Abstract
Purpose of Review:
Executive functions represent a constellation of cognitive abilities that drive goal-oriented behavior and are critical to the ability to adapt to an ever-changing world. This article provides a clinically oriented approach to classifying, localizing, diagnosing, and treating disorders of executive function, which are pervasive in clinical practice.
Recent Findings:
Executive functions can be split into four distinct components: working memory, inhibition, set shifting, and fluency. These components may be differentially affected in individual patients and act together to guide higher-order cognitive constructs such as planning and organization. Specific bedside and neuropsychological tests can be applied to evaluate components of executive function. While dysexecutive syndromes were first described in patients with frontal lesions, intact executive functioning relies on distributed neural networks that include not only the prefrontal cortex, but also the parietal cortex, basal ganglia, thalamus, and cerebellum. Executive dysfunction arises from injury to any of these regions, their white matter connections, or neurotransmitter systems. Dysexecutive symptoms therefore occur in most neurodegenerative diseases and in many other neurologic, psychiatric, and systemic illnesses. Management approaches are patient specific and should focus on treatment of the underlying cause in parallel with maximizing patient function and safety via occupational therapy and rehabilitation.
Summary:
Executive dysfunction is extremely common in patients with neurologic disorders. Diagnosis and treatment hinge on familiarity with the clinical components and neuroanatomic correlates of these complex, high-order cognitive processes.
INTRODUCTION
The term executive functions refers to a constellation of cognitive abilities that enable and drive adaptive, goal-oriented behavior. These include the ability to generate thought and think flexibly, to update and manipulate information mentally, to inhibit what is irrelevant to current goals, to self-monitor, and to plan and adjust behavior as appropriate to the present context.1 Intact executive functions are critical to the ability to adapt to an ever-changing world, and deficits in executive functioning lead to disproportionate impairment in function and activities of daily living.2 The cognitive construct of executive functions was originally described in the 1970s based on patterns of deficits observed in patients with frontal lobe lesions.3,4 Executive abilities evolve over childhood and adolescence, paralleling myelination and synaptogenesis of the frontal lobes5 and then decline with age in relation to loss of prefrontal function.6 Tasks requiring executive function activate distributed neural networks that prominently involve the prefrontal cortex, but also include the parietal cortex, basal ganglia, thalamus, and cerebellum.7–10 Executive functions are vulnerable to white matter injury11 and to perturbations in the cholinergic, noradrenergic, serotonergic, and dopaminergic neurotransmitter systems.12,13 As a result, executive functions are sensitive to a broad array of neurologic, psychiatric, and medical conditions. Disorders of executive function are pervasive in clinical practice, and it is critical for neurologists to be skillful in their assessment and treatment.
This article defines the related but dissociable components of executive functioning that can be variably affected in individual patients, introduces bedside and neuropsychological tests used in the assessment of these components and describes their proposed neuroanatomic correlates. This article provides a clinical approach to assessing patients with disorders of executive functions, illustrating the diversity of clinical presentations and the broad differential diagnosis, and concludes with a brief review of current approaches to therapy.
COMPONENTS OF EXECUTIVE FUNCTION
Since the initial description of a central executive by Baddeley and Hitch in 1974, considerable debate has occurred about whether executive function can be explained by a single mechanism or whether executive abilities are driven by distinct, although related, processes.3,14,15 From a clinical perspective, it is useful to split executive functions into specific components that can be differentially affected in individual patients. This article emphasizes four components that make distinct clinical contributions: information updating and monitoring (referred to in this article as working memory), inhibition of prepotent responses, mental set shifting, and fluency.14,15
Working Memory
