Abstract
The arylesterase (ARE) activity linked to the paraoxonase-1 (PON1) gene is known to protect lipoproteins from oxidation and provide defense against metabolic syndrome (MetS) and cardiovascular diseases. The epigenetic regulation of enzymatic activities is gaining importance nowadays. This research aimed to assess the potential relationships between the ARE activity with the methylation levels of the PON1 gene transcriptional regulatory region, anthropometrics, biochemical markers and antioxidant dietary components. Forty-seven subjects (47 ± 10 y.o; BMI 36.2 ± 3.8 kg/m2; 46.8 % female) with MetS features, who followed a six-month energy-restricted dietary weight-loss intervention, were included in this study (www.clinicaltrials.gov; NCT01087086). Anthropometric, biochemical, enzymatic and dietary data were assessed using validated procedures. PON1 transcriptional regulatory region methylation was analyzed by a microarray technical approach. Volunteers reduced ARE activity in parallel with body weight (p = 0.005), BMI (p = 0.006), total fat mass (p = 0.020), diastolic blood pressure (p = 0.018), mean blood pressure (p = 0.022) and triglycerides (p = 0.014). Methylation levels of some CpG sites of the PON1 gene correlated negatively with ARE activity (p < 0.05). Interestingly, dietary vitamin C (p = 0.001), tocopherols (p = 0.009) and lycopene (p = 0.038) were positively associated with ARE activity and showed an inverse correlation (p = 0.004, p = 0.029 and p = 0.021, respectively) with the methylation of some selected CpG sites of the PON1 gene. In conclusion, ARE activity decreased in parallel with MetS-related markers associated to the energy restriction, while dietary antioxidants might enhance the ARE activity by lowering the PON1 gene methylation in patients with MetS features.
Keywords: DNA methylation, ARE, PON1 gene, obesity, metabolic syndrome, energy restriction, antioxidants
Introduction
Paraoxonase-1 (PON1) is a calcium-dependent glycoprotein hepatically synthesized that belongs to the paraoxonase family (Precourt et al., 2011[35]). This enzyme is related to high density lipoprotein-cholesterol (HDL-c) and has been described to protect lipoproteins, particularly low density lipoprotein-cholesterol (LDL-c), from oxidation (Kim et al., 2013[18]; Kumar et al., 2013[22]). Thus, some of the antiatherogenic properties of HDL-c are attributed to PON1 functions (Cohen et al., 2012[9]). The precise mechanisms by which the PON1 acts remains unclear, but several in vitro activities have been attributed to this enzyme (Nus et al., 2008[32]): paraoxonase (hydrolysis of organophosphates), lactonase (hydrolysis of lactones), and arylesterase or ARE (hydrolysis of aromatic carboxylic acid esters).
Obesity and metabolic syndrome (MetS) features are main risk factors for the development of atherosclerosis and cardiovascular diseases (CVD) onset (Garg et al., 2014[15]). Due to the increasing prevalence of obese people, these accompanying diseases are considered a major concern for public health authorities and many scientific efforts are being carried out to detect, treat and prevent them. Hypocaloric diets are one of the most common treatments employed to combat MetS and related diseases (Straznicky et al., 2010[39]). However, more investigation is needed to understand the PON1 activity role as part of these nutritional strategies. In this context, some research have been performed focusing on the relationship between the PON1 activity levels and the MetS/ obesity states (Ferretti et al., 2012[12]; Koncsos et al., 2011[19]; Kota et al., 2013[20]; Tabur et al., 2010[40]) while other authors have investigated the influence of some specific dietary factors on the activity levels of this enzyme (Canales et al., 2011[5]; Jarvik et al., 2002[16]; Vazquez-Velasco et al., 2011[44]). But the studies carried out are scarce and reported controversial results.
Epigenetics is defined as heritable changes in gene expression that cannot be explained by changes in DNA sequence (Christensen et al., 2011[7]). Among the different possible epigenetic modifications, DNA methylation is probably the most widely studied (Milagro et al., 2011[28]). In this context, dietary factors, specially antioxidants, have become agents of strong interest in the field of epigenetics due to their prominent role as potent modulators of epigenome-regulated gene expression through regulation of DNA methylation (Bartels, 2007[4]; Malireddy et al., 2012[24]; Milagro et al., 2009[29]).
Thus, within this scenario, the current study aimed to assess the potential relationships between the PON1 ARE activity and anthropometric and biochemical markers, dietary antioxidants intake, and the cytosine methylation levels of PON1 gene transcriptional regulatory region, in volunteers with MetS symptoms after following an energy-restricted dietary program.
Materials and methods
Subjects and study protocol
The current analysis was an ancillary study conducted within the RESMENA (Metabolic Syndrome Reduction in Navarra) project, a randomized controlled trial (Zulet et al., 2011[48]) where a subsample of 47 obese adults (47 ± 10 y.o; BMI 36.2 ± 3.8 kg/m2; 46.8 % female) who presented MetS features was selected. The study lasted a total of 6 months divided in two sequential stages: an initial 8-week nutritional-learning intervention period, during which nutritional assessment was carried out for the participants every 15 days (Lopez-Legarrea et al., 2013[23]) and a 4-month self-control period, during which the participants followed the previously acquired dietary habits (de la Iglesia et al., 2013[11]).
The study was approved by the Ethics Committee of the University of Navarra (065/2009) and appropriately registered at www.clinicaltrials.gov (NCT01087086). Consequently, all the participants gave written informed consent for participation in agreement with the Declaration of Helsinki. More details about the procedures and protocols have been previously reported (Zulet et al., 2011[48]).
