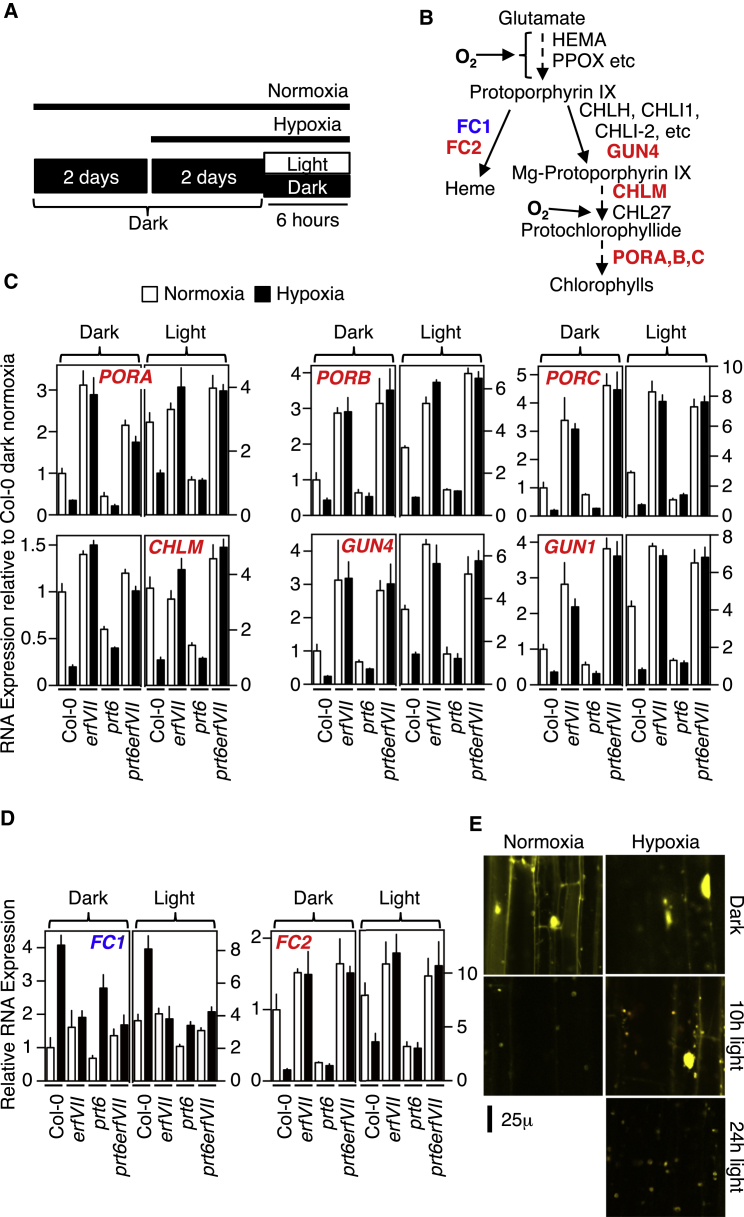
We analyzed the effect of oxygen availability and light on seedling growth following germination in species representing distinct branches of eudicot phylogeny: Papaver somniferum, Nicotiana benthamiana, and Arabidopsis thaliana (Arabidopsis). Apical hook development is characterized by three phases: formation, maintenance, and opening [5, 9]. Contrary to the observation that seedlings kept in darkness under normal oxygen levels (normoxia) eventually open the hook (Figure 1A), we found that hypoxic conditions (2% oxygen imposed following the hook maintenance phase) strongly inhibited opening in all species, which was reverted following transfer back to normoxia (Figures 1A and 1B). The response of final hook angle to increasing oxygen tensions in Arabidopsis is linear, suggesting stochastic cumulative sensing and response to oxygen (Figure 1C) [10]. Etiolated seedlings maintained under hypoxia were able to complete hook opening when exposed to light but had defective unfolding and greening of cotyledons, indicating that oxygen is required for the complete response to light (Figure 1D). Oxygen sensing in flowering plants is therefore a major component of skotomorphogenic development and the transition to photomorphogenesis.
Figure 1.
