Abstract
Background
Cognitive impairments occur frequently after stroke and contribute to significant disability. Strategy training shows promise but has not been examined in the acute phase of recovery.
Objective
We conducted a single-blind randomized pilot study estimating the effect of strategy training, relative to reflective listening (attention control), for reducing disability and executive cognitive impairments.
Methods
Thirty participants with acute stroke who were enrolled in inpatient rehabilitation and had cognitive impairments were randomized to receive strategy training (n=15, 10 sessions as adjunct to usual inpatient rehabilitation) or reflective listening (n=15, same dose). The Functional Independence Measure assessed disability at baseline, rehabilitation discharge, 3 and 6 months. The Color Word Interference Test of the Delis-Kaplan Executive Function System assessed selected executive cognitive impairments (inhibition, flexibility) at baseline, 3 and 6 months.
Results
Changes in Functional Independence Measure scores for the two groups over 6 months showed significant effects of group (F1,27=9.25, p=0.005), time (F3.74=96.00, p<0.001), and group*time interactions (F3,74=4.37, p<0.007) after controlling for baseline differences in stroke severity (F1,27=6.74, p=0.015). Color Word Interference Inhibition scores showed significant effects of group (F1,26=6.50, p=0.017), and time (F2,34=4.74, p=0.015), but the group*time interaction was not significant (F2,34=2.55, p=0.093). Color Word Interference Cognitive Flexibility scores showed significant effects of group (F1,26=23.41, p<0.001), and time (F2,34=12.77, p<0.001), and group*time interactions (F2,34=7.83, p<0.002). Interaction effects suggested greater improvements were associated with strategy training.
Conclusions
Strategy training shows promise for addressing disability in the first 6 months after stroke. Lessons from this pilot study may inform future clinical trials.
Introduction
Meta-cognitive strategy training (hereafter referred to as strategy training) is an intervention designed to harness one’s ability to observe, assess, and positively alter one’s own behavior.1 Strategy training teaches individuals to identify and prioritize problematic daily activities, identify barriers impeding performance, generate and evaluate strategies addressing these barriers, and generalize learning through practice.1,2 Thus, strategy training teaches skills that can be used to address disability in “real-life” activities, and is therefore worth considering in rehabilitation.
The hallmark of strategy training is its delineation between the therapist’s role and the participant’s role in the rehabilitation process. Therapists assume a role of guided discovery,3 systematically facilitating participants’ learning through prompts and questions, rather than directly instructing participants. In doing so, therapists guide participants, allowing participants to learn through their experiences. Through strategy training, participants learn to work through or work around specific problems in selected daily activities. In addition, participants learn how to apply the process to novel activities and situations, with the goal of promoting additional recovery of independence with daily activities long after rehabilitation is completed.
Strategy training has been examined in a variety of clinical populations (e.g., attention deficit hyperactivity disorder, learning disability, major depressive disorder, acquired brain injury) as an intervention to address a variety of clinical outcomes (e.g., independence, productivity, self-regulation).4–10 Originally conceived as a method for self-monitoring,11 strategy training has been examined as an intervention for teaching problem solving skills.12–13 In fact, selected studies suggest that strategy training helps adults with acquired brain injury (i.e., stroke, traumatic brain injury) reduce disability,1–2,14–15 and may even address some of the cognitive sequelae, particularly impairments in executive cognitive functions, associated with acquired brain injury.16–17
These studies have examined strategy training in clinical populations with stable behavioral and cognitive symptoms, including individuals with acquired brain injury in the chronic phase of recovery. However, there are compelling reasons to suspect that strategy training administered in the acute phase of recovery may have a robust effect on long-term outcomes. Experience-dependent synaptic changes that occur in the acute phase are likely to shape subsequent trajectories of recovery.18–19 These trajectories of recovery establish patterns of behavior, or habits, that are difficult to change.20 This is important because even small to moderate improvements in behavior can substantially reduce caregiver burden and health care costs.21–22 Furthermore, rehabilitation programs that have the potential to shape biological and behavioral patterns of recovery are most accessible during the acute phase of recovery. Combined, these factors support early intervention.
We previously examined the rationale for and feasibility of strategy training in adults with acute cognitive impairments after acquired brain injury (specifically, adults with stroke).1–2, 23 The purpose of this pilot study was to obtain preliminary data estimating the effect of strategy training, relative to an attention control condition, in the acute phase of recovery after stroke. The primary aim was to estimate the effect of strategy training on activities of daily living outcomes. The secondary aim was to explore the effects of strategy training on selected measures of executive cognitive functions. Our intent was to use these pilot data, and our experiences, to inform the design of future larger scale clinical trials examining the efficacy of strategy training in acute rehabilitation.