Working memory is a limited capacity system that enables us to temporarily process, store, and manipulate information in conscious awareness. Examples from everyday life include rehearsing a phone number as we prepare to dial and registering a long sentence spoken in conversation as we process its meaning. Patients with working memory deficits may report absentmindedness and trouble focusing (Case 4-1). Intact working memory is critical to higher-level tasks such as planning and decision making as it allows us to actively keep track of all of the necessary information. An early model proposed by Baddeley and Hitch3 split working memory into a phonologic loop that maintains auditory and verbal information and a visuospatial sketchpad that maintains visual information. This model has gained credence from functional neuroimaging studies showing left-lateralized activations when performing verbal working memory tasks and right lateralized activity in response to visually oriented tasks.17
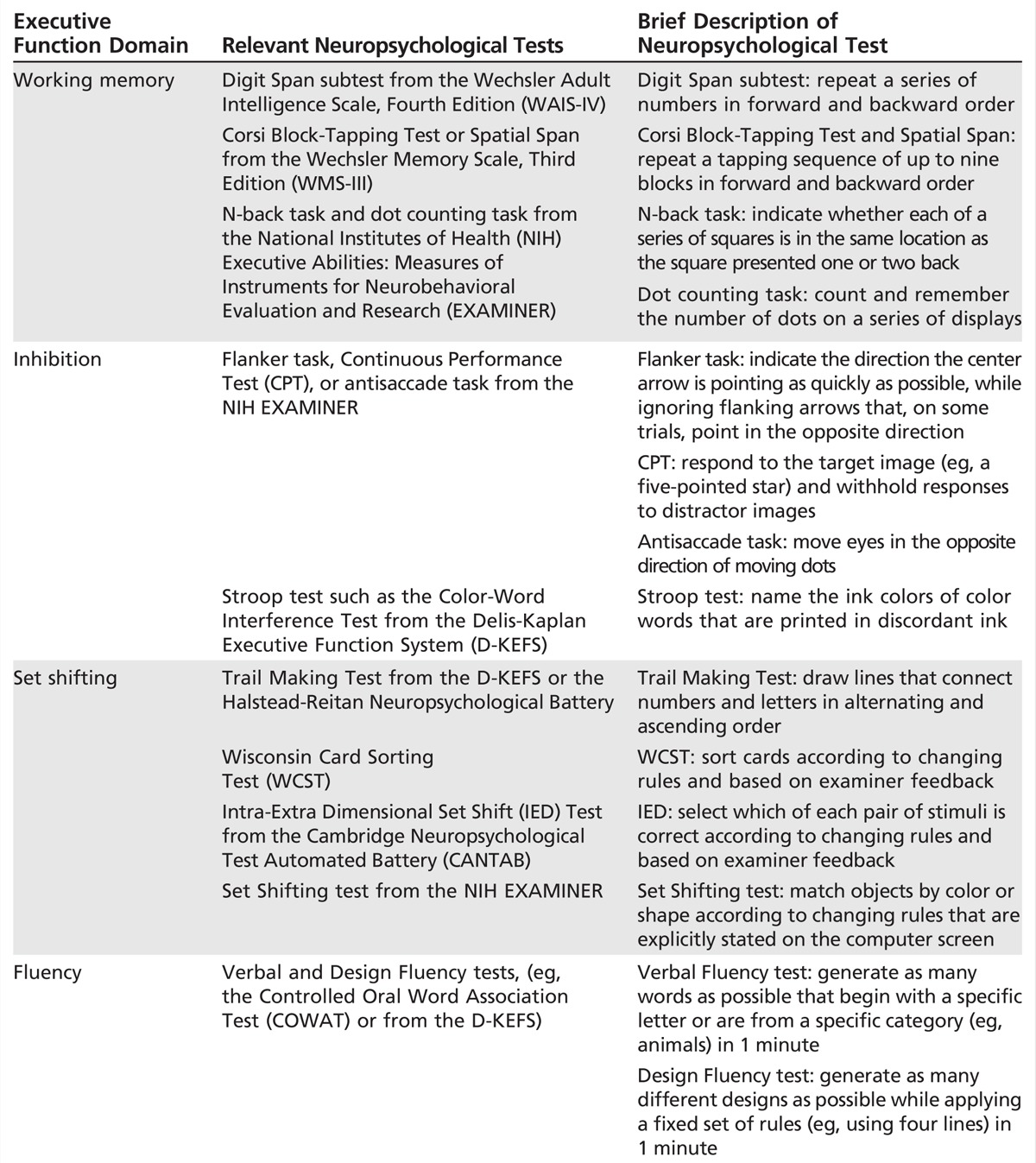
Working memory can be further divided into tasks that require simple maintenance of information (eg, forward digit span, which is a task that requires repeating a chain of numbers) and those that require active manipulation of information (eg, backward digit span, in which numbers are repeated in reverse order). Either spelling WORLD backward or performing serial 7s on the Mini-Mental State Examination (MMSE) are examples of working memory tests that require information manipulation. Additional examples of working memory paradigms, including tasks specific for visual information, are listed in Table 4-1.
Table 4-1.
Neuropsychological Tests of Executive Functions
Case 4-1
A 74-year-old retired college professor was self-referred for memory loss that he had been experiencing over the previous year. He retired 3 years ago when his wife fell ill with metastatic breast cancer, and he served as her primary caregiver until she passed away 9 months before this evaluation. He noted difficulty staying focused and concentrating. He found himself reading the same page in a book multiple times without retaining the information and has often lost track of his purpose when entering a room. The patient reported anhedonia, crying spells, insomnia, and weight loss. His past medical history was significant for an episode of major depression in his 30s. On mental status testing he was a good historian, but his affect was restricted. He became tearful when discussing his wife’s death. He scored 30/30 on the Montreal Cognitive Assessment (MoCA), and the remainder of his neurologic examination was normal. On formal neuropsychological testing, performance was generally in the above-average to superior range. However, he scored below average on backward digit span (repetition of numbers in reverse order) and letter fluency (generating as many words as possible beginning with a specific letter in 1 minute). He scored 18/30 on the Geriatric Depression Scale,17 a screening questionnaire in which scores over 10 raise concern for clinically significant depression. Brain MRI was normal. The patient was referred to a psychiatrist with a presumptive diagnosis of major depression and was started on a selective serotonin reuptake inhibitor (SSRI) and cognitive-behavioral therapy. In follow-up 6 months later, he reported that his mood and sleep were much improved. He started volunteering at a local museum. He felt that his cognition had improved, and repeat cognitive testing confirmed this improvement.
Comment. Executive dysfunction can accompany mood disorders as well as other psychiatric conditions. Patients may misrepresent their symptoms as related to memory, even though the primary problem is in attention and executive functioning. Screening for depression should be included as part of the neuropsychological evaluation, as illustrated here by use of the Geriatric Depression Scale. This patient was at high risk for depression given his wife’s illness and death. The diagnosis of major depression was made based on both mood and vegetative symptoms and signs. His functional deficits were out of proportion to his formal test scores, which is common in depression. Although his cognitive symptoms responded to treatment for depression, he should continue to be followed neurologically since late-life depression can represent a prodrome of a neurodegenerative condition.
Inhibition
Inhibition is the ability to hold back a predominant, automatic, or previously learned response that may be inappropriate or irrelevant in the present context. Certain stimuli are loaded to stimulate an automatic behavioral response due to either familiarity or immediate reward.18 However, adaptive behavior may require inhibition of the prepotent (ie, automatic or habitual) response in order to meet the current goal. Failure of cognitive inhibition should be distinguished from behavioral disinhibition, which, while related, suggests a distinct anatomy and differential diagnosis. For more information on this topic, refer to the article “Dysfunction of Social Cognition and Behavior” by Bradford C. Dickerson, MD, in this issue of CONTINUUM
Patients who are cognitively disinhibited have difficulty actively ignoring irrelevant or even penalizing stimuli. They may appear easily distracted, stimulus bound, and impulsive. In advanced cases, patients exhibit utilization behavior (picking up and using objects they observe for no clear purpose), echolalia (involuntarily repeating what is heard), or echopraxia (involuntarily imitating actions).
