Introduction
Erectile dysfunction (ED) is defined as the inability to achieve and maintain a penile erection adequate for satisfactory sexual intercourse.1 Up to 150 million men worldwide suffer from ED and this figure is likely to double by the year 2025.2
A number of studies have attempted to characterise the true prevalence of ED. In a Danish study, Ventegodt reported that 5.4% of all patients had a decreased ability to achieve an erection.3 The prevalence was reported to be highest (18%) in those aged over 58 years. The Massachusetts Male Aging Study (MMAS)4 reported the results of a regional survey of 1709 men aged 40–69 years. In this study 52% reported some degree of ED, with 10% having complete ED. Moreover, the results suggest that the probability of complete ED at age 70 was threefold compared to that at age 40; the probability of moderate ED was two-fold.
Physiology of penile erection
Penile erection is a complex neurovascular event. The degree of contraction or relaxation of the cavernosal smooth muscle determines the degree of tumescence or detumescence.5 The balance between the contractile and relaxant factors is known to be controlled by both central and peripheral mechanisms and involves the interaction of three different systems:6 (a) the central nervous system (CNS); (b) the peripheral nervous system; and (c) the vascular and cavernosal smooth muscle in the penis.
The CNS
The CNS coordinates incoming sensory information from a variety of sources which may be visual, auditory, cognitive/imaginative, tactile or olfactory. The central pathways integrating these inputs and controlling erectile function are complex and only partially understood. However, there is strong evidence to support the involvement of the paraventricular nucleus (PVN) and the medial pre-optic area (MPOA) within the hypothalamus in the control of erectile function. The MPOA has been postulated to be an integrative centre that collects the input and redistributes to other structures within the CNS such as the PVN. The PVN in turn has been suggested to activate selective autonomic pathways resulting in an erection.7 The neurons from the PVN have been reported to project onto the spinal cord either directly or via the median forebrain bundle, pons and medulla. The descending pathways from the PVN to the spinal cord have been reported to contain a variety of neurotransmitters such as oxytocin, vasopressin, encephalin and dopamine.7
The peripheral nervous system
Within the spinal cord, there are various specific areas which contain integral components of the erectile system. These are known as the “erection centres” (Figure 1). The thoraco-lumbar erection centre is located between T1 and L2 and gives rise to the sympathetic outflow pathway. This connects to the urogenital tract via the pelvic, cavernosal and pudendal nerves. The sacral erection centre is located between the S2 and S4 segments of the spinal cord and gives rise to the parasympathetic outflow pathway. These fibres reach the penis via the pelvic, cavernosal and pudendal nerves. Furthermore, the penis receives dense somatic input from sensory branches of the dorsal nerve, a branch of the pudendal nerve.5
Figure 1.
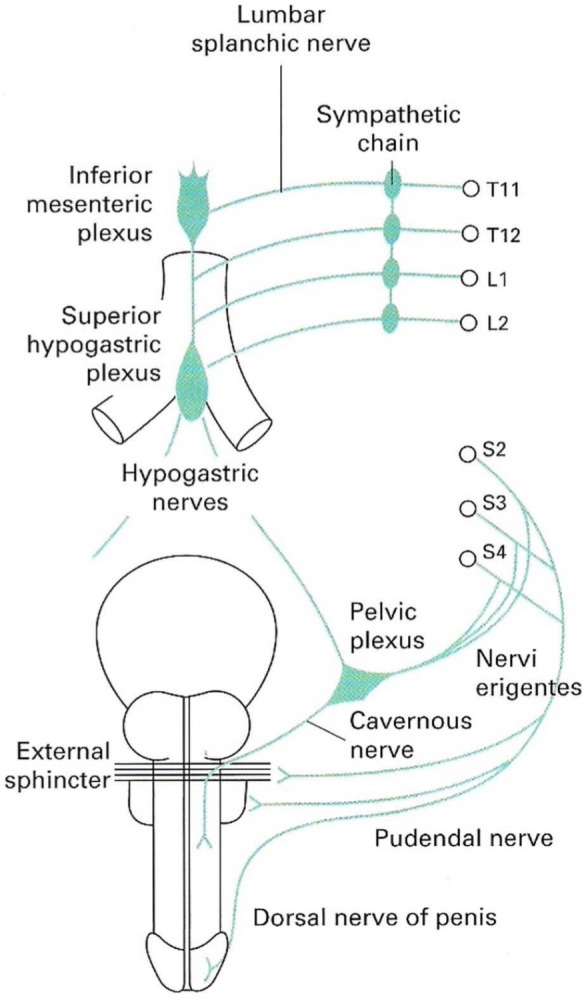
The nerve supply of the penis. From Eardley and Sethia.8
The individual nerves innervating the penis may contain a number of different neurotransmitters and as a result the nerves are categorised as either being adrenergic or cholinergic according to the predominant transmitter present. However, non-adrenergic non-cholinergic (NANC) neurotransmitters may be found and indeed be co-localised with either adrenergic or cholinergic nerves. Nitric oxide (NO) is one of the NANC neurotransmitters which has now been widely accepted to be the major mediator eliciting relaxation of the penile smooth muscle.9
During sexual arousal, NO has been reported to be released from parasympathetic nerve terminals,10 and these nerves are therefore called nitrergic nerves.11 NO release results in relaxation of the cavernosal smooth muscle and vasodilation. Simultaneous compression of the subtunical venules results in an erection. Noradrenaline released from sympathetic nerves causes contraction of the blood vessels and smooth muscle of the corpus cavernosum, thus leading to detumescence of the penis. Erection of the penis is therefore regulated by a balance between pro- and anti-erectile mediators (Figure 2). Studies with human corpus cavernosum suggest that when the two systems are simultaneously active, the nitrergic system is dominant over the sympathetic system.12
Figure 2.
