Abstract
Barley varieties have similar chemical composition but exhibit different rumen degradation kinetics and nutrient availability. These biological differences may be related to molecular, structural, and chemical makeup among the seed endosperm tissue. No detailed study was carried out. The objectives of this study were: (1) to use a molecular spectroscopy technique, synchrotron-based Fourier transform infrared microspectroscopy (SFTIRM), to determine the microchemical–structural features in seed endosperm tissue of six developed barley varieties; (2) to study the relationship among molecular–structural characteristics, degradation kinetics, and nutrient availability in six genotypes of barley. The results showed that inherent microchemical–structural differences in the endosperm among the six barley varieties were detected by the synchrotron-based analytical technique, SFTIRM, with the univariate molecular spectral analysis. The SFTIRM spectral profiles differed (P < 0.05) among the barley samples in terms of the peak ratio and peak area and height intensities of amides I (ca. 1650 cm−1) and II (ca. 1550 cm−1), cellulosic compounds (ca. 1240 cm−1), CHO component peaks (the first peak at the region ca. 1184–1132 cm−1, the second peak at ca. 1132–1066 cm−1, and the third peak at ca. 1066–950 cm−1). With the SFTIRM technique, the structural characteristics of the cereal seeds were illuminated among different cultivars at an ultraspatial resolution. The structural differences of barley seeds may be one reason for the various digestive behaviors and nutritive values in ruminants. The results show weak correlations between the functional groups’ spectral data (peak area, height intensities, and ratios) and rumen biodegradation kinetics (rate and extent of nutrient degradation). Weak correlations may indicate that limited variations of these six barley varieties might not be sufficient to interpret the relationship between spectroscopic information and the nutrient value of barley grain, although significant differences in biodegradation kinetics were observed. In conclusion, the studies demonstrated the potential of ultraspatially resolved synchrotron based technology (SFTIRM) to reveal the structural and chemical makeup within cellular and subcellular dimensions without destruction of the inherent structure of cereal grain tissue.
Keywords: Synchrotron, degradation kinetics, microchemical structure, endosperm
INTRODUCTION
Barley varieties exhibit different ruminal degradation rates and kinetics features (1,2). Knowledge of their chemical and structural differences may lead to an understanding of the reasons for these differences in degradation kinetics. Further study of the relationship of biological characteristics (in terms of biodegradation kinetics) of barley grain in relation to spectral–structural characteristics may provide useful understanding of how to best utilize the various barley varieties.
In conventional chemical analysis, harsh chemical reagents can unavoidably destroy or even obliterate the histological structure of the biological sample (3–5). The detection of inherent structural and chemical information is beyond the capacity of traditional chemical analysis, owing to the destructive pretreatment and the treatment during chemical analysis. These disadvantages necessitate a new and rapid tool to overcome the limitations.
Conventional (globar-sourced) FTIR microspectroscopy shows comparable performance to synchrotron only when working with large aperture size (>30–50 μm) (5, 6). However, it is inferior to synchrotron at an ultraspatial resolution when detecting biological or biomedical specimens (5–20 μm) (7–10). To overcome the weakness of conventional (thermal) light source, brilliant synchrotron light (which is million times brighter than sun light) is used because it is extremely advantageous for small aperture settings (11).
Synchrotron radiation allows simultaneous sample viewing and IR data collection at ultraspatial resolution. Using the synchrotron source to substitute for a conventional thermal (globar) source results in the potential to improve the S/N ratio and reach the diffraction limit (8, 11–13). The combining of microscopy with FTIR spectroscopy enables observations both visually and spectroscopically. Ultraspatially resolved synchrotron FTIR microspectroscopy is used to monitor the intrinsic distribution of chemical compounds in biological and biomedical specimens (14). Accordingly, taking advantage of the brilliance, broadband, and concentration (11), the application of synchrotron light associated with the FTIR technique makes it possible to probe further on the inherent structure of biological materials at a cellular or subcellular dimension without thermal noise (15).
The synchrotron-based FTIRM technique may enable us to establish a correlation of spectral information to structural and chemical features of plant/seed tissues, furthermore, to feed/food quality. The objectives of this study were (1) to use synchrotron-based FTIR microspectroscopy to determine structural makeup features and identify the structural differences in chemical functional groups in endosperm tissue within a cellular dimension among the six developed barley varieties; (2) to analyze the correlation between biodegradation kinetics (rate and extent of rumen degradation) and chemical functional group features determined by the synchrotron-based FTIR microspectroscopy.
MATERIALS AND METHODS
Barley Varieties and Growth Conditions
Six barley varieties were used in this study, including AC Metcalfe (for malting purposes), CDC Dolly (for feed purposes), McLeod (for feed purposes), CDC Helgason (for feed purposes), CDC Trey (for feed purposes), and CDC Cowboy (for feed and forage dual purposes). All are two-row hulled spring barley varieties. AC Metcalfe has a higher yield and larger and more plump seeds than Harrington. CDC Dolly is widely grown in Canada for its high test weight and plumpness. McLeod is developed with good yield and higher test and kernel weight than Harrington. CDC Helgason has similar plumpness but a higher yield and test weight compared to those of CDC Dolly. CDC Trey is suitable for growing in the eastern Canada prairies with large, plump seeds. CDC Cowboy has high biomass but somewhat lower grain yield (Rossnagel, B. G., personal communication (16, 17)).
The year of the barley grain harvest was 2005. The climate conditions during the year were 17.5 °C mean maximum daily temperature and 455 mm rainfall. Barley samples were obtained from Crop Development Center (CDC), University of Saskatchewan. All barley varieties were grown without irrigation at Kernen Crop Research Farm, University of Saskatchewan, Canada, following standard agronomic production practices for barley production.
In situ rumen degradation kinetics (degradation rate and extent) were significantly different among these barley varieties, as presented in Table 1. These data were used for the correction study between the degradation rate and extent and spectral structural features at the endosperm region in the barley varieties, revealed by the ultraspatially resolved synchrotron-based FTIR microspectroscopy within a cellular dimension.
Table 1.