Cognitive inhibition can be tested at the bedside with go/no go tasks that require the patient to respond to a certain stimulus while withholding a response if presented with an alternative stimulus. For example, the examiner can instruct the patient to clap once if he or she claps twice, but not to clap at all if the examiner claps once.19 In the antisaccade task, the patient is instructed to look away from an engaging stimulus (eg, the examiner’s moving finger).20 Figure 4-1 illustrates tests of cognitive inhibition administered during neuropsychological testing. In the response inhibition portion of the Stroop test21 (Figure 4-1A), the subjects must inhibit the prepotent response to read words and, instead, name the color of ink that the word is printed in (the correct responses in the first row are blue, red, and green). In incongruent trials on the Flanker task, patients must indicate the direction the central arrow is pointed, ignoring the adjacent arrows that are pointed in the opposite direction (Figure 4-1B). Examples of additional tests are provided in Table 4-1.
Figure 4-1.
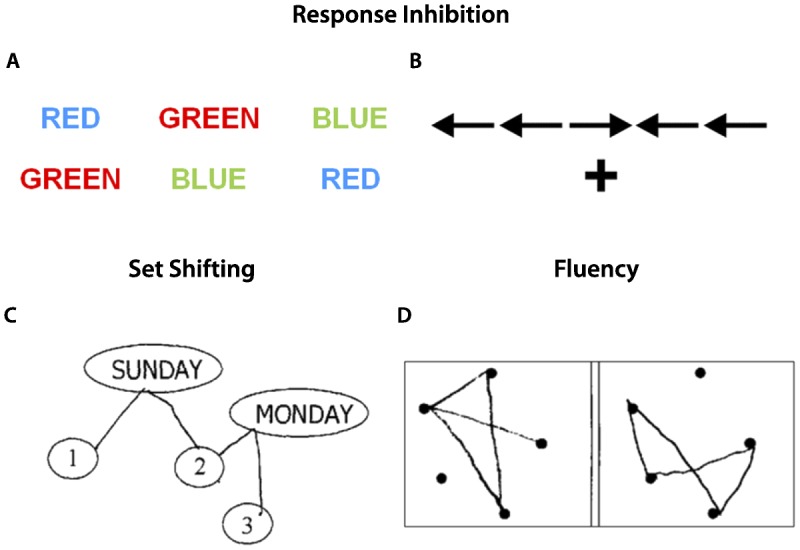
Example tasks to assess response inhibition, set shifting, and fluency. A, For the Stroop test, patients are asked to first read the words ignoring the colors (color naming), then name the colors ignoring the word (interference). B, For the Flanker task, patients are asked to identify the direction of the central arrow, with the adjacent arrows pointing either in the same direction (congruent) or opposite direction (incongruent). Stroop interference and incongruent trials on the Flanker task are considered tests of response inhibition. C, Trail making is a classic set shifting task in which patients write lines alternating in order between numbers and days of the week. D, Design fluency is an example of a nonverbal fluency task, in which patients are asked to draw as many unique designs as possible connecting the dots with four lines in 1 minute.
Set Shifting
Set shifting reflects the ability to modify attention and behavior in response to changing circumstances and demands. Set shifting inherently also relies on working memory (in order to keep in mind the current goals) and response inhibition (in order to ignore a previously relevant goal or focus of attention), illustrating the interdependence of different components of executive function. Patients with deficits in set shifting may report difficulties with multitasking and appear rigid in their thinking. On clinical evaluation, they may exhibit perseverative thoughts or behaviors. The Luria manual sequencing task, in which patients are asked to alternate sequentially between three hand positions (closed fist, fingers extended parallel to the ground, and fingers extended perpendicular to the ground), is a useful bedside test of set shifting. Trail-making tasks are often employed on neuropsychological testing. In the trail-making example shown in Figure 4-1C, patients are asked to draw alternating lines connecting numbers and days of the week in ascending order as rapidly and accurately as possible. The number of correct lines, time of completion, and number of shifting errors are recorded. Performance on shifting trials are often compared to sequencing trials without a shifting component (eg, connecting numbers only). Additional classic set shifting paradigms are listed in Table 4-1.
Fluency
Fluency represents the ability to maximize the production of verbal or visual information in a specific time period, while avoiding repeating responses. The three most common types of fluency tasks are category, letter, and design. For category fluency (also known as semantic fluency), subjects are asked to generate as many words as possible from a specified category (eg, animals or groceries). For letter fluency (also referred to as phonemic fluency), subjects are asked to generate as many words as possible that start with a specified letter, excluding names of people and places or grammatic variants of previous responses. For design fluency, subjects are asked to generate as many designs as possible while applying a fixed set of rules (eg, using four lines to connect the dots) (Figure 4-1D). Clinically, deficits on these measures may correlate with “tip-of-the-tongue” word retrieval deficits (in the absence of a true anomia), lack of initiation or inertia, or disorganization.
The components of executive function act in concert to enable planning and organization, which are higher-order cognitive constructs that allow an individual to identify, prioritize, and properly sequence the individual steps needed to achieve a goal in an efficient manner and adapt as needed to changes along the way.22 The functional consequences of executive dysfunction in daily life include ineffective planning and disorganization. On neuropsychological testing, planning can be evaluated via more complex tasks (eg, The Tower of London test, which requires moving colored beads across pegs to reproduce a target design in as few moves as possible).23
NEUROANATOMY OF EXECUTIVE FUNCTIONS
The concept of executive function first arose from deficits observed in patients with frontal lobe lesions. With the advent of functional neuroimaging, it has become clear that executive functioning relies on distributed neural networks that encompass the prefrontal cortex, but also engage the parietal cortex, basal ganglia, thalamus, and cerebellum. Overlapping activations in these regions are seen in paradigms that engage working memory, set shifting, response inhibition, fluency, and planning (Figure 4-224).7–10,25 Executive dysfunction can, therefore, arise from any neurologic process that involves these regions, their white matter connections, or neurotransmitter systems (Table 4-2). Executive functioning is also vulnerable to toxic-metabolic insults that lead to diffuse, bihemispheric dysfunction.