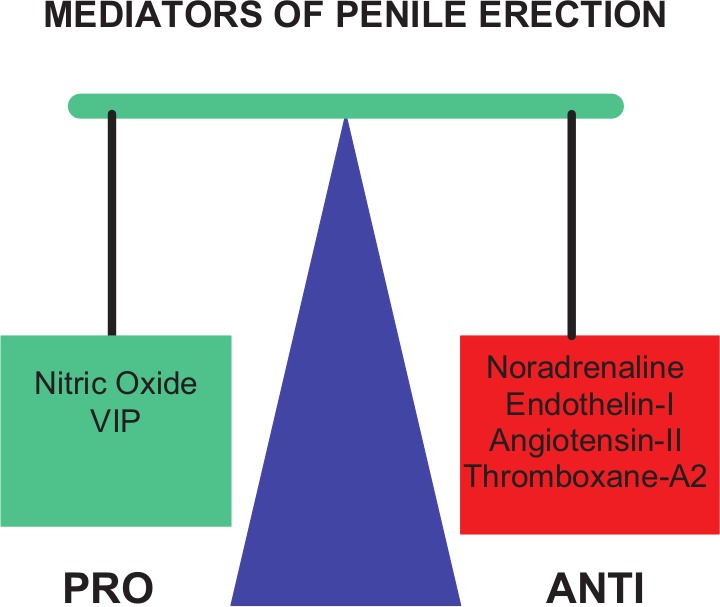
Penile erection is regulated by two opposing systems: pro-erectile mediators such as nitric oxide (NO) and vasoactive intestinal peptide (VIP) and anti-erectile mediators such as noradrenaline (NA), endothelin-I, angiotensin II and thromboxane A2.
Vascular and cavernosal smooth muscle in the penis
The human penis is composed of paired corpora cavernosa and the single corpus spongiosum (Figure 3). The corpus cavernosum consists of a meshwork of sinusoidal spaces lined by endothelial cells.5 In order for an erection to occur, relaxation of penile smooth muscle is required to allow blood to flow into the penile structures. The resulting increase in intracavernosal pressure (ICP) leads to compression of the subtunical venules against the tunica albuginea.5 This process reduces venous drainage from the corpora cavernosa and increases pressure within the corpora, resulting in an erection. In full rigidity the ICP reaches values considerably higher than systemic (systolic) blood pressure with the contribution of the skeletal muscles of the pelvic floor.
Figure 3.
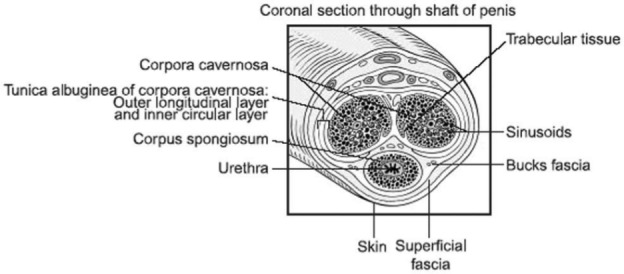
Cross-section through shaft of penis demonstrating the sinusoids, subtunical venules and tunica albuginea. From Eardley and Sethia.8
Relaxant factors: the NO-soluble guanylate cyclase (sGC)-cyclic guanosine monophosphate (cGMP) pathway
In 1990 Ignarro et al.13 reported that electrical field stimulation (EFS) of isolated strips of rabbit corpus cavernosum resulted in the endogenous generation and release of NO, nitrite and cGMP. These were the first published results to suggest that penile erection is mediated by NO generated in response to NANC stimulation.
Immuno-histochemical studies have demonstrated that the enzyme neuronal nitric oxide synthase (nNOS) is present in the nerve fibres of the pelvic plexus, corpus cavernosum and around blood vessels.14
NO may be released from both the endothelium via eNOS and the nitrergic nerves via nNOS. Nitrergic-derived NO may be functionally more important, as nitrergic relaxation of the corpus cavernosum has been reported to not require a functional endothelium after removal by either physical15 or chemical means.15,16
Once released, NO exerts its action on smooth muscle cells by activating the enzyme sGC. The activation of sGC results in an increase in intracellular cGMP concentrations (Figure 4).17 This in turn activates a number of second messenger systems which ultimately result in smooth muscle relaxation.
Figure 4.
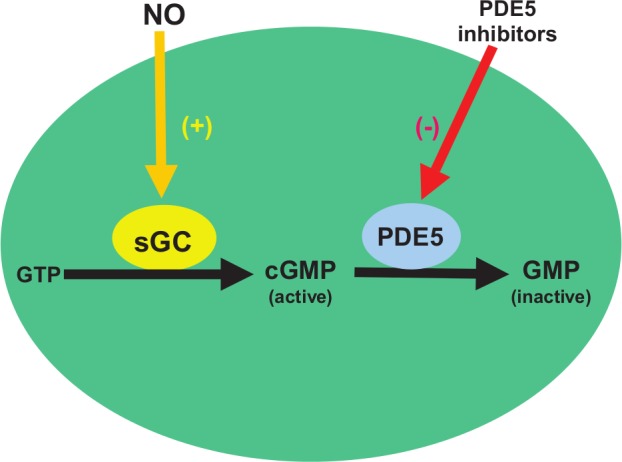
Nitric oxide (NO) released from nitrergic nerves and endothelium stimulates smooth muscle relaxation by activation of soluble guanylate cyclase (sGC), which in turn catalyses the conversion of guanine triphosphate (GTP) to the active intracellular second messenger cyclic guanosine monophosphate (cGMP). cGMP is then metabolised by phosphodiesterase type 5 (PDE5) to inactive GMP.
Pathophysiology of erectile dysfunction
ED can be caused by either psychogenic or organic factors; however, in many patients the disorder is of mixed aetiology with both factors contributing. The psychogenic component of ED is reported to be especially important in younger men (aged less than 35 years)18 and in elderly men who start a relationship with a new partner. Diseases which become more prevalent with age such as diabetes and vascular disease are major risk factors in the ageing male. It has been reported that in patients older than 50, up to 50% may have ED secondary to vascular disease. The presence of ED of any aetiology is itself associated with psychological distress. This of course may in turn reduce the probability of achieving satisfactory erectile function.
Endothelial dysfunction
As ED and coronary artery disease share common risk factors, the concept of endothelial dysfunction has developed. Here ED is considered another manifestation of vascular disease specific to small vessels. ED can be considered to be an early manifestation of systemic endothelial dysfunction.19
It is now well established that ED often precedes and predisposes subsequent atherosclerosis. Moreover, endothelial dysfunction is a reflection of the loss of NO activity or biosynthesis at the endothelial level. This is associated with vasoconstriction, coagulation, leucocyte adhesion and smooth muscle cell hyperplasia, which is central to the process of atherosclerosis. The inhibition of eNOS (via impaired hydrolysis of dimethyl arginine) and the uncoupling of eNOS activity increase the oxidative stress in the endothelial cells. This in turn results in further oxidative catabolism of NO and formation of peroxynitrite.
