Abstract
AIM: To investigate the utility of photodynamic diagnosis (PDD) using 5-aminolevulinic acid (5-ALA) to detect gastric/colorectal tumors.
METHODS: This prospective single-center study investigated inter-subject variability in patients with early-stage gastric/colorectal tumor indicated for endoscopic resection. Subjects were patients with gastric or colorectal tumors who had undergone endoscopic resection between November 2012 and November 2013. Selection criteria included age 20-80 years, either sex, and provision of informed consent. Patients were orally administered 20 mg/kg of 5-ALA enteric-coated capsules (SBI ALApromo Co., Tokyo, Japan). Administration of 5-ALA was followed by endoscopic resection of gastric or colorectal tumors, and the resected specimens were examined using a video autofluorescence processor and a fluorescence endoscope (SAFE-3000 and EB-1970AK, respectively; Pentax, Tokyo, Japan). The primary endpoint was the presence of fluorescence in tumors. Endoscopic, macroscopic, and histopathologic findings of tumors were assessed. We also evaluated adverse events of the present procedure as a secondary endpoint and examined each patient for the presence of known adverse effects of 5-ALA, namely, hematocytopenia, liver dysfunction, hypotension, nausea, and photosensitivity.
RESULTS: We enrolled 10 patients (7 men, 3 women) (n = 13 lesions: 10 gastric/3 colorectal tumors). Fluorescence was detected in 7/13 (53.8%) lesions. No significant differences in sex (male: 55.6% vs female: 50.5%, P = 1.00), age (67.1 ± 1.9 years vs 65.0 ± 2.0 years, P = 0.45), tumor color (reddish: 60.0% vs discolored: 33.3%, P = 0.56), tumor diameter (15.0 ± 2.1 mm vs 14.2 ± 2.3 mm, P = 0.80), macroscopic type (protruded: 70.0% vs depressed 0%, P = 0.07), histologic type (differentiated type: 58.3% vs 0%, P = 0.46), invasion depth (mucosal layer: 55.6% vs submucosal layer: 33.3%, P = 1.00), lymphatic invasion (present: 33.3% vs absent: 50.0%, P = 1.00), venous invasion (present: 0% vs absent: 54.5%, P = 1.00) or procedure time of endoscopic resection (36.3 ± 8.3 min vs 36.7 ± 9.0 min, P = 0.98) were observed between the patients with and without fluorescence. Fluorescence detection rate tended to be high for elevated lesions. Liver dysfunction developed in 4/10 (40.0%) patients. The extent of the liver dysfunction was a slight increase in transaminases and total bilirubin levels, which spontaneously improved in the patients. None of the patients developed photosensitivity.
CONCLUSION: Results of this preliminary study suggest the utility of PDD using 5-ALA for screening of gastric and colorectal cancers.
Keywords: Photodynamic diagnosis; 5-aminolevulinic acid; Protoporphyrin IX, Gastric tumor; Colorectal tumor
Core tip: We took advantage of the high tumor specificity of fluorescent protoporphyrin IX to conduct this preliminary study to apply photodynamic diagnosis (PDD) using 5-aminolevulinic acid (5-ALA) for gastrointestinal endoscopy. This study of PDD using 5-ALA showed the presence of fluorescence in early gastric cancers. This is the first study using enteric capsules for administration of 5-ALA. The findings in this study suggest that PDD with the oral administration of 5-ALA is useful and safe for the screening of early gastric and colorectal cancers.
INTRODUCTION
The detection rate of early-stage gastric and colorectal tumors continues to improve owing to technological advances in endoscopy, yet some tumors are still being missed[1]. Image-enhanced endoscopy (IEE) was recently developed to detect lesions by changing the wavelength of irradiated light and by processing the acquired endoscopic images, and the utility of IEE in the clinical setting is thus anticipated[2]. However, this technique has not been sufficient to improve the detection rate of gastric and colorectal tumors.
The clinical usefulness of photodynamic diagnosis (PDD) using 5-aminolevulinic acid (5-ALA) has been reported[3]. 5-ALA is a precursor of fluorescence-emitting protoporphyrin IX (PpIX), which accumulates specifically in tumor cells and emits fluorescence when the tumor is irradiated with excitation light[4]. This property of 5-ALA might improve the endoscopic diagnosis of gastric and colorectal tumors.