Figure 4-2.
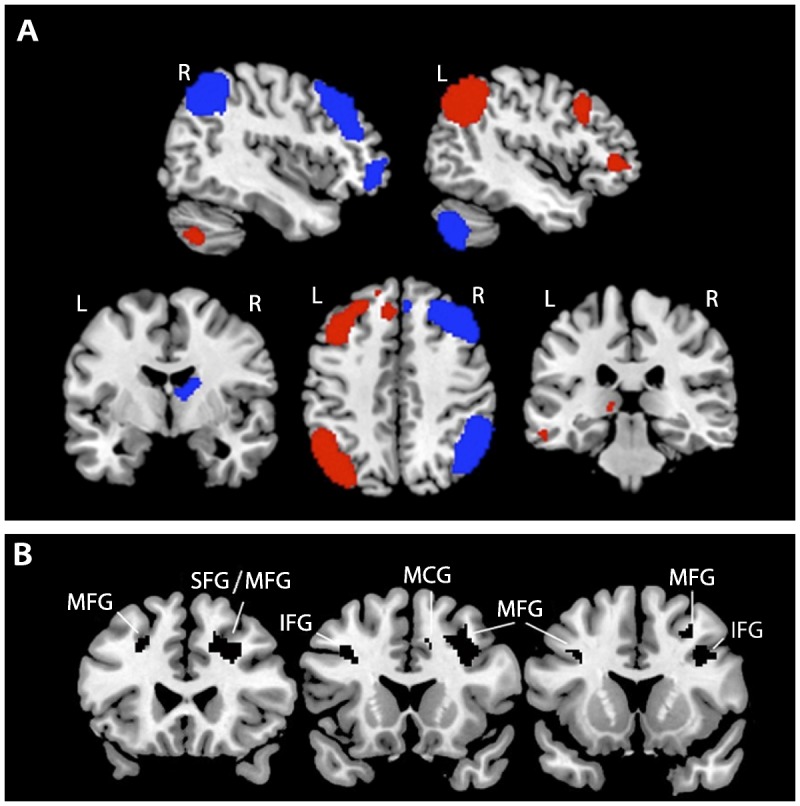
Neuroimaging correlates of executive functions. A, Left (red) and right (blue) hemisphere functional networks that are activated on functional MRI during executive control tasks are shown on a template brain in neurologic orientation. B, Gray matter correlates of a composite executive function score derived from the Executive Abilities: Measures and Instruments for Neurobehavioral Evaluation and Research (EXAMINER) battery identified via voxel-based morphometry. Black clusters represent brain regions in which, across subjects, higher gray matter volumes were correlated with higher composite executive function scores. All results are thresholded at P<.001 and corrected for multiple comparisons using permutation analysis at P<.05.
R = right; L = left; MFG = middle frontal gyrus; SFG = superior frontal gyrus; IFG = inferior frontal gyrus; MCG = middle cingulate gyrus. Panel A network templates courtesy of Michael Greicius, MD, and the Functional Imaging in Neuropsychiatric Disorders Laboratory, Stanford University. Panel B is reprinted with permission from Possin KL, et al, J Int Neuropsychol Soc.24 © 2013 The International Neuropsychological Society. journals.cambridge.org/action/displayAbstract?fromPage=online&aid=9135793&fileId=S1355617713000611.
Table 4-2.
Differential Diagnosis of Executive Dysfunction

The nature of executive deficits can assist with more precise localization. The dorsolateral prefrontal cortex is engaged during set shifting, planning, and working memory.26 The right prefrontal cortex is specialized for self-monitoring and spatial tasks, whereas left hemisphere regions are engaged in verbal processing. For example, greater deficits in verbal versus design fluency indicate greater left frontal dysfunction,27 whereas greater impairment in design fluency indicates right frontal or parietal injury.28 On working memory tasks, the ventrolateral prefrontal cortex is active during retrieval and maintenance of information, while dorsolateral prefrontal regions are activated when either active manipulation or updating of information is required.10 The pre–supplementary motor area is engaged during response selection, whereas the anterior cingulate cortex plays a critical role in error detection.29,30 Inferior frontal regions (ie, ventrolateral prefrontal and orbitofrontal cortex) may be particularly critical for assessing shifting reward-punishment contingencies and inhibiting inappropriate responses.31,32 Subcortical structures support executive functions via their roles in cortico-basal ganglia–thalamocortical circuits.33 The network engaged in executive control is distinct from the salience network, which is a network composed of frontal insula, anterior cingulate, and ventromedial prefrontal cortex, with connections to limbic and subcortical structures.34 The salience network mediates decision making related to social and emotional as well as autonomic and interoceptive processing (ie, monitoring the internal state as reflected by heart and respiratory rates or homeostatic needs such as hunger and thirst) and is targeted early and specifically in behavioral variant frontotemporal dementia. In contrast, involvement of executive control regions of the prefrontal cortex is characteristic of many neurodegenerative disorders and is not specific for frontotemporal dementia (Case 4-2). For more information on this topic, refer to the article “Dysfunction of Social Cognition and Behavior” by Bradford C. Dickerson, MD, in this issue of CONTINUUM.