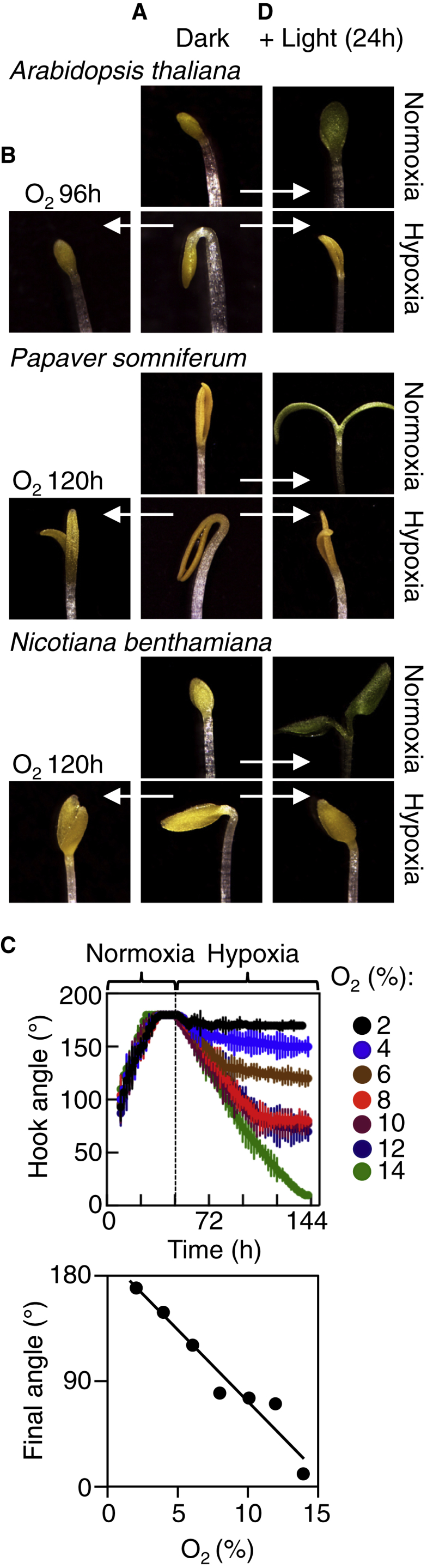
Skotomorphogenic Traits Are Regulated by Oxygen Sensing in Flowering Plants
(A) Images of final apical hook angle of etiolated seedlings of Arabidopsis thaliana, Papaver somniferum, and Nicotiana benthamiana in normoxia or hypoxia (168 hr, 168 hr, and 144 hr in the dark, respectively). Hypoxia treatment was continuously applied following initiation of the hook maintenance phase.
(B) Response of the apical hook after transfer from hypoxia (A) to normoxia for the indicated times.
(C) Response of the apical hook angle of Arabidopsis to increasing oxygen levels.
(D) Images of cotyledon greening of seedlings following transfer to light under normoxia or hypoxia (24 hr).
Error bars indicate SD from the mean.
Oxygen is sensed in plants by the Cysteine (Cys) branch of the Arginine (Arg)/N-end rule pathway of targeted proteolysis, using group VII Ethylene Response Factors (ERFVIIs) as substrates [11, 12] (Figure 2A). The N-end rule pathway relates the in vivo stability of a protein to the nature of its N terminus, which may be stabilizing or destabilizing (the N-degron) [14]. In normoxia, ERFVIIs are destabilized through oxidation of N-terminal (Nt)-Cys, which targets the proteins for degradation via the N-end rule pathway. Under hypoxia, Nt-Cys is not oxidized and substrates are stable, enhancing growth and development [11–13]. This mechanism is also used to sense nitric oxide (NO), an essential component of Nt-Cys oxidation [15, 16]. There are five ERFVIIs in Arabidopsis: RELATED TO AP (RAP)2.12, RAP2.2, RAP2.3, HYPOXIA RESPONSIVE (HRE)1, and HRE2 [17].
Figure 2.
Oxygen Sensing during Skotomorphogenesis and Photomorphogenesis Is Controlled by ERFVII Transcription Factors and the N-End Rule Pathway
(A) Schematic of the Cys-Arg/N-end rule pathway. Single amino acid abbreviations are used. ox, oxidized; NO, nitric oxide; PCO, plant-specific Plant Cysteine Oxidase [13]; MAP, Methionine Amino-Peptidase; ATE Arginyl Transferase; PRT6, PROTEOLYSIS E3 ligase. Protein substrates are indicated as blue ovals.