Anthropometric, body composition, blood pressure and dietary intake assessment
Anthropometric measurements were conducted in fasting conditions according to previously described procedures (Zulet et al., 2011[48]). Body mass index (BMI) was calculated as the body weight divided by height squared (kg/m2). Body composition analyses were carried out by Dual Energy X-ray Absorptiometry (DXA) following validated protocols as reported elsewhere (Zulet et al., 2011[48]). Systolic (SBP) and diastolic (DBP) blood pressures were measured following standardized World Health Organization criteria (Whitworth et al., 2004[45]). Mean blood pressure (MBP) was calculated as: [(DPB x 2) + SBP]/3 as advised elsewhere (Shapiro et al., 2010[37]).
Information about dietary intake was collected using a 48 h weighed food record and analysed using the DIAL (Alce Ingeniería) software (http://www.alceingenieria. net/nutricion) as previously reported (Perez-Cornago et al., 2013[33]).
Biochemical assessments
Venous blood samples were drawn after a 12 h overnight fast by venipuncture. The EDTA–plasma and serum samples as well as WBC were separated from whole blood by centrifugation at 3,500 rpm, 5 °C, 15 min (Model 5804R, Eppendorf, Germany), and were frozen immediately at -80 °C until assay (WBC in buffy-coat).
Plasma concentrations of triglycerides (TG), total cholesterol (TC), HDL-c (Wako Chemicals, GmbH, Nuiss, Germany) and glucose (Horiba ABX Diagnostics, Montpellier, France) were measured by specific colorimetric assays, using an automated analyzer system Pentra C-200 (HORIBA ABX, Madrid, Spain). LDL-c levels were calculated using the Friedewald formula: LDL-c = TC - HDL-c – TG/5 (Friedewald et al., 1972[13]). Apolipoprotein B (Apo B) was measured with a specific kit (Tina-quant Apolipoprotein B ver.2, Mannheim, Germany) using a Model 904 Modular Roche/Hitachi autoanalyser (Roche Diagnostics, Tokio, Japan).
Plasma concentrations of ARE activity were measured with simulated body fluid (SBF) as buffer and phenylacetate as substrate at pH 7.34–7.4 and 37 °C, as published elsewhere (Nus et al., 2006[31]). Reaction rates of ARE were followed at 270 nm in thermostatically controlled 10-mm Lightpath quartz cuvettes using a Shimadzu UV-2401PC spectrophotometer (Tokio, Japan). The final reaction volume in the cuvettes was 2.0 mL, and the total time was 3 min. One unit of ARE activity was defined as the mmol phenol formed from phenyl acetate per min.
DNA isolation and DNA methylation study
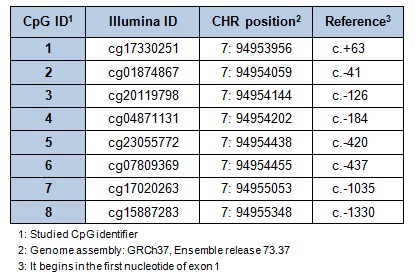
Genomic DNA from WBC was extracted using the Master Pure kit (Epicenter, Madison, WI, USA), whose quality was assessed with PicoGreen dsDNA Quantitation Reagent (Invitrogen, Carlsbad, CA, USA). A total of 500 ng of DNA was modified by using EZ-96 DNA Methylation Kit (Zymo Research Corporation, USA) according to the manufacturer's instructions, converting thus cytosine into uracil. Array-based specific DNA methylation analysis was performed with the Infinium Human Methylation 450K bead chip technology (Illumina, USA). Bisulfite-treated genomic DNA was whole-genome amplified, hybridized to HumanMethylation450 BeadChips (Illumina, USA) and scanned using the Illumina iScanSQ platform (Mansego et al., 2013[25]). The intensity of the images was extracted with the GenomeStudio Methylation Software Module (v 1.9.0, Illumina, USA). Eight Cytosine-phosphate-guanine (CpG) sites of the PON1 gene that codes for the PON1 enzyme were selected. CpG sites located in the transcriptional regulatory region (promoter, 5’-untranslated region and exon 1) were included (Figure 1(Fig. 1)). Reference names and characteristics of the selected CpG sites are shown in Table 1(Tab. 1).
Figure 1. Genomic localization and nucleotide sequence of 8 CpG sites covered by the Illumina probe for the study of DNA methylation levels of PON1 promoter (from - 1330 to +104 pb). Transcription Start Site (TSS).
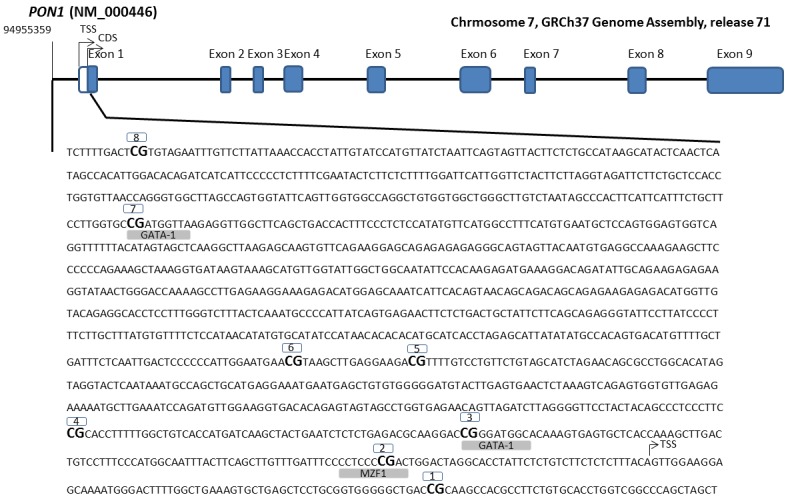
Table 1. Information of the selected CpG sites for each PON1 gene.
