Abstract
Peptic ulcer disease is a multifactorial and complex disease involving gastric and duodenal ulcers. Despite medical advances, the management of peptic ulcer and its complications remains a challenge, with high morbidity and death rates for the disease. An accumulating body of evidence suggests that, among a broad reach of natural molecules, dietary polyphenols with multiple biological mechanisms of action play a pivotal part in the management of gastric and duodenal ulcers. The current review confirmed that dietary polyphenols possess protective and therapeutic potential in peptic ulcer mediated by: improving cytoprotection, re-epithelialization, neovascularization, and angiogenesis; up-regulating tissue growth factors and prostaglandins; down-regulating anti-angiogenic factors; enhancing endothelial nitric oxide synthase-derived NO; suppressing oxidative mucosal damage; amplifying antioxidant performance, antacid, and anti-secretory activity; increasing endogenous mucosal defensive agents; and blocking Helicobacter pylori colonization associated gastric morphological changes and gastroduodenal inflammation and ulceration. In addition, anti-inflammatory activity due to down-regulation of proinflammatory cytokines and cellular and intercellular adhesion agents, suppressing leukocyte-endothelium interaction, inhibiting nuclear signaling pathways of inflammatory process, and modulating intracellular transduction and transcription pathways have key roles in the anti-ulcer action of dietary polyphenols. In conclusion, administration of a significant amount of dietary polyphenols in the human diet or as part of dietary supplementation along with conventional treatment can result in perfect security and treatment of peptic ulcer. Further well-designed preclinical and clinical tests are recommended in order to recognize higher levels of evidence for the confirmation of bioefficacy and safety of dietary polyphenols in the management of peptic ulcer.
Keywords: Anti-inflammatory, Dietary supplement, Helicobacter pylori, Inflammatory cytokines, Natural medicine, Non-steroidal anti-inflammatory drugs, Peptic ulcer, Polyphenols, Ulcer healing
Core tip: Polyphenols ubiquitously present in vegetables and fruits are progressively viewed as natural dietary ingredients vital for a balanced diet. An accumulating body of evidence indicates that dietary polyphenols with multiple biological mechanisms of action play a pivotal part in the management of gastric and duodenal ulcers. Administration of a sufficient amount of dietary polyphenols in the human diet or as part of dietary supplementation along with conventional treatment can result in perfect prevention and treatment of peptic ulcer.
INTRODUCTION
Peptic ulcer disease is a multifactorial and complex disease involving gastric and duodenal ulcers[1,2]. For many years, peptic ulcer was considered one of the main reasons for performing gastrointestinal surgery, owing to its high prevalence of morbidity and mortality. Peptic ulcer disease affects a wide range of people worldwide, and is one of the most common diseases of the twenty-first century[3].
PATHOPHYSIOLOGICAL PROCESSES IN PEPTIC ULCER
Peptic ulcer results from a pathological condition in which the biological balance between defensive and offensive factors in the gastrointestinal tract is disturbed. Gastric hydrochloric acid, pepsin, reactive free radicals and oxidants, leukotrienes, refluxed bile, and endothelins are among the main endogenous aggressive factors[4,5]. In addition, gastric mucus barrier, bicarbonate, mucosal blood flow, surface active phospholipids, prostaglandins (PG), nitric oxide (NO), as well as enzymatic and non-enzymatic antioxidant performance are considered defensive factors[4-6]. The exact pathogenesis of peptic ulcers is not clear, but diverse factors, including consumption of non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, stressful lifestyle, alcohol consumption, Helicobacter pylori (H. pylori) infection, smoking, and family history are considered as risk factors in the pathogenesis of peptic ulcer[2,7].
Oxidative damage
The pivotal role of antioxidants in the prevention and healing of peptic ulcer has been widely studied in numerous investigations. Tissue damage is always associated with intense generation of free radicals such as reactive oxygen species (ROS) that cause oxidative stress and subsequent mucosal injury. Likewise, oxidative mucosal damage contributes with DNA fragmentation, lowering cellular DNA content, and impairment of protein expression which is mediated by the intense generation of ROS. These free radicals also disturb the cellular antioxidant enzyme function that acts as an important cellular defense against oxidative stress, which leads to aggravated tissue damage during gastroduodenal ulceration. Leukocytes (particularly neutrophils) encompass a nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, which has an oxidative action; reduction of molecular oxygen to superoxide anion radical, one of the main type of ROS[2,5,8,9]. Free radicals initiate microvascular permeability, leading to extra infiltration of plasma cells and macrophages to gastric endothelial cells. Numerous endogenous anti-oxidative agents like sulfhydryl (SH) non-protein compounds, enzymatic antioxidants [including superoxide dismutase (SOD) and catalase (CAT)], as well as non-enzymatic proteins like glutathione (GSH) block biological production of ROS and consequent tissue damage[9,10].
Gastric mucus
Gastric mucosal layers act as a barrier which restricts the exposure of the gastric cells to various injurious agents of both exogenous and endogenous origin. Enhancement of mucus production could be assumed as a central factor in ulcer healing by protecting the damaged tissue against various aggressors like harmful drugs and oxidants. In fact, secretion of mucus is an obvious contributor in improving epithelial recovery after acute injury through formation of a mucoid cover beneath initializing re-epithelization. Additionally, the formation of buffering capacity and neutralization of luminal acid are main physiological functions of the gastric mucus[11,12].
NSAIDs
NSAID-induced peptic ulcer is the most serious complication of any synthetic drug therapy[13]. It is now well-recognized that ulceration induced by NSAIDs is mediated by suppression of the cyclooxygenase (COX)-dependent pathway and subsequently blocking synthesis of PG. Different factors, such as leukocyte-endothelium interaction, neutrophils infiltration, cytokine imbalance, and oxidative mucosal stress, contribute to the pathogenesis of gastric mucosal injury initiated by NSAIDs. Activation of adhesion molecules, including cellular adhesion molecules (CAMs) and intercellular adhesion molecule (ICAM-1) is associated with neutrophil infiltration[14-17]. In addition, NSAIDs delay the healing process of gastric injury resulting from suppressing angiogenesis by down-regulation of the pro-angiogenic agents along with up-regulation of anti-angiogenic proteins. Angiogenesis possess a key role in ulcer healing due to elevating mucosal blood flow and providing vital substances to the injured tissue, resulting in the promotion of re-epithelialization and healing[13,17,18].
Helicobacter pylori
Chronic infection of gastric mucosa with H. pylori is generally associated with gastric lesions. H. pylori is a prevalent human pathogen with an incidence of 90% in some developing countries. H. pylori undergoes asymptomatic gastric colonization in approximately 70% of the population, with a 10%-20% susceptibility of developing into peptic ulcer. The pathogenesis and pattern of H. pylori-induced gastritis is intensely associated with the morbidity of mucosal atrophy and duodenal/gastric ulcers. Eradication of H. pylori from the gastric mucosa of infected patients is considered to be the best therapeutic approach for complete remission of H. pylori associated gastritis and its consequent ulcers[19,20]. H. pylori infection stimulates overexpression of pro-inflammatory cytokines [e.g., interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF)-α, and IL-8] in gastric epithelial cells, which act as neutrophil-activating chemokines and lead to leukocyte infiltration. It is possible that H. pylori-associated IL-8 activation results in disturbing mucosal integrity due to generation of ROS, as well as the activation of proteolytic enzymes[21]. Various factors are involved in the severity of H. pylori-associated disease, such as age of acquisition, host immune system, and bacterial strain. Strains of H. pylori that carry the Cag-PAI (Cag Pathogenicity Island) show a more potent inflammatory and chemoattractant response than those that are Cag-negative. This early and severe response of Cag-positive strains is mediated by activating protein-1, as well as nuclear transcriptional factors [e.g., nuclear factor (NF)-κB][19,22,23].
CONVENTIONAL TREATMENT FOR PEPTIC ULCER
Several chemical drugs are presently available for the prevention and treatment of peptic ulcer, but none without adverse effects. Headache, anti-androgenic effect, pancreatitis, and confusion in elderly patients have been reported for H2 receptor antagonists. Proton pump inhibitors have demonstrated itching, skin rash, diarrhea, dizziness, and inactivation of some anti-fungal drugs. Likewise, indigestion, drowsiness, nausea, constipation, dizziness, and back pain are among the main side effect of sucralfate. Misoprostol, which is administrated for the prevention and treatment of NSAIDs-induced gastric ulcer, has shown different side effects, such as: abdominal pain, diarrhea, vaginal bleeding, constipation, and abortion in pregnant women. Prevention and effective remission of gastrointestinal ulcers remains a concern for both researchers and physicians[4,5,24]. Given the obvious role of H. pylori in gastritis and gastrointestinal ulcers, different combination therapies have been used in order to eradicate this pathogen, in addition to offering protective effects or remission of gastrointestinal diseases. Although combination therapy of a proton pump inhibitor with two antibiotics improves the symptoms and status of the disease via enhancement of eradication rate, this therapeutic approach is not commonly successful, with ulcer recurrence often occurring. Several investigations have reported treatment failure in antibiotic-resistant H. pylori infection (more specifically for antibiotics containing clarithromycin and metronidazole), which results in serious problems in the management of peptic ulcer[23,25].
Although various pharmacotherapeutic choices which suppress one or more processes involved in the pathogenesis of peptic ulcer are under evaluation, a perfect cure for this disease has not yet been achieved.
HEALTH BENEFITS OF POLYPHENOLS
Polyphenols are one of the largest secondary plant metabolites ubiquitously present in fruits and vegetables considered an integral part of the human diet. Polyphenols are characterized by the presence of different numbers of phenolic rings, along with two or more hydroxyl substitutions. Polyphenols consist of a wide variety of chemical structures based on the substitutions of the basic chemical structure of polyphenols, polymerization, and the degree of oxidation. Polyphenols are classified as flavonoids and non-flavonoids, based on their chemical structure. Flavonoids are comprised of two benzene rings linked by a linear three-carbon chain, which form an oxygenated heterocycle. Flavonoid-type polyphenols are divided into different subclasses according to the oxidation state of the central pyran ring, including anthocyanins, anthocyanidins, flavones, flavanols, catechins, isoflavones, flavanones, and flavonols. Non-flavonoids are comprised of stilbenes, phenolic acids, lignans, and hydroxycinnamic acids[26-28]. Their basic phenolic structure, known as “aglycone”, can be connected with different carbohydrates and organic acids to form “glycone” structures, as well as with various polyphenols to form “polymers”. Phenolic compounds with pharmaceutical interest are simple phenolic compounds such as gallic acid, ellagic acid, catechin, eugenol, vanillin, caffeic acid, ferulic acid, apigenin, quercetin, gingerol, kaempferol, myricetin, resveratrol, rutin, naringenin, and cyanidin[26,29].
Mounting evidence suggests that polyphenols are health promoting phytochemicals. A large number of human studies have demonstrated that polyphenol intake is associated with a reduced risk of various chronic illnesses, including cardiovascular disease, neurodegenerative disorders, diabetes, cancers, osteoarthritis, and gastrointestinal diseases. The biological activities of polyphenols vary due to numerous parameters, such as food processing methods, metabolism, absorption, bioavailability, derived compounds, and bioefficacy[26-28,30]. At the cellular and molecular levels, a wide range of pharmacological functions have been confirmed for polyphenols, including as an antioxidant, anti-apoptotic, anti-carcinogenic, anti-aging, antiviral, anti-allergic, antiplatelet, anti-estrogenic, anticarcinogenic, anti-inflammatory, and anti-proliferative properties. These therapeutic beneficial effects of polyphenols are associated with molecular mechanisms of action on intracellular signaling pathways; regulation of NFκB, mitogen-activated protein kinases (MAPKs), extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and signal transduction pathway interference [e.g., toll-like receptor 4 (TLR4)]. The antioxidant potential of polyphenols are performed by their direct action on free radical scavenging, breaking of radical chain reactions, peroxide reduction, and metal chelating properties, or indirectly by interacting with definite proteins of oxidative stress signaling pathways and by activating anti-oxidative enzymes like CAT and SOD[26-28,31].
DIETARY SUPPLEMENTATION OF POLYPHENOLS
There is ample evidence in the literature suggesting the beneficial role of a healthy diet in the prevention and remission of various diseases. One of the most sustainable approaches in order to protect against various chronic diseases and alleviate symptoms is removing patients from their habitual diet and providing them with natural dietary agents[32]. As a disorder of the GI tract, pathological conditions in peptic ulcer could be alleviated by nutritional factors. Dietary consumption of a significant amount of “natural” protective supplements in early life leads to prevention or delayed peptic ulcer[4,5,33].