(B–E) Dynamics of apical hook angle for etiolated seedlings of WT (Col-0), N-end rule pathway and NO synthesis mutants, and transgenics, containing stabilized ERFVIIs expressed under endogenous promoters (promERFVII:MA-ERFVII).
(F) Chlorophyll content of 4-day-old seedlings transferred to the light for 6 hr.
Error bars indicate SD from the mean.
We investigated whether early seedling growth in Arabidopsis is controlled by oxygen sensing through the N-end rule pathway. We analyzed apical hook development in mutant seedlings lacking either E3 ligase (PRT6) or Arginyl-transferase (ATE) functions (Figure 2A). In these mutants, substrates of the Cys-Arg/N-end rule pathway are constitutively stable, but with different N termini. In contrast to wild-type (WT; accession Col-0) etiolated seedlings, apical hooks of prt6 and ate1 ate2 did not fully open in the dark under normoxia (Figure 2B). Remarkably, the prt6 rap2.12 rap2.2 rap2.3 hre1 hre2 sextuple mutant (prt6 erfVII), which lacks the function of all five ERFVIIs, reverted the prt6 phenotype (Figures 2B and S1), indicating that ERFVIIs act redundantly to repress hook opening. Opening of apical hooks was also inhibited in NO-deficient mutants, a phenotype reverted in the nia1 nia2 erfVII mutant or by treatment with NO (Figures 2C and S1). To confirm that ERFVIIs are an integral component of oxygen sensing controlling early seedling development, we tested the ability of individual ERFVIIs to repress hook opening by expressing mutant stable versions (in which Cys-2 was changed to Alanine, a stabilizing residue; Figure 2A) driven by their endogenous promoters (promERFVII:MA-ERFVII) providing MA-ERFVII protein. All five MA-ERFVIIs were able to inhibit hook opening compared to WT, indicating that Cys-Arg-N-end rule-mediated degradation of all ERFVIIs contributes to hook opening (Figure 2D). In order to provide unequivocal evidence that oxygen sensing by ERFVIIs controls hook opening, we analyzed hook development in WT and rap2.12 rap2.2 rap2.3 hre1 hre2 (erfVII) pentuple mutant (which lacks all ERFVII activity) seedlings under conditions where hypoxia was imposed at the end of the hook maintenance phase (Figure 2E). Under hypoxia, WT etiolated seedlings were completely unable to open their hooks. This, however, was not the result of a loss of respiratory energy but a specific consequence of oxygen sensing, as erfVII mutant seedlings completed hook opening under hypoxia with similar kinetics to WT seedlings under normoxia. In addition, return to normoxia following a prolonged hypoxic treatment allowed hooks of WT seedlings to open (Figure S1), reaffirming a role for a fast-responding oxygen-sensing mechanism. We observed no effect of hypoxia on hypocotyl elongation, demonstrating that the observed phenotypes are not a consequence of hypoxia-induced quiescence (Figure S1). Apical hook development has been intimately linked to the dynamics of auxin levels [2]. Accordingly, we observed a strong correlation between the presence of a gradient of auxin activity across the apical hook and the maintenance of the hook under hypoxia (Figure S1), suggesting oxygen-mediated regulation of localized auxin responses.
Given that oxygen is required for complete response to light (Figure 1D), we investigated whether greening of cotyledons, another key component of photomorphogenesis, is also mediated by oxygen-dependent ERFVII degradation. Etiolated seedlings of WT and N-end rule mutants were analyzed following transfer to light under hypoxic or normoxic conditions. After transfer to light under normoxia, total chlorophyll levels were much lower in prt6, ate1 ate2, and nia1 nia2 mutants compared to WT, but similar in prt6 erfVII (Figure 2F). Chlorophyll levels were also reduced in WT seedlings under hypoxia following transfer to light, but not in erfVII. Individual stable ERVIIs MA-HRE1 and MA-RAP2.3 driven by their endogenous promoters were not able to repress chlorophyll accumulation, indicating that they act in combination, perhaps as part of heteromeric complexes, with other ERFVIIs, unlike their roles in apical hook maintenance.
