Abstract
We evaluated the performance of Time to Clinical Stability (TCS), a longitudinal outcome measure using four physiologic parameters (temperature, heart rate, respiratory rate, and use of supplemental oxygen), among children enrolled in a prospective study of pneumonia hospitalizations. We calculated the time from admission to normalization for each of the four parameters individually and various combinations of these parameters (>2 parameters). We assessed for agreement between the combined TCS measures and both hospital length of stay and an ordinal severity scale (non-severe, severe, and very severe). Overall, 323 (96.7%) of 334 included children had ≥1 parameter abnormal on admission; 70 (21%) children had ≥1 parameter abnormal at discharge. For the four combined measures, median TCS decreased with increasing age. Increasing TCS was associated with both longer length of stay and increasing disease severity. The simplest combined measure incorporating only respiratory rate and need for supplemental oxygen performed similarly to more complex measures including additional parameters. Our study demonstrates that longitudinal TCS measures may be useful in children with pneumonia, both in clinical settings to assess recovery and readiness for discharge, and as an outcome measure in research and quality assessments. Additional study is needed to further validate our findings.
Introduction
National guidelines for the management of childhood pneumonia highlight the need for the development of objective outcome measures to inform clinical decision making, establish benchmarks of care, and compare treatments and interventions.1 Time to Clinical Stability (TCS) is a measure reported in adult pneumonia studies that incorporates vital signs, ability to eat, and mental status to objectively assess readiness for discharge.2–4 TCS has not been validated among children as it has in adults5–8, although such measures could prove useful for assessing discharge readiness with applications in both clinical and research settings. The objective of our study was to test the performance of pediatric TCS measures among children hospitalized with pneumonia.
Methods
Study Population
We studied children hospitalized with community-acquired pneumonia at Monroe Carell Jr. Children's Hospital at Vanderbilt between January 6, 2010 and May 9, 2011. Study children were enrolled as part of the Centers for Disease Control (CDC) Etiology of Pneumonia in the Community (EPIC) study, a prospective, population-based study of community-acquired pneumonia hospitalizations. Detailed enrollment criteria for the EPIC study were reported previously.9 Institutional Review Boards at Vanderbilt University and CDC approved this study. Informed consent was obtained from enrolled families.
Data Elements and Study Definitions
Baseline data, including demographics, illness history, co-morbidities, and clinical outcomes (e.g. length of stay (LOS), intensive care admission), were systematically and prospectively collected. Additionally, data for four physiologic parameters, including temperature, heart rate, respiratory rate, and use of supplemental oxygen were obtained from the electronic medical record. These parameters were measured at least every 6 hours from admission through discharge as part of routine care. Readmissions within 7 calendar days of discharge were also obtained from the electronic medical record.
Stability for each parameter was defined as follows: normal temperature (36.0-37.9 degrees Celsius), normal respiratory and heart rates in accordance with Pediatric Advanced Life Support age-based values (supplemental table 1)10 and no administration of supplemental oxygen. If the last recorded value for a given parameter was abnormal, that parameter was considered unstable at discharge. Otherwise, the time and date of the last abnormal value for each parameter was subtracted from admission time and date to determine TCS for that parameter in hours.
To determine overall stability, we evaluated four combination TCS measures, each incorporating ≥2 individual parameters. All combinations included respiratory rate and need for supplemental oxygen, as these parameters are the most explicit clinical indicators of pneumonia. Stability for each combination measure was defined as normalization of all included measures.
Clinical Outcomes for the Combined TCS Measures
The four combined TCS measures were compared against clinical outcomes including hospital LOS (measured in hours) and an ordinal severity scale. The ordinal scale categorized children into three mutually exclusive groups as follows: non-severe (hospitalization without need for intensive care or empyema requiring drainage), severe (intensive care admission without invasive mechanical ventilation or vasopressor support and no empyema requiring drainage), and very severe (invasive mechanical ventilation, vasopressor support, or empyema requiring drainage).
Statistical Analysis
Categorical and continuous variables were summarized using frequencies and percentages and median and interquartile range (IQR) values, respectively. Analyses were stratified by age (<2 years, 2-4 years, 5-17 years). We also plotted summary statistics for the combined measures and LOS, and computed the median absolute difference between these measures for each level of the ordinal severity scale. Analyses were conducted using Stata 13 (College Station, TX).