Regarding the wide range of positive biological effects of polyphenols, as well as their crucial role in human health, incorporation of these dietary supplements in a balanced diet is necessary for the prevention and management of diseases. The average daily dietary intake of polyphenols in a balanced diet is estimated at nearly 1 g/d[34]. Oral consumption of polyphenols typically produces very few side effects, and no significant toxicity was reported in the literature. The gastrointestinal absorption of polyphenols varies based on their chemical structure; aglycone forms are absorbed through passive diffusion, whereas some glycoside forms, generally glucosides, interact with proteins such as cytosolic β-glucosidase, glucose transporter, or sodium-glucose transporter, resulting in limited absorption of glycosylated polyphenols[35,36].
Considering the wide variety of therapeutic benefits which has been reported for polyphenols, the current study was conducted to discuss the potential effects of commonly used dietary polyphenols in the prevention and/or treatment of peptic ulcer based on cellular, preclinical, and clinical studies, along with the possible molecular and intracellular mechanisms.
POLYPHENOLS FOR THE MANAGEMENT OF PEPTIC ULCER
It has been reported that polyphenols possess various pharmacological effects in the gastrointestinal tract. Numerous studies have been carried out to investigate the efficacy of polyphenols in the management of peptic ulcer. Some of the most efficacious dietary polyphenols with well-established preclinical studies on the prevention and treatment of peptic ulcer are discussed below.
Allylpyrocatechol
Allylpyrocatechol is a phenolic compound elicited from the leaves of Piper betel (P. betel) commonly grows in a tropical humid climate and the leaves are widely-used as a mouth freshener in South East Asia. Allylpyrocatechol is an excellent antioxidant agent and also possess anti-inflammatory, antibacterial, and anti-arthritic effects. Various investigations have confirmed the therapeutic potential of allylpyrocatechol for the management of peptic ulcer via different molecular mechanisms. Yadav et al[37] showed the significant protection of allylpyrocatechol against indomethacin-induced gastric ulcer in an animal model in comparison with that of standard drugs (misoprostol and omeprazole). In a histopathological assay, allylpyrocatechol demonstrated a protective effect on mucosa and submucosa. Indomethacin-induced gastric damage is mediated by a decrease in COX-1 expression, along with simultaneous elevation of COX-2 expression, which is reversed by allylpyrocatechol. Prostaglandins have a pivotal role in the prevention and treatment of peptic ulcer. They alleviate indomethacin-induced gastric mucosal damage and promote ulcer healing via the enhancement of bicarbonate and mucus secretion, as well as the acceleration of angiogenesis and mucosal blood flow[12]. Allylpyrocatechol markedly increases the level of PGE2 and PGEM, a metabolite of PGE2 produced by 15-hydroxyprostaglandin dehydrogenase (15-PGDH). Indomethacin-induced peptic ulcer is also associated with hypermotility and compression of the stomach, causing impairment of mucosal blood flow and angiogenesis, as well as leukocytes adhesion, which is attributed to lessoned pro-angiogenic/anti-angiogenic ratio and von Willebrand factor (VWF) expression. Allylpyrocatechol enhances angiogenesis through improvement of vascular endothelial growth factor (VEGF) and VWF, as well as by the reduction of endostatin. It also inhibits leukocyte infiltration and interactions with the endothelial layer via the suppression of adhesion molecules, including E-selectin, P-selectin, and VCAM-1[13-15,37].
Inflammatory situations exacerbate gastric lesions and prohibit healing parameters. Allylpyrocatechol prevents TNF-α stimulated expression of pro-inflammatory cytokines such as NF-κB through the inhibition of the intracellular inflammatory signaling pathway, IκB phosphorylation, MAPK-a signaling cascade regulating transcription, and translation of proteins involved in the inflammatory process (JNK and ERK1/2)[13-16].
The cytoprotective potential of allylpyrocatechol is through improvement of prostaglandin expression and activation, which is mediated by the stimulation of COX-1. Antioxidant activity is another protective mechanism which is mediated by preventing monocytes and macrophages from producing ROS, as well as inhibiting the ROS/NF-κB dependent pathway within gastric tissue[37,38].
Seven-day administration of allylpyrocatechol significantly alleviated histopathological damage of the gastric mucosa in rats with indomethacin-induced peptic ulcer. Bhattacharya et al[38] showed that the healing capacity of allylpyrocatechol is not significantly different from misoprostol, and is mediated by antioxidant activity. Allylpyrocatechol improves the levels of the antioxidant enzymes SOD and CAT, as well as preventing lipid peroxidation and protein and DNA oxidative damage in gastric tissue. Likewise, allylpyrocatechol significantly enhances mucin, a macromolecular glycoprotein which is the major component of gastric mucus that accelerates epithelial recovery and forms a mucoid layer that promotes tissue repair[38]. It has been demonstrated that the ulcer healing function of allylpyrocatechol is attributed to the augmentation of cellular growth factors including VEGF and epidermal growth factor (EGF)[39]. This polyphenol improves angiogenesis through the enhancement of VWF/factor VIII complex, as well as growth factors. Healing potential of the molecule is mainly through enhancement of PGE2 expression and reduction of COX cascade[39].
Apple polyphenols
Apples (Malus spp., Rosaceae) represent an excellent source of polyphenols. Various studies suggest that dietary intake of apple polyphenols and apple-derived polyphenolic products can prevent such chronic diseases as oxidative stress-associated disorders, cardiovascular disease, and arthritic diseases, and also reinforce gastrointestinal protection from drug damage[40]. Apple polyphenols encompass different structural classes, including hydroxycinnamic acids, flavonols (quercetin glycosides), dihydrochalcones (phloretin glycosides), and flavan-3-ols (oligomeric pro-cyanidins). Among the abundant apple polyphenols are chlorogenic acid, procyanidins, and dihydrochalcones[41,42]. Several preclinical investigations have revealed the protective and healing potential of apple polyphenols on peptic ulcer through various intracellular and molecular mechanisms[43-45].
Paturi et al[43] reported 10-d dietary intake of apple polyphenols following aspirin-induced gastric ulcer in rats has significant gastroprotective effects, which were confirmed by histopathological examination. The main mechanism of action is antioxidant activity in gastric mucosa via up-regulation of GSH. Consistent with this, apple polyphenol enhances glutathione-S-transferase P1 expression, acting as an enzymatic antioxidant in gastric tissue. Apple polyphenols also up-regulate mucin-2 and trefoil factor-2 gene expression, which have an important role in the defensive function of the stomach barrier. D’Argenio et al[44] demonstrated pretreatment with apple polyphenols reduce acute and chronic aspirin-induced ulceration, which confirmed by microscopic gastric tissue examination. Apple polyphenols suppress overexpression of mRNA and protein of COX-2 in gastric tissue. They also inhibit peroxidation of tissue lipids and reduce the level of malondialdehyde (MDA) in gastric mucosa. Likewise, apple polyphenol protects human gastric epithelial cells against xanthine-xanthine oxidase-induced damage, as well as indomethacin-induced oxidative damage, in vitro via enhancement of antioxidant capacity and preventing lipid peroxidation[45]. However, polyphenols have no significant effect on gastric acid secretion[44].
Anti-H. pylori properties are another main biological mechanism of apple polyphenols in the management of peptic ulcer. Apple peel polyphenol-rich extract exhibited inhibitory activity on H. pylori growth, infection, and tissue adhesion, as well as its vacuolation induced by vacuolating bacterial toxin. Likewise, in an animal model of H. pylori infection, the polyphenols ameliorate H. pylori-associated inflammatory reaction and gastritis[46].
Curcumin
Curcumin (diferuloylmethane) is the major constituent of turmeric (a spice derived from the rhizome of Curcuma longa), which is extensively consumed in Asian foods as an additive, coloring, and preservative agent. Curcumin is the active constituent responsible for its vibrant yellow color. It has been characterized that curcumin is a strong antioxidant and anti-inflammatory substance[47]. It has been shown that curcumin possesses several positive biological activities in different types of tissue, including hepatoprotective and anti-hepatotoxic, anti-arthritic, and protective effects in cardiovascular tissue, in addition to hypocholesterolemic and hypolipidemic properties[48]. Positive functions of curcumin, including free radical scavenging, as an antioxidant, modulating of key enzymes regulating cell life, and anti-inflammatory properties, resulted in considering this molecule as one of the most promising natural agents in the prevention and treatment of peptic ulcer[49-53].
Pretreatment with intraperitoneal (ip) administration of curcumin lessened indomethacin-induced small intestinal ulcer significantly. The role of curcumin as an antioxidant indicates the mechanism of protective effect in intestinal lumen. Curcumin enhance the level of antioxidant enzymes CAT and glutathione peroxidase (GPx). It also prevents peroxidation of membrane lipids and promotes maintaining integrity of cellular membrane. It suppresses xanthine oxidase activity, which generates superoxide radicals via catalytic function, as well as myeloperoxidase (MPO) activity as a source of ROS. Thus, curcumin via antioxidant performance suppresses oxidative stress-associated cellular apoptosis in the small intestine[49].
Pretreatment with curcumin can alleviate gastric lesions induced by indomethacin through amelioration of oxidative damage, scavenging ROS, suppressing thiol depletion and lipid peroxidation, and protecting gastric mucosal peroxidase against drug-associated inactivation resulting in inhibiting the accumulation of endogenous H2O2 and its OH derivative. This polyphenol ameliorates ulcer aggravation by suppressing elevated gastric acid secretion. Chattopadhyay et al[50] showed that anti-ulcer activity of curcumin is comparable to the standard drugs ranitidine and omeprazole. Likewise, Swarnakar et al[51] demonstrated both ip and oral administration of curcumin prevented gastric lesions induced by indomethacin dose, dependently. Prevention of GSH depletion and protein oxidation are among curcumin’s anti-oxidative stress mechanisms in peptic ulcer.
Matrix metalloproteinase (MMPs) are a developing group of zinc-dependent endopeptidases which selectively degrade constituents of the extracellular matrix. MMPs possess dynamic function in remodeling the extracellular matrix and regulation of matrix proteins like collagens. Curcumin possesses gastroprotective and healing properties through the enhancement of MMP-2 expression, along with reduction of MMP-9 activity in gastric tissue causing re-epithelialization and remodeling of endothelial tissue[14-16,52]. Furthermore, angiogenesis and collagenization within gastric tissues are among the main molecular mechanisms of curcumin, which is modulated via the up-regulation of VEGF, membrane type (MT) 1-MMP, MMP-2, and transforming growth factor (TGF)-β expression at both protein and mRNA levels[52].
Pretreatment with curcumin (200 mg/kg BW dissolved in olive oil) in rats with indomethacin-induced peptic ulcer markedly attenuated tissue erosions, leukocyte infiltration, and adherence to postcapillary venule compared to control. This activity is mediated by suppressing the pro-inflammatory cytokine TNF-α, as well the intercellular adhesion molecule ICAM-1[53].
It has been reported that curcumin has a strong antibacterial effect against 65 clinical isolates of H. pylori with the MIC range of 5-50 μg/mL, in vitro. The antibacterial potential of curcumin on metronidazole-resistant H. pylori strains revealed different mechanisms from the mode of action of current antibiotics for the prevention of H. pylori infection. Curcumin also demonstrated a therapeutic effect in an animal model of H. pylori infection. It was able to significantly eradicate H. pylori from infected mice and improve H. pylori-associated gastric damage. Inhibiting shikimate dehydrogenase, an essential pathway for the synthesis of vital metabolites in bacteria, seems to be among the main biological mechanisms of curcumin in the eradication of H. pylori[54].
Several clinical investigations have evaluated the therapeutic effect of curcumin in the management of peptic ulcer, as well as the eradication of H. pylori infection (Table 1)[55-58].
Table 1.
Clinical evidence of dietary polyphenols in the management of peptic ulcer
| Polyphenol |
Preparations |
Study design | Disease | No. of patients | Treatment duration | Outcomes | Level of evidence1 | Ref. | |
| Treatment group | Control group | ||||||||
| Curcumin | Turmeric 250 mg, four times per day | Liquid antacid: aluminum hydroxide (333 g) and magnesium hydroxide (33.3 g) per 1000 mL (30 mL, four times per day | Randomized controlled clinical | Benign gastric ulcers | 60 | 12 wk | Both antacid drug and turmeric significantly reduced the formation of gastric ulcer. The former was more effective in reducing the ulcers | 1b | Kositchaiwat et al[55] |
| Curcumin | Capsule-filled turmeric 3 g/d | - | Uncontrolled clinical trial | Peptic ulcer | 54 | 4 wk | 12 (48%) of patients showed remission after 4 wk, 18 of patients after 8 wk of treatment and 19 of patients after 12 wk. There was no significant alteration in hematological parameters, liver, or renal functions after treatment | 4 | Prucksunand et al[56] |
| Curcumin | Turmeric tablet (40 mg curcumin) | Omeprazole (20 mg), amoxicillin (1 g), metronidazole (800 mg), twice a day for 1 wk | Randomized controlled trial | Chronic gastritis with H. pylori infection | 36 | 4 wk | The eradication rate of H. pylori in curcumin group was 5.9% while in control group was 78.9%. IL-8 mRNA expression in control group reduced significantly in comparison with before trial, while curcumin had no effect on IL-8 | 1b | Koosirirat et al[57] |
| Curcumin | Curcumin (30 mg), bovine lactoferrin (100 mg), N-acetylcysteine (600 mg), and pantoprazole (20 mg), twice daily | - | Uncontrolled clinical trial | Peptic ulcer | 25 | 7 d | 12% of patients showed eradication of H. pylori infection, overall severity of gastrointestinal symptoms was significantly decreased after treatment in comparison with baseline (P < 0.001). Serum pepsinogens were significantly reduced after treatment in comparison with baseline (P < 0.05) | 4 | Di Mario et al[58] |
| Green tea | More than 1 d in week | - | Cohort study | Peptic ulcer | 150 | - | The incidence of H. pylori infection was lower in patients consuming green tea (45.2%) compared with the other patients (64.8%) | 4 | Boyanova et al[87] |
Level of evidence was calculated based on Oxford Centre for Evidence-based Medicine - Levels of Evidence (March 2009).
