Abstract
Psychological stress has been linked empirically with dysregulation of facets of the human immune system, yet these effects are not the same in every situation or population. Recent research has made strides towards understanding risk factors for immune dysregulation as well as why these risks occur. This review discusses mechanisms and mediators underlying the stress-immune relation, the role of context in determining whether an immunologic responses to stress is adaptive versus maladaptive, and the stress-immune relation in populations including children exposed to early adversity, older adults, and individuals with clinical diagnoses. The reviewed work holds great promise for further elucidating the circumstances under which psychological stress has immunological consequences, and provides new directions for work in this field.
Introduction
Stress is a broad concept that comprises challenging or difficult circumstances (stressors) or the physiological or psychological response to such circumstances (stress responses). In humans, among other species, one of the systems that responds to challenging circumstances is the immune system. Broadly, the immune system comprises cells, proteins, organs, and tissues that work together to provide protection against bodily disease and damage (see Box for explanations of relevant immunological parameters). Several facets of the human immune system have been empirically associated with stress. During acute stress lasting a matter of minutes, certain kinds of cells are mobilized into the bloodstream, potentially preparing the body for injury or infection during “fight or flight” [1]. Acute stress also increases blood levels of pro-inflammatory cytokines [2]. Chronic stress lasting from days to years, like acute stress, is associated with higher levels of pro-inflammatory cytokines, but with potentially different health consequences [3]. Inflammation is a necessary short-term response for eliminating pathogens and initiating healing, but chronic, systemic inflammation represents dysregulation of the immune system and increases risk for chronic diseases, including atherosclerosis and frailty [4]. Another consequence of chronic stress is activation of latent viruses. Latent virus activation can reflect the loss of immunological control over the virus, and frequent activation can cause wear-and-tear on the immune system [5].
Interestingly, these responses may not be the same for everyone. Those who have experienced early adversity, for example, may be more likely to exhibit exaggerated immune reactions to stress [6, 7]. Currently, the field is moving toward a greater understanding of who might be most at risk for chronic inflammation and other forms of immunological dysregulation, and why. This question is important not only for health, but also for longevity, as evidence suggests that the immunological effects of chronic stress can advance cellular aging and shorten telomere length [8].
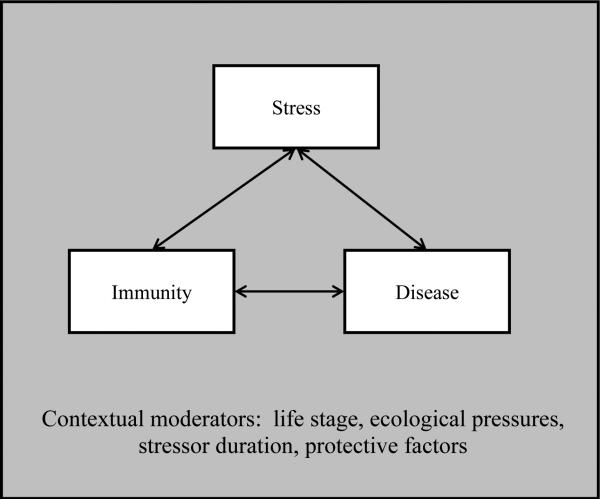
Meta-analyses provide a look backward at this research and summarize what has been learned about the relationship between stress and human immunity since it was first studied in the 1960s [1, 2, 9]. This review describes recent, groundbreaking work on the stress-immune relation in humans, including the immunological consequences of stress in early and late life, mediators of the stress-immunity link, ecological perspectives, and how the relationship between stress and immunity is manifest in clinical populations (see Figure).
Figure.
Stress, immunity, and disease can affect each other in reciprocal ways, but these relationships can be moderated by life stage, other ecological pressures and goals, stressor duration, and protective factors such as good sleep.
