Abstract
Rationale
When ad libitum fed (AL) rats undergo cocaine place preference conditioning (CPP) but are switched to food-restriction (FR) for testing, CPP is enhanced and preference scores correlate with phosphorylation of AMPA receptor GluA1 at Ser845 in nucleus accumbens (NAc) core.
Objectives
The present study tested whether a similar association exists in AL rats, and whether an inhibitor of Ca2+-permeable AMPARs blocks CPP expression in either diet group.
Materials and methods
In Experiments 1-3, AL rats were conditioned with cocaine (12.0 mg/kg, i.p.). Three weeks later, CPP was tested daily and brains were harvested after the 5th test. Western analyses were used to probe for levels of AMPA receptors in NAc. In Experiment 4, AL rats were conditioned, half were switched to FR for testing, and half in each diet group received NAc core microinjection of NASPM (25.0 μg) prior to each test.
Results
In Experiment 1, CPP expression in AL rats was associated with elevated pSer845-GluA1, GluA1 and GluA2 in NAc. In Experiment 2, the correlation between pSer845-GluA1 and CPP was localized to NAc core. In Experiment 3, pSer845-GluA1 following a CPP test was higher in NAc synaptic membranes of FR relative to AL rats. In Experiment 4, NASPM blocked CPP expression in both diet groups.
Conclusions
Results support a scheme in which pSer845-GluA1 in NAc core underlies expression of cocaine CPP, and does so by stabilizing or trafficking Ca2+-permeable AMPARs to the synaptic membrane. The more robust CPP of FR rats may result from upregulation of stimulus-induced pSer845-GluA1.
Keywords: Food restriction, nucleus accumbens, cocaine, conditioned place preference, GluA1
One consequence of the overlap in neural substrates for ingestive behavior and drug abuse is the increased self-administration of abused drugs by food-restricted subjects (Carroll and Meisch 1984). This well-established effect is due, at least in part, to increased drug reward magnitude which has been attributed to increased D1 dopamine (DA) receptor signaling and calcium-permeable AMPA receptors (CP-AMPARs) in nucleus accumbens (NAc) shell (Cabeza de Vaca and Carr 1998; Carr et al. 2003; Carr et al. 2010). From a clinical standpoint, an equally if not more hazardous effect of food restriction (FR) is the enhanced behavioral responsiveness to a drug-paired context, as demonstrated in cocaine conditioned place preference (CPP) studies (Liu et al. 2011; Zheng et al. 2012; Zheng et al. 2013). Context-induced craving is a common relapse trigger in addicts (Childress et al. 1993; See 2002), and it may be presumed that the hyperphagia and body weight gain that accompany psychostimulant abstinence sometimes provoke weight loss dieting (Orsini et al. 2014). Moreover, up to one third of cocaine abusers meet diagnostic criteria for an eating disorder (Jonas et al. 1987). Thus, food restriction initiated for health or cosmetic reasons, as well as when borne of psychopathology, may increase risk of relapse by enhancing the incentive effects of environments associated with subjective effects of drug.
It was previously shown that cocaine CPP is more persistent in FR than ad libitum fed (AL) rats, and that persistence is dependent on FR during CPP expression testing (Liu et al. 2011; Zheng et al. 2012). Further, CPP magnitude and persistence did not vary according to degree of acute food deprivation or circulating levels of ghrelin, insulin, or corticosterone, suggesting involvement of stable neuroadaptations (Zheng et al. 2013). When brains of FR subjects were harvested immediately after the 16th CPP test, those still expressing a CPP had higher levels of glutamatergic AMPA receptors, test, those still expressing a CPP had higher levels of glutamatergic AMPA receptors, pSer845-GluA1 and total GluA1, in the NAc core relative to those that did not express a CPP. In addition, there was a strong positive correlation between NAc core levels of pSer845-GluA1 and magnitude of preference for the cocaine-paired side of the CPP apparatus (Zheng et al. 2013). In light of the role of GluA1-containing AMPA receptors in fast excitatory synaptic transmission and activity-dependent synaptic plasticity (Barry and Ziff 2002; Malinow 2003), and the effect of Ser845 phosphorylation on increasing channel open probability, stabilizing surface GluA1 and facilitating its trafficking to the synaptic membrane (Esteban et al. 2003; He et al. 2009; Man et al. 2007; Mangiavacchi and Wolf 2004), these observations point to GluA1-containing AMPARs in NAc core as a candidate mechanistic basis of increased cocaine CPP expression in FR rats. Should this be the case, it would parallel evidence that the increased reward magnitude of psychostimulants in FR rats is mediated by Ca2+-permeable (i.e., GluA2-lacking) AMPARs in NAc shell (Carr et al. 2010; Peng et al. 2014). The main goals of the present study were to determine whether upregulated AMPAR mechanisms are unique to FR rats that express cocaine CPP, and whether NAc core microinjection of an antagonist of CP-AMPARs blocks CPP expression in FR, but not AL, rats.
Materials and methods
Subjects and food restriction
Experimental procedures were approved by the Institutional Animal Care and Use Committee at the New York University School of Medicine and were consistent with the Principles of Laboratory Care (NIH Publication no. 85-23). All subjects were mature male Sprague-Dawley rats (Taconic Farms, Germantown, NY) weighing 350-400 g (10-12 weeks old) at the start of each experiment. Rats were housed on a 12-hour light:dark photoperiod with lights on between 6am and 6pm in a central animal facility. All subjects were singly housed in individual plastic cages with bedding and free access to water.
Ad libitum-fed animals had free access to standard lab pellets (Rodent Diet #5001, Lab Diet, St. Louis, MO). Food restricted animals followed a feeding regimen described previously (e.g.,Cabeza de Vaca and Carr 1998), in which rats received 40% of ad libitum intake of chow (10 g) daily, delivered to the home cage at 5 pm, until body weight was reduced by 20% (~2 weeks). Daily feeding was then titrated to maintain the new body weight for one week before resuming behavioral testing. Restricted feeding and body weight were maintained until the end of experimentation.
