Key Points
Infused human megakaryocytes release young platelets in the lungs with characteristics similar to donor platelets.
Platelets released from ex vivo–derived megakaryocytes are preactivated and compare poorly to donor platelets.
Abstract
Thrombopoiesis is the process by which megakaryocytes release platelets that circulate as uniform small, disc-shaped anucleate cytoplasmic fragments with critical roles in hemostasis and related biology. The exact mechanism of thrombopoiesis and the maturation pathways of platelets released into the circulation remain incompletely understood. We showed that ex vivo–generated murine megakaryocytes infused into mice release platelets within the pulmonary vasculature. Here we now show that infused human megakaryocytes also release platelets within the lungs of recipient mice. In addition, we observed a population of platelet-like particles (PLPs) in the infusate, which include platelets released during ex vivo growth conditions. By comparing these 2 platelet populations to human donor platelets, we found marked differences: platelets derived from infused megakaryocytes closely resembled infused donor platelets in morphology, size, and function. On the other hand, the PLP was a mixture of nonplatelet cellular fragments and nonuniform-sized, preactivated platelets mostly lacking surface CD42b that were rapidly cleared by macrophages. These data raise a cautionary note for the clinical use of human platelets released under standard ex vivo conditions. In contrast, human platelets released by intrapulmonary-entrapped megakaryocytes appear more physiologic in nature and nearly comparable to donor platelets for clinical application.
Introduction
Platelets have major roles in hemostasis, thrombosis, inflammation, and vascular biology.1-3 Platelets are circulating anucleate cytoplasmic discs derived from differentiated megakaryocytes.4,5 The details by which a megakaryocyte releases 103-4 platelets6 are beginning to be understood, but this knowledge has been hampered by limitations of ex vivo culture systems.4,5 In situ examination of this process has provided key insights7-9: intramedullar, mature megakaryocytes migrate to a perivascular site and extend a single process through the endothelium, releasing variable size cytoplasmic fragments that may or may not remain continuous.9 Whole megakaryocytes have been noted to escape.7 Based on lung histology10 and differential measurements of pulmonary arterial vs venous platelet counts, enough megakaryocytes travel to the lungs to release platelets to maintain the steady-state platelet count.11 At the moment, the relative contributions of intramedullar- vs pulmonary-released platelets is unclear.
There are multiple thrombocytopenic states that necessitate therapeutic intervention. Thrombopoietin mimetics increase platelet counts in some thrombocytopenias.12 In others, donor platelet transfusions remain the mainstay of care.13 The need for platelet transfusions has been increasing so understanding thrombopoiesis and developing platelets from ex vivo–derived (EV) human megakaryocytes, especially from self-renewing cells such as embryonic stem cells and induced pluripotent stem cells (iPSCs),14-18 have been investigated. To date, a major limitation in generating EV-platelets has been their low yield and functional limitations. We show that EV-platelets include a mixture of CD41+ and CD41− particles of various sizes. These mixtures will be designated “platelet-like particles” or EV-PLPs. Xenotransfusion of EV-PLPs into immunocompromized, severely thrombocytopenic mice has resulted in modest numbers of circulating human platelets,14-16 and functional studies have shown incorporation into thrombi in recipient mice.14-18 Advancements in the generation of EV-PLPs have clearly been made, but limited studies have been performed comparing EV-PLP to infused, donor platelets.16,18
Using murine megakaryocytes generated from adult marrow cells (AMCs) and fetal liver cells (FLCs), we previously showed that intravenous infusion of these cells resulted in pulmonary entrapment and release of ∼102 in vivo–generated (IV) platelets per megakaryocyte, after an ∼1.5-hour delay.19 IV-platelet levels of ∼20% of total circulating platelets were achieved in nonthrombocytopenic mice. In thrombocytopenic mice, increases in platelet counts comparable to infused donor platelets were achieved, normalizing hemostasis.19 We now show that human EV-megakaryocytes xenotransfused into NOD/SCID/γ-interferon-deficient (NSG) mice generate IV-platelets independent of the starting source of cells (eg, AMCs, FLCs, or iPSCs). We describe 2 pools of human platelets in the recipient mice: (1) IV-platelets released intrapulmonary from EV-megakaryocytes, which are similar to infused donor platelets and (2) EV-PLP-derived platelets that differ markedly from donor platelets. Therefore, clinical application of stem cell-derived megakaryocytes will either require further optimization of EV-platelet formation with a focus on generating nonactivated platelets or to infuse EV-megakaryocytes and allow platelet shedding in vivo.
Materials and methods
Derivation of human EV-megakaryocytes, EV-PLPs, and donor platelets
AMC EV-megakaryocytes
Human CD34+ cells from bone marrow or that were granulocyte colony-stimulating factor mobilized were purchased from Fred Hutchinson Cancer Research Center Cell Processing Shared Resource and expanded and differentiated to megakaryocytes in Serum-Free Expansion Medium (SFEM; Stemcell Technologies) for 14 days.15,20
FLC EV-megakaryocytes
Hematopoietic mononuclear cell fractions of human fetal livers were obtained from week 13 to 23 abortuses.21 For megakaryocyte differentiation, these cells were cultured for 14 days in SFEM containing 100 ng/mL thrombopoietin, 40 μg/mL low-density lipoprotein, and 1% penicillin/streptomycin (all from Life Technologies).
iPSC EV-megakaryocytes
An iPSC line (WTBM1-8) was generated using a lentivirus expressing OCT4, SOX2, KLF4, and MYC22 and were analyzed for pluripotency by teratoma formation, flow cytometry, and gene expression.23 iPSCs were differentiated into megakaryocytes as previously described.24
For all, large megakaryocytes were isolated using a 2-step bovine serum albumin density gradient described for murine megakaryocytes19 and counted by hematocytometer before retro-orbital infusion in 200 µL phosphate-buffered saline (PBS; Invitrogen). Similar AMC growth conditions were used to isolate EV-PLPs.15 Briefly, EV-PLPs were collected from the culture medium and 1/10th volume of 3.8% sodium citrate was added. EV-PLP supernatant was obtained by centrifugation at 1000 rpm for 10 minutes. The PLPs were pelleted at 2000 rpm for 10 minutes and resuspended in 200 µL PBS for infusion. In some studies, EV-PLPs were labeled with 4 µM calcein violet (Invitrogen) for 30 minutes, washed with Tyrode buffer, and then pelleted and resuspended for infusion. Micrographs of processed cells and EV-PLPs before infusion were obtained using a CKX41 microscope (Olympus) equipped with an Axiocam camera (Carl Zeiss). Megakaryocyte DNA ploidy distribution was assessed on CD41+CD42a+ megakaryocytes using propidium iodide,19 and mean ploidy megakaryocytes were calculated.25
Donor platelets were isolated from venous blood of healthy volunteers after informed consent.26 Platelet counts were determined using a HemaVet counter (Triad Associates) before infusion into recipient mice. Human studies were approved by the Institutional Review Board at Children’s Hospital of Philadelphia (CHOP) in accordance with the Declaration of Helsinki.
Mice studied
NSG mice were produced pathogen-free at CHOP using Jackson Laboratory breeders. NSG male mice at 8 to 12 weeks of age were used for infusion with human megakaryocytes by retro-orbital injection. Animal experiments were approved by CHOP’s Institutional Animal Care and Use Committee.
