Abstract
Chronic lymphocytic leukemia (CLL) is a malignancy arising from immune cells (B-lymphocytes) endowed with intrinsic antigen-presenting capabilities. Such a function however is lost during malignant transformation and CLL cells are well known for their inability to process and present antigens to the T-cell arm of the immune system. Instead, malignant CLL cells elicit a vast array of immune regulatory mechanisms conducive to T-cell dysfunction and immunosupression. Previously, we have shown that treatment of CLL cells with the demethylating agent 5-aza-2′-deoxycytidine unleashed target antigen expression. Here we show for the first time that combining two epigenetic modifiers, 5-aza-2′-deoxycytidine and the histone deacetylase inhibitor LAQ824 effectively restores the immunogenicity of CLL cell lines as well as primary cells obtained from CLL patients. Indeed, such a combination induces the expression of novel and highly antigenic cancer testis antigens (CTAs) and co-stimulatory molecules. These changes facilitate the formation of robust supramolecular activation complexes (SMAC) between CLL cells and responder T-cells leading to intracellular signaling, lytic granule mobilization, and polarization of functional and relevant T-cell responses. This cascade of T-cell activating events triggered by CLL cells with restored APC function, points to combined epigenetic modifier treatment as a potential immunotherapeutic strategy for CLL patients.
Keywords: 5-aza-2′-deoxycytidine, Cancer-germline, Immunotherapy, Histone Deacetylase Inhibitor, Costimulatory
Introduction
Chronic lymphocytic leukemia (CLL) is the most common B-cell leukemia in the western world and is characterized by the progressive accretion of long-lived mature B-lymphocytes which maintain a low proliferation rate [1–4]. Despite recent advances in disease pathogenesis and treatment CLL remains incurable using conventional treatments and an allogenic stem cell transplant is the only curative approach [4–7]. The classic clinical staging system developed by Rai and Binet has been able to predict long term survival of patients with CLL and has been directly correlated with a worsening immune dysfunction [1, 8, 9]. Given a prolonged disease course immunotherapeutic approaches are at the forefront of novel treatments which have shown some promise in CLL [5]. Immunotherapeutic strategies which complement standard therapies, such as monoclonal antibodies, are already routine and have the potential to alter disease course [10, 11]. Since the majority of patients are of advanced age, an immunotherapeutic approach may be more suitable than aggressive chemotherapeutic regimens that exacerbate an already compromised immune system [1]. A recent example is the emergence of novel immunomodulatory compounds (iMIDs) that has brought to light the importance of immunoregulation and stromal interactions in the management of CLL [12–15].
In healthy individuals B-lymphocytes are proficient stimulators of antigen specific Th1 T-cell responses capable of eliminating virally infected or neoplastic cells via direct cellular cytotoxicity [16]. However, in CLL an increasingly immunosuppressive phenotype enables the malignant B-cell to evade immune detection [3, 17–19]. Mounting evidence points to specific defects in the antigen presenting cell (APC) functions including decreased self-peptide presentation, improper T-cell synapse formation, deficient costimulation, and diversionary cytokine signaling.
Although the distinct molecular mechanisms which elicit CLL-induced immunosupression remain unclear, recent evidence supports the notion that chromatin constriction of critical immunostimulatory factors may be involved [20, 21]. Moreover, promoter hypermethylation and histone hypoacetylation are primary mechanisms for silencing highly antigenic protein products which promote the immune recognition of malignant cells. Novel strategies have been suggested which may supplant immunosupression with effective cancer-antigen presentation leading to robust T-cell activation and prolonged killing [11, 22, 23]. The hallmarks of such strategies hinge upon functionally increasing the APC capacity of CLL cells by reducing the levels of immunosuppressive factors, generating a robust response against a non-tolerized cancer-specific antigen, and properly polarizing the resulting primed T-cells. Recent studies have demonstrated increased expression of MHC class I class II, immunoproteosome subunits, peptide processing machinery, pro-apoptotic molecules such as APO2L/TRAIL, and various costimulatory molecules after treatment with a histone deacetylase inhibitor (HDACi) leading to enhanced cancer-specific immune responses [24–26]. Additionally, both HDACi and DNA methyltransferase inhibitors (DNMTi), such as 5-aza-2′-deoxycytidine (5A2), have proven capable of inducing long lasting, cancer-specific, expression of a highly antigenic class of proteins, termed cancer-testis antigens (CTAs)[27]. Published studies demonstrate that this effect alone may potentiate effective anti-cancer immune responses [28]. Furthermore, a role for epigenetic modifiers in the regulation of pro-inflammatory cytokines has also been demonstrated [29, 30]. There currently exists a single clinical trial investigating the efficacy of 5-aza-2′-deoxycytidine in combination with the HDACi valproic acid in previously treated CLL patients, the final results of this trial are still pending [31].
Based on studies in other malignancies and our prior investigations we hypothesized that treating CLL cells with HDACi and DNMTi would potentiate APC function via restoring antigenic protein expression, costimulatory signaling, synapse formation, and cytokine polarization, leading to robust anti-CLL immune responses. Our studies demonstrate in both cell lines and primary CLL samples that combination therapy utilizing 5A2 and LAQ824 (LAQ) significantly increased the expression of multiple CTAs and costimulatory molecules. This facilitates the formation of robust supramolecular activation complexes (SMAC) between CLL cells and responder T-lymphocytes leading to signaling, lytic granule mobilization, Th1 polarization, and induction of functionally relevant T-cell responses.