In addition, curcumin mucoadhesive microspheres enhance the stomach residence time of curcumin, leading to an improvement in its ability to eradicate H. pylori and its related gastropathy[59].
Gallic acid
Gallic acid (3,4,5-trihydroxybenzoic acid) is a polyhydroxy phenolic compound commonly used as a dietary herbal supplement. Gallic acid is naturally found in various vegetables and fruits, such as gallnuts, Tragopogon species, sumach, grapes, pineapple, Phyllanthus spp., and zinger[60-62]. It is a strong antioxidant and possesses various therapeutic effects, including anti-cancer, anti-obesity, hepatoprotective, and anti-asthmatic effects[63]. Gallic acid is considered an essential active constituent of a wide range of herbal preparations used for the management of peptic ulcer[8]. The bioavailability of gallic acid in humans is low and its plasma concentration subsequent to 50 mg oral consumption of gallic acid is 1.8 μmol/L. This polyphenol is generally metabolized into 4-O-methylgallic acid, which is presumed to play an important role in its therapeutic action in peptic ulcer[8,61,62].
Gallic acid encompasses the ability to prevent and heal gastric mucosal damage induced by NSAIDs by its antihistaminic activity, as well as inhibiting up-regulation of pro-inflammatory cytokines and modulating inflammatory reactions[63,64]. Iron-chelating, free radicals scavenging, and preventing OH-mediated oxidative endothelial damage have been assumed to be its main mechanisms in preventing and healing peptic ulcers. In addition, gallic acid diminishes diclofenac- and indomethacin-associated mitochondrial oxidative stress by suppressing mitochondrial protein carbonyl generation, thiol depletion, and lipid peroxidation.
It is suggested that apoptosis is the main cause of cellular injury in gastropathy. Gallic acid significantly suppresses apoptosis induced by indomethacin in preclinical studies. In fact, this polyphenol prevents apoptosis via suppressing activation of caspase-3, a key mediator in the execution level of indomethacin-associated gastric mucosal cell apoptosis, and caspase-9, which is a mediator of the mitochondrial pathway of apoptosis[14-17,63]. In addition, gallic acid modulates oxidative damage-induced mitochondrial dysfunction by regulating mitochondrial transmembrane potential and mitochondrial dehydrogenase activity[63].
Seven days of pretreatment with gallic acid in an animal model of gastric ulcer induced by aspirin plus pylorus ligation reduced mucosal injury and gastric juice volume, which indicated the anti-secretory potential of this polyphenol. It prevented the release of endogenous offensive factors, pepsin, and free and total acidity, along with the enhancement of pH and carbohydrate concentration in the stomach. Likewise, it inhibited protein and DNA leakage from gastric mucosa through reinforcement the mucosal barrier. Enzymatic and non-enzymatic antioxidant proteins encompass an obvious role in the prevention of peptic ulcer. Phenolic acid elevated the level and activity of SOD, CAT, reduced GSH, GPx, glutathione reductase, and glucose-6-phosphate dehydrogenase, while peroxidation of lipids was significantly lessened in mucosal tissue. It suppresses MPO level in mucosal tissue, indicating diminishing neutrophil migration and infiltration within damaged tissue[8].
One week treatment with gallic acid enriched herbal extract (Phyllanthus emblica) significantly alleviated indomethacin-induced ulcer in animals. The healing potential of gallic acid enriched extract was reversed by the nonspecific NOS inhibitor, N-nitro-L-arginine methyl ester (L-NAME), while the i-NOS-specific inhibitor, L-N6-(1-iminoethyl) lysine hydrochloride (L-NIL) did not prevent healing activity, indicating the important role of eNOS in its therapeutic action[65]. It is now well-recognized that gastric ulceration induced by indomethacin is associated with elevated expression and activity of mucosal iNOS, while eNOS activity is lessened. Considering the key role of nitrogen metabolizing enzymes in the ulcer healing process, it is confirmed that gallic acid-enriched extract enhances eNOS/iNOS ratio along with eNOS-derived NO in gastric mucosal tissue significantly[65].
Gallic acid extract enhances blood vessel formation and microvessel number via the elevation of vWF/factor VIII complex. Moreover, augmentation of tissue growth factors, including VEGF, which improves cell proliferation, as well as hepatocyte growth factor, which regulates cell migration and re-epithelialization, are key contributors in the gastric mucosal repairment of gallic acid. It is suggested that the cytoprotective potential of gallic acid extract’s anti-ulcer mechanism is mediated by COX dependent cascade and up-regulation of PGE2 expression. These finding support the positive effect of dietary gallic acid in the vasodilation and angiogenesis pathways of ulcer healing[11,17,65].
Grape polyphenols
The grape, a fruiting berry of the woody vines (Vitis spp.) from the family Vitaceae, is an essential source of nutritional phytochemicals such as flavonoids and anthocyanins. A wide variety of anthocyanins containing glycosidic forms of cyanidin, delphinidin, peonidin, malvidin, pelargonidin, and petunidin have been identified in grapes. Several experimental investigations reported that grape polyphenol encompass positive medicinal functions like antioxidant, anti-inflammatory, antimicrobial and anti-platelet activities[66,67]. Grape seed extract is rich in flavonoids (generally proanthocyanidin) and possesses preventive activity against numerous diseases, such as atherosclerosis, vascular injury, cardiovascular disease, and cancer, as well as combating the aging process. It has been shown that proanthocyanidin of grape seeds has an adequate bioavailability in the vital organs. Its protective potential against biochemically-produced free radicals, ROS-induced peroxidation of tissue lipid, and oxidation of protein is significantly higher than vitamin E, vitamin C, β-carotene, and a combination of vitamins C and E[68-70]. Saito et al[71] showed grape seed extract and its proanthocyanidin constituents possess a preventive effect on ethanol/hydrochloride-induced gastric injury. Physical stress has been demonstrated to associate with oxidative tissue damage, resulting in severe gastrointestinal mucosal injury in animal models. Grape seed proanthocyanidin extract mitigates gastrointestinal mucosal injury induced by acute or chronic stress via the prevention of superoxide anion production, DNA fragmentation, lipid peroxidation, and membrane microviscosity[69].
Ten-day pretreatment with proanthocyanidin-enriched grape seed extract decreased gastric lesions and gastric MDA level induced by ethanol more effectively than vitamins E and C. Likewise, it diminishes aspirin-induced gastric injury and peroxidation of mucosal lipid in a similar manner to antioxidants agents and vitamins E and C[70]. Dietary supplementation of grape seed proanthocyanidin (89.3%) for 2 wk protected against gastric ulcer induced by stress (water-immersion restraint) and reduced the secretion of somatostatin, gastrin, and histamine significantly. The protective effect of dietary grape proanthocyanidin is mediated by hormonal mechanisms, including anti-gastrin and anti-histaminic potential, as well as stimulation of PGE2 secretion. Iwasaki et al[72] reported that inhibition of neutrophil infiltration to gastric mucosal tissue (reduction of gastric MPO) and enhancement of enzymatic antioxidant defense (e.g., SOD) are among the main contributors of dietary grape proanthocyanidin in the prevention of peptic ulcer.
Grape extract (seed and skin) showed antibacterial effect on both cagA-negative and cagA-positive H. pylori clinical strains, in vitro[73]. It also inhibits H. pylori attachment to gastric cells, which is considered to be a key step in the colonization and virulence of this bacterium. Moreover, dietary supplementation of grape polyphenols mitigated H. pylori infection associated gastritis via the reduction of lymphocyte, plasma cell, eosinophil, and neutrophil infiltration in the lamina propria, as well as alleviating inflammatory response in infected mice. This therapeutic function of dietary grape polyphenols is mediated through the suppression of pro-inflammatory cytokines IL-1β, TNF-α, and IFN-γ, which are elevated by H. pylori infection. Likewise, polyphenols promoted H. pylori eradication from the gastric tissue of infected mice[74,75].
Green tea polyphenols
Green tea (Camellia sinensis) is an evergreen shrub which has been used for approximately 4000 years. Green tea is the most popular beverage in the world after water and encompasses a wide range of polyphenols, including catechin, epicatechin, epicatechin gallate, epigallocatechin, and epigallocatechin gallate[76]. Epigallocatechin gallate (EGCG), the main green tea polyphenol (about 40% of tea polyphenols), possesses anti-diabetic, anti-cancer, and cardioprotective functions. Various studies indicated EGCG as the greatest antioxidant and anti-inflammatory substance of tea polyphenols, as well as having a higher antioxidant effect than vitamins C and E, and promising anti-inflammatory activity[77]. Numerous studies have shown that the positive anti-ulcer action of EGCG is based on its ability to alleviate the production of key inflammatory mediators within the pathological condition of ulceration. Adhikary et al[78] evaluated the role of COX-independent pro-inflammatory pathway in the gastroprotective effects of EGCG and showed that this polyphenol significantly reduces neutrophil infiltration and MPO activity. MPO is an enzyme produced by activated neutrophils which has strong pro-oxidative and pro-inflammatory activities.
iNOS-derived NO is involved in oxidative epithelium damage and the exacerbation of ulceration. The ability of EGCG in the reduction of NO level and altering the iNOS/ eNOS ratio at the gastric tissue has an important role in its therapeutic benefit for peptic ulcer. Moreover, EGCG inhibits neutrophil-endothelial cell interactions via the suppression of soluble E-selectin (sE-selectin) and P-selectin (sP-selectin) levels in damaged gastric tissue. Consistent with this, indomethacin up-regulates soluble ICAM-1 (sICAM-1) and VCAM-1 (sVCAM-1) in gastric tissue, which is significantly modulated by EGCG. Thus, this polyphenol suppresses both the early rolling phase through the selectins down-regulation as well as the late firm adhesion phase through the CAMs down-regulation[78,79].
During the pathological process of ulceration, the pro-inflammatory Th1 cytokines are elevated and the anti-inflammatory Th2 cytokines are lessened. This cytokine imbalance caused stimulation of oxidative stress and up-regulation of adhesion molecules. EGCG modulates the cytokine imbalance and reinforces anti-inflammatory cytokines, leading to the promotion of ulcer healing. Such anti-inflammatory potential of EGCG in damaged gastric tissue via the modulating COX-independent pro-inflammatory pathway is significantly more potent than the standard drug omeprazole[78,80].
Given the importance of COX isoforms and PGs in the pathogenesis of ulceration, the effect of EGCG on these inflammatory mediators was evaluated by Adhikary et al[80]. Treatment with this polyphenol in indomethacin-induced ulcerated animals enhanced the levels of COX enzyme, as well as PGE level, significantly (P < 0.001). It also improves mucin content in gastric tissue, causing enhancement of mucosal membrane protection. Oxidative damage in gastric tissue, which possesses a strong effect in initializing and exacerbating peptic ulcers, is modulated by EGCG; it enhances thiol defense, suppresses lipid peroxidation, and protein oxidation in ulcerated gastric tissue. Mei et al[81] reported that 16-wk intake of tea polyphenol as a dietary supplement in drinking water protects against gastric lesions induced by N-methyl-N’-nitro-N-nitrosoguanidine with inhibition of apoptosis and cell proliferation in an animal model.
Inhibitory function on H. pylori infection is another pharmacological mechanism of tea polyphenol for the management of peptic ulcer[82]. EGCG alleviate H. pylori-associated gastropathy, which is mediated by the TLR4 signaling pathway. TLR-4 is activated by the lipopolysaccharide of H. pylori and plays a key role in initializing and adapting the immune reaction to this pathogen. Lee et al[83] showed EGCG inhibits glycosylation of TLR-4. H. pylori induced host intracellular signaling and its subsequent pathways. EGCG suppresses NF-κB-DNA binding, which is a major transcription mediator involved in inflammation and redox. Likewise, it prevents phosphorylation of ERK1/2, one of three major subfamilies of MAPK cellular inflammatory signaling. In addition, tea polyphenols significantly diminish gastric mucosal cytotoxicity and DNA damage of epithelial cells induced by H. pylori infection. They also have inhibitory effects against urease enzyme, a virulence factor of H. pylori which is vital for its colonization and establishment in gastric mucosa[84].