Methods
We conducted a single-blind randomized Phase II pilot study examining strategy training among adults with cognitive impairments after acute stroke. We approached consecutive admissions to inpatient rehabilitation and obtained written informed consent from the individual, if cognitive status permitted, or from a proxy, consistent with approved Institutional Review Board procedures.
We screened participants for the following criteria: 1) primary diagnosis of acute stroke, 2) impairment of cognitive functions (Quick Executive Interview ≥3),24–25 3) absence of severe aphasia (Boston Diagnostic Aphasia Examination Severity Rating Scale ≤1),26 4) absence of dementia diagnosis (indicated in the medical record), 5) absence of current major depressive disorder, bipolar, or psychotic disorder (Primary Care Evaluation of Mental Disorders),27 6) absence of drug and alcohol abuse within 3 months (Mini-International Neuropsychiatric Interview),28 and 7) anticipated length of stay greater than 5 days. Participants who provided consent and were eligible were randomized to intervention groups (strategy training, attention control) using a simple randomization scheme derived from a random number table (1:1 ratio).
Intervention
For both groups, 45 minute research intervention sessions were administered in addition to usual inpatient rehabilitation therapy daily 5 days per week for the duration of inpatient rehabilitation. Given the goal of conducting a randomized controlled trial in acute inpatient rehabilitation, the ethical imperative to not withhold usual rehabilitation services in lieu of an unproven intervention, and the need to control the administration of the experimental interventions to ensure rigor of the pilot study, we chose to augment rather than replace usual rehabilitation. Within each group, research intervention sessions were administered according to standardized procedures described elsewhere1–2 by trained rehabilitation personnel who were masked to the opposing intervention protocol. Fidelity procedures for both intervention protocols were completed using a protocol described elsewhere.2
Strategy training sessions addressed four steps (self-selected goals, self-evaluation, strategy development, and generalization) applied iteratively throughout the sessions via guided discussion and workbook materials. The first step focused on the identification and derivation of self-selected goals. Strategy training therapists used the Canadian Occupational Performance Measure to help participants identify activities that were important to them, and were difficult to perform since the stroke.29 Participants prioritized these activities and picked 4 to 6 activities to address in the context of strategy training sessions. This step required in depth interview and facilitation and usually required 1 to 2 sessions. Next, the participants selected one activity to start the process; they performed that activity, and they identified barriers to performance. Participants then learned a global strategy that they applied to the barriers to performance that they identified (“Goal-Plan-Do-Check”). In short, participants set a goal to address identified barriers (i.e., set criterion for performance outcome), developed a plan to address the goal, completed the plan, and checked whether the plan worked or requires revising. These steps were repeated iteratively until the goal was met (and thus participants moved on to the next activity). At the completion of each session, the strategy training therapists prompted the participants to identify key principles that they learned during the session, and to discuss ways to apply these key principles during the next session.
Attention control sessions were designed to control for the non-specific effects of strategy training (i.e., dose, attention). Attention control therapists utilized the first 1 to 2 sessions to help the participants identify and prioritize self-selected goals, using same procedure that was employed in the strategy training intervention. The remaining sessions focused on the use of journal entries in a workbook and discussions using scripted open-ended questions to stimulate participants’ reflections on their rehabilitation goals, activities, and experiences. Should participants ask direct questions seeking guidance on addressing their rehabilitation goals, attention control therapists suggested that the participants discuss their questions with their rehabilitation team. All participants rated both the strategy training and attention control sessions with moderate to high satisfaction.
Measures
We collected demographic (age, gender, race) and medical information (stroke onset, type, hemisphere) from the medical record at study admission. We characterized stroke severity with the National Institutes of Health Stroke Scale,30 and co-morbid medical burden severity with the Cumulative Illness Rating Scale.31
Trained and reliable raters who were masked to study intent and design administered a standardized measure of activities of daily living and a battery of neuropsychological tests at baseline, 3 months and 6 months later. The primary outcome measure, the Functional Independence Measure, was administered at study admission, inpatient rehabilitation discharge, 3 months after study admission, and 6 months after study admission.32 We selected this measure because it is the industry standard for assessing activities of daily living outcomes in acute inpatient rehabilitation populations.
The secondary outcome measure, the Color Word Interference Test from the Delis-Kaplan Executive Functioning System, was used to assess selected executive cognitive functions.33 Specifically, we examined the scaled scores from the inhibition subscale (Condition 3) and the cognitive flexibility (switching) subscale (Condition 4). We selected these subscales because they measure two domains of executive cognitive functions that are likely to be influenced by strategy training. Inhibition, or the process of ignoring irrelevant stimuli to complete a task, may be influenced through self-monitoring and self-evaluation processes inherent in strategy training. Cognitive flexibility, or the ability to hold more than one concept in mind, and switch between concepts, may be influenced through iterative application and revision of the global strategy. The Color Word Interference Test is valid and reliable and has demonstrated sufficient psychometric performance in adults with stroke.33 For this study, we used the revised scoring procedures for the inhibition subscale. (Unpublished standardization data from the Delis-Kaplan Executive Function System. Copyright ©, 2013, 2001 NCS Pearson, Inc. Used with permission. All rights reserved).