Our results indicate that oxygen availability is sensed by ERFVIIs during early seedling development and prompt the hypothesis that low oxygen levels would also enhance seedling survival during exposure to prolonged darkness. As previously reported [4], we found that after extended exposure to dark under normoxia, WT seedlings died, whereas here we show that hypoxia allowed complete recovery of cotyledon greening and even the capacity for primary leaf growth (Figures 3A, 3B, and 3C). Hypoxia-mediated survival was completely dependent on ERFVIIs (Figures 3B and 3C), which restricted the accumulation of reactive oxygen species (ROS) (Figure S2). As a confirmation for the involvement of ERFVIIs in survival after long exposure to darkness, only prt6, but not prt6 erfVII, seedlings were able to survive long-term exposure to dark normoxia (Figure 3D). Photo-oxidative damage caused by high ROS levels has been linked to excessive accumulation of protochlorophyllide (PC) in darkness [4]; therefore, observed reduced levels of PC in prt6 mutants compared with WT, erfVII and prt6 erfVII (Figure 3E), are the likely explanation for the enhanced survival. These results demonstrate that stabilized ERFVIIs protect seedlings from prolonged exposure to dark conditions and permit subsequent growth of the apical meristem following transfer to light.
Figure 3.
Stabilized ERFVIIs Enhance Long-Term Survival in the Dark
(A) Schematic of experimental design to analyze the effect of long-term development in the dark followed by transfer to the light.
(B and C) Quantification and images of cotyledon greening and primary leaf expansion in WT and erfVII seedlings in response to light following exposure to dark under hypoxic or normoxic conditions.
(D) Quantification of cotyledon greening and primary leaf expansion in WT and mutant seedlings in response to light following exposure to increased periods of dark under normoxic conditions.
(E) Relative fluorescence of protochlorophyllide in 4-day etiolated seedlings.
Error bars indicate SD from the mean.
In darkness, chlorophyll biosynthesis is known to be repressed by PIF transcription factors [18]. However, our analysis of PC accumulation suggested that ERFVIIs repress expression of chlorophyll biosynthesis genes in low-oxygen environments. We therefore analyzed the influence of light and hypoxia on mRNA transcript accumulation for enzymes of tetrapyrrole synthesis (Figures 4A–4D). For several genes, expression in etiolated seedlings was not influenced by hypoxia (Figure S3). However, expression of the chloroplastic form of heme synthase, FC2, and CHLM, PORA, PORB, PORC GUN1, and GUN4 was greatly repressed in WT by hypoxia both in the dark and, following transfer, in the light, and was also constitutively repressed in prt6 independent of oxygen availability (Figures 4C and 4D). Furthermore, this repression was not observed in erfVII or prt6 erfVII mutants, indicating that downregulation is achieved by stabilized ERFVIIs. In contrast, FC1 (encoding chloroplastic and mitochondrial heme synthase [19]) expression was enhanced under hypoxia, in an ERFVII-dependent manner (Figure 4D). Our results suggest a homeostatic mechanism whereby hypoxia-stabilized ERFVIIs repress several steps of the oxygen-requiring tetrapyrrole pathway. Together, these results demonstrate an important role for oxygen sensing in chlorophyll biosynthesis by N-end rule control of ERFVII stability.
Figure 4.
Control of Tetrapyrrole Synthesis Gene Expression by Oxygen and ERFVIIs
(A) Schematic of experimental design. Etiolated seedlings were grown for 4 days in the dark, the final 2 days being in either normoxia or hypoxia. Then, seedlings were left in the dark or exposed to light for 6 hr.
(B) Diagram of chlorophyll biosynthesis pathway. Enzymes are shown next to intermediate compounds of the pathway. Oxygen (O2)-dependent sections of the pathway are indicated.
(C and D) Expression of tetrapyrrole and heme synthesis genes in WT and mutant lines in response to hypoxia and light.
(E) Changes in subcellular location and abundance of constitutively expressed YFP-RAP2.3 protein in response to oxygen and light.
Error bars indicate SD from the mean.
