Abstract
Background
Several risk factors for pacemaker (PM) related complications have been reported. However, no study has investigated the impact of lead characteristics on pacemaker-related complications.
Methods and Results
Patients who received a new pacemaker implant from January 1997 to December 2011 were selected from the Taiwan National Health Insurance Database. This population was grouped according to the pacemaker lead characteristics in terms of fixation and insulation. The impact of the characteristics of leads on early heart perforation was analyzed by multivariable logistic regression analysis, while the impact of the lead characteristics on early and late infection and late heart perforation over a three-year period were analyzed using Cox regression. This study included 36,104 patients with a mean age of 73.4±12.5 years. In terms of both early and late heart perforations, there were no significant differences between groups across the different types of fixation and insulations. In the multivariable Cox regression analysis, the pacemaker-related infection rate was significantly lower in the active fixation only group compared to either the both fixation (OR, 0.23; 95% CI, 0.07–0.80; P = 0.020) or the passive fixation group (OR, 0.26; 95% CI, 0.08–0.83; P = 0.023).
Conclusions
There was no difference in heart perforation between active and passive fixation leads. Active fixation leads were associated with reduced risk of pacemaker-related infection.
Introduction
The pacemaker (PM) is standard therapy for bradyarrhythmias [1,2] and the population with implanted devices continues to grow [3], PM-related complications such as infection, and cardiac perforation will increase as well [4,5]. These complications not only result in prolonged hospitalization and increased costs, but also accrue worse outcomes and mortality [6]. For these reasons, many studies have attempted to investigate factors causing PM-related complications by evaluating baseline characteristics, implant procedures, and medications. Among the evaluated risks of the PM-related infection, several risk factors have been reported, such as diabetes mellitus, end-stage renal disease, corticosteroid use, and so on [7–11]. Although heart perforation is a relatively rare PM-related complication with a reported incidence ranging from 0.09% to 1.2% in the literature [12], some studies have pointed out certain risk factors for heart perforation such as temporary pacemakers, steroid use within 7 days prior to implantation, and helical screws [13].
Although a number of studies have reported many risk factors contributing to PM-related complications [13,14], most of which being mechanical complications, the relationship between the characteristics of pacemaker leads (in terms of fixation types or insulation materials) and PM-related complications remains unclear. Only a handful of case report studies [15] have mentioned this relationship. Accordingly, this study investigated the relationship between the characteristics of PM leads, fixation type and insulation, and PM-related complications in a large number of patients from a nationwide database
Materials and Methods
Data Source
This retrospective national population-based cohort study was retrieved from the National Health Insurance Research Database (NHIRD) released by the Taiwan National Health Research Institute (NHRI) ( http://nhird.nhri.org.tw/en/index.htm ). The data of NHIRD contain registration files and original claim data for reimbursement, which were derived from the National Health Insurance Administration, Ministry of Health and Welfare and maintained by the NHRI. The NHIRD contains health care information of the 99.9% of the Taiwanese population (about 23.20 million in 2012) enrolled in the NHI program [16]. Previous studies have described the NHIRD in detail and validated the accuracy of its diagnostic data [17]. The insurance has since 1997 reimbursed all the new implantation, replacement, revision and removal expenses of cardiac implantable electronic devices (CIEDs), with the appropriate indications according to the clinical practice guidelines of the CIED. In addition, the expenses of the insurance include all the generators, CIED leads and physician fees. The original data of the NHIRD are unstructured data, which all the contents are string variables and are not able to be analyzed directly. According to the coded book provided by the NHRI (http://nhird.nhri.org.tw/date_02.htm), the original unstructured data were transformed into structured data, which were composed of numeric variables by using SAS Version 9.3 (SAS Institute, Cary, NC). Analyzing statistical analysis becomes feasible when the data are numeric rather than string type.
Study design
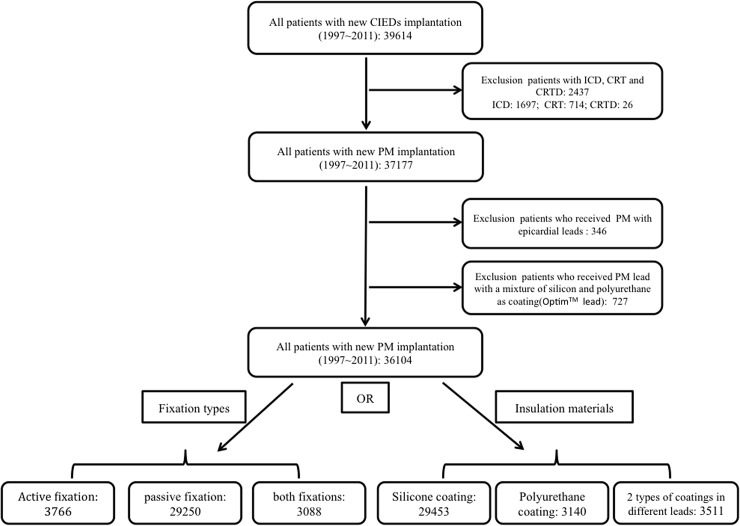
In the initial selection of the population set, the patients who received CIED implantation between January 1, 1997 and December 31, 2011 were included (Fig 1). We excluded patients who received implantable cardiac defibrillators, cardiac resynchronization therapy and cardiac resynchronization therapy defibrillator implantation. Furthermore, patients who received PM with epicardial leads were also excluded in order to reduce the procedure related bias. Moreover, we excluded patients who received leads with a mixture of silicone and polyurethane as insulation (Optim). Finally, we analyzed the impact of the fixation types or insulation materials of the leads on PM-related complications (Fig 1).