Data Analyses
Statistical analyses were conducted using SAS version 9.3 statistical software (SAS Institute, INC, Cary, North Carolina). All analyses were performed using the intent-to-treat principle so that comparisons were made according to the assigned intervention group regardless of study completion. We began by examining and testing relevant baseline characteristics to make sure there were no differences between groups prior to starting intervention and to identify possible covariates. We conducted repeated measures mixed effects model analyses for the primary outcome (Functional Independence Measure) and the exploratory outcomes (inhibition, cognitive flexibility). Repeated measures mixed model analyses allow for missing data without imputation. We examined group (strategy training, attention control) and time (baseline, discharge, 3 and 6 months) as main fixed effects, including the group*time interaction and significant baseline characteristics as covariates. We examined group variances and allowed the residual matrix to have different variances for each group when necessary. The optimal fit model was selected by comparing the Bayesian Criterion between nested models. Post hoc analyses were completed using planned contrasts in the repeated measures mixed models to interpret significant fixed effects. All available data were used in analyses for the first pass, and we also completed sensitivity analyses by repeating the mixed model analyses for participants with complete data. For the secondary outcomes examining inhibition and cognitive flexibility, the lowest score within the group was used if a participant was too cognitively impaired to engage in the task. For all statistically significant models, Cohen’s d effect sizes were computed using model-derived change score estimates.
Results
Sixty participants enrolled in the study (Figure 1). Of these, 27 participants were excluded because they did not meet eligibility criteria, and 3 participants withdrew during the screening process prior to randomization. Thus, we randomized 30 participants to the intervention groups.
Figure 1.
CONSORT Flow Diagram
The two groups did not differ at baseline with the exception of baseline stroke severity (t28=−2.99, p<0.006, Table 1). The strategy training group had significantly worse neurological impairment at baseline than the attention control group, and thus baseline National Institutes of Health Stroke Scale scores were considered as a potential covariate in subsequent analyses.
Table 1.
Baseline Characteristics of Participants
| Strategy Training (n=15) |
Attention Control (n=15) |
||
|---|---|---|---|
| Sex, Male, n(%) | 9 (60) | 11 (73) | χ21=0.60 |
| Age, Years, M(SD) | 64.87 (16.59) | 71.80 (13.19) | t28=1.26 |
| Race, White, n(%) | 12 (80) | 14 (93) | χ21=1.15 |
| Stroke onset, Days, M(SD) | 16.80 (15.58) | 18.47 (21.29) | t28=0.25 |
| Stroke type, Ischemic, n(%) | 10 (67) | 11 (73) | χ21=0.16 |
| Hemisphere, Right, n(%) | 10 (67) | 10 (67) | χ21=0.00 |
| Stroke severity, NIHSS, M(SD)* | 8.87 (2.77) | 5.87 (2.72) | t28=2.99† |
| Medical burden, CIRS, M(SD)* | 2.37 (0.47) | 2.36 (0.57) | t28=0.04 |
| Cognitive status, EXIT, M(SD)* | 8.13 (3.00) | 9.31 (3.54) | t28=1.20 |
| Communication status, BDAE, M(SD) | 4.27 (1.03) | 4.67 (0.62) | t28=1.29 |
| Functional Independence Measure, M(SD) | 65.27 (10.85) | 62.60 (16.56) | t28=0.52 |
| Inhibition, CWI, M(SD) | 4.85 (4.10) | 4.00 (2.98) | t26=0.63 |
| Cognitive Flexibility, CWI, M(SD) | 3.69 (2.36) | 2.73 (2.34) | t26=1.08 |
| Research intervention sessions, M(SD) | 9.40 (5.31) | 9.00 (4.89) | t28=0.21 |
NIHSS= National Institutes of Health Stroke Scale. CIRS=Cumulative Illness Rating Scale. EXIT=Quick Executive Interview (14-item version). BDAE=Boston Diagnostic Aphasia Examination Severity Index. CWI=Color Word Interference.
Lower scores=better health.
p<.05
All participants received and demonstrated at least an acceptable understanding of the intervention they received (1=minimal understanding, 2=acceptable understanding, 3=excellent understanding; strategy training M=2.28 SD=0.35, attention control M=2.87, SD=0.28). Understanding was modestly correlated with Quick Executive Interview scores for the strategy training group (Spearman’s ρ=0.06) and moderately correlated with Quick Executive Interview scores for the attention control group (Spearman’s ρ=0.51). Understanding was modestly correlated with Boston Diagnostic Aphasia Examination Severity Index scores in both groups (strategy training Spearman’s ρ=0.26; attention control Spearman’s ρ=0.12).