In diabetes-associated endothelial dysfunction, elevated free fatty acids which are seen in patients with insulin resistance may induce endothelial dysfunction through the activation of protein kinase C (PKC), the increased production of reactive oxygen species (ROS), elevation in triglyceride and low-density lipoprotein (LDL), and decrease in high-density lipoprotein (HDL) levels.20 More recent evidence suggests that the effects of hyperglycaemia and insulin resistance on endothelial cells are additive, since defects in both glucose and lipid metabolism produce similar effects with the resultant decrease in endothelial NO availability.21 The association between metabolic syndrome, insulin resistance and obesity and ED in men are now well characterised and understood.22
Diabetes mellitus
Many epidemiologic studies have reported an increased risk of ED in diabetic men.23 The prevalence of ED has been reported to affect between 35% to 50% of diabetic patients.24 Furthermore, a positive relationship was demonstrated between ED, poor metabolic control and age.25 These results indicate that diabetes is a significant risk factor for the development of ED.
In diabetes, the severity of ED has been demonstrated to be related to both the severity26 and duration of diabetes.27 However, it is likely that the aetiology of ED in diabetes is multifactorial. It is now well established that there is a higher incidence of peripheral neuropathy, autonomic neuropathy, microangiopathy and arterial insufficiency in diabetic patients with ED than in potent diabetic patients.
The proposed mechanisms of ED in diabetics include: elevated advanced glycation end-products (AGEs) and increased levels of oxygen free radicals,28 impaired (NO synthesis, decreased and impaired cGMP-dependent kinase-1 (PKG-1),29 increased endothelin B (ETB) receptor binding sites and ultrastructural changes,30 upregulated RhoA/Rho-kinase pathway,31 endothelial dysfunction32 and NO-dependent selective nitrergic nerve degeneration.33
Therapeutic options for ED
Lifestyle modifications
The identification of specific risk factors associated with ED provides an opportunity for conservative measures in patients with mild to moderate ED. Cessation of smoking, weight loss and exercise are associated with an improvement in erectile function, as well as improving endothelial function and reducing long-term cardiovascular risk.22
Pharmacological treatments for ED
There are a number of options available for the management of ED. They include oral agents, intracavernosal injection (papaverine, phentolamine, prostaglandin E1, vasoactive intestinal polypeptide (VIP)), transurethral vasoactive agents (prostaglandin E1), vacuum erection devices, penile revascularisation surgery and insertion of a penile prosthesis.
Oral agents are the least invasive option and are the most accepted form of first-line treatment (Table 1).
Table 1.
Oral agents for the treatment of male erectile dysfunction.
| Oral treatments | Mechanism of action |
|---|---|
| Sildenafil citrate (ViagraTM) | PDE5 inhibitor |
| Tadalafil (CialisTM) | PDE5 inhibitor |
| Vardenafil hydrochloride (LevitraTM) | PDE5 inhibitor |
| Avanafil (StendraTM) | PDE5 inhibitor |
| Udenafil | PDE5 inhibitor |
| Yohimbine | α-adrenoceptor antagonist |
| Apomorphine (UprimaTM, IxenseTM and TaluvianTM) | Dopamine receptor agonist |
PDE5: phosphodiesterase type 5.
PDE5 inhibitors
The second messenger cGMP is metabolised to GMP by a superfamily of enzymes called phophodiesterases (PDEs). Among all of the PDEs, PDE5, 6 and 9 are specific for cGMP, and PDE5 is the predominant PDE found in the corpus cavernosum.34
Sildenafil (ViagraTM), vardenafil (LevitraTM) and tadalafil (CialisTM) are the currently available PDE5 inhibitors. Vardenafil is now also available as an orodispersible tablet (ODT).35 Newer PDE5 inhibitors avanafil (StendraTM) and udenafil have recently been approved by the Food and Drug Administration (FDA) in the United States (US). Tadalafil (once daily 5mg) has also recently been licensed for the treatment of both ED and symptoms related to benign prostatic hyperplasia (BPH) by the FDA in the US.36 Udenafil, unlike the other PDE5 inhibitors, may also have a second mechanism of action. In animal models it has been found to increase ICP whilst reducing levels of the pro-contractile mediator endothelin 1, acting as an inhibitor of nNOS (asymmetric dimethylarginine (ADMA)).37
All of the PDE5 inhibitors have the same mechanism of action. However, they differ in their efficacy for the inhibition of the enzyme, in their selectivity for PDE5 over other isoenzymes such as PDE6 and in their pharmacological properties.
Potency and selectivity
The potency of the PDE5 inhibitors can be measured in vitro by assessing the IC50 value (concentration at which the enzyme activity is 50% inhibited). Using these values, vardenafil exhibits a PDE5 inhibitory potential approximately five times higher than that of sildenafil38 (Table 2). PDE6 plays an important role in the conversion of light impulses into nerve impulses in the retina. For PDE6, sildenafil and vardenafil show a lower selectivity than tadalafil. With respect to PDE11, tadalafil shows only five times greater selectivity than does PDE5. PDE11 has been detected in a variety of human tissues, e.g. in the heart, pituitary gland, brain and testes. The physiological significance of PDE11 and the possible consequences of its inhibition have not yet been fully established.
Table 2.
Pharmacological properties of three PDE5 inhibitors: Sildenafil, vardenafil, tadalafil and avanafil are shown as “time to onset” and “duration of action” obtained from clinical studies.
| Sildenafil | Vardenafil | Tadalafil | Avanafil | |
|---|---|---|---|---|
| Time to onset | 30–60 min | 25–40 min | 45 min | 15–30 min |
| Duration of action | 4–8 hours | Up to 6 hours | 24–36 hours | 4–6 hours |
| IC50 for PDE5 (nM) | 3.5–3.7 | 0.1–0.7 | 0.9–1.8 | N/A |
| aPDE1 | 80 | 500 | >4450 | |
| aPDE2 | >8570 | 44,290 | >14,800 | |
| aPDE3 | 4630 | >7140 | >14,800 | |
| aPDE4 | 2190 | 43,570 | >14,800 | |
| aPDE5 | 1 | 1 | 1 | |
| aPDE6 | 10 | 16 | 190 | |
| aPDE7 | 6100 | >214,000 | >14,800 | |
| aPDE8 | 8500 | >214,000 | >14,800 | |
| aPDE9 | 750 | 4150 | >14,800 | |
| aPDE10 | 2800 | 21,200 | >14,800 | |
| aPDE11 | 780 | 1160 | 5 |
PDE5: phosphodiesterase type 5; IC50: concentration at which the enzyme activity is 50% inhibited. IC50 values are from in vitro enzyme studies.
