Abstract
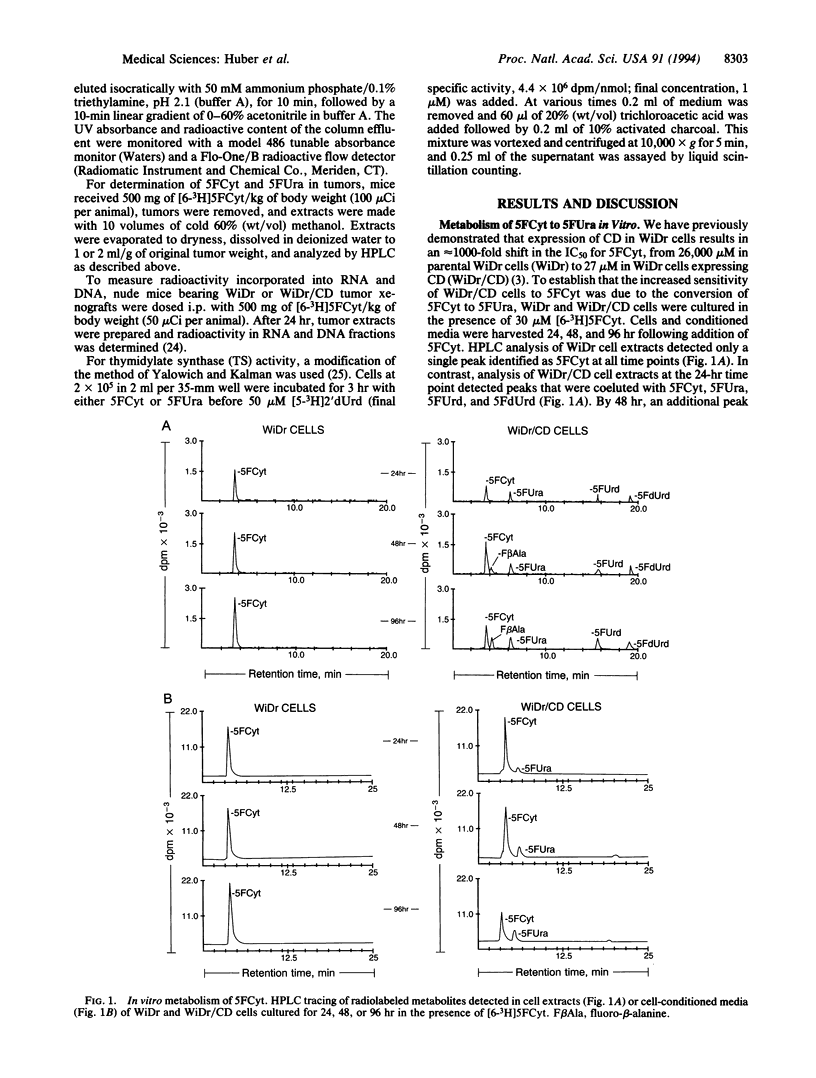
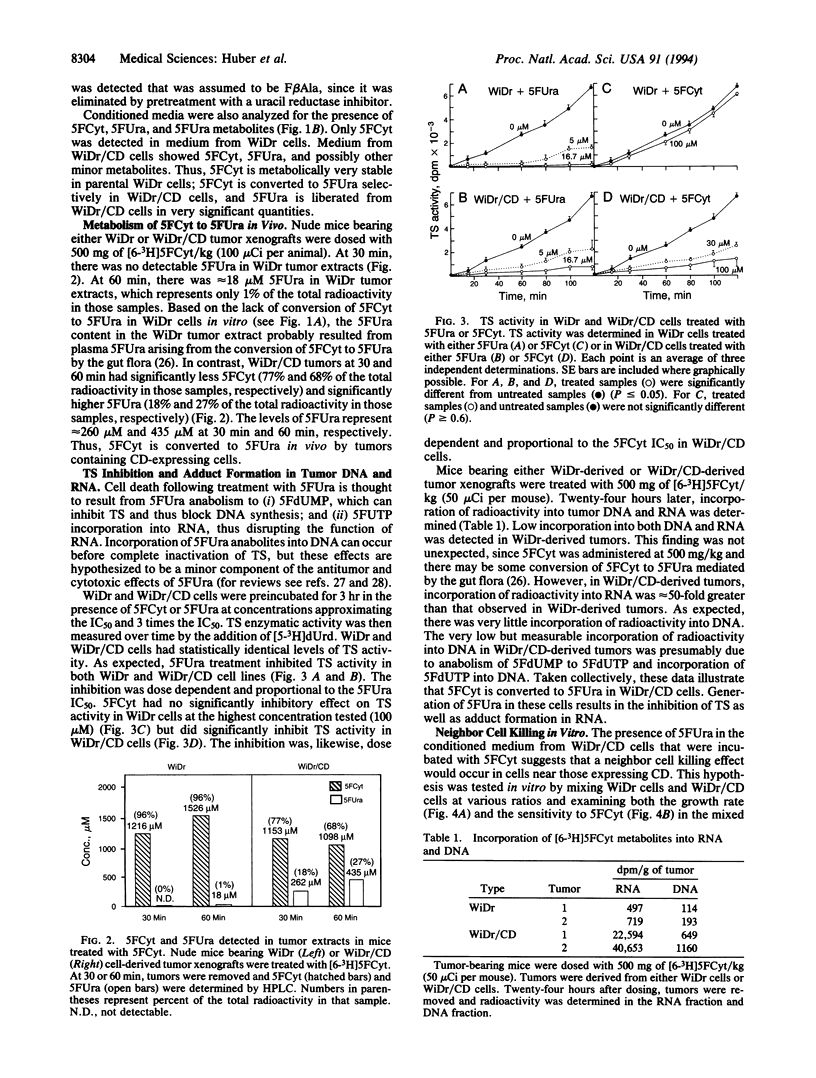
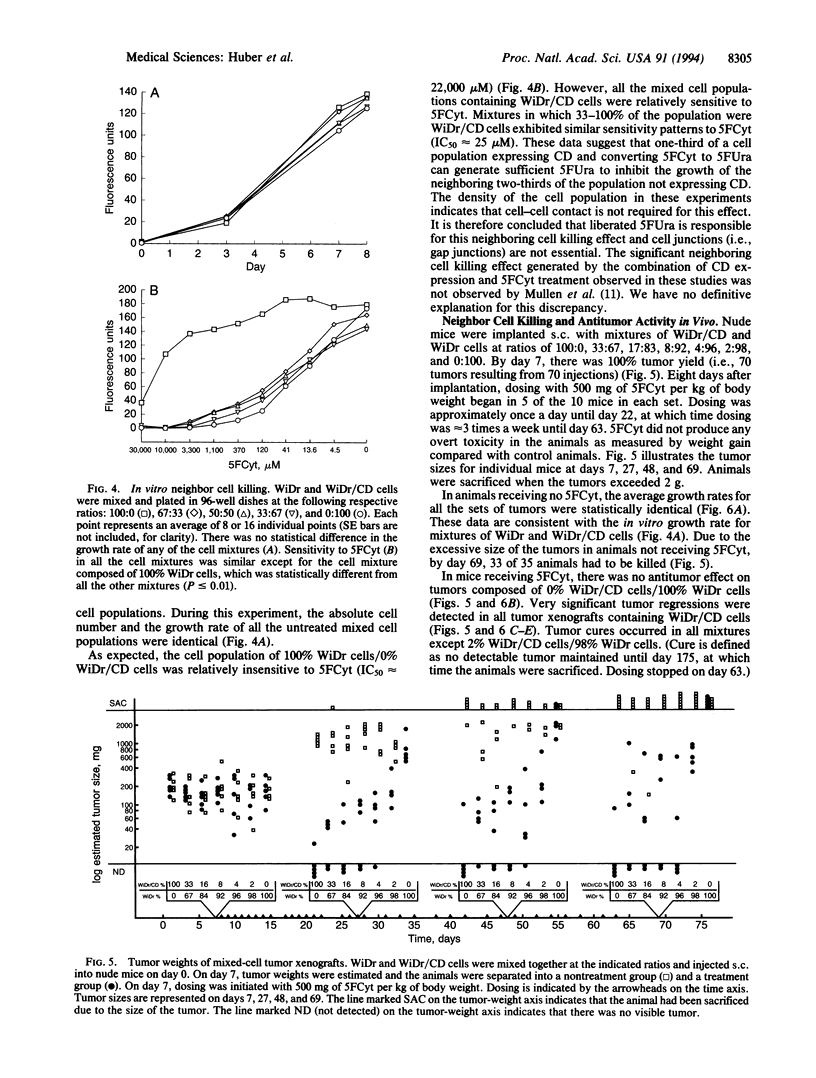
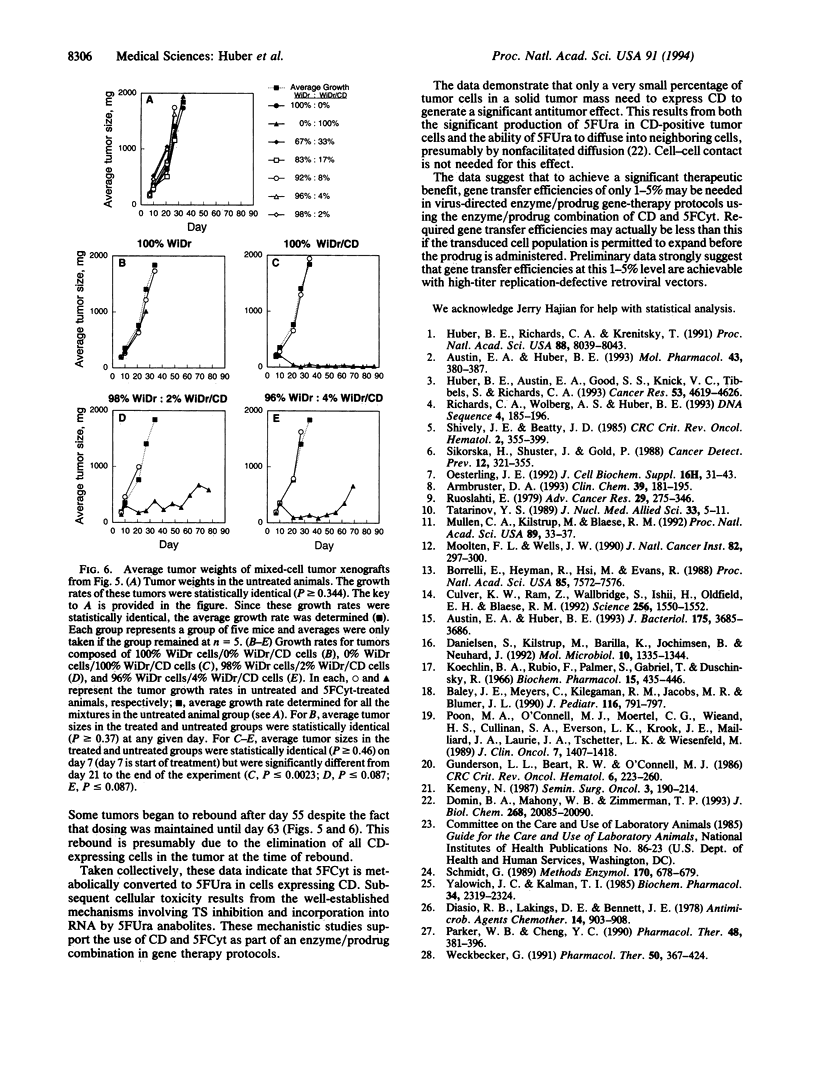
The gene encoding cytosine deaminase (CD) has been expressed in the human colorectal carcinoma cell line WiDr. Metabolism studies confirm that tumor cells expressing CD convert the very nontoxic prodrug 5-fluorocytosine (5FCyt) to 5-fluorouracil (5FUra) and 5FUra metabolites. Tumor xenografts composed of CD-expressing cells can selectively generate tumor levels of > 400 microM 5FUra when the host mouse is dosed with nontoxic levels of 5FCyt. The selective metabolic conversion of 5FCyt to 5FUra in CD-expressing tumor cells results in the inhibition of thymidylate synthase and incorporation of 5FUra into RNA. 5FUra is also liberated into the surrounding environment when CD-expressing tumor cells are treated with 5FCyt. The liberated 5FUra is able to kill neighboring, non-CD-expressing tumor cells in vitro and in vivo. Most importantly, when only 2% of the tumor mass contains CD-expressing cells (98% non-CD-expressing cells), significant regressions in all tumors are observed when the host mouse is dosed with nontoxic levels of 5FCyt.