Anti-secretory potential of catechin derivatives have a crucial role in green tea’s protective ability against peptic ulcer, which is mediated by the inhibition of gastric H+, K+-ATPase activity. Murakami et al[85] demonstrated (+)-catechin, (-)-epicatechin, (-)-epicatechin gallate, (-)-epigallocatechin, and (-)-epigallocatechin gallate significantly inhibit this gastric enzyme. Crude catechin, as one of the main phenolics in green tea, possesses a remarkable preventive effect on stress-associated peptic ulcer. Dietary supplementation of crude catechins [52.6% (w/w) epigallocatechin gallate and 16.7% (w/w) epicatechin gallate] for 14 d significantly alleviated water immersion restraint stress-induced gastric mucosal lesions in an animal model. Endocrine parameters are involved in the gastroprotective effect of dietary catechin due to the reduction of somatostatin, gastrin, and histamine levels[86]. In addition, dietary consumption of green tea reduced the incidence of H. pylori infection in patients with peptic ulcer (Table 1)[87].
Pomegranate polyphenols and ellagic acid
Pomegranate (Punica granatum L., Punicaceae) is a popular fruit that is native to Persia, but grown and consumed across the world[88]. Polyphenolic compounds are the main constituent of this fruit in which ellagitannins and anthocyanins are represented as the predominant polyphenols. Degradation of ellagitannins does not occur in the presence of hydrochloric acid and the gastric enzymes of the stomach, indicating its stability under stomach conditions. In addition to ellagitannins, anthocyanins, as another main polyphenol group of pomegranates, are quite stable under gastric acidic conditions. Pomegranate anthocyanins contain 3,5-diglucosides and 3-glucosides of cyanidin, delphinidin, and pelargonidin[89,90].
Pomegranate polyphenols possess various therapeutic beneficial effects, such as neuroprotective, anti-colitis, hypoglycemic, cardiovascular protective, antiproliferative, apoptotic, anti-cancer potential (especially against colon, prostate, and breast cancer), hypolipidemic, hypocholesterolemic, and anti-atherosclerosis effects, as well as a reduction of the LDL/HDL ratio. A wide variety of polyphenol-enriched pomegranate products have commonly been introduced into the pharmaceutical market and consumed as a beneficial complementary health drug. Various investigations support the remarkable antioxidant and anti-inflammatory properties of pomegranate polyphenols[91-93]. Ajaikumar et al[94] showed polyphenol enriched fruit rind extract prevents gastric injury and necrosis induced by ethanol in animal models. Ethanol-associated gastric mucosal injury is mediated by ROS, in addition to decreasing endogenous antioxidants, leading to the endothelial layer becoming more prone to oxidative damage. Pomegranate extract possess the ability to scavenge the ROS formation, as well as reinforce the performance of antioxidant enzymes and proteins, including SOD, CAT, GSH, and GPx. Likewise, oral administration of aspirin (400 mg/kg) in fasting animals caused severe gastric damage and intraluminal bleeding, which was completely alleviated by pretreatment with pomegranate polyphenolic extract[94].
Ellagic acid, a phenolic lactone compound, is the main in vivo hydrolysis product of pomegranate polyphenols, and possesses anti-inflammatory, antioxidant, hepatoprotective, and anti-mutagenic effects. In addition, intestinal microbiota metabolizes the most active pomegranate phenolics (ellagitannins) to urolithins, which are subsequently absorbed and reach different tissue. Urolithins have an anti-inflammatory function (i.e., suppressing the inflammatory response, pro-inflammatory cytokines expression, and inflammatory cells infiltration), which acts through the MAPK intracellular signaling pathway[95].
Beserra et al[96] reported the gastroprotective effect of ellagic acid on ethanol-induced ulcers was attributed to the enhancement of endogenous NO production, in addition to anti-oxidative damage potential, by restoring depleted non-protein sulfhydryls and suppressing the pro-inflammatory cytokine TNF-α. Ellagic acid also reduces gastric ulcer and bleeding induced by indomethacin in which suppressing leukotriene (LT)-B4 expression is a key molecular factor. It showed remarkable healing properties in chronic gastric ulcer associated with acetic acid as well, which is due to the mitigation of the pro-inflammatory cytokines INF-γ, IL-4, IL-6, and TNF-α.
Pretreatment with ellagic acid prevents hemorrhagic lesions induced by stomach ischemia, followed by reperfusion, which is considered oxidative-associated mucosal damage. The preventive potential of this oxidative mucosal injury was more effective than the antioxidant enzyme SOD. Iino et al[97] exhibited that free radical scavenging potential, as well as inhibiting lipid peroxidation, has a pivotal role in the protective effect of ellagic acid. Ellagic acid also protects gastric tissue against injury and damage of mucosal blood flow induced by stomach ischemia followed by ammonia (NH4OH), which is mediated by replenishing anti-oxidative performance[97,98]. Murakami et al[99] demonstrated ip administration of ellagic acid mitigates stress-induced gastric lesions and suppresses acid secretion significantly. Anti-secretory activity of ellagic acid is mediated by lowering gastric H+, K+-ATPase activity.
Polyphenolic extract of peel and fruit rind has a strong growth inhibitory effect on H. pylori clinical strains in vitro, which was comparable with metronidazole. Investigations revealed that pomegranate extract possess both bacteriostatic and bactericidal effects, as well as possessing the potential to modulate H. pylori cell surface hydrophobicity[100,101].
Quercetin
Quercetin (3,5,7,3′,4′-pentahydroxy flavone) is a flavone present in a wide variety of fruits and vegetables, and is the major flavonoid in the human diet. The daily intake of this phenolic compound in a healthy diet has been estimated as 5-40 mg/d[102]. Quercetin is one of the most potent scavengers of free radicals (e.g., nitrogen radicals, hydroxyl, and superoxide) and is more active than the well-known antioxidants vitamins E and C, which are naturally present in glycoside form; 3-rhamnosylquercetin (quercitrin) or 3-O-rhamnosyl-glucosyl-quercetin (rutin)[102,103]. This phenolic compound possesses a wide range of positive biological activities, such as: cardioprotective, anti-atherosclerotic, hypotensive, anti-diabetic, anti-tumor, anti-proliferative, and anti-metastasis effects[102]. Bioavailability of quercetin depends on the type of glycosides present in various food sources. Subsequent to intake of a balanced diet, total amount of quercetin in plasma is less than 100 nmol/L. However, the dietary supplementation of quercetin can improve the plasma level (i.e., 28 d intake of its supplementation at 1 g/d reaches the plasma concentrations to 1.5 μmol/L)[104,105]. It is worth mentioning that the absorption of quercetin glycosides is also influenced by gut microflora, which metabolizes to the locally active aglycone forms[104,105]. Various experimental investigations have studied the therapeutic benefits of quercetin and its derivatives on models of peptic ulcer disease. Its antioxidant and anti-inflammatory properties are contributing factors to therapeutic efficacy for peptic ulcers[104-106].
Accumulating evidence suggests that ethanol-associated ROS lead to disruption of cellular function and vital cell constituents, and produces toxic agents such as lipid hydroperoxides, lipid peroxyl radicals, and other lipid fragmentations. These toxic agents disturb the mucosal and interstitial matrix, causing gastric injury, which is mitigated by pretreatment with oral administration of quercetin. This phenolic substance lowers the tissue level of thiobarbituric acid reactive substances, indicating the inhibitory effect on lipid peroxidation. It also suppresses free radical-associated damaging of proteins and the production of protein carbonyl content significantly. Its ability to lessen histamine levels as the final chemo-stimulant of gastric secretion reveals its anti-secretory potential in gastroprotective function. Quercetin also elevates non-enzymatic and enzymatic antioxidant agents (protein sulfhydryl, SOD, and CAT). It also inhibits neutrophil infiltration and the gastric tissue level of MPO[9,106].
Min et al[107] reported quercetin-3-O-β-D-glucuronopyranoside prevents indomethacin-induced gastric ulcer dose-dependency. It modulates gastric acid secretion via the reduction of gastric volumes and total acidity. It also lessens the level of end-product lipid peroxidation, indicating the protective role of this polyphenol against indomethacin-associated oxidative mucosal stress. The gastroprotective, anti-secretory, and anti-oxidative effect of quercetin-3-O-β-D- glucopyranoside was significantly higher than that of quercetin.
Rutin is a quercetin glycoside with strong radical scavenging, anti-inflammatory, and vasoactive activity. It has been reported to possess cytoprotective and gastroprotective effects in animal models of gastroduodenal ulcer, including restraint stress, ethanol, and reserpine[108]. The protective potential of rutin against absolute ethanol-associated gastric mucosal ulceration and necrosis is due to its anti-lipoperoxidant effect, as well as an improvement in enzymatic antioxidant performance[108]. Likewise, mitigating gastric MPO activity, which is attributed to neutrophil infiltration, elevation of nitrite/nitrate which shows NO production, and enhanced activity of gastric antioxidant enzymes (SOD) and proteins (GSH), is among the main protective mechanisms of rutin on indomethacin-associated degenerative mucosal damage[109].
Olaleye et al[110] compared the healing potential of cimetidine and a low dose of rutin in animal experimental models of gastric ulcer induced by acetic acid, stress, or ethanol. Post-treatment with rutin in low doses diminished ulcer severity in all animal models of mucosal injury significantly, and showed higher therapeutic potential than cimetidine (300 mg/kg) in the management of gastric damage. It is reported that an increase in the antioxidant activity of vitamin C and GPx and inhibiting peroxidation of cellular lipid (reduction of MDA) is a crucial contributor in the current therapeutic action of rutin.
RESVERATROL
Resveratrol (3,5,4’-trihydroxy-trans-stilbene), a natural phytoalexin from the stilbenes subgroup, is isolated from berries, grape skin, and peanuts[111]. Stilbenes are phytoalexins, a plant stress-inducible metabolite, which is only produced in response to pathogen attacks as a part of plant’s defensive mechanism. Resveratrol is a natural dietary phenolic compound with various reported therapeutic benefits, including anti-carcinogenic, chemopreventive, and anti-aging effects, and is useful for the management of cardiovascular diseases, cancer, and neurodegenerative diseases[112]. It has been reported that resveratrol has beneficial effects in various biological tissues, despite its rapid metabolism and elimination. In addition, a wide range of investigations has been executed to enhance resveratrol bioavailability to overcome its normally poor bioavailability. These strategies are generally based on encapsulations, inhibition of CYPs by means of specific inhibitors such as piperine, and modifications of resveratrol’s structure[113]. The potential of resveratrol in the management of gastric and intestinal injury and inflammation, due to its strong antioxidant, anti-oxidative stress, free radical scavenging, and anti-inflammatory activities, is widely evaluated[112,114].
The protective effect of resveratrol in the models of peptic ulcer is reasonably well-reported, with most investigations being based on its potential to suppress the production of key inflammatory mediators. In vitro studies demonstrated that resveratrol inhibits the expression and activation of nuclear factors (NF-κB) and intracellular transcriptional enzymes (MAPKs)[114]. In an experimental study by Solmaz et al[115], resveratrol exhibited both protective and therapeutic effects in an animal model of gastric ulcer induced by acetic acid, which was due to attenuating gastric MPO activity (inhibition of leukocyte infiltration), MDA (suppressing tissue lipid peroxidation), elevating collagen content, and restoring depleted GSH. Suppressing the key pro-inflammatory agent TNF-α has an essential role in its therapeutic function in gastric tissue. This polyphenol also diminished the level of lucigenin- and luminol-enhanced chemiluminescence, indicating its significant suppression on intracellular and extracellular oxidative events in gastric mucosal tissue.
In another experimental study executed by Li et al[116], this polyphenol protects against gastric injury induced by acidified ethanol, which is attributed to the enhancement of dimethylarginine dimethylaminohydrolase (DDAH) activity and the subsequent reduction of asymmetric dimethylarginine (ADMA) content, resulting in blocked methylarginines accumulation, the induction of NO synthesis, and vasodilation in mucosal tissue. Thus, augmentation of gastric NO production is among the most important abilities of resveratrol in the protection of gastric mucosa. However, Dey et al[117] reported that resveratrol has a biphasic protective effect. This polyphenol, in low doses, showed preventive potential on indomethacin-induced gastric ulcer due to the enhancement of eNOS expression and, in higher doses, blocked PGE2 synthesis in gastric tissue, which is mediated by inhibiting COX-1 enzyme activity and expression.