Comparison of Six Barley Varieties: Chemical Profile and in Situ Rumen Degradation Kinetics of Dry Matter (DM), Crude Protein, and Starch (ST)
| items | barley varietya
|
SEMb | P value | |||||
|---|---|---|---|---|---|---|---|---|
| AC Metcalfe | CDC Dolly | McLeod | CDC Helgason | CDC Trey | CDC Cowboy | |||
| in situ rumen degradation kinetics of DMc | ||||||||
| Kd (%/h) | 10.04 a | 7.84 b | 9.63 ab | 5.62 c | 9.72 a | 8.29 ab | 0.671 | 0.0025 |
| EDDM (%) | 48.66 a | 39.49 c | 45.00 b | 35.98 d | 49.80 a | 45.46 b | 0.999 | <0.0001 |
| in situ rumen degradation kinetics of CPc | ||||||||
| Kd (%/h) | 6.72 a | 6.18 ab | 6.99 a | 4.55 b | 6.95 a | 5.73 ab | 0.551 | 0.0560 |
| EDCP (%CP) | 44.69 a | 39.04 bc | 42.56 ab | 36.41 c | 45.30 a | 42.17 ab | 1.250 | 0.0022 |
| in situ rumen degradation kinetics of starch (ST)c | ||||||||
| Kd (%/h) | 7.05 a | 4.24 d | 5.28 c | 3.67 e | 7.11 a | 6.16 b | 0.176 | <0.0001 |
| EDST (%ST) | 55.04 a | 41.67 d | 46.79 c | 38.99 e | 54.27 a | 50.69 b | 0.870 | <0.0001 |
All six varieties of barley were grown at the Kernen Crop Research Farm (University of Saskatchewan, Saskatoon, Canada) and were managed using the same and standard agronomic production practices for all barley production (Crop Development Center).
SEM = standard error of mean. Means with different letters in the same row are significantly different (P < 0.05).
Kd = degradation rate; EDDM, EDCP, and EDST = effective degradability of dry matter, crude protein, and starch, respectively.
Sample Preparation for Synchrotron-Based FTIR Microspectroscopy
Five seeds of each barley variety (six varieties) were randomly selected to cut transversely across the endosperm tissue using amicrotome at Western College of Veterinary, University of Saskatchewan, Saskatoon, Canada. The thin cross-sections of tissues (6 μm) were unstained and mounted on barium fluoride (BaF2) discs (2 mm thick, 13 mm diameter; Spectral Systems, Hopewell Junction, NY, USA) for synchrotron FTIR microspectroscopic work in transmission mode. The detailed methodology was reported by Yu et al. (14, 18).
Synchrotron Radiation Sources and Synchrotron FTIR Microspectroscopy
The synchrotron FTIRM experiment was performed using a Thermo Nicolet Magna 860 Step-Scan FTIR (Thermo Fisher Scientific Inc., Waltham, MA) spectrometer equipped with a Spectra Tech Continuum IR Microscope (Spectra-Tech, Inc., Shelton, CT) and liquid nitrogen-cooled mercury cadmium telluride (MCT) detector. The IR microspectroscopy instrument was coupled with synchrotron radiation from U2B beamline, National Synchrotron Light Source, Brookhaven National Laboratory, U.S. Department of Energy (NSLS-BNL, Upton, NY).
Synchrotron FTIRM Data Collection and Processing
The spectra were collected from five seeds of each barley variety. Spot samples (30–60) were randomly selected in the endosperm region between 100 and 600 μm from outside of the seed section on each window according to the method reported by Walker et al. (19). The spectra were collected through an aperture of 10×10 μmin transmission mode within mid-IR spectral range of ca. 4000–800 cm−1. The spatial resolution was set as 4 cm−1, and 128 scans were co-added to each spot to produce an IR spectrum. Background spectra were collected using the same measurement setting. Atlus software (Thermo Nicolet, Madison, WI, USA) was used to produce the visual image of the sample. Nicolet OMNIC software 7.3 (Spectra Tech, Madison, WI, USA) was used to collect and analyze spectral data. After baseline correction, the absorption peak parameters (baseline, region, relative height, and area)were recorded for further univariate analysis (13). More details on SFTIRM data collection were documented in Marinkovic et al. (8) and Yu et al. (20).
Statistical Analysis
Statistical analyses were performed using the MIXED procedure of SAS 9.1.3 (SAS Institute, Inc., Cary, NC). Fisher’s protected test was used to compare means with P<0.05, which were considered significant.
The synchrotron FTIRM data were analyzed using a completed nested design. The model used for the analysis was: Yij = μ + Ti + S(T)j + eij, where Yij was an observation of the dependent variable ij; μ was the population mean for the variable; Ti was the effect of the barley varieties, as a fixed effect; S(T)j was the seeds nested within treatments, as a random effect; and eij was the random error associated with the observation ij. The detailed methodology was reported in Yu (9) and Doiron et al. (21, 22).
The correlation between the degradation rate and extent of dry matter (DM), crude protein (CP), starch (ST), and synchrotron FTIRM spectral characteristics among the six barley varieties was analyzed using the CORR procedure of SAS software.
RESULTS AND DISCUSSION
Using Synchrotron-Based FTIR Microspectroscopy to Characterize and Compare Microchemical–Structural Makeup of Seed Endosperm Tissue within a Cellular Dimension among Six Barley Varieties
The synchrotron FTIRM technique was used to characterize inherent seed microchemical structure (23) among these six barley varieties. The spectra observed in the region of ca. 4000–800 cm−1 were analyzed to show the absorption characteristics attributed to the chemical functional groups of barley grains.
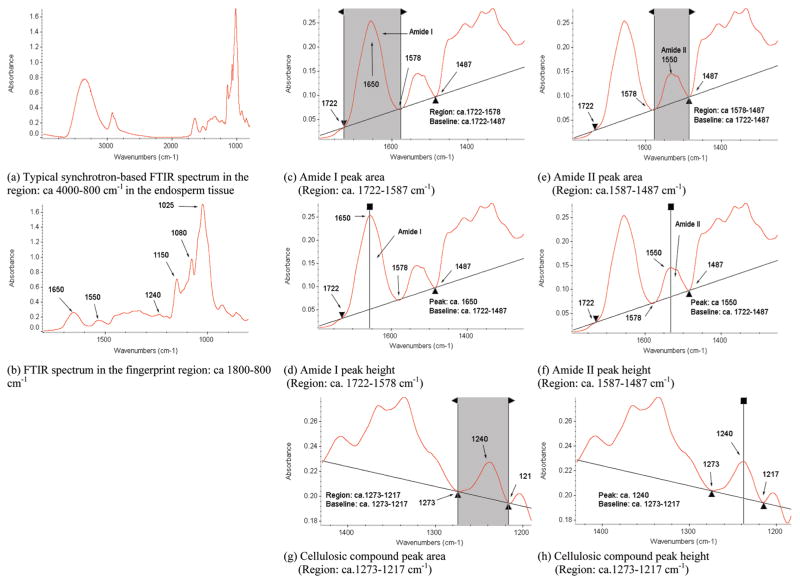
For synchrotron FTIRM, the barley seed specimens were optically observed with the coupled microscope, and spot sampling was done randomly on endosperm tissue (between 100 and 600 μm from outside of the seed section) of each barley variety to produce the spectral data. Figure 1a illustrates the typical SFTIRM spectrum of the endosperm tissue in the region of ca. 4000–800 cm−1. The IR spectra of characteristic bands associated with specific nutrients (protein, cellulosic compounds in Figure 1, and carbohydrates in Figure 2) are shown as well.
