Abstract

We report on a chemical platform to generate site-specific, homogeneous, antibody–antibody conjugates by targeting and bridging disulfide bonds. A bispecific antibody construct was produced in good yield through simple reduction and bridging of antibody fragment disulfide bonds, using a readily synthesized bis-dibromomaleimide cross-linker. Binding activity of antibodies was maintained, and in vitro binding of target antigens was observed. This technology is demonstrated through linking scFv and Fab antibody fragments, showing its potential for the construction of a diverse range of bispecifics.
The limited ability of conventional monoclonal antibody therapies to induce significant antitumor activity has led to the development of bispecifics: antibodies that can simultaneously bind two different antigens. In 2009, catumaxomab became the first bispecific therapeutic to be clinically approved, combining EpCAM targeting with T-cell recruitment for the treatment of malignant ascites.1 Recombinant technologies have produced a diverse range of bispecific antibodies, generating 45 formats in the past two decades.2 Despite this variety of topologies, the approach is not suited to every protein combination. The fusion of proteins via their N or C termini can result in a reduction or loss of bioactivity and variable expression yields can be observed due to complications in folding and processing.3−5
An alternative and potentially more versatile approach to generating bispecific therapeutics is chemical conjugation. Until now, this has been a less successful method of producing such conjugates. A fundamental flaw in the chemical techniques employed in this area has been their dependence on modifying lysine residues. There is an average of 100 lysine residues per antibody, and their distribution is uniform throughout the surface topology of the Fab and Fc regions. As such, conjugation techniques using lysine residues will randomly cross-link to virtually all areas of the antibody molecule, resulting in a highly heterogeneous mixture of products with unpredictable properties. One strategy to overcome this issue is provided by site-directed mutagenesis, which enables a single nucleophilic cysteine residue to be introduced at a desired site in an antibody. However, this approach is limited, as cysteine mutagenesis commonly leads to reduced expression yields and undesirable properties such as susceptibility to dimerization, mixed disulfide formation, or disulfide scrambling.6−8
Recently the site-specific introduction of chemical linkers has been reported through unnatural amino acid insertion.9,10 Using this approach, Schultz et al. described the synthesis of a homogeneous anti-HER2/anti-CD3 bispecific in good yield.10 This technology, while elegant, is not readily transferred; each antibody to be conjugated must undergo prior investigation to determine appropriate mutation sites, substitution for the unnatural amino acid is often incomplete, and expression yields are generally low due to the cellular toxicity of artificial amino acids at the high concentrations necessary.11,12 To avoid these difficulties, an ideal site-directed conjugation technique would use residues natural to the protein that are revealed for modification only under defined conditions.
Cysteine residues have a low natural abundance in proteins, and are often found tied up in disulfide bonds.13 In the case of antibodies and antibody fragments there are no free cysteine residues, and site-directed conjugation has been attempted via interchain disulfide bond reduction and subsequent conjugation of the free cysteines. However, conjugation of chemical entities to the generated cysteine residues results in significant physical instability of conjugates, particularly under times of stress.14 Furthermore, targeting the cysteine residues responsible for interchain disulfides using chemical cross-linking reagents results in poor yields of bispecific due to the formation of homodimers and intrachain coupling.15 Therefore, the ideal solution would be to use reagents that bridge disulfide bonds, maintaining this key stabilizing feature, and preventing the opportunity for product heterogeneity.16−23 Herein we propose a conjugation strategy using simple chemical reagents that selectively bridge disulfide bonds. Through rapid reduction and bridging of disulfides, homogeneous bispecific antibodies could be readily generated with no effect on stability or activity. To demonstrate the versatility of this chemical conjugation approach to varying antibody fragment formats, we aimed to generate a homogeneous scFv-Fab conjugate (Scheme 1).
Scheme 1. Strategy for the Production of a Homogeneous Bispecific through Disulfide Bridging of Two Antibody Fragments.

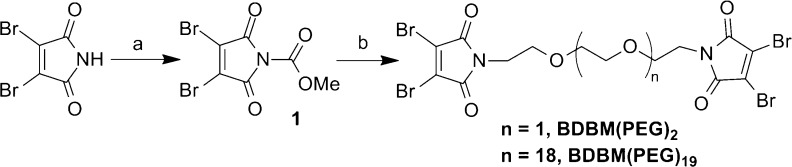
In prior work we have demonstrated that next generation maleimides can be used for the extremely efficient rebridging of disulfide bonds in Fab and disulfide-stabilized scFv antibody fragments, to produce fully active, homogeneous protein conjugates in near-quantitative yields.20,21 Antibody fragments including Fabs and scFvs are commonly used in a range of bispecific topologies. Thus, we envisaged that next generation maleimide based cross-linking reagents could be used to produce homogeneous bispecific constructs. To this end homobifunctional linkers were designed, incorporating two dibromomaleimide moieties linked by a PEG chain, conferring some flexibility to the molecule (Scheme 2). Using commercially available dibromomaleimide and diamine PEG, two linkers of distinct length were readily synthesized. The reaction proceeds under mild conditions in good yield, requiring only a single purification step.24
Scheme 2. Synthetic Route to Linkers.