It is now well-recognized that resveratrol has an excellent antibacterial effect against H. pylori infection. Resveratrol inhibits the virulence factor of H. pylori and urease enzyme activity significantly. The inhibitory property of resveratrol on this enzyme is non-competitive and dose-dependent[118,119]. H. pylori infection stimulates pro-inflammatory mediators, causing disturbance of surface epithelial cell integrity, irregularity of the luminal border, damage to the gastric microvilli, and inducing mucosal vacuolation. Among the pro-inflammatory cytokines, IL-8 possesses an obvious effect in the pathogenesis of H. pylori diseases by activating neutrophils and inducing cellular chemoattraction. Resveratrol prevents H. pylori-induced gastric mucosal damage and gastritis by suppressing the secretion of IL-8 from H. pylori-infected cells. H. pylori infection associates with ROS generation, oxidative DNA damage, and epithelial proliferation. Pretreatment with this polyphenol mitigates intracellular ROS generation and oxidative DNA fragmentation induced by H. pylori infection dose-dependently in gastric epithelial cell. The interaction of H. pylori with gastric epithelial cells is associated with morphological changes that result in the dysregulation of host cell functions, cell motility, and the “hummingbird phenomenon” (altered migration, motility, and adhesion of the gastric endothelial cells), leading to gastropathy. Cellular studies showed that H. pylori-initiated gastric morphological changes were noticeably suppressed by resveratrol[21,118,119].
CONCLUSION
Peptic ulcer disease has been a major threat to human health over the past two centuries. Despite medical advances, the management of peptic ulcer and its relevant complications remains a medical challenge, due to its high morbidity and mortality[1,4,93]. Therefore, there is a growing interest in dietary factors as supplements for the prevention or remission of gastritis and gastrointestinal ulcers.
Polyphenols, secondary plant metabolites ubiquitously present in vegetables and fruits, are progressively viewed as natural dietary ingredients which must be present in a balanced diet, owing to their wide range of biological effects. Natural polyphenols have been reported to possess numerous beneficial roles in the gastrointestinal tract, including antispasmodic, anti-colitis, anti-secretory, anti-diarrheal, anti-ulcer, and anti-oxidative stress properties[26-28]. Additionally, it is possible that the therapeutic benefits of various traditional and complementary medicines in the treatment of peptic ulcer are related to the presence of polyphenol constituents[10,120].
An accumulating body of evidence suggests that, among a wide range of natural molecules, dietary polyphenols with multiple biological mechanisms of action can play a pivotal role in the management of gastric and intestinal ulcers. Figure 1 illustrates the chemical structure of the most relevant dietary polyphenols with a potential role for the management of peptic ulcer. Recently, several distinct molecular mechanisms that possibly commensurate with peptic ulcer pathogenesis have been demonstrated. A growing body of in vitro and preclinical studies suggests a beneficial role of dietary polyphenols in peptic ulcer, in terms of their well-established intracellular and molecular pharmacological mechanisms of action. The ulcer healing process is a complex one that includes a combination of injury retraction and re-epithelization. The complex sequence of events in the ulcer healing process needs a high degree of coordination modulated by significant factors, of which the growth factors and PGs are considered a key contributors that are involved in the essential cell functions of tissue healing containing tissue restoration and angiogenesis[39,120]. Improving cytoprotection, re-epithelialization, neovascularization, and angiogenesis, which are mediated by the up-regulation of PGs, tissue growth factors, and vWF/ factor VIII complex, along with the down-regulation of anti-angiogenic factors, possess a pivotal role in the anti-ulcer potential of dietary polyphenols. In addition, polyphenols suppress vascular permeability, leucocyte infiltration, and leukocyte-endothelium interaction mediated by the down-regulation of cellular and intercellular adhesion agents, including CAMs and selectins. Dietary polyphenols mitigate inflammatory reactions and down-regulate pro-inflammatory cytokines such as TNF-α and IL-1β within mucosal ulcers by inhibiting intracellular signaling pathways of the inflammatory process (JNK, ERK, and MAPK), as well as modulating intracellular transcriptional factors. Regarding the important role of oxidative stress in ulceration, the remarkable ability of polyphenols in the suppression of oxidative mucosal damage and the scavenging of gastric free radicals mediated by enzymatic and non-enzymatic antioxidant substances are among its main anti-ulcer mechanisms. It has been reported that antacid and anti-secretory effects mediated by reducing H+, K+-ATPase activity and antihistaminic function, along with the enhancement of mucosal defensive agents (mucin and hexosamine), are among the other polyphenol abilities in the management of gastric ulceration. Additionally, modulating cellular events and functions leading to the enhancement of tissue eNOS-derived NO level possesses a crucial role in the management of peptic ulcer. Figure 2 illustrates the potential cellular mechanisms of dietary polyphenols as preventive or therapeutic factors in the management of peptic ulcer. It is worthwhile mentioning that Figure 2 is elicited from the pharmacological mechanisms demonstrated in the scientific literature with different dietary polyphenols, and not all of these mechanisms apply to each dietary polyphenol.
Figure 1.
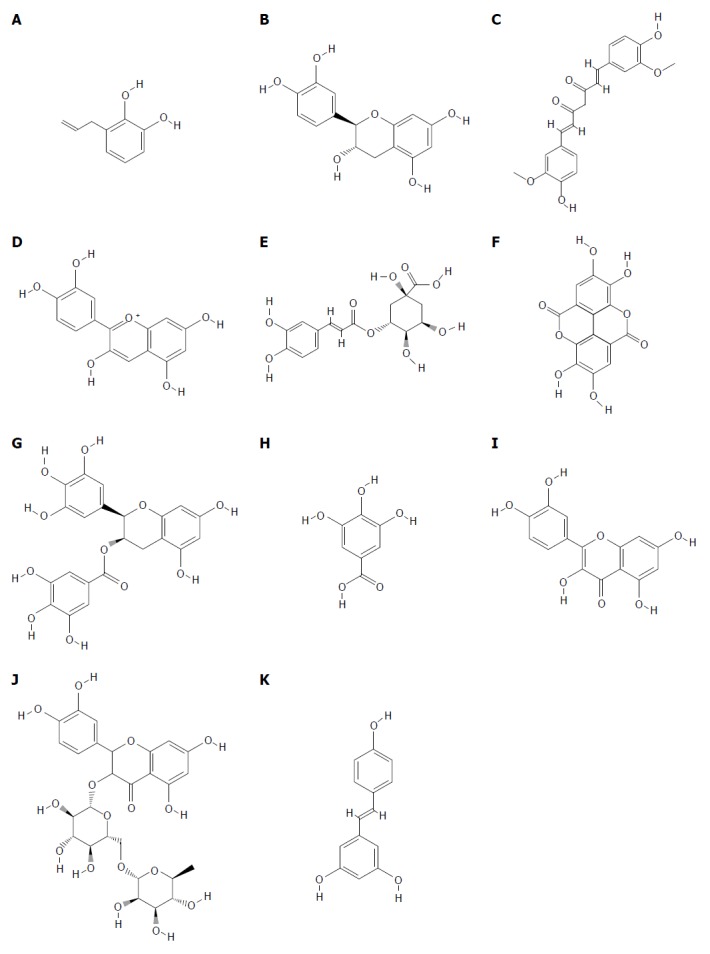
Structure of the most relevant polyphenol with a potential role for the management of peptic ulcer. A: Allylpyrocatechol; B: Catechin; C: Curcumin; D: Cyanidin; E: Chlorogenic acid; F: Ellagic acid; G: Epigallocatechin gallate; H: Gallic acid; I: Quercetin; J: Rutin; K: Resveratrol.
Figure 2.
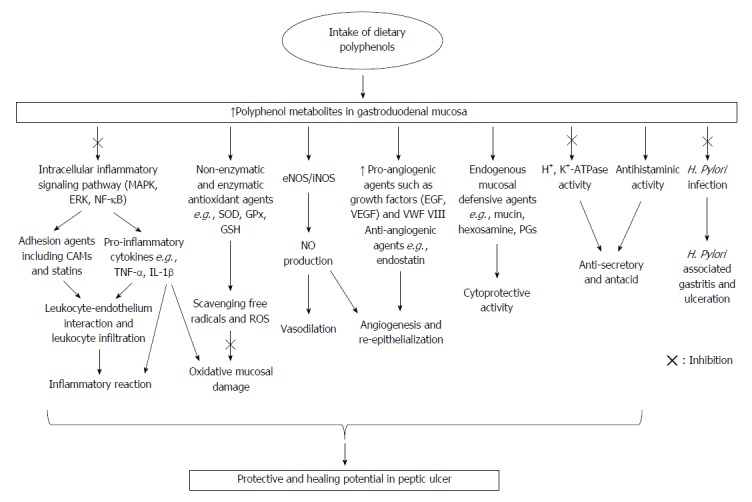
Potential cellular mechanisms of dietary polyphenols as preventive or therapeutic factors in the management of peptic ulcer. IL: Interleukin; NF: Nuclear factor; MAPK: Mitogen-activated protein kinases; ERK: Extracellular signal-regulated kinase; TNF: Tumor necrosis factor; SOD: Superoxide dismutase; GSH: Glutathione; GPx: Glutathione peroxidase; EGF: Epidermal growth factor; VEGF: Vascular endothelial growth factor; NO: Nitric oxide; vWF: Von Willebrand factor.
Due to the increase in antibacterial resistance, a diet-based treatment against pathogens infection has an important role in the management of various diseases. The accumulated data confirms that H. pylori infection can lead to severe gastritis and gastroduodenal ulceration, and its eradication is considered the therapeutic approach for peptic ulcer. Hence, host nutritional factors are thought to play a significant role in the management of H. pylori colonization and subsequent diseases[19,121]. A growing body of evidence shows the strong therapeutic potential of dietary polyphenols on H. pylori infection and its associated disease, which is mediated by inhibiting the activity of vital enzymes of this pathogen like urease and shikimate dehydrogenase, preventing its adhesion and vacuolation in gastric tissue, in addition to blocking H. pylori-induced morphological changes of gastric epithelial cells, causing dysregulation of host cell functions. Polyphenols prevent H. pylori-initiated inflammatory response in gastric tissue via suppressing pro-inflammatory cytokines TNF-α, IFN-γ, and IL-8 and cellular and nuclear inflammatory signaling (MAPK), as well as modulating intracellular transduction and transcription pathways (TLR4). In addition, dietary polyphenols protect against H. pylori associated oxidative mucosal damage and ROS generation. Figure 3 summarizes the possible biological mechanisms of dietary polyphenols in the management of H. pylori-associated gastritis and gastroduodenal ulceration.
Figure 3.
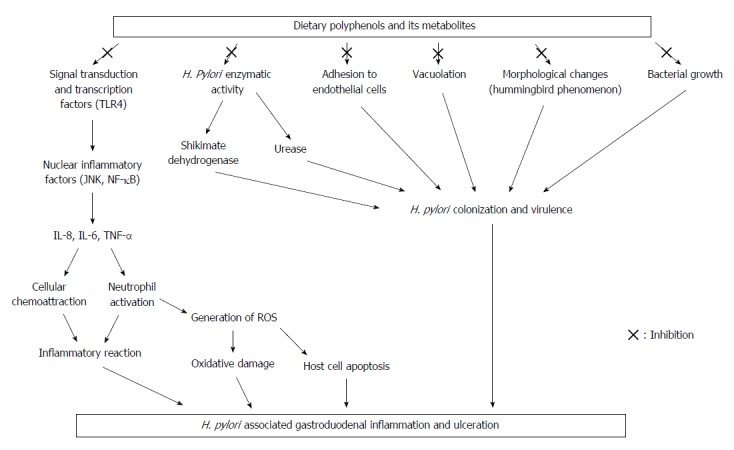
Summary of the possible biological mechanisms of dietary polyphenols in the management of Helicobacter pylori associated gastritis and gastroduodenal ulceration. TLR4: Toll like receptor; IL: Interleukin; ROS: Reactive oxygen species; TNF: Tumor necrosis factor; JNK: c-Jun N-terminal kinase; NF: Nuclear factor.
The current paper calls attention to a group of dietary natural phytochemicals, polyphenols, with a growing body of evidence regarding their beneficial association in human health. The evidence suggests that polyphenols, as non-essential dietary supplements, probably possess a pivotal role in improving the inflammatory process, oxidative reactions, degenerative mucosal events, and recurrence gastroduodenal ulceration. A wide range of preclinical investigations support the therapeutic benefits of dietary natural polyphenols in the management of peptic ulcer, although there are few clinical trials evaluating the efficacy of dietary polyphenols in peptic ulcer. Table 1 shows the clinical evidence of dietary polyphenols in the treatment of peptic ulcers. Among the dietary polyphenols with therapeutic potential for managing peptic ulcer, curcumin has the higher level of evidence, and there are different clinical studies evaluating its beneficial effect in gastrointestinal ulcers and H. pylori infection. Further well-designed clinical trials are necessary to evaluate the role of dietary polyphenols as promising preventive and therapeutic dietary factors in the management of peptic ulcer. In addition, our review exhibited that further in vitro and preclinical investigations are mandatory to understand the absorption, metabolism, bioavailability, bioefficacy, and cellular mechanisms of dietary polyphenols.