In this preliminary study, we investigated the utility of 5-ALA using PDD in the detection of gastric and colorectal tumors. After oral administration of 5-ALA, we examined fluorescence in the endoscopically resected tumors. A limitation of this study is that we examined the resected specimens ex vivo.
MATERIALS AND METHODS
Study design
This was a prospective single-center investigation of inter-subject variability. A flow chart of the study is shown in Figure 1. Subjects provided their informed consent on hospital admission, and the research was carried out in accordance with the Declaration of Helsinki. Administration of 5-ALA was followed by endoscopic resection of gastric or colorectal tumors, and the resected specimens were examined using a video autofluorescence processor and a fluorescence endoscope (SAFE-3000 and EB-1970AK, respectively; Pentax, Tokyo, Japan). The processor emits excitation light at 408 nm, which is filtered out during imaging. This study was approved by the Institutional Review Board of Yamaguchi University Hospital, and this clinical trial was registered in the UMIN clinical trial registry (UMIN ID 000010086). The authors have no conflict of interest directly relevant to the content of this article.
Figure 1.
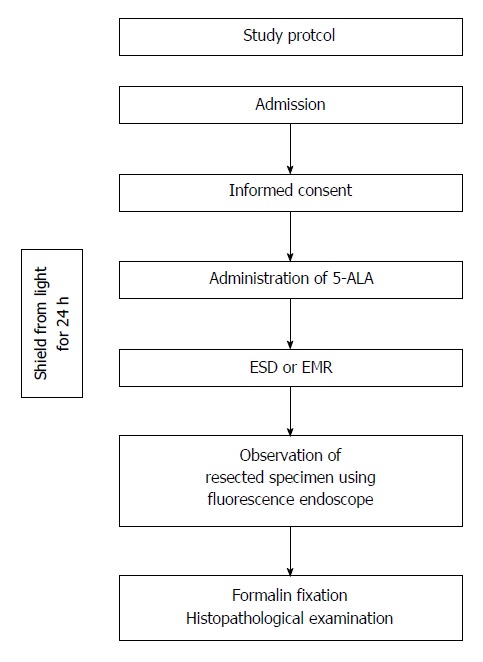
Flow chart of the study. 5-ALA: 5-aminolevulinic acid; ESD: Endoscopic submucosal dissection; EMR: Endoscopic mucosal resection.
Patients
Subjects were patients with gastric or colorectal tumors who had undergone endoscopic resection between November 2012 and November 2013. Patient selection criteria were age 20-80 years, either sex, and provision of informed consent after the content and purpose of the study were explained. Ten patients were enrolled. The gastric and colorectal lesions were selected on the basis of the indications for endoscopic resection in the guidelines for the treatment of gastric and colorectal cancer developed by the Japanese Gastric Cancer Association and the Japanese Society for Cancer of the Colon and Rectum, respectively[5,6]. Exclusion criteria were a predicted operation time of 2 h or longer, liver dysfunction, hypersensitivity to porphyrin, preexisting medical conditions, nyctophobia, or active bleeding from the tumor.
5-Aminolevulinic acid
Patients were orally administered 20 mg/kg of 5-ALA enteric-coated capsules (SBI ALApromo Co., Tokyo, Japan). 5-ALA is a precursor of PpIX, which is a biological substance that emits red fluorescence when excited by light in the wavelength of 400-410 nm. Endoscopic observation of each resected specimen was scheduled to take approximately 4 h after the 5-ALA was administered, and patients were shielded from light for 24 h after its administration, as described previously[7].