(a) ClCO2Me, NMM, THF, 97%; (b) For BDBM(PEG)2: NH2CH2CH2(OCH2CH2)2NH2, DCM, 70%. For BDBM(PEG)19: NH2CH2CH2(OCH2CH2)19NH2, DCM, 65%.
To examine the feasibility of this approach to producing a homogeneous protein–protein conjugate, we decided to first work with the anti-CEA disulfide-stabilized scFv fragment. Carcinoembryonic antigen (CEA) is a cell surface glycoprotein overexpressed in a wide range of cancers, particularly in colorectal carcinoma.25 A scFv is the smallest antibody fragment which retains full binding activity, and is a common component of many bispecific antibody formats, e.g., BiTEs (Bispecific T-cell Engager) and DARTs (Dual-Affinity Re-Targetting). Initially, the ability of the linkers to generate scFv homodimer was investigated (Scheme 3).
Scheme 3. Generation of scFv Homodimer.

The scFv was reduced (DTT, 20 equiv with respect to antibody) and, following buffer exchange, incubated with 0.5 equiv of linker BDBM(PEG)2 or BDBM(PEG)19. The conjugation reaction was monitored by SDS-PAGE, and a distinct band at ∼50 kDa was observed on incubation of BDBM(PEG)19 with reduced scFv after only 1 h at room temperature (Supporting Information Figure 1). This corresponds to the molecular weight of scFv dimer. In contrast, the reduced scFv incubated with linker BDBM(PEG)2, although rebridged, reveals only a faint band at ∼50 kDa, suggesting poor dimer formation.
The longer length of linker BDBM(PEG)19 (19 PEG units, ∼1 kDa), in combination with its flexibility, is likely to significantly reduce steric hindrance which creates difficulty when attempting to link two large proteins. This is observed in the far greater conversion to homodimer obtained compared to linker BDBM(PEG)2 (Supporting Information Figure 1). It was therefore decided that conditions for homogeneous protein–protein conjugation should be optimized using linker BDBM(PEG)19. As a model system, generation of homogeneous scFv dimer was further pursued in order to determine ideal conditions for maintaining antibody fragments in their reduced form while promoting bridging.
Following reduction, the scFv was eluted over a Sephadex column to remove DTT and to buffer exchange the protein into conditions suitable for maintaining the reduced antibody (20 mM phosphate buffer, 5 mM EDTA, pH 7.4). The antibody fragment was then concentrated to no greater than 1 mg/mL, since higher concentrations promoted disulfide reoxidation. A series of optimization experiments revealed incubation with 0.42 equiv of bis-dibromomaleimide cross-linker BDBM(PEG)19 for 1 h at room temperature, or 4 °C overnight, yielded the highest levels of homogeneous dimer (Supporting Information Figure 2). In fact, after 1 h approximately 80% of starting material was consumed (Figure 1[i], lane 3, measured using ImageJ). After purification of the homodimer by size exclusion chromatography, an excellent 64% yield of pure, homogeneous scFv–scFv conjugate was obtained from 1 mg of scFv (Figure 1[iii], 75% yield with respect to the limiting reagent BDBM(PEG)19). This yield represents a significant improvement on previous reports of direct chemical cross-linking of antibodies using natural amino acids, which have achieved yields in the 10–40% range.26−29 Notably the conjugate is homogeneous due to the site-selectivity of the methodology.
Figure 1.