Fig 1. Study design and flow chart of patient selection.
CIED = cardiac implantable electronic devices; ICD = implantable cardiac defibrillator; CRT = cardiac resynchronization therapy; CRTD = cardiac resynchronization therapy defibrillator; PM = pacemaker.
There were two outcomes specifically evaluated in this study. One was heart perforation that included early and late heart perforation; the other was PM-related infection, which included early and late infection and infection that either needed lead extraction or without lead extraction. This cohort study was followed up for three years after the index date. In the cohort dataset from NHIRD, the patients' original identification numbers have been encrypted to protect their privacy and the encrypted data were for research purposes only. We used NHIRD data set for this study and obtained ethical approval from the Institutional Review Board of Chang Gung Memorial Hospital (101-2055B).
Definitions
PM-related heart perforation was defined as a heart perforation treated with heart repair during admission for any implantation procedure, replacement, revision, or removal. Early heart perforation was defined as the events occurring within one month [12] after implantation, while late heart perforation was defined as events developing more than one month after implantation. Pacemaker infection was defined as an infection (S1 Table) that occurred during admission for implantation, replacement, revision, or removal. Early infection was defined as the PM-related infection developing within one year after the new implantation, while late infection was defined as PM-related infection developing more than one year after the new implantation [9].
The analyzed characteristics of leads were fixation types and insulation materials. In the analysis of fixation type, the population was divided into the three groups of active fixation (screw lead), passive (tined lead) and both-types, which meant that one lead was active fixation and the other was passive fixation in the dual chamber pacemaker. In the analysis of insulation, the population was divided into the three groups of silicone coating, polyurethane coating and both-coating which meant that one lead had a silicone coating and the other a polyurethane coating in the dual chamber pacemaker.
Statistical Analysis
The clinical characteristics of the study participants are presented as number and percentage for categorical variables or as mean and standard deviation for continuous variables. The association between study groups (different fixation types and insulation types) and early heart perforation was examined using multivariable logistic regression analysis. The three-year event-free survival rates of late heart perforation and PM-related infection among the study groups were compared using a multivariable Cox proportional hazard analysis. The results are presented as an adjusted odds ratio (OR) for logistic regression or adjusted hazard ratio (HR) for Cox regression with its corresponding 95% confidence intervals (CI). To rule out the confounding effects, both logistic and Cox regression analyses were performed with adjustment of patient’s characteristics, such as gender, age group, hospital level, device type, diabetes mellitus, liver cirrhosis, obstructive lung disease, chronic kidney disease, heart failure, hypertension, coronary artery disease, malignant neoplasm, antibiotics use, steroid use, and combination of warfarin and antiplatelet use. Data analyses were conducted using SPSS software version 15.0 (SPSS Inc., Chicago, IL, USA).
Results
Patient Characteristics
There were 36,104 PM patients with the mean age of 73.4 years (SD = 12.5 years) (range: 8 years to 105 years) were enrolled in this study and the majority of the population were older than 70 years. When the population was divided into three groups according to the fixation types, pacemakers with only active fixation leads were implanted in 3,766 patients, with only passive fixation leads in 29,250 patients, and with both fixation leads in 3,088 patients (Table 1). When the population was divided into three groups by different insulation of leads, the pacemakers with silicone-coating leads were implanted in 29,453 patients, with polyurethane-coating leads in 3,140 patients, and with one silicone-coating and the other polyurethane-coating leads in 3,511 patients (Table 1). There were similar distributions in age-scale, gender, comorbidities in each group and nearly all patients were prescribed intravenous antibiotics during the procedures. Furthermore, in the passive fixation group, most of the patients received a single chamber PM, while in the active fixation group, most received a dual chamber PM.
Table 1. Patient Characteristics at Implant Stratified by Lead Fixation Method and Insulation (n = 36,104).
