Abstract
Purpose of the review
B-cell tumors originating from the transformation of germinal center (GC) B-cells frequently harbor genetic mutations leading to constitutive activation of the nuclear factor-κB (NF-κB) signaling pathway. This review highlights recent insights into the roles of separate NF-κB transcription factors in GC B-cell development and discusses implications of the results for GC-lymphomagenesis.
Recent findings
Understanding how aberrant NF-κB-activation promotes tumorigenesis requires the understanding of the role of NF-κB in the tumor-precursor cells. Despite extensive knowledge on NF-κB biology, the function of this complex signaling pathway in the differentiation of GC B-cells is largely unknown. This review will discuss recent findings that revealed distinct roles of separate NF-κB transcription factors during the GC-reaction in the context of GC-lymphomagenesis. Most notably, a single NF-κB subunit, c-REL, was found to be required for the maintenance of the GC-reaction and was associated with the activation of a metabolic program that promotes cell growth.
Summary
Identifying the biological roles of the separate NF-κB transcription factor subunits in GC-biology will help to better understand the pathogenic consequences of their constitutive activation in B-cell tumors. This knowledge may be exploited for the development of targeted anti-tumor therapies aimed at inhibiting selectively those components of aberrant NF-κB activity which contribute to pathogenesis.
Keywords: B-cell lymphoma, lymphomagenesis, germinal center, NF-κB
Introduction
The hallmark of T cell-dependent antibody responses is the generation of high-affinity memory B-cells and plasma cells during the germinal center (GC)-reaction. The GC-reaction is essential for our immunity against invading microorganisms; however, this reaction is inherently dangerous, as suggested by the observation that the majority of human B-cell malignancies originate from the transformation of antigen-experienced B-cells that have undergone the GC-reaction[1,2].
The GC-reaction
GCs are specialized microenvironments that form upon T-dependent activation of antigen-specific B-cells within lymph nodes or spleen[3,4]. A mature GC comprises two functionally distinct compartments, a dark zone (DZ) and a light zone (LZ)[5,6]. DZ B-cells undergo rapid proliferation during which their antibody genes are modified by activation-induced cytidine deaminase (AID)-mediated immunoglobulin variable region (IgV) gene somatic hypermutation to generate a repertoire of antibody mutants with varying affinities to the immunizing antigen. Those with improved affinity to the antigen are positively selected in the LZ, where the cells may also undergo AID-mediated class switch recombination. Selected B-cells can recirculate to the DZ to undergo additional rounds of hypermutation and affinity-selection, or they are instructed to differentiate into memory B-cells or plasma cells and exit the GC.
GC-reaction and lymphomagenesis
Occasional errors during the AID-mediated, DNA-modifying processes of IgV hypermutation and class switch recombination can cause genetic alterations in GC B-cells, leading to the deregulated or ectopic expression of oncogenes or the inactivation of tumor suppressor genes[7]. Importantly, it has emerged that the vast majority of genetic aberrations target genes with essential functions during GC B-cell development, such as MYC, BCL2, BCL6, and PRDM1 (encoding BLIMP1)[8,9]. As a result, the deregulated expression or inactivation of those genes disturbs the normal physiology of the GC-reaction by exerting pro-survival or pro-proliferative effects, or by inhibiting the differentiation into post-GC memory B-cells and plasma cells. In order to better understand how oncogenes or tumor suppressors contribute to GC-lymphomagenesis, it is important to identify the functions of these genes during normal GC B-cell development. The recent finding that deregulated activity of NF-κB is a major contributor to the pathogenesis of various GC-derived B-cell malignancies underscores the need to dissect the function of the complex NF-κB pathway in GC B-cell development.
The NF-κB pathway
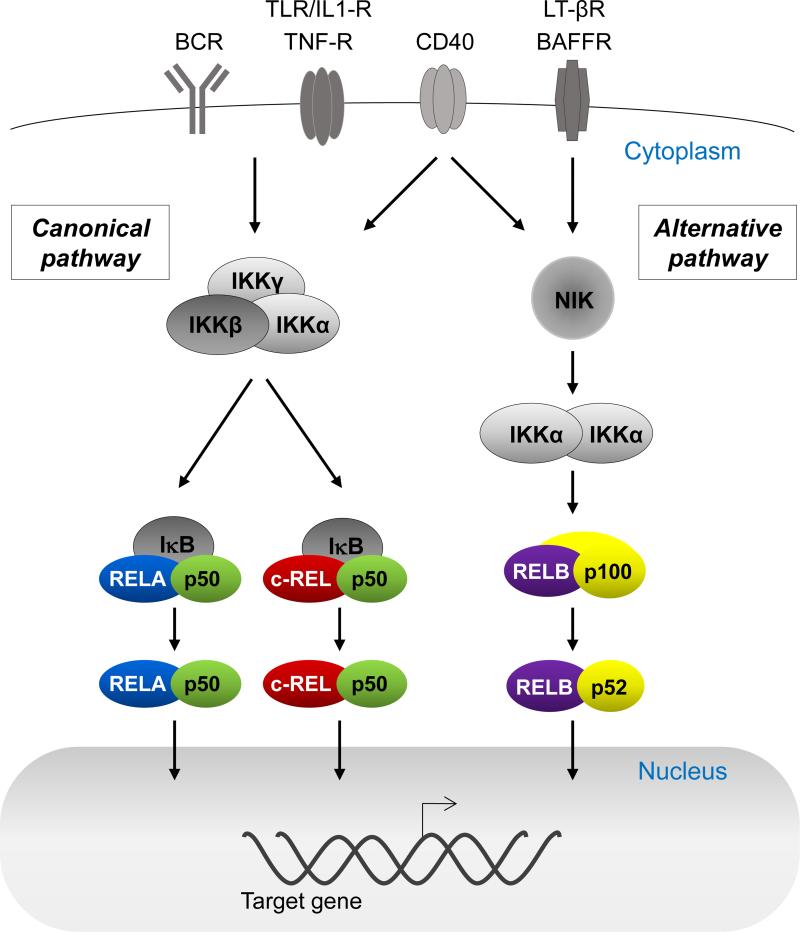
NF-κB transcription factors are associated with controlling the expression of genes involved in cell survival, growth, stress responses and inflammation in a cell-type or context-dependent fashion[10,11]. The five different NF-κB subunits c-REL, RELA, RELB, p50 and p52 occur as heterodimers or homodimers (p50 and p52 are generated by proteolytic cleavage from their precursors p105 and p100, respectively). Only the c-REL, RELA and RELB subunits can drive transcription through transactivation domains (TAD). Upon activation, the dimers translocate from the cytoplasm to the nucleus, where they activate transcription of hundreds of target genes. Functionally, NF-κB transcription factors form two groups (Fig. 1). RELA, c-REL and p50 comprise the subunits of the canonical NF-κB pathway that in B-cells is mainly activated in response to stimulation through the antigen and Toll-like receptors as well as CD40-ligation[12,13]. The major heterodimers are RELA/p50 and c-REL/p50 (Fig. 1). RELB and p52 comprise the factors of the alternative pathway that in B-cells is activated by CD40-ligation or BAFF[12,13] and occur as heterodimers.