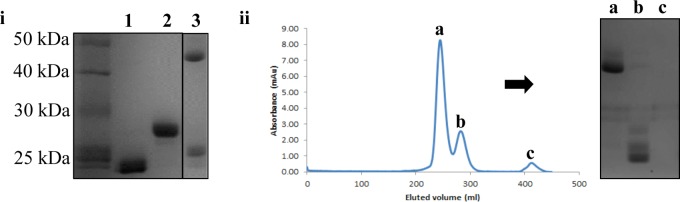
Application of optimized conditions to dimerization of scFv: [i] SDS-PAGE analysis (1) scFv; (2) scFv + 20 equiv DTT to afford reduced scFv (note - scFv with intact disulfide runs faster through gel due to more compact structure); (3) reduced scFv + 0.42 equiv BDBM(PEG)19, 1 h rt to afford scFv homodimer (∼50 kDa) and disulfide bridged scFv monomer (∼25 kDa). [ii] Size exclusion chromatography purification of pure scFv homodimer (peak a, ∼50 kDa) from monomer starting materials (peak b) and unreacted linker (peak c) (Superdex 75, GE Healthcare).
Given the success of our technology in generating homogeneous scFv homodimer in high yield, we targeted the generation of a bispecific antibody conjugate. The monoclonal IgG1 antibody Traztuzumab (Herceptin) targets the HER2/neu receptor and has successfully been used to treat HER2+ breast cancer patients.30 The Fab fragment of this clinically relevant antibody can be readily obtained by enzymatic digest, and its incorporation into bispecific formats has therapeutic potential.10 A Fab fragment contains one variable and one constant region of each of the heavy and light antibody chains, and so is twice the size of a scFv fragment. The heavy and light chains are covalently linked via a single interchain disulfide bond. Thus, we envisaged linking the disulfide-stabilized scFv to the Fab fragment using BDBM(PEG)19, generating a conjugate that could simultaneously bind two different antigens (CEA and HER2) as a proof of concept (Scheme 4).
Scheme 4. Generation of anti-CEA/anti-HER2 scFv-Fab Bispecific.
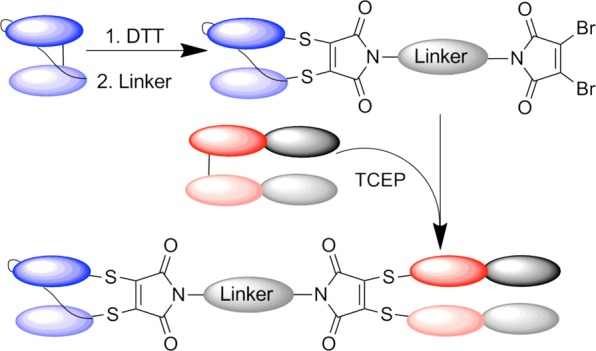
To efficiently link these two different antibody fragments, we decided to first functionalize the scFv with BDBM(PEG)19. This was cleanly achieved by reducing the scFv as previously described, purifying by Sephadex column and subsequently adding 30 equiv of linker to scFv. This reaction was complete in less than 5 min at room temperature, yielding bromomaleimide functionalized scFv in quantitative yield (Figure 2[i], lane 3). Excess linker was removed by buffer exchange. In tandem with this, the Fab fragment was reduced (TCEP, 10 equiv relative to antibody) and purified from reducing agent into pH 7.4 phosphate buffer containing 5 mM EDTA. The concentrations of bridged scFv and reduced Fab were adjusted to 37.2 μM (1 mg/mL for the scFv), and the antibodies mixed in a 2:1 ratio by volume (2 equiv bridged scFv to 1 equiv reduced Fab). This slight excess of functionalized scFv was found to be sufficient to promote bridging of the reduced Fab. The reaction was monitored by SDS-PAGE, and after 1 h at room temperature a strong band at ∼80 kDa could be observed, corresponding to the scFv-Fab conjugate. After purification by size exclusion chromatography, a pleasing 52% yield of homogeneous scFv-Fab bispecific was achieved.
Figure 2.

Production of homogeneous scFv-Fab: [i] SDS-PAGE analysis (1) scFv; (2) scFv + 20 equiv DTT to afford reduced scFv (note - scFv with intact disulfide runs faster through gel due to more compact structure); (3) Reduced scFv + 30 equiv BDBM(PEG)19 to afford disulfide bridged scFv monomer (note - dibromomaleimide functionalized scFv runs faster than reduced through gel as compact structure is recovered through disulfide bridging); (4) Fab; (5) Fab +10 equiv TCEP to afford component heavy and light Fab chains; (6) Bridged scFv + Reduced Fab (2:1), 1 h rt to afford scFv-Fab conjugate (∼80 kDa), reoxidized Fab (∼50 kDa) and scFv starting material (∼25 kDa) . [ii] Size exclusion chromatography purification of pure scFv-Fab heterodimer (peak a) from starting materials (peak b and c) (Superdex 75, GE Healthcare).