| Fixation method | Insulation | |||||||
|---|---|---|---|---|---|---|---|---|
| Variable | Active fixation | Passive fixation | Both fixation | P Value | Silicone | Polyurethane | Both insulation | P Value |
| Number of patient | 3,766 | 29,250 | 3,088 | — | 29,453 | 3,140 | 3,511 | — |
| Gender––no. (%) | <0.001 a b | 0.001 d f | ||||||
| Male | 2,058 (54.6) | 14,871 (50.8) | 1,518 (49.2) | 15,142 (51.4) | 1,502 (47.8) | 1,803 (51.4) | ||
| Female | 1,708 (45.4) | 14,379 (49.2) | 1,570 (50.8) | 14,311 (48.6) | 1,638 (52.2) | 1,708 (48.6) | ||
| Age––yr±SD | 74.6±11.8 | 73.1±12.7 | 74.7±10.5 | <0.001 | 73.4±12.4 | 73.5±12.6 | 73.5±12.6 | 0.849 |
| Age group––no. (%) | <0.001 a b c | 0.003 e | ||||||
| < 20 yrs | 14 (0.4) | 252 (0.9) | 4 (0.1) | 229 (0.8) | 19 (0.6) | 22 (0.6) | ||
| 20~49 yrs | 130 (3.5) | 1,090 (3.7) | 77 (2.5) | 1,019 (3.5) | 130 (4.1) | 148 (4.2) | ||
| 50~59 yrs | 258 (6.9) | 2,013 (6.9) | 203 (6.6) | 2,005 (6.8) | 202 (6.4) | 267 (7.6) | ||
| 60~69 yrs | 597 (15.9) | 5,504 (18.8) | 533 (17.3) | 5,483 (18.6) | 553 (17.6) | 598 (17.0) | ||
| 70~79 yrs | 1,431 (38.0) | 11,676 (39.9) | 1,262 (40.9) | 11,769 (40.0) | 1,264 (40.3) | 1,336 (38.1) | ||
| >80 yrs | 1,336 (35.5) | 8,715 (29.8) | 1,009 (32.7) | 8,948 (30.4) | 972 (31.0) | 1,140 (32.5) | ||
| Device type––no. (%) | <0.001 a b c | <0.001 d e f | ||||||
| Single chamber PPM | 547 (14.5) | 21,567 (73.7) | 0 (0.0) | 19,566 (66.4) | 2,548 (81.1) | 0 (0.0) | ||
| Dual chamber PPM | 3,219 (85.5) | 7,683 (26.3) | 3,088 (100.0) | 9,887 (33.6) | 592 (18.9) | 3,511 (100.0) | ||
| Indication––no. (%) | ||||||||
| AV block | 1,462 (38.8) | 10,517 (36.0) | 986 (31.9) | <0.001 a b c | 10,405 (35.3) | 1,158 (36.9) | 1,402 (39.9) | <0.001 e f |
| Congenital AV block | 1 (0.0) | 60 (0.2) | 0 (0.0) | 0.002 a c | 57 (0.2) | 3 (0.1) | 1 (0.0) | 0.046 e |
| AF | 1,016 (27.0) | 5,842 (20.0) | 667 (21.6) | <0.001 a b c | 6,038 (20.5) | 610 (19.4) | 877 (25.0) | <0.001 e f |
| Sick sinus syndrome | 1,813 (48.1) | 14,742 (50.4) | 1,726 (55.9) | <0.001 a b c | 14,956 (50.8) | 1,620 (51.6) | 1,705 (48.6) | 0.024 e f |
| Comorbidity––no. (%) | ||||||||
| Diabetes | 1,235 (32.8) | 8,139 (27.8) | 947 (30.7) | <0.001 a c | 8,236 (28.0) | 970 (30.9) | 1,115 (31.8) | <0.001 d e |
| Liver cirrhosis | 103 (2.7) | 808 (2.8) | 89 (2.9) | 0.920 | 807 (2.7) | 100 (3.2) | 93 (2.6) | 0.317 |
| Obstructive lung disease | 401 (10.6) | 2,491 (8.5) | 318 (10.3) | <0.001 a c | 2,561 (8.7) | 309 (9.8) | 340 (9.7) | 0.022 d |
| CKD | 486 (12.9) | 3,216 (11.0) | 362 (11.7) | 0.002 a | 3,208 (10.9) | 414 (13.2) | 442 (12.6) | <0.001 d e |
| Heart failure | 547 (14.5) | 3,773 (12.9) | 376 (12.2) | 0.007 a b | 3,812 (12.9) | 378 (12.0) | 506 (14.4) | 0.012 e f |
| Hypertension | 2,701 (71.7) | 18,500 (63.2) | 2,169 (70.2) | <0.001 a c | 18,846 (64.0) | 2,041 (65.0) | 2,483 (70.7) | <0.001 e f |
| CAD | 1,412 (37.5) | 10,365 (35.4) | 1,288 (41.7) | <0.001 a b c | 10,588 (35.9) | 1,234 (39.3) | 1,243 (35.4) | 0.001 d f |
| Malignant neoplasm | 530 (14.1) | 4,217 (14.4) | 459 (14.9) | 0.650 | 4,239 (14.4) | 501 (16.0) | 466 (13.3) | 0.008 d f |
| Medication––no. (%) | ||||||||
| Antibiotics | 3,653 (97.0) | 28,216 (96.5) | 2,952 (95.6) | 0.007 b c | 28,415 (96.5) | 2,993 (95.3) | 3,413 (97.2) | <0.001 d e f |
| Warfarin | 362 (9.6) | 2,205 (7.5) | 202 (6.5) | <0.001 a b c | 2,192 (7.4) | 243 (7.7) | 334 (9.5) | <0.001 e f |
| Steroid | 447 (11.9) | 3,120 (10.7) | 358 (11.6) | 0.033 a | 3,065 (10.4) | 445 (14.2) | 415 (11.8) | <0.001 d e f |
| Anti-platelet | 1,638 (43.5) | 12,161 (41.6) | 1,380 (44.7) | 0.001 a c | 12,075 (41.0) | 1,565 (49.8) | 1,539 (43.8) | <0.001 d e f |
| Hospital level––no. (%) | <0.001 a b c | <0.001 d f | ||||||
| Medical center | 2,347 (62.3) | 16,821 (57.5) | 1,382 (44.8) | 16,999 (57.7) | 1,524 (48.5) | 2,027 (57.7) | ||
| Metropolitan | 1,355 (36.0) | 11,667 (39.9) | 1,591 (51.5) | 11,652 (39.6) | 1,556 (49.6) | 1,405 (40.0) | ||
| Local community | 64 (1.7) | 762 (2.6) | 115 (3.7) | 802 (2.7) | 60 (1.9) | 79 (2.3) | ||
Data are presented as mean ± SD or number (percentage).