Materials and Methods
Subject Populations
Sera and peripheral blood mononuclear cells (PBMCs) were obtained from patients with CLL. All subjects gave written institutional review board (IRB)-approved informed consent for their blood products to be used for research. Blood was collected at the H. Lee Moffitt Cancer Center (Tampa, FL). PBMCs were stored in 1ml aliquots at −140°C and sera were stored in aliquots at −80°C until used.
Cell Culture and Drug Treatments
Unless otherwise stated cells were cultured in-vitro at 37°C and 5%CO2 using RPMI1640 medium supplemented with 10% fetal calf serum and antibiotics. The MEC1, MEC2, and WaC3 cell lines were kindly provided by Dr. John Byrd at Ohio State University and were previously characterized in the following references [32, 33]. Drug treatments were carried out on the CLL cells (separate from T-cells) for 72hr in complete medium using either 1uM 5-aza-2′-deoxycytidine (5A2), 25nM LAQ824 (LAQ), 1uM5A2 + 25nM LAQ, or neither. At the conclusion of treatment cells were washed twice using pre-warmed serum-free RPMI1640 and subjected to the various assays. For treatments involving primary CLL cells, cultures of purified B-CLL cells (>95% purity) were maintained in 6 well plates atop irradiated (30Gy) monolayers of CD40L expressing murine fibroblasts (a kind gift from Dr. John Gordon at the University of Birmingham) in the presence of 500U/ml recombinant human IL-4 (Research Diagnostic Inc., Concord, MA)
Reverse Transcriptase-PCR (RT-PCR)
Total RNA was prepared from pelleted cells (RNeasy mini columns and RNAse free DNAse, Qiagen, Valencia, CA) or was commercially obtained (BioChain, Hayward, CA). RT-PCR reactions were conducted using the Qiagen one-step RT-PCR kit (Qiagen) with transcript-specific primers and total RNA from CLL cell lines as templates. RT-PCR amplification reactions were resolved on 2% agarose gels and the size of the amplified transcript confirmed by comparison with a standard DNA ladder (GelPilot 1Kb Plus Ladder, Qiagen). Heatmap depictions were created using Image Quant 5.1 software (Molecular Dynamics) and the Heatmap Builder tool kindly provided by the Quertermous lab at Stanford.
Cell Conjugation Assays
Healthy or malignant B-cells were stained with CellTracker Blue (PKH26 for flow cytometric analysis) following the manufacturer’s instructions and pulsed with or without 2ug/ml of a cocktail of staphylococcal superantigens (SEA and SEB; Toxin Technologies; Sarasota, FL) for 30min at 37°C. B-cells were then centrifuged (200g for 5min) with 5 times the number of T cells stained with mitotracker deep-red (Invitrogen, Carlsbad, CA) (or CFSE for flow cytometric analysis) and were incubated at 37°C for 10min unless otherwise stated, then plated onto poly-L-lysine slides and fixed using 3.7% formaldehyde in PBS for 15min. Additional antibody staining was conducted using 1:50 anti-perforin (Pierce, Rockford, IL) followed by 1:125 anti-mouse FITC (Sigma). Microscopic acquisition and analysis of immune synapses was conducted using a Leica upright fluorescent confocal microscope with the associated software according to the methods developed by Ramsay et. al. [1]. Controls which lacked sufficient conjugation to acquire microscopic images (controls without SEA/SEB, without APCs, or without T-cells) were investigated and documented in low-resolution wide-field images showing no significant conjugation, unless otherwise stated. All microscopy experiments were repeated three independent times unless otherwise stated.
Flow Cytometry Immunophenotyping
Flow cytometric analysis of cultured cells was performed using fluorochrome-labeled monoclonal antibodies (mAbs; anti-HLA-A,B,C, -HLA-DR, -CD40, -CD80, -CD86, -CD19, and –CD20, Becton Dickinson, San Jose, CA and eBiosciences, San Diego, CA) and the viability dye 4′,6-diamidino-2-phenylindole (DAPI, Sigma). For CFDA-SE (CFSE) staining cells were resuspended in 0.5μM CFSE (Invitrogen) in RPMI medium for 15 minutes at 37°C followed by a wash and additional 30 minute incubation in serum supplemented medium prior to resuspension in culture medium. For conjugation based T-cell phenotype experiments cells were allowed to conjugate according to the methods described in cell conjugation assays and conjugated cells were stained for anti-CD20 and anti-CD4. Conjugation events were sorted using a FACS Aria cell sorter and separate populations were assayed for cytokine production using CBA array. Cytokine bead array (CBA) (Becton Dickinson) was conducted according to the manufacturers published protocol using cellular supernatant from three replicate experiments. PKH26 staining was carried out and data was acquired on an LSRII cytometer (Beckman Coulter), and analyzed with FlowJo software (Tree Star, Ashland, OR). For analysis
Mixed Lymphocyte Proliferation
CLL cells or healthy B-cells were incubated in serum supplemented RPMI1640 (Invitrogen) with allogenic, ficoll density gradient separated, PBMCs from a healthy donor which had been stained 30 minutes prior with 0.5μM CFSE at a 1:10 effector:target (E:T) ratio. Cultures were maintained at 37°C for 60 hours and were subsequently subjected to flow cytometric analysis. Viability dye was used to gate out dead cells, CD4 and CD8 cells were stained using spectrally separate fluorophores and were individually investigated for CFSE dilution resulting from proliferation.
FACS Based Cytotoxicity Assay
Healthy allogenic T-cells were stained with CFSE as previously described. CLL cells were pulsed with SEA and SEB superantigens at 2ug/ml for 2 hours and were centrifugally collected and incubated with T-cells at a 1:10 E:T ratio for 45 minutes at 37°C in serum-free RPMI1640. Cells were then carefully chilled to 4°C, centrifuged, and resuspended in FACS buffer containing the viability dye propidium iodide. FACS analysis was immediately conducted and the percentage of T-cells conjugated with dead cells was tracked and compared to a T-cell only control. Cytotoxicity analyses were conducted in triplicate and experiments were repeated at least twice.