Statistical analyses
Results are shown as mean value ± standard deviation. Variable distribution was determined by the Shapiro-Wilk test and no normal variables (glucose, HDL-c, LDL-c, TG, ARE and ARE/HDL-c) were logarithmically transformed for statistical purposes. Differences between the beginning and the end of the complete study were analyzed by paired Student t-test. Pearson correlations adjusted for age and sex were fitted to evaluate the potential associations of PON1 ARE activity and anthropometric, body composition, blood pressure and biochemical variables, and also to assess the relationships between PON1 transcriptional regulatory region methylation and ARE activity and specific dietary factors. Pearson correlations adjusted for sex and age were also used to study the association between the ARE activity and the dietary factors intake at the end of the study. Moreover, multiple testing correction (Benjamini–Hochberg) analyses were performed when appropriate. The SPSS 19.0 software (SPSS Inc., Chicago, IL, USA) for Windows XP (Microsoft, USA) was used for statistical analyses. Globally, p < 0.050 was considered as statistically significant.
Results
Information about selected anthropometric and biochemical measurements was recorded at baseline and at day 180 (Table 2(Tab. 2)). After the 6-month-long trial, participants presented significantly lower (p < 0.001) values concerning anthropometric and body composition variables, such as body weight, BMI, waist circumference, WHR, total fat mass and android fat mass. Volunteers also significantly reduced the SBP, DBP and MBP values as well as the plasma glucose (p < 0.001), Apo B (p < 0.001), TG (p = 0.001). On the other hand, LDL-c (p < 0.001) and HDL-c (p = 0.011) values were significantly higher at the end of the study.
Table 2. Changes in anthropometric, body composition, blood pressure, biochemical parameters and arylesterase (ARE) activity after 6 month-study (N = 47).
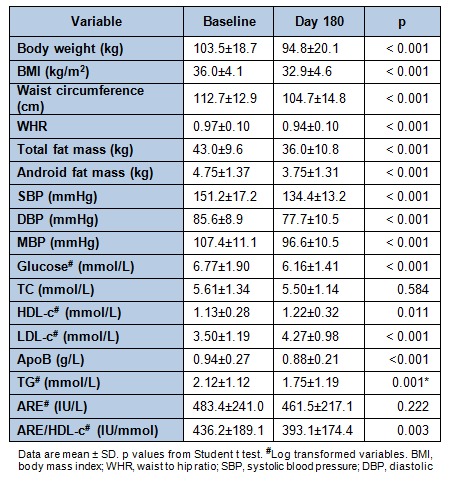
Plasma TC and ARE activity tended to be reduced, although did not reach significance, while the ARE/HDL-c ratio was significantly decreased (p = 0.003).
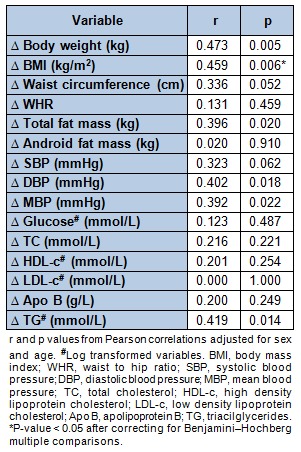
Pearson correlation analyses adjusted for sex and age were performed to assess the possible associations between the PON1 ARE activity and the anthropometric, body composition, blood pressure and biochemical variables. The PON1 ARE activity levels significantly correlated with TC (r = 0.362, p = 0.016), HDL-c (r = 0.346, p = 0.021) and Apo B (r = 0.446, p = 0.002) concentrations, at baseline. Moreover, significant positive associations were found between changes in ARE activity and variation of body weight, BMI, total fat mass, DBP, MBP and plasma TG levels (Table 3(Tab. 3)). The association with waist circumference and SBP resulted in a trend towards significance. Some of these relationships should be considered as explorative since after applying a multiple comparison correction, the statistical significance was toned down. Interestingly, in the case of BMI variation, which is one of the main variables, remained statistically associated with the change of PON1 ARE activity.
Table 3. Pearson correlation analyses between arylesterase (ARE) activity variation and changes in selected anthropometric, body composition, blood pressure and biochemical variables.