Early life stress
Stress that occurs early in development (e.g., maltreatment, poverty, and other adverse experiences) has immunological consequences that can be observed both in the near and long term after the stressor occurs. Early life stress (ELS) in children associates with immunological dysregulation, including low basal levels of cytokines that control immune responses [10]. When immune cells were stimulated in vitro (e.g., with tetanus toxoid), those cells from children who experienced ELS produced more pro-inflammatory cytokines [10]. Whereas much of the extant research focuses on maltreatment or poverty, a recent study into the effects of a less-studied adversity, bullying, also suggests that chronic peer victimization predicts a steeper increase in CRP from childhood into young adulthood [11]. EBV antibody levels in a younger adult sample were also found to differ based on the type, timing, and frequency of exposure to ELS. Individuals exposed to sexual abuse more than 10 times, as well as those physically abused starting between ages 3 and 5, had elevated levels of antibodies against EBV as adults, a signal of viral reactivation [12]. In adults, a meta-analysis of ELS and inflammation found a positive association between maltreatment and several inflammatory markers, with the most robust association for circulating CRP [13]. Recent work has investigated mechanisms linking ELS to immune alterations over time (e.g., self-control, adiposity, smoking, and stress; 14, 15] as well as examining inflammatory dysregulation as a pathway through which ELS affects adult disease prevalence and outcomes [16]. Finally, empirically based interventions to target immunological consequences of ELS are a necessary next step; recent evidence suggests the plausibility of such interventions to improve inflammatory profiles for youth raised in low-income families [17].
Stress, immunity, and aging
As people age, they are less able to mount appropriate immune responses to stressors. These could be physical stressors, such as injury, or psychological stressors such as caregiving. In addition, psychological stress affects organisms in a manner similar to the effects of chronological age, and chronological aging coupled with chronic stress accelerates immunological aging [18]. Research has suggested that older adults are unable to terminate cortisol production in response to stress. Cortisol is ordinarily anti-inflammatory and contains the immune response, but chronic elevations can lead to the immune system becoming “resistant,” an accumulation of stress hormones, and increased production of inflammatory cytokines that further compromise the immune response [18]. Older adults often have to provide long term care for an ailing spouse or partner. Caregiving has been implicated in significantly lower antibody and cell-mediated immune responses after vaccination [19, 20]. Caregivers also experience longer wound healing times, lower lymphocyte proliferation, increased proinflammatory cytokine levels, and more reactivation of latent viruses [21].
An important direction in aging research involves an examination of telomeres. Telomere length has been used as a measure of biological aging and is associated with psychological, physiological, and social factors. Chronic stress is linked to shortened telomere length along with increased disease in older adults [22]. Socioeconomic factors such as marital status and income have been linked with telomere length: those married for longer periods of time and who make more money are biologically younger than others in their cohort [22, 23]. However, studies thus far have found this link only in Caucasians and Hispanics, but not African Americans. This suggests that low socioeconomic status (SES) may accelerate aging in some populations [23]. Interestingly, health behaviors can moderate this effect by protecting individuals from accelerated aging during stress exposure [24]. It is unclear how this moderation occurs, and more work is needed.
Collectively, recent work points to new discoveries into how biological aging and stress interact to influence the immune response. This will lead to a better understanding of mechanisms of immunosenescence caused by stress and chronological aging that are presently unclear.
Biological and behavioral mediators of the relationship between stress and immunity
How does stress get “under the skin” to influence immunity? Immune cells have receptors for neurotransmitters and hormones such as norepinephrine, epinephrine, and cortisol, which mobilize and traffic immune cells, ideally preparing the body to mount an immune response if needed [25]. Recent evidence shows that immunological cells (e.g., lymphocytes) change their responsiveness to signaling from these neurotransmitters and hormones during stress [26]. However, immunological responses are biologically and energetically costly, and over time, chronic stress produces negative systemic changes both in immune trafficking and in target tissues [6].
The linkages between stress and immunity may be mediated by specific health behaviors, psychosocial factors, or both. For instance, stress has been linked to being in troubled relationships, having negative or competitive social interactions, and feeling lonely, which have each in turn been linked to increases in pro-inflammatory responses to stress [27-29]. Other potential mediators, like getting good sleep, are increasingly being recognized as important pieces of the stress-immunity puzzle [30]. Even one night of total sleep deprivation was recently found to significantly increase neutrophil counts and decrease neutrophil function in healthy men [31].
Taken together, these examples highlight a better understanding of the factors that mediate or moderate stress's influence on immunity. This direction may serve to one day develop targeted behavioral or pharmacotherapies to those at highest risk for poor health outcomes.