Changes in Functional Independence Measure scores for the two groups over the 6 months showed significant effects of group (F1,27=9.25, p=0.005), time (F3.74=96.00, p<0.001), and group*time interactions (F3,74=4.37, p<0.007) after controlling for baseline differences in stroke severity (F1,27=6.74, p=0.015; Figure 2). Post hoc analyses using modeled data suggest that strategy training was superior to attention control in reducing disability in activities of daily living over the 6 month time period (t74=2.94, p=0.004, d=1.11; Table 2). While both groups improved more than 3 times the clinically meaningful 10 point change35 at both 3 and 6 months from baseline, the strategy training participants showed 15 points greater change or 1.5 times greater than the meaningful change compared to attention control at 3 and at 6 months. Furthermore, groups differed by more than 24 points at 6 months (strategy training: M=116.31, SE=2.99; attention control: M=91.81, SE=5.81; t74=3.64, p<0.0001). Given that a 1 point change on the Functional Independence Measure has been associated with 2.19 minutes of caregiver assistance per day,22 strategy training participants in this small sample required approximately 1 hour less caregiver assistance per day than attention control participants. Sensitivity analysis findings were similar to the original analyses, the main effect of group (F1,21=6.15, p=0.02), time (F3,66=89.72, p<0.01), and group*time interaction effect (F3,66=4.50, p<0.01) remained significant after controlling for baseline differences in stroke severity (F1,21=4.33, p<0.05).
Figure 2.
Functional Independence Measure Total Scores, By Group
Table 2.
Post Hoc Contrasts Using Model-derived Change Score Estimates
| Strategy Training (n=15) |
Attention Control (n=15) |
Cohen’s d Effect Size |
||
|---|---|---|---|---|
| Functional Independence Measure | ||||
| Baseline to Month 3, M(SE) | 44.65 (2.68) | 29.91 (4.52) | t74=2.81, p=0.006 | 1.06 |
| Baseline to Month 6, M(SE) | 48.27 (2.68) | 32.12 (4.78) | t74=2.94, p=0.004 | 1.11 |
| Color Word Interference Inhibition (Condition 3) | ||||
| Baseline to Month 3, M(SE) | 3.89 (1.10) | 0.87 (1.16) | t34=1.89, p=0.07 | 0.74 |
| Baseline to Month 6, M(SE) | 3.23 (1.13) | 0.04 (1.22) | t34=1.92, p=0.06 | 0.76 |
| Color Word Interference Cognitive Flexibility (Condition 4) | ||||
| Baseline to Month 3, M(SE) | 5.50 (0.93) | 0.72 (0.98) | t34=3.52, p=0.001 | 1.38 |
| Baseline to Month 6, M(SE) | 4.91 (0.96) | 0.52 (1.03) | t34=3.12, p=0.004 | 1.23 |
We experienced difficulties in administering neuropsychological assessments with this population, resulting in missing data on the Color Word Interference subscales. At baseline, we were unable to administer the subscales with 2 participants in the strategy training group (aphasia impeded in one case, and severe visual perceptual impairment impeded in the other case). Both of these issues resolved over time, and these participants completed 3 and 6 month follow-ups. Among the 26 participants still enrolled in the study at 3 months, 6 participants did not complete the subscales (3 in the strategy training group refused, and 3 in the attention control group were too medically ill). Among the 24 participants still enrolled in the study at 6 months, the same 6 participants did not complete the subscales for the same reasons. These 6 participants did not differ from the rest of the sample with respect to baseline Color Word Interference subscale scores (Inhibition, t24=0.16; Cognitive Flexibility, t24=0.39). Given the sample size we elected to run the analyses without imputing data.