Denotes the ratio of IC50 for that PDE enzyme over IC50 for PDE5.39
Pharmacokinetics
All four drugs are rapidly absorbed from the gastrointestinal tract, with peak plasma levels being attained within one hour in the case of sildenafil and vardenafil and after two hours in the case of tadalafil.39 For avanafil, it has been reported that peak levels are achieved within 33 minutes.40 Food intake causes no delay or reduction in tadalafil absorption, whereas it is known to reduce and delay absorption of sildenafil. The mean half-lives (t 1/2) of sildenafil and vardenafil are three to four hours; for avanafil it is five to 10 hours whereas that of tadalafil is approximately 18 hours.39 The elimination of sildenafil, vardenafil and tadalafil takes place predominantly via cytochrome enzyme P450 (CYP3A4) in the liver.39
Clinical efficacy
Results from clinical trials suggest that all three currently available PDE5 inhibitors are effective in a wide range of patient groups.41–43 Treatment with vardenafil at a dose of 20 mg produced an improvement in the ability to achieve an erection in 80% of ED patients. In a comparable study of sildenafil (100 mg dose), 84% of ED patients were successfully treated. Treatment with tadalafil 20 mg produced an improvement in the ability to achieve an erection in 81% of ED patients. Comparative studies between vardenafil and sildenafil44 and more recently tadalafil and sildenafil45 suggest that patients do not have a significant preference among the PDE5 inhibitors.
Adverse effects
The most common side effects seen with sildenafil include headache, flushing, dyspepsia and rhinitis.46 The adverse effects with tadalafil and vardenafil are similar to sildenafil; however, tadalafil is associated with a higher incidence of back pain (4%–9%) and myalgia (1%–7%).
Difficult treatment groups
Diabetes mellitus
The sildenafil Diabetes Study Group reported that 56% of men with ED and diabetes who received sildenafil (25–100 mg) for 12 weeks reported improved erections (Global Assessment Questionnaire (GAQ)). With placebo only 10% reported better erections.47 In a more recent double-blind, placebo-controlled, flexible-dose study patients were randomised to receive sildenafil or placebo for 12 weeks. The erectile function domain of the International Index of Erectile Function (IIEF) showed only a six-point increase in the mean score over placebo (Figure 5). However, men with mild/moderate ED achieved a higher overall score compared with men with severe ED.48
Figure 5.
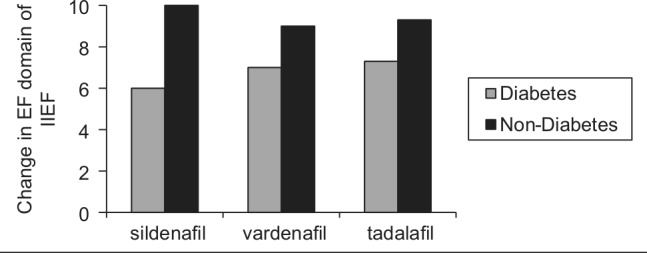
The change in erectile function (EF) domain of the IIEF in non-diabetic patients compared with diabetic patients after treatment with sildenafil 100 mg,48,51 vardenafil 20 mg43,50 and tadalafil 20 mg.52,53
IIEF: International Index of Erectile Function.
In a multicentre, double-blind, placebo-controlled, fixed-dose trial, patients with diabetes and ED were randomized to take vardenafil or placebo as needed for 12 weeks. With respect to the erectile function domain, the dose-dependent final scores for the 10- and 20-mg dose were 17.1 and 19.0 compared with 12.6 for placebo.49 Similar results have been reported in a prospective, randomised study in PDE5 inhibitor-naive patients with type 1 diabetes. Vardenafil treatment significantly improved the erectile function domain score of the IIEF (from 13 to 20)50 (Figure 5).
For tadalafil, at doses of 10 mg or 20 mg, the erectile function domain score was improved by 6.4 and 7.3, respectively, regardless of baseline HbA1c levels51 (Figure 5).
For the newly launched PDE5 inhibitor avanafil, the results of the REVIVE ED study were recently presented. These results suggest that erections sufficient for penetration (SEP2) were found in 63% of patients versus 42% with placebo, and successful intercourse was possible (SEP3) in 40% (versus 20% for placebo). Moreover, it was reported that over 70% of participants were able to achieve an erection within 15 minutes.
Testosterone replacement therapy
Androgens are known to be involved in both the central and peripheral pathways associated with penile erection. Testosterone is required for NOS expression in the corpus cavernosum and also for the maintenance of the neural pathway. Although controversial, testosterone supplementation is an option in patients with erectile dysfunction who are non-responders to PDE5 inhibitors and have a low serum testosterone level.54,55 Studies have suggested that up to 60% of non-responders may be converted to responders following combination treatment.56
Alternative oral treatment options
Apomorphine
The PVN in the hypothalamus is involved in initiating the erectile response. Apomorphine hydrochloride is a dopaminergic receptor agonist (D1 and D2 receptors) that has been developed as a sublingual agent to activate oxytocinergic neurons in the PVN. The median onset to action is 19 minutes and the half-life is one hour. An open-label, randomised, flexible-dose comparison of apomorphine and sildenafil demonstrated the superior efficacy of sildenafil to apomorphine (75% versus 35%, respectively). The side effect profile – nausea (7%), dizziness (6.5%) and yawning (8.1%) – combined with a high non-responder rate has limited the therapeutic acceptance of this drug and has led to its withdrawal from the market.
α-adrenoceptor antagonists
The aim of these agents is to reduce the corpus cavernosum smooth muscle tone by inhibiting the innervation of the sympathetic nervous system. These agents are not routinely used in clinical practice as we now know that the NO-cGMP signalling system is the predominant pathway in achieving smooth muscle relaxation.