Lactate Dehydrogenase (LDH) Cytotoxicity Assay
Allogenic CD8 T-cells (or B-cells) were separated from healthy donor ficoll density purified PBMCs using magnetic negative selection on an MACS LS magnetic column (Miltenyi Biotec, Auburn, CA) using the manufacturer specified protocol. CD8 T-cells and purified primary CLL B-cells (>95%) were washed twice with 37°C RPMI1640 without phenol red/1%FCS. Cells were then incubated together at 1:100, 1:50, and 1:25 E:T ratios in RPMI1640 without phenol red/1%FCS in 96 well round bottom plates for 4 hours at 37°C/5%CO2. Cytotoxicity was measured using the cytotox96 non-radioactive LDH assay kit according to the manufacturers published protocol (Promega, Madison, WI). Control wells corresponding to minimum cytotoxicity (targets alone), maximum cytotoxicity (Triton X100 lysed targets), and culture medium alone were used to calculate experimental sample percent lysis.
Statistical Analysis
Unless otherwise stated, all continuous data conducted in replicates were analyzed using a two tailed Students T test. P values are displayed on the graph to indicate the level of significance. Error bars presented on graphical representations of data indicate standard deviation unless otherwise stated.
Results
Epigenetic modifiers synergistically induce CTA expression in CLL cells
Our prior studies in CLL indicate that 5A2 treatment alone is capable of inducing the expression of highly antigenic CTAs [34]. Given this prior knowledge we wanted to examine the combined effect of 5A2 and histone deacetylase inhibitors in this regard. Prior to initiating experimentation, we first wanted to confirm that our epigenetic modifiers were capable of altering chromatin structure in a CLL cell line. Previous studies have documented acetylation changes at conserved interferon gamma distal regulatory elements [35, 36]. To confirm the activity of 5A2, LAQ, and 5A2/LAQ in MEC1 cells, we profiled the acetylation changes occurring at the regulatory CNS+18 locus of the IFNG gene. As expected, our results demonstrated increased H4 acetylation in treated MEC1 cells indicating that both drugs were capable altering chromatin structure in a CLL cell line (Supplementary Figure 1).
We therefore compared the mRNA expression pattern of 29 known CTAs between our CLL cell lines WaC3, MEC1, and MEC2 and primary CLL samples by RT-PCR. As shown in Figure 1A there is diverse low-level expression of multiple CTAs including SPANXC, MAD-CT-1 and -2, LIP1, SPA-17, and many members of the MAGE CTA family. Additionally, mRNA expression of SSX-1, -2, and -4, NY-SAR-35, GAGE-2, -4, and -7, XAGE-1, NXF2, LAGE-1, NY-ESO-1, TPX-1, FATE-1, ADAM-2, and TSP50 was extremely low or nonexistent in the majority of CLL samples and the CLL cell lines. The observed CTA expression profile indicated that interpatient expression patterns are somewhat variable. Nevertheless, our CLL cell lines accurately mirror the CTA expression trends seen in ten random patients with primary CLL making them valuable tools for in-vitro studies.
Figure 1.

We next sought to improve upon basal expression patterns by treating our CLL cell lines with 5A2, LAQ, or a combination treatment of both 5A2 and LAQ (Figure 1B). mRNA expression data from these studies indicated that while both 5A2 and LAQ were capable of inducing the expression of varied CTAs only the combination treatment was capable of eliciting robust expression amongst virtually all of the CTAs tested including SSX-1, -2, and -4, NY-SAR-35, GAGE-2, and -7, XAGE-1, NXF2, LAGE-1, NY-ESO-1, TPX-1, FATE-1, ADAM-2, and TSP50, antigens which were nonexistent prior to treatment. While these results indicate a cooperative nature between the two drugs we also observed synergistic activity with respect to certain CTAs, namely NY-ESO-1, FATE-1, TPX-1, PAGE-1, and TSP-50.
Given that primary CLL cells do not proliferate ex-vivo and thus do not incorporate 5A2 into their DNA it has previously been challenging to study the demethylating effects of nucleoside analogs outside of in-vivo or cell culture systems. To circumvent this challenge we utilize a CD40L expressing murine fibroblast feeder cell line which, in conjunction with IL-4, induces limited proliferation in some human CLL primary cells in-vitro. Using this system we characterized the epigenetic changes to the CTA expression pattern in primary CLL cells. As shown in Figure 1C, we saw a marked increase in CTA transcription with 5A2 or LAQ alone, although maximal effects were only observed with the combination treatment. These data confirm our initial hypothesis by demonstrating robust CTA inducibility in primary cells using epigenetic modifiers.
Epigenetic modifiers modulate the costimulatory profile and cytokine signaling of B-CLL
Prior studies indicate that treatment of specific solid tumors with HDACi can improve the costimulatory phenotype and can increase tumor immunogenicity [22–25, 37]. These data along with our prior studies using 5A2 on CLL led us to investigate the possibility that 5A2 and LAQ may potentiate an improved costimulatory phenotype in CLL. Our flow cytometry profiling experiments revealed modest increases in the CLL cell line surface expression of CD86, CD80, HLA-DR, CD40, and MHC class I (HLA-A,B,C) after treatment with 1μM 5A2 and 25nM LAQ (Figure 2A). In two additional repeat experiments this modest upregulation was consistent. Given that CLL cells are inherently derived from APCs, it is possible that even slight increases in the costimulatory phenotype may tilt the balance towards the formation of an anti-CLL immune response.