Flow cytometric studies
Levels of human platelets in recipient mice were determined by using a combination of phycoerythrin (PE)-conjugated rat anti-mouse CD41 monoclonal antibody (moAb), fluorescein isothiocyanate (FITC), or allophycocyanin (APC)-conjugated mouse anti-human CD42a, APC- or FITC-conjugated mouse anti-human CD42b antibody, and isotype controls (all BD Biosciences). For detecting young platelets in recipient mice, freshly drawn whole blood was stained with PE-conjugated rat anti-mouse CD41 moAb (BD Biosciences) and APC-conjugated mouse anti-human CD41 antibody plus 50 ng/mL thiazole orange (TO; Becton Dickinson) for 15 minutes.27
Human platelet activation in whole mouse blood was assessed using PE-conjugated mouse anti-human P-selectin (CD62p) moAb (BD Biosciences) in the absence or presence of convulxin (CVX, 24 nM;Enzo Life Sciences) for 15 minutes.28 In vitro activation of donor platelets, IV-platelets, and EV-PLPs were assessed by flow cytometry using retro-orbital whole blood stained with APC-conjugated mouse anti-human CD41 antibody and PE-conjugated mouse anti-human P-selectin (CD62P moAb; BD Biosciences). In some studies, donor platelets and EV-PLPs were treated with CVX (24 nM) or thrombin receptor agonist peptide (TRAP, 20 nM) for 10 minutes at 37°C and stained for Annexin V binding.19
Flow cytometry was performed using a BD FACSCalibur (BD Biosciences). Data were analyzed using FlowJo version 9.5.2 (TreeStar). In some experiments, clodronate liposomes (Encapsula NanoSciences) were infused at 100 µL/mouse 24 hours before megakaryocyte or platelet infusions.29 Depletion of macrophages was monitored after red cell lysis using PerCP-conjugated rat anti-mouse CD45 moAb (BD Pharmingen), FITC-conjugated rat anti-mouse CD11b moAb (BD Pharmingen), PE-conjugated anti-mouse CD115 moAb (eBiosciences), and Alexa Fluor 647 (Molecular Probes)-conjugated anti-mouse Ly-6C Ab (BioLegend).
Immunofluorescence microscopy studies
Donor platelets or EV-megakaryocytes were resuspended in 200 µL PBS before infusion into NSG mice who had been irradiated 10 days earlier with 300 centigray. Two hours later, whole mice blood was taken from the vena cava, and mice platelet-rich plasma (PRP) was isolated. Details regarding isolation of PRP, fixation, and immunostaining are as previously decribed.30 Immunolabeled cells were analyzed on a Leica DM16000B inverted microscope equipped with a 100× plan apo lens, Hamamatsu Orca Flash 4.0 V2 camera, and Leica Application Suite software for image capture and deconvolution. Sequential z planes were captured at 0.2-μm intervals and deconvolved using Gold’s algorithm for 3 iterations. Images were assembled and optimized for brightness and contrast using Adobe Photoshop (Mountainview, CA). Species specificity of the LAMP1 antibodies (1D4B and H4A3 for mouse and human LAMP1, respectively; Developmental Studies Hybridoma Bank, Iowa City, IA) was verified using separately isolated human and mouse platelets.
Tissue immunohistochemistry studies
To address where human platelets were released from infused EV-megakaryocytes, cells were exposed to 10 µM bromodeoxyuridine (BrdU; Sigma-Aldrich) for 48 hours. Recipient mice were killed after infusion, and tissues were isolated and fixed.18 BrdU-labeled nuclei were detected using a rat anti-BrdU moAb (Abcam) and biotinylated goat anti-rat IgG (Vector Laboratories) as secondary antibody. Human megakaryocyte cell membrane in the lungs was also studied using a rabbit human anti-CD41a moAb (Epitopics) and secondary biotinylated goat anti-rabbit IgG antibody (Vector Laboratories). Tissues were counterstained with hematoxylin (Fisher Scientific).19 NSG mice infused with PBS served as negative controls.
Cremaster laser injury functional studies
To exam whether human platelets incorporate into developing thrombi, laser-induced injuries of cremaster arterioles (30-40 µm diameter) were performed.19 Donor platelets or EV-megakaryocytes were labeled with 2 µM calcein AM (Invitrogen) at 37°C for 30 minutes, washed twice with PBS, and resuspended in 200 µL PBS before jugular infusion. Alexa 647-conjugated rat anti-mouse CD41 Fab fragments (BD Pharmingen) were injected intravenously 5 minutes before injury. Thirty minutes after cell infusion, laser injuries were induced in the cremaster arterioles, and thrombosis was recorded. To determine selective human platelet incorporation, 100 µL of 250 µM tirofiban (Ben Venue Laboratories), a species-specific inhibitor of human CD41,31 was injected after control injuries had been recorded. After tirofiban infusion, additional injuries were made. To exam whether young platelets were incorporated into developing thrombosis, donor platelets or AMC EV-megakaryocytes were labeled with 4 µM calcein violet and 50 ng/mL TO before infusion. Injuries were then induced, and thrombi were captured as Z-stack images.
Statistical analysis
Statistical analysis was performed using a 2-tailed Student t test of means ± standard error of the mean (SEM). Differences were considered significant when P < .05.
Results
Circulating human platelets derived from EV-megakaryocytes in xenotransfused mice
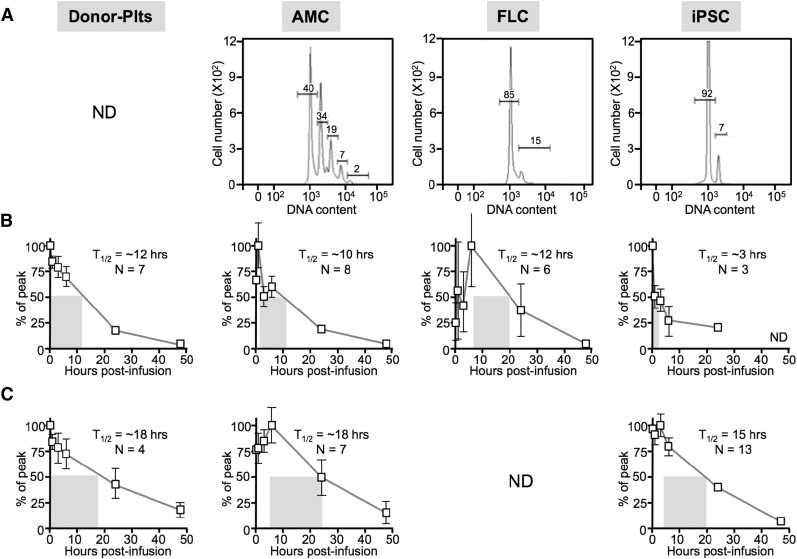
Previously, our group had shown that murine AMC- or FLC-derived EV-megakaryocytes infused into mice became entrapped in the pulmonary microvasculature, releasing functional platelets into the circulation.19 We asked whether this phenomenon would also apply to infused human megakaryocytes. For this work, we used a previously described platelet-xenotransfusion model in which human platelets infused into naïve, immunodeficient mice circulated with a half-life of >12 hours32 (Figure 1). We used NSG mice because of their success in a marrow xenotransplantation model in which human platelets are detectable only from 2 to 4 weeks after transplantation 29 (supplemental Figure 1, available on the Blood Web site). We analyzed EV-megakaryocytes from 3 different cell sources; AMCs, FLCs, and iPSCs. Infused donor platelets had a 12-hour half-life. Comparable to our mouse study,19 infused human AMC EV-megakaryocytes (average ploidy = 5.8 ± 0.6; Figure 1A) had a delayed platelet peak and an ∼10-hour half-life (Figure 1B). Peak yield was 32 ± 4 platelets per megakaryocyte (Table 1). FLC-derived EV-megakaryocytes had a more limited ploidy (2.4 ± 0.1; Figure 1A), but similar half-life (Figure 1B), yielding ∼15 platelets per megakaryocyte (Table 1). Finally, iPSC EV-megakaryocytes also had low ploidy (2.4 ± 0.2; Figure 1A), but in addition had a low yield of platelets (∼3 platelets per megakaryocyte; Table 1). Here, platelet survival was more complex, including a short, immediate half-life of ∼3 hours, followed by a longer half-life (Figure 1B).