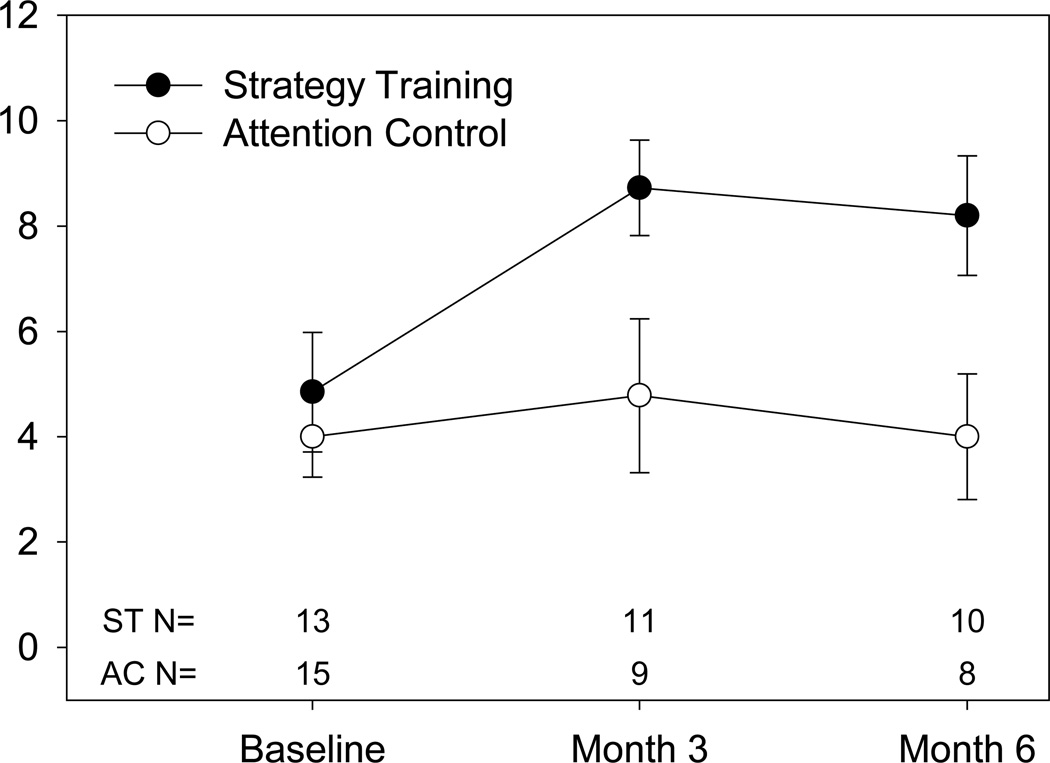
Stroke severity was not a significant covariate of Color Word Interference subscale scores (Inhibition, F1,25=0.95, p=0.34; Cognitive Flexibility, F1,25=0.05, p=0.83). Thus we removed it from both models and reran the analyses. For inhibition, the model showed significant effects of group (F1,26=6.50, p=0.017), and time (F2,34=4.74, p=0.015), but the group*time interaction was not significant (F2,34=2.55, p=0.093; Figure 3). For cognitive flexibility, the model showed significant effects of group (F1,26=23.41, p<0.001), and time (F2,34=12.77, p<0.001), and group*time interactions (F2,34=7.83, p<0.002; Figure 4). Post hoc analyses of modeled data suggest that strategy training was superior to attention control for improving inhibition (t34=1.92, p=0.06, d=0.76; Table 2) and cognitive flexibility (t34=3.12, p=0.004, d=1.23; Table 2), although the differences improvements in inhibition were not statistically significant. Nonetheless, given that these are scaled scores that have a population mean of 10, and standard deviation of 3, change greater than 3 points could be considered clinically meaningful.
Figure 3.
Color Word Interference Inhibition Scaled Scores, By Group
Figure 4.
Color Word Interference Cognitive Flexibility Scaled Scores, By Group
Discussion
This early phase trial is a critical step toward future trials examining the efficacy of targeted interventions designed to address disability among adults with cognitive impairments in the acute phase of recovery. Stinear and colleagues reported in a recent systematic review that while stroke rehabilitation begins early in the acute phase of recovery, stroke rehabilitation research does not.36 The most obvious reasons for this discrepancy are the challenges in responsibly delivering standardized interventions and accurately assessing outcomes that can be attributed to these interventions in this setting. Nonetheless, there are scientific and practical considerations that emerge when attempting to translate evidence developed in the chronic phase of recovery to practice in the acute phase of recovery. Furthermore, Stinear and colleagues argue that while the study of interventions in the chronic phase allows us to parse out the effects of an intervention without the confounding of spontaneous recovery, the study of interventions in the acute phase may allow us to harness the synergistic benefit of potent interventions and spontaneous recovery in this critical period of recovery. These points argue for concerted efforts in the rigorous design and implementation of trials in the acute phase. The present study addresses these efforts in several ways.
The primary aim of this early phase trial was to obtain preliminary data to estimate the effect of strategy training among individuals with stroke-related cognitive impairments engaged in acute inpatient rehabilitation. This estimate may be used to inform the design of the next phase of studies. In this small sample, strategy training resulted in greater improvements in independence with activities of daily living than attention control (large effect size). Of note, we observed an increasing magnitude of difference between groups in activities of daily living scores over time. Although these findings are consistent with studies examining strategy training among individuals with chronic cognitive impairments after stroke and traumatic brain injury,12–15 the authors urge caution in generalizing these findings. Others have commented that small pilot studies, while an important first step in the design of rigorous clinical trials, tend to yield inflated effect sizes.37–38 In addition, there may be many other factors contributing to these findings, including differences in social support, post-acute care, or other factors that require closer examination in a larger study. Thus, these findings suggest that strategy training shows promise, but requires confirmatory testing in larger, more rigorous studies.