AF = atrial fibrillation; AV block = atrioventricular block; CAD = coronary artery disease; CKD = chronic kidney disease; PPM = permanent pacemaker; SD = standard deviation
a P < 0.05 for active fixation vs. passive fixation
b P < 0.05 for active fixation vs. both fixation
c P < 0.05 for passive fixation vs. both fixation
d P< 0.05 for silicon vs. polyurethane
e P < 0.05 for silicon vs. both
f P < 0.05 for polyurethane vs. both.
Pacemaker-Related Heart Perforation
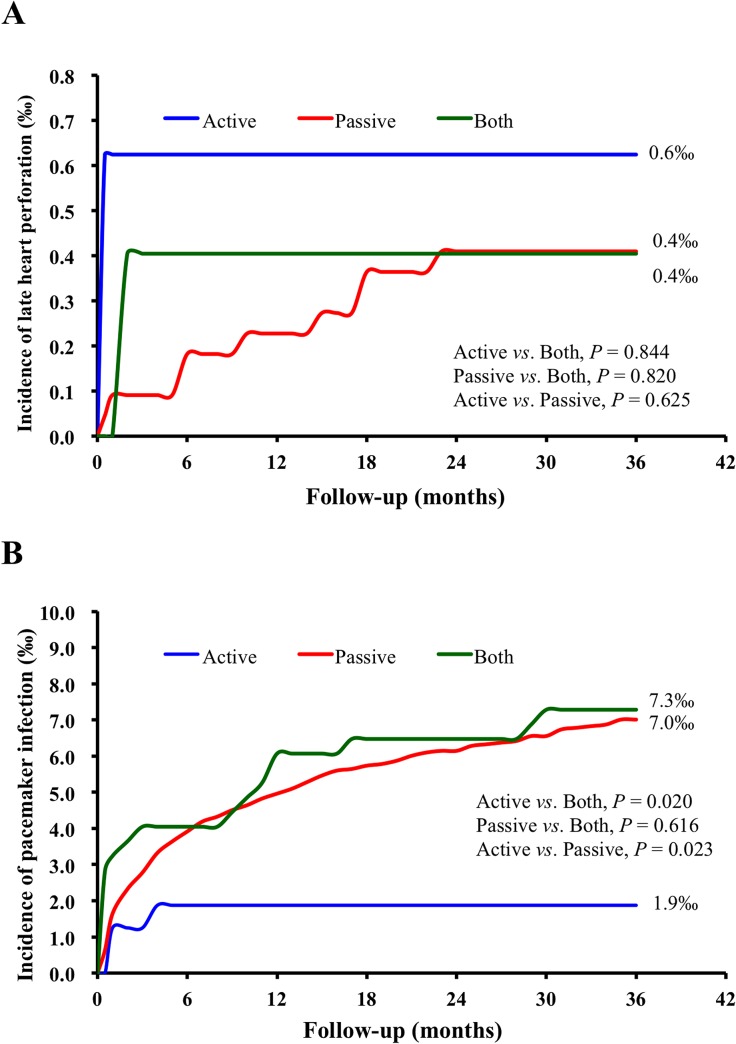
Regarding early heart perforation, during the 14-year study period, there were 22 heart perforation (0.06%) events. In the multivariable logistic regression analysis, the incidence of early heart perforation was not significantly different between groups with different fixation methods, and there were also no significant differences between groups with different insulations (Table 2). Regarding the late heart perforation, the incidence of complications did not differ year by year according to the new implant year (S2 and S3 Tables). In the 3-year follow-up period, the incidence of late heart perforation was 0.04% (11 out of 26,047). The cumulative incidence of late heart perforation did not differ for the different types of fixation (Fig 2 and Table 3) and insulations (Fig 3 and Table 4).