Conditioned Place Preference (CPP) apparatus
An unbiased three-compartment apparatus was used as described previously (e.g., Zheng et al., 2013). Automated data collection was accomplished through 24 infrared photo-beam detectors along the length of the test chamber (VersaMax System, Accuscan, Columbus, OH), and provided a record of horizontal activity and time spent in each compartment.
Habituation and pre-conditioning
All rats were habituated to transport and handling on at least five occasions. On the first experimental day, each rat was pre-exposed to the CPP apparatus. Pre-exposure involved placing the rat in the center compartment and allowing free movement for 20 min. Time spent in each compartment was recorded. Based on the absence of initial preference for either conditioning compartment, rats were randomly assigned to receive cocaine in one of the two compartments. One rat each in Experiments 2, 4a, and 4b expressed an unconditioned side preference (<30% or >70% time spent in one compartment). These subjects were assigned to be conditioned against their bias.
Conditioning
At least 48 hours after pre-exposure, rats underwent a series of 8 consecutive daily conditioning sessions, with the exception of Experiment 4a in which there were 12 sessions. Sessions were of 20 minutes duration, with partitions in place and animals confined to one side of the apparatus. On alternate days, rats were injected with cocaine HCl (12.0 mg/kg, i.p.) immediately prior to placement in the paired compartment. On intervening days, rats were injected with saline-vehicle (1.0 ml/kg, i.p.) before confinement to the opposite compartment.
Testing
At least 48 hours after the last conditioning session, rats were tested for CPP expression. On test days, rats were placed in the center compartment with partitions in place for 15s. Partitions were removed and rats allowed to move freely for 20 min. Additional CPP tests were conducted after a three-week interval, sometimes including a change in feeding regimen that was maintained throughout the interval and subsequent expression testing. Extinction of a CPP was defined as three consecutive test sessions in which time spent on the cocaine-paired side did not differ from time spent on the saline-paired side, as determined by t-test. To ensure a conservative approach to establishing extinction, t-tests were each one-tailed with the α-level set a at 0.05 (i.e., no correction for multiple tests); this maximized the likelihood of concluding a difference between time spent on the cocaine- and saline-paired sides of the chamber, and thus no extinction.
In experiments conducted for purposes of analyzing the relationship between NAc biochemistry and behavior, subjects were classified as CPP+ or CPP−. CPP+ was defined as having spent at least 50 sec more on the cocaine-paired side than the vehicle-paired side in the test that preceded brain harvesting (see Zheng et al., 2013).
Drugs
Cocaine HCl was obtained via the National Institute on Drug Abuse (NIDA), Research Triangle Institute, Research Triangle Park, NC, and dissolved in sterile 0.9% saline for intraperitoneal injection. 1-Naphthylacetyl spermine (NASPM; Sigma-Aldrich, St. Louis, MO) was dissolved in sterile 0.9% saline for intracerebral microinjection.
Whole-cell and synaptosomal preparations
Subjects were briefly exposed to CO2 and decapitated by guillotine. In Experiments 1 and 3, NAc was dissected from fresh brain on ice and 2-3 samples per treatment condition were pooled for fractionation. All subjects’ CPP expression in the final test was inspected and NAc samples from 2-3 rats with similar CPP expression were pooled per sample tube. The purpose of pooling was to ensure an adequate yield of synaptosomal fraction. The methods of subcellular fractionation were as described previously (e.g., Peng et al., 2014) and fractions were stored at −80°C until use.
In Experiment 2, brains were extracted and rapidly frozen in powdered dry ice. A series of 500-μm sections were cut using an IEC Minotome cryostat, and NAc core and shell were dissected using a combination of micropunch and microknife under an Olympus dissecting microscope. Tissue lysates were prepared as described previously (e.g., Zheng et al., 2013) and stored in sample buffer in aliquots at −80°C. The protein content was determined using the BCA reagent kit with bovine serum albumin as a standard (Pierce, Rockford, IL).
Western blotting
Proteins (6-10 μg/lane) were separated by electrophoresis on precast 4-12% sodium dodecyl sulfate polyacrylamide gels (Lonza, Rockland, ME). Precision Plus protein standard molecular weight markers (Bio-Rad) were loaded to estimate the size of the target proteins and to ensure complete transfer of proteins from gel to membrane. Proteins were electrophoretically transferred to Protran nitrocellulose membranes (Whatman, Mobile, AL) for 1.75 hours at a constant voltage of 100V.
Membranes were blocked for 60 minutes with Odyssey Blocking Buffer (LI-COR Biosciences, Lincoln, NE) with shaking at room temperature. Membranes were then probed overnight at 4°C using primary antibodies fo r target proteins or the protein loading control, α-tubulin. Primary antibodies were diluted in Odyssey Blocking Buffer with 0.2% Tween 20. After probing with primary antibodies and washing with TBST buffer (3 x 5 min), membranes were incubated for 1 hour at room temperature with IRDye 680RD Goat anti-Mouse IgG (H+L) and IRDye 800CW Goat anti-Rabbit IgG (H+L) (1:16,000; LI-COR Biosciences) in Odyssey Blocking Buffer with 0.2% Tween 20. Proteins were visualized using an Odyssey CLx infrared imaging system, and bands were quantified using the Image Studio software (LI-COR Biosciences).
For Experiments 1 and 3, samples from each of the four test groups were loaded in each gel in an alternating order. The pattern of alternation differed between the multiple gels run to obtain averaged results for each target protein in each test group. For Experiment 2, samples from different subjects were loaded in different orders between the multiple gels run to obtain averaged results, and separate gels were run for NAc core and shell samples. GluA1, pSer845-GluA1, and GluA2 were normalized to total tubulin in the corresponding lane. In order to account for gel-to-gel variability, the average intensity of all bands in a gel was set at 100%. GluA1 and pSer845-GluA1 are reported separately for a more complete picture.
Primary antibodies used included mouse monoclonal anti-GluA1 (1:1,000; MAB2263, Millipore, Temecula, CA); rabbit polyclonal antiphospho-Ser845-GluA1 (1:1,000; AB5849, Millipore), rabbit polyclonal anti-GluA2 (1:1,000; PA1-4659, Thermo Scientific, Rockford IL), and mouse monoclonal anti-α-tubulin (1:10,000; T6199, Sigma-Aldrich).