Case 4-2
A 54-year-old woman sought evaluation after experiencing 2 years of progressive cognitive difficulties. Her symptoms were first apparent during her work as a high-level executive. She reported difficulty comprehending complex written material. She made a number of impulsive decisions with poor outcomes, and she subsequently felt “paralyzed” when faced with multifaceted problems. She lost her job 1 year ago and has been unable to “get things together” to apply for a new position. Her husband, interviewed separately, noted no inappropriate behavior, loss of empathy, stereotyped behaviors, or dietary changes. Her elemental neurologic examination was normal. She scored 28/30 on the Mini-Mental State Examination (MMSE), but on formal neuropsychologic testing she showed moderate impairment on all tests of executive function (including digit span forward and backward, a modified Trail Making Test, lexical fluency, semantic fluency, design fluency, and Stroop inhibition), with milder deficits noted in naming and figure copying. On memory testing she showed mild to moderate impairment in encoding and spontaneous retrieval but intact recognition. Fluorodeoxyglucose positron emission tomography (FDG-PET) showed marked hypometabolism in bilateral temporoparietal, dorsolateral, and dorsomedial prefrontal cortex (Figure 4-3). CSF biomarker studies revealed low amyloid-β 42 and high total and phosphorylated tau levels. The patient was diagnosed with probable Alzheimer disease dementia, started on a cholinesterase inhibitor, and referred to a social worker to assist with future planning.
Figure 4-3.
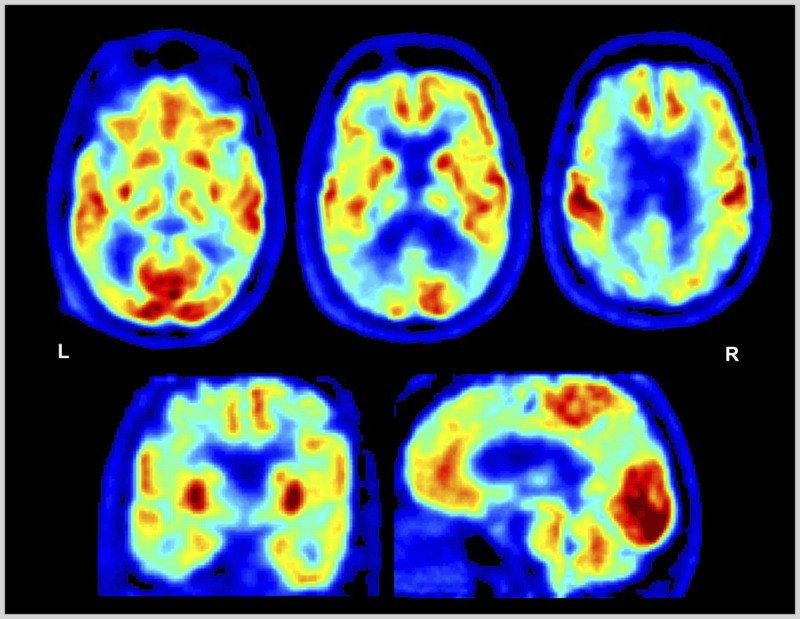
Fluorodeoxyglucose positron emission tomography (FDG-PET) of the patient in Case 4-2. Images displayed in the National Institutes of Health color scale in neurologic orientation. Hypometabolism is noted in bilateral temporoparietal and dorsal prefrontal cortex. Temporoparietal hypometabolism is the FDG pattern associated with Alzheimer disease, while additional involvement of the prefrontal cortex may explain the prominent executive dysfunction experienced by the patient.
L = left; R = right.
Comment. While Alzheimer disease most commonly presents with episodic memory loss, patients can present with primary deficits in executive functions, and this is particularly true of patients with an early age of onset (under age 65). Executive dysfunction can also be the presenting cognitive deficit in vascular dementia, Lewy body diseases, and frontotemporal dementia, among other neurodegenerative conditions (Table 4-2). In this patient, preservation of emotional and social function (prominent early features in frontotemporal dementia) and a normal motor examination pointed to Alzheimer disease as the likely diagnosis. FDG-PET and CSF biomarkers confirmed the diagnosis. This case also highlights the poor sensitivity of the MMSE to executive dysfunction and the importance of obtaining neuropsychological testing to better characterize the cognitive correlates of this individual’s devastating functional disability.
CLINICAL APPROACH TO PATIENTS WITH EXECUTIVE DYSFUNCTION
The clinical evaluation of patients with executive dysfunction should follow practice parameters for the assessment of cognitive impairment, beginning with a comprehensive history and neurologic examination.35 Patients are unlikely to present a chief complaint of executive dysfunction and may misrepresent their cognitive problem as memory loss. Specific questions that capture executive deficits include inquiries about the patient’s difficulty with planning or organization, problems with multitasking, poor judgment or decisions, impaired concentration/short attention span, difficulty with problem solving, mental rigidity/inflexibility, and impulsivity. When testing global cognitive function, the Montreal Cognitive Assessment (MoCA) (www.mocatest.org) is more sensitive than the MMSE for detecting executive dysfunction (Case 4-3).36 Bedside testing and neuropsychological batteries should test all of the executive components described above. For individual tests and reference to commonly used batteries, see Table 4-1. The Executive Abilities: Measures and Instruments for Neurobehavioral Evaluation and Research (EXAMINER) (examiner.ucsf.edu) was designed as an efficient (30-minute) battery to test the spectrum of executive functions across a broad range of ages and disorders in clinical research and therapeutic trials.37 The composite EXAMINER score correlates with informant-based measures of real-world executive functioning and with gray matter volumes in the prefrontal cortex (Figure 4-2).24
A differential diagnosis for executive dysfunction is shown in Table 4-2, although a completely comprehensive list cannot be provided given that executive dysfunction can accompany most processes that impact the brain. To address the broad differential diagnosis, the clinician must characterize the relative involvement of other cognitive domains, motor and psychiatric symptoms and signs, comorbid neuropsychiatric and medical conditions, and medication use. A laboratory evaluation for systemic disorders and structural brain imaging is needed to assess for treatable conditions (Case 4-3). In selected cases, CSF or imaging biomarkers for Alzheimer disease may be helpful (Case 4-2).