Endoscopic resection
Endoscopic resection was performed by 3 certified endoscopists of the Japan Gastroenterological Endoscopy Society (Nishikawa J, Hashimoto S, and Okamoto T). Endoscopic submucosal dissection (ESD) or endoscopic mucosal resection (EMR) was performed for endoscopic resection. ESD was performed using an endoscope (GIF-H260Z, -Q260J; Olympus, Tokyo, Japan) and high-frequency electrical generator (VIO 300D; Erbe Elektromedizin, Tübingen, Germany). The ESD procedure was performed as follows. First, several dots were marked on the surrounding normal mucosa 5 mm from the tumor. After injection of saline solution with epinephrine (0.025 mg/mL) into the submucosa, a mucosal incision was made with a needle knife (KI-1L-1; Olympus) outside the line of dots surrounding the tumor. Electrosurgical current was applied to complete the incision around the tumor using an insulation-tipped 2 knife (KD-611L; Olympus). After the circumferential cut was completed, the submucosa was dissected until the lesion was removed.
EMR was performed using an endoscope (CF-H260AZI; Olympus) and high-frequency electrical generator (ESG-100; Olympus). After injection of saline solution into the submucosa, mucosal resection was performed with a high-frequency snare (SD-210U-10; Olympus).
Evaluation items
The primary endpoint was the presence of fluorescence in tumors. After we administered 5-ALA and performed endoscopic resection, the resected specimens were subjected to fluorescence endoscopy to examine for the presence of red fluorescence. After endoscopic observation, specimens were fixed in formalin for histopathologic analysis and subjected to endoscopic, macroscopic, and pathologic comparisons. The presence of fluorescence was assessed by the principal investigator, who is a certified endoscopist of the Japan Gastroenterological Endoscopy Society.
We also evaluated adverse events of the present procedure as a secondary endpoint and examined each patient for the presence of known adverse effects of 5-ALA, namely, hematocytopenia, liver dysfunction, hypotension, nausea, and photosensitivity. Evaluation items were age, sex, tumor location, macroscopic type, tumor color, invasion depth, tumor differentiation, treatment method, and procedure time.
Statistical analysis
Results were analyzed statistically with the Student t-test or Fisher’s exact test, and a value of P < 0.05 was considered to indicate statistical significance. Statistical analysis was performed using statistical software (JMP program for Windows, version 9; SAS Institute, Cary, NC, United States).
RESULTS
Ten patients (7 men and 3 women) with a total of 13 lesions, consisting of 10 gastric tumors from 8 patients and 3 colorectal tumors from 2 patients, were enrolled in this study. Patient characteristics are shown in Table 1. The lesions included 9 gastric cancers, 1 gastric adenoma, and 3 colorectal cancers of which invasion depth was limited to within the mucosal or submucosal layer.
Table 1.
Characteristics of early gastric and colorectal tumors in this study
| Patient | Age (yr) | Sex | Location | Macroscopic type | Tumor size (mm) | Tumor color | Tumor depth | Tumor differentiation |
Treatment |
Adverse effect | FL | |
| Method | Time (min) | |||||||||||
| 1 | 71 | F | M | Protruded | 20 | Reddish | m | tub1 | ESD | 50 | No | Yes |
| 2 | 65 | M | L | Protruded | 20 | Reddish | m | tub1 | ESD | 23 | Yes | Yes |
| 2 | 65 | M | U | Protruded | 15 | Reddish | sm | tub2 | ESD | 82 | Yes | No |
| 3 | 75 | M | L | Protruded | 14 | Reddish | m | tub1 | ESD | 34 | Yes | Yes |
| 4 | 56 | M | M | Protruded | 10 | Reddish | m | tub1 | ESD | 54 | No | Yes |
| 5 | 70 | M | M | Protruded | 8 | Discolored | m | tub1 | EMR | 7 | No | Yes |
| 6 | 66 | M | U | Protruded | 10 | Reddish | adenoma | ESD | 60 | Yes | Yes | |
| 7 | 59 | M | L | Depressed | 12 | Reddish | m | tub1 | ESD | 43 | No | No |
| 8 | 65 | M | M | Depressed | 15 | Reddish | sm | tub1 | ESD | 23 | No | No |
| 8 | 65 | M | M | Depressed | 5 | Discolored | m | sig | ESD | 37 | No | No |
| 9 | 69 | F | R | Protruded | 20 | Reddish | m | tub1 | EMR | 25 | Yes | No |
| 10 | 67 | F | S | Protruded | 18 | Discolored | m | tub1 | EMR | 10 | No | No |
| 10 | 67 | F | S | Protruded | 23 | Reddish | sm | pap | EMR | 26 | No | Yes |
| Total | 66.3 ± 5.91 | 7M3F | 2U/5M/ | 10 Protruded | 14.6 ± 5.41 | 10 Reddish | 9 m/ | 1 adenoma | 9 ESD | 36.5 ± 21.11 | 4 Yes | 7 Yes |
| 3L/2S/1R | 3 Depressed | 3 Discolored | 3 sm | 9 tub1/1 tub2 1 pap/1 sig | 4 EMR | 6 No | 6 No | |||||
mean ± SD. FL: Fluorescence; U: Upper third of stomach; M: Middle third of stomach; L: Lower third of stomach; S: Sigmoid colon; R: Rectum; m: Mucosal layer; sm: Submucosal layer; tub1: Well-differentiated adenocarcinoma; tub2: Moderate differentiated adenocarcinoma; pap: Papillary adenocarcinoma; sig: Signet cell adenocarcinoma; ESD: Endoscopic submucosal dissection; EMR: Endoscopic mucosal resection.