Results
Study Population
Among 336 children enrolled in the EPIC study at Vanderbilt during the study period, 334 (99.4%) with complete data were included. Median age was 33 months (IQR 14, 80). Median LOS was 56.4 hours (IQR 41.5, 91.7). There were 249 (74.5%) children classified as non-severe, 39 (11.7) as severe and 46 (13.8) as very severe (age-based characteristics appear in supplemental table 2). Overall, 12 (3.6%) children were readmitted within 7 days of discharge.
Individual Stability Parameters
Overall, 323 (96.7%) children had ≥1 parameter abnormal on admission. Respiratory rate (81.4%) was the most common abnormal parameter, followed by abnormal temperature (71.4%), use of supplemental oxygen (63.8%), and abnormal heart rate (54.4%). Overall, use of supplemental oxygen had the longest TCS, followed by respiratory rate (Table 1A). In comparison, heart rate and temperature stabilized relatively quickly.
Table 1A.
Time to Stability for Four Physiologic Parameters in Children Hospitalized with Community-acquired Pneumonia
| <2 years (n=130) | 2-4 years (n=90) | 5-17 years (n=101) | ||||
|---|---|---|---|---|---|---|
| Parameter | No. (%)1 | Median (IQR) TCS, in hours | No. (%) | Median (IQR) TCS, in hours | No. (%) | Median (IQR) TCS, in hours |
| Respiratory rate (RR) | 97 (74.6) | 38.6 (18.7, 68.9) | 63 (70.0) | 31.6 (9.5, 61.9) | 63 (62.4) | 24.3 (10.8, 59.2) |
| Oxygen (O2) | 90 (69.2) | 39.5 (19.2, 73.6) | 58 (64.4) | 44.2 (24, 77.6) | 61 (60.4) | 38.3 (18, 70.6) |
| Heart rate (HR) | 21 (16.2) | 4.52 (0.3, 18.4) | 73 (81.1) | 21.8 (5.7, 51.9) | 62 (61.4) | 18 (5.8, 42.2) |
| Temperature (T) | 101 (77.7) | 14.5 (4.5, 45.3) | 61 (67.8) | 18.4 (2.8, 42.8) | 62 (61.4) | 10.6 (0.8, 34) |
For each parameter, time to clinical stability (TCS) was calculated by subtracting the time and date of the last abnormal value for that parameter from admission time and date to determine time to stability in hours; children stable on admission for all four parameters not included (n=11)
Number (%) of children who reached stability more than six hours prior to discharge
Likely influenced by the wide upper range of this parameter for children <2 years (84% of children in this age group classified as stable on admission for heart rate)
abbreviations: TCS, time to clinical stability; IQR, interquartile range; T, temperature; HR, heart rate; RR, respiratory rate; O2, supplemental oxygen
Seventy children (21.0%) had ≥1 parameter abnormal at discharge, including abnormal respiratory rate in 13.7%, heart rate in 7.0%, and temperature in 3.3%. One child (0.3%) was discharged with supplemental oxygen. Ten children (3.0%) had ≥2 parameters abnormal at discharge. There was no difference in 7 day readmissions for children with ≥1 parameter abnormal at discharge (1.4%) compared to those with no abnormal parameters at discharge (4.4%, p=.253).