Figure 1.
Typical synchrotron-based FTIR spectrum of endosperm tissue within a cellular dimension. (a) Whole mid-IR region: ca. 4000–800 cm−1. (b) Fingerprint region: ca. 1800–800 cm−1. (c,d) Amide I peak area and height: ca. 1650 cm−1. (e,f) Amide II peak area and height: ca. 1550 cm−1. (g,h) Cellulosic compounds peak area and height: ca. 1240 cm−1.
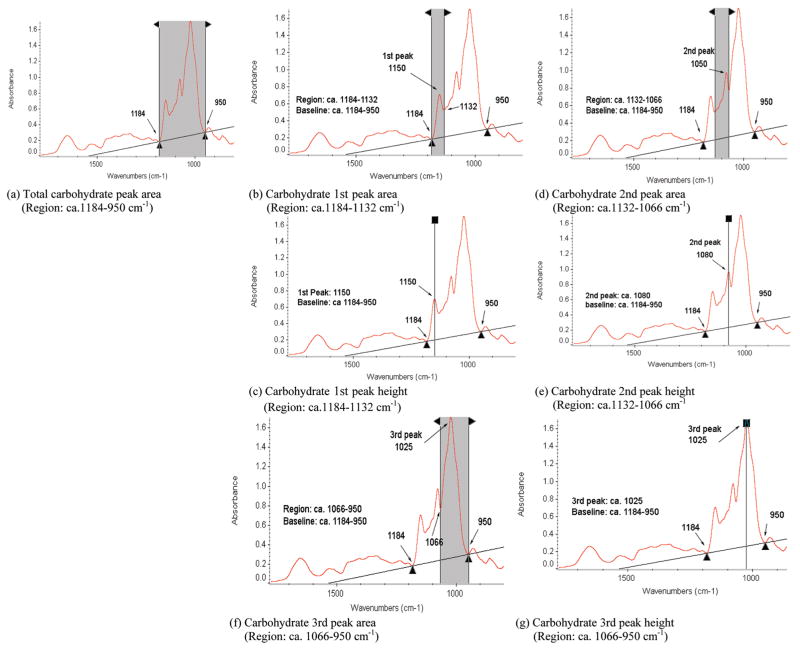
Figure 2.
Typical synchrotron-based FTIR spectrum of endosperm tissue within a cellular dimension. (a) Total carbohydrate peak area region: ca. 1184– 950 cm−1. (b,c) Carbohydrate first component peak area and height: ca. 1150 cm−1. (d,e) Carbohydrate second component peak area and height: ca. 1080 cm−1. (f,g) Carbohydrate third component peak area and height: ca. 1025 cm−1.
The characteristic absorbance bands of the amide groups can be used to probe and characterize the structural and chemical makeup of the plant tissue. The amide I (ca. 1650 cm−1) and amide II (ca. 1550 cm−1) peak area and height definition used for the spectral univariate analysis are shown in Figure 1c,d,e,f. The peak centered at approximately 1240 cm−1 (Figure 1g,h) was considered to represent the structural carbohydrates (cellulosic compounds) (12, 24, 25). The total carbohydrate peak area is located at the region from ca. 1184 to 951 cm−1 because of C–O and C–C stretching vibrations and C–O–H deformation (12, 24, 25). The CHO band intensities were determined by calculating the peak heights and areas of specific bands: the carbohydrate first component peak area occurs at the region of ca. 1184–1132 cm−1, the carbohydrate second component peak area occurs at the region of ca. 1132–1066 cm−1, and the carbohydrate third peak area occurs at the region of ca. 1066– 950 cm−1. These three peaks are regarded to represent total carbohydrate compounds (Figure 2).
To detect the structural features associated with nutrients, synchrotron-based FTIR microspectroscopy was used to examine structure features of barley endosperm tissue. Tables 2 and 3 show spectroscopic data from the spot samples within cellular dimensions. It needs to bementioned that the peak intensities (e.g., amide I area and height) should not be considered as a representation of accurate biological compound content (e.g., starch and protein).
Table 2.
Structural Characteristics of Protein Amides I and II and Carbohydrates, and Structural Carbohydrate (Cellulosic Compounds) in the Endosperm Tissue of Barley Varieties, Revealed Using Synchrotron-Based FTIR Microspectroscopy: Comparison of Six Barley Varieties
| items | peak (cm−1) | region (cm−1) | baseline (cm−1) | characteristics of whole barley seed in terms of the mid-IR absorbed peak area, height, and their ratios (infrared absorbed intensity unit)
|
SEMa | P value | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| AC Metcalfe | CDC Dolly | McLeod | CDC Helgason | CDC Trey | CDC Cowboy | ||||||
| amide molecular spectral features | |||||||||||
| based on the amide I and II peak area, and height | |||||||||||
| amide I peak area | ~1650 | 1722–1578 | 1722–1487 | 7.99 bc | 8.23 abc | 10.02 a | 7.33 c | 9.01 ab | 9.23 ab | 0.582 | 0.004 |
| amide I peak height | ~1650 | 1722–1578 | 1722–1487 | 0.11 cd | 0.12 bcd | 0.16 a | 0.11 d | 0.14 ab | 0.13 bc | 0.091 | <0.0001 |
| amide II peak area | ~1550 | 1578–1487 | 1722–1487 | 1.94 b | 1.97 abc | 2.57 a | 1.86 b | 2.20 ab | 2.34 a | 0.182 | 0.024 |
| amide II peak height | ~1550 | 1578–1487 | 1722–1487 | 0.04 b | 0.04 ab | 0.05 a | 0.04 b | 0.04 ab | 0.04 ab | 0.004 | 0.133 |
| amide I and II area | 1722–1487 | 1722–1487 | 9.93 bc | 10.20 abc | 12.59 a | 9.19 c | 11.22 ab | 11.56 a | 0.754 | 0.005 | |
| carbohydrate (CHO) molecular spectral features | |||||||||||
| based on the carbohydrate peak area and height | |||||||||||
| cellulosic compound area | ~1240 | 1273–1217 | 1273–1217 | 0.54 a | 0.53 ab | 0.48 b | 0.54 a | 0.47 b | 0.49 b | 0.020 | 0.003 |
| cellulosic compound height | ~1240 | 1273–1217 | 1273–1217 | 0.02 ab | 0.02 a | 0.02 b | 0.02 ab | 0.02 b | 0.02 b | 0.002 | 0.121 |
| total carbohydrate peak area | 1184–951 | 1184–951 | 71.10 a | 75.20 a | 62.68 b | 76.71 a | 72.30 a | 72.31 a | 2.889 | 0.022 | |
| CHO: 1st component peak area | ~1150 | 1184–1132 | 1184–951 | 8.25 b | 8.67 ab | 7.03 c | 9.02 a | 8.14 b | 8.19 b | 0.374 | 0.006 |
| CHO: 1st component peak height | ~1150 | 1184–1132 | 1184–951 | 0.29 a | 0.30 ab | 0.24 b | 0.27 ab | 0.28 a | 0.28 a | 0.012 | 0.048 |
| CHO: 2nd component peak area | ~1080 | 1132–1066 | 1184–951 | 10.41 ab | 11.24 a | 9.54 b | 11.14 a | 10.74 ab | 10.64 ab | 0.460 | 0.175 |
| CHO: 2nd component peak height | ~1080 | 1132–1066 | 1184–951 | 0.42 ab | 0.45 a | 0.38 b | 0.44 a | 0.42 ab | 0.42 ab | 0.017 | 0.144 |
| CHO: 3rd component peak area | ~1025 | 1066–951 | 1184–951 | 44.92 b | 47.38 ab | 39.59 c | 48.66 a | 46.08 ab | 46.30 ab | 1.791 | 0.014 |
| CHO: 3rd component peak height | ~1025 | 1066–951 | 1184–951 | 0.66 ab | 0.69 ab | 0.59 b | 0.72 a | 0.72 a | 0.72 a | 0.029 | 0.014 |
SEM = pooled standard error of means. Means with different letters in the same row are significantly different (P < 0.05).