Fluorescence was observed in 6 of the 10 (60.0%) gastric lesions. A representative case of an early gastric cancer with fluorescence is shown in Figure 2. ESD was performed for a 20-mm elevated lesion located in the incisura angularis of the lesser curvature (Figure 2A and B). The resected specimen was irradiated by white light (Figure 2C) and fluorescent light (Figure 2D). The TWIN mode system, in which white-light and fluorescent images are displayed concurrently, was used. Red fluorescence was observed in the cancerous area, whereas virtually no fluorescence was observed in the non-cancerous areas. In this case, we evaluated the spectra of the emitted fluorescence with a spectroscope (HSC1700; EBA JAPAN Co., Ltd., Tokyo, Japan), which records the reflectance in the wavelength range of 400-800 nm. The spectra showed a bimodal reflectance curve with peaks at 635 and 704 nm, the characteristic reflectance peaks of PpIX (Figure 3)[8-10].
Figure 2.
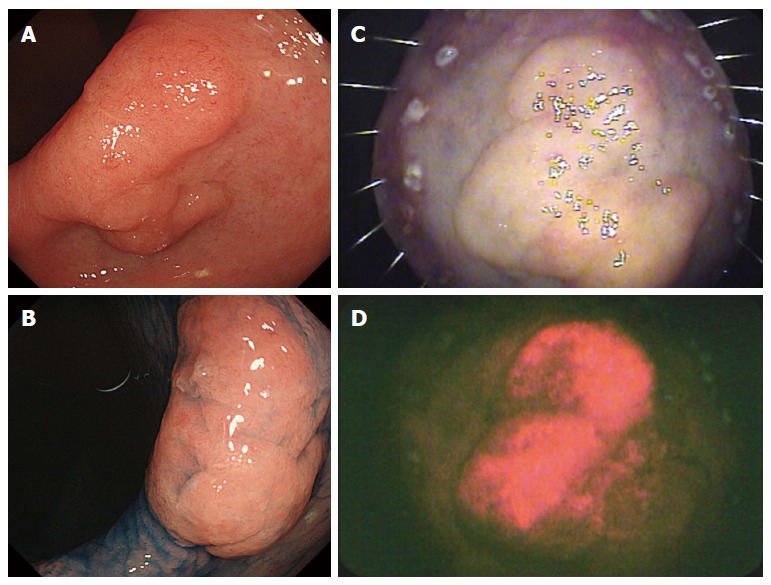
Early gastric cancer with fluorescence. A: Endoscopic image; B: Endoscopic image obtained after the spraying of indigo carmine dye; C: Resected specimen irradiated by white light; D: Resected specimen showing fluorescence.
Figure 3.
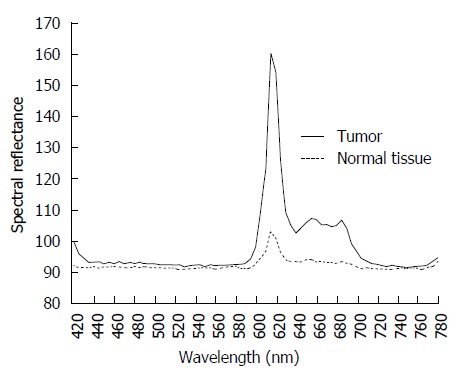
Spectral reflectance in the range of 400-800 nm obtained using a hyperspectral camera. Two reflectance peaks in the tumor were detected at 635 and 700 nm; this bimodal reflectance is the characteristic reflectance pattern of protoporphyrin IX.
