Abstract
To evaluate the association of either propylthiouracil or methimazole treatment for hyperthyroidism during pregnancy with congenital malformations, relevant studies were identified by searching Medline, PubMed, the Cochrane Library and EMBASE.
We intended to include randomized controlled trials, but no such trials were identified. Thus, we included cohort studies and case-control studies in this meta-analysis.
A total of 7 studies were included in the meta-analyses. The results revealed an increased risk of birth defects among the group of pregnant women with hyperthyroidism treated with methimazole compared with the control group (odds ratio 1.76, 95% confidence interval 1.47–2.10) or the non-exposed group (odds ratio 1.71, 95% confidence interval 1.39–2.10). A maternal shift between methimazole and propylthiouracil was associated with an increased odds ratio of birth defects (odds ratio 1.88, 95% confidence interval 1.27–2.77). An equal risk of birth defects was observed between the group of pregnant women with hyperthyroidism treated with propylthiouracil and the non-exposed group (odds ratio 1.18, 95% confidence interval 0.97–1.42). There was only a slight trend towards an increased risk of congenital malformations in infants whose mothers were treated with propylthiouracil compared with in infants whose mothers were healthy controls (odds ratio 1.29, 95% confidence interval 1.07–1.55). The children of women receiving methimazole treatment showed an increased risk of adverse fetal outcomes relative to those of mothers receiving propylthiouracil treatment.
We found that propylthiouracil was a safer choice for treating pregnant women with hyperthyroidism according to the risk of birth defects but that a shift between methimazole and propylthiouracil failed to provide protection against birth defects.
Keywords: Hyperthyroidism, Congenital anomalies, Propylthiouracil, Methimazole, Pregnancy, Meta-analysis
INTRODUCTION
Hyperthyroidism during pregnancy is uncommon, affecting approximately 1 in 500 pregnancies 1. Overt hyperthyroidism has well-documented adverse impacts on pregnancy outcomes, such as preterm birth, congenital anomalies and pre-eclampsia 2–4. Therefore, despite its rarity, proper management of hyperthyroidism during pregnancy is of utmost importance. Medical therapy is preferred by most authorities because radioiodine is contra-indicated and because thyroidectomy requires pre-treatment with antithyroid drugs and may be complicated by surgical adverse effects 5. The available antithyroid drugs (ATDs) are propylthiouracil (PTU) and methimazole (MMI)/carbimazole (CZ). These drugs are equivalent in terms of their efficacy for the treatment of clinical hyperthyroidism 6. Concerning hyperthyroidism during pregnancy, some reports suggest an association between a specific congenital malformation (MMI embryopathy) and prenatal exposure to MMI 7. The specific malformation pattern related to prenatal MMI exposure consists of choanal and esophageal atresia, scalp defects, minor facial anomalies and psychomotor delay 8. The reported facial anomalies typically include upward slanted palpebral fissures, arched flared eyebrows and a small nose with a broad bridge 9. A recent study conducted by Anderson et al. demonstrated that ventricular septal defect (VSD) is another component of MMI embryopathy 10. Compared with non-exposure, exposure of children to MMI/CMZ during early pregnancy (up to and including gestational week 10) is associated with an increased risk of VSD. PTU may be safer than MMI, and PTU should be considered as the first-line agent for the treatment of hyperthyroidism during pregnancy 11. In contrast, other studies have shown an equal risk of birth defects among pregnant women with hyperthyroidism treated with PTU or with MMI 12, and several major birth defects have been observed in the offspring of women treated with PTU 13. Unilateral kidney agenesis and malformations of the face and neck region have been associated with in utero exposure to PTU. In a case-control study conducted by Clementi et al., PTU exposure was significantly associated with an increased risk of situs inversus, with or without dextrocardia 6. Recent animal studies have demonstrated that PTU is teratogenic during the late blastula, gastrulation and neurulation stages. PTU can alter ciliary-driven flow and can disrupt the normal genetic program involved in left-right axis determination 14. Thus, there is no consensus regarding the best method of therapy for hyperthyroidism during pregnancy. In this study, we evaluated the association of either PTU or MMI treatment for hyperthyroidism during pregnancy with congenital malformations and conducted a systematic review.