The secondary aim of this pilot study was to explore the effects of strategy training on selected indices of executive cognitive functions. This exploration was necessary to gather preliminary data on theorized mechanisms underlying strategy training. In this sample, strategy training was associated with greater improvements in cognitive flexibility (large effect size). Cognitive flexibility has been defined as the ability to alter behavior in response to changes in circumstances and is considered one of the executive cognitive function domains that guide goal-directed behavior.39 Strategy training, as implemented in this study, focused on identifying and prioritizing problems with everyday activities, as well as identifying, trialing, and evaluating solutions to these problems. One may suppose that strategy training supports improvements in cognitive flexibility through multiple reiteration of these steps. Once participants identify a solution to a problem, and determine whether it requires modification or replacement with another solution, they may increase their ability to think more flexibly about the problems they encountered in everyday life. Indeed some forms of strategy training improve selected cognitive functions through training of meta-cognitive awareness.15–16 However, the potential effects of strategy training approaches on cognitive functions, and the mechanisms through which these effects may emerge remain important areas of study, particularly as we seek to identify key principles to augment rehabilitation for individuals with cognitive impairments after acquired brain injury.
The findings from this early phase clinical trial are not definitive. First, the small size of the sample does not rule out potential factors that may have influenced study outcomes (i.e., differences in baseline characteristics, differences in inpatient rehabilitation and post-acute care among participants). There was a statistically significant difference between groups in baseline stroke severity (the difference favored the attention control group). Although not statistically significant, the attention control group was older than the strategy training group, with the difference potentially favoring the strategy training group.
Second, 20% of participants were missing 6 month data for the primary outcome due to refusal of assessments (n=1), withdrawal (n=2), and loss to follow-up (n=3). While sensitivity analyses of the primary outcome, the Functional Independence Measure, indicates a limited effect of this attrition, replication in a larger sample is warranted. Of greater concern are the challenges that we encountered when attempting to administer the subscales of the Color Word Interference Test. Aphasia, visual perceptual impairment, and fatigue are all common problems after acute stroke, and all influenced the administration of this assessment at various timepoints. Thus, we urge caution when interpreting the executive cognitive assessment findings from this pilot study.
Nonetheless, we learned many valuable lessons about the opportunities and challenges of conducting trials in the acute phase of rehabilitation that may be used to inform larger, well-controlled trials. First, while randomization in larger samples may account for non-specific individual differences that influence intervention outcomes, stratification on selected variables, such as stroke severity and communication status, may be warranted. Second, careful characterization of the content and duration of inpatient and post-acute rehabilitation sessions may be useful for separating out the effects of the usual care from those of the experimental intervention. Third, improved staff training and the use of mid-interval calls to participants may reduce attrition after participants leave inpatient rehabilitation. Finally, exploration of simpler, less time intensive methods of assessing executive cognitive functions may be warranted for this population. The primary challenge that we encountered with the neuropsychological assessments was the amount of time and energy these assessments required, and subsequent participant refusal. Based on our experiences, we recommend administering neuropsychological assessments in a separate test session at baseline and at follow-up, if necessary. We also recommend improved methods for assessing executive cognitive functions for individuals with mild to moderate aphasia and individuals with visual perceptual impairments. These lessons may be used to inform the design of future, well-controlled trials examining the efficacy of interventions such as strategy training.
Conclusion
Strategy training shows promise for addressing disability and executive cognitive impairments within the first 6 months of stroke. Valuable lessons learned in the conduct of this early phase clinical trial may be useful in the design of future confirmatory trials.
Acknowledgments
Supported by the National Institutes of Health (K12 HD055931), the University of Pittsburgh Medical Center Rehabilitation Institute, and the University of Pittsburgh Office of Research Health Sciences.