Yohimbine is an orally administered indolalquinolonic alkaloid agent with both peripheral α2 adrenergic receptor blockade and central noradrenergic agonist activity. Phentolamine is a non-selective α-adrenoceptor antagonist, but has not gained acceptance – partly because of the associated systemic effects. The selective α1 antagonists doxazosin and terazosin are routinely utilised for patients with bladder outflow obstruction and can improve erectile function in patients with very mild symptoms.
Intracavernosal and intraurethral prostaglandins
The synthetic PGE1 analogue alprostadil can be administered as a second-line therapy in patients failing oral pharmacotherapies or who have a specific contraindication to treatment with oral agents. PGE1 increases the intracellular concentrations of the second messenger cAMP, resulting in corpus cavernosum smooth muscle relaxation. Currently, two methods of administration are available: direct intracavernosal injection (80% response rate) or intraurethral application of a small pellet (MUSE® dose 250–1000 μg, 65% response rate). This second-line treatment is useful in patients with long-standing diabetes or ED secondary to pelvic surgery, who have a higher incidence of ED refractory to oral pharmacotherapies.
A recent study suggests that a poor response to intracavernosal alprostadil is associated with small vessel disease and a higher risk of cardiovascular events.57
Mechanical interventions
Vacuum devices
These devices are useful in patents with psychogenic or organic ED and can be used alone or in combination with other therapies. An external cylinder is utilised to create a negative pressure and penile tumescence is maintained by means of a constriction ring at the base of the penis. The reported patient satisfaction rate is 50%–70%.58
Penile prosthesis surgery
The insertion of a penile prosthesis is suitable for patients with severe organic ED. Two main subtypes of prosthesis are available: malleable (or semi-rigid) and inflatable. The malleable devices have the advantages of decreased mechanical breakdown, easier placement and lower cost. Inflatable devices are available as two- (Ambicor, American Medical Systems) or three-piece devices (AMS 700CX or Coloplast Titan, Titan Zero Degree). The two- and three-piece devices have a pump placed in the scrotum that controls the inflation and deflation of the device and therefore requires an element of patient dexterity.
Complications include infection rates of up to 2%–3% and re-operation rates for mechanical failure of 15% by 10 years. The overall satisfaction rates have been reported as over 90% from the patients and partners, respectively.59 Using a minimal handling approach and antibiotic-coated implants, infection rates have fallen to less than 1%60 (Figure 6).
Figure 6.
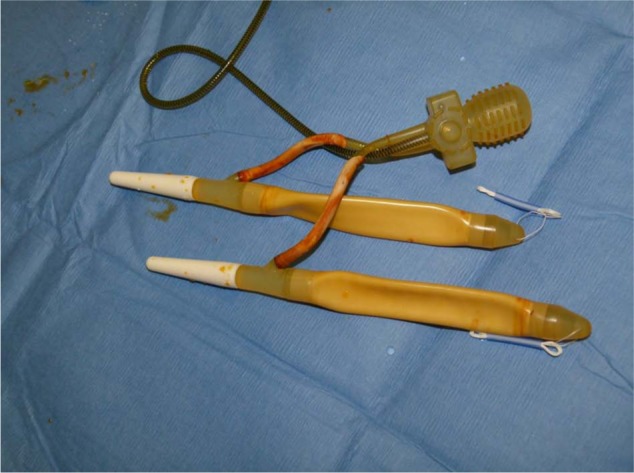
The AMS 700CX three-piece inflatable implant.
The implant is made up of two cylinders that are placed inside the corpus cavernosum, a momentary squeeze pump which is placed in the scrotum and a reservoir which is located in the retropubic space.
Future therapeutic options
Rho-kinase inhibitors
Rho-A is a small monomeric G protein that activates rho-kinase and is involved in the sensitisation of the smooth muscle contractile elements to Ca2+. Therefore, smooth muscle relaxation can be modulated without a change in the intracellular Ca2+ levels. Rho-kinase inhibitors provide an alternative pathway to produce smooth muscle relaxation, and in vitro studies have shown that specific inhibitors of rho-kinase such as Y-27632 can cause a concentration-dependent relaxation of the corpus cavernosum.61
Direct sGC activators
Patients with significant endothelial dysfunction or nitrergic nerve impairment are unable to produce adequate endogenous NO. Therefore, direct NO-independent activation of sGC provides a novel approach. The benzylindazole derivative YC-1 has been investigated as a potential sGC activator, but has been found to have non-specific phosphodiesterase inhibitory activity – although alternative compounds based on this prototype have been developed and are under investigation.62
NO-releasing PDE5 inhibitors
Sildenafil nitrate is an NO-releasing derivative of sildenafil citrate that can release NO spontaneously and can also inhibit PDE5. This compound is more potent than sildenafil citrate and can release NO in the absence of endogenous NO. Further research is required before any clinical application is proposed.63
Further strategies for the treatment of ED
ED is invariably the result of a number of pathophysiological events which result in a reduction in the bioavailability of NO. We believe that future strategies for the treatment of ED should be aimed at correcting or treating the underlying mechanisms involved in the pathogenesis of ED as well as finding more specific and effective sGC activators and NO-releasing compounds.
Novel research areas include gene therapy with neurotrophic factors, eNOS, nNOS and superoxide dismutase. Through the use of an appropriate vector, diabetic animals have already been successfully transfected with these agents. Direct injections into the cavernous sheath of diabetic rats with neurotrophin-3 (NT3) using the herpes simplex virus as the vector have been performed. Subsequent immunoreactive strains have demonstrated a significant increase in nNOS neurons in the major pelvic ganglia. Moreover, this was associated with significant increases in the ICP following cavernous nerve stimulation.64
Moreover, diabetic rats injected with adenoviruses containing eNOS into the corpus cavernosum have produced significant rises in ICP secondary to cavernous nerve stimulation. This was further associated with a rise in eNOS (measured by Western blot analysis) and an increase in NOS biosynthesis (measured by an increase in cavernous nitrate and nitrite formation).65
Further studies have examined the intracavernous injection of adenoviruses containing superoxide dismutase into diabetic rats. The results indicate a decrease in superoxide anion levels, an increase in NO bioavailability and an increase in cGMP levels.28
More recently, the effects of gene transfer on erectile function and sexual behaviour have evaluated in male cynomolgus monkeys with ED and an ageing rat model. The animals were injected intracavernously with a smooth-muscle-specific gene transfer vector (pSMAA-hSlo) encoding the pore-forming subunit of the human large-conductance, calcium-sensitive potassium channels (Maxi-K). The results have a shown a significant improvement in erectile function and sexual behaviour66 or increased ICP responses to cavernous nerve stimulation67 after the intracorporeal gene transfer. These results support the concept that intracorporeal Maxi-K-channel gene transfer may be a novel way of improving erectile function.