Case 4-3
A 62-year-old woman presented with progressive cognitive decline. She worked as a nursing assistant for a home health company, and over the past year, she had been noted to be increasingly inefficient with poor prioritization of tasks. She had recently been placed on personal leave after she had missed a number of appointments and on one occasion attempted to administer a treatment to the wrong patient. She acknowledged being easily distracted and having problems staying on task. She also noted frequent headaches. On mental status testing, she scored 21/30 on the Montreal Cognitive Assessment (MoCA), losing points on trail making, backward digit span, vigilance, abstract reasoning, and delayed recall (the latter improving with cuing). Physical examination revealed a subtle left hemiparesis with left-side hyperreflexia and a mute plantar response. Brain MRI revealed a dural-based enhancing extraaxial mass along the right anterior clinoid process, suggestive of a meningioma, with displacement of the right inferior frontal cortex, and surrounding vasogenic edema (Figure 4-4). She was referred for neurosurgical consultation.
Figure 4-4.
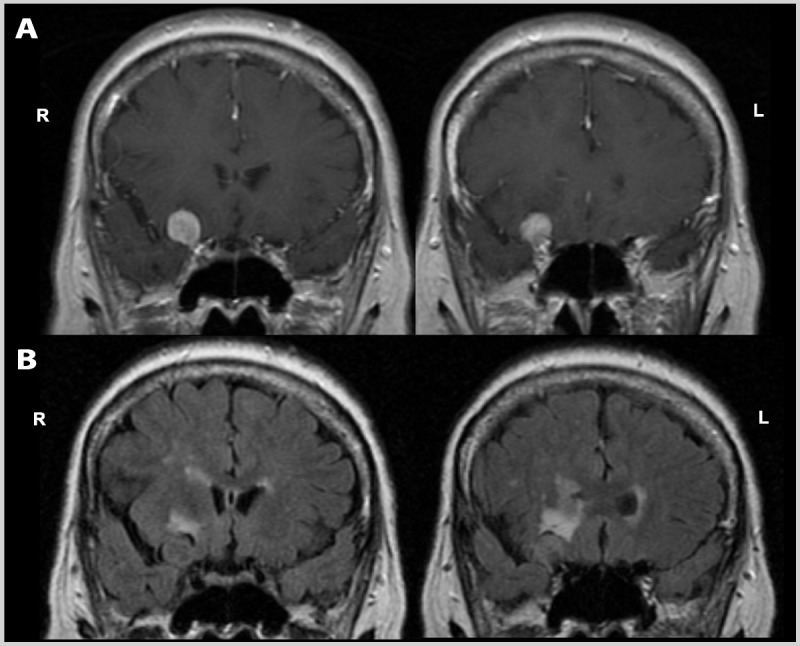
Brain MRI of the patient in Case 4-3. Coronal postcontrast T1-weighted (A) and noncontrast fluid-attenuated inversion recovery (FLAIR) (B) images show a dural-based enhancing extraaxial mass along the right anterior clinoid process, suggestive of a meningioma, with displacement of the right inferior frontal cortex, and surrounding vasogenic edema. R = right; L = left.
Comment. This patient presented with dysexecutive symptoms, including difficulty with planning, organization, multitasking, and attention. The neurologist appropriately used the MoCA to screen global cognitive function, as this test includes several items that directly test executive functions. The subacute presentation, presence of headaches, and focal motor findings increased suspicion for an underlying mass lesion, which was confirmed on MRI. The lesion and associated edema were within executive control networks in the right hemisphere and, thus, were very likely to be responsible for the patient’s clinical presentation.
TREATMENT
Treatment of dysexecutive disorders needs to be tailored to the individual patient. The first step is to identify and treat the primary underlying condition. For example, performance on executive function tests responds to cholinesterase inhibitors in Alzheimer disease,38 whereas patients with Parkinson disease perform better on dopamine replacement therapy.39 An evaluation by occupational therapy can help clarify the real-world functional implications in order to ensure patient safety and maximize function.40 Cognitive rehabilitation strategies include environmental manipulation (eg, minimizing distractors and simplifying tasks), compensatory techniques (eg, increased use of daily planners or smartphones), and direct interventions (eg, repetitive training to improve a skill).41 Experimental approaches include dopamine agonist therapy in patients without known dopamine deficiency (eg, posttraumatic brain injury and stroke),42 computer-based brain exercises,43 and transcranial magnetic stimulation.44
CONCLUSION
Intact executive functions are critical for adaptive behavior and day-to-day function and rely on widespread frontal, parietal, and subcortical brain networks. Executive functions are vulnerable to a broad array of neurologic, psychiatric, and medical processes, including many reversible or treatable conditions. Familiarity with the components of executive functions, underlying neuroanatomy, and differential diagnosis can help guide the clinician toward an accurate diagnosis and optimal treatment plan.
KEY POINTS
Executive functions include a constellation of cognitive abilities that drive goal-oriented behavior and allow the individual to adapt to an ever-changing world. The concept of executive functions first arose from deficits seen in patients with frontal lesions.
Executive functions can be split into four distinct components that can be differentially affected in individual patients: working memory, response inhibition, set shifting, and fluency.
Working memory enables us to temporarily process, store, and manipulate information in conscious awareness, an example of which is the rehearsal of a phone number as we prepare to dial. Patients with working memory deficits may report absentmindedness or trouble focusing.
Inhibition represents the ability to hold back a predominant, automatic, or previously learned response, which may be inappropriate or irrelevant in the present context. Patients with deficits in cognitive inhibition may appear stimulus bound or exhibit utilization behavior.
Set shifting reflects the ability to modify attention and behavior in response to changing circumstances and demands. Patients with deficits exhibit perseverative behavior and rigid thinking.
Fluency is the ability to maximize the production of verbal or visual information in a specific time period, examples of which include category fluency (eg, animals), letter fluency (eg, “D” words), and design fluency.
The components of executive function act together to enable planning and organization, which are higher-order cognitive constructs that allow an individual to identify, prioritize, and properly sequence the individual steps needed to achieve a goal in an adaptive and efficient manner.