Fluorescence was observed in one of three colorectal lesions. A representative case of an early colorectal cancer with fluorescence is shown in Figure 4. EMR was performed for a 23-mm elevated lesion located in the sigmoid colon (Figure 4A and B). Red fluorescence was detected in the tumor (Figure 4C and D).
Figure 4.
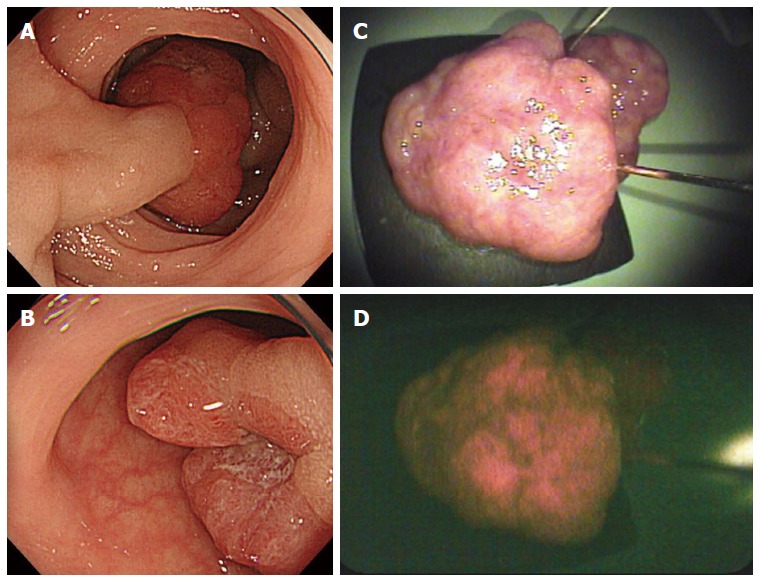
Early colorectal cancer with fluorescence. A and B: Endoscopic images; C: Resected specimen irradiated by white light; D: Resected specimen showing fluorescence.
No significant differences in sex (male: 55.6% vs female: 50.5%, P = 1.00), age (67.1 ± 1.9 years vs 65.0 ± 2.0 years, P = 0.45), tumor color (reddish: 60.0% vs discolored: 33.3%, P = 0.56), tumor diameter (15.0 ± 2.1 mm vs 14.2 ± 2.3 mm, P = 0.80), macroscopic type (protruded: 70.0% vs depressed 0%, P = 0.07), histologic type (differentiated type: 58.3% vs 0%, P = 0.46), invasion depth (mucosal layer: 55.6% vs submucosal layer: 33.3%, P = 1.00), lymphatic invasion (present: 33.3% vs absent: 50.0%, P = 1.00), venous invasion (present: 0% vs absent: 54.5%, P = 1.00) or procedure time of endoscopic resection (36.3 ± 8.3 min vs 36.7 ± 9.0 min, P = 0.98) were observed between the patients with and without fluorescence (Table 2). The detection rate of fluorescence tended to be high for elevated lesions of the macroscopic type.
Table 2.