References
- 1.Skidmore ER, Holm MB, Whyte EM, Dew MA, Dawson D, Becker JT. The feasibility of meta-cognitive strategy training in acute inpatient stroke rehabilitation: case report. Neuropsychol Rehabil. 2011;23:208–223. doi: 10.1080/09602011.2011.552559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Skidmore ER, Dawson DR, Whyte EM, Butters MA, Dew MA, Grattan ES, Becker JT, Holm MB. Developing complex interventions: lessons learned from a pilot study examining strategy training in acute rehabilitation. Clin Rehabil. 2014;28:378–378. doi: 10.1177/0269215513502799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wales CE, Nardi AH, Stager RA. Decision making: new paradigm for education. Educ Leadersh. 1986;43:203–215. [Google Scholar]
- 4.Kennedy MR, Coelho C, Turkstra L, Ylvisaker M, Moore Sohlberg M, Yorkston K, Chiou HH, Kan PF. Intervention for executive functions after traumatic brain injury: a systematic review, meta-analysis and clinical recommendations. Neuropsychol Rehabil. 2008;18:257–299. doi: 10.1080/09602010701748644. [DOI] [PubMed] [Google Scholar]
- 5.Webster JS, Scott RR. The effect of self-instructional training on attention deficits following head injury. Clin Neuropsychol. 1983;5:69–74. [Google Scholar]
- 6.Alderman N, Fry RK, Youngson HA. Improvement of self-monitoring skills, reduction of behaviour disturbance and the dysexecutive syndrome: comparison of response cost and a new programme of self-monitoring training. Neuropsychol Rehabil. 1995;5:193–221. [Google Scholar]
- 7.Nezu AM, Perry MG. Social problem-solving therapy for unipolar depression: an initial dismantling investigation. J Consult Clin Psychol. 1989;57:408–413. [PubMed] [Google Scholar]
- 8.Arean P, Hegel M, Vannoy S, Fan MY, Unuzter J. Effectiveness of problem solving therapy for older, primary care patients with depression: results from the IMPACT project. Gerontol. 2008;48:311–323. doi: 10.1093/geront/48.3.311. [DOI] [PubMed] [Google Scholar]
- 9.Feeney T, Ylvisaker M. Context-sensitive behavioral supports for young children with TBI: short-term effects and long-term outcome. J Head Trauma Rehabil. 2003;18:33–51. doi: 10.1097/00001199-200301000-00006. [DOI] [PubMed] [Google Scholar]
- 10.Feeney T, Ylvisaker M. Context-sensitive cognitive-behavioral supports for young children with TBI: a replication study. Brain Injury. 2006;20:629–645. doi: 10.1080/02699050600744194. [DOI] [PubMed] [Google Scholar]
- 11.Meichenbaum D. Cognitive-behavior modification: an integrative approach. New York: Plenum Press; 1977. [Google Scholar]
- 12.von Cramon D, Matthes-von Cramon G, Mai N. Problem-solving deficits in brain-injured patients: a therapeutic approach. Neuropsych Rehabil. 1991;1:45–64. [Google Scholar]
- 13.Cicerone KD, Giacino JT. Remediation of executive function deficits after traumatic brain injury. Neurorehabil. 1992;2:12–22. [Google Scholar]
- 14.Dawson D, Gaya A, Hunt A, Levine B, Lemsky C, Polatajko H. Using the cognitive orientation to occupational performance (CO-OP) with adults with executive dysfunction following traumatic brain injury. Can J Occup Ther. 2009;76:74–86. doi: 10.1177/000841740907600209. [DOI] [PubMed] [Google Scholar]
- 15.McEwen S, Huijbregts M, Ryan J, Polatajko H. Cognitive strategy use to enhance motor skill acquisition post-stroke: a critical review. Brain Injury. 2009;23:263–277. doi: 10.1080/02699050902788493. [DOI] [PubMed] [Google Scholar]
- 16.Levine B, Schweizer TA, O'Connor C, Turner G, Gillingham S, Stuss DT, Manly T, Robertson IH. Rehabilitation of executive functioning in patients with frontal lobe brain damage with goal management training. Frontiers Human Neurosci. 2011;5 doi: 10.3389/fnhum.2011.00009. Article 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Novakovic-Agopian T, Chen AJ, Rome S, Abrams G, Castelli H, Rossi A, McKim R, Hills N, D'Esposito M. Rehabilitation of executive functioning with training in attention regulation applied to individually defined goals: a pilot study bridging theory, assessment and treatment. J Head Trauma Rehabil. 2011;26:325–328. doi: 10.1097/HTR.0b013e3181f1ead2. [DOI] [PubMed] [Google Scholar]
- 18.Kleim JA, Jones TA, Schallert T. Motor enrichment and the induction of plasticity before or after brain injury. Neurochemical Research. 2003;28:1757–1769. doi: 10.1023/a:1026025408742. [DOI] [PubMed] [Google Scholar]
- 19.Jones JA, Jefferson SC. Reflections of experience-expectant development in repair of the adult damaged brain. Developmental Psychobiol. 2011;53:466–475. doi: 10.1002/dev.20557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Taub E, Uswatte G, Elbert T. New treatments in neurorehabilitation founded on basic research. Nature. 2002;3:228–236. doi: 10.1038/nrn754. [DOI] [PubMed] [Google Scholar]