Table 3.
Structural Characteristics of the Ratios of Amides I and II, and Structural (Cellulosic Compounds) and Nonstructural (NSC, Starch) Carbohydrates (CHO) in the Endosperm Tissue of Barley Varieties, Revealed Using Synchrotron-Based FTIR Microspectroscopy: Comparison of Six Barley Varieties
| items | peaks area regions (baseline) (cm−1) | molecular characteristics of barley seed in terms of the mid-IR absorbed peak area and height and their ratios (IR absorbed intensity unit)
|
SEMa | P value | |||||
|---|---|---|---|---|---|---|---|---|---|
| AC Metcalfe | CDC Dolly | McLeod | CDC Helgason | CDC Trey | CDC Cowboy | ||||
| based on the amide I and CHO peak area and height ratios | |||||||||
| ratio of total CHO peak area/amides I and II peak area | 1184–951 (baseline: 1184–951)/1722–1487 (baseline 1722–1487) | 8.90 b | 9.15 abc | 7.11 c | 9.89 ab | 10.81 a | 9.49 ab | 0.618 | 0.002 |
| ratio of NSC (starch) peak area (CHO 3rd peak)/amides I peak area | 1066–951 (baseline: 1184–951)/1722–1487 (baseline 1722–1487) | 6.91 b | 6.86 abc | 5.35 c | 7.68 ab | 8.25 a | 7.58 ab | 0.505 | 0.003 |
| ratio of NSC (starch) peak height (CHO 3rd peak)/amide I height | ~1025 (baseline: 1184–951)/~1650 (baseline: 1722–1487) | 6.91 bc | 7.24 bc | 5.16 c | 7.90 b | 8.41 ab | 9.97 a | 0.789 | <0.001 |
| ratio of amides I and II peak area/cellulosic compound peak area | 1722–1487 (baseline: 1722–1487)/1273–1217 (baseline: 1273–1217) | 19.41 b | 20.98 ab | 26.44 a | 17.99 b | 23.64 a | 24.29 a | 1.568 | <0.0001 |
| ratio of amide I peak area/cellulosic compound peak area | 1722–1587 (baseline: 1722–1487)/1273–1217 (baseline: 1273–1217) | 15.44 b | 16.92 ab | 21.03 a | 14.27 b | 18.96 a | 19.36 a | 1.190 | <0.0001 |
| ratio of amide I peak height/cellulosic compound peak height | ~1650 (baseline: 1722–1487)/1273–1217 (baseline: 1273–1217) | 5.90 c | 6.36 bc | 9.77 a | 5.67 c | 7.87 b | 7.67 b | 0.501 | <0.0001 |
| ratio of total CHO peak area/cellulosic compound peak area | 1184–951 (baseline: 1184–951)/1273–1217 (baseline: 1273–1217) | 137.14 bc | 144.80 abc | 130.89 c | 144.99 abc | 157.57 a | 148.60 ab | 5.963 | 0.022 |
| ratio of NSC (starch) peak area (CHO 3rd peak)/cellulosic peak area | 1066–951 (baseline: 1184–951)//1273–1217 (baseline: 1273–1217) | 87.08 bc | 91.26 abc | 82.65 c | 92.71 abc | 101.24 a | 95.32 ab | 3.936 | 0.011 |
| ratio of NSC (starch) peak height (CHO 3rd peak)/cellulosic peak height | ~1025 (baseline: 1184–951)/1273–1217 (baseline: 1273–1217) | 34.56 d | 36.47 cd | 35.82 cd | 38.45 bc | 42.47 a | 40.93 ab | 1.271 | <0.0001 |
SEM = pooled standard error of means. Means with different letters in the same row are significantly different (P < 0.05).
Structural Characteristics of Protein Amides I and II in Barley Endosperm
Table 2 shows that McLeod had the largest (P<0.05) amide I peak area (10.02 IR absorbed intensity unit), indicating a greater concentration of protein in the endosperm tissue. In contrast, CDC Helgason showed the lowest value (7.33). The other four barley varieties were in between. Six barley varieties differed (P<0.05) in amide I peak height and varied from 0.11 to 0.16. McLeod exhibited the highest amide II peak area and height (2.57 and 0.05, respectively), implying a greater concentration of protein in the endosperm tissue. By analyzing the peak of the total amides I and II areas (Table 2), McLeod was still the greatest (12.59), while CDC Helgason was the smallest (9.19). This may partially explain the lowest effective degradability of CP of CDC Helgason among the six barley varieties (Table 1). If the six barley varieties are ranked on the basis of their amide group intensities, all of the amide group parameters (amide I, amide II, and total amide I and II) show a similar trend that CDC Helgason exhibited the smallest value, whereas McLeod showed the greatest peak intensity. As for the remaining four, AC Metcalfe and CDC Dolly had relatively smaller values, and CDC Trey and CDC Cowboy were relatively higher. These results agree with the previous work that McLeod was developed as a high protein content barley (26, 27). The protein can play a protector role when it combined with structural and nonstructural carbohydrates (e.g., starch granule) to form a protein matrix, which is related with barley rumen degradability and total tract digestibility.