Figure 2.
Along with costimulatory molecules B-cells also provide critical immunostimulatory and immunosuppressive cytokine signals. B-CLL cells in particular, secrete high levels of immunosuppressive factors which inhibit the generation of anti-CLL T-cell responses. One such potent immunosuppressive factor is IL-10 [38]. In light of this we decided to investigate the IL-10 secretion by CLL cell lines after treatment with 5A2 and LAQ. Results from our cytokine analysis indicated that 5A2 and LAQ had the potential to independently inhibit IL-10 secretion in particular cell lines, however the combination of both inhibitors was essential to obtain significant inhibition in all cell lines, indicating that combination 5A2+LAQ therapy may release CLL-induced T-cell suppression (Figure 2B).
DNA demethylation and histone acetylation cooperate to increase the potency of the CLL cell – T-cell interaction
Our results thus far point to improved antigen expression, costimulatory phenotype, and cytokine signaling which have been postulated to directly correlate with the formation of a healthy immunological synapse, or SMAC. To further investigate the changes in the resulting immune synapse we utilized superantigen (sAg) stimulation via staphylococcal enterotoxins A and B to induce a TCR mediated response in healthy allogenic donor CD8 or CD4 purified T-cells. As APCs we utilized 5A2 and LAQ treated CLL cell lines or alternatively healthy allogenic B-cells as a control. Our confocal imaging data suggested that the untreated CLL cells rarely formed robust interaction complexes with either CD8 or CD4 T-cells. In sharp contrast, after treatment with 5A2, LAQ, or both, SMAC complexes were readily abundant, robust, symmetrical, and concave, indicative of more effective and prolonged APC signaling (Figure 3A and 3B). To quantify these data we manually counted and scored 100 synaptic events for each experiment (Figure 3C) according to the methodology developed by Ramsay et. al. and demonstrated in Supplementary Figure 2 [1]. Data showed a significant shift towards increasingly numerous and robust interaction zones after treatment, with optimal actin polarization observed with combination 5A2+LAQ treatment.
Figure 3.
To further confirm our microscopy data, we conducted flow cytometric sAg-induced conjugation assays between healthy allogenic CD8 or CD4 T-cells and our CLL cell lines. As depicted in an example flow plot (Figure 4A) the percentage of TCR specific conjugated cells was determined by subtracting the conjugates forming in an unstimulated sample from those forming in the sAg stimulated sample. Data obtained in this manner indicated that robust TCR mediated conjugation occurred with greater frequency in both CD8 and CD4 conjugates after treatment with 5A2 or LAQ. In all cases maximal conjugation was observable after combined therapy (Figure 4B).
Figure 4.
Epigenetic alteration can restore T-cell signaling capacity and lytic granule mobilization in the context of CLL
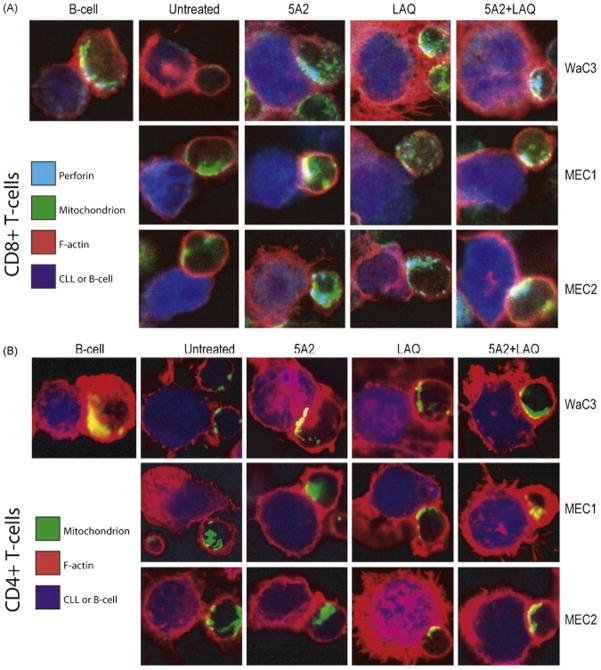
To this point our data has shown that epigenetic modifiers such as 5A2 and LAQ could enhance CLL cell – T-cell SMAC formation. However, we also sought to demonstrate that intracellular signaling, a key component of a functional APC – T-cell interaction, was improved. It has been well demonstrated that mitochondrial localization at the synaptic interface is directly associated with CRAC mediated calcium entry, an essential downstream signal required for T-cell activation [39, 40]. To determine if mitochondria were localizing along the SMAC complex in our experiments we performed immunofluorescent confocal microscopy of CLL cell – allogenic T-cell conjugates after treatment with our epigenetic modifiers (Figure 5A and 5B). Our observations led us to conclude that mitochondrial localization was deficient in untreated CLL cell conjugates, but was qualitatively restored by treatment with 5A2 plus LAQ in both CD4 and CD8 T-cell conjugations.
Figure 5.
Primed CD8 T-cells utilize TCR mediated signaling to mobilize and eventually release lytic granules containing perforin, granzyme-B, and interferon gamma. By immunofluorescent staining of perforin we found that appropriate SMAC F-actin polarization, mitochondrial localization, and lytic granule mobilization was improved in the CD8 T-cell conjugates after CLL cells had been treated with both epigenetic modifiers (Figure 5A). These data serve to confirm our prior results and indicate a definitive signaling enhancement.