Executive functioning relies on distributed neural networks that include the prefrontal cortex, parietal cortex, basal ganglia, thalamus, and cerebellum. Executive dysfunction arises from injury to any of these regions, their white matter connections, or neurotransmitter systems.
Executive control networks are impacted by many neurodegenerative diseases, whereas the salience network, which mediates social and emotional behavior, is specifically targeted in frontotemporal dementia.
When probing about executive functions, clinicians should ask about the patient’s difficulty with planning or organization, problems with multitasking, poor judgment or decisions, impaired concentration/short attention span, difficulty with problem solving, mental rigidity/inflexibility, and impulsivity.
The Montreal Cognitive Assessment is more sensitive to deficits in executive function than the Mini-Mental State Examination.
Treatment of dysexecutive disorders needs to be tailored to the individual patient, often requiring a combination of disease-specific pharmacologic treatments, occupational therapy, and cognitive rehabilitation.
REFERENCES
- 1. Jurado MB, Rosselli M. The elusive nature of executive functions: a review of our current understanding. Neuropsychol Rev 2007; 17 (3): 213– 233. doi:10.1007/s11065-007-9040-z. [DOI] [PubMed] [Google Scholar]
- 2. Cahn-Weiner DA, Boyle PA, Malloy PF. Tests of executive function predict instrumental activities of daily living in community-dwelling older individuals. Appl Neuropsychol 2002; 9 (3): 187– 191. doi:10.1207/S15324826AN0903_8. [DOI] [PubMed] [Google Scholar]
- 3. Baddeley AD, Hitch GJ. Working memory. In: Bower G, ed. Recent advances in learning and motivation. London: Academic Press, 1974: 47– 90. [Google Scholar]
- 4. Stuss DT, Benson DF. The frontal lobes. New York: Raven Press, 1986. [Google Scholar]
- 5. Anderson VA, Anderson P, Northam E, et al. Development of executive functions through late childhood and adolescence in an Australian sample. Dev Neuropsychol 2001; 20 (1): 385– 406. doi:10.1207/S15326942DN2001_5. [DOI] [PubMed] [Google Scholar]
- 6. Buckner RL. Memory and executive function in aging and AD: multiple factors that cause decline and reserve factors that compensate. Neuron 2004; 44 (1): 195– 208. doi:10.1016/j.neuron.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 7. Collette F, Van der Linden M, Laureys S, et al. Exploring the unity and diversity of the neural substrates of executive functioning. Hum Brain Mapp 2005; 25 (4): 409– 423. doi:10.1002/hbm.20118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Monchi O, Petrides M, Strafella AP, et al. Functional role of the basal ganglia in the planning and execution of actions. Ann Neurol 2006; 59 (2): 257– 264. doi:10.1002/ana.20742. [DOI] [PubMed] [Google Scholar]
- 9. Wager TD, Jonides J, Reading S. Neuroimaging studies of shifting attention: a meta-analysis. Neuroimage 2004; 22 (4): 1679– 1693. doi:10.1016/j.neuroimage.2004.03.052. [DOI] [PubMed] [Google Scholar]
- 10. Wager TD, Smith EE. Neuroimaging studies of working memory: a meta-analysis. Cogn Affect Behav Neurosci 2003; 3 (4): 255– 274. doi:10.3758/CABN.3.4.255. [DOI] [PubMed] [Google Scholar]
- 11. Kerchner GA, Racine CA, Hale S, et al. Cognitive processing speed in older adults: relationship with white matter integrity. PLoS One 2012; 7 (11): e50425 doi:10.1371/journal.pone.0050425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Barnes JJ, Dean AJ, Nandam LS, et al. The molecular genetics of executive function: role of monoamine system genes. Biol Psychiatry 2011; 69 (12): e127– e143. doi:10.1371/journal.pone.0050425. [DOI] [PubMed] [Google Scholar]
- 13. Robbins TW, Roberts AC. Differential regulation of fronto-executive function by the monoamines and acetylcholine. Cereb Cortex 2007; 17 (suppl 1): i151– i160. doi:10.1093/cercor/bhm066. [DOI] [PubMed] [Google Scholar]
- 14. Miyake A, Friedman NP, Emerson MJ, et al. The unity and diversity of executive functions and their contributions to complex “Frontal Lobe” tasks: a latent variable analysis. Cogn Psychol 2000; 41 (1): 49– 100. doi:10.1006/cogp.1999.0734. [DOI] [PubMed] [Google Scholar]
- 15. Fisk JE, Sharp CA. Age-related impairment in executive functioning: updating, inhibition, shifting, and access. J Clin Exp Neuropsychol 2004; 26 (7): 874– 890. doi:10.1080/13803390490510680. [DOI] [PubMed] [Google Scholar]
- 16. Yesavage JA, Brink TL, Rolse TL, et al. Development and validity of a geriatric depression scale: a preliminary report. J Psychiatr Res 1982–1983; 17 (1): 37– 49. [DOI] [PubMed] [Google Scholar]
- 17. Smith EE, Jonides J, Koeppe RA. Dissociating verbal and spatial working memory using PET. Cereb Cortex 1996; 6 (1): 11– 20. doi:10.1093/cercor/6.1.11. [DOI] [PubMed] [Google Scholar]
- 18. Friedman NP, Miyake A. The relations among inhibition and interference control functions: a latent-variable analysis. J Exp Psychol Gen 2004; 133 (1): 101– 135. doi:10.1037/0096-3445.133.1.101. [DOI] [PubMed] [Google Scholar]
- 19. Luria AR. Human brain and psychological processes. New York: Harper Row, 1966. [Google Scholar]
- 20. Hellmuth J, Mirsky J, Heuer HW, et al. Multicenter validation of a bedside antisaccade task as a measure of executive function. Neurology 2012; 78 (23): 1824– 1831. doi:10.1212/WNL.0b013e318258f785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Golden C. Stroop color and word test: manual for clinical and experimental uses. Chicago, IL: Stoelting, 1978. [Google Scholar]