Combination TCS Measures
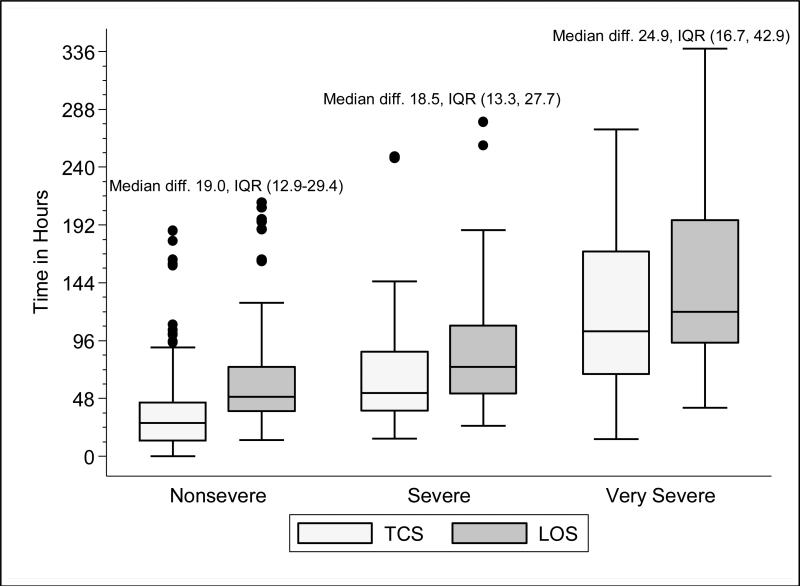
Within each age group, the percentage of children achieving stability was relatively consistent across the four combined TCS measures (Table 1B); however, more children were considered unstable at discharge (and fewer classified as stable on admission) as the number of included parameters increased. More children <5 years of age reached stability (range 80.0-85.6%) compared to children ≥5 years of age (range 68.3-72.3%). We also noted increasing median TCS with increasing disease severity (Figure 1A, p<0.01 and Supplemental Figures 1A-C); TCS was only slightly shorter than LOS across all three levels of the severity scale.
Table 1B.
Progression to Stability for Four TCS Measures Among Children Hospitalized with Community-acquired Pneumonia
| <2 years (n=130) | 2-4 years (n=90) | 5-17 years (n=101) | |||||
|---|---|---|---|---|---|---|---|
| TCS Measures | No. (%)1 | Median (IQR) TCS, in hours | No. (%) | Median (IQR) TCS, in hours | No. (%) | Median (IQR) TCS, in hours | p-value2 |
| RR + O2 | 108 (83.1) | 40.5 (20.1, 75.0) | 72 (80.0) | 39.6 (15.6, 79.2) | 69 (68.3) | 30.4 (14.7, 59.2) | .08 |
| RR + O2 + HR | 109 (83.8) | 40.2 (19.5, 73.9) | 73 (81.1) | 35.9 (15.9, 77.6) | 68 (67.3) | 29.8 (17.2, 56.6) | .11 |
| RR + O2 + T | 110 (84.6) | 40.5 (20.7, 70.1) | 77 (85.6) | 39.1 (18.4, 77.6) | 73 (72.3) | 28.2 (14.7, 44.7) | .03 |
| RR + O2 + HR + T | 110 (84.6) | 40.5 (20.7, 70.1) | 72 (80.0) | 39.7 (20.1, 77.5) | 71 (70.3) | 29.2 (18.2, 54) | .05 |
For each measure, time to clinical stability was calculated by subtracting the time and date of the last abnormal value for the included parameters from admission time and date to determine time to stability for each parameter in hours; children stable on admission for all four parameters not included (n=11).
Number (%) of children who reached stability more than six hours prior to discharge
p-value comparing median TCS by age group, estimated using non-parametric test of trend
abbreviations: TCS, time to clinical stability; IQR, interquartile range; T, temperature; HR, heart rate; RR, respiratory rate; O2, supplemental oxygen
Figure 1A. Time to Clinical Stability (Respiratory Rate and Supplemental Oxygen Need) and Length of Stay According to Disease Severity Among Children Hospitalized with Pneumonia.
Time to clinical stability (TCS measure incorporating respiratory rate and supplemental oxygen need) and length of stay (LOS) according to pneumonia disease severity. The median absolute difference between LOS and time to clinical stability along with interquartile range values by disease severity is also presented. The ordinal severity scale categorized children into three mutually-exclusive groups as follows: non-severe, severe, and very severe. Box and whisker plots represent the median, interquartile range, and 1.5 times the interquartile range. P-value <0.01 for non-parametric test of trend comparing time to stability according to disease severity. Abbreviations: diff., absolute difference; IQR, interquartile range; TCS, time to clinical stability; LOS, length of stay
Discussion
Our study demonstrates that longitudinal TCS measures consisting of routinely collected physiologic parameters may be useful for objectively assessing disease recovery and clinical readiness for discharge among children hospitalized with pneumonia. A simple TCS measure incorporating respiratory rate and oxygen requirement performed similarly to the more complex combinations and classified fewer children as unstable at discharge. However, we also note several challenges that deserve additional study prior to the application of a pediatric TCS measure in clinical and research settings.