Table 2. Lead Fixation Method and Lead Insulation vs. Early Heart Perforation Under Logistic Regression Analysis.
| Number of Event––no. (%) | Adjusted Odds Ratio and 95% CI | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Active | Passive | Both | Active vs. Both | Passive vs. Both | Active vs. Passive | ||||
| Outcome | (n = 3,766) | (n = 29,250) | (n = 3,088) | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P |
| Early heart perforation | 3 (0.08) | 17 (0.06) | 2 (0.06) | 1.16 (0.19–7.23) | 0.870 | 0.66 (0.11–3.93) | 0.647 | 1.77 (0.41–7.54) | 0.441 |
| Silicone | Polyurethane | Both Insulation | Silicone vs. Both Insulation | Polyurethane vs. Both Insulation | Silicone vs. Polyurethane | ||||
| Outcome | (n = 29,453) | (n = 3,140) | (n = 3,511) | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P |
| Early heart perforation | 16 (0.05) | 2 (0.06) | 4 (0.11) | 0.54 (0.18–1.64) | 0.275 | 0.47 (0.08–2.60) | 0.384 | 1.15 (0.26–5.10) | 0.850 |
The odds ratios (OR) were adjusted for all covariates listed in Table 1.
Fig 2. Kaplan–Meier estimates of the cumulative incidence of complications with different fixation types during three years of follow-up.
(A) The cumulative incidence of late heart perforation; (B) The cumulative incidence of early and late complications of pacemaker infection. The blue line indicates active fixation group, red line the passive fixation group, and the green line the both-fixation group.
Table 3. Lead Fixation Method vs. Late Heart Perforation and Infection Under Cox Logistic Regression Analysis.
| Number of Event––no. (%) | Adjusted Hazard Ratio and 95% CI | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Active | Passive | Both | Active vs. Both | Passive vs. Both | Active vs. Passive | ||||
| Outcome | (n = 1,602) | (n = 21,973) | (n = 2,472) | HR (95% CI) | P | HR (95% CI) | P | HR (95% CI) | P |
| Late heart perforation | 1 (0.06) | 9 (0.04) | 1 (0.04) | 1.33 (0.08–22.02) | 0.844 | 0.79 (0.10–6.26) | 0.820 | 1.69 (0.21–13.77) | 0.625 |
| Infection | 3 (0.19) | 154 (0.70) | 18 (0.73) | 0.23 (0.07–0.80) | 0.020 | 0.88 (0.54–1.44) | 0.616 | 0.26 (0.08–0.83) | 0.023 |
| Early vs. late | |||||||||
| Early infection | 3 (0.19) | 106 (0.48) | 13 (0.53) | 0.31 (0.09–1.08) | 0.066 | 0.85 (0.47–1.51) | 0.571 | 0.36 (0.11–1.14) | 0.083 |
| Late infection | 0 (0.00) | 48 (0.22) | 5 (0.20) | NA | NA | 0.99 (0.39–2.50) | 0.977 | NA | NA |
| Need of lead-extraction vs. without lead extraction | |||||||||
| Need of lead extraction | 2 (0.12) | 90 (0.41) | 13 (0.53) | 0.24 (0.05–1.06) | 0.060 | 0.77 (0.43–1.39) | 0.394 | 0.31 (0.08–1.25) | 0.099 |
| Without lead extraction | 1 (0.06) | 64 (0.29) | 5 (0.20) | 0.24 (0.03–2.04) | 0.189 | 1.17 (0.47–2.93) | 0.734 | 0.20 (0.03–1.46) | 0.112 |
The hazard ratios (HR) were adjusted for all covariates listed in Table 1.
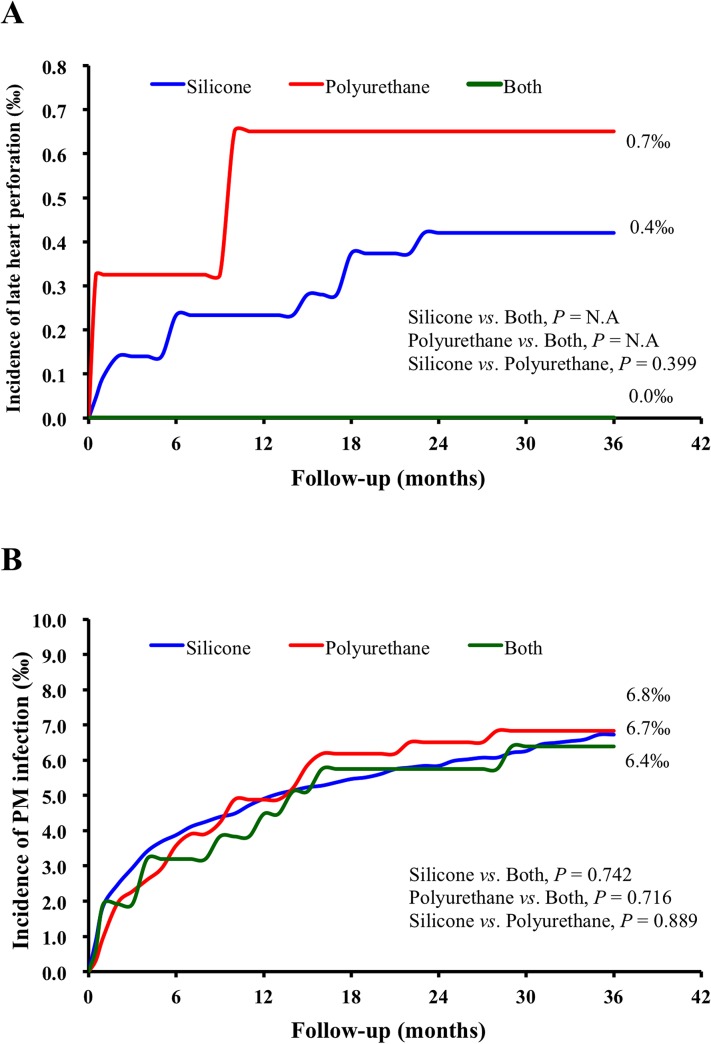
Fig 3. Kaplan–Meier estimates of the cumulative incidence of complications with different insulation materials during three years follow-up.