Structural Characteristics of Carbohydrates and Structural Carbohydrate in Barley Endosperm
Table 2 shows the absorbance peak area and height intensities of the nonstructural and structural carbohydrates (cellulosic compounds) in the endosperm tissue of the six barley varieties. There were different peak intensities found in the region ca. 1184–951 cm−1, revealing the various CHO contents of the endosperm tissue of barley varieties. CDC Trey had the relatively smallest peak area (0.47) at ca. 1240 cm−1 (cellulosic compounds band) (12). McLeod had the lowest (P<0.05) value in total CHO peak area (62.68), and CDC Helgason had the highest total carbohydrate peak area (76.71).
The three characteristic absorption peaks that fell in the CHO region (ca. 1184–951 cm−1) (Figure 2) were analyzed, and similar trends was observed. McLeod had the smallest (P<0.01) area of the first CHO peak (7.03), in contrast to CDC Dolly and CDC Helgason (8.67 and 9.02, respectively). Compared to the other varieties, McLeod and CDC Helgason exhibited relatively lower (P<0.05) peak heights of the first CHO peak (0.24 and 0.27, respectively). There was no difference (P>0.05) in the second CHO peak intensity shown here. At ca. 1066–951 cm−1, there was an absorption band, which was assigned to nonstructural starch. The peak maxima appeared at ca. 1025 cm−1 (12). Consistent with other CHO peak parameters, McLeod had the smallest (P < 0.05) third CHO peak area and relatively smaller peak height (0.59), suggesting the lowest enrichment of starch in endosperm tissue among the six barley varieties. Starch is abundant in endosperm tissue with different granule size, type, and content (28, 29). These variable molecular characteristics of barley endosperm tissue may have a strong influence on ruminal fermentation and degradation.
Structural Characteristics of the Ratios of Amides I and II, and Structural and Nonstructural Carbohydrates (CHO) in Barley Endosperm
In order to further understand the structural and chemical makeup of the barley endosperm, relative peak intensity ratios of specific bands associated with nutrients were calculated and presented in Table 3. The ratio analysis was calculated using the infrared absorbance intensity (area or height) of one characteristic peak observed in the spectrum to divide that of another one. The results show that McLeod had the relatively smaller ratio of total CHO peak area to amides I and II peak area (7.11). The ratios of nonstructural carbohydrate starch (ca. 1205 cm−1) to amide I (at 1650 cm−1) were significantly different among the six barley varieties. McLeod showed a relatively smaller value for both the area and height ratio comparisons (5.35 and 5.16, respectively). According to the chemical composition analysis, McLeod has relatively more protein and less CHO content, which may be a reason for the smallest CHO/amide ratio. Varlier, feed-type barley variety, was found to have a lower starch/protein ratio in an earlier study, implying that protein protects starch from degradation in rumen (30).
There was also a notable difference in the ratio of the amide band to cellulosic compounds in the 1273–1217 cm−1 region. CDC Helgason, AC Metcalfe, and CDC Dolly had relatively smaller ratios of amide I/cellulosic peak area (14.27, 15.44, and 16.92, respectively). The same trend was also found in the peak area ratio comparison of amide I/cellulosic compounds. McLeod had the greatest (P<0.05) height ratio of amide I/cellulosic compounds (9.77). This is further evidence to support the opinion that cellulosic compounds significantly contribute to the lower digestibility and degradability.
Another large difference (P<0.05) was seen at the peak area ratio of total CHO/cellulosic compounds. The higher starch content and lower undigestible fibrous materials may result in the better nutrient value, which has been noted in an earlier study (31). In comparison with CDC Trey (157.57) and CDC Cowboy (148.60), McLeod was merely 130.89. The ratio of nonstructural CHO (starch) peak area (CHO third peak)/cellulosic compounds peak area exhibited a similar trend among these six barley varieties. CDC Trey and CDC Cowboy had a relatively higher (P<0.05) peak height ratio of nonstructural CHO (starch) peak area (CHO third peak)/cellulosic compounds (42.47 and 40.93, respectively). Others ranged from 34.56 to 38.45.
Protein matrix provides adhesion for starch granules in endosperm tissue (32). Cellulosic compounds in the cell wall also make an effort to support and maintain microstructure. This association may influence the hardness of cereal, the particle size distribution, and, as a result, nutrition value. With protein encompassing the starch granules, cultivars containing more protein may be associated with harder structure. The protein content, structure, distribution, and conjunction may partially explain the grain characteristics. Whereas, documents are lacking in individual spot sampling on barley grain using IR spectroscopic analysis. Little information is comparable. Further study is needed to examine whether this can be used to predict the nutrient availability of grain.
We can draw the conclusion that in conjunction with synchrotron light source, chemical–structural information can be obtained in situ via the synchrotron FTIRM technique with intact tissue sections. Compared to traditional chemical analysis methodologies, for which grinding is always required, synchrotron SFTIRM is obviously superior in probing the internal biological matrix with minimal artificial influence within a cellular dimension. Again, the results confirm that with the advanced synchrotron FTIRM techniques, chemical–structural information of feedstuffs can be revealed. The synchrotron FTIRM data can provide the structural–chemical makeup information of original biological tissue.
Correlations between in Situ Degradation Kinetics (Rate and Extent) and Spectral Characteristics among Barley Varieties
Pearson correlation was computed to investigate the relationship between the synchrotron FTIRM spectral data and the rumen degradation rate and extent of DM, CP, and ST (Tables 4 and 5), and the soluble fraction (S) and potentially degradable fraction (D) of DM, CP, and ST in Table 6 for the evaluation of nutrient availability. The results showed that the correlation tendency appeared between the synchrotron FTIRM data and degradation features, though not all of them were significant (Tables 4–6). For CHO, the first component peak area was correlated with the degradation rate (Kd) of CP (P=0.101) (Table 4). Cellulosic compound area was correlated with soluble fractions (S, %) of CP (P=0.068) and ST (P=0.041) (Table 6). Additional spectroscopic analysis is necessary to clarify this assumption as to whether the chemical–structural makeup of the endosperm tissue is linked to the degradation kinetics of cereals grain.
Table 4.