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armbruster D. A. Prostate-specific antigen: biochemistry, analytical methods, and clinical application. Clin Chem. 1993 Feb;39(2):181–195. [PubMed] [Google Scholar]
- Austin E. A., Huber B. E. A first step in the development of gene therapy for colorectal carcinoma: cloning, sequencing, and expression of Escherichia coli cytosine deaminase. Mol Pharmacol. 1993 Mar;43(3):380–387. [PubMed] [Google Scholar]
- Austin E. A., Huber B. E. Localization of the codA gene on the Escherichia coli chromosome. J Bacteriol. 1993 Jun;175(11):3685–3686. doi: 10.1128/jb.175.11.3685-3686.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baley J. E., Meyers C., Kliegman R. M., Jacobs M. R., Blumer J. L. Pharmacokinetics, outcome of treatment, and toxic effects of amphotericin B and 5-fluorocytosine in neonates. J Pediatr. 1990 May;116(5):791–797. doi: 10.1016/s0022-3476(05)82674-5. [DOI] [PubMed] [Google Scholar]
- Borrelli E., Heyman R., Hsi M., Evans R. M. Targeting of an inducible toxic phenotype in animal cells. Proc Natl Acad Sci U S A. 1988 Oct;85(20):7572–7576. doi: 10.1073/pnas.85.20.7572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Culver K. W., Ram Z., Wallbridge S., Ishii H., Oldfield E. H., Blaese R. M. In vivo gene transfer with retroviral vector-producer cells for treatment of experimental brain tumors. Science. 1992 Jun 12;256(5063):1550–1552. doi: 10.1126/science.1317968. [DOI] [PubMed] [Google Scholar]
- Danielsen S., Kilstrup M., Barilla K., Jochimsen B., Neuhard J. Characterization of the Escherichia coli codBA operon encoding cytosine permease and cytosine deaminase. Mol Microbiol. 1992 May;6(10):1335–1344. doi: 10.1111/j.1365-2958.1992.tb00854.x. [DOI] [PubMed] [Google Scholar]
- Diasio R. B., Lakings D. E., Bennett J. E. Evidence for conversion of 5-fluorocytosine to 5-fluorouracil in humans: possible factor in 5-fluorocytosine clinical toxicity. Antimicrob Agents Chemother. 1978 Dec;14(6):903–908. doi: 10.1128/aac.14.6.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffith D. A., Jarvis S. M. High affinity sodium-dependent nucleobase transport in cultured renal epithelial cells (LLC-PK1). J Biol Chem. 1993 Sep 25;268(27):20085–20090. [PubMed] [Google Scholar]
- Gunderson L. L., Beart R. W., O'Connell M. J. Current issues in the treatment of colorectal cancer. Crit Rev Oncol Hematol. 1986;6(3):223–260. doi: 10.1016/s1040-8428(86)80057-9. [DOI] [PubMed] [Google Scholar]
- Huber B. E., Austin E. A., Good S. S., Knick V. C., Tibbels S., Richards C. A. In vivo antitumor activity of 5-fluorocytosine on human colorectal carcinoma cells genetically modified to express cytosine deaminase. Cancer Res. 1993 Oct 1;53(19):4619–4626. [PubMed] [Google Scholar]
- Huber B. E., Richards C. A., Krenitsky T. A. Retroviral-mediated gene therapy for the treatment of hepatocellular carcinoma: an innovative approach for cancer therapy. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):8039–8043. doi: 10.1073/pnas.88.18.8039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemeny N. Role of chemotherapy in the treatment of colorectal carcinoma. Semin Surg Oncol. 1987;3(3):190–214. doi: 10.1002/ssu.2980030312. [DOI] [PubMed] [Google Scholar]