- 21.Granger CV, Cotter AC, Hamilton BB, Fiedler RC. Functional assessment scales: a study of persons after stroke. Arch Phys Med Rehabil. 1993;74:133–138. [PubMed] [Google Scholar]
- 22.Mahler MP, Zuger K, Kaspar K, Haefeli A, Jenni W, Leniger T, Beer JH. A cost analysis of the first year after stroke - early triage and inpatient rehabilitation may reduce long term costs. Swiss Medical Weekly. 2008;138:459–468. doi: 10.4414/smw.2008.11845. [DOI] [PubMed] [Google Scholar]
- 23.Skidmore ER. Activity interventions for cognitive impairments. Ped Blood Oncol. doi: 10.1002/pbc.24781. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Royall DR, Mahurin RK, Gray KF. Bedside assessment of executive cognitive impairment: the executive interview. J Am Geriatr Soc. 1992;40:1221–1226. doi: 10.1111/j.1532-5415.1992.tb03646.x. [DOI] [PubMed] [Google Scholar]
- 25.Larson EB, Heinemann AW. Rasch analysis of the Executive Interview (the EXIT-25) and introduction of an abridged version (the Quick EXIT) Arch Phys Med Rehabil. 2010;91:389–394. doi: 10.1016/j.apmr.2009.11.015. [DOI] [PubMed] [Google Scholar]
- 26.Goodglass H, Kaplan E, Barressi B. The assessment of aphasia and related disorders. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2001. [Google Scholar]
- 27.Spitzer RL, Williams JB, Kroenke K, KLinzer M, deGruy FV, Hahn SR, Brody D, Johnson JG. Utility of a new procedure for diagnosing mental disorders in primary care: the Prime-MD 1000 study. JAMA. 1994;272:1749–1756. [PubMed] [Google Scholar]
- 28.Sheehan DV. The Mini-International Neuropsychiatric Interview (M.I.N.I): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatr. 1998;59:22–57. [PubMed] [Google Scholar]
- 29.Law M, Baptiste S, Carswell A, et al. Canadian Occupational Performance Measure. third edition. Ottawa, ON: CAOT Publications ACE; 1998. [Google Scholar]
- 30.Brott T, Adams HP, Olinger CP, Marler JR, Barsan WG, Biller J, Spilker J, Holleran R, Eberle R, Hertzberg V, Rorick M, Moomaw CJ, Walker M. Measurements of acute cerebral infarction: a clinical examination scale. Stroke. 1989;20:864–870. doi: 10.1161/01.str.20.7.864. [DOI] [PubMed] [Google Scholar]
- 31.Miller MD, Paradis CF, Houck PR, Mazumdar S, Stack JA, Rifai AH, Mulsant B, Reynolds CF. Rating chronic medical illness burden in geropsychiatric practice and research: Application of the Cumulative Illness Rating Scale. Psychiatr Res. 1992;41:237–248. doi: 10.1016/0165-1781(92)90005-n. [DOI] [PubMed] [Google Scholar]
- 32.Stineman MG, Shea JA, Jette A, Tassoni CJ, Ottenbacher KJ, Fiedler R, Granger CV. The Functional Independence Measure: tests of scaling assumptions, structure and reliability across 20 diverse impairment categories. Arch Phys Med Rehabil. 1996;77:1101–1108. doi: 10.1016/s0003-9993(96)90130-6. [DOI] [PubMed] [Google Scholar]
- 33.Delis DC, Kaplan EF, Kramer JH. Delis-Kaplan Executive Function System (D-KEFS) London: Harcourt Assessment Inc; 2001. [Google Scholar]
- 34.Lenze EJ, Munin MC, Quear T, et al. The Pittsburgh Rehabilitation Participation Scale: reliability and validity of a clinician-rated measure of participation in acute rehabilitation. Arch Phys Med Rehabil. 2004;85:380–384. doi: 10.1016/j.apmr.2003.06.001. [DOI] [PubMed] [Google Scholar]
- 35.Granger CV, Cotter AC, Hamilton BB, Fiedler RC. Functional assessment scales: a study of persons after stroke. Arch Phys Med Rehabil. 1993;74:133–138. [PubMed] [Google Scholar]
- 36.Stinear C, Ackerley S, Byblow W. Rehabilitation is initiated early after stroke, but most motor rehabilitation trials are not: a systematic review. Stroke. 2013;44:2039–2045. doi: 10.1161/STROKEAHA.113.000968. [DOI] [PubMed] [Google Scholar]
- 37.Dobin BH. Progressive staging of pilot studies to improve phase III trials for motor interventions. Neurorehabil Neural Repair. 2009;23:197–206. doi: 10.1177/1545968309331863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Moore CG, Carter RE, Nietert PJ, Stewart PW. Recommendations for planning pilot studies in clinical and translational research. Clin Trans Sci. 2011;4:332–337. doi: 10.1111/j.1752-8062.2011.00347.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Robbins TW. Optimizing the executive: neurochemical modulation of the fronto-executive “toolbox.”. In: Stuss DT, Knight RT, editors. Principles of frontal lobe function (2nd ed) Oxford: Oxford University Press; 2013. [Google Scholar]