Surgical procedures and intracranial injections
Several days after arrival in the central animal facility, rats were anesthetized with ketamine (100 mg/kg, i.p.) and xylazine (10 mg/kg, i.p.). They were stereotaxically implanted with chronically indwelling guide cannulae. Two guide cannulae (26 ga) were placed bilaterally 2.0 mm dorsal to the injection sites in the NAc core (coordinates: 1.6 mm anterior to bregma; 2.9 mm lateral to the sagittal suture, tips angled 8°toward the m idline, 5.6 mm ventral to the skull surface). Cannula patency was maintained with occlusion stylets. The cannulae and four mounting screws were then permanently secured to the skull by flowing dental acrylic around them. Postsurgical analgesia was achieved by administration of Banamine (flunixin, 2.0 mg/kg, s.c.).
Intracerebral injections
Solutions were loaded into two 30-cm lengths of PE-50 tubing. The tubing was attached at one end to a 5 μl Hamilton syringe filled with distilled water, and at the other end to a 31-ga injector cannula, which extended 2.0 mm beyond the implanted guides. The 0.5 μl injection volumes were delivered over a period of 90 seconds at a rate of 0.05 μl/10 s. One minute following completion of the microinjection, injector needles were removed from the guides, stylets were replaced, and animals were placed in test chambers for CPP expression testing.
Histology
Subjects were euthanized with CO2 and decapitated by guillotine. Brains were removed and fixed in 10% buffered formalin for at least 48 hours. Frozen coronal sections, 40 μm thick, were cut on a Reichert-Jung Cryostat, thaw-mounted on gelatin-coated glass slides, and stained with cresyl violet. Injection sites were determined by visual inspection of sections under an Olympus SZ40 microscope. Rats whose cannula placements were not bilaterally accurate were excluded from behavioral data analysis.
Experiment 1
One aim of the present study was to obtain NAc samples from AL rats displaying a wide range of CPP test outcomes and assess whether a relationship exists between levels of pSer845-GluA1, total GluA1, and behavior. Thus, subjects remained in this study whether they displayed a CPP in the first post-conditioning test or not, and were sacrificed immediately after the 5th expression test – a time when CPP of AL rats, as a group, approached extinction in previous studies. Levels of pSer845-GluA1, total GluA1 and total GluA2 were measured with comparison between those expressing and not expressing a CPP. Because FR rats had not previously been tested using this specific protocol, a group of FR rats were included.
All subjects underwent conditioning in the AL-fed state. Following conditioning and a single CPP expression test, subjects were divided into two groups matched for CPP expression. Approximately half were assigned to the FR regimen while the others remained AL. In the initial CPP expression test, 10 of the 26 rats assigned to the AL condition and 12 of the 28 assigned to the FR condition did not express a CPP. CPP expression testing resumed only after FR subjects had stabilized at a 20% lower body weight relative to pre-restriction (~ 3 weeks later). Subjects were sacrificed immediately after removal from the CPP apparatus on the fifth expression test. In this terminal test session, 14 of the AL rats displayed a CPP (CPP+), which yielded 6 samples for biochemistry (based on pooling of 2-3 rat NAc per sample); 12 did not display a CPP (CPP−), yielding 4 samples. Thirteen of the FR rats displayed a CPP (CPP+), yielding 5 samples; 15 did not display a CPP (CPP−), yielding 6 samples. Based on the previous findings in FR subjects (Zheng et al. 2013), a unidirectional hypothesis was tested: that levels of pSer845-GluA1 and total GluA1 would be greater in CPP+ relative to CPP− subjects, and a positive correlation would exist between these AMPAR levels and magnitude of CPP.
Experiment 2
To elaborate on results of Experiment 1, a second set of AL rats were tested in Experiment 2 with separate analyses of NAc core and shell. Subjects were conditioned and tested as in Experiment 1, including the 3 week hiatus between the first CPP test and resumption of testing, with separate analyses of NAc core and shell. All subjects remained AL throughout the experiment. A total of 9 rats were tested.
Experiment 3
Results of Experiments 1 and 2, in combination with previous findings, indicated a strong association between pSer845-GluA1 in NAc core and CPP expression irrespective of diet. In order to assess whether AMPA receptor levels associated with CPP expression in Experiments 1 and 2 are localized to the synaptic membrane, Experiment 3 compared levels in the NAc synaptosomal fractions obtained from samples collected in Experiment 1.
Experiment 4
Present and prior results suggested that trafficking effects of pSer845-GluA1 may differ between the two diet groups, with GluA2-lacking AMPARs playing a role in FR rats and GluA2-containing AMPARs playing the corresponding role in AL rats. In Experiment 4, 1-napththyl acetyl spermine (NASPM), a selective antagonist of CP-AMPARs (Koike et al. 1997; Tsubokawa et al. 1995), was microinjected into NAc core prior to each CPP expression test; it was predicted that NASPM would selectively decrease CPP in FR rats.
All subjects were conditioned in the AL state. In Experiment 4a, conducted several months before Experiment 4b, a single expression test followed conditioning to confirm CPP; subjects not displaying a CPP were removed from the experiment. Subjects were then split into two groups matched for CPP expression, and all were placed on the FR regimen. Three weeks later, when subjects were stabilized at a 20% lower body weight relative to pre-restriction, expression testing resumed at 48 hr intervals with intra-NAc microinjection of NASPM (25.0 μg) or vehicle (0.9% saline) prior to each expression test. Eight subjects were injected with NASPM and 8 were injected with saline. In Experiment 4b, a single expression test again followed conditioning to confirm CPP. Subjects were then split into two groups matched for CPP expression, and all remained in home cages for 3 weeks continuing to feed on an AL basis. Expression testing resumed at 48 hr intervals with intra-NAc microinjection of NASPM (25.0 μg) or vehicle (0.9% saline) immediately preceding each expression test. Ten subjects were injected with NASPM and eleven were injected with saline.