- 22. Grafman J. Planning and the brain. In: Miller BL, Cummings JL, editors. The human frontal lobes: functions and disorders. 2nd ed New York: The Guilford Press, 2007: 249– 261. [Google Scholar]
- 23. Shallice T. Specific impairments of planning. Philos Trans R Soc Lond B Biol Sci 1982; 298 (1089): 199– 209. [DOI] [PubMed] [Google Scholar]
- 24. Possin KL, LaMarre AK, Wood KA, et al. Ecological validity and neuroanatomical correlates of the NIH EXAMINER executive composite score. J Int Neuropsychol Soc 2014; 20 (1): 20– 28. doi:10.1017/S1355617713000611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Criaud M, Boulinguez P. Have we been asking the right questions when assessing response inhibition in go/no-go tasks with fMRI? A meta-analysis and critical review. Neurosci Biobehav Rev 2013; 37 (1): 11– 23. doi:10.1016/j.neubiorev.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 26. Stuss DT, Alexander MP, Floden D, et al. Fractionation and localization of distinct frontal lobe processes: evidence from focal lesions in humans. In: Stuss DT, Knight RT, eds. Principles of frontal lobe function. New York: Oxford University Press, 2002: 392– 407. [Google Scholar]
- 27. Baldo JV, Shimamura AP, Delis DC, et al. Verbal and design fluency in patients with frontal lobe lesions. J Int Neuropsychol Soc 2001; 7 (5): 586– 596. [DOI] [PubMed] [Google Scholar]
- 28. Possin KL, Chester SK, Laluz V, et al. The frontal-anatomic specificity of design fluency repetitions and their diagnostic relevance for behavioral variant frontotemporal dementia. J Int Neuropsychol Soc 2012; 18 (5): 834– 844. doi:10.1017/S1355617712000604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lau H, Rogers RD, Passingham RE. Dissociating response selection and conflict in the medial frontal surface. Neuroimage 2006; 29 (2): 446– 451. doi:10.1016/j.neuroimage.2005.07.050. [DOI] [PubMed] [Google Scholar]
- 30. Devinsky O, Morrell MJ, Vogt BA. Contributions of anterior cingulate cortex to behaviour. Brain 1995; 118 (pt 1): 279– 306. doi:10.1093/brain/118.1.279. [DOI] [PubMed] [Google Scholar]
- 31. Rolls ET. The functions of the orbitofrontal cortex. Brain Cogn 2004; 55 (1): 11– 29. doi:10.1016/S0278-2626(03)00277-X. [DOI] [PubMed] [Google Scholar]
- 32. Levy BJ, Wagner AD. Cognitive control and right ventrolateral prefrontal cortex: reflexive reorienting, motor inhibition, and action updating. Ann N Y Acad Sci 2011; 1224: 40– 62. doi:10.1111/j.1749-6632.2011.05958.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Schmahmann JD, Pandya DN. Disconnection syndromes of basal ganglia, thalamus, and cerebrocerebellar systems. Cortex 2008; 44 (8): 1037– 1066. doi:10.1016/j.cortex.2008.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Seeley WW, Menon V, Schatzberg AF, et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 2007; 27 (9): 2349– 2356. doi:10.1523/JNEUROSCI.5587-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Knopman DS, DeKosky ST, Cummings JL, et al. Practice parameter: diagnosis of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2001; 56 (9): 1143– 1153. doi:10.1212/WNL.56.9.1143. [DOI] [PubMed] [Google Scholar]
- 36. Lam B, Middleton LE, Masellis M, et al. Criterion and convergent validity of the Montreal cognitive assessment with screening and standardized neuropsychological testing. J Am Geriatr Soc 2013; 61 (12): 2181– 2185. doi:10.1111/jgs.12541. [DOI] [PubMed] [Google Scholar]
- 37. Kramer JH, Mungas D, Possin KL, et al. NIH EXAMINER: conceptualization and development of an executive function battery. J Int Neuropsychol Soc 2014; 20 (1): 11– 19. doi:10.1017/S1355617713001094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Behl P, Lanctot KL, Streiner DL, et al. Cholinesterase inhibitors slow decline in executive functions, rather than memory, in Alzheimer’s disease: a 1-year observational study in the Sunnybrook dementia cohort. Curr Alzheimer Res 2006; 3 (2): 147– 156. doi:10.2174/156720506776383031. [DOI] [PubMed] [Google Scholar]
- 39. Oswal A, Litvak V, Sauleau P, Brown P. Beta reactivity, prospective facilitation of executive processing, and its dependence on dopaminergic therapy in Parkinson’s disease. J Neurosci 2012; 32 (29): 9909– 9916. doi:10.1523/JNEUROSCI.0275-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Cramm HA, Krupa TM, Missiuna CA, et al. Executive functioning: a scoping review of the occupational therapy literature. Can J Occup Ther 2013; 80 (3): 131– 140. [DOI] [PubMed] [Google Scholar]
- 41. D’Esposito M, Gazzaley A. Neurorehabilitation of executive function. In: Textbook of neural repair and rehabilitation. Cambridge: Cambridge University Press, 2005: 475– 486. [Google Scholar]
- 42. McDowell S, Whyte J, D’Esposito M. Differential effect of a dopaminergic agonist on prefrontal function in traumatic brain injury patients. Brain 1998; 121 (pt 6): 1155– 1164. doi:10.1093/brain/121/6/1155. [DOI] [PubMed] [Google Scholar]
- 43. Anguera JA, Boccanfuso J, Rintoul JL, et al. Video game training enhances cognitive control in older adults. Nature 2013; 501 (7465): 97– 101. doi:10.1038/nature12486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Luber B, Lisanby SH. Enhancement of human cognitive performance using transcranial magnetic stimulation (TMS). Neuroimage 2014; 85 (pt 3): 961– 970. doi:10.1016/j.neuroimage.2013.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]