Figure 1. NF-κB signaling pathway.
NF-κB is a complex signaling cascade that comprises two separate pathways. The canonical pathway (left) is activated via signals through a range of cell surface receptors, most importantly the B-cell receptor (BCR) and CD40. The activation results in the inactivation of IκB, thereby releasing the heterodimers formed by the canonical NF-κB subunits RELA, c-REL and p50 to translocate into the nucleus and to activate transcription of target genes. The alternative pathway (right) is activated by a more limited set of signals, including CD40. Proteasomal degradation of the precursor protein p100 results in the generation of the major heterodimer of the alternative pathway RELB/p52, which can then enter the nucleus and activate transcription of target genes. Only RELA, RELB, and c-REL can drive transcription of target genes due to transactivation domains.
Aberrant activation of NF-κB in B-cell malignancies
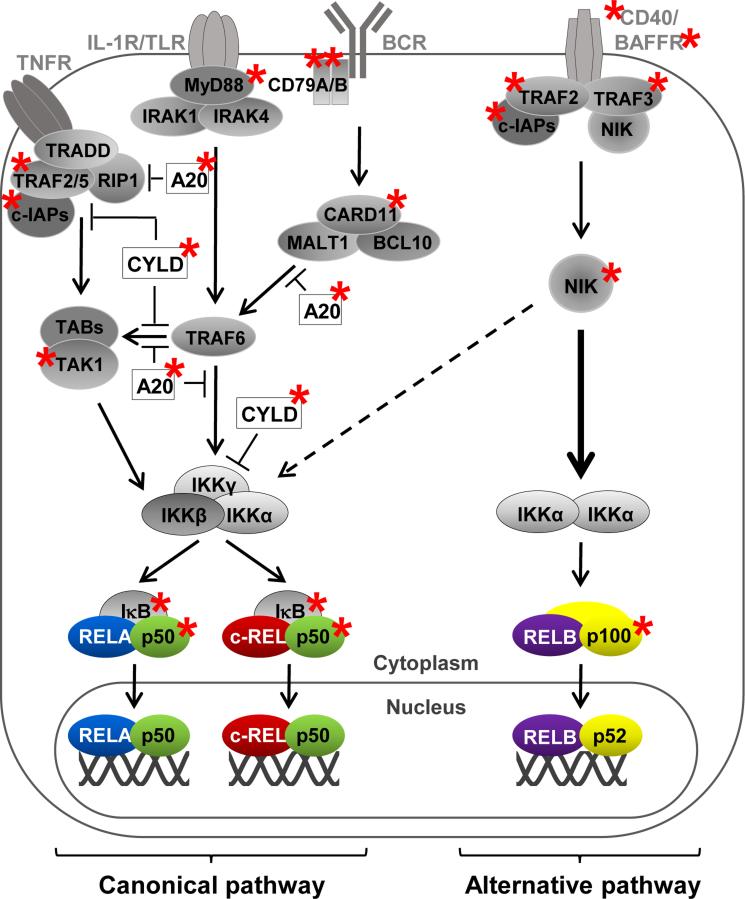
Constitutive NF-κB activity occurs in several B cell-derived malignancies[9,14-19]. NF-κB can be activated by the tumor microenvironment[20] or as a result of infection with tumor viruses such as EBV or KSHV[21]. Recently, it emerged that a large percentage of Hodgkin lymphoma (HL), non-Hodgkin lymphoma (NHL) and multiple myeloma (MM) cases show constitutive NF-κB signaling as the result of genetic alterations in NF-κB pathway components[9,22-32] (Fig. 2). Importantly, the inhibition of abnormal NF-κB responses has effectively suppressed tumorigenesis in in vitro and in vivo assays[22-24], consistent with the demonstration of an oncogenic role for NF-κB in mice[33].
Figure 2. Multiple mutations in NF-κ pathway components can constitutively activate the canonical or alternative pathway.
Summary of genetic mutations in NF-κB pathway components that result in the constitutive activation of the canonical or alternative pathway (see text). Genes in which mutations leading to constitutive activation of NF-κB were identified are marked with an asterisk.
Mutations have been identified in NF-κB pathway components that lead to the preferential activation of either the canonical or the alternative pathway. Particular tumor subtypes seem to be associated with an activation of particular NF-κB pathways, although this distinction is not absolute. The vast majority of cases of activated B cell-type (ABC)-diffuse large B-cell lymphoma (DLBCL), the more aggressive subtype of DLBCL, carry genetic mutations that lead to the activation of the canonical pathway[9,22-24]; only ~10% of mutations are associated with aberrantly activating the alternative pathway[34-36], although the percentage of cases with nuclear p52 is higher[23], suggesting a broader role for this pathway. Also HL shows predominantly mutations leading to the activation of the canonical pathway[25,32], with a subset of HL harboring mutations in components of the alternative pathway[37,38]. Splenic marginal zone lymphoma harbors genetic aberrations that activate the canonical or alternative pathway at almost equal fractions[39]. Interestingly, a recent study reports that a fraction of mantle cell lymphoma cases that were insensitive to the treatment of inhibitors of the canonical pathway showed genetic aberrations in regulators of the alternative pathway and were susceptible to pharmacologic downmodulation of this pathway[40]. Whereas most mutations in MM occur in upstream components associated with inducing the alternative pathway[27-29], it has been established that in this malignancy of transformed plasma cells, both NF-κB pathways are activated in the majority of cases[27,29,41]. Certain lymphoma subtypes including germinal center B cell-type (GC)-DLBCLs, HL and mediastinal large B-cell lymphoma (MLBCL) show amplification of the REL (c-REL) locus[17,42-44]. It has been noted that HL and MLBCL are associated with predominant nuclear translocation of c-REL[15-17], suggesting unique functions for single canonical NF-κB subunits in the pathogenesis of different lymphoma subtypes. Finally, chronic lymphocytic leukemia (CLL) tumor cells show activation of the canonical route[20,45], which is thought to be due to chronic antigen-stimulation of the B-cell receptor[46] and stimulation of CLL cells mediated by the tumor environment[20]. Recurrent mutations were identified in a negative regulator of the alternative NF-κB pathway in about ~10% of CLL cases[47] as well as in two components of the canonical pathway at a similar rate[48-50].