METHODS
Literature search and study selection
Relevant studies published until July 2014 were identified by searching Medline, PubMed, the Cochrane Library and EMBASE. The applied search criteria were related to thyroid function and pregnancy outcomes. Specifically, the following search items were used: thyroid*, hyperthyr*, Graves' disease, PTU, propylthiouracil, MMI, methimazole, congenital malformation, birth defect and congenital anomalies. There were no language limitations for the initial search. Randomized controlled trials (RCTs), cohort studies and case-control studies were included. Data on the effects of combinational therapy were excluded.
Data extraction
The titles and the abstracts of the articles were independently screened by two reviewers (Li X and Zhou L). The articles to be included for full-text screening were compared during a consensus meeting. In cases of disagreement, a third reviewer (Ma JL) was consulted regarding the decision about inclusion or exclusion for full-text evaluation. Articles that did not contribute to the resolution of our research questions after full-text evaluation were excluded. Hyperthyroidism was defined as a low TSH level accompanied by a high free T4 level. After a consensus was achieved, the remaining articles were included for critical appraisal and were independently assessed by two reviewers. Articles (RCT studies) were judged on scientific quality according to the CONSORT and STROBE statements 15,16.
Data analysis and statistical methods
The significance of each pooled odds ratio (OR) was determined using a Z-test, in which p<0.05 was considered to indicate a significant difference. The χ2-based Q statistical test was employed for the assessment of between-study heterogeneity, which was considered to be significant at p<0.1. In the analyses, if heterogeneity was low, then we used a fixed effects model; otherwise, we applied a random effects model. Review Manager 5.3 software (available from Cochrane) was used to perform the meta-analyses.
RESULTS
Figure 1 shows the selection process after the search. Of the 7 articles included in this systematic review, 6 reported on MMI use, 6 reported on PTU use and 2 reported on shifts between MMI and PTU. The patients in the included studies were pregnant women with hyperthyroidism who required treatment with antithyroid medication to maintain their thyroid hormone levels within the normal range. All controls were pregnant women who either exhibited euthyroidism or presented with hyperthyroidism that was observed late in pregnancy; the latter group of patients delivered before therapy and did not require any antithyroid medication therapy 12. The characteristics of the included articles are reported in Table 1.
Figure 1.
Flow chart of the literature search and article selection.
Table 1.
-Characteristics of the 7 studies included in the review.
| Study | Year | Study type | Participants | Controls | Treatment | Birth defects |
| Andersen et al. | 2013 | Cohort | 1,661 women treated with PTU or MMI159 women who shifted between MMI and PTU treatment | 811,730 women without hyperthyroidism3,543 women not receiving any ATD1,066 women without hyperthyroidism | Treated with antithyroid medication in early pregnancy | Urinary system malformation, malformations in the face and neck region, choanal atresia, esophageal atresia |
| Chen et al. | 2011 | Case-control | 703 women treated with PTU or MMI | 14,150 women without hyperthyroidism2,127 women not receiving any ATD | Treated with PTU or MMI for at least 30 days | Cleft lip and palate, limb defects, heart defects, Down syndrome, hypospadias |