In conclusion, the administration of a significant content of dietary polyphenols in the human diet or as dietary supplementation along with conventional treatment can resulted in perfect prevention and treatment for peptic ulcer. Regarding the role of NSAID intake in the pathogenesis of ulceration, dietary polyphenols can be considered as adjuvants with NSAID-therapy for the protection of peptic ulcer. It is suggested that, according to the hierarchy pyramid of evidence-based medicine, well-designed controlled clinical trials must be performed in order to recognize higher levels of evidence for the confirmation of dietary polyphenol efficacy and safety in the management of peptic ulcer.
Footnotes
Supported by National Elites Foundation of Iran (partly).
Conflict-of-interest: The authors have no conflicts of interest.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: November 26, 2014
First decision: February 10, 2015
Article in press: April 3, 2015
P- Reviewer: Baik GH, Guadagni S S- Editor: Qi Y L- Editor: Rutherford A E- Editor: Wang CH
References
- 1.Malfertheiner P, Chan FK, McColl KE. Peptic ulcer disease. Lancet. 2009;374:1449–1461. doi: 10.1016/S0140-6736(09)60938-7. [DOI] [PubMed] [Google Scholar]
- 2.Sumbul S, Ahmad MA, Mohd A, Mohd A. Role of phenolic compounds in peptic ulcer: An overview. J Pharm Bioallied Sci. 2011;3:361–367. doi: 10.4103/0975-7406.84437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.O’Malley P. Gastric ulcers and GERD: the new “plagues” of the 21st century update for the clinical nurse specialist. Clin Nurse Spec. 2003;17:286–289. doi: 10.1097/00002800-200311000-00008. [DOI] [PubMed] [Google Scholar]
- 4.Borrelli F, Izzo AA. The plant kingdom as a source of anti-ulcer remedies. Phytother Res. 2000;14:581–591. doi: 10.1002/1099-1573(200012)14:8<581::aid-ptr776>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 5.Repetto MG, Llesuy SF. Antioxidant properties of natural compounds used in popular medicine for gastric ulcers. Braz J Med Biol Res. 2002;35:523–534. doi: 10.1590/s0100-879x2002000500003. [DOI] [PubMed] [Google Scholar]
- 6.Cryer B. Mucosal defense and repair. Role of prostaglandins in the stomach and duodenum. Gastroenterol Clin North Am. 2001;30:877–894, v-vi. doi: 10.1016/s0889-8553(05)70218-1. [DOI] [PubMed] [Google Scholar]
- 7.Bandyopadhyay D, Biswas K, Bhattacharyya M, Reiter RJ, Banerjee RK. Gastric toxicity and mucosal ulceration induced by oxygen-derived reactive species: protection by melatonin. Curr Mol Med. 2001;1:501–513. doi: 10.2174/1566524013363483. [DOI] [PubMed] [Google Scholar]
- 8.Sen S, Asokkumar K, Umamaheswari M, Sivashanmugam AT, Subhadradevi V. Antiulcerogenic effect of gallic Acid in rats and its effect on oxidant and antioxidant parameters in stomach tissue. Indian J Pharm Sci. 2013;75:149–155. [PMC free article] [PubMed] [Google Scholar]
- 9.Tandon R, Khanna HD, Dorababu M, Goel RK. Oxidative stress and antioxidants status in peptic ulcer and gastric carcinoma. Indian J Physiol Pharmacol. 2004;48:115–118. [PubMed] [Google Scholar]
- 10.Farzaei MH, Rahimi R, Abbasabadi Z, Abdollahi M. An evidence-based review on medicinal plants used for the treatment of peptic ulcer in traditional Iranian medicine. Int J Pharmacol. 2013;9:108–124. [Google Scholar]
- 11.Arakawa T, Watanabe T, Tanigawa T, Tominaga K, Fujiwara Y, Morimoto K. Quality of ulcer healing in gastrointestinal tract: its pathophysiology and clinical relevance. World J Gastroenterol. 2012;18:4811–4822. doi: 10.3748/wjg.v18.i35.4811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Halter F, Tarnawski AS, Schmassmann A, Peskar BM. Cyclooxygenase 2-implications on maintenance of gastric mucosal integrity and ulcer healing: controversial issues and perspectives. Gut. 2001;49:443–453. doi: 10.1136/gut.49.3.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wallace JL. NSAID gastropathy and enteropathy: distinct pathogenesis likely necessitates distinct prevention strategies. Br J Pharmacol. 2012;165:67–74. doi: 10.1111/j.1476-5381.2011.01509.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Souza MH, Mota JM, Oliveira RB, Cunha FQ. Gastric damage induced by different doses of indomethacin in rats is variably affected by inhibiting iNOS or leukocyte infiltration. Inflamm Res. 2008;57:28–33. doi: 10.1007/s00011-007-7089-z. [DOI] [PubMed] [Google Scholar]
- 15.Yadav SK, Adhikary B, Chand S, Maity B, Bandyopadhyay SK, Chattopadhyay S. Molecular mechanism of indomethacin-induced gastropathy. Free Radic Biol Med. 2012;52:1175–1187. doi: 10.1016/j.freeradbiomed.2011.12.023. [DOI] [PubMed] [Google Scholar]
- 16.Langenbach R, Morham SG, Tiano HF, Loftin CD, Ghanayem BI, Chulada PC, Mahler JF, Lee CA, Goulding EH, Kluckman KD, et al. Prostaglandin synthase 1 gene disruption in mice reduces arachidonic acid-induced inflammation and indomethacin-induced gastric ulceration. Cell. 1995;83:483–492. doi: 10.1016/0092-8674(95)90126-4. [DOI] [PubMed] [Google Scholar]
- 17.Tarnawski AS, Jones MK. Inhibition of angiogenesis by NSAIDs: molecular mechanisms and clinical implications. J Mol Med (Berl) 2003;81:627–636. doi: 10.1007/s00109-003-0479-y. [DOI] [PubMed] [Google Scholar]
- 18.Bahramsoltani R, Farzaei MH, Rahimi R. Medicinal plants and their natural components as future drugs for the treatment of burn wounds: an integrative review. Arch Dermatol Res. 2014;306:601–617. doi: 10.1007/s00403-014-1474-6. [DOI] [PubMed] [Google Scholar]
- 19.Wang YC. Medicinal plant activity on Helicobacter pylori related diseases. World J Gastroenterol. 2014;20:10368–10382. doi: 10.3748/wjg.v20.i30.10368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vítor JM, Vale FF. Alternative therapies for Helicobacter pylori: probiotics and phytomedicine. FEMS Immunol Med Microbiol. 2011;63:153–164. doi: 10.1111/j.1574-695X.2011.00865.x. [DOI] [PubMed] [Google Scholar]
- 21.Zaidi SF, Ahmed K, Yamamoto T, Kondo T, Usmanghani K, Kadowaki M, Sugiyama T. Effect of resveratrol on Helicobacter pylori-induced interleukin-8 secretion, reactive oxygen species generation and morphological changes in human gastric epithelial cells. Biol Pharm Bull. 2009;32:1931–1935. doi: 10.1248/bpb.32.1931. [DOI] [PubMed] [Google Scholar]
- 22.Crabtree JE, Xiang Z, Lindley IJ, Tompkins DS, Rappuoli R, Covacci A. Induction of interleukin-8 secretion from gastric epithelial cells by a cagA negative isogenic mutant of Helicobacter pylori. J Clin Pathol. 1995;48:967–969. doi: 10.1136/jcp.48.10.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Toracchio S, Cellini L, Di Campli E, Cappello G, Malatesta MG, Ferri A, Ciccaglione AF, Grossi L, Marzio L. Role of antimicrobial susceptibility testing on efficacy of triple therapy in Helicobacter pylori eradication. Aliment Pharmacol Ther. 2000;14:1639–1643. doi: 10.1046/j.1365-2036.2000.00870.x. [DOI] [PubMed] [Google Scholar]
- 24.Miederer SE. [Will anti-ulcer drugs soon differ only in their side effects?] Fortschr Med. 1986;104:918–920. [PubMed] [Google Scholar]
- 25.Gisbert JP, Calvet X. Review article: the effectiveness of standard triple therapy for Helicobacter pylori has not changed over the last decade, but it is not good enough. Aliment Pharmacol Ther. 2011;34:1255–1268. doi: 10.1111/j.1365-2036.2011.04887.x. [DOI] [PubMed] [Google Scholar]
- 26.Daglia M. Polyphenols as antimicrobial agents. Curr Opin Biotechnol. 2012;23:174–181. doi: 10.1016/j.copbio.2011.08.007. [DOI] [PubMed] [Google Scholar]
- 27.Tsao R. Chemistry and biochemistry of dietary polyphenols. Nutrients. 2010;2:1231–1246. doi: 10.3390/nu2121231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Farzaei MH, Rahimi R, Abdollahi M. The role of dietary polyphenols in the management of inflammatory bowel disease. Curr Pharm Biotechnol. 2015;16:196–210. doi: 10.2174/1389201016666150118131704. [DOI] [PubMed] [Google Scholar]
- 29.Antolovich M, Prenzler P, Robards K, Ryan D. Sample preparation in the determination of phenolic compounds in fruits. Analyst. 2000;125:989–1009. [Google Scholar]
- 30.Farzaei MH, Khanavi M, Moghaddam G, Dolatshahi F, Rahimi R, Shams-Ardekani MR, Amin G, Hajimahmoodi M. Standardization of Tragopogon graminifolius DC. extracts based on phenolic compounds and antioxidant activity. J Chem. 2014;2014:425965. [Google Scholar]
- 31.Cardona F, Andrés-Lacueva C, Tulipani S, Tinahones FJ, Queipo-Ortuño MI. Benefits of polyphenols on gut microbiota and implications in human health. J Nutr Biochem. 2013;24:1415–1422. doi: 10.1016/j.jnutbio.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 32.Ferguson LR, Shelling AN, Browning BL, Huebner C, Petermann I. Genes, diet and inflammatory bowel disease. Mutat Res. 2007;622:70–83. doi: 10.1016/j.mrfmmm.2007.05.011. [DOI] [PubMed] [Google Scholar]
- 33.McCarty MF. Dietary nitrate and reductive polyphenols may potentiate the vascular benefit and alleviate the ulcerative risk of low-dose aspirin. Med Hypotheses. 2013;80:186–190. doi: 10.1016/j.mehy.2012.11.025. [DOI] [PubMed] [Google Scholar]
- 34.Scalbert A, Williamson G. Dietary intake and bioavailability of polyphenols. J Nutr. 2000;130:2073S–2085S. doi: 10.1093/jn/130.8.2073S. [DOI] [PubMed] [Google Scholar]
- 35.Romier B, Schneider YJ, Larondelle Y, During A. Dietary polyphenols can modulate the intestinal inflammatory response. Nutr Rev. 2009;67:363–378. doi: 10.1111/j.1753-4887.2009.00210.x. [DOI] [PubMed] [Google Scholar]
- 36.Németh K, Plumb GW, Berrin JG, Juge N, Jacob R, Naim HY, Williamson G, Swallow DM, Kroon PA. Deglycosylation by small intestinal epithelial cell beta-glucosidases is a critical step in the absorption and metabolism of dietary flavonoid glycosides in humans. Eur J Nutr. 2003;42:29–42. doi: 10.1007/s00394-003-0397-3. [DOI] [PubMed] [Google Scholar]
- 37.Yadav SK, Adhikary B, Bandyopadhyay SK, Chattopadhyay S. Inhibition of TNF-α, and NF-κB and JNK pathways accounts for the prophylactic action of the natural phenolic, allylpyrocatechol against indomethacin gastropathy. Biochim Biophys Acta. 2013;1830:3776–3786. doi: 10.1016/j.bbagen.2013.03.013. [DOI] [PubMed] [Google Scholar]
- 38.Bhattacharya S, Banerjee D, Bauri AK, Chattopadhyay S, Bandyopadhyay SK. Healing property of the Piper betel phenol, allylpyrocatechol against indomethacin-induced stomach ulceration and mechanism of action. World J Gastroenterol. 2007;13:3705–3713. doi: 10.3748/wjg.v13.i27.3705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Banerjee D, Bhattacharya S, Bandyopadhyay SK, Chattopadhyay S. Biochemical mechanism of healing activity of the natural phenolic, allylpyrocatechol against indomethacin-induced gastric ulceration in mice. Dig Dis Sci. 2008;53:2868–2877. doi: 10.1007/s10620-008-0266-2. [DOI] [PubMed] [Google Scholar]