In summary, aberrant NF-κB activation has been linked to the pathogenesis of many B cell-derived malignancies, and the picture emerges that the canonical or alternative pathways, and potentially the separate NF-κB subunits, have specific roles in the oncogenic process. This specificity may be exploited for the development of targeted anti-tumor therapies, thus reducing systemic side effects of ubiquitous NF-κB inhibition. An important step toward this goal is to understand the function of NF-κB in the tumor precursor cells.
NF-κB transcription factors in GC B-cell development
While the roles of NF-κB in early B-cell development and naïve B-cell survival are well defined[12,13], the precise function of the separate NF-κB pathways and their subunits during the GC-reaction and the differentiation into memory or plasma cells is less clear. While NF-κB is known to be required during the initial antigen-dependent B-cell activation phase[12,13] that leads to GC-formation, gene expression profiling analyses and immunohistochemistry studies in the human revealed that the vast majority of GC B-cells, i.e. DZ and most LZ B-cells, are not subjected to NF-κB signaling[51-53]. However, NF-κB is activated in a subset of LZ B-cells presumably by stimulation of the B-cell receptor and the CD40-signaling pathway resulting in nuclear translocation of the canonical NF-κB subunits[52] (and likely also of the alternative subunits as CD40 is a strong activator of this pathway). Together, this indicates that NF-κB follows a biphasic activation pattern in B-cells during the GC-reaction, which prevented the study of the role of NF-κB transcription factors in GC B-cells using constitutional knockout mice. Recently, the development of mouse models for conditional inactivation of NF-κB transcription factors has allowed to investigate this issue. We here focus on recent results from the analysis of the canonical NF-κB subunits c-REL and RELA in GC B-cell development.
c-REL is required for the maintenance of the GC-reaction
Constitutional rel (encoding c-REL) knockout mice generate a normal mature B-cell repertoire[54-56], indicating that c-REL is not required for the maintenance of naïve B-cells, or that this subunit is functionally redundant with RELA. However, in vitro-stimulation experiments clearly documented a role for c-REL during B-cell activation[54-57]. In accordance, c-REL-deficient mice are impaired in the formation of GCs upon T-dependent immunization[56,58] in a B cell-intrinsic fashion (own unpublished observations). The question of how deletion of rel in the small subset of LZ B-cells that exhibit nuclear translocation of c-REL affects GC development was addressed by crossing a conditional rel allele to mice that express the Cre-recombinase in GC B-cells. These experiments revealed that deletion of rel in GC B-cells led to the gradual collapse of mature GCs until the structure almost completely disappeared several days later[59]. The observation that both DZ and LZ B-cells disappeared at equal fractions suggests that c-REL is essential for the maintenance of the mature GC by controlling the cyclic reentry of antigen-selected LZ B-cells back to the DZ.
The GC collapse observed upon deletion of rel in GC B-cells could not be rescued by constitutive anti-apoptotic stimuli via a bcl2-transgene[59], indicating that prevention of apoptosis is unlikely to be c-REL's major function during selection in the GC. Instead, gene expression profile analysis indicated that rel-deleted GC B-cells lacked the expression of a metabolic program that enables cell growth by providing the increased demands of rapidly cycling cells for energy and building blocks for anabolic reactions[59]. In accordance with these findings, in vitro-stimulated c-REL-deficient B-cells failed to upregulate a metabolic program, were characterized by strongly impaired metabolic functions as measured by extracellular flux assays, and had a smaller cell size compared to c-REL-proficient B-cells[59]. It therefore seems that c-REL's primary role in LZ B-cells may be the establishment of a transcriptional program that mediates cell growth through facilitating enhanced biosynthesis of DNA, protein and lipids. These findings add c-REL to a growing list of transcription factors that over the last few years have been found to be involved in the control of metabolic reprogramming during the differentiation of activated lymphocytes[60-63], and provide an additional example for the emerging role of NF-κB transcription factors in the regulation of metabolic processes[64,65].
The gradual disappearance of GCs upon rel deletion is strikingly reminiscent of the GC collapse observed upon functional inactivation of the c-MYC proto-oncogene in mature GCs[66,67]. It therefore seems that both transcription factors are required for sustaining the GC-reaction by instructing positively selected B-cells to recycle from the LZ back to the DZ. The interplay between c-REL and c-MYC in the LZ B-cells is currently unclear. A NF-κB signature is present in c-MYC+ LZ B-cells[66], and vice versa, c-REL-deficient B-cells lack a c-MYC signature[59], suggesting that both transcription factors are active in the same LZ B-cells. While c-MYC can be a NF-κB target gene[64], it seems unlikely that c-REL exerts its function in LZ B-cells solely through the control of c-MYC. Rather, c-MYC and c-REL may jointly regulate a particular set of target genes in addition to their specific transcriptional targets. Future work is needed to dissect the relation between c-MYC and c-REL in GC B-cell development.