The observation that in the light chlorophyll biosynthesis is not completely impaired in hypoxia suggests light either compensates for repression of chlorophyll biosynthesis by ERFVIIs or promotes inactivation of ERFVIIs. It has been shown that cytosolic RAP2.12 moves to the nucleus in response to the hypoxic signal [12]. In agreement with this result, we found that in etiolated seedlings under hypoxia, the subcellular location of constitutively expressed stabilized RAP2.3 (35S:YFP-RAP2.3) changed from cytoplasm and nucleus to exclusively nucleus (Figures 4E and S3). However, under normoxia and following transfer to light, this stabilized ERFVII is removed from the nucleus, through a mechanism unrelated to the ERFVII N-degron, as Nt-Cys is not present in the analyzed protein due to the Nt-YFP fusion. However, it is interesting to note that this degradation occurs much more slowly in hypoxia. These results show that light can override ERFVII function even under hypoxia, eventually becoming the dominant environmental signal.
In addition to its metabolic requirement for aerobic respiration, higher eukaryotes use specific sensing of molecular oxygen as a mechanism to control physiology and development [20, 21]. Animals use a different Hypoxia-Inducible Factor (HIF) system to sense oxygen [20]. It has been shown that embryos of Caenorhabditis elegans exhibit diapause (arrested development) in response to hypoxia, controlled by maternal nonautonomous expression of neural HIF-1 activity [22]. The rice Submergence1A (Sub1A) locus (encoding an ERFVII) provides tolerance to submergence-induced hypoxia through a quiescence strategy [21], and work reported here demonstrates that etiolated seedlings of higher plants restrict photomorphogenic development under low-oxygen stress and survive extended low-oxygen conditions through stabilization of ERFVIIs. Thus, morphogenetic and biochemical adaptations to survive hypoxic environments in animals and plants may share common features.
Our data demonstrate that oxygen sensing, in addition to light perception, is a key component of early seedling development, functioning to protect the apical meristem stem cell niche and prevent photo-oxidative damage. We show that long-term survival of seedlings in the dark depends on their ability to sense oxygen and that counter-intuitively hypoxic conditions are an important environmental component enhancing long-term seedling survival. In addition to previous work demonstrating a role for ERFVIIs in plant responses to waterlogging and hypoxia, our current work highlights how sensing of the gaseous environment may play a more general role in plant growth and development. Low light and high ethylene production are similarities between the phenomenologies of flooding and skotomorphogenesis [23, 24]. However, the response of etiolated seedlings described here is not related directly to escape or quiescence strategies associated with long-term submergence [21] but to rapid and non-permanent hypoxic conditions that could occur, for example, after heavy rain in soil with good draining capacity. In agreement with this hypothesis, oxygen sensing does not appear to influence one skotomorphogenic trait, hypocotyl elongation, as etiolated prt6 seedlings, and WT and erfVII seedlings under hypoxia, elongate hypocotyls similarly to WT under normoxia [15] (Figure S1), suggesting that oxygen sensing is specifically related to protection of the apical meristem stem cell niche.
Recently it was shown that oxygen acts as an internal developmental positional cue in plants [25]; in this work, we have shown that it also acts as an environmental positional cue. The underground environment combines both oxygen and light gradients. We suggest that whereas PIF function integrates responses to light in early seedling growth [26, 27], ERFVII function integrates responses to the gaseous environment, and the extent of overlap between the two pathways remains to be determined. Oxygen sensing may therefore provide an adaptive advantage to seedlings growing through the soil, allowing changes in the gaseous atmosphere to be sensed prior to the irreversible transition to photomorphogenic growth. Ultimately, removal of ERFVII-repressive function is assured when seedlings reach the soil surface through oxygen and NO-mediated destruction. Together with light perception, this allows an integrated response to the complex and changing physical microenvironment encountered by the growing etiolated seedling as it struggles through the soil to reach the surface.
Author Contributions
M.J.H., M.A.B., D.A., J.L., M.A., G.W.B., and S.B. conceived and designed experiments. M.A., S.B., J.V.C., D.J.G., D.R., C.S.C., G.W.B., N.M-d.l.R., J.L., D.A., and M.J.H. performed the experiments. M.J.H., M.A.B., D.A., J.L., and M.A. analyzed the data. M.J.H and M.A.B wrote the manuscript.