| Korelitz et al. | 2012 | Case-control | 1,023 women treated with PTU or MMI126 women who shifted between MMI and PTU treatment | 634,858 women without hyperthyroidism5,932 women not receiving any ATD | Prescription (PTU or MMI) filled within the last 6 months of pregnancy | Congenital anomalies of the eye, complex heart anomalies, atrial ventricular septal defects, anomalies of the respiratory system, anomalies of the congenital organs |
| Rosenfeld et al. | 2009 | Cohort | 80 women treated with PTU | 1,066 women without hyperthyroidism | Treated with PTU between 4 and 13 weeks of gestation | Developmental dysplasia of the hip |
| Yoshihara et al. | 2012 | Case-control | 2,630 women treated with PTU or MMI | 1,906 women not receiving any ATD | Treated with PTU or MMI during the first trimester | Aplasia cutis congenital, omphalocele, symptomatic omphalomesenteric duct anomaly |
| Wing et al. | 1994 | Case-control | 135 women treated with PTU or MMI | 43 women not receiving any ATD99 women without hyperthyroidism | Treated with PTU or MMI | Severe pulmonary stenosis, ventricular septal defect, patent ductus arteriosus |
| Momotani et al. | 1984 | Case-control | 117 women treated with MMI | 350 women without hyperthyroidism | Treated with MMI during the first trimester | Malformation of the ear lobe, omphalocele, imperforate anus, anencephaly, harelip, polydactyly |
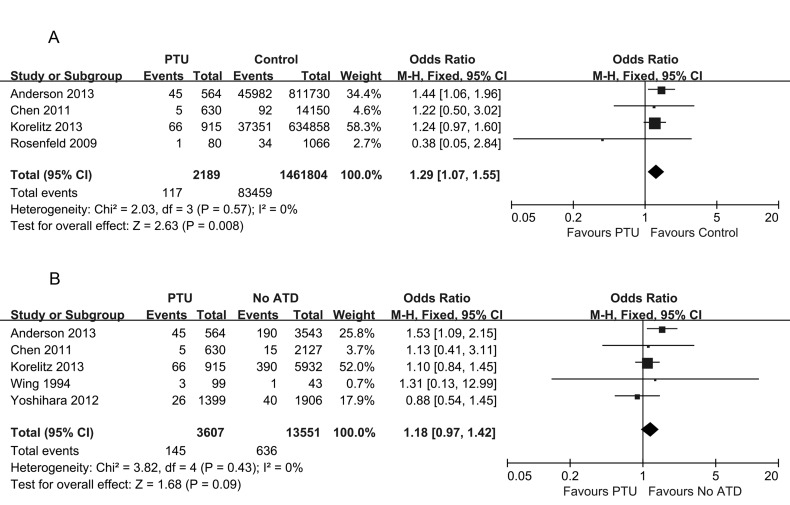
The effect of PTU use on congenital malformations
A total of 7 studies reported on congenital malformations after exposure to PTU. Compared with healthy pregnant women, only one study showed an increased risk of congenital malformations in pregnant women treated with PTU 17. Three other studies did not detect any difference in the congenital malformation rate 13,18,19. A meta-analysis of these four studies concerning the association of exposure to PTU with congenital malformations resulted in a pooled OR of 1.29, with a 95% CI of 1.07–1.55, indicating a mild difference (Figure 2A). We further compared the risk of congenital malformations in pregnant women exposed to PTU with that in women who were not exposed to any ATD during pregnancy. The pregnant women in the non-ATD-exposed group either exhibited euthyroidism throughout pregnancy, thus requiring no medication, or presented with hyperthyroidism that was observed late in pregnancy. The latter group of patients delivered before therapy. There was no statistically significant association between exposure to PTU and the risk of birth defects (pooled OR 1.18, 95% CI 0.97–1.42) (Figure 2B).
Figure 2.
Forest plot of the odds ratios and 95% confidence intervals of the pooled studies. (A) PTU-treated pregnant hyperthyroid women compared with healthy pregnant women according to the risk of congenital anomalies. (B) PTU-treated pregnant hyperthyroid women compared with pregnant hyperthyroid women not receiving any ATD treatment according to the risk of congenital anomalies.
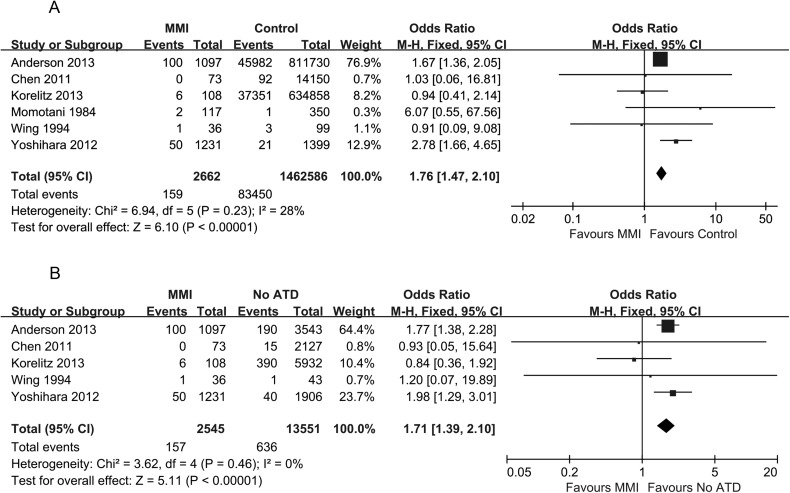
The effect of MMI use on congenital malformations
As shown in Figure 3A, six studies reported on congenital malformations after exposure to MMI. Three studies showed an increased risk of congenital malformations in the group of pregnant women treated with MMI compared with the control group 11,17,20. Three other studies did not detect any difference in the congenital malformation rate 12,13,18. A meta-analysis of these six studies concerning the association of exposure to MMI with congenital malformations resulted in a pooled OR of 1.76, with a 95% CI of 1.47–2.10, indicating a significant difference. Even compared with women with hyperthyroidism who were not exposed to any ATD during pregnancy, there was a significantly increased risk of birth defects in women exposed to MMI (pooled OR 1.71, 95% CI 1.39–2.10) (Figure 3B).