Conclusion
Despite major advances in the understanding of the physiology of penile erection and the pathophysiology of ED, together with an increase in the available pharmacotherapies, ED remains a significant global male health problem. This condition has an impact on the patients’ and partners’ quality of life and self-esteem.
Oral tablets, in particular PDE5 inhibitors, have revolutionised the treatment of ED by decreasing reliance on more invasive options. Three potent selective PDE5 inhibitors, sildenafil (Viagra; Pfizer), tadalafil (Cialis; Lilly) and vardenafil (Levitra; Bayer) are currently available in the United Kingdom. Although large multicentre clinical trials have shown the efficacy and tolerability of these drugs in ED with various aetiologies and a broad range of severity, 30%–35% of patients fail to respond to oral pharmacotherapies, especially in difficult treatment groups such as diabetic patients. The possible reasons for failure include severe ED at presentation, worsening of endothelial dysfunction, ED after radical prostatectomy or diabetes, unrecognised or untreated hypogonadism, inadequate patient education or incorrect drug usage or the development of drug tolerance. However, end-stage surgical treatment using penile prosthesis surgery is still associated with a high patient and partner satisfaction rate of over 80%. The continued refinement of penile prostheses has resulted in reduced infection and mechanical failure rates.
Footnotes
Conflict of interest: None declared.
Funding: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
- 1. Consensus development conference statement. National Institutes of Health. Impotence. December 7–9, 1992. Int J Impot Res 1993; 5: 181–284. [PubMed] [Google Scholar]
- 2. Ayta IA, McKinlay JB, Krane RJ. The likely worldwide increase in erectile dysfunction between 1995 and 2025 and some possible policy consequences. BJU Int 1999; 84: 50–56. [DOI] [PubMed] [Google Scholar]
- 3. Ventegodt S. Sex and the quality of life in Denmark. Arch Sex Behav 1998; 27: 295–307. [DOI] [PubMed] [Google Scholar]
- 4. Feldman HA, Goldstein I, Hatzichristou DG, et al. Impotence and its medical and psychosocial correlates: Results of the Massachusetts Male Aging Study. J Urol 1994; 151: 54–61. [DOI] [PubMed] [Google Scholar]
- 5. Andersson KE, Wagner G. Physiology of penile erection. Physiol Rev 1995; 75: 191–236. [DOI] [PubMed] [Google Scholar]
- 6. Andersson KE. Pharmacology of penile erection. Pharmacol Rev 2001; 53: 417–450. [PubMed] [Google Scholar]
- 7. Giuliano F, Rampin O. Central neural regulation of penile erection. Neurosci Biobehav Rev 2000; 24: 517–533. [DOI] [PubMed] [Google Scholar]
- 8. Erectile Dysfunction: Current Investigation and Management. 2nd Ed, Eds: Eardley I, Sethia K. 2003, Mosby, UK. [Google Scholar]
- 9. Lundberg JM. Pharmacology of cotransmission in the autonomic nervous system: Integrative aspects on amines, neuropeptides, adenosine triphosphate, amino acids and nitric oxide. Pharmacol Rev 1996; 48: 113–178. [PubMed] [Google Scholar]
- 10. Hedlund P, Ny L, Alm P, et al. Cholinergic nerves in human corpus cavernosum and spongiosum contain nitric oxide synthase and heme oxygenase. J Urol 2000; 164 (3 Pt 1): 868–875. [DOI] [PubMed] [Google Scholar]
- 11. Moncada S, Higgs A, Furchgott R. International Union of Pharmacology Nomenclature in Nitric Oxide Research. Pharmacol Rev 1997; 49: 137–142. [PubMed] [Google Scholar]
- 12. Cellek S, Moncada S. Modulation of noradrenergic responses by nitric oxide from inducible nitric oxide synthase. Nitric Oxide 1997; 1: 204–210. [DOI] [PubMed] [Google Scholar]
- 13. Ignarro LJ, Bush PA, Buga GM, et al. Nitric oxide and cyclic GMP formation upon electrical field stimulation cause relaxation of corpus cavernosum smooth muscle. Biochem Biophys Res Commun 1990; 170: 843–850. [DOI] [PubMed] [Google Scholar]
- 14. Alm P, Larsson B, Ekblad E, et al. Immunohistochemical localization of peripheral nitric oxide synthase-containing nerves using antibodies raised against synthesized C- and N-terminal fragments of a cloned enzyme from rat brain. Acta Physiol Scand 1993; 148: 421–429. [DOI] [PubMed] [Google Scholar]
- 15. Kim N, Azadzoi KM, Goldstein I, et al. A nitric oxide-like factor mediates nonadrenergic-noncholinergic neurogenic relaxation of penile corpus cavernosum smooth muscle. J Clin Invest 1991; 88: 112–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Okamura T, Ayajiki K, Fujioka H, et al. Effects of endothelial impairment by saponin on the responses to vasodilators and nitrergic nerve stimulation in isolated canine corpus cavernosum. Br J Pharmacol 1999; 127: 802–808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Schmidt HH, Lohmann SM, Walter U. The nitric oxide and cGMP signal transduction system: Regulation and mechanism of action. Biochim Biophys Acta 1993; 1178: 153–175. [DOI] [PubMed] [Google Scholar]
- 18. Melman A, Gingell JC. The epidemiology and pathophysiology of erectile dysfunction. J Urol 1999; 161: 5–11. [PubMed] [Google Scholar]
- 19. Billups KL, Bank AJ, Padma-Nathan H, et al. Erectile dysfunction as a harbinger for increased cardiometabolic risk. Int J Impot Res 2008; 20: 236–242. [DOI] [PubMed] [Google Scholar]
- 20. Creager MA, Luscher TF, Cosentino F, et al. Diabetes and vascular disease: Pathophysiology, clinical consequences, and medical therapy: Part I. Circulation 2003; 108: 1527–1532. [DOI] [PubMed] [Google Scholar]