RELA is required for the generation of GC-derived plasma cells
Mainly due to the embryonic lethality observed upon constitutional rela deletion[68], relatively little is known about the role of the canonical NF-κB subunit RELA in mature B-cell development. Evidence suggests that, similar to c-REL, RELA is not required for the generation of a normal mature B-cell repertoire[69]. However, data obtained from the study of conditional knockout mice suggest that RELA and c-REL play distinct roles during the GC-reaction. Specifically, in contrast to c-REL inactivation, deletion of rela in GC B-cells did not affect GC maintenance, but impaired the generation of GC-derived plasma cells[59]. The precise mechanism by which RELA induces terminal differentiation in concert with other transcriptional regulators required for plasma cell development remains to be determined. However, in vitro experiments suggest that RELA contributes to the transcription factor network that controls plasma cell differentiation by upregulating the expression of the plasma cell regulator BLIMP1[59].
Implications for GC lymphomagenesis
REL has been identified as a viral oncogene causing reticuloendotheliosis in birds[70]. The amplification of the REL locus in several types of B-cell lymphomas[17,42-44] and the occurrence in lymphomas of genetic mutations leading to constitutive activation of the canonical NF-κB pathway suggest an oncogenic function for c-REL also in humans. The finding that in normal GC B-cells, c-REL-deficiency results in the failure to establish a metabolic program required for cell growth[59] may in turn suggest that aberrantly elevated c-REL activity could be oncogenic by enhancing cellular metabolism with direct effects on cell growth (Fig. 3). Gene expression profiling has allowed the classification of DLBCL cases into subtypes that show differences in the particular metabolic program of the tumor cells[18]. One subgroup is characterized by the expression of genes involved in oxidative phosphorylation (OxPhos), and it has been noted that DLBCL cases corresponding to a non-OxPhos subgroup (mostly comprising cases with a signature relating to B-cell receptor signaling/proliferation) show enhanced glycolytic flux[71]. It will be interesting to determine the extent to which c-REL activation contributes to the respective metabolic phenotypes.
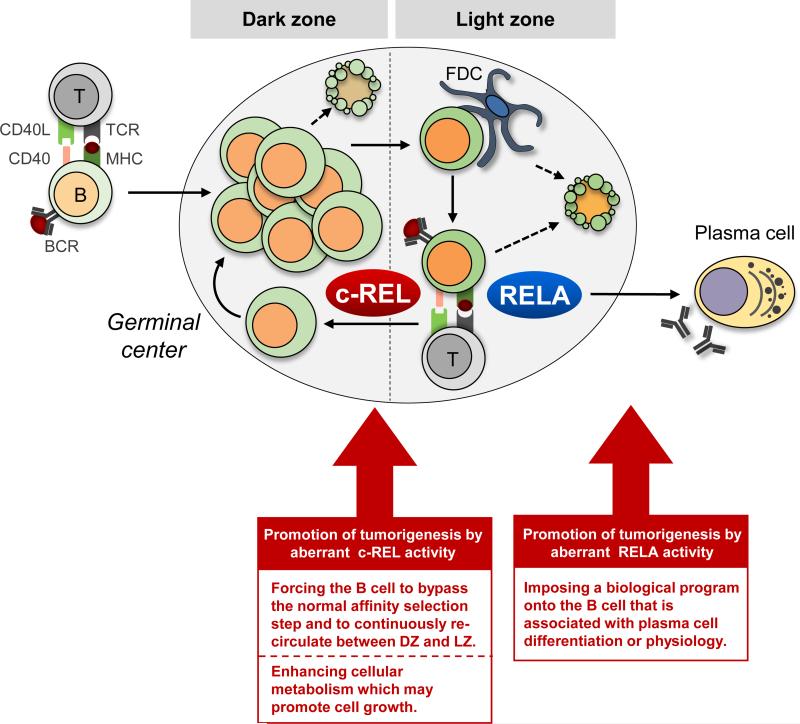
Figure 3. Hypothesis: consequences of c-REL or RELA deregulation in GC B-cells or plasma cells.
Due to its role in GC B-cell metabolism and growth, aberrant c-REL activity might promote tumorigenesis by allowing the cell to bypass affinity selection in the GC and recirculate between DZ and LZ in an uncontrolled fashion. In contrast, aberrant RELA activity in GC B-cells may disturb the physiological processes controlling the development of plasma cells. B − B-cell; T − T-cell; FDC − follicular dendritic cell.
c-REL appears to maintain the GC-reaction by licensing the recirculation of antigen-selected B-cells from the LZ back to the DZ. Such a LZ-DZ reentry function has previously been established for the c-MYC proto-oncogene[66,67], whose expression is deregulated in several GC-lymphoma subtypes. It has been suggested that in GC-lymphomas with MYC-translocations, constitutive expression of c-MYC may promote tumorigenesis by forcing the B-cell to bypass the normal affinity-selection step in the LZ and to continuously recirculate between LZ and DZ[66]. Deregulated c-REL activity may similarly perturb the normal dynamics of the GC-reaction (Fig. 3). It is tempting to speculate that the aberrant activities of these transcription factors exert oncogenic roles specifically in the subpopulation of LZ B-cells that undergo antigen selection by preventing their further differentiation into memory or plasma cells. During GC development, this likely occurs at a cellular stage that precedes the plasmablastic stage in which PRDM1 inactivation or constitutive BCL6 activity is thought to inhibit terminal differentiation[9].
Among DLBCL cases, MYC translocations and REL amplifications occur predominantly in the GC-subtype. It has been noted that in GC-DLBCL with amplification of REL, there is a lack of correlation between REL amplification and nuclear translocation of the subunit[72]. Clearly, increased levels of c-REL are unlikely to be biologically active unless the canonical pathway is induced. In GC-DLBCL, that, in contrast to ABC-DLBCL, is rarely associated with activating mutations in the canonical NF-κB pathway, one could envision that stimuli by the tumor microenvironment in a small fraction of cells may drive c-REL into the nucleus. Enhanced protein levels of c-REL would then lead to an altered NF-κB response in thus stimulated GC-DLBCL tumor cells. With regard to other GC-derived tumor types, c-REL is found in the nucleus of a large fraction of tumor cells in many DLBCLs mostly of the ABC-type[72] as well as in HL and MLBCL regardless of amplification of the REL locus[15-17]. Mutations in upstream components of the canonical NF-κB pathway such as A20 may lead to the continuous translocation of c-REL/p50 heterodimers into the nucleus. It will be interesting to determine the specific biological programs controlled by c-REL in the corresponding tumor cells.