Statistical analysis of clinicopathologic characteristics between tumors with and without fluorescence
|
Fluorescence |
||||
| No (n = 6) | Yes (n = 7) | Total (n = 13) | P value | |
| Sex | ||||
| F | 2 | 2 | 4 | |
| M | 4 | 5 | 9 | 1.00 |
| Age (yr) | ||||
| mean ± SD | 65.0 ± 2.0 | 67.1 ± 1.9 | 66.2 ± 4.9 | 0.45 |
| Tumor color | ||||
| Reddish | 4 | 6 | 10 | 0.56 |
| Discolored | 2 | 1 | 3 | |
| Tumor size (mm) | ||||
| mean ± SD | 14.2 ± 2.3 | 15.0 ± 2.1 | 14.6 ± 5.4 | 0.80 |
| Macroscopic type | ||||
| Protruded | 3 | 7 | 10 | 0.07 |
| Depressed | 3 | 0 | 3 | |
| Tumor differentiation | ||||
| Differentiated | 5 | 7 | 12 | 0.46 |
| Undifferentiated | 1 | 0 | 1 | |
| Tumor depth | ||||
| m | 4 | 5 | 9 | 1.00 |
| sm | 2 | 1 | 3 | |
| Lymphatic invasion | ||||
| Absent | 5 | 5 | 10 | |
| Present | 1 | 1 | 2 | 1.00 |
| Venous invasion | ||||
| Absent | 5 | 6 | 11 | 1.00 |
| Present | 1 | 0 | 1 | |
| Procedure time (min) | ||||
| mean ± SD | 36.7 ± 9.0 | 36.3 ± 8.3 | 36.5 ± 21.1 | 0.98 |
Differentiated: Adenoma, tub1, tub2, and pap; Undifferentiated: sig; m: Mucosal layer; sm: Submucosal layer; tub1: Well-differentiated adenocarcinoma; tub2: Moderate differentiated adenocarcinoma; pap: Papillary adenocarcinoma; sig: Signet cell adenocarcinoma.
With regard to adverse events, 4 of the 10 patients (40.0%) developed liver dysfunction. The extent of the liver dysfunction was a slight increase in transaminases and total bilirubin levels, which spontaneously improved in the patients. None of the patients developed photosensitivity.
DISCUSSION
The utility of PDD using 5-ALA has been demonstrated in various cancers since its first clinical application in brain tumors by Moore et al[11] in 1948. Recently, PDD with 5-ALA has been used clinically to verify both tumor extent and surgical residues in urologic and cerebral surgery[3]. 5-ALA is an essential amino acid synthesized from succinyl-CoA and glycine by ALA synthase in mitochondria[4]. Then, 5-ALA is converted into PpIX in mitochondria, followed by conversion to heme in normal cells. PpIX induced by 5-ALA is photoactive and emits red fluorescence when irradiated by an excitation light in the 400-410 nm range[8]. In tumors, PpIX synthesis is highly activated, and the enzyme that converts PpIX into heme is downregulated; therefore, PpIX tends to accumulate specifically in tumors[4]. The SAFE-3000 processor uses a semiconductor laser diode that emits light at 408 nm, which is in the excitation wavelength range of PpIX, and detects fluorescence images. We took advantage of the high tumor specificity of fluorescent PpIX to conduct this preliminary study to apply PDD using 5-ALA for gastrointestinal endoscopy[2].
Red fluorescence was observed in the cancerous area, whereas virtually no fluorescence was observed in the non-cancerous areas. We confirmed with a spectroscope that the spectra of the emitting fluorescence showed a bimodal reflectance curve with peaks at 635 and 704 nm, which are the characteristic reflectance peaks of PpIX (Figure 3)[8-10]. We disregarded the autofluorescence property of the background mucosa because its intensity is well below that emitted by PpIX and is too weak to affect endoscopic observation with the SAFE-3000. No fluorescence was observed in tumors resected from patients who had not taken 5-ALA (data not shown). Previous reports showed PDD using 5-ALA provides good visualization and detection of gastric cancers[12,13]. This report also indicated that PDD using 5-ALA was useful to detect early gastric cancers. We used enteric capsules to avoid non-specific background fluorescence in the gastric mucosa. This is the first study using enteric capsules for administration of 5-ALA. The findings in this study suggest that PDD with the oral administration of 5-ALA is useful and safe for the screening of early gastric and colorectal cancers.
The endoscopic view during the fluorescent mode became dark compared to the usual view by white-light observation, as shown in Figures 2D and Figure 4D. It is difficult to evaluate findings of the mucosal surface with only fluorescent images. The SAFE-3000 uses a TWIN mode system in which white-light and fluorescent images are displayed concurrently. The TWIN mode system made it possible to compare fluorescence accumulation and macroscopic findings by white-light images. We believe that TWIN mode will be of great help when performing actual endoscopic observation by PDD using 5-ALA.