Results
Experiment 1 AMPAR subunit levels in NAc of animals that display CPP+ vs CPP−
Prior to conditioning, the average body weight of rats in this experiment (all AL-fed at that time) was 402 ± 3.2 g. At the time of CPP test #1, which followed conditioning and preceded diet manipulation, the mean body weight of subjects in the group that would remain AL was 435 ± 4.5 g, and the mean body weight in the group to be switched to FR was 435 ± 5.7 g. When testing resumed ~3 wks later, mean body weights were 499 ± 6.3 g in the AL group and 344 ± 3.8 g in the FR group; body weights of FR subjects remained fixed for the remainder of the experiment.
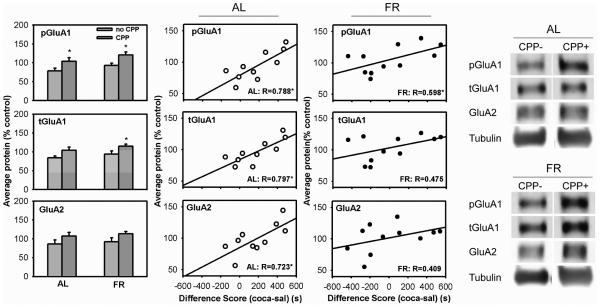
In AL rats, pSer845-GluA1 levels were higher in CPP+ relative to CPP− (t(8)=1.96, 1-tail, p<0.05). Because NAc samples from 2-3 rats were pooled for biochemical analyses, the CPP scores used for correlational analyses were the means of the 2-3 subjects whose samples were pooled. There was a strong positive correlation between pSer845-GluA1 levels and preference for the cocaine-paired side of the apparatus (r=0.788, t(8)=3.62, p<.01). Analysis of total GluA1 revealed a trend toward increased levels in the CPP+ group (t(8)=1.76, p=0.06), with a strong positive correlation between GluA1 levels and preference for the cocaine-paired side (r=0.797, t(8)=3.74, p<0.01). Levels of total GluA2 did not differ between CPP+ and CPP− groups, but there was a strong positive correlation between GluA2 levels and preference for the cocaine-paired side (r=0.723, t(8)=2.96, p<0.02) (Figure 1).
Figure 1. Levels of AMPA receptor subunits in NAc (WC) of rats expressing a CPP (CPP+) vs. rats not expressing a CPP (CPP−) in the test immediately preceding brain harvesting.
Bar graph: results (mean ±SEM) for AL (left) and FR rats (right) are normalized to tubulin; values are expressed in comparison to average intensity across all groups, which was set at 100%. Scatter plot: correlation between AMPAR protein levels and CPP difference scores (time spent in the cocaine-paired side minus time spent in the saline-paired side) in AL and FR subjects. *statistically significant, see text for p-values.
In FR animals, pSer845-GluA1 levels were higher in CPP+ relative to CPP− subjects (t(9)=2.89, p<0.01), and there was a positive correlation between pSer845-GluA1 levels and preference for the cocaine-paired side of the apparatus (r=0.598, t(9)=2.24, p<0.05). In addition, levels of total GluA1 were higher in CPP+ than CPP− (t(9)=1.96, p<0.05). These effects were seen previously in FR subjects sacrificed after the 16th expression test (Zheng et al. 2013). Correlations between total AMPAR levels and preference for the cocaine-paired side were positive but not statistically significant (Figure 1).
Expression of locomotor activity during CPP testing did not differ between feeding groups nor between subjects that did and did not display CPP. For example, total horizontal activity counts in the test session that followed diet switch (i.e., test #2) was 2293 ± 110 in FR rats and 2512 ± 138 in AL rats. Further, there was no correlation between CPP difference score and horizontal activity counts in FR (r=0.02) or AL (r=0.003) rats.
Experiment 2 AMPA receptor subunit levels in NAc core and shell of AL rats after the last test session
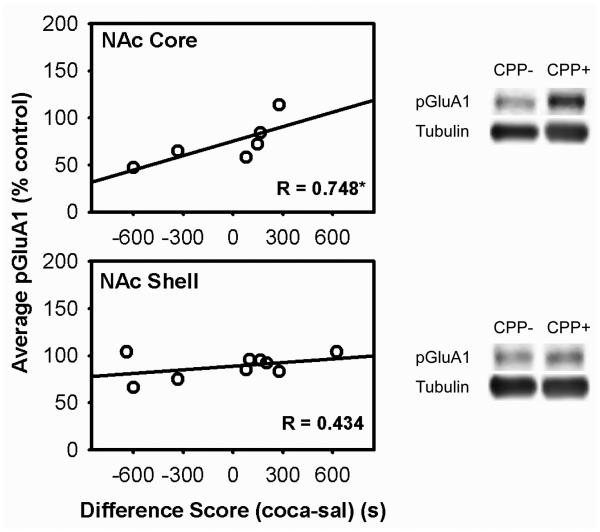
This experiment tested the prediction that the association between pSer845-GluA1 and CPP expression seen in AL rats in Experiment 1 is localized to NAc core as previously seen in FR rats (Zheng et al. 2013). Of the 9 subjects tested, 3 NAc core samples could not be analyzed due to insufficient protein. Nevertheless, regression analysis revealed a strong positive correlation between levels of pSer845-GluA1 and preference for the cocaine-paired side of the apparatus in NAc core (r=0.748, t(4)=2.25, one-tailed p<.05) but not NAc shell.
Correlations between total GluA1 and GluA2 and preference for the cocaine-paired side were positive and greater in the core than shell but none was statistically significant (Figure 2).
Figure 2. pSer845-GluA1 levels in NAc core and shell in relation to difference score in the cocaine CPP expression test that immediately preceded brain harvesting in AL rats.
Scatter plots depict correlation between levels of pSer845-GluA1 in nucleus accumbens core (top), shell (bottom) and CPP difference scores (time spent in the cocaine-paired side minus time spent in the saline-paired side). pSer845-GluA1 was normalized to tubulin.