Acknowledgments
M.J.H., D.J.G., J.V.C., and C.S.C. were supported by BBSRC grants BB/G010595/1 and BB/K000144/1 (including financial support from SABMiller). G.W.B. was supported by a Marie Curie International Incoming Fellowship. S.B. was supported by a University of Nottingham PhD fellowship. D.R. was supported by a BBSRC DTP PhD fellowship. J.L. and M.A.B., D.A., and M.A. were supported, respectively, through grants from MICINN (Spain) BIO2011-27526 and BIO2010-15071. We thank Christian Fankhauser and François Parcy for comments on the manuscript.
Footnotes
This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Contributor Information
Miguel A. Blázquez, Email: mblazquez@ibmcp.upv.es.
Michael J. Holdsworth, Email: michael.holdsworth@nottingham.ac.uk.
Supplemental Information
References
- 1.Wu S.H. Gene expression regulation in photomorphogenesis from the perspective of the central dogma. Annu. Rev. Plant Biol. 2014;65:311–333. doi: 10.1146/annurev-arplant-050213-040337. [DOI] [PubMed] [Google Scholar]
- 2.Abbas M., Alabadí D., Blázquez M.A. Differential growth at the apical hook: all roads lead to auxin. Front. Plant Sci. 2013;4:441. doi: 10.3389/fpls.2013.00441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mazzella M.A., Casal J.J., Muschietti J.P., Fox A.R. Hormonal networks involved in apical hook development in darkness and their response to light. Front. Plant Sci. 2014;5:52. doi: 10.3389/fpls.2014.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sperling U., van Cleve B., Frick G., Apel K., Armstrong G.A. Overexpression of light-dependent PORA or PORB in plants depleted of endogenous POR by far-red light enhances seedling survival in white light and protects against photooxidative damage. Plant J. 1997;12:649–658. doi: 10.1046/j.1365-313x.1997.00649.x. [DOI] [PubMed] [Google Scholar]
- 5.Raz V., Ecker J.R. Regulation of differential growth in the apical hook of Arabidopsis. Development. 1999;126:3661–3668. doi: 10.1242/dev.126.16.3661. [DOI] [PubMed] [Google Scholar]
- 6.Crawford R.M.M. Oxygen availability as an ecological limit to plant distribution. In: Begon M., Fitter A.H., editors. Volume 23. Academic Press; 1992. pp. 93–185. (Advances in Ecological Research). [Google Scholar]
- 7.Liptzin D., Silver W.L., Detto M. Temporal dynamics in soil oxygen and greenhouse gases in two humid tropical forests. Ecosystems. 2011;14:171–182. [Google Scholar]
- 8.Silver W.L., Lugo A.E., Keller M. Soil oxygen availability and biogeochemistry along rainfall and topographic gradients in upland wet tropical forest soils. Biogeochemistry. 1999;44:301–328. [Google Scholar]
- 9.Gallego-Bartolomé J., Arana M.V., Vandenbussche F., Zádníková P., Minguet E.G., Guardiola V., Van Der Straeten D., Benkova E., Alabadí D., Blázquez M.A. Hierarchy of hormone action controlling apical hook development in Arabidopsis. Plant J. 2011;67:622–634. doi: 10.1111/j.1365-313X.2011.04621.x. [DOI] [PubMed] [Google Scholar]
- 10.Kosmacz M., Parlanti S., Schwarzländer M., Kragler F., Licausi F., VAN Dongen J.T. The stability and nuclear localization of the transcription factor RAP2.12 are dynamically regulated by oxygen concentration. Plant Cell Environ. 2014 doi: 10.1111/pce.12493. [DOI] [PubMed] [Google Scholar]
- 11.Gibbs D.J., Lee S.C., Isa N.M., Gramuglia S., Fukao T., Bassel G.W., Correia C.S., Corbineau F., Theodoulou F.L., Bailey-Serres J., Holdsworth M.J. Homeostatic response to hypoxia is regulated by the N-end rule pathway in plants. Nature. 2011;479:415–418. doi: 10.1038/nature10534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Licausi F., Kosmacz M., Weits D.A., Giuntoli B., Giorgi F.M., Voesenek L.A.C.J., Perata P., van Dongen J.T. Oxygen sensing in plants is mediated by an N-end rule pathway for protein destabilization. Nature. 2011;479:419–422. doi: 10.1038/nature10536. [DOI] [PubMed] [Google Scholar]