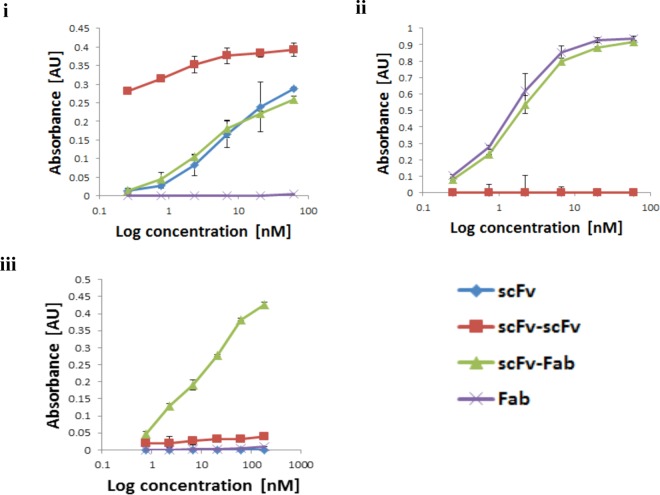
Next, we used enzyme-linked immunosorbent assay (ELISA) to assess the immuno-reactivity of the purified conjugates, in comparison to the unmodified scFv and Fab fragments that bound either CEA or HER2, respectively (Figure 3[i] and [ii]). Against CEA, the activity of the scFv homodimer was not only maintained, but remained high at low dilutions, suggesting dimerization has successfully increased the avidity of the antibody. Most importantly, the scFv-Fab conjugate showed comparable antigen binding activity against both CEA and HER2. Given this success, we wanted to demonstrate that our heterodimeric conjugate could simultaneously bind its two target antigens. To achieve this we developed a sandwich ELISA. A 96-well plate was coated with CEA and the sample to be tested, e.g., scFv-Fab conjugate, was applied. Subsequent incubation with HER2-Biotin and Extravadin-Peroxidase would thus only lead to a signal if the sample successfully bound both CEA and HER2 antigens. Pleasingly, simultaneous binding activity was confirmed (Figure 3[iii]). Hence we have successfully produced a homogeneous antibody conjugate with bispecific ability using our disulfide-bridging technology.
Figure 3.
ELISA analysis of conjugates and unmodified antibody fragments: [i] ELISA against full length CEA. [ii] ELISA against HER2. [iii] Sandwich ELISA using full length CEA coated plates and HER2 conjugated to biotin, followed by Extravadin-Peroxidase.
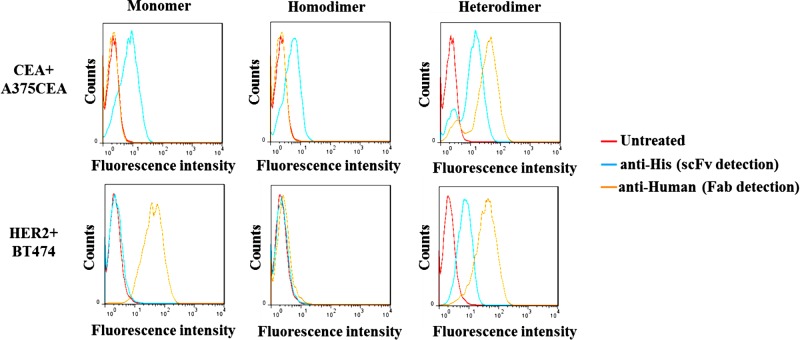
In a final investigation, we assessed the binding of our antibody conjugates to a CEA-positive cell line, A375 CEA, and a HER2-positive cell line, BT-474 (controls shown in Supporting Information Figure 3). The monomer controls demonstrate that anti-CEA scFv binds only to A375CEA cells and anti-HER2 Fab only to BT474 cells (Figure 4, monomer). Application of our scFv homodimer conjugate reveals that CEA binding activity and selectivity is maintained after conjugation, with a shift in fluorescence being observed only on the A375CEA cell line (Figure 4, homodimer). Following this success, we tested our bispecific construct (Figure 4, heterodimer). In this case, scFv and Fab were detected in both the CEA-positive cell line A375CEA and the HER2-positive BT474. This demonstrates that the antibody fragments have maintained their distinct selectivity and binding activity in vitro, while being successfully chemically linked.
Figure 4.
Flow cytometry based binding assay of unmodified scFv and Fab (monomer), scFv dimer (homodimer), and scFv-Fab conjugate (heterodimer) to a CEA-positive cell line (A375CEA) and HER2-positive cell line (BT474).