Figure 1.
Infused human donor platelets and EV-megakaryocytes from different developmental stages into NSG mice. Donor platelets (plts) and megakaryocytes grown from AMC-, FLC-, and iPSC-derived hematopoietic progenitors were infused into NSG recipient mice. ND, not determined. (A) Degree of ploidy determined for the infused megakaryocytes before infusion showing representative studies. (B) Mean ± SEM for percent of peak human platelet counts determined over the first 48 hours after infusion. Each mouse was infused with 1 to 2.2 × 106 megakaryocytes. The half-life is indicated by a gray rectangle beginning at peak platelet count. The half-life value and the number of independent studies done are indicated in each graph. N = number of independent studies done in duplicate. (C) Same as in B, but in mice that had been pretreated intravenously with clodronate-ladened liposomes.
Table 1.
Yield of human platelets per megakaryocyte infused into NSG mice with or without liposomal clodronate treatment
| Clodronate liposomes | Per AMC-derived cell | Per FLC-derived cell | Per iPSC-derived cell |
|---|---|---|---|
| No treatment | 32 ± 4 (N = 8) | 15 ± 3 (N = 6) | 3 ± 1 (N = 6) |
| Treated | 54 ± 6 (N = 7) | Not done | 15 ± 3 (N = 13) |
The number of released platelets per total cells infused determined by assuming that a 20-g mouse has a blood volume of 2 mL and that every nucleated cell infused releases platelets. Based on peak percent of platelets that are infused. Mean ± SEM is shown with the number (N) of separate experiments, each done in duplicate, in parentheses.
A major reason for the limited window of detectable circulating platelets in xenotransplanted NSG mice is macrophage-mediated clearance of human platelets (supplemental Figure 1).29 We therefore tested what effect macrophage removal would have on platelet half-life in xenotransfused mice. Studies were performed using naïve NSG mice treated with clodronate liposomes. Human donor platelets half-life modestly increased to ∼18 hours. These data suggest that clearance of human platelets is occurring in naïve NSG mice, but to a much lesser degree than in the xenotransplantation model (Figure 1C; supplemental Figure 1). A similar prolongation of the half-life of platelets derived from infused EV-megakaryocytes was observed, especially for iPSC EV-megakaryocytes (Figure 1C).
We had found that infused murine EV-megakaryocytes were entrapped in the pulmonary microvessels and disappeared concurrent with the appearance of circulating platelets.19 Pulmonary entrapment and platelet release also occurred after infusion of human EV-megakaryocytes into NSG mice as detected by BrdU-labeled nuclei and human CD41 staining (Figure 2A-B). The spreading of human cellular processes from entrapped megakaryocytes into perialveolar microvessels was notable (supplemental Figure 2, arrows). Based on differences in organ size,33-35 but with a similar degree of megakaryocyte entrapment, we estimate that 5% to 10% of entrapment occurred in the spleen (Figure 2C), but in no other studied tissue (supplemental Figure 3). Comparative confocal microscopy of human donor platelets circulating in mice blood and of released human platelets 2 hours after infusing AMC EV-megakaryocytes showed similar morphology, were equally greater in size than the mice platelets,36-38 had discoid shape, and had a similar number and distribution of α-granules and lysosomes (supplemental Figure 4).
Figure 2.
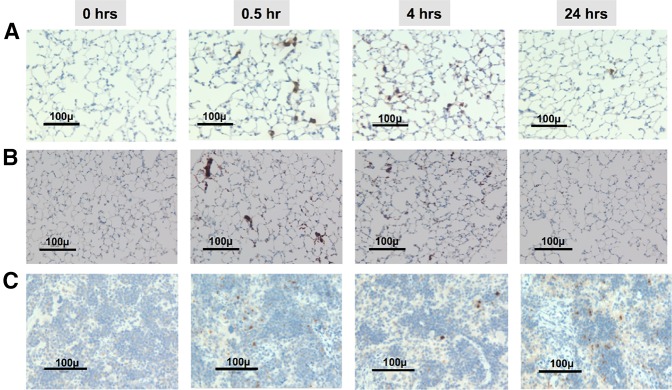
Histologic examination of the lungs and spleens after infusion of AMC EV-megakaryocytes into NSG mice. (A) Temporal representative sections of the lungs in recipient animals after the infusion of human BrdU-labeled megakaryocytes into NSG mice. Counterstained with hematoxylin. Black bar = 100 µm at an original magnification of 40×. (B) Same as in A, but stained for human CD41. (C) Same as in A, but splenic tissue is presented.
Size characterization of human platelets derived from infused EV-megakaryocytes
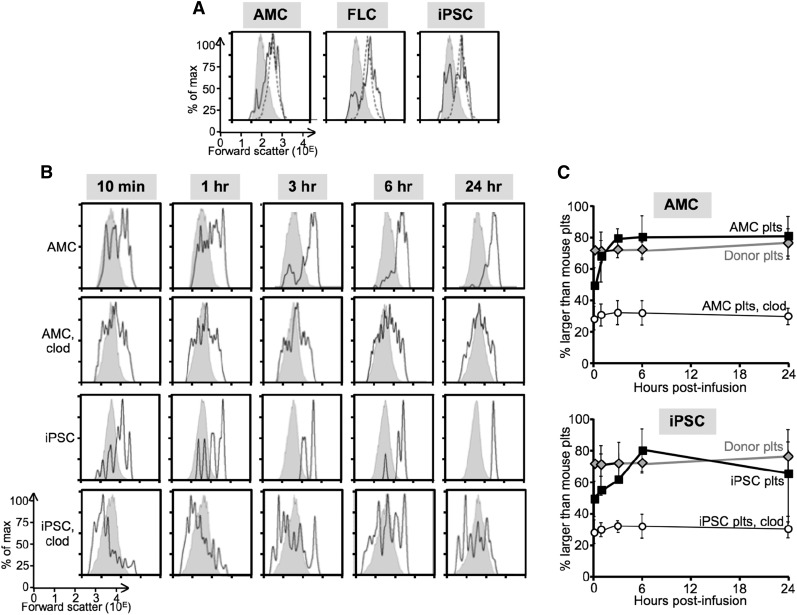
It has been proposed that proplatelets become preplatelets and eventually platelets.39 The physical environment, including shear forces and vascular architecture, are thought to be important in this processing.7,40 Murine platelets have approximately one third of the diameter of human platelets, with both displaying bell-shaped size distributions.36-38 Whether higher shear forces in mice and differences in microvasculature contribute to small murine platelet size is unknown. We examined the size range (as determined by forward scatter on flow cytometry41) of human platelets released from all 3 EV-megakaryocytes (Figures 3A) and compared their size distribution to that of murine platelets and infused donor platelets. Both endogenous murine platelets and infused human platelets showed bell-shaped size distributions (Figure 3A, gray areas and dashed lines, respectively) with human platelets being ∼10-fold greater. Platelets generated from all 3 sources of human EV-megakaryocytes showed mostly the same peak size distribution as donor platelets, consistent with supplemental Figure 4, with no pool of CD41+ fragments larger than donor platelets (Figure 3A, solid lines). In addition, there was a broader peak in the murine platelet size range. We asked whether these size distributions remained constant over time and noted that the small human platelets (defined as having a forward scatter ≤90% of the mouse platelet size range) disappeared over the first few hours after infusion of EV-megakaryocytes (Figure 3B-C), whereas large platelets (defined as having forward scatter >90% of the mouse platelet size range) predominated by 6 hours with their size distribution near identical to infused donor platelets. We then examined the effect of macrophage depletion on the platelet size distribution after AMC and iPSC EV-megakaryocyte infusion and noted that the initial broad distribution of platelet sizes did not change over time (Figure 3B-C). A peak in the large platelet size range never emerged.