- 40.Hyson DA. A comprehensive review of apples and apple components and their relationship to human health. Adv Nutr. 2011;2:408–420. doi: 10.3945/an.111.000513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vrhovsek U, Rigo A, Tonon D, Mattivi F. Quantitation of polyphenols in different apple varieties. J Agric Food Chem. 2004;52:6532–6538. doi: 10.1021/jf049317z. [DOI] [PubMed] [Google Scholar]
- 42.Jung M, Triebel S, Anke T, Richling E, Erkel G. Influence of apple polyphenols on inflammatory gene expression. Mol Nutr Food Res. 2009;53:1263–1280. doi: 10.1002/mnfr.200800575. [DOI] [PubMed] [Google Scholar]
- 43.Paturi G, Butts CA, Bentley-Hewitt KL, McGhie TK, Saleh ZS, McLeod A. Apple polyphenol extracts protect against aspirin-induced gastric mucosal damage in rats. Phytother Res. 2014;28:1846–1854. doi: 10.1002/ptr.5210. [DOI] [PubMed] [Google Scholar]
- 44.D’Argenio G, Mazzone G, Tuccillo C, Grandone I, Gravina AG, Graziani G, Fogliano V, Romano M. Apple polyphenol extracts prevent aspirin-induced damage to the rat gastric mucosa. Br J Nutr. 2008;100:1228–1236. doi: 10.1017/S0007114508988747. [DOI] [PubMed] [Google Scholar]
- 45.Graziani G, D’Argenio G, Tuccillo C, Loguercio C, Ritieni A, Morisco F, Del Vecchio Blanco C, Fogliano V, Romano M. Apple polyphenol extracts prevent damage to human gastric epithelial cells in vitro and to rat gastric mucosa in vivo. Gut. 2005;54:193–200. doi: 10.1136/gut.2004.046292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pastene E, Speisky H, García A, Moreno J, Troncoso M, Figueroa G. In vitro and in vivo effects of apple peel polyphenols against Helicobacter pylori. J Agric Food Chem. 2010;58:7172–7179. doi: 10.1021/jf100274g. [DOI] [PubMed] [Google Scholar]
- 47.Jurenka JS. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: a review of preclinical and clinical research. Altern Med Rev. 2009;14:141–153. [PubMed] [Google Scholar]
- 48.Vecchi Brumatti L, Marcuzzi A, Tricarico PM, Zanin V, Girardelli M, Bianco AM. Curcumin and inflammatory bowel disease: potential and limits of innovative treatments. Molecules. 2014;19:21127–21153. doi: 10.3390/molecules191221127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sivalingam N, Hanumantharaya R, Faith M, Basivireddy J, Balasubramanian KA, Jacob M. Curcumin reduces indomethacin-induced damage in the rat small intestine. J Appl Toxicol. 2007;27:551–560. doi: 10.1002/jat.1235. [DOI] [PubMed] [Google Scholar]
- 50.Chattopadhyay I, Bandyopadhyay U, Biswas K, Maity P, Banerjee RK. Indomethacin inactivates gastric peroxidase to induce reactive-oxygen-mediated gastric mucosal injury and curcumin protects it by preventing peroxidase inactivation and scavenging reactive oxygen. Free Radic Biol Med. 2006;40:1397–1408. doi: 10.1016/j.freeradbiomed.2005.12.016. [DOI] [PubMed] [Google Scholar]
- 51.Swarnakar S, Ganguly K, Kundu P, Banerjee A, Maity P, Sharma AV. Curcumin regulates expression and activity of matrix metalloproteinases 9 and 2 during prevention and healing of indomethacin-induced gastric ulcer. J Biol Chem. 2005;280:9409–9415. doi: 10.1074/jbc.M413398200. [DOI] [PubMed] [Google Scholar]
- 52.Sharma AV, Ganguly K, Paul S, Maulik N, Swarnakar S. Curcumin heals indomethacin-induced gastric ulceration by stimulation of angiogenesis and restitution of collagen fibers via VEGF and MMP-2 mediated signaling. Antioxid Redox Signal. 2012;16:351–362. doi: 10.1089/ars.2011.4232. [DOI] [PubMed] [Google Scholar]
- 53.Veillette A, Fournel M. The CD4 associated tyrosine protein kinase p56lck is positively regulated through its site of autophosphorylation. Oncogene. 1990;5:1455–1462. [PubMed] [Google Scholar]
- 54.De R, Kundu P, Swarnakar S, Ramamurthy T, Chowdhury A, Nair GB, Mukhopadhyay AK. Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrob Agents Chemother. 2009;53:1592–1597. doi: 10.1128/AAC.01242-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kositchaiwat C, Kositchaiwat S, Havanondha J. Curcuma longa Linn. in the treatment of gastric ulcer comparison to liquid antacid: a controlled clinical trial. J Med Assoc Thai. 1993;76:601–605. [PubMed] [Google Scholar]
- 56.Prucksunand C, Indrasukhsri B, Leethochawalit M, Hungspreugs K. Phase II clinical trial on effect of the long turmeric (Curcuma longa Linn) on healing of peptic ulcer. Southeast Asian J Trop Med Public Health. 2001;32:208–215. [PubMed] [Google Scholar]
- 57.Koosirirat C, Linpisarn S, Changsom D, Chawansuntati K, Wipasa J. Investigation of the anti-inflammatory effect of Curcuma longa in Helicobacter pylori-infected patients. Int Immunopharmacol. 2010;10:815–818. doi: 10.1016/j.intimp.2010.04.021. [DOI] [PubMed] [Google Scholar]
- 58.Di Mario F, Cavallaro LG, Nouvenne A, Stefani N, Cavestro GM, Iori V, Maino M, Comparato G, Fanigliulo L, Morana E, et al. A curcumin-based 1-week triple therapy for eradication of Helicobacter pylori infection: something to learn from failure? Helicobacter. 2007;12:238–243. doi: 10.1111/j.1523-5378.2007.00497.x. [DOI] [PubMed] [Google Scholar]
- 59.Ali MS, Pandit V, Jain M, Dhar KL. Mucoadhesive microparticulate drug delivery system of curcumin against Helicobacter pylori infection: Design, development and optimization. J Adv Pharm Technol Res. 2014;5:48–56. doi: 10.4103/2231-4040.126996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Roberts AT, Martin CK, Liu Z, Amen RJ, Woltering EA, Rood JC, Caruso MK, Yu Y, Xie H, Greenway FL. The safety and efficacy of a dietary herbal supplement and gallic acid for weight loss. J Med Food. 2007;10:184–188. doi: 10.1089/jmf.2006.272. [DOI] [PubMed] [Google Scholar]
- 61.Madlener S, Illmer C, Horvath Z, Saiko P, Losert A, Herbacek I, Grusch M, Elford HL, Krupitza G, Bernhaus A, et al. Gallic acid inhibits ribonucleotide reductase and cyclooxygenases in human HL-60 promyelocytic leukemia cells. Cancer Lett. 2007;245:156–162. doi: 10.1016/j.canlet.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 62.Farzaei MH, Rahimi R, Attar F, Siavoshi F, Saniee P, Hajimahmoodi M, Mirnezami T, Khanavi M. Chemical composition, antioxidant and antimicrobial activity of essential oil and extracts of Tragopogon graminifolius, a medicinal herb from Iran. Nat Prod Commun. 2014;9:121–124. [PubMed] [Google Scholar]
- 63.Pal C, Bindu S, Dey S, Alam A, Goyal M, Iqbal MS, Maity P, Adhikari SS, Bandyopadhyay U. Gallic acid prevents nonsteroidal anti-inflammatory drug-induced gastropathy in rat by blocking oxidative stress and apoptosis. Free Radic Biol Med. 2010;49:258–267. doi: 10.1016/j.freeradbiomed.2010.04.013. [DOI] [PubMed] [Google Scholar]
- 64.Bhattacharya S, Chatterjee S, Bauri A, Bandivdeker AH, Chattopadhyay S, Bandyopadhyay SK, Poduval TB. Immunopharmacological basis of the healing of indomethacin-induced gastric mucosal damage in rats by the constituents of Phyllanthus emblica. Curr Sci. 2007;93:47–53. [Google Scholar]
- 65.Chatterjee A, Chatterjee S, Biswas A, Bhattacharya S, Chattopadhyay S, Bandyopadhyay SK. Gallic Acid Enriched Fraction of Phyllanthus emblica Potentiates Indomethacin-Induced Gastric Ulcer Healing via e-NOS-Dependent Pathway. Evid Based Complement Alternat Med. 2012;2012:487380. doi: 10.1155/2012/487380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Flaten TP. A nation-wide survey of the chemical composition of drinking water in Norway. Sci Total Environ. 1991;102:35–73. doi: 10.1016/0048-9697(91)90307-z. [DOI] [PubMed] [Google Scholar]
- 67.Park YK, Park E, Kim JS, Kang MH. Daily grape juice consumption reduces oxidative DNA damage and plasma free radical levels in healthy Koreans. Mutat Res. 2003;529:77–86. doi: 10.1016/s0027-5107(03)00109-x. [DOI] [PubMed] [Google Scholar]
- 68.Bagchi D, Garg A, Krohn RL, Bagchi M, Tran MX, Stohs SJ. Oxygen free radical scavenging abilities of vitamins C and E, and a grape seed proanthocyanidin extract in vitro. Res Commun Mol Pathol Pharmacol. 1997;95:179–189. [PubMed] [Google Scholar]
- 69.Bagchi M, Milnes M, Williams C, Balmoori J, Ye X, Stohs S, Bagchi D. Acute and chronic stress-induced oxidative gastrointestinal injury in rats, and the protective ability of a novel grape seed proanthocyanidin extract. Nutr Res. 1999;19:1189–1199. [Google Scholar]
- 70.Cuevas VM, Calzado YR, Guerra YP, Yera AO, Despaigne SJ, Ferreiro RM, Quintana DC. Effects of grape seed extract, vitamin C, and vitamin e on ethanol- and aspirin-induced ulcers. Adv Pharmacol Sci. 2011;2011:740687. doi: 10.1155/2011/740687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Saito M, Hosoyama H, Ariga T, Kataoka S, Yamaji N. Antiulcer activity of grape seed extract and procyanidins. J Agric Food Chem. 1998;46:1460–1464. [Google Scholar]
- 72.Iwasaki Y, Matsui T, Arakawa Y. The protective and hormonal effects of proanthocyanidin against gastric mucosal injury in Wistar rats. J Gastroenterol. 2004;39:831–837. doi: 10.1007/s00535-004-1399-5. [DOI] [PubMed] [Google Scholar]
- 73.Martini S, D’Addario C, Braconi D, Bernardini G, Salvini L, Bonechi C, Figura N, Santucci A, Rossi C. Antibacterial activity of grape extracts on cagA-positive and -negative Helicobacter pylori clinical isolates. J Chemother. 2009;21:507–513. doi: 10.1179/joc.2009.21.5.507. [DOI] [PubMed] [Google Scholar]
- 74.Brown JC, Huang G, Haley-Zitlin V, Jiang X. Antibacterial effects of grape extracts on Helicobacter pylori. Appl Environ Microbiol. 2009;75:848–852. doi: 10.1128/AEM.01595-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Brown JC, Wang J, Kasman L, Jiang X, Haley-Zitlin V. Activities of muscadine grape skin and quercetin against Helicobacter pylori infection in mice. J Appl Microbiol. 2011;110:139–146. doi: 10.1111/j.1365-2672.2010.04870.x. [DOI] [PubMed] [Google Scholar]
- 76.Sharma VK, Bhattacharya A, Kumar A, Sharma HK. Health benefits of tea consumption. Trop J Pharm Res. 2007;6:785–792. [Google Scholar]
- 77.Yang F, Oz HS, Barve S, de Villiers WJ, McClain CJ, Varilek GW. The green tea polyphenol (-)-epigallocatechin-3-gallate blocks nuclear factor-kappa B activation by inhibiting I kappa B kinase activity in the intestinal epithelial cell line IEC-6. Mol Pharmacol. 2001;60:528–533. [PubMed] [Google Scholar]
- 78.Adhikary B, Yadav SK, Bandyopadhyay SK, Chattopadhyay S. Role of the COX-independent pathways in the ulcer-healing action of epigallocatechin gallate. Food Funct. 2011;2:338–347. doi: 10.1039/c0fo00183j. [DOI] [PubMed] [Google Scholar]
- 79.Glowinska B, Urban M, Peczynska J, Florys B. Soluble adhesion molecules (sICAM-1, sVCAM-1) and selectins (sE selectin, sP selectin, sL selectin) levels in children and adolescents with obesity, hypertension, and diabetes. Metabolism. 2005;54:1020–1026. doi: 10.1016/j.metabol.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 80.Adhikary B, Yadav SK, Bandyopadhyay SK, Chattopadhyay S. Epigallocatechin gallate accelerates healing of indomethacin-induced stomach ulcers in mice. Pharmacol Rep. 2011;63:527–536. doi: 10.1016/s1734-1140(11)70519-9. [DOI] [PubMed] [Google Scholar]