Aberrant RELA activity in GC B-cells may impose a biological program onto the cell that is associated with plasma cell differentiation or physiology (Fig. 3). Besides ABC-DLBCL, constitutive RELA activation has been associated with MM[27,28], where it may render the tumor cells less dependent on NF-κB activation mediated by ligands that are required for the survival of plasma cells within the bone-marrow niches, allowing stromal-independent tumor cell growth. Future work is needed to define the precise function of RELA in GC-lymphomas and MM.
A role for the alternative NF-κB pathway during the GC-reaction is highly likely in light of the fact that CD40-stimulation (which occurs in a subset of light zone B-cells) strongly activates this pathway, and since several genetic aberrations lead to the predominant activation of this signaling route. Indeed, the contribution of this pathway to lymphomagenesis is actively being explored[73,74]. Importantly, a recent study provides functional evidence for a role of aberrant activation of the alternative NF-κB pathway in GC lymphomagenesis. Mice with constitutive activation of NF-κB inducing kinase (NIK), a critical upstream component of the alternative pathway, specifically in GC B-cells led to DLBCL development upon simultaneous deregulation of BCL6-expression[36]. The study further provides evidence that constitutive NIK activity in GC B-cells promotes plasma cell hyperplasia; upon blocking terminal differentiation by deregulated BCL6-expression, lymphomas developed. It will be interesting to identify the precise roles of the alternative NF-κB subunits RELB and p100/p52 in GC-development.
Finally, the ability to identify the targets to which the individual NF-κB subunits bind will help to better understand the selectivity of the NF-κB response[75]. In addition, the precise dissection of the signaling network upstream of the transcription factor subunits will help to better define NF-κB pathway activation[76,77]. The ultimate goal should be to inhibit only those components of aberrant NF-κB activity that are pathogenic, ideally at the level of NF-κB subunits to ensure the highest specificity[78]. In this regard, the recent characterization of a small molecule c-REL inhibitor may hold promise for the development of anti-cancer therapies that enable targeting of individual NF-κB subunits[79].
Conclusion
It is now firmly established that aberrant activity of the NF-κB pathway plays an important role in B-cell lymphomagenesis. It is also becoming clear that distinct B-cell tumors are characterized by a preferential activity of the canonical or alternative NF-κB pathways and presumably also individual NF-κB subunits. This specificity may be exploited for the development of targeted therapies aimed at inhibiting selectively those components of the NF-κB pathway that directly contribute to pathogenesis. Unraveling the selectivity of the NF-κB response in normal and cancerous B-cells will be instrumental in this undertaking.
Key Points.
A large percentage of Hodgkin lymphoma (HL), non-Hodgkin lymphoma (NHL) and multiple myeloma (MM) cases show constitutive NF-κB signaling as the result of genetic alterations in NF-κB pathway components.
Particular tumor subtypes seem to be associated with a preferential activation of either the canonical or the alternative NF-κB pathway or individual NF-κB transcription factors.
Identifying the biological roles of the separate NF-κB transcription factor subunits in GC-biology will help to better understand the pathogenic consequences of their constitutive activation in B-cell tumors.
A single NF-κB subunit, c-REL, was found to be required for the maintenance of the GC-reaction and was associated with the activation of a metabolic program that promotes cell growth.
The findings that the two NF-κB pathways and potentially the separate NF-κB subunits seem to have specific roles in the oncogenic process may be exploited for the development of targeted anti-tumor therapies, thus reducing systemic side effects of ubiquitous NF-κB inhibition.
Acknowledgements
We thank Laura Pasqualucci for comments on the manuscript and the members of the Klein lab for discussions.
Financial support and sponsorship
This work was supported by NCI/NIH grant R01-CA157660 to UK and by a fellowship from the German Research Council (DFG) to NH.
Footnotes
Conflicts of interest
The authors declare no conflicts of interest.
References
- 1.Stevenson F, Sahota S, Zhu D, et al. Insight into the origin and clonal history of B-cell tumors as revealed by analysis of immunoglobulin variable region genes. Immunol Rev. 1998;162:247–259. doi: 10.1111/j.1600-065x.1998.tb01446.x. [DOI] [PubMed] [Google Scholar]
- 2.Küppers R, Klein U, Hansmann ML, et al. Cellular origin of human B-cell lymphomas. N Engl J Med. 1999;341:1520–1529. doi: 10.1056/NEJM199911113412007. [DOI] [PubMed] [Google Scholar]
- 3.MacLennan IC. Germinal centers. Annu Rev Immunol. 1994;12:117–139. doi: 10.1146/annurev.iy.12.040194.001001. [DOI] [PubMed] [Google Scholar]
- 4.Rajewsky K. Clonal selection and learning in the antibody system. Nature. 1996;381:751–758. doi: 10.1038/381751a0. [DOI] [PubMed] [Google Scholar]
- 5.Allen CD, Okada T, Cyster JG. Germinal-center organization and cellular dynamics. Immunity. 2007;27:190–202. doi: 10.1016/j.immuni.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Victora GD, Nussenzweig MC. Germinal centers. Annu Rev Immunol. 2012;30:429–457. doi: 10.1146/annurev-immunol-020711-075032. [DOI] [PubMed] [Google Scholar]
- 7.Küppers R. Mechanisms of B-cell lymphoma pathogenesis. Nat Rev Cancer. 2005;5:251–262. doi: 10.1038/nrc1589. [DOI] [PubMed] [Google Scholar]
- 8.Klein U, Dalla-Favera R. Germinal centres: role in B-cell physiology and malignancy. Nat Rev Immunol. 2008;8:22–33. doi: 10.1038/nri2217. [DOI] [PubMed] [Google Scholar]