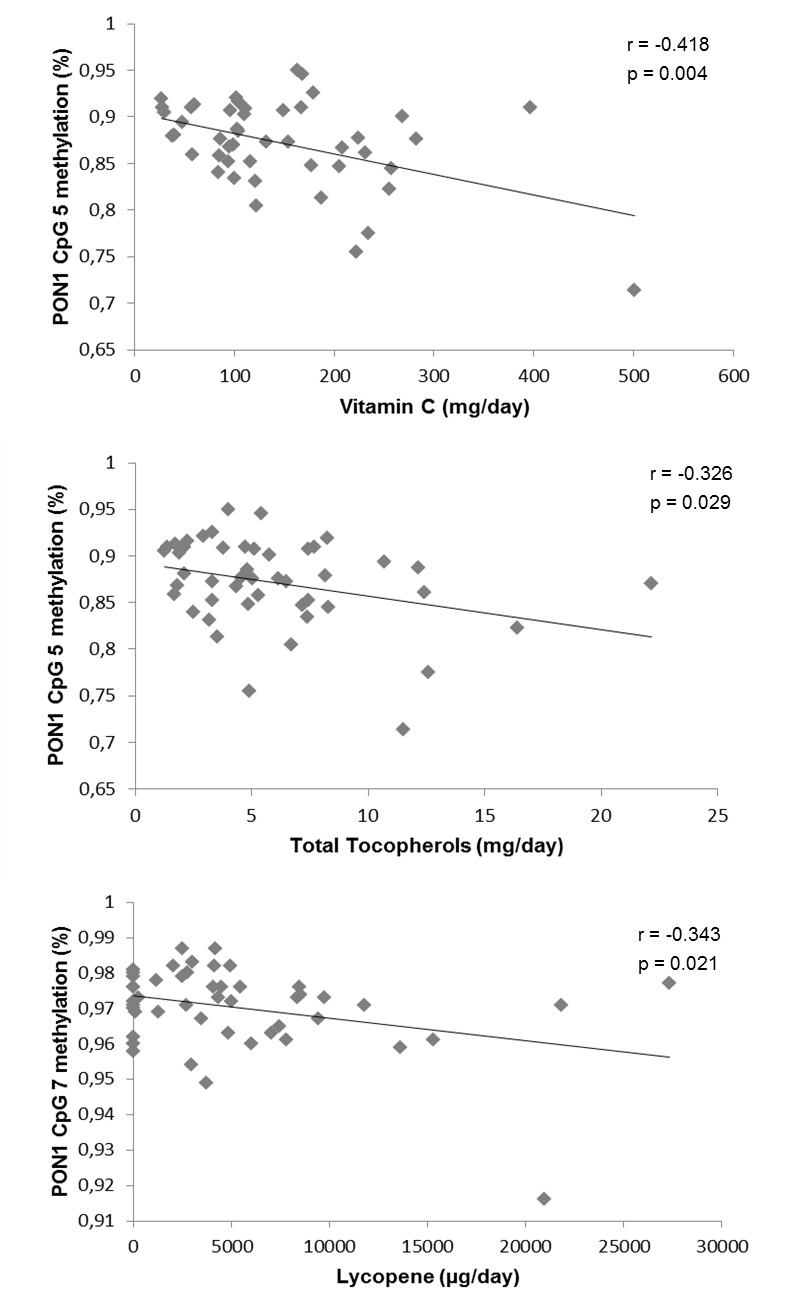
At the end of the intervention, the PON1 ARE activity measurements showed positive correlations with the intake of the selected antioxidants vitamin C (p = 0.001), total tocopherols (p = 0.009) and lycopene (p = 0.038) recorded by the 48h weighed food record (Figure 2(Fig. 2)). Interestingly, when assessing the association between the selected CpG sites methylation of the PON1 gene and the related ARE activity at baseline (Table 4(Tab. 4)), a significant inverse correlation was found concerning the PON1 CpG 1, CpG 2, CpG 3 and CpG 4 sites with the enzymatic ARE activity. Moreover, these associations remained significant after applying the Benjamini-Hochberg test for multiple comparisons. Finally, the analysis of the possible relationships between the antioxidant dietary intake and the percentage of methylation of the different PON1 gene CpG sites, revealed a correlation between the selected antioxidants: vitamin C, total tocopherols and lycopene and the selected CpG sites at baseline (Figure 3(Fig. 3)).
Figure 2. Associations between antioxidant dietary components intake and arylesterase (ARE) activity.
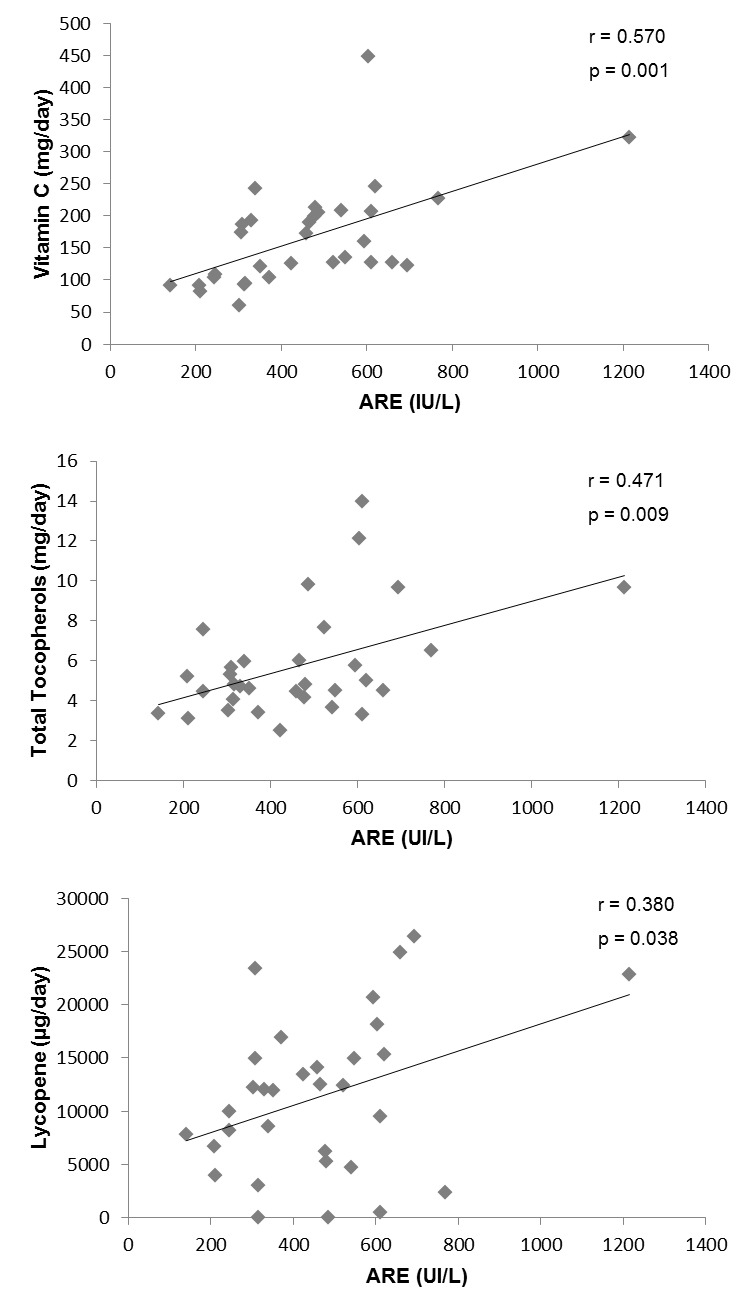
Table 4. Pearson correlation analyses between arylesterase (ARE) activity and the selected CpG sites methylation (%) of PON1 gene at baseline.