(A) The cumulative incidence of late heart perforation; (B) The cumulative incidence of early and late complications of pacemaker infection. The blue line indicates the silicone group, the red line the polyurethane group, and the green line the both-coatings group.
Table 4. Lead Insulation vs. Late Heart Perforation and Infection Under Cox Logistic Regression Analysis.
| Number of Event––no. (%) | Adjusted Hazard Ratio and 95% CI | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Silicone | Polyurethane | Combined | Silicone vs. Combined | Polyurethane vs. Combined | Silicone vs. Polyurethane | ||||
| Outcome | (n = 21,409) | (n = 3,073) | (n = 1,565) | HR (95% CI) | P | HR (95% CI) | P | HR (95% CI) | P |
| Late heart perforation | 9 (0.04) | 2 (0.07) | 0 (0.00) | NA | NA | NA | NA | 0.51 (0.11–2.44) | 0.399 |
| Infection | 144 (0.67) | 21 (0.68) | 10 (0.64) | 1.11 (0.58–2.13) | 0.742 | 1.15 (0.54–2.47) | 0.716 | 0.97 (0.61–1.54) | 0.889 |
| Early vs. late | |||||||||
| Early infection | 101 (0.47) | 15 (0.49) | 6 (0.38) | 1.38 (0.60–3.16) | 0.447 | 1.43 (0.55–3.72) | 0.467 | 0.97 (0.56–1.67) | 0.906 |
| Late infection | 43 (0.20) | 6 (0.20) | 4 (0.26) | 0.73 (0.26–2.06) | 0.553 | 0.76 (0.21–2.77) | 0.683 | 0.95 (0.40–2.26) | 0.915 |
| Need of lead-extraction vs. without lead extraction | |||||||||
| Need of lead-extraction | 82 (0.38) | 17 (0.55) | 6 (0.38) | 1.05 (0.45–2.41) | 0.915 | 1.51 (0.59–3.88) | 0.390 | 0.69 (0.41–1.17) | 0.172 |
| Without lead-extraction | 62 (0.29) | 4 (0.13) | 4 (0.26) | 1.25 (0.45–3.47) | 0.670 | 0.61 (0.15–2.47) | 0.487 | 2.06 (0.74–5.68) | 0.165 |
The hazard ratios (OR) were adjusted for all covariates listed in Table 1.
Pacemaker-Related Infection
During the 3-year follow-up period, the incidence of PM-related infection was 0.67% (175 out of 26,047). In the multivariable Cox-regression analysis, the PM-related cumulative infection rate was significantly lower in the active fixation group compared to either the both fixation group (OR, 0.23; 95% CI, 0.07–0.80; P = 0.020) or the passive fixation group (OR, 0.26; 95% CI, 0.08–0.83; P = 0.023) (Fig 2 and Table 3). In the subgroup analysis, the active fixation group had a lower early PM-related infection rate and a lower rate of PM-related infection that required lead extractions compared to either the both fixation group or the passive fixation group (Table 3). Moreover, there was no late infection (0%) in the active fixation group compared to 0.20% in the both fixation group and 0.22% in the passive fixation group (Table 3).
Regarding the insulations, the types of lead insulation had no impact on the cumulative incidence of PM-related infection (Fig 3 and Table 4). In the subgroup analysis, the insulation types of leads also did not differ in terms of the incidence of early and late infection and infection that required lead extraction or infection without lead extraction (Table 4).
Discussion
This is the first study to investigate the impact of characteristics of leads on the major PM-related complications. In this nationwide cohort study, there was no difference in heart perforations between active and passive fixation leads. The active fixation leads were associated with reduced risk of pacemaker-related infection.
Pacemaker-Related Heart Perforation
In the literature, most reported heart perforation events occurred in implantable cardiac defibrillator [18] patients with the correlation between heart perforation and lead characteristics usually being studied in implantable cardiac defibrillator patients [19]. In PM patients, no large cohort study has ever been conducted to investigate the clinical impact of lead characteristics on heart perforation, except for some series of case reports [12, 20–22]. In general, the active fixation type was reported to be a risk factor of PM-related heart perforation [13,21,22]. Furthermore, other than the patient’s characteristics, other reported risk factors for PM-related heart perforation include the location of lead tip [22], temporal pacemaker lead [14], and the operator experience. Although the details of procedures, such as tip location, were not available in this study, some case control studies showed that the location of tip had no statistically significant impact on incidence of heart perforation [13,22]. In this study, we evaluated the relationship between the characteristics of leads and heart perforation after adjusting for all patient characteristics, medication, and operator’s volume, and we found that both the fixation type and insulation materials of leads did not differ in the incidence of either early or late heart perforation.