- 9.Shaffer AL, 3rd, Young RM, Staudt LM. Pathogenesis of human B cell lymphomas. Annu Rev Immunol. 2012;30:565–610. doi: 10.1146/annurev-immunol-020711-075027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hayden MS, Ghosh S. Signaling to NF-kappaB. Genes Dev. 2004;18:2195–2224. doi: 10.1101/gad.1228704. [DOI] [PubMed] [Google Scholar]
- 11.Vallabhapurapu S, Karin M. Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol. 2009;27:693–733. doi: 10.1146/annurev.immunol.021908.132641. [DOI] [PubMed] [Google Scholar]
- 12.Gerondakis S, Siebenlist U. Roles of the NF-kappaB pathway in lymphocyte development and function. Cold Spring Harb Perspect Biol. 2010;2:a000182. doi: 10.1101/cshperspect.a000182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kaileh M, Sen R. NF-kappaB function in B lymphocytes. Immunol Rev. 2012;246:254–271. doi: 10.1111/j.1600-065X.2012.01106.x. [DOI] [PubMed] [Google Scholar]
- 14.Alizadeh AA, Eisen MB, Davis RE, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000;403:503–511. doi: 10.1038/35000501. [DOI] [PubMed] [Google Scholar]
- 15.Rosenwald A, Wright G, Leroy K, et al. Molecular diagnosis of primary mediastinal B cell lymphoma identifies a clinically favorable subgroup of diffuse large B cell lymphoma related to Hodgkin lymphoma. J Exp Med. 2003;198:851–862. doi: 10.1084/jem.20031074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Savage KJ, Monti S, Kutok JL, et al. The molecular signature of mediastinal large B-cell lymphoma differs from that of other diffuse large B-cell lymphomas and shares features with classical Hodgkin lymphoma. Blood. 2003;102:3871–3879. doi: 10.1182/blood-2003-06-1841. [DOI] [PubMed] [Google Scholar]
- 17.Feuerhake F, Kutok JL, Monti S, et al. NFkappaB activity, function, and target-gene signatures in primary mediastinal large B-cell lymphoma and diffuse large B-cell lymphoma subtypes. Blood. 2005;106:1392–1399. doi: 10.1182/blood-2004-12-4901. [DOI] [PubMed] [Google Scholar]
- 18.Monti S, Savage KJ, Kutok JL, et al. Molecular profiling of diffuse large B-cell lymphoma identifies robust subtypes including one characterized by host inflammatory response. Blood. 2005;105:1851–1861. doi: 10.1182/blood-2004-07-2947. [DOI] [PubMed] [Google Scholar]
- 19.Hideshima T, Chauhan D, Richardson P, et al. NF-kappa B as a therapeutic target in multiple myeloma. J Biol Chem. 2002;277:16639–16647. doi: 10.1074/jbc.M200360200. [DOI] [PubMed] [Google Scholar]
- 20.Herishanu Y, Perez-Galan P, Liu D, et al. The lymph node microenvironment promotes B-cell receptor signaling, NF-kappaB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood. 2011;117:563–574. doi: 10.1182/blood-2010-05-284984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sun SC, Cesarman E. NF-kappaB as a target for oncogenic viruses. Curr Top Microbiol Immunol. 2011;349:197–244. doi: 10.1007/82_2010_108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lenz G, Davis RE, Ngo VN, et al. Oncogenic CARD11 mutations in human diffuse large B cell lymphoma. Science. 2008;319:1676–1679. doi: 10.1126/science.1153629. [DOI] [PubMed] [Google Scholar]
- 23.Compagno M, Lim WK, Grunn A, et al. Mutations of multiple genes cause deregulation of NF-kappaB in diffuse large B-cell lymphoma. Nature. 2009;459:717–721. doi: 10.1038/nature07968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kato M, Sanada M, Kato I, et al. Frequent inactivation of A20 in B-cell lymphomas. Nature. 2009;459:712–716. doi: 10.1038/nature07969. [DOI] [PubMed] [Google Scholar]
- 25.Schmitz R, Hansmann ML, Bohle V, et al. TNFAIP3 (A20) is a tumor suppressor gene in Hodgkin lymphoma and primary mediastinal B cell lymphoma. J Exp Med. 2009;206:981–989. doi: 10.1084/jem.20090528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Novak U, Rinaldi A, Kwee I, et al. The NF-{kappa}B negative regulator TNFAIP3 (A20) is inactivated by somatic mutations and genomic deletions in marginal zone lymphomas. Blood. 2009;113:4918–4921. doi: 10.1182/blood-2008-08-174110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Annunziata CM, Davis RE, Demchenko Y, et al. Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell. 2007;12:115–130. doi: 10.1016/j.ccr.2007.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Keats JJ, Fonseca R, Chesi M, et al. Promiscuous mutations activate the noncanonical NF-kappaB pathway in multiple myeloma. Cancer Cell. 2007;12:131–144. doi: 10.1016/j.ccr.2007.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Demchenko YN, Glebov OK, Zingone A, et al. Classical and/or alternative NF-kappaB pathway activation in multiple myeloma. Blood. 2010;115:3541–3552. doi: 10.1182/blood-2009-09-243535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Davis RE, Ngo VN, Lenz G, et al. Chronic active B-cell-receptor signalling in diffuse large B-cell lymphoma. Nature. 2010;463:88–92. doi: 10.1038/nature08638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ngo VN, Young RM, Schmitz R, et al. Oncogenically active MYD88 mutations in human lymphoma. Nature. 2011;470:115–119. doi: 10.1038/nature09671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jungnickel B, Staratschek-Jox A, Brauninger A, et al. Clonal deleterious mutations in the IkappaBalpha gene in the malignant cells in Hodgkin's lymphoma. J Exp Med. 2000;191:395–402. doi: 10.1084/jem.191.2.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Calado DP, Zhang B, Srinivasan L, et al. Constitutive canonical NF-kappaB activation cooperates with disruption of BLIMP1 in the pathogenesis of activated B cell-like diffuse large cell lymphoma. Cancer Cell. 2010;18:580–589. doi: 10.1016/j.ccr.2010.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pasqualucci L, Trifonov V, Fabbri G, et al. Analysis of the coding genome of diffuse large B-cell lymphoma. Nat Genet. 2011;43:830–837. doi: 10.1038/ng.892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bushell KR, Kim Y, Chan FC, et al. Genetic inactivation of TRAF3 in canine and human B-cell lymphoma. Blood. 2015;125:999–1005. doi: 10.1182/blood-2014-10-602714. [DOI] [PubMed] [Google Scholar]