Conclusion
To date, the generation of bispecifics for the clinic by chemical cross-linking has been unsuccessful, due to low yields and product heterogeneity. Here we have presented the rapid production of two homogeneous conjugates in high yield: an anti-CEA scFv homodimer and an anti-CEA/anti-HER2 scFv-Fab heterodimer. Our chemical conjugation approach uses bis-dibromomaleimide PEG linker readily synthesized over two steps from the commercially available dibromomaleimide. Through targeting and bridging the disulfide bond of antibody fragments, conjugates were produced which exhibit retention of activity by ELISA and cell binding assays. This platform has the potential to enable the facile generation of bispecifics from a range of antibody fragment formats, and could be readily translated to other protein conjugates of choice, exploiting the versatility of the chemical conjugation approach.
Experimental Procedures
N,N-PEG2-bis-3,4-dibromomaleimide (BDBM(PEG)2)24
2,2′-(Ethylenedioxy)bis(ethylamine) (0.2 mL, 1.38 mmol) was added to a stirred solution of N-(methoxycarbonyl)-3,4-dibromomaleimide24 (1.08 g, 3.45 mmol) in DCM (8 mL). After 20 min EtOAc (80 mL) was added and the organic layer extracted with saturated NH4Cl solution (2 × 40 mL) and H2O (3 × 40 mL). The organic layer was dried with MgSO4 and concentrated in vacuo. Purification by column chromatography (gradient elution from 1:9 to 3:7 EtOAc/Petrol) yielded BDBM(PEG)2 as a pale yellow solid (600 mg, 70%): mp 116–117 °C; 1H NMR (500 MHz, CDCl3) δ 3.82 (4H, t, J = 5.6 Hz), 3.65 (4H, t, J = 5.6 Hz), 3.58 (4H, s); 13C NMR (125 MHz, CDCl3) δ 164.2 (C), 129.8 (C), 70.4 (CH2), 68.0 (CH2), 39.3 (CH2); IR (solid) 3489, 2913, 1784, 1720, 1597; LRMS (ES+) 651 ([81,81,81,81M+Na], 20), 649 ([81,81,81,79M+Na], 70), 647 ([81,81,79,79M+Na], 100), 645 ([81,79,79,79M+Na], 70), 643 ([79,79,79,79M+Na], 20); HRMS (ES+) calculated for C14H12N2O6Na79Br4 642.7327, observed 642.7355.
N,N-PEG20-bis-3,4-dibromomaleimide (BDBM(PEG)19)
O,O′-Bis(2-aminoethyl)octadecaethylene glycol (50 mg, 0.06 mmol) was added to a stirred solution of N-(methoxycarbonyl)-3,4-dibromomaleimide24 (44 mg, 0.14 mmol) in DCM (2 mL). The reaction mixture was left at room temperature overnight, and then the solvent removed in vacuo. Purification by column chromatography (gradient elution from DCM to 20:1 DCM/MeOH) yielded BDBM(PEG)19 as a pale yellow oil (53 mg, 65%). 1H NMR (600 MHz, CDCl3) δ 3.81 (4H, t, J = 6.0), 3.67–3.59 (76H, m); 13C NMR (150 MHz, CDCl3) δ 164.0 (C), 129.6 (C), 70.7 (CH2), 70.2 (CH2), 67.7 (CH2), 39.0 (CH2); IR (oil, cm–1) 2865, 1722; LRMS (EI) 1394 ([81,81,81,81M+NH4], 20), 1392 ([81,81,81,79M+NH4], 70), 1390 ([81,81,79,79M+NH4], 100), 1388 ([81,79,79,79M+NH4], 70), 1386 ([79,79,79,79M+NH4], 20), 1346 (40), 1300 (10); HRMS (EI) calculated for C48H8081,81,79,79Br4N2O3NH4 1390.2192, observed 1390.2181.
Disulfide Bridging of scFv with Linker BDBM(PEG)2 to Generate scFv–scFv Homodimer
Anti-CEA ds-scFv in PBS (1 mg/mL, 37.2 μM) was reduced with DTT (20 equiv relative to scFv, 37.2 mM stock in PBS) for 1 h at room temperature. DTT was then removed using a desalting column (PD-10, GE Healthcare) and the reduced scFv buffer exchanged into conjugation buffer (20 mM phosphate buffer, 5 mM EDTA, pH 7.4). The scFv was concentrated to approximately 1 mg/mL and linker BDBM(PEG)2 added (0.42 equiv relative to scFv, 3.72 mM stock in DMF). After 1 h at room temperature or overnight at 4 °C, the reaction was purified by size exclusion (500 mL Superdex 75 column, GE Healthcare).