Ecological immunology
Over the last several years, there has been greater attention paid to the relevance of ecological immunity to the relationship between stress and immunity. Ecological immunity is based on the premise that mounting immune responses is energetically costly and that the (mal)adaptiveness of immune responses to stress is determined by cost:benefit ratios [32-34]. In early human history many stressors were life-threatening: being eaten by a predator, being excluded by one's peer group, or being faced with starvation, to name a few. Appropriately responding to some of these stressors (e.g., predation) required activating the energetically costly fight or flight response, including immunological changes that could protect against infection secondary to wounding. However, energetic costs of the immune system during other kinds of stressors (e.g., social exclusion) that resulted in less availability of energetic resources (e.g., shared food) might have been counterproductive. Thus, downregulating immune responses might have been evolutionarily adaptive. Research in bumblebees finds that under conditions of starvation, immune responses to an immune challenge accelerated time to death from starvation, suggesting that allocating energy to the immune system under those conditions was maladaptive [35]. Although energetic resources are abundant in the modern environment, physiological evidence of these ecological tradeoffs in the ancestral environment can still be found. For example, in contemporary humans, costly endeavors such as building and maintaining a large social network or persisting on unsolvable challenges can be associated with decreases in some immune parameters [36, 37]. Taken together, these and other findings [for reviews, see 33, 38] suggest that ecological conditions and resource availability may shape immune functioning in ways that remain relatively underexplored.
Stress, immunity, and clinical health
Psychological stress has been implicated in altered immune functioning in many diseases. Stress induces chronic immune activation and altered health outcomes that resemble those seen in chronic inflammatory diseases such as RA [39, 40]. Altered immune function can lead to exacerbated symptoms of both physical and psychological illnesses. In irritable bowel syndrome, sustained cortisol activity during stress is associated with an increase in gastrointestinal symptoms [41]. High levels of proinflammatory cytokines resulting from stress have recently been implicated in the etiology of schizophrenia and schizophrenia-related brain alterations [42]. Chronic stress has been shown to enhance risk for developing autoimmune disease [e.g., 43]. Individuals with autoimmune disease also appear to have difficulty down-regulating their immune responses after exposure to stressors. In MS, neuropeptides secreted under stress (e.g., corticotropin-releasing hormone) activate glial cells in the brain to release inflammatory molecules that result in brain inflammation and worsen MS pathology [44]. Similar immune activation and symptom exacerbation is evidenced in those with other autoimmune diseases [40]. Currently, possible mechanisms by which autoimmune diseases alter individual responses to stress are being explored. This knowledge may lead to interventions that decrease stress-induced immune responses and improve outcomes in autoimmune diseases.
Conclusions and future directions
Research on the immunological effects of stress has burgeoned over the past decade following Segerstrom and Miller's meta-analysis [1]. This research has explored new avenues, including the areas reviewed here, that show particular promise for illuminating the conditions under which stress impacts the immune system. Research on stressors occurring early (i.e., childhood and adolescence) and late (i.e., aging) in the lifespan have suggested that individuals exposed to chronic stressors (e.g., abuse, caregiving) can exhibit immune dysregulation that may be persistent and severe. Stressor qualities (e.g., type, timing) as well as individual characteristics that make individuals more or less susceptible to these effects are targets for future work. Examinations of mediators and mechanisms of the stress-immune relation can also determine how and for whom exposure to stress impacts the immune response. Ecological immunology suggests that downregulating the immune response may sometimes be adaptive, and future work building from this perspective will help to further elucidate contexts in which immunosuppression may occur but progress toward superordinate goals is facilitated. Finally, research into the effects of stress on inflammation in clinical populations has demonstrated that stress exposure can increase the likelihood of developing disease, as well as exacerbating preexisting conditions. Further work in this area may help to treat or even prevent morbidity. Overall, this area of research is broad, rapidly developing, and holds promise for improving human health.
Box: Guide to some immunological parameters related to stress.
Antibodies: Proteins produced by immune cells that can bind to pathogens such as viruses, bacteria, and parasites. Bound pathogens are inactivated or marked for killing by other immune cells.
Autoimmune disease: Caused when the immune system misidentifies self tissue as foreign and mounts an attack against it. Examples include rheumatoid arthritis (RA), lupus, and multiple sclerosis (MS).
C-reactive protein (CRP): A downstream product of pro-inflammatory signaling and marker of systemic inflammation.
Cell-mediated immunity: The arm of the immune system that protects against pathogens residing inside cells (e.g., viruses) and other “sick” cells such as cancer cells.