Epigenetically altered CLL APC signaling results in T-cells with Th1 polarization, increased proliferative capacity, and lytic activity
The functional significance of an improved CLL APC is, in part, determined by the polarization of the responding T-cells. To characterize this polarization we sorted conjugates formed between CD4+ T-cells and CLL cells and restimulated them with PHA following a 48 hour rest period. The cytokine profile was then measured using cytokine bead array. Our data suggest that, the balance between Th1 and Th2 signaling ([Th1]:[Th2]) was improved (increasingly Th1) after treatment with both inhibitors (Figure 6A). For the MEC1 cell line, treatment completely restored cytokine signaling, while for the WaC3 cell line treatment constricted Th2 signaling predominantly decreasing IL-10, IL-4, IL-6 and IFNγ. These data serve to highlight clinically observed differences in cytokine signaling amongst CLL patients as well as a potential methodology to restore therapeutic T-cell polarization.
Figure 6.
Proliferative self-renewal of antigen specific T-cells is a requirement in the context of sustaining any potential anti-tumor effect. To determine the effects of 5A2 and LAQ have on the proliferation of T-cells stimulated with drug-treated MEC1 CLL cells we CFSE stained healthy donor allogenic CD8 and CD4 T-cells and subjected them to mixed lymphocyte reactions. As shown in Figure 6B a normal allogenic response between healthy B-lymphocytes and healthy allogenic T-lymphocytes (both CD8 and CD4) induces approximately 30% proliferation. On the other hand, only CLL cells which had been treated with a combination of 5A2 and LAQ were capable of stimulating appreciable proliferation (22.7% for CD8 and 21.1% for CD4). This proliferation data served as evidence that the combined 5A2+LAQ treatment could potentiate significant functional improvements when compared to either drug alone.
As a final indicator of proper APC function of CLL cells we wanted to examine the TCR mediated lytic ability of CD8 cytotoxic T-cells. To investigate this we utilized sAg mediated killing and analyzed the results by flow cytometry, quantifying the percentage of T-cells specifically conjugating with dead CLL cells in a sAg specific (or TCR specific) manner [41, 42]. Results from this analysis showed improved lytic function of allogenic CD8 T-cells after being stimulated with 5A2+LAQ treated CLL cells (Figure 6C). Significance (P<0.01) was achieved in the WaC3 and MEC1 cell lines, correlating with and corroborating prior evidence of APC functionality.
5-aza-2′-deoxycytidine and LAQ824 enhance allogenic CD8 cytotoxicity against early stage primary CLL cells
It is well established that CLL cells are deficient in their ability to stimulate T-cell responses, even in allogenic MLR (Figure 6B). It has also been established that the level of dysfunction is directly correlated to increased RAI staging [1]. To ensure that our in-vitro results hold true in primary CLL, we conducted cytotoxicity assays using allogenic CD8 T-cells and 5A2 or LAQ treated primary CLL cells. We found that when primary purified CLL cells were treated with 5A2 and LAQ, there was a significant increase in allogenic cytotoxic potential above that seen in either drug treatment alone (Figure 6D). These results indicate that combined 5A2+LAQ therapy may increase the APC potential of primary CLL.
Discussion
Our studies unveil a previously unknown effect of 5A2 and LAQ on the immunobiology of B-CLL. As anticipated, our results show a therapeutic improvement in antigenic protein expression, costimulatory potential, cytokine signaling, SMAC synapse formation, and T-cell stimulation. These changes reinforce the three signals of APC function: antigenic peptide (signal 1), costimulation (signal 2), and cytokine stimulation (signal 3), resulting in functional changes which may benefit current immunotherapeutic approaches for CLL.
One caveat to our studies is that the majority of mechanistic experimentation has been carried out in CLL cell lines. In our experience, the treatment of primary CLL cells in-vitro with proliferation-dependent epigenetic modifiers such as 5A2 can be quite tedious, requiring the addition of external cytokines, growth factors, and feeder cells, making primary cells inappropriate for large scale reproducible molecular assays. To avoid such pitfalls, our approach focused on validating the CLL cell lines as appropriate epigenetic surrogates of primary CLL cells and utilizing them for the short term in-vitro treatments necessary to foster an enhanced understanding of the mechanisms behind CLL cell epigenetic dysfunction. Subsequently we confirm increased immunogenicity and epigenetic upregulation of potential target antigens using primary CLL cells to ensure the accuracy of our final conclusions. In addition to our independent validation, other investigators have found these cell lines ideal for studying CLL epigenetics [21, 43].
We postulate that the B-CLL cells are capable of directly presenting cancer antigens to the immune system, however it is likely that cross-presentation of CLL antigens via dendritic cells (DCs) is also occurring. In our experience, treatment of DCs with epigenetic modifiers consistently inhibited the production of IL-10. Such an effect was accompanied by an increased expression of co-stimulatory molecules and enhanced production of pro-inflammatory mediators. In addition, HDACi-treated APCs were capable of effectively priming naïve antigen-specific CD4 T-cells and restoring the responsiveness of anergic CD4 T-cells isolated from tumor bearing mice (Wang et. al. unpublished observations). These studies led us to unveil a novel role for HDAC11, in particular, as a transcriptional repressor of IL-10 in murine and human APCs, providing one potential molecular mechanism which we plan to specifically examine in the setting of CLL [29].