Figure 3.
Size distribution of platelets in recipient mice. (A) Representative flow cytometric studies from ≥5 studies 1 hour after infusion showing forward scatter distribution as an indicator of size for the recipient mice platelets (shaded gray) compared with infused human donor platelets (dotted line) to mice studied in parallel receiving infused EV-megakaryocytes (solid line) grown from AMCs (left), FLCs (center), and iPSCs (right). (B) Representative size distribution studies after infused AMC- or iPSC-derived EV-megakaryocytes as indicated ± clodronate-ladened liposome (clod) pretreatment of the recipient NSG mice as determined by forward scatter with recipient mice platelet size as an internal size standard. (C) Mean ± SEM of ≥5 studies per arm of changes after infused AMC (top) or iPSC (bottom) EV-megakaryocytes or donor platelets that are >90% of recipient mouse platelet size. In some studies, the recipient mice had been pretreated with clodronate-ladened liposomes.
TO studies of young vs old platelets from infused EV-megakaryocytes
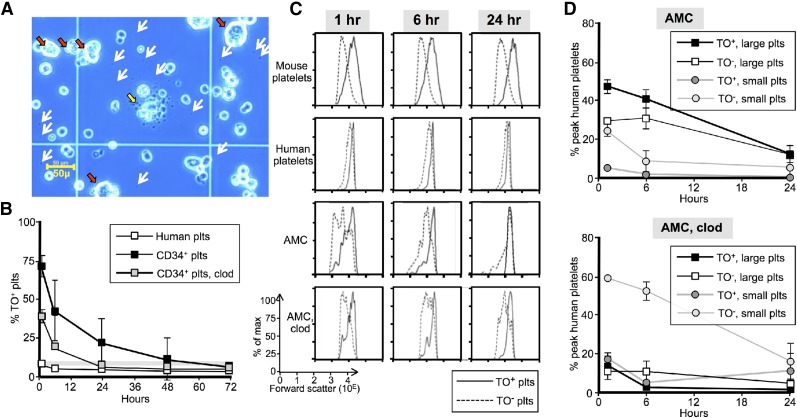
We hypothesized that these small platelets should be predominantly old, having been released from EV-megakaryocytes during culture and/or processing. The large platelets should initially all be young. We addressed this hypothesis using available AMC EV-megakaryocytes. Visual examination of an AMC EV-megakaryocyte preparation before infusion showed PLPs (Figure 4A, white arrows), variably sized nucleated cells (red arrows), and a large cell disaggregating in the center (yellow arrow) consistent with the concept that EV-PLPs are of varied age. Platelet age was measured using TO, which stains platelet RNA, ADP, and ATP, all of which correlate with age.27,42,43 Murine platelets had a steady-state level of ∼8% TO+ platelets (Figure 4B, horizontal gray bar). Infused donor platelets initially showed the same initial level of staining, but staining was lost by 24 hours (Figure 4B, open squares). On the other hand, >70% of platelets derived from infused EV-megakaryocytes were initially TO+, and positivity decreased with an ∼12-hour half-life (Figure 4B, black squares).
Figure 4.
Characterization young vs old platelets. (A) Representative field of AMC EV-megakaryocyte preparation on a hematocytometer before infusion. White arrows represent some of the visible platelet-size particles. Red arrows indicate large megakaryocyte-like cells, whereas a large cell with extended proplatelets/platelets is shown in the center (yellow arrow). Bar in left lower corner indicates size of the observed field. (B) Temporal profile of the percent of a circulating platelet population that are TO+. Thin gray horizontal bar is percent TO+ recipient murine platelets. Open squares are percent of infused human platelets that are TO+. Black squares are percent of derived platelets from infused AMC EV-megakaryocytes that are TO+, and gray squares are the same when infused into clodronate liposome-pretreated mice. Mean ± SEM are shown. N ≥ 5 studies per arm. (C) Representative temporal profiles of size as determined by forward scatter for TO+ (solid lines) and TO− (dashed lines) platelets. Top row is recipient mice platelets after infused AMC EV-megakaryocytes. Second row is human platelets after infusion of donor platelets. Third and bottom row are human platelets derived from AMC EV-megakaryocytes without and with clodronate liposome pretreatment, respectively. (D) Graphs are percent of the maximum human platelet number (which occurred 1-hour after EV-megakaryocyte infusion) at each time point separated out by both TO staining and size. The AMC EV-megakaryocytes were infused into either mice that were untreated (top) or pretreated (bottom) with clodronate liposomes. Mean ± SEM are shown. N ≥ 5 studies per arm.
Both TO− and TO+ mouse and human platelets followed bell-shaped curves of size distribution, with the mean size of the TO+ platelets larger as anticipated44,45 (Figure 4C, top two rows). Human platelets seen after infused AMC EV-megakaryocytes followed a different pattern (Figure 4C, third row, and D, top): initial TO+ platelet size distribution had a left shoulder of small-sized, presumably young EV-platelets. This shoulder disappeared rapidly. The remaining TO+ platelets were the size of donor platelets. TO− platelet size distribution was initially broad, but by 6 hours, a bell-shaped size curve emerged that markedly overlapped with the TO+ platelets and presumably was derived from them. In macrophage-depleted mice, small, TO− platelets always predominated, although now a bell-shaped curve of large TO+ platelets can be seen (Figure 4C, bottom, and D, bottom).
Understanding the rapid clearance of the small platelets
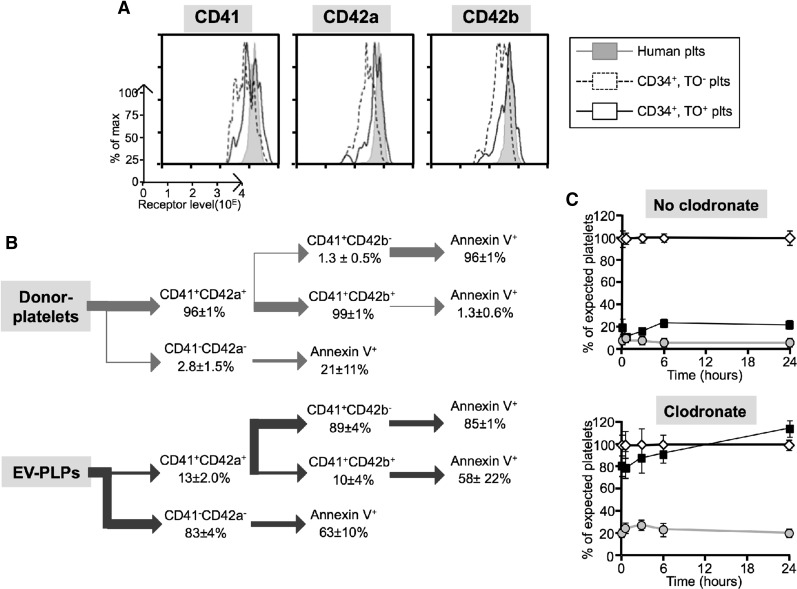
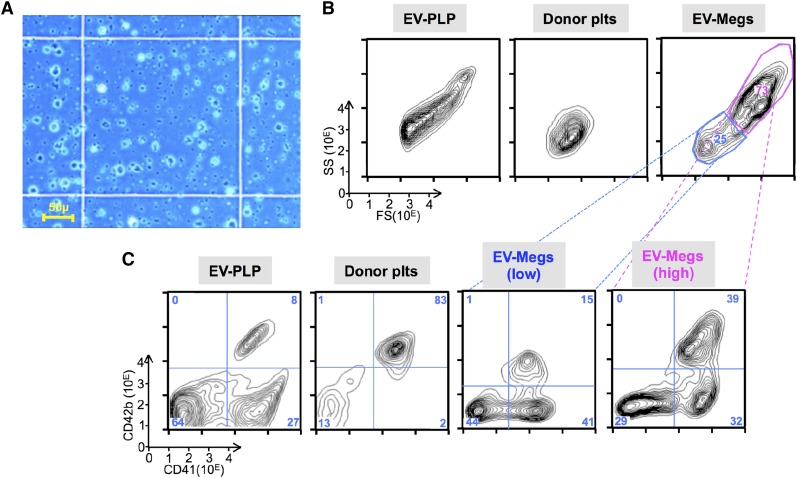
Prior studies suggest that murine EV-platelets can be damaged by extracellular metalloproteinases, resulting in short circulating times in recipient mice.46 One indicator of such metalloproteinase activity is loss of surface CD42b expression relative to CD41 (αIIb) and CD42a (GP9). We compared surface expression levels of CD42b with CD42a and CD41 on IV-platelets from infused AMC EV-megakaryocytes relative to infused donor platelets. There was no deficiency in surface levels of CD42b to CD42a on TO+ IV-platelets from infused EV-megakaryocytes compared with each other and to levels on concurrently studied donor platelets (Figure 5A). However, although >99% of donor platelets are CD42b+, in studies of isolated EV-PLPs, only ∼10% of its CD41+ subpopulation were CD42b+ (Figure 5B). This low level on EV-PLPs contrasts to EV-megakaryocytes, where ∼48% to 65% of CD41+ megakaryocytes are CD42b+ (supplemental Table 1).