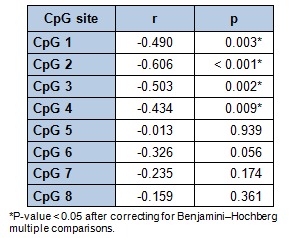
Figure 3. Statistically significant correlations between antioxidant dietary components intake and methylation (%) of PON1 gene CpG sites at baseline.
Discussion
The assessment of processes associated to weight loss may contribute to a better understanding of the metabolic and genetic machinery, which will benefit the implementation of personalized dietary treatments.
Indeed, as reported in other studies concerning hypocaloric diets (Straznicky et al., 2010[39]), the energy-restricted intervention proved to be effective in improving MetS abnormalities except for LDL-c, which was higher at the end of the study. This outcome agrees with Clifton et al. who described that in some cases, LDL-c may increase despite weight loss (Clifton et al., 2007[8]). Nevertheless, Apo B, which has been considered a better predictor of CVD onset than any other lipid measurement (McQueen et al., 2008[27]), evidenced statistically significant decreased levels.
Concerning PON1 ARE activity, it seems logical the positive correlation found with the HDL-c levels at baseline since it is an HDL-c related enzyme, as other studies have previously reported (Aksoy et al., 2009[2]; Mascarenhas-Melo et al., 2013[26]; Tabur et al., 2010[40]). The positive correlation with LDL-c and TC also agree with previous studies (Aksoy et al., 2009[2]; Younis et al., 2013[46]).
The PON1 ARE activity was reduced in parallel to some anthropometric (body weight, BMI), body composition (total fat mass), blood pressure and biochemical biomarkers (TG) related to the MetS. Although there is scarce research about the relationship of PON1 activity and obesity or MetS, the studies carried out point to relate obese/MetS status to lower levels of ARE activity (Ferretti et al., 2012[12]; Kota et al., 2013[20]). Nevertheless, when it comes to clinical trials, in accordance to our results, a significant decrease of ARE activity and serum PON1 protein levels after a weight loss intervention, as well as a significant association between reduced PON1 and reductions in body fat has been reported (Rector et al., 2007[36]). Similarly, it has been shown that significant depletions of all metabolic outcomes in addition to a significant reduction of PON1 activity correlated with BMI reduction, after a low calorie diet intervention (Kotani et al., 2009[21]). In this last study, a significant association of PON1 activity changes and LDL-c depletion was also observed; however, no significant relationships were found in the present experimental trial. On the other hand, to our knowledge, this is the first research that has found an association of ARE activity and TG changes after a dietary intervention. With regards to the significant association of PON1 activity depletion and reduction of blood pressure levels, it has been reported significantly higher ARE levels in hypertensive than in normotensive children (Akis et al., 2009[1]), while in other studies carried out in different adult populations no association was found (Tabur et al., 2010[40]; Usta et al., 2011[42]). However, the studies mentioned above were cross-sectional studies, while the present work is a clinical intervention. Taking into account the previous issues and the lack of research in the area, the findings observed in the present study may indicate that reductions on PON1 general activity might be indicative of the improvement in the overall MetS/obese related features, in patients with MetS manifestations under an energy-restricted programme.
The activity of PON1 is under genetic and environmental regulation (Jarvik et al., 2002[16]). Within the environmental factors that may alter PON1 activity, dietary antioxidants are of major importance, since PON1 is an oxidative stress related enzyme (de la Iglesia et al., 2014[10]; Kheir-Eldin et al., 2008[17]). Among the different antioxidants that can be incorporated in the diet, the antioxidant capacity of vitamins C and E and lycopene is well-established (Chen et al., 2013[6]; Mustacich et al., 2007[30]; Story et al., 2010[38]). The positive relationships between the ARE activity and the selected antioxidant components reported in the present work may be explained by the capacity of the dietary antioxidants to scavenge free-oxygen radical products that may depress PON1 activity, as previous studies have suggested (Jarvik et al., 2002[16]; Tsakiris et al., 2009[41]).
DNA methylation is one of the major epigenetic mechanisms considered to regulate gene expression, together with histone modifications and noncoding RNA activity (Portela et al., 2010[34]). Methylation of the CpG-rich region (CpG island) overlapping a gene’s promoter is a generally accepted mechanism for silencing expression (Vanderkraats et al., 2013[43]). Our findings confirm this outcome since four CpG sites and the average methylation of all the CpG sites studied inversely correlated with the ARE activity.