Disulfide Bridging of scFv with Linker BDBM(PEG)19 to Generate scFv–scFv Homodimer
Anti-CEA ds-scFv in PBS (1 mg/mL, 37.2 μM) was reduced with DTT (20 equiv relative to scFv, 37.2 mM stock in PBS) for 1 h at room temperature. DTT was then removed using a desalting column (PD-10, GE Healthcare) and the reduced scFv buffer exchanged into conjugation buffer (20 mM phosphate buffer, 5 mM EDTA, pH 7.4). The scFv was concentrated to approximately 1 mg/mL and linker BDBM(PEG)19 added (0.42 equiv relative to scFv, 3.72 mM stock in water). After 1 h at room temperature or overnight at 4 °C, the reaction was purified by size exclusion (500 mL Superdex 75 column, GE Healthcare).
Disulfide Bridging of scFv and Fab to Generate scFv-Fab Heterodimer
Anti-CEA ds-scFv in PBS (1 mg/mL, 37.2 μM) was reduced with DTT (20 equiv relative to scFv, 37.2 mM in PBS) for 1 h at room temperature. DTT was then removed using a desalting column (PD-10, GE Healthcare) and the reduced scFv buffer exchanged into conjugation buffer (20 mM phosphate buffer, 5 mM EDTA, pH 7.4). The scFv was concentrated to approximately 1 mg/mL and linker BDBM(PEG)19 added (30 equiv relative to scFv, 37.2 mM in water). After 10 min excess linker was removed by buffer exchange (repeat 3 times, Amicon Ultra-4 Centrifugal Filter Units, 10 kDa cutoff) into conjugation buffer, and the bridged scFv concentrated to approximately 1 mg/mL. Meanwhile, Herceptin Fab in Borate buffer (1 mg/mL, 25 mM sodium borate, 25 mM NaCl, 1 mM EDTA, pH 8.0) was reduced with TCEP (10 equiv relative to Fab, 37.2 mM in borate buffer) for 1 h at room temperature. TCEP was then removed using a desalting column (PD-10, GE Healthcare) and the reduced Fab buffer exchanged into conjugation buffer (20 mM phosphate buffer, 5 mM EDTA, pH 7.4). The reduced Fab concentration was adjusted to 37.2 μM, and bridged scFv (37.2 μM) mixed with the reduced Fab in a 2:1 ratio by volume, respectively. After 1 h at room temperature or overnight at 4 °C, the reaction was purified by size exclusion (500 mL Superdex 75 column, GE Healthcare).
Acknowledgments
We gratefully acknowledge UCL, the Wellcome Trust, BBSRC, UCLB, the KCL and UCL Comprehensive Cancer Imaging Centre (CCIC) Grant C1519/A10331, The Department of Health and Cancer Research UK Experimental Cancer Medicine Centre (ECMC) Grant C34/A7279 and The EU Seventh Framework Programme (IMAGINT Project) Grant 259881 for support of our programme. We also thank Dr Berend Tolner for kindly providing the anti-CEA ds-scFv, Dr Kersti Karu for the MALDI data and the EPSRC UK National Mass Spectrometry Facility (NMSF), Swansea.
Supporting Information Available
Full experimental procedures and data are provided. This material is available free of charge via the Internet at http://pubs.acs.org.
The authors declare the following competing financial interest(s): J.R.B. and M.E.B.S. are founders and directors of Thiologics, a company developing homogeneous protein modification technology.