Pacemaker-Related Infection
Pacemaker-related infection has attracted more attention and many studies have investigated the risk factors of PM-related infection. In some research, PM-related infection was sub-grouped into early and late infection and risks related to early and late infection were found to be different [23]. Among the well-known risks [7–11, 23–25] all procedure-related factors contributed to the early PM-related infection and some comorbidities were associated with late infection. Unfortunately, the characteristics of leads have been paid scant attention in PM-related infection. In our study, we investigated the relationship between the characteristics of lead and early/late infection and whether or not the infection necessitated lead extraction. We found that the majority of PM-related infection events were early infection, and the PM-related cumulative infection rate was significantly lower in the active fixation group compared to the both fixation group and the passive fixation group. In the subgroup analysis, the active fixation group had a lower early PM-related infection rate and a lower rate of PM-related infection that required lead extraction compared to either the both fixation group or the passive fixation group. Other than active fixation leads, we could not exclude some procedure-related risk factors that might have impacted on PM-related infection. However, we were unable to obtain the details of the procedure in this insurance database. Further studies are thus warranted to examine the biological mechanisms of our observations.
Limitations
This retrospective analysis bears the inherent limitations of these types of studies. One major limitation was that procedure details could not be obtained, since data were from an insurance system where diseases were classified according to ICD-9 code (Table 1), and payment was just made according to procedure type. Therefore, procedure-related variables, which might affect the early complications, could not be analyzed, thus possibly introducing some bias. In the analysis of the early infection, such issues did in fact exist. Further study should be conducted to confirm these observations that included procedure details in the analysis. Another limitation is that the severity of the complications could not be obtained in this insurance database, and some complications may have been underestimated. Nevertheless, the complications requiring treatment were more important clinically than those that required no therapy.
Conclusions
In bradyarrhythmic patients, there was no difference in heart perforations between active and passive fixation leads, and active fixation leads were associated with reduced risk of pacemaker-related infection.
Supporting Information
(DOCX)
(DOCX)
(DOCX)
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
The authors have no support or funding to report.
References
- 1. Brignole M, Auricchio A, Baron-Esquivias G, Bordachar P, Boriani G, Breithardt OA, et al. 2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy: the Task Force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA). Eur Heart J 2013;29:2281–329. 10.1093/eurheartj/eht150 [DOI] [PubMed] [Google Scholar]
- 2. Dickstein K, Vardas PE, Auricchio A, Daubert JC, Linde C, McMurray J, et al. 2010 Focused Update of ESC Guidelines on device therapy in heart failure: an update of the 2008 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure and the 2007 ESC guidelines for cardiac and resynchronization therapy. Developed with the special contribution of the Heart Failure Association and the European Heart Rhythm Association. Eur Heart J 2010;31:2677–87. 10.1093/eurheartj/ehq337 [DOI] [PubMed] [Google Scholar]
- 3. Mond HG, Proclemer A. The 11th world survey of cardiac pacing and implantable cardioverter-defibrillators: calendar year 2009—a World Society of Arrhythmia's project. PACE 2011;34:1013–27. 10.1111/j.1540-8159.2011.03150.x [DOI] [PubMed] [Google Scholar]
- 4. Voigt A, Shalaby A, Saba S. Continued rise in rates of cardiovascular implantable electronic device infections in the United States: temporal trends and causative insights. PACE 2010;33:414–9. 10.1111/j.1540-8159.2009.02569.x [DOI] [PubMed] [Google Scholar]
- 5. Cabell CH, Heidenreich PA, Chu VH, Moore CM, Stryjewski ME, Corey GR, et al. Increasing rates of cardiac device infections among Medicare beneficiaries: 1990–1999. Am Heart J 2004;147:582–6. [DOI] [PubMed] [Google Scholar]
- 6. Sohail MR, Henrikson CA, Braid-Forbes MJ, Forbes KF, Lerner DJ. Mortality and cost associated with cardiovascular implantable electronic device infections. Arch Intern Med 2011;171:1821–8. 10.1001/archinternmed.2011.441 [DOI] [PubMed] [Google Scholar]