Figure 5.
Rapid clearance of small human platelets. (A) Representative flow cytometric studies of surface receptor level for CD41, CD42a, and CD42b on infused human platelets (gray graph) or young and old platelets after AMC EV-megakaryocytes (dashed and solid line graph, respectively) at 1 hour after infusion. (B) Data summary of flow cytometric studies of isolated human donor platelets (top) or EV-PLPs (bottom) stained for the indicated surface markers or Annexin V binding. Mean ± SEM for each subpopulation. N = 3 separate studies for donor platelets and N = 4 for EV-PLPs, each in duplicate. (C) Human platelets either nonactivated (open diamonds) or after TRAP (filled squares) or CVX activation (gray circles) were infused into recipient NSG mice that were or were not preinfused with clodronate-ladened liposomes. The counts in the activated studies were normalized to parallel studies with nonactivated donor platelets. Mean ± SEM is shown. N = 5 studies, each done in duplicate.
An alternative explanation for the rapid clearance of the small platelets is that they may be preactivated during culture. To examine whether preactivation of platelets could also account for their rapid clearance, donor platelets were activated with either TRAP or CVX before infusion. Both agonists lead to rapid clearance of the platelets after infusion (Figure 5C, top). Following macrophage depletion, TRAP-activated platelets had a marked improved half-life similar to that seen with small platelets (Figure 5C, bottom). This improvement was not observed with the stronger agonist CVX. Consistent with platelet activation contributing to rapid, small platelet clearance, we found that both CD42b+ and CD42b− EV-platelets expressed Annexin V (indicative of activation) (Figure 5B). These data suggest that ≥2 mechanisms may account for the rapid clearance of EV-PLP in NSG mice: loss of CD42b and preactivation.
Reactivity of EV and IV platelets
How responsive are IV-platelets to agonists? As expected, infused donor platelets were quiescent before agonist exposure and highly responsive to CVX independent of platelet age or size (supplemental Figure 4A-B). In contrast, baseline platelet activation status after EV-megakaryocyte infusion was high when measured 1-hour after infusion. By 6 hours, the remaining platelets were quiescent and showed a robust response to CVX, although not as vigorous as seen with infused donor platelets.
Active incorporation of human platelets into thrombi
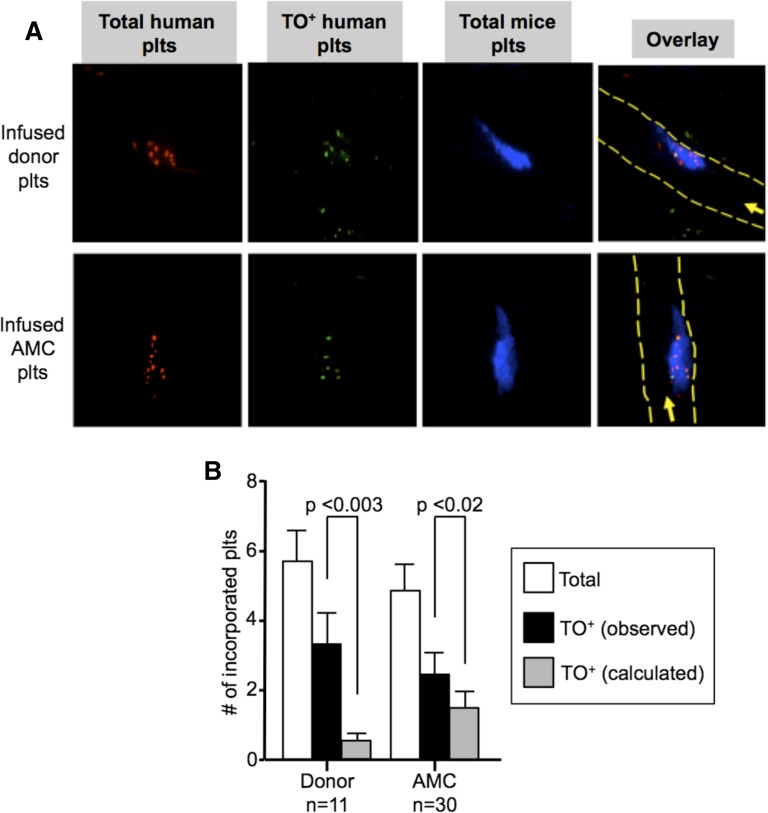
Are the small, old, and preactivated EV-platelets or the large, young, and quiescent IV-platelets better incorporated into thrombi? We studied human platelet incorporation into growing thrombi in mice in the absence or presence of tirofiban, a selective blocker of human αIIbβ3 binding to fibrinogen.19 Donors platelets were selectively incorporated into growing thrombi (supplemental Figure 5A) as were platelets derived from infused EV-megakaryocytes (supplemental Figure 5B). For both, we demonstrate that circulating TO+ platelets are selectively incorporated into thrombi over TO− platelets (Figure 6A-B).
Figure 6.
In situ studies of incorporation of TO+ vs TO− platelets into growing thrombi. (A) Representative confocal images after cremaster arteriole laser injuries in NSG mice done 0.5 to 1 hour after infusion of (top) human donor platelets or (bottom) AMC EV-megakaryocytes double-labeled with calcein violet (red) and TO (green) with double-labeled human platelets being yellow in the overlay (right). Incorporated murine platelets into the thrombi are in blue. In the overlay, the direction of flow is indicated by an arrow and the outline of the vessels by dashed yellow lines. (B) Mean ± SEM of total number of human platelets per thrombus (open bars), observed number of TO+ platelets per thrombus (black bars), and calculated number of TO+ platelets per thrombus based on the percent of human platelets in the circulation based on level of circulating human platelets determined on concurrent flow cytometric studies (gray bars). On the left are studies done after infused human donor platelets and on the right are the same after infused AMC EV-megakaryocytes.