- 21. La Vignera S, Calogero AE, Cannizzaro MA, et al. Tadalafil and modifications in peak systolic velocity (Doppler spectrum dynamic analysis) in the cavernosal arteries of patients with type 2 diabetes after continuous tadalafil treatment [in English and Italian]. Minerva Endocrinol 2006; 31: 251–261. [PubMed] [Google Scholar]
- 22. Esposito K, Giugliano D. Lifestyle for erectile dysfunction: A good choice. Arch Intern Med 2012; 172: 295–296. [DOI] [PubMed] [Google Scholar]
- 23. Aytac IA, Araujo AB, Johannes CB, et al. Socioeconomic factors and incidence of erectile dysfunction: Findings of the longitudinal Massachusetts Male Aging Study. Soc Sci Med 2000; 51: 771–778. [DOI] [PubMed] [Google Scholar]
- 24. Akkus E, Kadioglu A, Esen A, et al. Prevalence and correlates of erectile dysfunction in Turkey: A population-based study. Eur Urol 2002; 41: 298–304. [DOI] [PubMed] [Google Scholar]
- 25. Fedele D, Coscelli C, Cucinotta D, et al. Incidence of erectile dysfunction in Italian men with diabetes. J Urol 2001; 166: 1368–1371. [PubMed] [Google Scholar]
- 26. Metro MJ, Broderick GA. Diabetes and vascular impotence: Does insulin dependence increase the relative severity? Int J Impot Res 1999; 11: 87–89. [DOI] [PubMed] [Google Scholar]
- 27. McCulloch DK, Campbell IW, Wu FC, et al. The prevalence of diabetic impotence. Diabetologia 1980; 18: 279–283. [DOI] [PubMed] [Google Scholar]
- 28. Bivalacqua TJ, Usta MF, Kendirci M, et al. Superoxide anion production in the rat penis impairs erectile function in diabetes: Influence of in vivo extracellular superoxide dismutase gene therapy. J Sex Med 2005; 2: 187–197. [DOI] [PubMed] [Google Scholar]
- 29. Chang S, Hypolite JA, Velez M, et al. Downregulation of cGMP-dependent protein kinase-1 activity in the corpus cavernosum smooth muscle of diabetic rabbits. Am J Physiol Regul Integr Comp Physiol 2004; 287: R950–R960. [DOI] [PubMed] [Google Scholar]
- 30. Khan MA, Thompson CS, Jeremy JY, et al. The effect of superoxide dismutase on nitric oxide-mediated and electrical field-stimulated diabetic rabbit cavernosal smooth muscle relaxation. BJU Int 2001; 87: 98–103. [DOI] [PubMed] [Google Scholar]
- 31. Chang S, Hypolite JA, Changolkar A, et al. Increased contractility of diabetic rabbit corpora smooth muscle in response to endothelin is mediated via Rho-kinase beta. Int J Impot Res 2003; 15: 53–62. [DOI] [PubMed] [Google Scholar]
- 32. Billups KL. Erectile dysfunction as an early sign of cardiovascular disease. Int J Impot Res 2005; 17 (Suppl 1): S19–S24. [DOI] [PubMed] [Google Scholar]
- 33. Cellek S, Rodrigo J, Lobos E, et al. Selective nitrergic neurodegeneration in diabetes mellitus – a nitric oxide-dependent phenomenon. Br J Pharmacol 1999; 128: 1804–1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Francis SH, Turko IV, Corbin JD. Cyclic nucleotide phosphodiesterases: Relating structure and function. Prog Nucleic Acid Res Mol Biol 2001; 65: 1–52. [DOI] [PubMed] [Google Scholar]
- 35. Sanford M. Vardenafil orodispersible tablet. Drugs 2012; 72: 87–98. [DOI] [PubMed] [Google Scholar]
- 36. Egerdie RB, Auerbach S, Roehrborn CG, et al. Tadalafil 2.5 or 5 mg administered once daily for 12 weeks in men with both erectile dysfunction and signs and symptoms of benign prostatic hyperplasia: Results of a randomized, placebo-controlled, double-blind study. J Sex Med 2012; 9: 271–281. [DOI] [PubMed] [Google Scholar]
- 37. Kang KK, Yu JY, Yoo M, et al. The effect of DA-8159, a novel PDE5 inhibitor, on erectile function in the rat model of hypercholesterolemic erectile dysfunction. Int J Impot Res 2005; 17: 409–416. [DOI] [PubMed] [Google Scholar]
- 38. Kim NN, Huang YH, Goldstein I, et al. Inhibition of cyclic GMP hydrolysis in human corpus cavernosum smooth muscle cells by vardenafil, a novel, selective phosphodiesterase type 5 inhibitor. Life Sci 2001; 69: 2249–2256. [DOI] [PubMed] [Google Scholar]
- 39. Corbin JD, Francis SH. Pharmacology of phosphodiesterase-5 inhibitors. Int J Clin Pract 2002; 56: 453–459. [PubMed] [Google Scholar]
- 40. Jung J, Choi S, Cho SH, et al. Tolerability and pharmacokinetics of avanafil, a phosphodiesterase type 5 inhibitor: A single- and multiple-dose, double-blind, randomized, placebo-controlled, dose-escalation study in healthy Korean male volunteers. Clin Ther 2010; 32: 1178–1187. [DOI] [PubMed] [Google Scholar]
- 41. Brock GB, McMahon CG, Chen KK, et al. Efficacy and safety of tadalafil for the treatment of erectile dysfunction: Results of integrated analyses. J Urol 2002; 168 (4 Pt 1): 1332–1336. [DOI] [PubMed] [Google Scholar]
- 42. Eardley I, Morgan R, Dinsmore W, et al. Efficacy and safety of sildenafil citrate in the treatment of men with mild to moderate erectile dysfunction. Br J Psychiatry 2001; 178:325–330. [DOI] [PubMed] [Google Scholar]
- 43. Hellstrom WJ, Gittelman M, Karlin G, et al. Vardenafil for treatment of men with erectile dysfunction: Efficacy and safety in a randomized, double-blind, placebo-controlled trial. J Androl 2002; 23: 763–771. [PubMed] [Google Scholar]
- 44. Rubio-Aurioles E, Porst H, Eardley I, et al. Comparing vardenafil and sildenafil in the treatment of men with erectile dysfunction and risk factors for cardiovascular disease: A randomized, double-blind, pooled crossover study. J Sex Med 2006; 3: 1037–1049. [DOI] [PubMed] [Google Scholar]