About the possible mechanisms by which the dietary antioxidants exert their effects, modulation of gene expression through regulation of DNA methylation is one of the main studied mechanisms (Bartels et al., 2007[4]; Malireddy et al., 2012[24]; Milagro et al., 2009[29]). In the present work, inverse correlations between dietary vitamin C, total tocopherols and lycopene with different CpG sites methylation was observed, which may influence in the ARE activity as other studies have suggested (Jarvik et al., 2002[16]; Tsakiris et al., 2009[41]).
This work has the limitations that we have not determined PON1 methylation levels at the end of the intervention and we have not measured expression levels. However, we found that CpG2 and CpG7 sites match a core-binding consensus motif for the GATA binding protein 1 (globin transcription factor 1) and CpG2 site matches for myeloid zinc finger 1 (MZF1), which are known transcriptional regulators for several pathways (Gaboli et al., 2001[14]; Zheng et al., 2010[47]). Furthermore, some statistical associations concerning changes in PON1 ARE activity and anthropometric and biochemical measurements variations were lost after the application of a multiple comparison correction test.
In conclusion, the present study reports that ARE activity decreased in parallel with MetS-related markers, while dietary antioxidants intake may enhance the ARE activity by lowering the PON1 gene methylation in patients with MetS features, under an energy-restricted intervention. These results suggest that further investigations into PON1 activity might be a good target to better understand the metabolic effects of the dietary intervention studies, in which epigenetic processes may be involved.
Acknowledgements
We wish to thank the physician Blanca E. Martinez de Morentin, the nurse Salome Perez, the technician Veronica Ciaurriz and the researcher Paloma Celada for excellent technical assistance as well as the volunteers who participated in this study.
This work was supported by Health Department of the Government of Navarra (48/2009) and the Linea Especial about Nutrition, Obesity and Health (University of Navarra LE/97). Also CIBERobn and RETICS schemes from the isciii are gratefully credited. Carlos III Health Institute provided a predoctoral grant to R. de la Iglesia (n° FI10/00587). M.L.M. holds a Juan de la Cierva fellowship from Spanish Ministry of Economic and Competitiveness.
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Akis N, Pala K, Meric-Utku A, Seyithan B, Sarandol E, Aytekin H. Hypertension in children (12-14 years) - a case-control study in Bursa, Turkey. Turk J Pediatr. 2009;51:437–443. [PubMed] [Google Scholar]
- 2.Aksoy H, Aksoy AN, Ozkan A, Polat H. Serum lipid profile, oxidative status, and paraoxonase 1 activity in hyperemesis gravidarum. J Clin Lab Anal. 2009;23:105–109. doi: 10.1002/jcla.20298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.ALCE IGENIERIA DIAL Programme. Evaluation and design diets programme; 2011. Available from: http://www.alceingenieria.net/nutricion.htm. [Google Scholar]
- 4.Bartels M, Meyer zu Vilsendorf A, Kassahun WT, von Gerstenbergk B, Engelhart K, Meyer zu Vilsendorf E, et al. Protective effect of antioxidative vitamins against lipid peroxidation in liver ischemia and reperfusion – an animal experimental study. EXCLI J. 2007;6:152–166. [Google Scholar]
- 5.Canales A, Sanchez-Muniz FJ, Bastida S, Librelotto J, Nus M, Corella D, et al. Effect of walnut-enriched meat on the relationship between VCAM, ICAM, and LTB4 levels and PON-1 activity in ApoA4 360 and PON-1 allele carriers at increased cardiovascular risk. Eur J Clin Nutr. 2011;65:703–710. doi: 10.1038/ejcn.2011.20. [DOI] [PubMed] [Google Scholar]
- 6.Chen GC, Lu DB, Pang Z, Liu QF. Vitamin C intake, circulating vitamin C and risk of stroke: a meta-analysis of prospective studies. J Am Heart Assoc. 2013;2:e000329. doi: 10.1161/JAHA.113.000329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Christensen BC, Marsit CJ. Epigenomics in environmental health. Front Genet. 2011;2:84. doi: 10.3389/fgene.2011.00084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Clifton PM, Keogh J. Metabolic effects of high-protein diets. Curr Atheroscler Rep. 2007;9:472–478. doi: 10.1007/s11883-007-0063-y. [DOI] [PubMed] [Google Scholar]
- 9.Cohen E, Aviram M, Khatib S, Rabin A, Mannheim D, Karmeli R, et al. Increased levels of human carotid lesion linoleic acid hydroperoxide in symptomatic and asymptomatic patients is inversely correlated with serum HDL and paraoxonase 1 activity. J Lipids. 2012;2012:762560. doi: 10.1155/2012/762560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.de la Iglesia R, Lopez-Legarrea P, Abete I, Bondia-Pons I, Navas-Carretero S, Forga L, et al. A new dietary strategy for long-term treatment of the metabolic syndrome is compared with the American Heart Association (AHA) guidelines: the MEtabolic Syndrome REduction in NAvarra (RESMENA) project. Br J Nutr. 2014;111:643–652. doi: 10.1017/S0007114513002778. [DOI] [PubMed] [Google Scholar]