Figure 3.
Forest plot of the odds ratios and 95% confidence intervals of the pooled studies. (A) MMI-treated pregnant hyperthyroid women compared with healthy pregnant women according to the risk of congenital anomalies. (B) MMI-treated pregnant hyperthyroid women compared with pregnant hyperthyroid women not receiving any ATD treatment according to the risk of congenital anomalies.
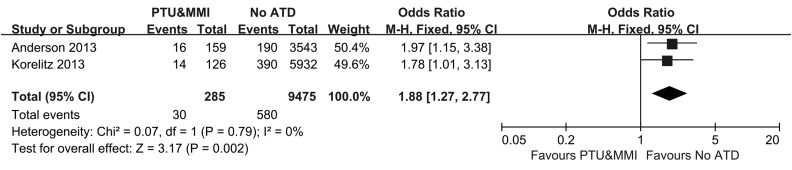
The effect of shifts between MMI and PTU use on congenital malformations
As shown in Figure 4, two studies reported on congenital malformations after shifting between MMI and PTU. Both studies showed an increased risk of congenital malformations in pregnant women whose treatment shifted between MMI and PTU compared with the controls 13,17. A meta-analysis of these two studies resulted in a pooled OR of 1.88, with a 95% CI of 1.27–2.77, indicating a significant difference.
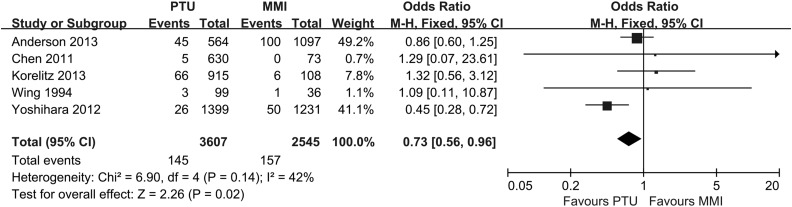
Figure 4.
Forest plot of the odds ratios and 95% confidence intervals of the pooled studies comparing pregnant hyperthyroid women who shifted between PTU and MMI with pregnant hyperthyroid women who did not receive any ATD treatment according to the risk of congenital anomalies.
The effect of antithyroid drugs on congenital malformations
As shown in Figure 5, five studies reported on congenital malformations after exposure to PTU or MMI. A meta-analysis of these five studies resulted in a pooled OR of 0.73, with a 95% CI of 0.56–0.96, indicating that PTU was a safer choice with respect to the risk of birth defects among pregnant women with hyperthyroidism.
Figure 5.
Forest plot of the odds ratios and 95% confidence intervals of the pooled studies comparing PTU-treated pregnant hyperthyroid women with MMI-treated pregnant hyperthyroid women according to the risk of congenital anomalies.
DISCUSSION
Several studies have linked hyperthyroidism during pregnancy with an increased risk of congenital anomalies. However, the evidence regarding the impact of ATDs such as PTU and MMI on pregnancy outcomes remains controversial. The preferred ATD during pregnancy in the USA is PTU, whereas in Europe, it is MMI 21,22.
In this study, we compared the risk of congenital anomalies among pregnant women with hyperthyroidism classified into three groups: those receiving PTU treatment, those receiving MMI treatment, and those receiving either PTU or MMI first and then shifting to MMI or PTU, respectively. In addition, we enrolled two control groups in this study: completely healthy pregnant women and women with hyperthyroidism who were not exposed to any ATD. Severe hyperthyroidism during pregnancy typically causes various complications for both the mother and the fetus 23. Thus, the pregnant women who were not exposed to any ATD either exhibited euthyroidism throughout pregnancy or exhibited hyperthyroidism that was observed late in pregnancy. The latter group of patients delivered before therapy.