- 81.Mei Y, Wei D, Liu J. Modulation effect of tea polyphenol toward N-methyl-N’-nitro-N-nitrosoguanidine-induced precancerous gastric lesion in rats. J Nutr Biochem. 2005;16:172–177. doi: 10.1016/j.jnutbio.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 82.Ankolekar C, Johnson D, Pinto Mda S, Johnson K, Labbe R, Shetty K. Inhibitory potential of tea polyphenolics and influence of extraction time against Helicobacter pylori and lack of inhibition of beneficial lactic acid bacteria. J Med Food. 2011;14:1321–1329. doi: 10.1089/jmf.2010.0237. [DOI] [PubMed] [Google Scholar]
- 83.Lee KM, Yeo M, Choue JS, Jin JH, Park SJ, Cheong JY, Lee KJ, Kim JH, Hahm KB. Protective mechanism of epigallocatechin-3-gallate against Helicobacter pylori-induced gastric epithelial cytotoxicity via the blockage of TLR-4 signaling. Helicobacter. 2004;9:632–642. doi: 10.1111/j.1083-4389.2004.00281.x. [DOI] [PubMed] [Google Scholar]
- 84.Yee YK, Koo MW. Anti-Helicobacter pylori activity of Chinese tea: in vitro study. Aliment Pharmacol Ther. 2000;14:635–638. doi: 10.1046/j.1365-2036.2000.00747.x. [DOI] [PubMed] [Google Scholar]
- 85.Murakami S, Muramatsu M, Otomo S. Gastric H+, K(+)-ATPase inhibition by catechins. J Pharm Pharmacol. 1992;44:926–928. doi: 10.1111/j.2042-7158.1992.tb03238.x. [DOI] [PubMed] [Google Scholar]
- 86.Sato H, Matsui T, Arakawa Y. The protective effect of catechin on gastric mucosal lesions in rats, and its hormonal mechanisms. J Gastroenterol. 2002;37:106–111. doi: 10.1007/s005350200004. [DOI] [PubMed] [Google Scholar]
- 87.Boyanova L, Ilieva J, Gergova G, Vladimirov B, Nikolov R, Mitov I. Honey and green/black tea consumption may reduce the risk of Helicobacter pylori infection. Diagn Microbiol Infect Dis. 2015;82:85–86. doi: 10.1016/j.diagmicrobio.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 88.Seeram NP, Adams LS, Henning SM, Niu Y, Zhang Y, Nair MG, Heber D. In vitro antiproliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. J Nutr Biochem. 2005;16:360–367. doi: 10.1016/j.jnutbio.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 89.Gil MI, Tomás-Barberán FA, Hess-Pierce B, Holcroft DM, Kader AA. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J Agric Food Chem. 2000;48:4581–4589. doi: 10.1021/jf000404a. [DOI] [PubMed] [Google Scholar]
- 90.Pérez-Vicente A, Gil-Izquierdo A, García-Viguera C. In vitro gastrointestinal digestion study of pomegranate juice phenolic compounds, anthocyanins, and vitamin C. J Agric Food Chem. 2002;50:2308–2312. doi: 10.1021/jf0113833. [DOI] [PubMed] [Google Scholar]
- 91.Lansky EP, Newman RA. Punica granatum (pomegranate) and its potential for prevention and treatment of inflammation and cancer. J Ethnopharmacol. 2007;109:177–206. doi: 10.1016/j.jep.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 92.Colombo E, Sangiovanni E, Dell’agli M. A review on the anti-inflammatory activity of pomegranate in the gastrointestinal tract. Evid Based Complement Alternat Med. 2013;2013:247145. doi: 10.1155/2013/247145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Farzaei MH, Shams-Ardekani MR, Abbasabadi Z, Rahimi R. Scientific evaluation of edible fruits and spices used for the treatment of peptic ulcer in traditional Iranian medicine. ISRN Gastroenterol. 2013;2013:136932. doi: 10.1155/2013/136932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ajaikumar KB, Asheef M, Babu BH, Padikkala J. The inhibition of gastric mucosal injury by Punicagranatum L. (pomegranate) methanolic extract. J Ethnopharmacol. 2005;96:171–176. doi: 10.1016/j.jep.2004.09.007. [DOI] [PubMed] [Google Scholar]
- 95.González-Sarrías A, Espín JC, Tomás-Barberán FA, García-Conesa MT. Gene expression, cell cycle arrest and MAPK signalling regulation in Caco-2 cells exposed to ellagic acid and its metabolites, urolithins. Mol Nutr Food Res. 2009;53:686–698. doi: 10.1002/mnfr.200800150. [DOI] [PubMed] [Google Scholar]
- 96.Beserra AM, Calegari PI, Souza Mdo C, Dos Santos RA, Lima JC, Silva RM, Balogun SO, Martins DT. Gastroprotective and ulcer-healing mechanisms of ellagic acid in experimental rats. J Agric Food Chem. 2011;59:6957–6965. doi: 10.1021/jf2003267. [DOI] [PubMed] [Google Scholar]
- 97.Iino T, Tashima K, Umeda M, Ogawa Y, Takeeda M, Takata K, Takeuchi K. Effect of ellagic acid on gastric damage induced in ischemic rat stomachs following ammonia or reperfusion. Life Sci. 2002;70:1139–1150. doi: 10.1016/s0024-3205(01)01493-x. [DOI] [PubMed] [Google Scholar]
- 98.Suzuki H, Yanaka A, Shibahara T, Matsui H, Nakahara A, Tanaka N, Muto H, Momoi T, Uchiyama Y. Ammonia-induced apoptosis is accelerated at higher pH in gastric surface mucous cells. Am J Physiol Gastrointest Liver Physiol. 2002;283:G986–G995. doi: 10.1152/ajpgi.00482.2001. [DOI] [PubMed] [Google Scholar]
- 99.Murakami S, Isobe Y, Kijima H, Nagai H, Muramatu M, Otomo S. Inhibition of gastric H+, K(+)-ATPase and acid secretion by ellagic acid. Planta Med. 1991;57:305–308. doi: 10.1055/s-2006-960103. [DOI] [PubMed] [Google Scholar]
- 100.Hajimahmoodi M, Shams-Ardakani M, Saniee P, Siavoshi F, Mehrabani M, Hosseinzadeh H, Foroumadi P, Safavi M, Khanavi M, Akbarzadeh T, et al. In vitro antibacterial activity of some Iranian medicinal plant extracts against Helicobacter pylori. Nat Prod Res. 2011;25:1059–1066. doi: 10.1080/14786419.2010.501763. [DOI] [PubMed] [Google Scholar]
- 101.Voravuthikunchai SP, Limsuwan S, Mitchell H. Effects of Punica granatum pericarps and Quercus infectoria nutgalls on cell surface hydrophobicity and cell survival of Helicobacter pylori. J health SCI. 2006;52:154–159. [Google Scholar]
- 102.Russo M, Spagnuolo C, Tedesco I, Bilotto S, Russo GL. The flavonoid quercetin in disease prevention and therapy: facts and fancies. Biochem Pharmacol. 2012;83:6–15. doi: 10.1016/j.bcp.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 103.Butković V, Klasinc L, Bors W. Kinetic study of flavonoid reactions with stable radicals. J Agric Food Chem. 2004;52:2816–2820. doi: 10.1021/jf049880h. [DOI] [PubMed] [Google Scholar]
- 104.Manach C, Williamson G, Morand C, Scalbert A, Rémésy C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr. 2005;81:230S–242S. doi: 10.1093/ajcn/81.1.230S. [DOI] [PubMed] [Google Scholar]
- 105.Conquer JA, Maiani G, Azzini E, Raguzzini A, Holub BJ. Supplementation with quercetin markedly increases plasma quercetin concentration without effect on selected risk factors for heart disease in healthy subjects. J Nutr. 1998;128:593–597. doi: 10.1093/jn/128.3.593. [DOI] [PubMed] [Google Scholar]
- 106.Kahraman A, Erkasap N, Köken T, Serteser M, Aktepe F, Erkasap S. The antioxidative and antihistaminic properties of quercetin in ethanol-induced gastric lesions. Toxicology. 2003;183:133–142. doi: 10.1016/s0300-483x(02)00514-0. [DOI] [PubMed] [Google Scholar]
- 107.Min YS, Lee SE, Hong ST, Kim HS, Choi BC, Sim SS, Whang WK, Sohn UD. The Inhibitory Effect of Quercetin-3-O-beta-D-Glucuronopyranoside on Gastritis and Reflux Esophagitis in Rats. Korean J Physiol Pharmacol. 2009;13:295–300. doi: 10.4196/kjpp.2009.13.4.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.La Casa C, Villegas I, Alarcón de la Lastra C, Motilva V, Martín Calero MJ. Evidence for protective and antioxidant properties of rutin, a natural flavone, against ethanol induced gastric lesions. J Ethnopharmacol. 2000;71:45–53. doi: 10.1016/s0378-8741(99)00174-9. [DOI] [PubMed] [Google Scholar]
- 109.Abdel-Raheem IT. Gastroprotective effect of rutin against indomethacin-induced ulcers in rats. Basic Clin Pharmacol Toxicol. 2010;107:742–750. doi: 10.1111/j.1742-7843.2010.00568.x. [DOI] [PubMed] [Google Scholar]
- 110.Olaleye MT, Akinmoladun AC. Comparative gastroprotective effect of post-treatment with low doses of rutin and cimetidine in rats. Fundam Clin Pharmacol. 2013;27:138–145. doi: 10.1111/j.1472-8206.2011.00972.x. [DOI] [PubMed] [Google Scholar]
- 111.Yadav M, Jain S, Bhardwaj A, Nagpal R, Puniya M, Tomar R, Singh V, Parkash O, Prasad GB, Marotta F, et al. Biological and medicinal properties of grapes and their bioactive constituents: an update. J Med Food. 2009;12:473–484. doi: 10.1089/jmf.2008.0096. [DOI] [PubMed] [Google Scholar]
- 112.Tomé-Carneiro J, Larrosa M, González-Sarrías A, Tomás-Barberán FA, García-Conesa MT, Espín JC. Resveratrol and clinical trials: the crossroad from in vitro studies to human evidence. Curr Pharm Des. 2013;19:6064–6093. doi: 10.2174/13816128113199990407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Santos AC, Veiga F, Ribeiro AJ. New delivery systems to improve the bioavailability of resveratrol. Expert Opin Drug Deliv. 2011;8:973–990. doi: 10.1517/17425247.2011.581655. [DOI] [PubMed] [Google Scholar]
- 114.Panaro MA, Carofiglio V, Acquafredda A, Cavallo P, Cianciulli A. Anti-inflammatory effects of resveratrol occur via inhibition of lipopolysaccharide-induced NF-κB activation in Caco-2 and SW480 human colon cancer cells. Br J Nutr. 2012;108:1623–1632. doi: 10.1017/S0007114511007227. [DOI] [PubMed] [Google Scholar]
- 115.Solmaz A, Sener G, Cetinel S, Yüksel M, Yeğen C, Yeğen BC. Protective and therapeutic effects of resveratrol on acetic acid-induced gastric ulcer. Free Radic Res. 2009;43:594–603. doi: 10.1080/10715760902977424. [DOI] [PubMed] [Google Scholar]
- 116.Li L, Luo XJ, Liu YZ, Zhang YS, Yuan Q, Tan N, Xiang DX, Peng J. The role of the DDAH-ADMA pathway in the protective effect of resveratrol analog BTM-0512 on gastric mucosal injury. Can J Physiol Pharmacol. 2010;88:562–567. doi: 10.1139/y10-027. [DOI] [PubMed] [Google Scholar]
- 117.Dey A, Guha P, Chattopadhyay S, Bandyopadhyay SK. Biphasic activity of resveratrol on indomethacin-induced gastric ulcers. Biochem Biophys Res Commun. 2009;381:90–95. doi: 10.1016/j.bbrc.2009.02.027. [DOI] [PubMed] [Google Scholar]
- 118.Mahady GB, Pendland SL, Chadwick LR. Resveratrol and red wine extracts inhibit the growth of CagA+ strains of Helicobacter pylori in vitro. Am J Gastroenterol. 2003;98:1440–1441. doi: 10.1111/j.1572-0241.2003.07513.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Paulo L, Oleastro M, Gallardo E, Queiroz JA, Domingues F. Anti-Helicobacter pylori and urease inhibitory activities of resveratrol and red wine. Food Res Int. 2011;44:964–969. [Google Scholar]
- 120.Farzaei MH, Abbasabadi Z, Shams-Ardekani , MR , Abdollahi M, Rahimi R. A comprehensive review of plants and their active constituents with wound healing activity in traditional Iranian medicine. Wounds. 2014;26:197–206. [PubMed] [Google Scholar]
- 121.Kalali B, Mejías-Luque R, Javaheri A, Gerhard M. H. pylori virulence factors: influence on immune system and pathology. Mediators Inflamm. 2014;2014:426309. doi: 10.1155/2014/426309. [DOI] [PMC free article] [PubMed] [Google Scholar]