Experiment 3 AMPAR subunit levels in the NAc synaptosomal fraction
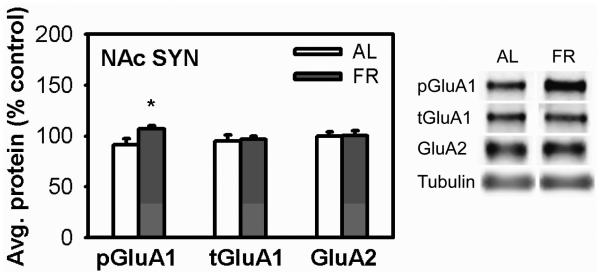
Unlike the whole cell preparation analyzed in Experiment 1, there were no differences in pSer845-GluA1 or total GluA1 between CPP+ and CPP− subjects in either the AL or FR groups. Further, there were no significant correlations between AMPAR levels and CPP expression. However, the level of pSer845-GluA1 was higher in FR than AL subjects (t(20)=2.42, p<0.05). Levels of total GluA1 and GluA2 did not differ between diet groups (Figure 3).
Figure 3. AMPA receptor levels in the synaptosomal fraction of NAc of AL and FR rats after CPP testing.
Results (mean ±SEM) are normalized to tubulin. *p<0.05.
Experiment 4a Intra-NAc core NASPM microinjection in FR rats
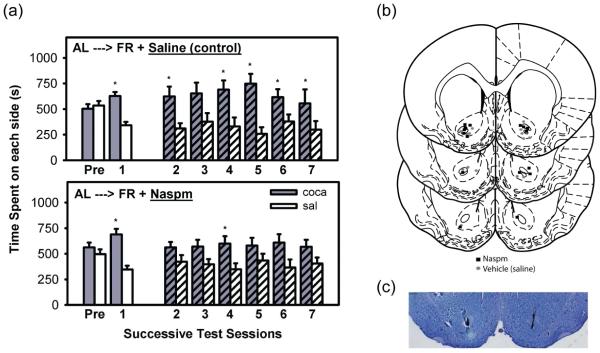
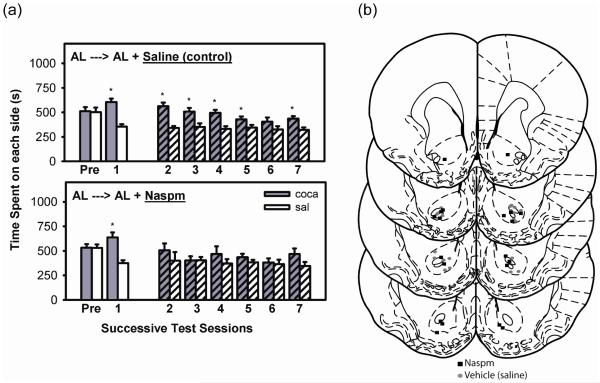
During pre-exposure (Pre), rats did not display a preference for either side of the conditioning chamber (Figure 4a). In the expression test conducted 48 hrs after the final conditioning session (test 1), rats displayed a strong preference for the cocaine-paired side (t(15)=5.95, p<0.001). All animals were then maintained on FR for 3 weeks to attain and stabilize at the target body weight. Upon resumption of testing, the group that received intra-NAc saline prior to each expression test displayed a cocaine-side preference (t(7)=2.22, p<0.05) and the group that received intra-NAc NASPM did not. With continued testing, fulfillment of extinction criterion was established in the NASPM-treated group, as defined by absence of a cocaine-side preference on the 5th, 6th, and 7th test days (p>0.05 for all test days). A 2-way ANOVA (side x day) with repeated measures on both variables confirmed that the NASPM-treated group failed to display a CPP over tests 2-7 (F(1,7)=2.92, p>0.10). In contrast, the saline-treated group continued to display a CPP over tests 2-7 (F(1,7)=7.65, p<0.05). Microinjection sites were histologically confirmed to lie within the NAc core or on its border with the shell or olfactory tubercle (Figure 4b). One FR and one AL rat were rejected from this study based on histology.
Figure 4. CPP expression in NASPM- and saline-injected FR rats.
(a) CPP expression in FR rats microinjected with saline (top) or NASPM (bottom) in the NAc core immediately prior to each expression test. Mean (±SEM) time spent (s) on the cocaine and saline-paired sides of the CPP apparatus are shown. *Results of t-tests performed to determine extinction; p<0.05. (b) Bilateral NAc microinjection sites in the NASPM (square) and saline (circle)-injected rats. (c) A representative stained coronal section displaying bilateral microinjection sites in NAc core.
Experiment 4b Intra-NAc core NASPM microinjection in AL rats
During pre-exposure (Pre), rats did not display a preference for either side of the conditioning chamber (Figure 5a). In the expression test conducted 48 hrs after the final conditioning session, rats displayed a strong preference for the cocaine-paired side (t(20)=6.25, p<0.001). All animals remained AL-fed in their home cages for three weeks in order to match the delay to expression testing in Experiment 4a. Upon resumption of testing, the group that received intra-NAc saline prior to each expression test displayed a cocaine-side preference (t(10)=5.57, p<0.001) and the group that received intra-NAc NASPM did not. While continued testing on days 3 and 4 revealed that the NASPM-injection group did not display a place preference by t-test, thereby fulfilling the criterion for CPP extinction, testing was continued to match testing of the FR subjects in Experiment 4a. A 2-way ANOVA (side x day) with repeated measures on both variables confirmed that over tests 2-7, the NASPM-injection group failed to display a CPP (F(1,9)=1.17, p>0.10). In contrast, the saline-injection group continued to display a CPP over tests 2-7 (F(1,10)=20.7, p<0.001). Microinjection sites were histologically confirmed to lie within the NAc core or on its border with the shell or olfactory tubercle (Figure 5b).
Figure 5. CPP expression in NASPM- and saline-injected AL rats.
(a) CPP expression in AL rats microinjected with saline (top) or NASPM (bottom) in the NAc core immediately prior to each expression test. Mean (±SEM) time spent (s) on the cocaine and saline-paired sides of the CPP apparatus are shown. *Results of t-tests performed to determine extinction; p<0.05 (b) Bilateral NAc microinjection sites in the NASPM (square) and saline (circle)-injected rats.