Compared with hyperthyroid women not receiving an ATD or healthy pregnant controls, a significant increase in the rate of major anomalies was observed in those receiving MMI treatment. There was only a slight trend towards an increased risk of congenital malformations in infants whose mothers were treated with PTU compared with those whose mothers were healthy controls. The risk of birth defects for hyperthyroid women treated with PTU and those not receiving any ATD was similar. As the non-exposed group of women was largely healthy, the similar rates between the two aforementioned groups indicated that PTU was the preferred option according to the risk of congenital anomalies. However, one limitation to these studies was the confounding between the effect of hyperthyroidism itself and the effect of the drug in producing adverse fetal outcomes. It is reasonable to expect that the more severe examples of hyperthyroidism would be observed in the very women for whom drug treatment was prescribed, particularly at higher doses. Thus, the adverse fetal outcomes could have resulted from either the underlying severe hyperthyroidism or the treatment. To control for this confounding factor, we considered the eligible subjects for this study to be pregnant women with hyperthyroidism who received ATD treatment. Then, we compared PTU use with MMI use in relation to the overall rate of anomalies. Our data also supported the use of PTU. Taken together, these results indicated that PTU is a safer choice for the treatment of pregnant women with hyperthyroidism according to the risk of birth defects. This conclusion is in accordance with an American Thyroid Association statement supporting the use of PTU as the first-line agent for the management of hyperthyroidism during pregnancy 24.
A recent systematic review conducted by Hackmon et al. also described the safety of MMI and PTU during pregnancy 25. These authors concluded that MMI is a teratogen, which is consistent with our findings. In most cases associated with MMI embryopathy included in the case reports reviewed by Hackmon et al., exposure to MMI indeed occurred in the first trimester during the critical embryogenic period. In the present study, among the 6 studies examining the relationship of MMI exposure to the risk of birth defects, a total of 3 studies (Anderson et al., Yoshihara et al., and Momotani et al.) reported on the risk of congenital anomalies following intrauterine exposure in the first trimester. The findings of these studies showed that MMI treatment significantly increased the risk of congenital malformations. The remaining studies (Chen et al., Korelitz et al., and Wing et al), which did not describe whether ATD treatment was administered in the first trimester, failed to confirm a causal relationship between MMI and congenital malformations. This result was probably due to exposure after the period of organogenesis.
Shifts between PTU and MMI are not common in the treatment of patients with hyperthyroidism. Two of the included articles did not explain the reason that the doctors preferred to change the treatment, although this preference was likely related to potential concerns about the teratogenic effects of MMI in the first trimester and the hepatotoxicity of PTU during pregnancy 26,27. Thus, it was suggested that in pregnant women, PTU use should be restricted to the first trimester, and MMI should then be used during the remainder of pregnancy to minimize potential risks to the fetus and the mother. Our study illustrated that shifts between MMI and PTU may confer little protection against birth defects. This conclusion is inconsistent with the previous study conducted by Hackmon et al., who demonstrated that PTU should be administered during the first trimester, followed by a switch to MMI for the remainder of the pregnancy. One possible explanation for this disagreement might be as follows: due to severe PTU-induced hepatotoxicity, women with hyperthyroidism might be treated with MMI, and the shift from MMI to PTU may only occur when they find out that they are pregnant. Thus, there is a high probability that exposure to MMI occurs during the critical embryogenic period (4 to 10 weeks) because under most circumstances, women are actually pregnant for more than one month before they find out that they are pregnant. For instance, one study included in our meta-analysis reported that the mean time from the beginning of pregnancy to the shift to PTU treatment was 44 days. In addition, considering shifts between PTU and MMI, there were two types of drug prescription patterns observed in the included studies: from PTU to MMI and from MMI to PTU. Therefore, we should examine whether the prevalence of birth defects differs according to type of ATD prescription pattern.
However, there is no evidence from current trials to guide the choice between PTU followed by a switch to MMI and vice versa. Thus, it is certain that changes in ATD prescription patterns are not necessary during pregnancy. To avoid shifts in ATD use, it is recommended that women of child-bearing age choose PTU treatment before pregnancy to minimize the potential risk of congenital malformations.