Cortisol: A steroid hormone produced by the adrenal gland with broad metabolic effects, including suppression of some facets of the immune system.
Cytokines: Proteins that coordinate immune responses. Examples include interleukins (IL). Some cytokines, such as IL-5 and IL-10, primarily control and contain immune responses. Others, such as IL-6 and tumor necrosis factor-α (TNF-α), induce inflammation.
Inflammation: Local inflammation is a part of the healing process that includes accumulation of immune cells, anti-pathogen activity, and initiation of tissue repair. Chronic, systemic inflammation, in contrast, can promote tissue damage across a number of systems.
Latent viruses: Viruses that reside in the body indefinitely after infection, often without overt disease consequences either acutely or chronically. Examples include Epstein-Barr virus (EBV) and cytomegalovirus (CMV).
Neutrophils: The first cells to infiltrate damaged or infected tissue and effect an inflammatory response.
Telomeres: The protective caps on the end of chromosomes that prevent deterioration.
Highlights.
Psychological stress can dysregulate the human immune system.
Stress can impact immunity differentially across individuals and contexts.
Recent work in this area has made strides towards elucidating these differences.
Future work holds promise for reducing stress's effects on physical health.
Acknowledgement
funding from AG026307-R01 (Segerstrom), AG048692-F31 (Boggero), & AG033629-K02 (Segerstrom)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
Papers of particular interest have been demarcated as:
*of special interest; **of outstanding interest
- 1.Segerstrom SC, Miller GE. Psychological stress and the human immune system: a meta-analytic study of 30 years of inquiry. Psychol Bull. 2004;130:601–630. doi: 10.1037/0033-2909.130.4.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Steptoe A, Hamer M, Chida Y. The effects of acute psychological stress on circulating inflammatory factors in humans: a review and meta-analysis. Brain Behav Immun. 2007;21:901–912. doi: 10.1016/j.bbi.2007.03.011. [DOI] [PubMed] [Google Scholar]
- 3.Gouin JP, Glaser R, Malarkey WB, Beversdorf D, Kiecolt-Glaser J. Chronic stress, daily stressors, and circulating inflammatory markers. Health Psychol. 2012;31:264–268. doi: 10.1037/a0025536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ershler WB. Interleukin-6: a cytokine for gerontologists. J Am Geriatr Soc. 1993;41:176–181. doi: 10.1111/j.1532-5415.1993.tb02054.x. [DOI] [PubMed] [Google Scholar]
- 5.Pawelec G, Akbar A, Caruso C, Solana R, Grubeck-Loebenstein B, Wikby A. Human immunosenescence: is it infectious? Immunol Rev. 2005;205:257–268. doi: 10.1111/j.0105-2896.2005.00271.x. [DOI] [PubMed] [Google Scholar]
- *6.McEwen BS. Brain on stress: how the social environment gets under the skin. Proc Natl Acad Sci USA. 2012;109:17180–17185. doi: 10.1073/pnas.1121254109. In this relatively short review, the author does a thorough job of explaining how stress and psychosocial factors get “under the skin” to influence health. The paper starts by reviewing the history and major theories of stress's influence on health and then discusses more contemporary work on the role of allostatic load, glucocorticoids, and brain function under stress. Finally, the review concludes with a section on interventions and what they can and cannot do. Overall, the paper provides a thorough yet accessible review on stress, social environments, and health. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carpenter LL, Gawuga CE, Tyrka AR, Lee JK, Anderson GM, Price LH. Association between plasma IL-6 responses to acute stress and early-life adversity in healthy adults. Neuropsychopharmacology. 2010;35:2617–2623. doi: 10.1038/npp.2010.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tomiyama AJ, O'Donovan A, Lin J, Puterman E, Lazaro A, Chan J, Dhabhar FS, Wolkowitz O, Kirschbaum C, Blackburn E, Epel E. Does cellular aging relate to patterns of allostasis?: an examination of basal and stress reactive HPA axis activity and telomere length. Physiol Behav. 2012;106:40–45. doi: 10.1016/j.physbeh.2011.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pedersen AF, Zachariae R, Bovbjerg DH. Psychological stress and antibody response to influenza vaccination: a meta-analysis. Brain Behav Immun. 2009;23:427–433. doi: 10.1016/j.bbi.2009.01.004. [DOI] [PubMed] [Google Scholar]