Supplementary Material
References
- Heiss M. M.; Murawa P.; Koralewski P.; Kutarska E.; Kolesnik O. O.; Ivanchenko V. V.; Dudnichenko A. S.; Aleknaviciene B.; Razbadauskas A.; Gore M.; Ganea-Motan E.; Ciuleanu T.; Wimberger P.; Schmittel A.; Schmalfeldt B.; Burges A.; Bokemeyer C.; Lindhofer H.; Lahr A.; Parsons S. L. (2010) The trifunctional antibody catumaxomab for the treatment of malignant ascites due to epithelial cancer: Results of a prospective randomized phase II/III trial. Int. J. Cancer 127, 2209–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrne H.; Conroy P. J.; Whisstock J. C.; O’Kennedy R. J. (2013) A tale of two specificities: bispecific antibodies for therapeutic and diagnostic applications. Trends Biotechnol. 31, 621–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt S. R. (2009) Fusion proteins as biopharmaceuticals – applications and challenges. Curr. Opin. Drug Discovery Dev. 12, 284–295. [PubMed] [Google Scholar]
- Baggio L. L.; Huang Q.; Brown T. J.; Drucker D. J. (2004) A recombinant human glucagon-like peptide (GLP)-1–albumin protein (Albugon) mimics peptidergic activation of GLP-1 receptor-dependent pathways coupled with satiety, gastrointestinal motility, and glucose homeostasis. Diabetes 53, 2492–2500. [DOI] [PubMed] [Google Scholar]
- Chames P.; Baty D. (2009) Bispecific antibodies for cancer therapy: the light at the end of the tunnel?. mAbs 1, 539–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmiedl A.; Breitling F.; Winter C. H.; Queitsch I.; Dübel S. (2000) Effects of unpaired cysteines on yield, solubility and activity of different recombinant antibody constructs expressed in E. coli. J. Immunol. Methods 242, 101–14. [DOI] [PubMed] [Google Scholar]
- Junutula J. R.; Raab H.; Clark S.; Bhakta S.; Leipold D. D.; Weir S.; Chen Y.; Simpson M.; Tsai S. P.; Dennis M. S.; Lu Y.; Meng Y. G.; Ng C.; Yang J.; Lee C. C.; Duenas E.; Gorrell J.; Katta V.; Kim A.; McDorman K.; Flagella K.; Venook R.; Ross S.; Spencer S. D.; Lee Wong W.; Lowman H. B.; Vandlen R.; Sliwkowski M. X.; Scheller R. H.; Polakis P.; Mallet W. (2008) Site-specific conjugation of a cytotoxic drug to an antibody improves the therapeutic index. Nat. Biotechnol. 26, 925–32. [DOI] [PubMed] [Google Scholar]
- Albrecht H., Burke P. A., Natarajan A., Xiong C.-Y., Kalicinsky M., DeNardo G. L., and DeNardo S. J.. Production of soluble ScFvs with C-terminal-free thiol for site-specific conjugation or stable dimeric ScFvs on demand. Bioconjugate Chem. 15, 16–26. [DOI] [PubMed] [Google Scholar]
- Hudak J. E.; Barfield R. M.; de Hart G. W.; Grob P.; Nogales E.; Bertozzi C. R.; Rabuka D. (2012) Synthesis of heterobifunctional protein fusions using copper-free click chemistry and the aldehyde tag. Angew. Chem., Int. Ed. 51, 4161–4165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim C. H.; Axup J. Y.; Dubrovska A.; Kazane S. A.; Hutchins B. A.; Wold E. D.; Smider V. V.; Schultz P. G. (2012) Synthesis of bispecific antibodies using genetically encoded unnatural amino acids. J. Am. Chem. Soc. 134, 9918–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loscha K. V.; Herlt A. J.; Qi R.; Huber T.; Ozawa K.; Otting G. (2012) Multiple-site labeling of proteins with unnatural amino acids. Angew. Chem., Int. Ed. 51, 2243–6. [DOI] [PubMed] [Google Scholar]
- Liebscher S.; Schöpfel M.; Aumüller T.; Sharkhuukhen A.; Pech A.; Höss E.; Parthier C.; Jahreis G.; Stubbs M. T.; Bordusa F. (2014) N-terminal protein modification by substrate-activated reverse proteolysis. Angew. Chem., Int. Ed. 53, 3024–8. [DOI] [PubMed] [Google Scholar]
- Fodje M. N.; Al-Karadaghi S. (2002) Occurrence, conformational features and amino acid propensities for the α-helix. Protein Eng., Des. Sel. 15, 353–358. [DOI] [PubMed] [Google Scholar]
- Adem Y. T.; Schwarz K. A.; Duenas E.; Patapoff T. W.; Galush W. J.; Esue O. (2014) Auristatin antibody drug conjugate physical instability and the role of drug payload. Bioconjugate Chem. 25, 656–64. [DOI] [PubMed] [Google Scholar]
- Ellerman D., and Scheer J. M. (2011) Generation of Bispecific Antibodies by Chemical Conjugation. In Bispecific Antibodies (Kontermann R. E., Ed.) pp 47–63, Chapter 3, Springer. [Google Scholar]