Discussion
Weight loss dieting is prevalent in the US (Bish et al. 2005; Rideout and Barr 2009; Serdula et al. 1999; Williamson et al. 1992), and calorie restriction is the primary method used (Kruger et al. 2004). Additionally, 20 million women and 10 million men suffer from a clinically significant eating disorder at some time in their life (Wade et al. 2011). For individuals with an eating disorder, the prevalence of substance abuse is 5-6 times higher than in the general population (Root et al. 2010; Merikangas and McClair 2012). Moreover, one-third of callers to the National Cocaine Hotline who met criteria for cocaine abuse also met criteria for bulimia and/or anorexia nervosa (Jonas and Gold 1987). Using a CPP paradigm, it was previously observed that FR in rats increases the incentive effect of a cocaine-paired environment in much the same way that it increases the rewarding effect of cocaine itself (Liu et al. 2011; Zheng et al. 2012; Zheng et al. 2013). This observation suggested that FR may not only increase vulnerability to initial drug use by enhancing reward magnitude but may increase relapse risk by enhancing the incentive effect of contexts associated with the subjective effects of cocaine. This possibility receives support from a finding that FR increased cue-induced heroin seeking in rats (D'Cunha et al. 2013). Evaluation of BDNF, ERK signaling, and AMPAR mechanisms in several brain regions led to identification of elevated pSer845-GluA1 and total GluA1 in NAc core as characteristic of FR rats that express a persistent CPP (Liu et al. 2011; Zheng et al. 2013). The main goals of the present study were to determine whether these AMPAR mechanisms are unique to FR rats that express cocaine CPP, and whether NAc core microinjection of an antagonist of CP-AMPARs selectively blocks CPP expression in FR rats.
The purpose of Experiment 1 was to capture NAc at a time when AL rats are expressing a range of CPP test outcomes in order to determine whether there is an association between CPP expression and levels of both pSer845-GluA1 and total GluA1 as previously seen in FR rats tested out to day 16 (Zheng et al. 2013). Similar to the previously tested FR rats, AL rats expressing a CPP showed higher levels of pSer845-GluA1 than those not expressing a CPP, and levels correlated with magnitude of preference for the cocaine-paired side. Whereas FR rats that expressed a CPP in the previous study also had elevated levels of total GluA1, AL rats displaying CPP showed a positive correlation between levels of both GLuA1 and GluA2 and preference for the cocaine-paired side. Experiment 1 included a group of FR subjects that differed from those in the previous study in two respects; a larger proportion did not display CPP in the first post-conditioning test (i.e., pre-FR), and they were sacrificed after the 5th, rather than 16th, expression test in order to match the AL group. The previous key findings were replicated: FR rats expressing a CPP showed higher levels of pSer845-GluA1 and total GluA1 in NAc than FR rats not expressing CPP, and levels of pSer845-GluA1 correlated with preference for the cocaine-paired side. Thus, results of Experiment 1 indicate that pSer845-GluA1 is associated with CPP expression irrespective of diet. A caveat is that the pSer845-GluA1 associated with CPP expression in FR rats was previously localized to NAc core, and Experiment 1 results were obtained in whole NAc. Consequently, Experiment 2 analyzed the NAc core and shell separately in AL subjects. Results supported mechanistic similarity between AL and FR rats in so far as AL rats in Experiment 2 displayed a strong positive correlation between preference for the cocaine-paired side and levels of pSer845-GluA1 in NAc core, but not shell.
If the more persistent cocaine CPP of FR relative to AL rats is mediated by AMPARs, the present and previous results suggest two possible mechanistic bases. The first is that FR upregulates basal or context-induced phosphorylation of GluA1 at Ser845 which may be sufficient to increase behavioral responsiveness to a psychostimulant-paired environment (Li et al. 2011). The second is that downstream of Ser845 phosphorylation, stabilization of CP-AMPARs in the membrane and synaptic incorporation of GluA1-containing AMPARs are greater in FR than AL and/or lead to insertion of AMPARs with different subunit composition in the two diet groups. The elevated levels of GluA1 in FR rats expressing CPP, and the positive correlation between levels of both GluA1 and GluA2 and CPP in AL rats raise the possibility that context-induced phosphorylation increases trafficking of GluA2-lacking AMPARs in FR rats and GluA1/GluA2 heteromers in AL rats, based on evidence that GluA1 trafficking rules prevail in both cases (Shi et al. 2001). A difference in AMPAR composition could explain the difference in CPP expression in as much as GluA2-lacking AMPARs enhance the summation of synaptic responses after a period of high input activity, and trigger signaling cascades involved in synaptic strengthening (Barry and Ziff 2002; Malinow 2003).
With regard to the first potential mechanism, previous studies have demonstrated that FR increases stimulus-induced phosphorylation of GluA1 at Ser845 in NAc (Carr et al. 2010; Peng et al. 2014). In fact, results obtained in Experiment 3 indicate that immediately following a CPP test, levels of pSer845-GluA1 in the NAc synaptosomal fraction are higher in FR than AL rats. Thus, an increase in synaptic pSer845-GluA1 seems a candidate mechanism underlying the more persistent cocaine CPP in FR rats. With regard to the second potential mechanism, the current biochemical measurements in the synaptosomal fraction did not reveal differences in levels of specific AMPAR subunits in FR relative to AL rats, or rats expressing CPP relative to rats not expressing CPP. However, the current methods are not capable of distinguishing differences in subunit composition. Moreover, it is possible that stimulus-induced AMPAR trafficking that accounts for differences in CPP expression is transient, stimulus-bound, and not subject to capture once subjects are removed from the CPP apparatus for brain harvesting (Choquet 2010). Consequently, a pharmacological approach was used to test whether CP-AMPARs are differentially involved in CPP expression in the two diet groups. As had been predicted, the CPP of FR subjects was eliminated when each expression test was preceded by intra-NAc core microinjection of NASPM. However, the relatively weak but significant CPP displayed by AL subjects was also eliminated by NASPM. These findings indicate that AMPARs in NAc core are necessary for expression of cocaine CPP, and this is the first evidence suggesting that CP-AMPARs mediate this behavior. What is not known is whether these receptors are in the synaptic membrane under basal conditions, whether they are incorporated as a consequence of conditioning, or whether they are rapidly trafficked into the membrane in response to context exposure. A further question regarding the time and circumstance of synaptic incorporation relates to the 3 week interval between conditioning and testing. The 3 week interval was necessary to assess the effect of FR during the expression phase of cocaine CPP. To control for passage of time, subjects that remained AL throughout were also subject to this 3 week interval. Three weeks is the approximate minimum interval for incubation of cocaine craving, which is accompanied by increased surface expression of CP-AMPARs in NAc (Conrad et al. 2008; Ferrario et al. 2011). It is conceivable that 3 weeks withdrawal from intermittent cocaine treatment contributed to CP-AMPAR involvement in CPP expression.