- 10.Carlsson E, Frostell A, Ludvigsson J, Faresjo M. Psychological stress in children may alter the immune response. J Immunol. 2014;192:2071–81. doi: 10.4049/jimmunol.1301713. [DOI] [PubMed] [Google Scholar]
- 11.Copeland WE, Wolke D, Lereya ST, Shanahan L, Worthman C, Costello EJ. Childhood bullying involvement predicts low-grade systemic inflammation into adulthood. Proc Natl Acad Sci USA. 2014;111:7570–7575. doi: 10.1073/pnas.1323641111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- **12.Slopen N, McLaughlin KA, Dunn EC, Koenen KC. Childhood adversity and cell-mediated immunity in young adulthood: does type and timing matter? Brain Behav Immun. 2013;28:63–71. doi: 10.1016/j.bbi.2012.10.018. In this work, the authors examine how adverse experiences earlier in the life span correspond with EBV antibody titers in young adulthood. This work has several notable strengths, including investigating the immune response to latent viruses, assessing experiences of both socioeconomic disadvantage and abuse, and incorporating frequency as well as timing of adversity exposure. This paper is an important read for those interested in how stressor characteristics influence immune system outcomes. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Coelho R, Viola TW, Walss-Bass C, Brietzke E, Grassi-Oliveira R. Childhood maltreatment and inflammatory markers: a systematic review. Acta Psychiatr Scand. 2014;129:180–92. doi: 10.1111/acps.12217. [DOI] [PubMed] [Google Scholar]
- 14.Hostinar CE, Ross KM, Chen E, Miller GE. Modeling the association between lifecourse socioeconomic disadvantage and systemic inflammation in healthy adults: the role of self-control. Health Psychol. 2014 doi: 10.1037/hea0000130. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Raposa EB, Bower JE, Hammen CL, Najman JM, Brennan PA. A developmental pathway from early life stress to inflammation: the role of negative health behaviors. Psychol Sci. 2014;25:1268–1274. doi: 10.1177/0956797614530570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fagundes CP, Way B. Early-life stress and adult inflammation. Curr Dir Psychol. 2014;23:277–283. In this relatively short but authoritative review, the authors provide support for inflammatory processes as a mechanism through which early life stress impacts physical health in adulthood. Literature linking low socio-economic status as well as maltreatment with inflammation is reviewed, and mechanisms for this linkage are discussed. The authors also suggest policy implications centering on the reduction of poverty and maltreatment to reduce adult disease prevalence and its associated costs. Readers interested in an accessible yet thorough review of early adversity's impact on the immune system will find this work beneficial. [Google Scholar]
- *17.Miller GE, Brody GH, Yu T, Chen E. A family-oriented psychosocial intervention reduces inflammation in low-SES African American youth. Proc Natl Acad Sci USA. 2014;111:11287–11292. doi: 10.1073/pnas.1406578111. In this paper, the authors describe results of an intervention designed to improve parenting and family relationships in a sample of low-SES, rural African-American 11-year-olds and their mothers. They found that youth from families who received the intervention exhibited lower levels of inflammatory markers (e.g., IL-6) at an 8-year follow-up visit than youth from families who did not receive the intervention, in part due to improved parenting within the intervention group. This work is highly important as it suggests that, with intervention at the family level, it may be possible to reduce the enduring physiological impact of early hardship. [DOI] [PMC free article] [PubMed] [Google Scholar]
- **18.Vitlic A, Lord JM, Philips AC. Stress, ageing and their influence on functional, cellular and molecular aspects of the immune system. Age. 2014;36:1169–1185. doi: 10.1007/s11357-014-9631-6. In this compelling article, the authors provide a definitive review of the effects aging, stress, and their interplay have on the robustness of the human immune response. The paper begins with a brief review of the stress and immune literature and adds in a compelling discussion of the beneficial and detrimental effects of both acute and chronic stress. Lastly, the review ends by thoroughly outlining the effects of aging on the immune system at the systems and molecular level. This review is a must read for those wanting a full but relatively brief look at the influences of aging on different aspects of the immune system. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wong SY, Wong CK, Chan FW, Chan PK, Ngai K, Mercer S, Woo J. Chronic psychosocial stress: does it modulate immunity to the influenza vaccine in Hong Kong Chinese elderly caregivers? Age. 2012;35:1479–1493. doi: 10.1007/s11357-012-9449-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Segerstrom SC, Schipper MA, Greenberg RN. Caregiving, repetitive thought, and immune response to vaccination in older adults. Brain Behav Immun. 2008;22:744–752. doi: 10.1016/j.bbi.2007.11.004. Currently, this may be the best study outlining the detrimental effects of chronic stress associated with caregiving on vaccine responses. The authors show through antibody titers and pre/post vaccine serum IL-6 levels that individual differences in repetitive thoughts can alter the way the body responds to vaccination. This article is an essential read for those interested in repetitive thoughts and immune system functioning. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kiecolt-Glaser JK, Glaser R, Christian LM. Omega-3 fatty acids and stress-induced immune dysregulation: implications for wound healing. Mil Med. 2014;179:129–133. doi: 10.7205/MILMED-D-14-00167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yen YC, Lung FW. Older adults with higher incomes or marriage have longer telomeres. Age Ageing. 2013;42:234–239. doi: 10.1093/ageing/afs122. Leukocyte telomere length has been used as a proxy for biological aging and is associated with many social, physical, and psychological factors. In this article, Yen and Lung present clear evidence that sociodemographic factors can actually alter the speed at which we age. In their study, a representative sample of older adults randomly selected from the census reported on their marital status and yearly income and had their leukocyte telomere length measured. The authors effectively show that older adults who are married and have higher income also have longer telomere length- indicating the large importance of these factors on our health and longevity. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Carroll JE, Diez-Roux AV, Adler NE, Seeman TE. Socioeconomic factors and leukocyte telomere length in a multi-ethnic sample: findings from the multi-ethnic study of artherosclerosis (MESA). Brain Behav Immun. 2013;28:108–114. doi: 10.1016/j.bbi.2012.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Puterman E, Lin J, Krauss J, Blackburn EH, Epel ES. Determinants of telomere attrition over 1 year in healthy older women: stress and health behaviors matter. Mol Psychiatry. 2014:1–7. doi: 10.1038/mp.2014.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dhabhar FS, Malarkey WB, Neri E, McEwen BS. Stress-induced redistribution of immune cells—From barracks to boulevards to battlefields: a tale of three hormones–Curt Richter Award Winner. Psychoneuroendocrinology. 2012;37:1345–1368. doi: 10.1016/j.psyneuen.2012.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rohleder N. Acute and chronic stress induced changes in sensitivity of peripheral inflammatory pathways to the signals of multiple stress systems–2011 Curt Richter Award Winner. Psychoneuroendocrinology. 2012;37:307–316. doi: 10.1016/j.psyneuen.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 27.Jaremka LM, Lindgren ME, Kiecolt-Glaser JK. Synergistic relationships among stress, depression, and troubled relationships: insights from psychoneuroimmunology. Depress Anxiety. 2013;30:288–296. doi: 10.1002/da.22078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chiang JJ, Eisenberger NI, Seeman TE, Taylor SE. Negative and competitive social interactions are related to heightened proinflammatory cytokine activity. Proc Natl Acad Sci USA. 2012;109:1878–1882. doi: 10.1073/pnas.1120972109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Uchino BN, Bowen K, Carlisle M, Birmingham W. Psychological pathways linking social support to health outcomes: a visit with the “ghosts” of research past, present, and future. Soc Sci Med. 2012;74:949–957. doi: 10.1016/j.socscimed.2011.11.023. In this paper, the authors provide an authoritative review of the literature linking social support to health outcomes. They argue that the mechanisms linking social support to health are poorly understood because of statistical and design issues and poor conceptualization in studies linking social support to health, among other reasons. The authors conclude by suggesting how future research in the area could be improved. This paper is an important read for those interested in how social support influences health outcomes. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Besedovsky L, Lange T, Born J. Sleep and immune function. Pflugers Arch. 2012;463:121–137. doi: 10.1007/s00424-011-1044-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ruiz FS, Andersen ML, Martins RC, Zager A, Lopes JD, Tufik S. Immune alterations after selective rapid eye movement or total sleep deprivation in healthy male volunteers. Innate Immun. 2012;18:44–54. doi: 10.1177/1753425910385962. [DOI] [PubMed] [Google Scholar]