- 11.de la Iglesia R, Lopez-Legarrea P, Celada P, Sanchez-Muniz FJ, Martinez JA, Zulet MA. Beneficial effects of the resmena dietary pattern on oxidative stress in patients suffering from metabolic syndrome with hyperglycemia are associated to dietary tac and fruit consumption. Int J Mol Sci. 2013;14:6903–6919. doi: 10.3390/ijms14046903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ferretti G, Bacchetti T, Masciangelo S, Grugni G, Bicchiega V. Altered inflammation, paraoxonase-1 activity and HDL physicochemical properties in obese humans with and without Prader-Willi syndrome. Dis Model Mech. 2012;5:698–705. doi: 10.1242/dmm.009209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- 14.Gaboli M, Kotsi PA, Gurrieri C, Cattoretti G, Ronchetti S, Cordon-Cardo C, et al. Mzf1 controls cell proliferation and tumorigenesis. Genes Dev. 2001;15:1625–1630. doi: 10.1101/gad.902301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Garg PK, Biggs ML, Carnethon M, Ix JH, Criqui MH, Britton KA, et al. Metabolic syndrome and risk of incident peripheral artery disease: The cardiovascular health study. Hypertension. 2014;63:413–419. doi: 10.1161/HYPERTENSIONAHA.113.01925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jarvik GP, Tsai NT, McKinstry LA, Wani R, Brophy VH, Richter RJ, et al. Vitamin C and E intake is associated with increased paraoxonase activity. Arterioscler Thromb Vasc Biol. 2002;22:1329–1333. doi: 10.1161/01.atv.0000027101.40323.3a. [DOI] [PubMed] [Google Scholar]
- 17.Kheir-Eldin A, Motawi T, Sadik N. Effect of some natural antioxidants on aflatoxin B1- induced hepatic toxicity. EXCLI J. 2008;7:119–131. [Google Scholar]
- 18.Kim DS, Marsillach J, Furlong CE, Jarvik GP. Pharmacogenetics of paraoxonase activity: elucidating the role of high-density lipoprotein in disease. Pharmacogenomics. 2013;14:1495–1515. doi: 10.2217/pgs.13.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Koncsos P, Seres I, Harangi M, Pall D, Jozsa L, Bajnok L, et al. Favorable effect of short-term lifestyle intervention on human paraoxonase-1 activity and adipokine levels in childhood obesity. J Am Coll Nutr. 2011;30:333–339. doi: 10.1080/07315724.2011.10719976. [DOI] [PubMed] [Google Scholar]
- 20.Kota SK, Meher LK, Jammula S, Krishna SV, Modi KD. Implications of serum paraoxonase activity in obesity, diabetes mellitus, and dyslipidemia. Indian J Endocrinol Metab. 2013;17:402–412. doi: 10.4103/2230-8210.111618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kotani K, Sakane N, Sano Y, Tsuzaki K, Matsuoka Y, Egawa K, et al. Changes on the physiological lactonase activity of serum paraoxonase 1 by a diet intervention for weight loss in healthy overweight and obese women. J Clin Biochem Nutr. 2009;45:329–334. doi: 10.3164/jcbn.09-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kumar D, Rizvi SI. Plasma paraoxonase 1 arylesterase activity in D-galactose-induced aged rat model: correlation with LDL oxidation and redox status. Aging Clin Exp Res. 2014;26:261–267. doi: 10.1007/s40520-013-0170-2. Available from: http://dx.doi.org/10.1007/s40520-013-0170-2. [DOI] [PubMed] [Google Scholar]
- 23.Lopez-Legarrea P, de la Iglesia R, Abete I, Bondia-Pons I, Navas-Carretero S, Forga L, et al. Short-term role of the dietary total antioxidant capacity in two hypocaloric regimes on obese with metabolic syndrome symptoms: the RESMENA randomized controlled trial. Nutr Metab. 2013;10:22. doi: 10.1186/1743-7075-10-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Malireddy S, Kotha SR, Secor JD, Gurney TO, Abbott JL, Maulik G, et al. Phytochemical antioxidants modulate mammalian cellular epigenome: implications in health and disease. Antioxid Redox Signal. 2012;17:327–339. doi: 10.1089/ars.2012.4600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mansego ML, Milagro FI, Campion J, Martinez JA. Techniques of DNA methylation analysis with nutritional applications. J Nutrigenet Nutrigenomics. 2013;6:83–96. doi: 10.1159/000350749. [DOI] [PubMed] [Google Scholar]
- 26.Mascarenhas-Melo F, Sereno J, Teixeira-Lemos E, Marado D, Palavra F, Pinto R, et al. Implication of low HDL-c levels in patients with average LDL-c levels: a focus on oxidized LDL, large HDL subpopulation, and adiponectin. Mediators Inflamm. 2013;2013:612038. doi: 10.1155/2013/612038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McQueen MJ, Hawken S, Wang X, Ounpuu S, Sniderman A, Probstfield J, et al. Lipids, lipoproteins, and apolipoproteins as risk markers of myocardial infarction in 52 countries (the INTERHEART study): a case-control study. Lancet. 2008;372:224–233. doi: 10.1016/S0140-6736(08)61076-4. [DOI] [PubMed] [Google Scholar]
- 28.Milagro FI, Campion J, Cordero P, Goyenechea E, Gomez-Uriz AM, Abete I, et al. A dual epigenomic approach for the search of obesity biomarkers: DNA methylation in relation to diet-induced weight loss. FASEB J. 2011;25:1378–1389. doi: 10.1096/fj.10-170365. [DOI] [PubMed] [Google Scholar]
- 29.Milagro FI, Campion J, Garcia-Diaz DF, Goyenechea E, Paternain L, Martinez JA. High fat diet-induced obesity modifies the methylation pattern of leptin promoter in rats. J Physiol Biochem. 2009;65:1–9. doi: 10.1007/BF03165964. [DOI] [PubMed] [Google Scholar]