Our studies did not directly focus on the direct cytotoxic effect of HDAC and DNMT inhibitors; instead we focused our efforts on the epigenetic changes which result in improved immunological function. In preliminary in-vitro testing we established a working concentration of 1μM for 5A2 and 25nM for LAQ which demonstrated negligible direct cytotoxicity over the 72 hour treatment, as measured by Annexin V and propidium iodide staining, yet yielded sustained epigenetic effect, as measured by CTA upregulation or histone acetylation. Although we cannot rule out the possibility that autophagy may occur [44], we have demonstrated quantifiable epigenetic changes which could conceivably be independently responsible for the demonstrated functional modifications. In addition, although our studies do not directly focus on treating T-cell and instead focused on the polarization of T-cells by treated CLL APCs, preliminary data from our laboratory suggest that these drugs do not negatively affect the proliferation or IFNγ production capacity of primary CD4 and CD8 T-cells. Future ongoing studies in our laboratory will focus on the direct effects of epigenetic modifiers on the T-cells themselves (Dubovsky et. al. unpublished observations).
It has been suggested that the survival of CLL hinges upon the balance between receiving lymphocyte activation, survival, and proliferation signals while maintaining immunosupression of the remaining healthy lymphocyte compartments [45, 46]. This model implies that a given CLL clone may preserve this balance in a different manner explaining the heterogeneity we see in the disease. Our results elucidate some of these subtle differences and provide a potential mechanism for tilting the balance towards immune stimulation. This balance is exemplified in our costimulatory phenotyping experiments (Figure 2A) which demonstrated that slight to moderate improvements in the surface expression of costimulatory and MHC molecules can facilitate the changes seen in the SMAC complex. Our studies confirm the current understanding that slight alterations to APC molecular machinery enable CLL cells to facilitate their own survival while circumventing or diverting T-cell stimulation. It is also possible that the epigenetic modifiers alter functional affinity and avidity of certain costimulatory molecules.
Our studies focused on drug treatment and its effects on B-CLL immunology. However, in the clinical setting it is unlikely that these inhibitors will be so exclusive in their actions. In prior studies we have demonstrated that the epigenetic effects of 5A2 do preferentially act on malignant cells, potentially due to deregulation of the chromatin packaging machinery such as DNA methyltransferase I (DNMT1) or brother of the regulator of imprinted sites (BORIS)[47–50]. Our previous efforts also indicate a potential for drug treatment to elicit long term molecular changes specifically in cancer cells, allowing for temporal separation of drug treatment and effector reaction. Additional evidence comes in the form of studies conducted by Guo et. al. demonstrating elimination of lung metastases by CTA specific cytolytic T-lymphocytes in a murine mammary carcinoma model after treatment with 5A2 [28].
Altogether, our results suggest that epigenetic modifiers which release chromatin constriction via DNA demethylation and histone acetylation may effectively restore the functional immunogenicity of chronic lymphocytic leukemia by inducing the expression of novel and highly antigenic tumor targets, increasing costimulatory potential, repairing defective SMAC formation, and rectifying cytokine stimulation. These effects could aid in the generation of more robust TCR mediated cytolytic responses in primary CLL.
Supplementary Material
References
- 1.Ramsay AG, Johnson AJ, Lee AM, et al. Chronic lymphocytic leukemia T cells show impaired immunological synapse formation that can be reversed with an immunomodulating drug. J Clin Invest. 2008 doi: 10.1172/JCI35017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71–96. doi: 10.3322/CA.2007.0010. [DOI] [PubMed] [Google Scholar]
- 3.Krackhardt AM, Witzens M, Harig S, et al. Identification of tumor-associated antigens in chronic lymphocytic leukemia by SEREX. Blood. 2002;100(6):2123–31. doi: 10.1182/blood-2002-02-0513. [DOI] [PubMed] [Google Scholar]
- 4.Ouillette P, Erba H, Kujawski L, et al. Integrated genomic profiling of chronic lymphocytic leukemia identifies subtypes of deletion 13q14. Cancer Res. 2008;68(4):1012–21. doi: 10.1158/0008-5472.CAN-07-3105. [DOI] [PubMed] [Google Scholar]
- 5.Kay NE, Rai KR, O’Brien S. Chronic lymphocytic leukemia: current and emerging treatment approaches. Clin Adv Hematol Oncol. 2006;4(11 Suppl 22):1–10. quiz 11–2. [PubMed] [Google Scholar]
- 6.Rassenti LZ, Jain S, Keating MJ, et al. Relative value of ZAP-70, CD38, and immunoglobulin mutation status in predicting aggressive disease in chronic lymphocytic leukemia. Blood. 2008;112(5):1923–30. doi: 10.1182/blood-2007-05-092882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fulci V, Chiaretti S, Goldoni M, et al. Quantitative technologies establish a novel microRNA profile of chronic lymphocytic leukemia. Blood. 2007;109(11):4944–51. doi: 10.1182/blood-2006-12-062398. [DOI] [PubMed] [Google Scholar]