Vital signs and supplemental oxygen use are used clinically to assess disease severity and response to therapy among children with acute respiratory illness. Since these objective parameters are routinely collected among hospitalized children, the systematization of these data could inform clinical decision making around hospital discharge. Similar to early warning scores used to detect impending clinical deterioration,11 TCS measures, by signaling normalization of stability parameters in a consistent and objective manner, could serve as an early signal of readiness for discharge. However, maximizing the clinical utility of TCS would require embedding the process within the electronic health record, a tool which could also have implications for the Center for Medicare and Medicaid Services’ meaningful use regulations.12
TCS could also serve as an outcome measure in research and quality efforts. Increased disease severity was associated with longer TCS for the four combined measures; TCS also demonstrated strong agreement with LOS. Further, TCS minimizes the influence of factors unrelated to disease that may impact LOS (e.g. frequency of hospital rounds, transportation difficulties, or social impediments to discharge), an advantage when studying outcomes for research and quality benchmarking.
The percentage of children reaching stability and the median TCS for the combined measures demonstrated little variation within each age group, likely because respiratory rate and need for supplemental oxygen, two of the parameters with the longest individual time to stability, were also included in each of the combination measures. This suggests that less complex measures incorporating only respiratory rate and need for supplemental oxygen may be sufficient to assess clinical stability, particularly since these parameters are objectively measured and possess a direct physiological link to pneumonia. In contrast, the other parameters may be more often influenced by factors unrelated to disease severity.
Our study also highlights several shortcomings of the pediatric TCS measures. Despite use of published, age-based reference values,13 we noted wide variation in the achievement of stability across individual parameters, especially for children >5 years. Overall, 21% of children had ≥1 abnormal parameter at discharge. Even the simplest combined measure classified 13.4% of children as unstable at discharge. Discharge with unstable parameters was not associated with 7 day readmission, although our study was underpowered to detect small differences. Additional study is therefore needed to evaluate less restrictive cut-off values on calculated TCS and the impact of hospital discharge prior to reaching stability. In particular, relaxing the upper limit for normal respiratory rate in adolescents (16 breaths per minute) to more closely approximate the adult TCS parameter (24 breaths per minute) should be explored. Refinement and standardization of age-based vital sign reference values specific to hospitalized children may also improve the performance of these measures.14
Several limitations deserve discussion. TCS parameters and readmission data were abstracted retrospectively from a single institution and our findings may not be generalizable. Although clinical staff routinely measured these data, measurement variation likely exists. Nevertheless, such variation is likely systematic, limiting the impact of potential misclassification. TCS was calculated based on the last abnormal value for each parameter; prior fluctuations between normal and abnormal periods of stability were not captured. We were unable to assess room air oxygen saturations. Instead, supplemental oxygen use served as a surrogate for hypoxia. At our institution, oxygen therapy is provided for children with pneumonia to maintain oxygen saturations of 90-92%. We did not assess work of breathing (a marker of severe pneumonia) or ability to eat (a component of adult TCS measures). We initially considered the evaluation of intravenous fluids as a proxy for ability to eat (addition of this parameter to the four parameter TCS resulted in a modest increase in median time to stability, data not shown); however, we felt the lack of institutional policy and subjective nature of this parameter detracted from our study's objectives. Finally, we were not able to determine clinical readiness for discharge beyond the measurement of vital sign parameters. Therefore, prospective evaluation of the proposed pediatric TCS measures in broader populations will be important to build upon our findings, refine stability parameters, and test the utility of new parameters (e.g., ability to eat, work of breathing) prior to use in clinical settings.
Our study provides an initial evaluation of TCS measures for assessing severity and recovery among children hospitalized with pneumonia. Similar to adults, such validated TCS measures may ultimately prove useful for improving the quality of both clinical care and research, although additional study to more clearly define stability criteria is needed prior to implementation.
Supplementary Material
Acknowledgments
This work was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health under Award Number K23AI104779 to Dr. Williams. The EPIC study was supported by the Influenza Division in the National Center for Immunizations and Respiratory Diseases at the Centers for Disease Control and Prevention through cooperative agreements with each study site and was based on a competitive research funding opportunity. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention or the National Institutes of Health.