Pearson Correlation between Structural Characteristics from Seed Endosperm Tissue and in Situ Rumen Degradation Rate and Extent of Six Barley Varietiesa
| items | in situ rumen degradation kinetics of DMb
|
in situ rumen degradation kinetics of CPb
|
in situ rumen degradation kinetics of STb
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Kd (%/h)
|
EDDM (%)
|
Kd (%/h)
|
EDCP (%)
|
Kd (%/h)
|
EDST (%)
|
|||||||
| correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | |
| amide I peak area | 0.610 | 0.198 | 0.529 | 0.281 | 0.644 | 0.168 | 0.538 | 0.271 | 0.381 | 0.456 | 0.394 | 0.440 |
| amide I peak height | 0.569 | 0.239 | 0.463 | 0.355 | 0.625 | 0.185 | 0.480 | 0.335 | 0.330 | 0.523 | 0.317 | 0.540 |
| amide II peak area | 0.498 | 0.315 | 0.434 | 0.390 | 0.513 | 0.298 | 0.437 | 0.386 | 0.290 | 0.578 | 0.312 | 0.547 |
| amide II peak height | 0.513 | 0.298 | 0.443 | 0.379 | 0.512 | 0.299 | 0.448 | 0.373 | 0.324 | 0.531 | 0.329 | 0.524 |
| amide I and II area | 0.587 | 0.220 | 0.510 | 0.302 | 0.617 | 0.192 | 0.517 | 0.293 | 0.362 | 0.480 | 0.377 | 0.461 |
| cellulosic compound area | −0.511 | 0.300 | −0.553 | 0.255 | −0.560 | 0.247 | −0.550 | 0.258 | −0.321 | 0.535 | −0.418 | 0.410 |
| cellulosic compound height | −0.349 | 0.497 | −0.0522 | 0.288 | −0.257 | 0.623 | −0.494 | 0.320 | −0.347 | 0.500 | −0.480 | 0.336 |
| CHO: 1st component peak area | −0.711 | 0.113 | −0.556 | 0.252 | −0.728 | 0.101 | −0.582 | 0.225 | −0.515 | 0.296 | −0.435 | 0.389 |
| CHO: 1st component peak height | −0.021 | 0.968 | 0.058 | 0.914 | −0.059 | 0.912 | 0.045 | 0.933 | 0.097 | 0.855 | 0.140 | 0.792 |
| CHO: 2nd component peak area | −0.646 | 0.166 | −0.505 | 0.307 | −0.609 | 0.199 | −0.528 | 0.282 | −0.527 | 0.283 | −0.426 | 0.399 |
| CHO: 2nd component peak height | −0.630 | 0.180 | −0.503 | 0.309 | −0.610 | 0.199 | −0.523 | 0.287 | −0.484 | 0.331 | −0.407 | 0.423 |
| CHO: 3rd component peak area | −0.660 | 0.154 | −0.449 | 0.372 | −0.687 | 0.132 | −0.486 | 0.329 | −0.471 | 0.346 | −0.336 | 0.515 |
| CHO: 3rd component peak height | −0.509 | 0.302 | −0.201 | 0.702 | −0.579 | 0.229 | −0.255 | 0.626 | −0.313 | 0.546 | −0.098 | 0.853 |
| Total carbohydrate peak area | −0.668 | 0.147 | −0.484 | 0.330 | −0.680 | 0.137 | −0.515 | 0.295 | −0.487 | 0.327 | −0.374 | 0.465 |
The structural characteristics include protein amides I and II and carbohydrates and structural carbohydrate (cellulosic compounds) in the endosperm tissue of barley varieties, revealed using synchrotron-based FTIR microspectroscopy.
Kd = degradation rate; EDDM, EDCP, and EDST = effective degradability of DM, CP, and starch, respectively.
Table 5.
Pearson Correlation Results between Structural Characteristic Ratios from Seed Endosperm Tissue and in Situ Rumen Degradation Rate and Extent of Six Barley Varietiesa
| items | in situ rumen degradation kinetics of DMb
|
in situ rumen degradation kinetics of CPb
|
in situ rumen degradation kinetics of STb
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Kd (%/h)
|
EDDM (%)
|
Kd (%/h)
|
EDCP (%)
|
Kd (%/h)
|
EDST (%)
|
|||||||
| correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | |
| ratio of total CHO peak area/amides macI and II peak area | −0.279 | 0.593 | 0.030 | 0.955 | −0.322 | 0.533 | −0.012 | 0.982 | −0.098 | 0.853 | 0.102 | 0.848 |
| ratio of NSC (starch) peak area (CHO 3rd peak)/amides I peak area | −0.295 | 0.571 | 0.045 | 0.933 | −0.380 | 0.457 | −0.007 | 0.990 | −0.081 | 0.879 | 0.138 | 0.795 |
| ratio of NSC (starch) peak height (CHO 3rd peak)/amide I height | −0.294 | 0.572 | 0.042 | 0.937 | −0.434 | 0.390 | −0.026 | 0.961 | −0.109 | 0.837 | 0.143 | 0.788 |
| ratio of amides I and II peak area/cellulosic compound peak area | 0.552 | 0.256 | 0.489 | 0.325 | 0.596 | 0.212 | 0.495 | 0.319 | 0.317 | 0.540 | 0.351 | 0.495 |
| ratio of amide I peak area/cellulosic compound peak area | 0.555 | 0.253 | 0.489 | 0.325 | 0.607 | 0.202 | 0.496 | 0.317 | 0.315 | 0.544 | 0.348 | 0.499 |
| ratio of amide I peak height/cellulosic compound peak height | 0.509 | 0.303 | 0.413 | 0.415 | 0.574 | 0.234 | 0.428 | 0.398 | 0.263 | 0.614 | 0.264 | 0.613 |
| ratio of total CHO peak area/cellulosic compound peak area | −0.171 | 0.746 | 0.117 | 0.826 | −0.175 | 0.741 | 0.076 | 0.887 | −0.089 | 0.867 | 0.136 | 0.797 |
| ratio of NSC (starch) peak area (CHO 3rd peak)/cellulosic peak area | −0.169 | 0.750 | 0.139 | 0.793 | −0.190 | 0.718 | 0.094 | 0.860 | −0.069 | 0.896 | 0.165 | 0.754 |
| ratio of NSC (starch) peak height (CHO 3rd peak)/cellulosic peak height | −0.125 | 0.813 | 0.185 | 0.726 | −0.155 | 0.770 | 0.134 | 0.801 | −0.085 | 0.873 | 0.177 | 0.737 |
The structural characteristics include protein amides I and II, and structural (cellulosic compounds) and nonstructural (NSC, starch) carbohydrates (CHO) in the endosperm tissue of barley varieties, revealed using synchrotron-based FTIR microspectroscopy.
Kd = degradation rate; EDDM, EDCP, and EDST = effective degradability of DM, CP, and starch, respectively.
Table 6.