- Koechlin B. A., Rubio F., Palmer S., Gabriel T., Duschinsky R. The metabolism of 5-fluorocytosine-2-14-C and of cytosine-14-C in the rat and the disposition of 5-fluorocytosine-2-14-C in man. Biochem Pharmacol. 1966 Apr;15(4):435–446. doi: 10.1016/0006-2952(66)90254-1. [DOI] [PubMed] [Google Scholar]
- Moolten F. L., Wells J. M. Curability of tumors bearing herpes thymidine kinase genes transferred by retroviral vectors. J Natl Cancer Inst. 1990 Feb 21;82(4):297–300. doi: 10.1093/jnci/82.4.297. [DOI] [PubMed] [Google Scholar]
- Mullen C. A., Kilstrup M., Blaese R. M. Transfer of the bacterial gene for cytosine deaminase to mammalian cells confers lethal sensitivity to 5-fluorocytosine: a negative selection system. Proc Natl Acad Sci U S A. 1992 Jan 1;89(1):33–37. doi: 10.1073/pnas.89.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oesterling J. E. Prostate-specific antigen and diagnosing early malignancies of the prostate. J Cell Biochem Suppl. 1992;16H:31–43. doi: 10.1002/jcb.240501209. [DOI] [PubMed] [Google Scholar]
- Poon M. A., O'Connell M. J., Moertel C. G., Wieand H. S., Cullinan S. A., Everson L. K., Krook J. E., Mailliard J. A., Laurie J. A., Tschetter L. K. Biochemical modulation of fluorouracil: evidence of significant improvement of survival and quality of life in patients with advanced colorectal carcinoma. J Clin Oncol. 1989 Oct;7(10):1407–1418. doi: 10.1200/JCO.1989.7.10.1407. [DOI] [PubMed] [Google Scholar]
- Richards C. A., Wolberg A. S., Huber B. E. The transcriptional control region of the human carcinoembryonic antigen gene: DNA sequence and homology studies. DNA Seq. 1993;4(3):185–196. doi: 10.3109/10425179309015631. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Seppälä M. alpha-Fetoprotein in cancer and fetal development. Adv Cancer Res. 1979;29:275–346. doi: 10.1016/s0065-230x(08)60849-0. [DOI] [PubMed] [Google Scholar]
- Shively J. E., Beatty J. D. CEA-related antigens: molecular biology and clinical significance. Crit Rev Oncol Hematol. 1985;2(4):355–399. doi: 10.1016/s1040-8428(85)80008-1. [DOI] [PubMed] [Google Scholar]
- Sikorska H., Shuster J., Gold P. Clinical applications of carcinoembryonic antigen. Cancer Detect Prev. 1988;12(1-6):321–355. [PubMed] [Google Scholar]
- Tatarinov Y. S. The past and the future for cancer testing by alpha-fetoprotein. A review. J Nucl Med Allied Sci. 1989 Jul-Sep;33(3 Suppl):5–11. [PubMed] [Google Scholar]
- Weckbecker G. Biochemical pharmacology and analysis of fluoropyrimidines alone and in combination with modulators. Pharmacol Ther. 1991;50(3):367–424. doi: 10.1016/0163-7258(91)90051-m. [DOI] [PubMed] [Google Scholar]
- Yalowich J. C., Kalman T. I. Rapid determination of thymidylate synthase activity and its inhibition in intact L1210 leukemia cells in vitro. Biochem Pharmacol. 1985 Jul 1;34(13):2319–2324. doi: 10.1016/0006-2952(85)90788-9. [DOI] [PubMed] [Google Scholar]