- Smith M. E. B.; Schumacher F. F.; Ryan C. P.; Tedaldi L. M.; Papaioannou D.; Waksman G.; Caddick S.; Baker J. R. (2010) Protein modification, bioconjugation, and disulfide bridging using bromomaleimides. J. Am. Chem. Soc. 132, 1960–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schumacher F. F.; Nobles M.; Ryan C. P.; Smith M. E. B.; Tinker A.; Caddick S.; Baker J. R. (2011) In situ maleimide bridging of disulfides and a new approach to protein PEGylation. Bioconjugate Chem. 22, 132–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan C. P.; Smith M. E. B.; Schumacher F. F.; Grohmann D.; Papaioannou D.; Waksman G.; Werner F.; Baker J. R.; Caddick S. (2011) Tunable reagents for multi-functional bioconjugation: reversible or permanent chemical modification of proteins and peptides by control of maleimide hydrolysis. Chem. Commun. 47, 5452–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khalili H.; Godwin A.; Choi J.; Lever R.; Brocchini S. (2012) Comparative binding of disulfide-bridged PEG-Fabs. Bioconjugate Chem. 23, 248–263. [DOI] [PubMed] [Google Scholar]
- Schumacher F. F.; Sanchania V. A.; Tolner B.; Wright Z. V. F.; Ryan C. P.; Smith M. E. B.; Ward J. M.; Caddick S.; Kay C. W. M.; Aeppli G.; Chester K. A.; Baker J. R. (2013) Homogeneous antibody fragment conjugation by disulfide bridging introduces “spinostics”. Sci. Rep. 3, 1525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castañeda L.; Maruani A.; Schumacher F. F.; Miranda E.; Chudasama V.; Chester K. A.; Baker J. R.; Smith M. E. B.; Caddick S. (2013) Acid-cleavable thiomaleamic acid linker for homogeneous antibody-drug conjugation. Chem. Commun. 49, 8187–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palanki M. S. S.; Bhat A.; Bolanos B.; Brunel F.; Del Rosario J.; Dettling D.; Horn M.; Lappe R.; Preston R.; Sievers A.; Stankovic N.; Woodnut G.; Chen G. (2013) Development of a long acting human growth hormone analog suitable for once a week dosing. Bioorg. Med. Chem. Lett. 23, 402–6. [DOI] [PubMed] [Google Scholar]
- Marculescu C.; Kossen H.; Morgan R. E.; Mayer P.; Fletcher S. A.; Tolner B.; Chester K. A.; Jones L. H.; Baker J. R. (2014) Aryloxymaleimides for cysteine modification, disulfide bridging and the dual functionalization of disulfide bonds. Chem. Commun. 54, 4–7. [DOI] [PubMed] [Google Scholar]
- Castañeda L.; Wright Z. V. F.; Marculescu C.; Tran T. M.; Chudasama V.; Maruani A.; Hull E. A.; Nunes J. P. M.; Fitzmaurice R. J.; Smith M. E. B.; Jones L. H.; Caddick S.; Baker J. R. (2013) A mild synthesis of N-functionalised bromomaleimides, thiomaleimides and bromopyridazinediones. Tetrahedron Lett. 54, 3493–3495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein M. J.; Mitchell E. P. (2005) Carcinoembryonic antigen in the staging and follow-up of patients with colorectal cancer. Cancer Invest. 23, 338–351. [DOI] [PubMed] [Google Scholar]
- Khalili H.; Godwin A.; Choi J.; Lever R.; Khaw S. P.; Brocchini S. (2013) Fab-PEG-Fab as a potential antibody mimetic Fab-PEG-Fab as a potential antibody mimetic. Bioconjugate Chem. 24, 1870–1882. [DOI] [PubMed] [Google Scholar]
- Karpovsky B. Y. B.; Titus J. A.; Stephany D. A.; Segal D. M. (1984) Production of target-specific effector cells using hetero-cross-linked aggregates containing anti-target cell and anti-Fc gamma receptor antibodies. J. Exp. Med. 160, 1686–1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reusch U.; Sundaram M.; Davol P. A.; Olson S. D.; Davis J. B.; Demel K.; Nissim J.; Rathore R.; Liu P. Y.; Lum L. G. (2006) Anti-CD3 x anti-epidermal growth factor receptor (EGFR) bispecific antibody redirects T-cell cytolytic activity to EGFR-positive cancers in vitro and in an animal model. Clin. Cancer Res. 12, 183–90. [DOI] [PubMed] [Google Scholar]
- Lee R. J.; Fang Q.; Davol P. a; Gu Y.; Sievers R. E.; Grabert R. C.; Gall J. M.; Tsang E.; Yee M. S.; Fok H.; Huang N. F.; Padbury J. F.; Larrick J. W.; Lum L. G. (2007) Antibody targeting of stem cells to infarcted myocardium. Stem cells 25, 712–7. [DOI] [PubMed] [Google Scholar]
- Hudis C. A. (2007) Trastuzumab – mechanism of action and use in clinical practice. N. Eng. J. Med. 357, 39–51. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.