- 8.Binet JL, Auquier A, Dighiero G, et al. A new prognostic classification of chronic lymphocytic leukemia derived from a multivariate survival analysis. Cancer. 1981;48(1):198–206. doi: 10.1002/1097-0142(19810701)48:1<198::aid-cncr2820480131>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 9.Rai KR, Sawitsky A, Cronkite EP, et al. Clinical staging of chronic lymphocytic leukemia. Blood. 1975;46(2):219–34. [PubMed] [Google Scholar]
- 10.Tam CS, O’Brien S, Wierda W, et al. Long-term results of the fludarabine, cyclophosphamide, and rituximab regimen as initial therapy of chronic lymphocytic leukemia. Blood. 2008;112(4):975–80. doi: 10.1182/blood-2008-02-140582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wierda WG, Kipps TJ. Gene therapy and active immune therapy of hematologic malignancies. Best Pract Res Clin Haematol. 2007;20(3):557–68. doi: 10.1016/j.beha.2007.03.006. [DOI] [PubMed] [Google Scholar]
- 12.Molica S. Immunomodulatory drugs in chronic lymphocytic leukemia: a new treatment paradigm. Leuk Lymphoma. 2007;48(5):866–9. doi: 10.1080/10428190601126636. [DOI] [PubMed] [Google Scholar]
- 13.Chanan-Khan A, Porter CW. Immunomodulating drugs for chronic lymphocytic leukaemia. Lancet Oncol. 2006;7(6):480–8. doi: 10.1016/S1470-2045(06)70723-9. [DOI] [PubMed] [Google Scholar]
- 14.Ferrajoli A, Lee BN, Schlette EJ, et al. Lenalidomide induces complete and partial remissions in patients with relapsed and refractory chronic lymphocytic leukemia. Blood. 2008;111(11):5291–7. doi: 10.1182/blood-2007-12-130120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu L, Adams M, Carter T, et al. lenalidomide enhances natural killer cell and monocyte-mediated antibody-dependent cellular cytotoxicity of rituximab-treated CD20+ tumor cells. Clin Cancer Res. 2008;14(14):4650–7. doi: 10.1158/1078-0432.CCR-07-4405. [DOI] [PubMed] [Google Scholar]
- 16.Mosmann TR, Coffman RL. TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. [Review] Annu Rev Immunol. 1989;7:145–73. doi: 10.1146/annurev.iy.07.040189.001045. [DOI] [PubMed] [Google Scholar]
- 17.Horna P, Sotomayor EM. Cellular and molecular mechanisms of tumor-induced T-cell tolerance. Curr Cancer Drug Targets. 2007;7(1):41–53. doi: 10.2174/156800907780006940. [DOI] [PubMed] [Google Scholar]
- 18.Mellstedt H, Choudhury A. T and B cells in B-chronic lymphocytic leukaemia: Faust, Mephistopheles and the pact with the Devil. Cancer Immunol Immunother. 2006;55(2):210–20. doi: 10.1007/s00262-005-0675-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Scrivener S, Goddard RV, Kaminski ER, et al. Abnormal T-cell function in B-cell chronic lymphocytic leukaemia. Leuk Lymphoma. 2003;44(3):383–9. doi: 10.1080/1042819021000029993. [DOI] [PubMed] [Google Scholar]
- 20.Rush LJ, Raval A, Funchain P, et al. Epigenetic profiling in chronic lymphocytic leukemia reveals novel methylation targets. Cancer Res. 2004;64(7):2424–33. doi: 10.1158/0008-5472.can-03-2870. [DOI] [PubMed] [Google Scholar]
- 21.Chen SS, Raval A, Johnson AJ, et al. Epigenetic changes during disease progression in a murine model of human chronic lymphocytic leukemia. Proc Natl Acad Sci U S A. 2009;106(32):13433–8. doi: 10.1073/pnas.0906455106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Khan AN, Magner WJ, Tomasi TB. An epigenetically altered tumor cell vaccine. Cancer Immunol Immunother. 2004;53(8):748–54. doi: 10.1007/s00262-004-0513-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Khan AN, Magner WJ, Tomasi TB. An epigenetic vaccine model active in the prevention and treatment of melanoma. J Transl Med. 2007;5(1):64. doi: 10.1186/1479-5876-5-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Khan AN, Tomasi TB. Histone deacetylase regulation of immune gene expression in tumor cells. Immunol Res. 2008;40(2):164–78. doi: 10.1007/s12026-007-0085-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Khan AN, Gregorie CJ, Tomasi TB. Histone deacetylase inhibitors induce TAP, LMP, Tapasin genes and MHC class I antigen presentation by melanoma cells. Cancer Immunol Immunother. 2008;57(5):647–54. doi: 10.1007/s00262-007-0402-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Borden EC. Augmentation of effects of interferon-stimulated genes by reversal of epigenetic silencing: potential application to melanoma. Cytokine Growth Factor Rev. 2007;18(5–6):491–501. doi: 10.1016/j.cytogfr.2007.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dubovsky JA, McNeel DG, Powers JJ, et al. Treatment of chronic lymphocytic leukemia with a hypomethylating agent induces expression of NXF2, an immunogenic cancer testis antigen. Clin Cancer Res. 2009;15(10):3406–15. doi: 10.1158/1078-0432.CCR-08-2099. [DOI] [PubMed] [Google Scholar]
- 28.Guo ZS, Hong JA, Irvine KR, et al. De novo induction of a cancer/testis antigen by 5-aza-2′-deoxycytidine augments adoptive immunotherapy in a murine tumor model. Cancer Res. 2006;66(2):1105–13. doi: 10.1158/0008-5472.CAN-05-3020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Villagra A, Cheng F, Wang HW, et al. The histone deacetylase HDAC11 regulates the expression of interleukin 10 and immune tolerance. Nat Immunol. 2009;10(1):92–100. doi: 10.1038/ni.1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sailhamer EA, Li Y, Smith EJ, et al. Acetylation: a novel method for modulation of the immune response following trauma/hemorrhage and inflammatory second hit in animals and humans. Surgery. 2008;144(2):204–16. doi: 10.1016/j.surg.2008.03.034. [DOI] [PubMed] [Google Scholar]