- 13.Weits D.A., Giuntoli B., Kosmacz M., Parlanti S., Hubberten H.M., Riegler H., Hoefgen R., Perata P., van Dongen J.T., Licausi F. Plant cysteine oxidases control the oxygen-dependent branch of the N-end-rule pathway. Nat. Commun. 2014;5:3425. doi: 10.1038/ncomms4425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gibbs D.J., Bacardit J., Bachmair A., Holdsworth M.J. The eukaryotic N-end rule pathway: conserved mechanisms and diverse functions. Trends Cell Biol. 2014;24:603–611. doi: 10.1016/j.tcb.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 15.Gibbs D.J., Md Isa N., Movahedi M., Lozano-Juste J., Mendiondo G.M., Berckhan S., Marín-de la Rosa N., Vicente Conde J., Sousa Correia C., Pearce S.P. Nitric oxide sensing in plants is mediated by proteolytic control of group VII ERF transcription factors. Mol. Cell. 2014;53:369–379. doi: 10.1016/j.molcel.2013.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hu R.G., Sheng J., Qi X., Xu Z., Takahashi T.T., Varshavsky A. The N-end rule pathway as a nitric oxide sensor controlling the levels of multiple regulators. Nature. 2005;437:981–986. doi: 10.1038/nature04027. [DOI] [PubMed] [Google Scholar]
- 17.Nakano T., Suzuki K., Fujimura T., Shinshi H. Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol. 2006;140:411–432. doi: 10.1104/pp.105.073783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Huq E., Al-Sady B., Hudson M., Kim C., Apel K., Quail P.H. Phytochrome-interacting factor 1 is a critical bHLH regulator of chlorophyll biosynthesis. Science. 2004;305:1937–1941. doi: 10.1126/science.1099728. [DOI] [PubMed] [Google Scholar]
- 19.Chow K.S., Singh D.P., Walker A.R., Smith A.G. Two different genes encode ferrochelatase in Arabidopsis: mapping, expression and subcellular targeting of the precursor proteins. Plant J. 1998;15:531–541. doi: 10.1046/j.1365-313x.1998.00235.x. [DOI] [PubMed] [Google Scholar]
- 20.Kaelin W.G., Jr., Ratcliffe P.J. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol. Cell. 2008;30:393–402. doi: 10.1016/j.molcel.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 21.Bailey-Serres J., Fukao T., Gibbs D.J., Holdsworth M.J., Lee S.C., Licausi F., Perata P., Voesenek L.A., van Dongen J.T. Making sense of low oxygen sensing. Trends Plant Sci. 2012;17:129–138. doi: 10.1016/j.tplants.2011.12.004. [DOI] [PubMed] [Google Scholar]
- 22.Miller D.L., Roth M.B. C. elegans are protected from lethal hypoxia by an embryonic diapause. Curr. Biol. 2009;19:1233–1237. doi: 10.1016/j.cub.2009.05.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bailey-Serres J., Voesenek L.A. Flooding stress: acclimations and genetic diversity. Annu. Rev. Plant Biol. 2008;59:313–339. doi: 10.1146/annurev.arplant.59.032607.092752. [DOI] [PubMed] [Google Scholar]
- 24.Zhong S., Shi H., Xue C., Wei N., Guo H., Deng X.W. Ethylene-orchestrated circuitry coordinates a seedling’s response to soil cover and etiolated growth. Proc. Natl. Acad. Sci. USA. 2014;111:3913–3920. doi: 10.1073/pnas.1402491111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kelliher T., Walbot V. Hypoxia triggers meiotic fate acquisition in maize. Science. 2012;337:345–348. doi: 10.1126/science.1220080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leivar P., Monte E. PIFs: systems integrators in plant development. Plant Cell. 2014;26:56–78. doi: 10.1105/tpc.113.120857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ni W., Xu S.L., Tepperman J.M., Stanley D.J., Maltby D.A., Gross J.D., Burlingame A.L., Wang Z.Y., Quail P.H. A mutually assured destruction mechanism attenuates light signaling in Arabidopsis. Science. 2014;344:1160–1164. doi: 10.1126/science.1250778. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.