EV-PLP infusion studies
Figure 5B shows that >80% of the EV-PLP population was CD41−CD42−, with ∼65% of these cells expressing Annexin V. Compared with EV-megakaryocytes (Figure 4A), EV-PLP preparations are dominated by PLPs and small cells (Figure 7A-B); a distribution comparable to studies by others of EV-PLPs.16-18 The EV-PLP CD41+/CD42b+ platelets made up <10% of the total EV-PLPs and <25% of total CD41+ platelets (Figure 7C). Infusions of EV-PLPs resulted in 2 circulating populations: small CD41+ platelets that cleared rapidly and a population of large platelets seen best at 6 hours, likely IV-platelets from residual EV-megakaryocytes (top half, supplement Figure 6A-B). Macrophage depletion improved the survival of the small platelets. The second population was the CD41− nonplatelets, which had a better circulating half-life than CD41+ cells. Here macrophage depletion worsened survival (bottom half; supplemental Figure 6A-B).
Figure 7.
Analysis of AMC EV-PLP preparation. (A) Representative field of an EV-PLP preparation on a hematocytometer before infusion. Bar in left lower corner indicates size of the observed field. (B) Representative size distribution of particles of the total preparation as determined by forward-scatter (FS) vs side-scatter (SS) for (left) EV-PLPs, (center) donor platelets, and (right) EV-megakaryocytes. (C) Representative surface expression of human CD41 and CD42b done on the same samples as in B. Percent of cells in each of 4 quadrants are noted. The AMC-megakaryocytes total preparation was also fractionated and gave low CD41+ (blue, 25% of the total) and high CD41+ (magenta, 73% of total) size preparations.
Discussion
EV-PLPs prepared from megakaryocytes have multiple functional deficiencies, some of which have been noted previously.16-18 The size range of PLPs is much wider than donor platelets; however, often studies have focused on a platelet-size gated window or selectively examined individual PLPs rather than the whole population for biology. Those studies still found limited reactivity of EV-PLPs similar to that observed here; however, this limited reactivity was not as notable in those studies as positive controls also showed limited reactivity, especially using cord blood platelets, which are known to have low reactivity.47 The short-half-life seen with EV-platelets in NSG mice was also noted previously,18 but in that study, donor platelet control also had a short half-life. In that study, both EV-platelet and donor platelet half-lives improved markedly after macrophage depletion. Although recipient mouse strain differences may explain why in our studies donor platelet half-lives was relatively long, we believe that baseline platelet preactivation better explains the difference between studies. Our data of a relatively long half-life for infused donor platelets in immunocompromized mice are consistent with others32 and are supported by Figure 5C, showing the effect of preactivation on infused donor-platelet half-lives.
In our studies, 85% of the EV-PLPs are CD41−, whereas others have reported this admixture to be 15% to 80%.17,18 We show that CD41− particles circulate well in recipient mice and are not cleared by macrophages. Whether they are incorporated into thrombi is unclear as is their other biologic impact. Certainly CD41− EV-PLPs complicate interpretation of proplatelet release in culture. Are the megakaryocytes present in these cultures releasing platelets physiologically or by a pathway shared with CD41− cells? CD41+ EV-PLPs often are preactivated and/or have lost CD42b antigenicity. Are these a consequence of the megakaryocytes shedding by a nonphysiologic pathway? Thrombopoiesis in a plastic environment without the presence of appropriate supportive cells and stroma48 and/or physical-chemical gradients8 may be nonphysiologic and may contribute to the many limitations of EV-PLPs.
On the other hand, IV-platelets released from infused EV-megakaryocytes are more similar to donor platelets in regard to shape, size, granule distribution, half-life, and surface marker expression, supporting the idea that megakaryocytes that have migrated out of the marrow space could release physiologic platelets intrapulmonary. These data must be interpreted with caution because the murine pulmonary bed and circulatory system have much higher shear and perhaps different branching that may change the details of pulmonary thrombopoiesis from what might be observed in humans. Nonetheless, the release of normal-sized platelets is consistent with studies that the specifics of the megakaryocyte rather than the pulmonary bed determine platelet size.40 In that case, would any vascular bed that entraps mature megakaryocytes support thrombopoiesis? It has been proposed that the cause of thrombocytopenia in cyanotic right-to-left congenital heart disease is that a portion of the blood bypasses the lungs,49 suggesting that the lung is uniquely designed for thrombopoiesis.
Although the biology of IV-platelets approaches infused donor platelets, they fall short in several ways. One would expect that newly released IV-platelets would have a longer half-life than donor platelets. The fact that the half-lives are nearly identical suggests that either platelet survival is not age dependent or that IV-platelets are less stable than donor platelets. CVX responsiveness by IV-platelets was less than that of donor platelets, and TO+ IV-platelets were incorporated relatively less than TO+ donor platelets into thrombi. As shown in supplemental Table 1, only ∼65% of EV-megakaryocytes are CD42b+, supporting that these megakaryocytes are injured to a degree. Also, EV-megakaryocytes are grown in media lacking many proteins normally incorporated into α-granules, which would could limit their reactivity. Differences between the individuals providing the donor platelets and the EV-megakaryocytes is an alternative explanation. Finally, approaches to optimize CD41High megakaryocyte purity and yield, as well as strategies to limit EV-megakaryocyte damage, would be needed to allow a more accurate determination of IV-platelet yield per infused megakaryocyte than the likely underestimates in Table 1 and to develop cells for potential clinical application.
Our studies show that infused human AMC, FLC, and iPSC EV-megakaryocytes can lead to IV-platelets. This paper focused on AMC EV-megakaryocytes, but also examined FLC and iPSC EV-megakaryocytes. The relative balance between EV- vs IV-platelets varied between sources, with IPSCs having the least number of IV-platelets, explaining their overall short initial half-life, which is similar to that of EV-PLP infusion (Figure 1B).
Presently, the mainstay of platelet support is donor-derived platelets. Concerns over quality and quantity issues with these platelets have been noted,50 but at the same time, experience with such donor platelets is extensive, and cost of product procurement is contained by voluntary donation. Any replacement should have important advantages over the present product. These advantages could include reduced risks of pathogen transmission, standardization of product, unlimited supply, and ability to modify the platelets to improve platelet survival or broaden functionality. Above all, these alternative products should meet high standards of reactivity and half-life. This paper shows that EV-PLPs in our culture conditions fall short in this regard: they are an admixture of CD41− and CD41+ particles. The CD41+ subpopulation is short lived, preactivated, weakly reactive, and underincorporated into thrombi. EV-PLPs generated in bioreactors51-55 have been described and such high-throughput devices are clearly needed to achieve large-scale EV-platelet production. Like infused EV-megakaryocytes, they would have the advantage of producing mostly young platelets. Bioreactor-generated platelets would not have some of the disadvantages of infusing EV-megakaryocytes, including transient occlusion of a portion of the pulmonary vascular bed, delayed platelet release, and introduction of nucleated cells rather than nuclear-free platelets. However, at present, no bioreactor-derived products have been infused into recipient mice and shown to have biological properties approaching well-prepared, donor platelets as has been achieved with infused EV-megakaryocytes.
Acknowledgments
This work was supported by funding from the National Institutes of Health, National Heart, Lung, and Blood Institute (NHLBI) grant U01 HL099656 and National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) grant P30 DK090969 (S.T.C., D.L.F., P.G., M.P., and S.K.S.), NHLBI grant U01 HL099993 and NIDDK grant P30 DK056465 (B.J.T.-S.), NHLBI grant P01 HL064190 (M.P.), NHLBI grant R01 HL121323 (M.S.M.), and the Jane Fishberg Grinberg Chair (M.P.).
Footnotes
The online version of this article contains a data supplement.