Pearson Correlation Results between Structural Characteristics from Seed Endosperm Tissue and in Situ Rumen Degradation Characteristics (Soluble and Potentially Degradable Fractions) of Six Barley Varietiesa
| items | degradation characteristics of DMb
|
degradation characteristics of CPb
|
degradation characteristics of STb
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S (%)
|
D (%)
|
S (%/)
|
D (%)
|
S (%/)
|
D (%)
|
|||||||
| correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | correlation coefficient | P-value | |
| cellulosic compound area | −0.614 | 0.195 | −0.281 | 0.590 | 0.779 | 0.068 | −0.507 | 0.304 | 0.830 | 0.041 | −0.677 | 0.140 |
| cellulosic compound height | −0.527 | 0.283 | −0.628 | 0.182 | 0.500 | 0.313 | −0.823 | 0.044 | 0.404 | 0.427 | −0.684 | 0.134 |
| ratio of amide I peak area/cellulosic compounds peak area | 0.442 | 0.380 | 0.035 | 0.948 | −0.648 | 0.164 | 0.213 | 0.685 | −0.860 | 0.028 | 0.531 | 0.278 |
The structural characteristics include structural carbohydrate (cellulosic compounds) and amide I: cellulosic compound ratio in the endosperm tissue of barley varieties, revealed using synchrotron-based FTIR microspectroscopy.
S = soluble fraction; D = potentially degradable fraction of DM, CP, and starch.
Weak correlations (Tables 4–6) indicate that limited variations of these six barley varieties might not be sufficient to interpret the relationship between spectroscopic information and nutrient value of barley grain, although significant differences in the biodegradation kinetics were observed as presented in Table 1. The weak correlations may be due to the similarities and fewer variations among the barley varieties and may also be due to the limited number barley varieties (only six barley varieties were examined). The differences among the samples may not be significant enough to reveal the actual relationship between the spectroscopic data and the ruminal degradation kinetics (rate and extent). More studies involving a diverse range of samples with biological differences will, thereby, be necessary to further understand the relationship between the mid-IR spectroscopic information and nutrition value of feedstuffs.
In conclusion, inherent structural differences in the endosperm tissue among the seeds of the six barley varieties were detected by the synchrotron-based FTIRM technique with the univariate molecular spectral analysis. The synchrotron FTIRM spectral profiles differed among the barley varieties. The studies demonstrated the potential of ultraspatially resolved synchrotron based analytical technology (SFTIRM) to reveal the structural and chemical makeup within cellular and subcellular dimensions without destruction of the inherent structure of cereal seed tissue. With the synchrotron FTIRM technique, the structural characteristics of the cereal seeds were illuminated among different cultivars at high spatial resolution. The structural differences of barley seeds may be one reason for the various digestive behaviors and nutritive values for ruminants. Examination of other tissues such as seed coat, aleurone layer, and pericarp in barley may provide further insight as to why barley varieties exhibit different biodegradation behaviors and provide a deeper and better indication of the chemical makeup and structural information. Weak correlations indicate that limited variations of these six barley varieties might be not sufficient to interpret the relationship between synchrotron-based mid-IR spectroscopic information and the nutrient value of barley grain, although significant differences in the biodegradation kinetics were observed. Further research will be focused on the relationship between mid-IR spectral information in a whole IR region from 4000 to 800 cm−1 (not individual peaks and ratios) and rumen degradation kinetics at the different tissues, not only endosperm but also pericarp, seed coat, and aleurone.
Acknowledgments
We are grateful to B. Rossnagel (University of Saskatchewan) for sitting in graduate student (N. Liu) committee and providing valuable suggestion and discussion, Z. Niu (University of Saskatchewan) for chemical analysis assistance, Jennifer Bohon, Megan Bourassa, Randy Smith, and Lisa Miller (National Synchrotron Light Source-Brookhaven National Laboratory (NSLSBNL), U.S. Department of Energy, Upton, New York) for helpful data collection at U10B and U2B experimental stations, TimMay, Tor Pederson and Luca Quaroni (Canadian Light Sources (CLS), University of Saskatchewan) for helpful plant/feed/food/seed structure study at 01B1-1 (Mid IR) experimental station.
This research has been supported by grants from the Natural Sciences and Engineering Research Council of Canada (NSERC-Individual Discovery Grant) and Saskatchewan Agricultural Development Fund (ADF). The Canadian Light Sources (CLS) is supported by various federal and provincial funding agencies in Canada. The National Synchrotron Light Source in Brookhaven National Laboratory (NSLS-BNL, New York, USA) is supported by the U.S. Department of Energy contract DE-AC02-98CH10886. The Center for Synchrotron Biosciences (U2B), Case Western Reserve University, is supported by the National Institute for Biomedical Imaging and Bioengineering under P41-EB-01979.
LITERATURE CITED
- 1.Hart KJ, Rossnagel BG, Yu P. Chemical characteristics and in situ ruminal parameters of barley for cattle: Comparison of the malting cultivar AC Metcalfe and five feed cultivars. Can J Anim Sci. 2008;88:711–719. [Google Scholar]
- 2.Liu N. MS Thesis. University of Saskatchewan; Saskatoon, Canada: 2009. Ruminal Nutrient Availability and Inherent Structural Features of Six Barley Varieties Using in Situ Technique and Mid-IR Spectroscopy. [Google Scholar]
- 3.Wetzel DL, LeVine SM. Infrared Microbeam Analysis of Intricate Biological Specimens. Institute of Physics; Philadelphia, PA: 2000. pp. 65–66. [Google Scholar]
- 4.Wetzel DL, LeVine SM. Biological Applications of Infrared Microspectroscopy. In: Gremlich H-U, Yan B, editors. Infrared and Raman Spectroscopy of Biological Materials. Marcel Dekker Inc; New York: 2001. pp. 101–142. [Google Scholar]
- 5.Budevska BO. Vibrational Spectroscopy Imaging of Agricultural Products. In: Chalmers JM, Griffiths PR, editors. Handbook of Vibrational Spectroscopy. J. Wiley; Hoboken, NJ: 2002. pp. 3720–3732. [Google Scholar]