The present fluorescence detection rate was 53.8%, which is lower than the 85% rate in a PDD study of esophageal tumors[14]. Although no significant differences were observed in patient or tumor factors between the tumors with and without fluorescence (Table 2), the fluorescence detection rate tended to be higher in elevated lesions (P = 0.070). This suggests that the formation of thick tumors by proliferation of cancer cells is required for the expression of fluorescence. To facilitate clinical application of PDD using 5-ALA, further studies are needed to elucidate the conditions for maximum fluorescence expression, for example, chronological changes in the emission of fluorescence after 5-ALA administration.
In previous studies, the adverse effects of 5-ALA were cytopenia, mild liver dysfunction, nausea, and photosensitivity[4,10], with an incidence of < 1% for each[4]. In the present study, temporary liver dysfunction was caused by 5-ALA in 4 of the 10 patients. Our patients were shielded from light for 24 h after the administration of 5-ALA, and none of them developed photosensitivity. Because the half-life of 5-ALA is 0.7-0.9 h, the incidence of photosensitivity would be expected to remain low[4]. We consider the clinical application of PDD using 5-ALA to be relatively safe.
A limitation of this study is that we examined the resected specimens ex vivo. Blood flow and oxygen saturation in the tumor would have differed from those observed in vivo.
In conclusion, fluorescence was localized consistently in the area of the gastric and colorectal tumors, and no serious adverse events were observed. With the development of a fluorescence endoscope for use in the stomach and colon, PDD using 5-ALA may become an effective screening tool for gastric and colorectal cancers.
COMMENTS
Background
Esophagogastroduodenoscopy is widely used for the screening of gastric and colorectal tumors in Japan. The detection rate of early-stage gastric and colorectal tumors continues to improve owing to technological advances in endoscopy, yet some tumors are still being missed. The reported miss rate for gastric cancers is approximately 20%.
Research frontiers
Image-enhanced endoscopy (IEE) was recently developed to detect lesions by changing the wavelength of irradiated light and by processing the acquired endoscopic images. IEE was reported to be useful for the diagnosis of gastric and colorectal tumors. However, this technique has not been sufficient to improve the detection rate of gastric and colorectal tumors.
Innovations and breakthroughs
The findings in this study suggest that photodynamic diagnosis (PDD) with the oral administration of 5-aminolevulinic acid (5-ALA) is useful and safe for the screening of early gastric and colorectal cancers.
Applications
With the development of a fluorescence endoscope for use in the stomach and colon, PDD using 5-ALA may become an effective screening tool for gastric and colorectal cancers.
Peer-review
This manuscript is well written and describes a comprehensible prospective single-center study investigating the applicability of photodynamic diagnosis using 5-ALA in resected specimens of patients with gastric and colorectal cancer.
Footnotes
Ethics approval: The study was reviewed and approved by the Yamaguchi University Hospital Institutional Review Board.
Clinical trial registration: This study is registered at UMIN-CTR: http://www.umin.ac.jp/ctr/. The registration identification number is 000010086.
Informed consent: All study participants, or their legal guardian, provided informed written consent prior to study enrollment.
Conflict-of-interest: All authors disclosed no financial relationships relevant to this publication.