- 45. Ahn TY, Lee SW, Kim SW, et al. Treatment preferences in men with erectile dysfunction: An open label study in Korean men switching from sildenafil citrate to tadalafil. Asian J Androl 2007; 9: 760–770. [DOI] [PubMed] [Google Scholar]
- 46. Fagelman E, Fagelman A, Shabsigh R. Efficacy, safety, and use of sildenafil in urologic practice. Urology 2001; 57: 1141–1144. [DOI] [PubMed] [Google Scholar]
- 47. Rendell MS, Rajfer J, Wicker PA, et al. Sildenafil for treatment of erectile dysfunction in men with diabetes: A randomized controlled trial. Sildenafil Diabetes Study Group. JAMA 1999; 281: 421–426. [DOI] [PubMed] [Google Scholar]
- 48. Stuckey BG, Jadzinsky MN, Murphy LJ, et al. Sildenafil citrate for treatment of erectile dysfunction in men with type 1 diabetes: Results of a randomized controlled trial. Diabetes Care 2003; 26: 279–284. [DOI] [PubMed] [Google Scholar]
- 49. Goldstein I, Young JM, Fischer J, et al. Vardenafil, a new phosphodiesterase type 5 inhibitor, in the treatment of erectile dysfunction in men with diabetes: A multicenter double-blind placebo-controlled fixed-dose study. Diabetes Care 2003; 26: 777–783. [DOI] [PubMed] [Google Scholar]
- 50. Ziegler D, Merfort F, van Ahlen H, et al. Efficacy and safety of flexible-dose vardenafil in men with type 1 diabetes and erectile dysfunction. J Sex Med 2006; 3: 883–891. [DOI] [PubMed] [Google Scholar]
- 51. Carson CC, Burnett AL, Levine LA, et al. The efficacy of sildenafil citrate (Viagra) in clinical populations: An update. Urology 2002; 60 (2 Suppl 2): 12–27. [DOI] [PubMed] [Google Scholar]
- 52. Sáenz de Tejada I, Anglin G, Knight JR, et al. Effects of tadalafil on erectile dysfunction in men with diabetes. Diabetes Care 2002; 25: 2159–2164. [DOI] [PubMed] [Google Scholar]
- 53. Seftel AD, Wilson SK, Knapp PM, et al. The efficacy and safety of tadalafil in United States and Puerto Rican men with erectile dysfunction. J Urol 2004; 172: 652–657. [DOI] [PubMed] [Google Scholar]
- 54. Rosenthal BD, May NR, Metro MJ, et al. Adjunctive use of AndroGel (testosterone gel) with sildenafil to treat erectile dysfunction in men with acquired androgen deficiency syndrome after failure using sildenafil alone. Urology 2006; 67: 571–574. [DOI] [PubMed] [Google Scholar]
- 55. Traish AM, Goldstein I, Kim NN. Testosterone and erectile function: From basic research to a new clinical paradigm for managing men with androgen insufficiency and erectile dysfunction. Eur Urol 2007; 52: 54–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Garcia JA, Sanchez PE, Fraile C, et al. Testosterone undecanoate improves erectile dysfunction in hypogonadal men with the metabolic syndrome refractory to treatment with phosphodiesterase type 5 inhibitors alone. Andrologia 2011; 43: 293–296. [DOI] [PubMed] [Google Scholar]
- 57. Rastrelli G, Corona G, Monami M, et al. Poor response to alprostadil ICI test is associated with arteriogenic erectile dysfunction and higher risk of major adverse cardiovascular events. J Sex Med 2011; 8: 3433–3445. [DOI] [PubMed] [Google Scholar]
- 58. Chen J, Mabjeesh NJ, Greenstein A. Sildenafil versus the vacuum erection device: Patient preference. J Urol 2001; 166: 1779–1781. [PubMed] [Google Scholar]
- 59. Chung E, Van CT, Wilson I, et al. Penile prosthesis implantation for the treatment for male erectile dysfunction: Clinical outcomes and lessons learnt after 955 procedures. World J Urol. Epub ahead of print 29 March 2012. [DOI] [PubMed] [Google Scholar]
- 60. Eid JF, Wilson SK, Cleves M, et al. Coated implants and “no touch” surgical technique decreases risk of infection in inflatable penile prosthesis implantation to 0.46%. Urology 2012; 79: 1310–1315. [DOI] [PubMed] [Google Scholar]
- 61. Rees RW, Ralph DJ, Royle M, et al. Y-27632, an inhibitor of Rho-kinase, antagonizes noradrenergic contractions in the rabbit and human penile corpus cavernosum. Br J Pharmacol 2001; 133: 455–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Kalsi JS, Rees RW, Hobbs AJ, et al. BAY41-2272, a novel nitric oxide independent soluble guanylate cyclase activator, relaxes human and rabbit corpus cavernosum in vitro. J Urol 2003; 169: 761–766. [DOI] [PubMed] [Google Scholar]
- 63. Kalsi JS, Kell PD, Cellek S, et al. NCX-911, a novel nitric oxide-releasing PDE5 inhibitor relaxes rabbit corpus cavernosum in the absence of endogenous nitric oxide. Int J Impot Res 2004; 16: 195–200. [DOI] [PubMed] [Google Scholar]
- 64. Bennett NE, Kim JH, Wolfe DP, et al. Improvement in erectile dysfunction after neurotrophic factor gene therapy in diabetic rats. J Urol 2005; 173: 1820–1824. [DOI] [PubMed] [Google Scholar]
- 65. Bivalacqua TJ, Usta MF, Champion HC, et al. Gene transfer of endothelial nitric oxide synthase partially restores nitric oxide synthesis and erectile function in streptozotocin diabetic rats. J Urol 2003; 169: 1911–1917. [DOI] [PubMed] [Google Scholar]
- 66. Christ GJ, Andersson KE, Williams K, et al. Smooth-muscle-specific gene transfer with the human maxi-k channel improves erectile function and enhances sexual behavior in atherosclerotic cynomolgus monkeys. Eur Urol 2009; 56: 1055–1066. [DOI] [PubMed] [Google Scholar]
- 67. Melman A, Biggs G, Davies K, et al. Gene transfer with a vector expressing maxi-K from a smooth muscle-specific promoter restores erectile function in the aging rat. Gene Ther 2008; 15: 364–370. [DOI] [PubMed] [Google Scholar]