- 30.Mustacich DJ, Bruno RS, Traber MG. Vitamin E. Vitam Horm. 2007;76:1–21. doi: 10.1016/S0083-6729(07)76001-6. [DOI] [PubMed] [Google Scholar]
- 31.Nus M, Sanchez-Muniz FJ, Sanchez-Montero JM. A new method for the determination of arylesterase activity in human serum using simulated body fluid. Atherosclerosis. 2006;188:155–159. doi: 10.1016/j.atherosclerosis.2005.10.016. [DOI] [PubMed] [Google Scholar]
- 32.Nus M, Sanchez-Muniz FJ, Sanchez-Montero JM. Arylesterase. Methodological and functional aspects of a key enzyme in the cardiovascular disease. An Real Acad Farm. 2008;74:5–27. [Google Scholar]
- 33.Perez-Cornago A, Lopez-Legarrea P, de la Iglesia R, Lahortiga F, Martinez JA, Zulet MA. Longitudinal relationship of diet and oxidative stress with depressive symptoms in patients with metabolic syndrome after following a weight loss treatment: The RESMENA project. Clin Nutr. Clin Nutr. 2013:S0261–5614(13)00317. doi: 10.1016/j.clnu.2013.11.011.. [DOI] [PubMed] [Google Scholar]
- 34.Portela A, Esteller M. Epigenetic modifications and human disease. Nat Biotechnol. 2010;28:1057–1068. doi: 10.1038/nbt.1685. [DOI] [PubMed] [Google Scholar]
- 35.Precourt LP, Amre D, Denis MC, Lavoie JC, Delvin E, Seidman E, et al. The three-gene paraoxonase family: physiologic roles, actions and regulation. Atherosclerosis. 2011;214:20–36. doi: 10.1016/j.atherosclerosis.2010.08.076. [DOI] [PubMed] [Google Scholar]
- 36.Rector RS, Warner SO, Liu Y, Hinton PS, Sun GY, Cox RH, et al. Exercise and diet induced weight loss improves measures of oxidative stress and insulin sensitivity in adults with characteristics of the metabolic syndrome. Am J Physiol Endocrinol Metab. 2007;293:E500–E506. doi: 10.1152/ajpendo.00116.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shapiro DS, Loiacono LA. Mean arterial pressure: therapeutic goals and pharmacologic support. Crit Care Clin. 2010;26:285–293. doi: 10.1016/j.ccc.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 38.Story EN, Kopec RE, Schwartz SJ, Harris GK. An update on the health effects of tomato lycopene. Annu Rev Food Sci Technol. 2010;1:189–210. doi: 10.1146/annurev.food.102308.124120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Straznicky NE, Lambert EA, Nestel PJ, McGrane MT, Dawood T, Schlaich MP, et al. Sympathetic neural adaptation to hypocaloric diet with or without exercise training in obese metabolic syndrome subjects. Diabetes. 2010;59:71–79. doi: 10.2337/db09-0934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tabur S, Torun AN, Sabuncu T, Turan MN, Celik H, Ocak AR, et al. Non-diabetic metabolic syndrome and obesity do not affect serum paraoxonase and arylesterase activities but do affect oxidative stress and inflammation. Eur J Endocrinol. 2010;162:535–541. doi: 10.1530/EJE-09-0732. [DOI] [PubMed] [Google Scholar]
- 41.Tsakiris S, Karikas GA, Parthimos T, Tsakiris T, Bakogiannis C, Schulpis KH. Alpha-tocopherol supplementation prevents the exercise-induced reduction of serum paraoxonase 1/arylesterase activities in healthy individuals. Eur J Clin Nutr. 2009;63:215–221. doi: 10.1038/sj.ejcn.1602918. [DOI] [PubMed] [Google Scholar]
- 42.Usta M, Turan E, Aral H, Inal BB, Gurel MS, Guvenen G. Serum paraoxonase-1 activities and oxidative status in patients with plaque-type psoriasis with/without metabolic syndrome. J Clin Lab Anal. 2011;25:289–295. doi: 10.1002/jcla.20471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vanderkraats ND, Hiken JF, Decker KF, Edwards JR. Discovering high-resolution patterns of differential DNA methylation that correlate with gene expression changes. Nucleic Acids Res. 2013;41:6816–6827. doi: 10.1093/nar/gkt482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vazquez-Velasco M, Esperanza Diaz L, Lucas R, Gomez-Martinez S, Bastida S, Marcos A, et al. Effects of hydroxytyrosol-enriched sunflower oil consumption on CVD risk factors. Br J Nutr. 2011;105:1448–1452. doi: 10.1017/S0007114510005015. [DOI] [PubMed] [Google Scholar]
- 45.Whitworth JA, Chalmers J. World Health Organization - International Society of Hypertension (WHO/ ISH) hypertension guidelines. Clin Exp Hypertens. 2004;26:747–752. doi: 10.1081/ceh-200032152. [DOI] [PubMed] [Google Scholar]
- 46.Younis NN, Soran H, Charlton-Menys V, Sharma R, Hama S, Pemberton P, et al. High-density lipoprotein impedes glycation of low-density lipoprotein. Diab Vasc Dis Res. 2013;10:152–160. doi: 10.1177/1479164112454309. [DOI] [PubMed] [Google Scholar]
- 47.Zheng R, Blobel GA. GATA transcription factors and cancer. Genes Cancer. 2010;1:1178–1188. doi: 10.1177/1947601911404223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zulet MA, Bondia-Pons I, Abete I, de la Iglesia R, Lopez-Legarrea P, Forga L, et al. The reduction of the metabolyc syndrome in Navarra-Spain (RESMENA-S) study: a multidisciplinary strategy based on chrononutrition and nutritional education, together with dietetic and psychological control. Nutr Hosp. 2011;26:16–26. [PubMed] [Google Scholar]