- 31.Blum KA, Liu Z, Lucas DM, et al. Phase I trial of low dose decitabine targeting DNA hypermethylation in patients with chronic lymphocytic leukaemia and non-Hodgkin lymphoma: dose-limiting myelosuppression without evidence of DNA hypomethylation. Br J Haematol. 2010;150(2):189–95. doi: 10.1111/j.1365-2141.2010.08213.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stacchini A, Aragno M, Vallario A, et al. MEC1 and MEC2: two new cell lines derived from B-chronic lymphocytic leukaemia in prolymphocytoid transformation. Leuk Res. 1999;23(2):127–36. doi: 10.1016/s0145-2126(98)00154-4. [DOI] [PubMed] [Google Scholar]
- 33.Wendel-Hansen V, Sallstrom J, De Campos-Lima PO, et al. Epstein-Barr virus (EBV) can immortalize B-cll cells activated by cytokines. Leukemia. 1994;8(3):476–84. [PubMed] [Google Scholar]
- 34.Dubovsky JA, McNeel DG, Powers JJ, et al. Treatment of Chronic Lymphocytic Leukemia with a Hypomethylating Agent Induces Expression of NXF2, an Immunogenic Cancer Testis Antigen. Clinical Cancer Research. 2009 doi: 10.1158/1078-0432.CCR-08-2099. In Press. [DOI] [PubMed] [Google Scholar]
- 35.Schoenborn JR, Wilson CB. Regulation of interferon-gamma during innate and adaptive immune responses. Adv Immunol. 2007;96:41–101. doi: 10.1016/S0065-2776(07)96002-2. [DOI] [PubMed] [Google Scholar]
- 36.Shnyreva M, Weaver WM, Blanchette M, et al. Evolutionarily conserved sequence elements that positively regulate IFN-gamma expression in T cells. Proc Natl Acad Sci U S A. 2004;101(34):12622–7. doi: 10.1073/pnas.0400849101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tomasi TB, Magner WJ, Khan AN. Epigenetic regulation of immune escape genes in cancer. Cancer Immunol Immunother. 2006;55(10):1159–84. doi: 10.1007/s00262-006-0164-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sjoberg J, Aguilar-Santelises M, Sjogren AM, et al. Interleukin-10 mRNA expression in B-cell chronic lymphocytic leukaemia inversely correlates with progression of disease. Br J Haematol. 1996;92(2):393–400. doi: 10.1046/j.1365-2141.1996.00358.x. [DOI] [PubMed] [Google Scholar]
- 39.Quintana A, Schwindling C, Wenning AS, et al. T cell activation requires mitochondrial translocation to the immunological synapse. Proc Natl Acad Sci U S A. 2007;104(36):14418–23. doi: 10.1073/pnas.0703126104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Oakes SA. Mitochondria control calcium entry at the immunological synapse. Proc Natl Acad Sci U S A. 2007;104(39):15171–2. doi: 10.1073/pnas.0707798104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vitale M, Zamai L, Neri LM, et al. Natural killer function in flow cytometry: identification of human lymphoid subsets able to bind to the NK sensitive target K562. Cytometry. 1991;12(8):717–22. doi: 10.1002/cyto.990120805. [DOI] [PubMed] [Google Scholar]
- 42.Morgan MM, Labno CM, Van Seventer GA, et al. Superantigen-induced T cell:B cell conjugation is mediated by LFA-1 and requires signaling through Lck, but not ZAP-70. J Immunol. 2001;167(10):5708–18. doi: 10.4049/jimmunol.167.10.5708. [DOI] [PubMed] [Google Scholar]
- 43.Plass C, Byrd JC, Raval A, et al. Molecular profiling of chronic lymphocytic leukaemia: genetics meets epigenetics to identify predisposing genes. Br J Haematol. 2007;139(5):744–52. doi: 10.1111/j.1365-2141.2007.06875.x. [DOI] [PubMed] [Google Scholar]
- 44.Fink SL, Cookson BT. Apoptosis, pyroptosis, and necrosis: mechanistic description of dead and dying eukaryotic cells. Infect Immun. 2005;73(4):1907–16. doi: 10.1128/IAI.73.4.1907-1916.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rossmann ED, Lewin N, Jeddi-Tehrani M, et al. Intracellular T cell cytokines in patients with B cell chronic lymphocytic leukaemia (B-CLL) Eur J Haematol. 2002;68(5):299–306. doi: 10.1034/j.1600-0609.2002.01612.x. [DOI] [PubMed] [Google Scholar]
- 46.Tangye SG, Raison RL. Human cytokines suppress apoptosis of leukaemic CD5+ B cells and preserve expression of bcl-2. Immunol Cell Biol. 1997;75(2):127–35. doi: 10.1038/icb.1997.17. [DOI] [PubMed] [Google Scholar]
- 47.Vatolin S, Abdullaev Z, Pack SD, et al. Conditional expression of the CTCF-paralogous transcriptional factor BORIS in normal cells results in demethylation and derepression of MAGE-A1 and reactivation of other cancer-testis genes. Cancer Res. 2005;65(17):7751–62. doi: 10.1158/0008-5472.CAN-05-0858. [DOI] [PubMed] [Google Scholar]
- 48.Klenova EM, Morse HC, 3rd, Ohlsson R, et al. The novel BORIS + CTCF gene family is uniquely involved in the epigenetics of normal biology and cancer. Semin Cancer Biol. 2002;12(5):399–414. doi: 10.1016/s1044-579x(02)00060-3. [DOI] [PubMed] [Google Scholar]
- 49.Loukinov DI, Pugacheva E, Vatolin S, et al. BORIS, a novel male germ-line-specific protein associated with epigenetic reprogramming events, shares the same 11-zinc-finger domain with CTCF, the insulator protein involved in reading imprinting marks in the soma. Proc Natl Acad Sci U S A. 2002;99(10):6806–11. doi: 10.1073/pnas.092123699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dubovsky JA, McNeel DG. Inducible expression of a prostate cancer-testis antigen, SSX-2, following treatment with a DNA methylation inhibitor. Prostate. 2007;67(16):1781–90. doi: 10.1002/pros.20665. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.