- 36.Zhang B, Calado DP, Wang Z, et al. A key oncogenic role for alternative NF-kappaB signaling in DLBCL, revealed upon interference with terminal B cell differentiation. Cell Rep. 2015;11:715–726. doi: 10.1016/j.celrep.2015.03.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Steidl C, Telenius A, Shah SP, et al. Genome-wide copy number analysis of Hodgkin Reed-Sternberg cells identifies recurrent imbalances with correlations to treatment outcome. Blood. 2010;116:418–427. doi: 10.1182/blood-2009-12-257345. [DOI] [PubMed] [Google Scholar]
- 38.Otto C, Giefing M, Massow A, et al. Genetic lesions of the TRAF3 and MAP3K14 genes in classical Hodgkin lymphoma. Brit J Hematol. 2012;157:702–708. doi: 10.1111/j.1365-2141.2012.09113.x. [DOI] [PubMed] [Google Scholar]
- 39.Rossi D, Deaglio S, Dominguez-Sola D, et al. Alteration of BIRC3 and multiple other NF- kappaB pathway genes in splenic marginal zone lymphoma. Blood. 2011;118:4930–4934. doi: 10.1182/blood-2011-06-359166. [DOI] [PubMed] [Google Scholar]
- 40*.Rahal R, Frick M, Romero R, et al. Pharmacological and genomic profiling identifies NF- kappaB-targeted treatment strategies for mantle cell lymphoma. Nat Med. 2014;20:87–92. doi: 10.1038/nm.3435. [This study identifies NF-κB-inducing kinase (NIK) as a new therapeutic target for mantle cell lymphoma treatment for the subset of lymphomas that are refractory to BCR pathway inhibitors.] [DOI] [PubMed] [Google Scholar]
- 41.Hideshima T, Chauhan D, Kiziltepe T, et al. Biologic sequelae of I{kappa}B kinase (IKK) inhibition in multiple myeloma: therapeutic implications. Blood. 2009;113:5228–5236. doi: 10.1182/blood-2008-06-161505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rosenwald A, Wright G, Chan WC, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med. 2002;346:1937–1947. doi: 10.1056/NEJMoa012914. [DOI] [PubMed] [Google Scholar]
- 43.Martin-Subero JI, Gesk S, Harder L, et al. Recurrent involvement of the REL and BCL11A loci in classical Hodgkin lymphoma. Blood. 2002;99:1474–1477. doi: 10.1182/blood.v99.4.1474. [DOI] [PubMed] [Google Scholar]
- 44.Barth TF, Martin-Subero JI, Joos S, et al. Gains of 2p involving the REL locus correlate with nuclear c-Rel protein accumulation in neoplastic cells of classical Hodgkin lymphoma. Blood. 2003;101:3681–3686. doi: 10.1182/blood-2002-08-2577. [DOI] [PubMed] [Google Scholar]
- 45.Hewamana S, Alghazal S, Lin TT, et al. The NF-kappaB subunit Rel A is associated with in vitro survival and clinical disease progression in chronic lymphocytic leukemia and represents a promising therapeutic target. Blood. 2008;111:4681–4689. doi: 10.1182/blood-2007-11-125278. [DOI] [PubMed] [Google Scholar]
- 46.Chiorazzi N, Rai KR, Ferrarini M. Chronic lymphocytic leukemia. N Engl J Med. 2005;352:804–815. doi: 10.1056/NEJMra041720. [DOI] [PubMed] [Google Scholar]
- 47.Rossi D, Fangazio M, Rasi S, et al. Disruption of BIRC3 associates with fludarabine chemorefractoriness in TP53 wild-type chronic lymphocytic leukemia. Blood. 2012;119:2854–2862. doi: 10.1182/blood-2011-12-395673. [DOI] [PubMed] [Google Scholar]
- 48.Puente XS, Pinyol M, Quesada V, et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature. 2011;475:101–105. doi: 10.1038/nature10113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wang L, Lawrence MS, Wan Y, et al. SF3B1 and other novel cancer genes in chronic lymphocytic leukemia. N Engl J Med. 2011;365:2497–2506. doi: 10.1056/NEJMoa1109016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50*.Damm F, Mylonas E, Cosson A, et al. Acquired initiating mutations in early hematopoietic cells of CLL patients. Cancer Discovery. 2014;4:1088–1101. doi: 10.1158/2159-8290.CD-14-0104. [This study identifies recurrent mutations of the NF-κB pathway component NFKBIE in CLL.] [DOI] [PubMed] [Google Scholar]
- 51.Shaffer AL, Rosenwald A, Hurt EM, et al. Signatures of the immune response. Immunity. 2001;15:375–385. doi: 10.1016/s1074-7613(01)00194-7. [DOI] [PubMed] [Google Scholar]
- 52.Basso K, Klein U, Niu H, et al. Tracking CD40 signaling during germinal center development. Blood. 2004;104:4088–4096. doi: 10.1182/blood-2003-12-4291. [DOI] [PubMed] [Google Scholar]
- 53.Li Z, Wang X, Yu RY, et al. BCL-6 negatively regulates expression of the NF-kappaB1 p105/p50 subunit. J Immunol. 2005;174:205–214. doi: 10.4049/jimmunol.174.1.205. [DOI] [PubMed] [Google Scholar]
- 54.Köntgen F, Grumont RJ, Strasser A, et al. Mice lacking the c-rel proto-oncogene exhibit defects in lymphocyte proliferation, humoral immunity, and interleukin-2 expression. Genes Dev. 1995;9:1965–1977. doi: 10.1101/gad.9.16.1965. [DOI] [PubMed] [Google Scholar]
- 55.Tumang JR, Owyang A, Andjelic S, et al. c-Rel is essential for B lymphocyte survival and cell cycle progression. Eur J Immunol. 1998;28:4299–4312. doi: 10.1002/(SICI)1521-4141(199812)28:12<4299::AID-IMMU4299>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 56.Carrasco D, Cheng J, Lewin A, et al. Multiple hemopoietic defects and lymphoid hyperplasia in mice lacking the transcriptional activation domain of the c-Rel protein. J Exp Med. 1998;187:973–984. doi: 10.1084/jem.187.7.973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Damdinsuren B, Zhang Y, Khalil A, et al. Single round of antigen receptor signaling programs naive B cells to receive T cell help. Immunity. 2010;32:355–366. doi: 10.1016/j.immuni.2010.