- 32.Bonneaud C, Mazuc J, Gonzalez G, Haussy C, Chastel O, Faivre B, Sorci G. Assessing the cost of mounting an immune response. Am Nat. 2003;161:367–379. doi: 10.1086/346134. [DOI] [PubMed] [Google Scholar]
- 33.Segerstrom SC. Resources, stress, and immunity: an ecological perspective on human psychoneuroimmunology. Ann Behav Med. 2010;40:114–125. doi: 10.1007/s12160-010-9195-3. [DOI] [PubMed] [Google Scholar]
- 34.Straub RH, Cutolo M, Buttgereit F, Pongratz G. Energy regulation and neuroendocrine–immune control in chronic inflammatory diseases. J Intern Med. 2010;267:543–560. doi: 10.1111/j.1365-2796.2010.02218.x. [DOI] [PubMed] [Google Scholar]
- 35.Moret Y, Schmid-Hempel P. Survival for immunity: the price of immune system activation for bumblebee workers. Science. 2000;290:1166–1168. doi: 10.1126/science.290.5494.1166. [DOI] [PubMed] [Google Scholar]
- 36.Hamrick N, Cohen S, Rodriguez MS. Being popular can be healthy or unhealthy: stress, social network diversity, and incidence of upper respiratory infection. Health Psychol. 2002;21:294–298. [PubMed] [Google Scholar]
- 37.Segerstrom SC. Optimism and immunity: do positive thoughts always lead to positive effects? Brain Behav Immun. 2005;19:195–200. doi: 10.1016/j.bbi.2004.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- **38.Demas GE, Carlton ED. Ecoimmunology for psychoneuroimmunologists: considering context in neuroendocrine–immune–behavior interactions. Brain Behav Immun. 2015;44:9–16. doi: 10.1016/j.bbi.2014.09.002. Drawing from a wide array of disciplines, the paper makes a compelling case that the adaptiveness or maladaptiveness of immunological functions are dependent on the energetic resource availability in a specific ecological context. The ideas from this paper have the power to change the way researchers think about what truly makes an immunological response adaptive. [DOI] [PMC free article] [PubMed] [Google Scholar]
- *39.Straub RH. Systemic disease sequelae in chronic inflammatory diseases and chronic psychological stress: comparison and pathophysiological model. Ann NY Acad Sci. 2014;1318:7–17. doi: 10.1111/nyas.12409. Straub provides an excellent review of how chronic psychological stress may predict long term disease sequelae in a manner similar to that of chronic inflammatory diseases. He also effectively describes the slight differences in these processes, namely the fact that chronic psychological stress does induce brain inflammation, but it is not strong enough to explain disease sequelae. He introduces the term the “selfish immune system,” stemming from its substantial requests for energy independent of the body, as well as a new pathophysiological theory that places inflammation and stress axes in the middle of the brain vs. body battle. [DOI] [PubMed] [Google Scholar]
- 40.de Brouwer SJ, van Middendorp H, Kraaimaat FW, Radstake TR, Joosten I, Donders AR, Eijsbouts A, Spillekom-van Koulil S, van Riel PL, Evers AW. Immune responses to stress after stress management training in patients with rheumatoid arthritis. Arthritis Res Ther. 2013;15:R200. doi: 10.1186/ar4390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kennedy PJ, Cryan JF, Quigley EMM, Dinan TG, Clarke G. A sustained hypothalamic-pituitary-adrenal axis response to acute psychosocial stress in irritable bowel syndrome. Psychol Med. 2014;44:3123–3134. doi: 10.1017/S003329171400052X. [DOI] [PubMed] [Google Scholar]
- 42.Fineberg AM, Ellman LM. Inflammatory cytokines and neurological and neurocognitive alterations in the course of schizophrenia. Biol Psychiatry. 2013;73:951–966. doi: 10.1016/j.biopsych.2013.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dhabhar FS. Effects of stress on immune function: the good, the bad, and the beautiful. Immunol Res. 2014;58:193–210. doi: 10.1007/s12026-014-8517-0. [DOI] [PubMed] [Google Scholar]
- 44.Karagkouni A, Alevizos M, Theoharides TC. Effect of stress on brain inflammation and multiple sclerosis. Autoimmun Rev. 2013;12:947–953. doi: 10.1016/j.autrev.2013.02.006. [DOI] [PubMed] [Google Scholar]