Our research provides important population-based estimates of medication use that are relevant to the care of pregnant women with hyperthyroidism. However, a definitive answer to this question remains unclear because there are no available RCTs comparing antithyroid treatments in pregnant women with hyperthyroidism. Additional human epidemiology studies are needed, and new evidence-based recommendations should be developed for the treatment of women with hyperthyroidism during pregnancy.
Despite the lack of RCTs, we believe that our study provides potentially useful data to physicians prescribing ATDs to pregnant women with hyperthyroidism. The limited available evidence shows that PTU is a safer choice for treating pregnant women with hyperthyroidism according to the risk of birth defects but that shifting between MMI and PTU fails to provide protection against birth defects.
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Mestman JH. Hyperthyroidism in pregnancy. Curr Opin Endocrinol Diabetes Obes. 2012;19((5)):394–401. doi: 10.1097/MED.0b013e328357f3d5. [DOI] [PubMed] [Google Scholar]
- 2.Inoue M, Arata N, Koren G, Ito S. Hyperthyroidism during pregnancy. Can Fam Physician. 2009;55((7)):701–3. [PMC free article] [PubMed] [Google Scholar]
- 3.Vissenberg R, van den Boogaard E, van Wely M, van der Post JA, Fliers E, Bisschop PH, et al. Treatment of thyroid disorders before conception and in early pregnancy: a systematic review. Hum Reprod Update. 2012;18((4)):360–73. doi: 10.1093/humupd/dms007. [DOI] [PubMed] [Google Scholar]
- 4.van den Boogaard E, Vissenberg R, Land JA, van Wely M, van der Post JA, Goddijn M, et al. Significance of (sub)clinical thyroid dysfunction and thyroid autoimmunity before conception and in early pregnancy: a systematic review. Hum Reprod Update. 2011;17((5)):605–19. doi: 10.1093/humupd/dmr024. [DOI] [PubMed] [Google Scholar]
- 5.Azizi F, Amouzegar A. Management of hyperthyroidism during pregnancy and lactation. Eur J Endocrinol. 2011;164((6)):871–6. doi: 10.1530/EJE-10-1030. [DOI] [PubMed] [Google Scholar]
- 6.Clementi M, Di Gianantonio E, Cassina M, Leoncini E, Botto LD, Mastroiacovo P. Treatment of hyperthyroidism in pregnancy and birth defects. J Clin Endocrinol Metab. 2010;95((11)):E337–41. doi: 10.1210/jc.2010-0652. [DOI] [PubMed] [Google Scholar]
- 7.Di Gianantonio E, Schaefer C, Mastroiacovo PP, Cournot MP, Benedicenti F, Reuvers M, et al. Adverse effects of prenatal methimazole exposure. Teratology. 2001;64((5)):262–6. doi: 10.1002/(ISSN)1096-9926. [DOI] [PubMed] [Google Scholar]
- 8.Clementi M, Di Gianantonio E, Pelo E, Mammi I, Basile RT, Tenconi R. Methimazole embryopathy: delineation of the phenotype. Am J Med Genet. 1999;83((1)):43–6. doi: 10.1002/(ISSN)1096-8628. [DOI] [PubMed] [Google Scholar]
- 9.Rivkees SA. Pediatric Graves' disease: controversies in management. Horm Res Paediatr. 2010;74((5)):305–11. doi: 10.1159/000320028. [DOI] [PubMed] [Google Scholar]
- 10.Bowman P, Osborne NJ, Sturley R, Vaidya B. Carbimazole embryopathy: implications for the choice of antithyroid drugs in pregnancy. QJM. 2012;105((2)):189–93. doi: 10.1093/qjmed/hcq248. [DOI] [PubMed] [Google Scholar]
- 11.Yoshihara A, Noh J, Yamaguchi T, Ohye H, Sato S, Sekiya K, et al. Treatment of graves' disease with antithyroid drugs in the first trimester of pregnancy and the prevalence of congenital malformation. J Clin Endocrinol Metab. 2012;97((7)):2396–403. doi: 10.1210/jc.2011-2860. [DOI] [PubMed] [Google Scholar]