Footnotes
Personal Disclosures:
Dr. Grijalva serves as a consultant to Glaxo-Smith-Kline and Pfizer outside of the scope of this manuscript. Dr. Edwards is supported through grants from Novartis for the conduction of a Group B strep vaccine study and serves as the Chair of the Data Safety and Monitoring Data Committee for Influenza Study outside the scope of this manuscript. Dr. Self reports grants from CareFusion, BioMerieux, Affinium Pharmaceuticals, Astute Medical, Crucell Holland BV, BRAHMS GmbH, Pfizer, Rapid Pathogen Screening, Venaxis, BioAegis Inc, Sphingotec GmbH, and Cempra Pharmaceuticals; personal fees from BioFire Diagnostics and Venaxis, Inc; and patent 13/632,874 (Sterile Blood Culture Collection System) pending; all outside the scope of this manuscript.
References
- 1.Healthcare Cost and Utilization Project (HCUP) < http://www.ahrq.gov/research/data/hcup/index.html>.
- 2.Halm EA, et al. Time to clinical stability in patients hospitalized with community-acquired pneumonia: implications for practice guidelines. JAMA J. Am. Med. Assoc. 1998;279:1452–7. doi: 10.1001/jama.279.18.1452. [DOI] [PubMed] [Google Scholar]
- 3.Menéndez R, et al. Reaching stability in community-acquired pneumonia: the effects of the severity of disease, treatment, and the characteristics of patients. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2004;39:1783–1790. doi: 10.1086/426028. [DOI] [PubMed] [Google Scholar]
- 4.Arnold F, et al. The pneumonia severity index predicts time to clinical stability in patients with community-acquired pneumonia. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Tuberc. Lung Dis. 2006;10:739–743. [PubMed] [Google Scholar]
- 5.Snijders D, et al. Efficacy of corticosteroids in community-acquired pneumonia: a randomized double-blinded clinical trial. Am. J. Respir. Crit. Care Med. 2010;181:975–982. doi: 10.1164/rccm.200905-0808OC. [DOI] [PubMed] [Google Scholar]
- 6.Silber SH, et al. Early administration of antibiotics does not shorten time to clinical stability in patients with moderate-to-severe community-acquired pneumonia. Chest. 2003;124:1798–1804. doi: 10.1378/chest.124.5.1798. [DOI] [PubMed] [Google Scholar]
- 7.Jaoude P, et al. A comparison between time to clinical stability in community-acquired aspiration pneumonia and community-acquired pneumonia. Intern. Emerg. Med. 2014;9:143–150. doi: 10.1007/s11739-012-0764-2. [DOI] [PubMed] [Google Scholar]
- 8.Arnold FW, et al. A worldwide perspective of atypical pathogens in community-acquired pneumonia. Am. J. Respir. Crit. Care Med. 2007;175:1086–1093. doi: 10.1164/rccm.200603-350OC. [DOI] [PubMed] [Google Scholar]
- 9.Jain S, et al. Community-acquired pneumonia requiring hospitalization among U.S. children. N Engl J Med. 2015;372:835–845. doi: 10.1056/NEJMoa1405870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.American Heart Association 2005 American Heart Association (AHA) guidelines for cardiopulmonary resuscitation (CPR) and emergency cardiovascular care (ECC) of pediatric and neonatal patients: pediatric basic life support. Pediatrics. 2006;117:e989–1004. doi: 10.1542/peds.2006-0219. [DOI] [PubMed] [Google Scholar]
- 11.Parshuram CS, Hutchison J, Middaugh K. Development and initial validation of the Bedside Paediatric Early Warning System score. Crit Care. 2009;13:R135. doi: 10.1186/cc7998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Center for Medicare and Medicaid Services Guidelines for Meaningful Use. < http://www.cms.gov/Regulations-and-Guidance/Legislation/EHRIncentivePrograms/index.html>.
- 13.Bonafide CP, et al. Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131:e1150–1157. doi: 10.1542/peds.2012-2443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cortoos PJ, et al. Length of stay after reaching clinical stability drives hospital costs associated with adult community-acquired pneumonia. Scand. J. Infect. Dis. 2013;45:219–226. doi: 10.3109/00365548.2012.726737. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.