There is an Inside Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: Y.W. designed and performed most of the studies and was primary author of the manuscript; V.H. performed the cremaster studies; R.F. helped with early studies for this manuscript; D.J. and X.S. carried out the characterization of the EV-PLPs; S.K.S., P.G., and D.L.F. helped with the induced pluripotent stem cell studies and with manuscript editing; S.T.C. helped with the fetal liver megakaryocytes studies and with manuscript editing; D.C.H. and M.S.M. performed the fluorescence microscopy studies; B.J.T.-S. helped with critical analysis of the data and manuscript editing; and M.P. provided overall scientific guidance and manuscript preparation.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Mortimer Poncz, Children’s Hospital of Philadelphia, 3615 Civic Center Blvd, 317 ARC, Philadelphia, PA 19104; e-mail: poncz@email.chop.edu.
References
- 1.Ware J, Corken A, Khetpal R. Platelet function beyond hemostasis and thrombosis. Curr Opin Hematol. 2013;20(5):451–456. doi: 10.1097/MOH.0b013e32836344d3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rondina MT, Weyrich AS, Zimmerman GA. Platelets as cellular effectors of inflammation in vascular diseases. Circ Res. 2013;112(11):1506–1519. doi: 10.1161/CIRCRESAHA.113.300512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ho-Tin-Noé B, Demers M, Wagner DD. How platelets safeguard vascular integrity. J Thromb Haemost. 2011;9(Suppl 1):56–65. doi: 10.1111/j.1538-7836.2011.04317.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Weyrich AS, Zimmerman GA. Platelets in lung biology. Annu Rev Physiol. 2013;75:569–591. doi: 10.1146/annurev-physiol-030212-183752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Machlus KR, Italiano JE., Jr The incredible journey: From megakaryocyte development to platelet formation. J Cell Biol. 2013;201(6):785–796. doi: 10.1083/jcb.201304054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kaufman RM, Airo R, Pollack S, Crosby WH. Circulating megakaryocytes and platelet release in the lung. Blood. 1965;26(6):720–731. [PubMed] [Google Scholar]
- 7.Junt T, Schulze H, Chen Z, et al. Dynamic visualization of thrombopoiesis within bone marrow. Science. 2007;317(5845):1767–1770. doi: 10.1126/science.1146304. [DOI] [PubMed] [Google Scholar]
- 8.Zhang L, Urtz N, Gaertner F, et al. Sphingosine kinase 2 (Sphk2) regulates platelet biogenesis by providing intracellular sphingosine 1-phosphate (S1P). Blood. 2013;122(5):791–802. doi: 10.1182/blood-2012-12-473884. [DOI] [PubMed] [Google Scholar]
- 9.Zhang L, Orban M, Lorenz M, et al. A novel role of sphingosine 1-phosphate receptor S1pr1 in mouse thrombopoiesis. J Exp Med. 2012;209(12):2165–2181. doi: 10.1084/jem.20121090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aschoff L. Ueber capillaire embolie von riesenkernhaltigen Zellen Virchow’s Arch Path Anat. 1893;134:11.
- 11.Howell WH, Donahue DD. The production of blood platelets in the lungs. J Exp Med. 1937;65(2):177–203. doi: 10.1084/jem.65.2.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kuter DJ. New drugs for familiar therapeutic targets: thrombopoietin receptor agonists and immune thrombocytopenic purpura. Eur J Haematol Suppl. 2008;(69):9–18. doi: 10.1111/j.1600-0609.2007.00999.x. [DOI] [PubMed] [Google Scholar]
- 13.Thiagarajan P, Afshar-Kharghan V. Platelet transfusion therapy. Hematol Oncol Clin North Am. 2013;27(3):629–643. doi: 10.1016/j.hoc.2013.03.004. [DOI] [PubMed] [Google Scholar]
- 14.Takayama N, Nishimura S, Nakamura S, et al. Transient activation of c-MYC expression is critical for efficient platelet generation from human induced pluripotent stem cells. J Exp Med. 2010;207(13):2817–2830. doi: 10.1084/jem.20100844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Takayama N, Eto K. In vitro generation of megakaryocytes and platelets from human embryonic stem cells and induced pluripotent stem cells. Methods Mol Biol. 2012;788:205–217. doi: 10.1007/978-1-61779-307-3_15. [DOI] [PubMed] [Google Scholar]
- 16.Nakamura S, Takayama N, Hirata S, et al. Expandable megakaryocyte cell lines enable clinically applicable generation of platelets from human induced pluripotent stem cells. Cell Stem Cell. 2014;14(4):535–548. doi: 10.1016/j.stem.2014.01.011. [DOI] [PubMed] [Google Scholar]
- 17.Lu SJ, Li F, Yin H, et al. Platelets generated from human embryonic stem cells are functional in vitro and in the microcirculation of living mice. Cell Res. 2011;21(3):530–545. doi: 10.1038/cr.2011.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Feng Q, Shabrani N, Thon JN, et al. Scalable generation of universal platelets from human induced pluripotent stem cells. Stem Cell Rev. 2014;3(5):817–831. doi: 10.1016/j.stemcr.2014.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fuentes R, Wang Y, Hirsch J, et al. Infusion of mature megakaryocytes into mice yields functional platelets. J Clin Invest. 2010;120(11):3917–3922. doi: 10.1172/JCI43326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cortin V, Pineault N, Garnier A. Ex vivo megakaryocyte expansion and platelet production from human cord blood stem cells. Methods Mol Biol. 2009;482:109–126. doi: 10.1007/978-1-59745-060-7_7. [DOI] [PubMed] [Google Scholar]
- 21.Chou ST, Opalinska JB, Yao Y, et al. Trisomy 21 enhances human fetal erythro-megakaryocytic development. Blood. 2008;112(12):4503–4506. doi: 10.1182/blood-2008-05-157859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Somers A, Jean JC, Sommer CA, et al. Generation of transgene-free lung disease-specific human induced pluripotent stem cells using a single excisable lentiviral stem cell cassette. Stem Cells. 2010;28(10):1728–1740. doi: 10.1002/stem.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sullivan SK, Mills JA, Koukouritaki SB, et al. High-level transgene expression in induced pluripotent stem cell-derived megakaryocytes: correction of Glanzmann thrombasthenia. Blood. 2014;123(5):753–757. doi: 10.1182/blood-2013-10-530725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mills JA, Wang K, Paluru P, et al. Clonal genetic and hematopoietic heterogeneity among human-induced pluripotent stem cell lines. Blood. 2013;122(12):2047–2051. doi: 10.1182/blood-2013-02-484444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lordier L, Bluteau D, Jalil A, et al. RUNX1-induced silencing of non-muscle myosin heavy chain IIB contributes to megakaryocyte polyploidization. Nat Commun. 2012;3:717. doi: 10.1038/ncomms1704. [DOI] [PubMed] [Google Scholar]
- 26.Rauova L, Hirsch JD, Greene TK, et al. Monocyte-bound PF4 in the pathogenesis of heparin-induced thrombocytopenia. Blood. 2010;116(23):5021–5031. doi: 10.1182/blood-2010-03-276964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kienast J, Schmitz G. Flow cytometric analysis of thiazole orange uptake by platelets: a diagnostic aid in the evaluation of thrombocytopenic disorders. Blood. 1990;75(1):116–121. [PubMed] [Google Scholar]