- 12.Wing DA, Millar LK, Koonings PP, Montoro MN, Mestman JH. A comparison of propylthiouracil versus methimazole in the treatment of hyperthyroidism in pregnancy. Am J Obstet Gynecol. 1994;170((1 Pt 1)):90–5. doi: 10.1089/thy.2012.0488. [DOI] [PubMed] [Google Scholar]
- 13.Korelitz JJ, McNally DL, Masters MN, Li SX, Xu Y, Rivkees SA. Prevalence of thyrotoxicosis, antithyroid medication use, and complications among pregnant women in the United States. Thyroid. 2013;23((6)):758–65. doi: 10.1096/fj.12-218073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.van Veenendaal NR, Ulmer B, Boskovski MT, Fang X, Khokha MK, Wendler CC, et al. Embryonic exposure to propylthiouracil disrupts left-right patterning in Xenopus embryos. FASEB J. 2013;27((2)):684–91. doi: 10.1096/fj.12-218073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370((9596)):1453–7. doi: 10.1016/S0140-6736(07)61602-X. [DOI] [PubMed] [Google Scholar]
- 16.Schulz KF, Altman DG, Moher D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. J Pharmacol Pharmacother. 2010;1((2)):100–7. doi: 10.4103/0976-500X.72352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Andersen SL, Olsen J, Wu CS, Laurberg P. Birth defects after early pregnancy use of antithyroid drugs: a Danish nationwide study. J Clin Endocrinol Metab. 2013;98((11)):4373–81. doi: 10.1210/jc.2013-2831. [DOI] [PubMed] [Google Scholar]
- 18.Chen CH, Xirasagar S, Lin CC, Wang LH, Kou YR, Lin HC. Risk of adverse perinatal outcomes with antithyroid treatment during pregnancy: a nationwide population-based study. BJOG. 2011;118((11)):1365–73. doi: 10.1111/bjo.2011.118.issue-11. [DOI] [PubMed] [Google Scholar]
- 19.Rosenfeld H, Ornoy A, Shechtman S, Diav-Citrin O. Pregnancy outcome, thyroid dysfunction and fetal goitre after in utero exposure to propylthiouracil: a controlled cohort study. Br J Clin Pharmacol. 2009;68((4)):609–17. doi: 10.1111/bcp.2009.68.issue-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Momotani N, Ito K, Hamada N, Ban Y, Nishikawa Y, Mimura T. Maternal hyperthyroidism and congenital malformation in the offspring. Clin Endocrinol (Oxf) 1984;20((6)):695–700. doi: 10.1111/j.1365-2265.1984.tb00119.x. [DOI] [PubMed] [Google Scholar]
- 21.Clark SM, Saade GR, Snodgrass WR, Hankins GD. Pharmacokinetics and pharmacotherapy of thionamides in pregnancy. Ther Drug Monit. 2006;28((4)):477–83. doi: 10.1097/00007691-200608000-00001. [DOI] [PubMed] [Google Scholar]
- 22.Cooper DS. Antithyroid drugs. N Engl J Med. 2005;352((9)):905–17. doi: 10.1056/NEJMra042972. [DOI] [PubMed] [Google Scholar]
- 23.Cooper DS, Laurberg P. Hyperthyroidism in pregnancy. Lancet Diabetes Endocrinol. 2013;1((3)):238–49. doi: 10.1016/S2213-8587(13)70086-X. [DOI] [PubMed] [Google Scholar]
- 24.Emiliano AB, Governale L, Parks M, Cooper DS. Shifts in propylthiouracil and methimazole prescribing practices: antithyroid drug use in the United States from 1991 to 2008. J Clin Endocrinol Metab. 2010;95((5)):2227–33. doi: 10.1210/jc.2009-2752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hackmon R, Blichowski M, Koren G. The safety of methimazole and propylthiouracil in pregnancy: a systematic review. J Obstet Gynaecol Can. 2012;34((11)):1077–86. doi: 10.1016/S1701-2163(16)35438-X. [DOI] [PubMed] [Google Scholar]
- 26.Clementi M, Gianantonio E. Therapeutic drug monitoring of antithyroid drugs in pregnancy: the knowledge gaps. Ther Drug Monit. 2006;28((4)):576. doi: 10.1097/00007691-200608000-00014. author reply 576. [DOI] [PubMed] [Google Scholar]
- 27.Diav-Citrin O, Ornoy A. Teratogen update: antithyroid drugs-methimazole, carbimazole, and propylthiouracil. Teratology. 2002;65((1)):38–44. doi: 10.1002/(ISSN)1096-9926. [DOI] [PubMed] [Google Scholar]