- 7. Tompkins C, McLean R, Cheng A, Brinker JA, Marine JE, Nazarian S, et al. End-stage renal disease predicts complications in pacemaker and ICD implants. J Cardiovasc Electrophysiol 2011;22:1099–104. 10.1111/j.1540-8167.2011.02066.x [DOI] [PubMed] [Google Scholar]
- 8. Nery PB, Fernandes R, Nair GM, Sumner GL, Ribas CS, Menon SM, et al. Device-related infection among patients with pacemakers and implantable defibrillators: incidence, risk factors, and consequences. J Cardiovasc Electrophysiol 2010;21:786–90. 10.1111/j.1540-8167.2009.01690.x [DOI] [PubMed] [Google Scholar]
- 9. Johansen JB, Jørgensen OD, Møller M, Arnsbo P, Mortensen PT, Nielsen JC. Infection after pacemaker implantation: infection rates and risk factors associated with infection in a population-based cohort study of 46299 consecutive patients. Eur Heart J 2011;32:991–8. 10.1093/eurheartj/ehq497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Sohail MR, Uslan DZ, Khan AH, Friedman PA, Hayes DL, Wilson WR, et al. Risk factor analysis of permanent pacemaker infection. Clin Infect Dis 2007;45:166–73. [DOI] [PubMed] [Google Scholar]
- 11. Klug D, Balde M, Pavin D, Hidden-Lucet F, Clementy J, Sadoul N, et al. Risk factors related to infections of implanted pacemakers and cardioverter-defibrillators: results of a large prospective study. Circulation 2007;116:1349–55. [DOI] [PubMed] [Google Scholar]
- 12. Piekarz J, Lelakowski J, Rydlewska A, Majewski J. Heart perforation in patients with permanent cardiac pacing—pilot personal observations. Arch Med Sci 2012;8:70–4. 10.5114/aoms.2012.27284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Mahapatra S, Bybee KA, Bunch TJ, Espinosa RE, Sinak LJ, McGoon MD, et al. Incidence and predictors of cardiac perforation after permanent pacemaker placement. Heart Rhythm 2005;2:907–11. [DOI] [PubMed] [Google Scholar]
- 14. Piekarz J, Lelakowski J, Rydlewska A, Majewski J. Heart perforation in patients with permanent cardiac pacing—pilot personal observations. Arch Med Sci. 2012;8:70–74. 10.5114/aoms.2012.27284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Selcuk H, Selcuk MT, Maden O, Ozeke O, Celenk MK, Turkvatan A, et al. Uncomplicated heart and lung perforation by a displaced ventricular pacemaker lead: a case report. PACE 2006;29:429–30. [DOI] [PubMed] [Google Scholar]
- 16.National Health Insurance Administration, Ministry of Health and Welfare, Taiwan, R.O.C. (2014). National Health Insurance Annual Report 2014–2015. Available: http://www.nhi.gov.tw/Nhi_E-LibraryPubWeb/CustomPage/P_Detail.aspx?CP_ID=129.
- 17. Cheng CL, Kao YH, Lin SJ, Lee CH, Lai ML. Validation of the National Health Insurance Research Database with ischemic stroke cases in Taiwan. Pharmacoepidemiol Drug Saf 2011;20:236–42. 10.1002/pds.2087 [DOI] [PubMed] [Google Scholar]
- 18. Refaat MM, Hashash JG, Shalaby AA. Late perforation by cardiac implantable electronic device leads: clinical presentation, diagnostic clues, and management. Clin Cardiol. 2010;33:466–75. 10.1002/clc.20803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Danik SB, Mansour M, Singh J, Reddy VY, Ellinor PT, Milan D, et al. Increased incidence of subacute lead perforation noted with one implantable cardioverter-defibrillator. Heart Rhythm 2007;4:439–42. [DOI] [PubMed] [Google Scholar]
- 20. Geyfman V, Storm RH, Lico SC, Oren JW 4th. Cardiac tamponade as complication of active-fixation atrial lead perforations: proposed mechanism and management algorithm. PACE 2007;30:498–501. [DOI] [PubMed] [Google Scholar]
- 21. Sterliński M, Przybylski A, Maciag A, Syska P, Pytkowski M, Lewandowski M, et al. Subacute cardiac perforations associated with active fixation leads. Europace 2009;11:206–12. 10.1093/europace/eun363 [DOI] [PubMed] [Google Scholar]
- 22. Ohlow MA, Lauer B, Brunelli M, Geller JC. Incidence and predictors of pericardial effusion after permanent heart rhythm device implantation: prospective evaluation of 968 consecutive patients. Circ J 2013;77;975–81. [DOI] [PubMed] [Google Scholar]
- 23. Sohail MR, Hussain S, Le KY, Dib C, Lohse CM, Friedman PA, et al. Risk factors associated with early- versus late-onset implantable cardioverter-defibrillator infections. J Interv Card Electrophysiol 2011;31:171–83. 10.1007/s10840-010-9537-x [DOI] [PubMed] [Google Scholar]
- 24. Dasgupta A, Montalvo J, Medendorp S, Lloyd-Jones DM, Ghossein C, Goldberger J, et al. Increased complication rates of cardiac rhythm management devices in ESRD patients. Am J Kidney Dis 2007;49:656–63. [DOI] [PubMed] [Google Scholar]
- 25. Lekkerkerker JC, van Nieuwkoop C, Trines SA, van der Bom JG, Bernards A, van de Velde ET, et al. Risk factors and time delay associated with cardiac device infections: Leiden device registry. Heart 2009;95:715–20. 10.1136/hrt.2008.151985 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.