- 28.Nieswandt B, Schulte V, Bergmeier W. Flow-cytometric analysis of mouse platelet function. Methods Mol Biol. 2004;272:255–268. doi: 10.1385/1-59259-782-3:255. [DOI] [PubMed] [Google Scholar]
- 29.Hu Z, Yang YG. Full reconstitution of human platelets in humanized mice after macrophage depletion. Blood. 2012;120(8):1713–1716. doi: 10.1182/blood-2012-01-407890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Meng R, Wu J, Harper DC, et al. Defective release of α granule and lysosome contents from platelets in mouse Hermansky-Pudlak syndrome models. Blood. 2015;125(10):1623–1632. doi: 10.1182/blood-2014-07-586727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Basani RB, Zhu H, Thornton MA, et al. Species differences in small molecule binding to alpha IIb beta 3 are the result of sequence differences in 2 loops of the alpha IIb beta propeller. Blood. 2009;113(4):902–910. doi: 10.1182/blood-2008-09-177337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Boylan B, Berndt MC, Kahn ML, Newman PJ. Activation-independent, antibody-mediated removal of GPVI from circulating human platelets: development of a novel NOD/SCID mouse model to evaluate the in vivo effectiveness of anti-human platelet agents. Blood. 2006;108(3):908–914. doi: 10.1182/blood-2005-07-2937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Canals M, Olivares R, Rosenmann M. A radiographic method to estimate lung volume and its use in small mammals. Biol Res. 2005;38(1):41–47. doi: 10.4067/s0716-97602005000100006. [DOI] [PubMed] [Google Scholar]
- 34.Economopoulos V, Noad JC, Krishnamoorthy S, Rutt BK, Foster PJ. Comparing the MRI appearance of the lymph nodes and spleen in wild-type and immuno-deficient mouse strains. PLoS ONE. 2011;6(11):e27508. doi: 10.1371/journal.pone.0027508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schagatay E, Richardson MX, Lodin-Sundström A. Size matters: spleen and lung volumes predict performance in human apneic divers. Front Physiol. 2012;3:173. doi: 10.3389/fphys.2012.00173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rowan RM, Fraser C, Gray JH, McDonald GA. The Coulter Counter Model S Plus—the shape of things to come. Clin Lab Haematol. 1979;1(1):29–40. doi: 10.1111/j.1365-2257.1979.tb00587.x. [DOI] [PubMed] [Google Scholar]
- 37.Eason CT, Pattison A, Howells DD, Mitcheson J, Bonner FW. Platelet population profiles: significance of species variation and drug-induced changes. J Appl Toxicol. 1986;6(6):437–441. doi: 10.1002/jat.2550060610. [DOI] [PubMed] [Google Scholar]
- 38.Weingand KW, Odioso LW, Dameron GW, Laytart MJ, Stitzel KA. Hematology analyzer comparison: Ortho ELT-8/ds vs. Baker 9000 for healthy dogs, mice,and rats. Vet Clin Pathol. 1992;21(1):10–14. doi: 10.1111/j.1939-165x.1992.tb00575.x. [DOI] [PubMed] [Google Scholar]
- 39.Thon JN, Montalvo A, Patel-Hett S, et al. Cytoskeletal mechanics of proplatelet maturation and platelet release. J Cell Biol. 2010;191(4):861–874. doi: 10.1083/jcb.201006102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Thon JN, Macleod H, Begonja AJ, et al. Microtubule and cortical forces determine platelet size during vascular platelet production. Nat Commun. 2012;3:852. doi: 10.1038/ncomms1838. [DOI] [PubMed] [Google Scholar]
- 41.Holme S, Heaton A, Konchuba A, Hartman P. Light scatter and total protein signal distribution of platelets by flow cytometry as parameters of size. J Lab Clin Med. 1988;112(2):223–231. [PubMed] [Google Scholar]
- 42.Joutsi-Korhonen L, Sainio S, Riikonen S, Javela K, Teramo K, Kekomäki R. Detection of reticulated platelets: estimating the degree of fluorescence of platelets stained with thiazole orange. Eur J Haematol. 2000;65(1):66–71. doi: 10.1034/j.1600-0609.2000.90184.x. [DOI] [PubMed] [Google Scholar]
- 43.Berny-Lang MA, Frelinger AL, III, Barnard MR, Michelson AD. Flow cytometry. In: Michelson AD, editor. Platelets. 3rd ed. Waltham, MA: Academic Press; 2012. pp. 581–602. [Google Scholar]
- 44.Diquattro M, Gagliano F, Calabrò GM, et al. Relationships between platelet counts, platelet volumes and reticulated platelets in patients with ITP: evidence for significant platelet count inaccuracies with conventional instrument methods. Int J Lab Hematol. 2009;31(2):199–206. doi: 10.1111/j.1751-553X.2007.01025.x. [DOI] [PubMed] [Google Scholar]
- 45.Koh KR, Yamane T, Ohta K, Hino M, Takubo T, Tatsumi N. Pathophysiological significance of simultaneous measurement of reticulated platelets, large platelets and serum thrombopoietin in non-neoplastic thrombocytopenic disorders. Eur J Haematol. 1999;63(5):295–301. doi: 10.1111/j.1600-0609.1999.tb01131.x. [DOI] [PubMed] [Google Scholar]
- 46.Nishikii H, Eto K, Tamura N, et al. Metalloproteinase regulation improves in vitro generation of efficacious platelets from mouse embryonic stem cells. J Exp Med. 2008;205(8):1917–1927. doi: 10.1084/jem.20071482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sitaru AG, Holzhauer S, Speer CP, et al. Neonatal platelets from cord blood and peripheral blood. Platelets. 2005;16(3-4):203–210. doi: 10.1080/09537100400016862. [DOI] [PubMed] [Google Scholar]
- 48.Kulawig R, Stegner D, Meyer I, Nieswandt B, Schulze H. Competitive collagen I-GPVI / collagen IV- α2β1-integrin signaling modulates proplatelet formation. Platelets. In: Proceedings of the 8th International Platelet Symposium; April 3-6, 2014; Maále Hachamisha, Israel. [Google Scholar]
- 49.Lill MC, Perloff JK, Child JS. Pathogenesis of thrombocytopenia in cyanotic congenital heart disease. Am J Cardiol. 2006;98(2):254–258. doi: 10.1016/j.amjcard.2006.01.083. [DOI] [PubMed] [Google Scholar]
- 50.Lambert MP, Sullivan SK, Fuentes R, French DL, Poncz M. Challenges and promises for the development of donor-independent platelet transfusions. Blood. 2013;121(17):3319–3324. doi: 10.1182/blood-2012-09-455428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dunois-Lardé C, Capron C, Fichelson S, Bauer T, Cramer-Bordé E, Baruch D. Exposure of human megakaryocytes to high shear rates accelerates platelet production. Blood. 2009;114(9):1875–1883. doi: 10.1182/blood-2009-03-209205. [DOI] [PubMed] [Google Scholar]
- 52.Pallotta I, Lovett M, Kaplan DL, Balduini A. Three-dimensional system for the in vitro study of megakaryocytes and functional platelet production using silk-based vascular tubes. Tissue Eng Part C Methods. 2011;17(12):1223–1232. doi: 10.1089/ten.tec.2011.0134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nakagawa Y, Nakamura S, Nakajima M, et al. Two differential flows in a bioreactor promoted platelet generation from human pluripotent stem cell-derived megakaryocytes. Exp Hematol. 2013;41(8):742–748. doi: 10.1016/j.exphem.2013.04.007. [DOI] [PubMed] [Google Scholar]
- 54.Thon JN, Mazutis L, Wu S, et al. Platelet bioreactor-on-a-chip. Blood. 2014;124(12):1857–1867. doi: 10.1182/blood-2014-05-574913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Di Buduo CA, Wray LS, Tozzi L, et al. Programmable 3D silk bone marrow niche for platelet generation ex vivo and modeling of megakaryopoiesis pathologies. Blood. 2015;125(14):2254–2264. doi: 10.1182/blood-2014-08-595561. [DOI] [PMC free article] [PubMed] [Google Scholar]