In the whole cell preparation of NAc examined in Experiment 1, there was an association between high levels of GluA1 and CPP expression in FR rats, replicating a previous result in NAc core (Zheng et al. 2013). In AL rats, however, there were equally strong associations between levels of GluA1 and GluA2 with CPP expression. These observations led to the suggestion that context exposure trafficks GluA1 homomers to the synapse in FR rats and GluA1/GluA2 heteromers to the synapse in AL rats, accounting for the weaker and less persistent cocaine CPP in the latter. The inhibitory effect of NASPM in both diet groups does not necessarily rule this out. It may be that the balance between activity-dependent trafficking of the two AMPAR types does differ between diet groups, and accounts for the difference in magnitude and persistence of CPP. Clearly, the present study raises a number of important questions for future investigation. Nevertheless, based on the present results and the previous related results, the current working hypothesis is that phosphorylation of GluA1 at Ser845 in NAc core underlies the expression and magnitude of cocaine CPP, that it does so by stabilizing or trafficking CP-AMPARs to the synaptic membrane, and that the more persistent cocaine CPP of FR rats results from increased stimulus-induced phosphorylation of GluA1 at Ser845 in the synaptic membrane. From the standpoint of translation, the present study provides another set of findings to support the potential value of AMPAR pharmacotherapies in drug addiction (Bowers et al. 2010) and its interaction with disordered eating (Guardia et al. 2011).
Acknowledgments
This research was supported by DA03956 from NIDA/NIH.
References
- Barry MF, Ziff EB. Receptor trafficking and the plasticity of excitatory synapses. Curr Opin Neurobiol. 2002;12:279–86. doi: 10.1016/s0959-4388(02)00329-x. [DOI] [PubMed] [Google Scholar]
- Bish CL, Blanck HM, Serdula MK, Marcus M, Kohl HW, 3rd, Khan LK. Diet and physical activity behaviors among Americans trying to lose weight: 2000 Behavioral Risk Factor Surveillance System. Obes Res. 2005;13:596–607. doi: 10.1038/oby.2005.64. [DOI] [PubMed] [Google Scholar]
- Bowers MS, Chen BT, Bonci A. AMPA receptor synaptic plasticity induced by psychostimulants: the past, present, and therapeutic future. Neuron. 2010;67:11–24. doi: 10.1016/j.neuron.2010.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cabeza de Vaca S, Carr KD. Food restriction enhances the central rewarding effect of abused drugs. J Neurosci. 1998;18:7502–10. doi: 10.1523/JNEUROSCI.18-18-07502.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr KD, Chau LS, Cabeza de Vaca S, Gustafson K, Stouffer M, Tukey DS, Restituito S, Ziff EB. AMPA receptor subunit GluR1 downstream of D-1 dopamine receptor stimulation in nucleus accumbens shell mediates increased drug reward magnitude in food-restricted rats. Neuroscience. 2010;165:1074–86. doi: 10.1016/j.neuroscience.2009.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr KD, Tsimberg Y, Berman Y, Yamamoto N. Evidence of increased dopamine receptor signaling in food-restricted rats. Neuroscience. 2003;119:1157–67. doi: 10.1016/s0306-4522(03)00227-6. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Meisch RA. Increased drug-reinforced behavior due to food-deprivation. Advances in Behavioral Pharmacology. 1984;4:47–88. [Google Scholar]
- Childress AR, Hole AV, Ehrman RN, Robbins SJ, McLellan AT, O'Brien CP. Cue reactivity and cue reactivity interventions in drug dependence. NIDA Res Monogr. 1993;137:73–95. [PubMed] [Google Scholar]
- Choquet D. Fast AMPAR trafficking for a high-frequency synaptic transmission. Eur J Neurosci. 2010;32:250–60. doi: 10.1111/j.1460-9568.2010.07350.x. [DOI] [PubMed] [Google Scholar]
- Conrad KL, Tseng KY, Uejima JL, Reimers JM, Heng LJ, Shaham Y, Marinelli M, Wolf ME. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature. 2008;454:118–21. doi: 10.1038/nature06995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Cunha TM, Sedki F, Macri J, Casola C, Shalev U. The effects of chronic food restriction on cue-induced heroin seeking in abstinent male rats. Psychopharmacology (Berl) 2013;225:241–50. doi: 10.1007/s00213-012-2810-1. [DOI] [PubMed] [Google Scholar]
- Esteban JA, Shi SH, Wilson C, Nuriya M, Huganir RL, Malinow R. PKA phosphorylation of AMPA receptor subunits controls synaptic trafficking underlying plasticity. Nat Neurosci. 2003;6:136–43. doi: 10.1038/nn997. [DOI] [PubMed] [Google Scholar]
- Ferrario CR, Loweth JA, Milovanovic M, Ford KA, Galinanes GL, Heng LJ, Tseng KY, Wolf ME. Alterations in AMPA receptor subunits and TARPs in the rat nucleus accumbens related to the formation of Ca(2+)-permeable AMPA receptors during the incubation of cocaine craving. Neuropharmacology. 2011;61:1141–51. doi: 10.1016/j.neuropharm.2011.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guardia D, Rolland B, Karila L, Cottencin O. GABAergic and glutamatergic modulation in binge eating: therapeutic approach. Curr Pharm Des. 2011;17:1396–409. doi: 10.2174/138161211796150828. [DOI] [PubMed] [Google Scholar]
- He K, Song L, Cummings LW, Goldman J, Huganir RL, Lee HK. Stabilization of Ca2+-permeable AMPA receptors at perisynaptic sites by GluR1-S845 phosphorylation. Proc Natl Acad Sci U S A. 2009;106:20033–8. doi: 10.1073/pnas.0910338106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonas JM, Gold MS. Naltrexone treatment of bulimia: clinical and theoretical findings linking eating disorders and substance abuse. Adv Alcohol Subst Abuse. 1987;7:29–37. [PubMed] [Google Scholar]