Data sharing: No additional data are available.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: December 23, 2014
First decision: January 27, 2015
Article in press: March 19, 2015
P- Reviewer: Hehlgans S S- Editor: Yu J L- Editor: O’Neill M E- Editor: Wang CH
References
- 1.Lee HL, Eun CS, Lee OY, Han DS, Yoon BC, Choi HS, Hahm JS, Koh DH. When do we miss synchronous gastric neoplasms with endoscopy? Gastrointest Endosc. 2010;71:1159–1165. doi: 10.1016/j.gie.2010.01.011. [DOI] [PubMed] [Google Scholar]
- 2.Kato M, Kaise M, Yonezawa J, Yoshida Y, Tajiri H. Autofluorescence endoscopy versus conventional white light endoscopy for the detection of superficial gastric neoplasia: a prospective comparative study. Endoscopy. 2007;39:937–941. doi: 10.1055/s-2007-966857. [DOI] [PubMed] [Google Scholar]
- 3.Nokes B, Apel M, Jones C, Brown G, Lang JE. Aminolevulinic acid (ALA): photodynamic detection and potential therapeutic applications. J Surg Res. 2013;181:262–271. doi: 10.1016/j.jss.2013.02.002. [DOI] [PubMed] [Google Scholar]
- 4.Ishizuka M, Abe F, Sano Y, Takahashi K, Inoue K, Nakajima M, Kohda T, Komatsu N, Ogura S, Tanaka T. Novel development of 5-aminolevurinic acid (ALA) in cancer diagnoses and therapy. Int Immunopharmacol. 2011;11:358–365. doi: 10.1016/j.intimp.2010.11.029. [DOI] [PubMed] [Google Scholar]
- 5.Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2010 (ver. 3) Gastric Cancer. 2011;14:113–123. doi: 10.1007/s10120-011-0042-4. [DOI] [PubMed] [Google Scholar]
- 6.Watanabe T, Itabashi M, Shimada Y, Tanaka S, Ito Y, Ajioka Y, Hamaguchi T, Hyodo I, Igarashi M, Ishida H, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Int J Clin Oncol. 2012;17:1–29. doi: 10.1007/s10147-011-0315-2. [DOI] [PubMed] [Google Scholar]
- 7.Zaak D, Sroka R, Khoder W, Adam C, Tritschler S, Karl A, Reich O, Knuechel R, Baumgartner R, Tilki D, et al. Photodynamic diagnosis of prostate cancer using 5-aminolevulinic acid--first clinical experiences. Urology. 2008;72:345–348. doi: 10.1016/j.urology.2007.12.086. [DOI] [PubMed] [Google Scholar]
- 8.Haj-Hosseini N, Richter J, Andersson-Engels S, Wardell K. Photobleaching behavior of protoporphyrin IX during 5-aminolevulinic acid marked glioblastoma detection. Proc SPIE. 2009;1:7161. [Google Scholar]
- 9.Kiyotoki S, Nishikawa J, Okamoto T, Hamabe K, Saito M, Goto A, Fujita Y, Hamamoto Y, Takeuchi Y, Satori S, et al. New method for detection of gastric cancer by hyperspectral imaging: a pilot study. J Biomed Opt. 2013;18:26010. doi: 10.1117/1.JBO.18.2.026010. [DOI] [PubMed] [Google Scholar]
- 10.Tope WD, Ross EV, Kollias N, Martin A, Gillies R, Anderson RR. Protoporphyrin IX fluorescence induced in basal cell carcinoma by oral delta-aminolevulinic acid. Photochem Photobiol. 1998;67:249–255. doi: 10.1562/0031-8655(1998)067<0249:pifiib>2.3.co;2. [DOI] [PubMed] [Google Scholar]
- 11.Moore GE, Peyton WT. The clinical use of fluorescein in neurosurgery; the localization of brain tumors. J Neurosurg. 1948;5:392–398. doi: 10.3171/jns.1948.5.4.0392. [DOI] [PubMed] [Google Scholar]
- 12.Namikawa T, Inoue K, Uemura S, Shiga M, Maeda H, Kitagawa H, Fukuhara H, Kobayashi M, Shuin T, Hanazaki K. Photodynamic diagnosis using 5-aminolevulinic acid during gastrectomy for gastric cancer. J Surg Oncol. 2014;109:213–217. doi: 10.1002/jso.23487. [DOI] [PubMed] [Google Scholar]
- 13.Nakamura T, Oinuma T, Yamagishi H, Masuyama H, Terano A. Evaluation of a novel high-resolution magnifying videoendoscope that is capable of photodynamic diagnosis and therapy for gastric cancer. Photodiagnosis Photodyn Ther. 2015;12:115–122. doi: 10.1016/j.pdpdt.2014.10.010. [DOI] [PubMed] [Google Scholar]
- 14.Mayinger B, Neidhardt S, Reh H, Martus P, Hahn EG. Fluorescence induced with 5-aminolevulinic acid for the endoscopic detection and follow-up of esophageal lesions. Gastrointest Endosc. 2001;54:572–578. doi: 10.1067/mge.2001.118642. [DOI] [PubMed] [Google Scholar]