- 6.Diem M, Romeo M, Matthäs C, Miljkovic M, Miller L, Lasch P. Comparison of Fourier transform infrared (FTIR) spectra of individual cells acquired using synchrotron and conventional sources. Infrared Phys Technol. 2004;45(5–6):331–338. [Google Scholar]
- 7.Miller LM, Carr GL, Jackson M, Dumas P, Williams GP. The impact of infrared synchrotron radiation in biology: Past, present and future. Synchrotron Radiation News. 2000;13:31–38. [Google Scholar]
- 8.Marinkovic NS, Huang R, Bromberg P, Sullivan M, Sperber E, Moshe S, Miller LM, Jones K, Chouparova E, Franzen S. Center for Synchrotron Biosciences’ U2B Beamline: An international resource for biological infrared spectroscopy. J Synchrotron Radiat. 2002;9(part 4):189–197. doi: 10.1107/s0909049502008543. [DOI] [PubMed] [Google Scholar]
- 9.Yu P. Application of advanced synchrotron radiation-based Fourier transform infrared (SR-FTIR) microspectroscopy to animal nutrition and feed science: a novel approach. Br J Nutr. 2004;92(6):869–885. doi: 10.1079/bjn20041298. [DOI] [PubMed] [Google Scholar]
- 10.Marinkovic NS, Chance MR. Synchrotron Infrared Microspectroscopy. In: Meyers R, editor. Encyclopedia of Molecular Cell Biology and Molecular Medicine. Wiley; New York: 2005. pp. 671–708. [Google Scholar]
- 11.Miller LM, Dumas P. Chemical imaging of biological tissue with synchrotron infrared light. Biochim Biophys Acta. 2006;1758(7):846–857. doi: 10.1016/j.bbamem.2006.04.010. [DOI] [PubMed] [Google Scholar]
- 12.Wetzel DL, Eilert AJ, Pietrzak LN, Miller SS, Sweat JA. Ultraspatially-resolved synchrotron infrared microspectroscopy of plant tissue in situ. Cell Mol Biol (Noisy-Le-Grand) 1998;44(1):145–168. [PubMed] [Google Scholar]
- 13.Miller LM. Infrared Microspectroscopy and Imaging. National Synchrotron Light Source, Brookhaven National Laboratory, Internal Publication; 2002. [accessed July, 2009]. http://www.nsls.bnl.gov/newsroom/publications/otherpubs/imaging/workshopmiller.pdf. [Google Scholar]
- 14.Yu P, Block H, Niu Z, Doiron K. Rapid characterization of molecular chemistry, nutrient make-up and microlocation of internal seed tissue. J Synchrotron Radiat. 2007;14(part 4):382–390. doi: 10.1107/S0909049507014264. [DOI] [PubMed] [Google Scholar]
- 15.Dumas P, Miller L. The use of synchrotron infrared microspectroscopy in biological and biomedical investigations. Vib Spectrosc. 2003;32(1):3–21. [Google Scholar]
- 16.Rossnagel BG, Harvey BL. Barley Newsletter. Vol. 38. American Malting Barley Association, Inc; Milwaukee, WI: 1994. CDC Dolly two-row feed barley; p. 109. [Google Scholar]
- 17.Legge WG. AC Metcalfe barley. Can J Plant Sci. 2003;83(2):381–384. [Google Scholar]
- 18.Yu P, Doiron K, Liu D. Shining light on the differences in molecular structural chemical make-up and the cause of distinct degradation behavior between malting- and feed-type barley using synchrotron FTIR microspectroscopy: a novel approach. J Agric Food Chem. 2008;56(9):3417–3426. doi: 10.1021/jf800015x. [DOI] [PubMed] [Google Scholar]
- 19.Walker AM, Yu P, Christensen CR, Christensen DA, McKinnon JJ. Fourier transform infrared microspectroscopic analysis of the effects of cereal type and variety within a type of grain on molecular structural make-up in relation to rumen degradation kinetics. J Agric Food Chem. 2009;57:6871–6878. doi: 10.1021/jf901461u. [DOI] [PubMed] [Google Scholar]
- 20.Yu P, Christensen DA, Christensen CR, Drew MD, Rossnagel BG, McKinnon JJ. Use of synchrotron FTIR microspectroscopy to identify chemical differences in barley endosperm tissue in relation to rumen degradation characteristics. Can J Anim Sci. 2004;84(3):523–528. [Google Scholar]
- 21.Doiron KJ, Yu P, Christensen CR, Christensen DA, McKinnon JJ. Detecting molecular changes in vimy flaxseed protein structure using synchrotron ftirm and drift spectroscopic techniques: structural and biochemical characterization. Spectroscopy. 2009;23:307–322. [Google Scholar]
- 22.Doiron KJ, Yu P, McKinnon JJ, Christensen DA. Heat-induced protein structures and protein subfractions in relation to protein degradation kinetics and intestinal availability in dairy cattle. J Dairy Sci. 2009;92:3319–3330. doi: 10.3168/jds.2008-1946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pietrzak LN, Miller SS. Microchemical structure of soybean seeds revealed in situ by ultraspatially resolved synchrotron Fourier transformed infrared microspectroscopy. J Agric Food Chem. 2005;53(24):9304–9311. doi: 10.1021/jf050608x. [DOI] [PubMed] [Google Scholar]
- 24.Stuart B. Infrared Spectroscopy: Fundamentals and Applications. J. Wiley; Chichester, England: 2004. [Google Scholar]
- 25.Yu P. Applications of hierarchical cluster analysis (CLA) and principal component analysis (PCA) in feed structure and feed molecular chemistry research, using synchrotron-based Fourier transform infrared (FTIR) microspectroscopy. J Agric Food Chem. 2005;53(18):7115–7127. doi: 10.1021/jf050959b. [DOI] [PubMed] [Google Scholar]
- 26.Camm GA. MS Thesis. University of Saskatchewan; Saskatoon, SK, Canada: 2008. Grain Hardness and Slow Dry Matter Disappearance Rate in Barley. [Google Scholar]
- 27.Mikel MA, Kolb FL. Genetic Diversity of Contemporary North American Barley. Crop Sci. 2008;48(4):1399–1407. [Google Scholar]
- 28.Takeda Y, Takeda C, Mizukami H, Hanashiro I. Structures of large, medium and small starch granules of barley grain. Carbohydr Polym. 1999;38(2):109–114. [Google Scholar]
- 29.You SG, Izydorczyk MS. Molecular characteristics of barley starches with variable amylose content. Carbohydr Polym. 2002;49(1):33–42. [Google Scholar]
- 30.Yu P. An emerging method for rapid characterization of feed structures and feed component matrix at a cellular level and relation to feed quality and nutritive value. Arch Anim Nutr. 2006;60(3):229–244. doi: 10.1080/17450390600679017. [DOI] [PubMed] [Google Scholar]
- 31.Bowman JGP, Blake TK, Surber LMM, Habernicht DK, Bockelman H. Feed-quality variation in the barley core collection of the USDA National Small Grains Collection. Crop Sci. 2001;41(3):863–870. [Google Scholar]
- 32.Holopainen UR, Wilhelmson A, Salmenkallio-Marttila M, Peltonen-Sainio P, Rajala A, Reinikainen P, Kotaviita E, Simolin H, Home S. Endosperm structure affects the malting quality of barley (Hordeum vulgare L.) J Agric Food Chem. 2005;53(18):7279–7287. doi: 10.1021/jf050349b. [DOI] [PubMed] [Google Scholar]