- Jonas JM, Gold MS, Sweeney D, Pottash AL. Eating disorders and cocaine abuse: a survey of 259 cocaine abusers. J Clin Psychiatry. 1987;48:47–50. [PubMed] [Google Scholar]
- Koike M, Iino M, Ozawa S. Blocking effect of 1-naphthyl acetyl spermine on Ca(2+)-permeable AMPA receptors in cultured rat hippocampal neurons. Neurosci Res. 1997;29:27–36. doi: 10.1016/s0168-0102(97)00067-9. [DOI] [PubMed] [Google Scholar]
- Kruger J, Galuska DA, Serdula MK, Jones DA. Attempting to lose weight: specific practices among U . adults. Am J Prev Med. 2004;26:402–6. doi: 10.1016/j.amepre.2004.02.001. [DOI] [PubMed] [Google Scholar]
- Li D, Herrera S, Bubula N, Nikitina E, Palmer AA, Hanck DA, Loweth JA, Vezina P. Casein kinase 1 enables nucleus accumbens amphetamine-induced locomotion by regulating AMPA receptor phosphorylation. J Neurochem. 2011;118:237–47. doi: 10.1111/j.1471-4159.2011.07308.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Zheng D, Peng XX, Cabeza de Vaca S, Carr KD. Enhanced cocaine-conditioned place preference and associated brain regional levels of BDNF, p-ERK1/2 and p-Ser845-GluA1 in food-restricted rats. Brain Res. 2011;1400:31–41. doi: 10.1016/j.brainres.2011.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malinow R. AMPA receptor trafficking and long-term potentiation. Philos Trans R Soc Lond B Biol Sci. 2003;358:707–14. doi: 10.1098/rstb.2002.1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Man HY, Sekine-Aizawa Y, Huganir RL. Regulation of {alpha}-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor trafficking through PKA phosphorylation of the Glu receptor 1 subunit. Proc Natl Acad Sci U S A. 2007;104:3579–84. doi: 10.1073/pnas.0611698104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangiavacchi S, Wolf ME. D1 dopamine receptor stimulation increases the rate of AMPA receptor insertion onto the surface of cultured nucleus accumbens neurons through a pathway dependent on protein kinase A. J Neurochem. 2004;88:1261–71. doi: 10.1046/j.1471-4159.2003.02248.x. [DOI] [PubMed] [Google Scholar]
- Merikangas KR, McClair VL. Epidemiology of substance use disorders. Hum Genet. 2012;131:779–89. doi: 10.1007/s00439-012-1168-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orsini CA, Ginton G, Shimp KG, Avena NM, Gold MS, Setlow B. Food consumption and weight gain after cessation of chronic amphetamine administration. Appetite. 2014;78:76–80. doi: 10.1016/j.appet.2014.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng XX, Cabeza de Vaca S, Ziff EB, Carr KD. Involvement of nucleus accumbens AMPA receptor trafficking in augmentation of D-amphetamine reward in food-restricted rats. Psychopharmacology (Berl) 2014;231:3055–63. doi: 10.1007/s00213-014-3476-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rideout CA, Barr SI. "Restrained eating" vs "trying to lose weight": how are they associated with body weight and tendency to overeat among postmenopausal women? J Am Diet Assoc. 2009;109:890–3. doi: 10.1016/j.jada.2009.02.009. [DOI] [PubMed] [Google Scholar]
- Root TL, Pisetsky EM, Thornton L, Lichtenstein P, Pedersen NL, Bulik CM. Patterns of co-morbidity of eating disorders and substance use in Swedish females. Psychol Med. 2010;40:105–15. doi: 10.1017/S0033291709005662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- See RE. Neural substrates of conditioned-cued relapse to drug-seeking behavior. Pharmacol Biochem Behav. 2002;71:517–29. doi: 10.1016/s0091-3057(01)00682-7. [DOI] [PubMed] [Google Scholar]
- Serdula MK, Mokdad AH, Williamson DF, Galuska DA, Mendlein JM, Heath GW. Prevalence of attempting weight loss and strategies for controlling weight. JAMA. 1999;282:1353–8. doi: 10.1001/jama.282.14.1353. [DOI] [PubMed] [Google Scholar]
- Shi S, Hayashi Y, Esteban JA, Malinow R. Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell. 2001;105:331–43. doi: 10.1016/s0092-8674(01)00321-x. [DOI] [PubMed] [Google Scholar]
- Tsubokawa H, Oguro K, Masuzawa T, Nakaima T, Kawai N. Effects of a spider toxin and its analogue on glutamate-activated currents in the hippocampal CA1 neuron after ischemia. J Neurophysiol. 1995;74:218–25. doi: 10.1152/jn.1995.74.1.218. [DOI] [PubMed] [Google Scholar]
- Wade TD, Keski-Rahkonen A, Hudson J. Epidemiology of eating disorders. In: Tsuang MT, Tohen M, editors. Textbook in Psychiatric Epidemiology. Wiley; New York: 2011. pp. 343–60. [Google Scholar]
- Williamson DF, Serdula MK, Anda RF, Levy A, Byers T. Weight loss attempts in adults: goals, duration, and rate of weight loss. Am J Public Health. 1992;82:1251–7. doi: 10.2105/ajph.82.9.1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng D, Cabeza de Vaca S, Carr KD. Food restriction increases acquisition, persistence and drug prime-induced expression of a cocaine-conditioned place preference in rats. Pharmacol Biochem Behav. 2012;100:538–44. doi: 10.1016/j.pbb.2011.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng D, Liu S, Cabeza de Vaca S, Carr KD. Effects of time of feeding on psychostimulant reward, conditioned place preference, metabolic hormone levels, and nucleus accumbens biochemical measures in food-restricted rats. Psychopharmacology (Berl) 2013;227:307–20. doi: 10.1007/s00213-013-2981-4. [DOI] [PMC free article] [PubMed] [Google Scholar]