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pohl T, Gugasyan R, Grumont RJ, et al. The combined absence of NF-kappa B1 and c- Rel reveals that overlapping roles for these transcription factors in the B cell lineage are restricted to the activation and function of mature cells. Proc Natl Acad Sci USA. 2002;99:4514–4519. doi: 10.1073/pnas.072071599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59*.Heise N, De Silva NS, Silva K, et al. Germinal center B cell maintenance and differentiation are controlled by distinct NF-kappaB transcription factor subunits. J Exp Med. 2014;211:2103–2118. doi: 10.1084/jem.20132613. [This study demonstrates that the canonical NF-κB subunit c-REL is required for the maintenance of the GC B-cell reaction, presumably by activating a metabolic program that promotes cell growth which may have implications for the potential role of c-REL in lymphomagenesis.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wang R, Dillon CP, Shi LZ, et al. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity. 2011;35:871–882. doi: 10.1016/j.immuni.2011.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.van der Windt GJ, Pearce EL. Metabolic switching and fuel choice during T-cell differentiation and memory development. Immunol Rev. 2012;249:27–42. doi: 10.1111/j.1600-065X.2012.01150.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Man K, Miasari M, Shi W, et al. The transcription factor IRF4 is essential for TCR affinity- mediated metabolic programming and clonal expansion of T cells. Nat Immunol. 2013;14:1155–1165. doi: 10.1038/ni.2710. [DOI] [PubMed] [Google Scholar]
- 63.Sinclair LV, Rolf J, Emslie E, et al. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat Immunol. 2013;14:500–508. doi: 10.1038/ni.2556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Grumont RJ, Strasser A, Gerondakis S. B cell growth is controlled by phosphatidylinosotol 3-kinase-dependent induction of Rel/NF-kappaB regulated c-myc transcription. Mol Cell. 2002;10:1283–1294. doi: 10.1016/s1097-2765(02)00779-7. [DOI] [PubMed] [Google Scholar]
- 65.Mauro C, Leow SC, Anso E, et al. NF-kappaB controls energy homeostasis and metabolic adaptation by upregulating mitochondrial respiration. Nat Cell Biol. 2011;13:1272–1279. doi: 10.1038/ncb2324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dominguez-Sola D, Victora GD, Ying CY, et al. The proto-oncogene MYC is required for selection in the germinal center and cyclic reentry. Nat Immunol. 2012;13:1083–1091. doi: 10.1038/ni.2428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Calado DP, Sasaki Y, Godinho SA, et al. The cell-cycle regulator c-Myc is essential for the formation and maintenance of germinal centers. Nat Immunol. 2012;13:1092–1100. doi: 10.1038/ni.2418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Beg AA, Sha WC, Bronson RT, et al. Embryonic lethality and liver degeneration in mice lacking the RelA component of NF-kappa B. Nature. 1995;376:167–170. doi: 10.1038/376167a0. [DOI] [PubMed] [Google Scholar]
- 69.Doi TS, Takahashi T, Taguchi O, et al. NF-kappa B RelA-deficient lymphocytes: normal development of T cells and B cells, impaired production of IgA and IgG1 and reduced proliferative responses. J Exp Med. 1997;185:953–961. doi: 10.1084/jem.185.5.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Gilmore TD. Multiple mutations contribute to the oncogenicity of the retroviral oncoprotein v-Rel. Oncogene. 1999;18:6925–6937. doi: 10.1038/sj.onc.1203222. [DOI] [PubMed] [Google Scholar]
- 71.Caro P, Kishan AU, Norberg E, et al. Metabolic signatures uncover distinct targets in molecular subsets of diffuse large B cell lymphoma. Cancer Cell. 2012;22:547–560. doi: 10.1016/j.ccr.2012.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Houldsworth J, Olshen AB, Cattoretti G, et al. Relationship between REL amplification, REL function, and clinical and biologic features in diffuse large B-cell lymphomas. Blood. 2004;103:1862–1868. doi: 10.1182/blood-2003-04-1359. [DOI] [PubMed] [Google Scholar]
- 73.Ramachandiran S, Adon A, Guo X, et al. Chromosome instability in diffuse large B cell lymphomas is suppressed by activation of the noncanonical NF-kappaB pathway. Int J Cancer. 2015;136:2341–2351. doi: 10.1002/ijc.29301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ranuncolo SM, Pittaluga S, Evbuomwan MO, et al. Hodgkin lymphoma requires stabilized NIK and constitutive RelB expression for survival. Blood. 2012;120:3756–3763. doi: 10.1182/blood-2012-01-405951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhao B, Barrera LA, Ersing I, et al. The NF-kappaB genomic landscape in lymphoblastoid B cells. Cell Rep. 2014;8:1595–1606. doi: 10.1016/j.celrep.2014.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76*.Nogai H, Wenzel SS, Hailfinger S, et al. IkappaB-zeta controls the constitutive NF- kappaB target gene network and survival of ABC DLBCL. Blood. 2013;122:2242–2250. doi: 10.1182/blood-2013-06-508028. [This study shows that IκB-ζ is essential for nuclear NF-κB activity in the ABC-type DLBCL, identifying IκB-ζ as a potential therapeutic target.] [DOI] [PubMed] [Google Scholar]
- 77.Rosebeck S, Rehman AO, Apel IJ, et al. The API2-MALT1 fusion exploits TNFR pathway-associated RIP1 ubiquitination to promote oncogenic NF-kappaB signaling. Oncogene. 2014;33:2520–2530. doi: 10.1038/onc.2013.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Perkins ND. The diverse and complex roles of NF-kappaB subunits in cancer. Nat Rev Cancer. 2012;12:121–132. doi: 10.1038/nrc3204. [DOI] [PubMed] [Google Scholar]
- 79*.Shono Y, Tuckett AZ, Ouk S, et al. A small-molecule c-Rel inhibitor reduces alloactivation of T cells without compromising antitumor activity. Cancer Discovery. 2014;4:578–591. doi: 10.1158/2159-8290.CD-13-0585. [This study provides evidence for the feasibility to pharmacologically target individual NF-κB subunits on the example of the c-REL subunit.] [DOI] [PMC free article] [PubMed] [Google Scholar]