Abstract
Background
Accurate, rapid detection of tuberculosis (TB) and TB drug resistance is critical for improving patient care and decreasing TB transmission. Xpert® MTB/RIF assay is an automated test that can detect both TB and rifampicin resistance, generally within two hours after starting the test, with minimal hands‐on technical time. The World Health Organization (WHO) issued initial recommendations on Xpert® MTB/RIF in early 2011. A Cochrane Review on the diagnostic accuracy of Xpert® MTB/RIF for pulmonary TB and rifampicin resistance was published January 2013. We performed this updated Cochrane Review as part of a WHO process to develop updated guidelines on the use of the test.
Objectives
To assess the diagnostic accuracy of Xpert® MTB/RIF for pulmonary TB (TB detection), where Xpert® MTB/RIF was used as both an initial test replacing microscopy and an add‐on test following a negative smear microscopy result.
To assess the diagnostic accuracy of Xpert® MTB/RIF for rifampicin resistance detection, where Xpert® MTB/RIF was used as the initial test replacing culture‐based drug susceptibility testing (DST).
The populations of interest were adults presumed to have pulmonary, rifampicin‐resistant or multidrug‐resistant TB (MDR‐TB), with or without HIV infection. The settings of interest were intermediate‐ and peripheral‐level laboratories. The latter may be associated with primary health care facilities.
Search methods
We searched for publications in any language up to 7 February 2013 in the following databases: Cochrane Infectious Diseases Group Specialized Register; MEDLINE; EMBASE; ISI Web of Knowledge; MEDION; LILACS; BIOSIS; and SCOPUS. We also searched the metaRegister of Controlled Trials (mRCT) and the search portal of the WHO International Clinical Trials Registry Platform to identify ongoing trials.
Selection criteria
We included randomized controlled trials, cross‐sectional studies, and cohort studies using respiratory specimens that allowed for extraction of data evaluating Xpert® MTB/RIF against the reference standard. We excluded gastric fluid specimens. The reference standard for TB was culture and for rifampicin resistance was phenotypic culture‐based DST.
Data collection and analysis
For each study, two review authors independently extracted data using a standardized form. When possible, we extracted data for subgroups by smear and HIV status. We assessed the quality of studies using QUADAS‐2 and carried out meta‐analyses to estimate pooled sensitivity and specificity of Xpert® MTB/RIF separately for TB detection and rifampicin resistance detection. For TB detection, we performed the majority of analyses using a bivariate random‐effects model and compared the sensitivity of Xpert® MTB/RIF and smear microscopy against culture as reference standard. For rifampicin resistance detection, we undertook univariate meta‐analyses for sensitivity and specificity separately to include studies in which no rifampicin resistance was detected.
Main results
We included 27 unique studies (integrating nine new studies) involving 9557 participants. Sixteen studies (59%) were performed in low‐ or middle‐income countries. For all QUADAS‐2 domains, most studies were at low risk of bias and low concern regarding applicability.
As an initial test replacing smear microscopy, Xpert® MTB/RIF pooled sensitivity was 89% [95% Credible Interval (CrI) 85% to 92%] and pooled specificity 99% (95% CrI 98% to 99%), (22 studies, 8998 participants: 2953 confirmed TB, 6045 non‐TB). As an add‐on test following a negative smear microscopy result, Xpert®MTB/RIF pooled sensitivity was 67% (95% CrI 60% to 74%) and pooled specificity 99% (95% CrI 98% to 99%; 21 studies, 6950 participants).
For smear‐positive, culture‐positive TB, Xpert® MTB/RIF pooled sensitivity was 98% (95% CrI 97% to 99%; 21 studies, 1936 participants).
For people with HIV infection, Xpert® MTB/RIF pooled sensitivity was 79% (95% CrI 70% to 86%; seven studies, 1789 participants), and for people without HIV infection, it was 86% (95% CrI 76% to 92%; seven studies, 1470 participants).
Among 180 specimens with nontuberculous mycobacteria (NTM), Xpert® MTB/RIF was positive in only one specimen that grew NTM (14 studies, 2626 participants).
Comparison with smear microscopy
In comparison with smear microscopy, Xpert® MTB/RIF increased TB detection among culture‐confirmed cases by 23% (95% CrI 15% to 32%; 21 studies, 8880 participants).
For TB detection, if pooled sensitivity estimates for Xpert® MTB/RIF and smear microscopy are applied to a hypothetical cohort of 1000 patients where 10% of those with symptoms have TB, Xpert® MTB/RIF will diagnose 88 cases and miss 12 cases, whereas sputum microscopy will diagnose 65 cases and miss 35 cases.
Rifampicin resistance
For rifampicin resistance detection, Xpert® MTB/RIF pooled sensitivity was 95% (95% CrI 90% to 97%; 17 studies, 555 rifampicin resistance positives) and pooled specificity was 98% (95% CrI 97% to 99%; 24 studies, 2411 rifampicin resistance negatives).
For rifampicin resistance detection, if the pooled accuracy estimates for Xpert® MTB/RIF are applied to a hypothetical cohort of 1000 individuals where 15% of those with symptoms are rifampicin resistant, Xpert® MTB/RIF would correctly identify 143 individuals as rifampicin resistant and miss eight cases, and correctly identify 833 individuals as rifampicin susceptible and misclassify 17 individuals as resistant. Where 5% of those with symptoms are rifampicin resistant, Xpert® MTB/RIF would correctly identify 48 individuals as rifampicin resistant and miss three cases and correctly identify 931 individuals as rifampicin susceptible and misclassify 19 individuals as resistant.
Authors' conclusions
In adults thought to have TB, with or without HIV infection, Xpert® MTB/RIF is sensitive and specific. Compared with smear microscopy, Xpert® MTB/RIF substantially increases TB detection among culture‐confirmed cases. Xpert® MTB/RIF has higher sensitivity for TB detection in smear‐positive than smear‐negative patients. Nonetheless, this test may be valuable as an add‐on test following smear microscopy in patients previously found to be smear‐negative. For rifampicin resistance detection, Xpert® MTB/RIF provides accurate results and can allow rapid initiation of MDR‐TB treatment, pending results from conventional culture and DST. The tests are expensive, so current research evaluating the use of Xpert® MTB/RIF in TB programmes in high TB burden settings will help evaluate how this investment may help start treatment promptly and improve outcomes.
Keywords: Adult; Humans; Drug Resistance, Bacterial; Antibiotics, Antitubercular; Antibiotics, Antitubercular/therapeutic use; Mycobacterium tuberculosis; Mycobacterium tuberculosis/drug effects; Mycobacterium tuberculosis/genetics; Mycobacterium tuberculosis/isolation & purification; Polymerase Chain Reaction; Polymerase Chain Reaction/methods; Rifampin; Rifampin/therapeutic use; Sensitivity and Specificity; Sequence Analysis, DNA; Sequence Analysis, DNA/methods; Tuberculosis, Pulmonary; Tuberculosis, Pulmonary/diagnosis; Tuberculosis, Pulmonary/drug therapy
Xpert MTB/RIF test for diagnosing pulmonary tuberculosis and rifampicin resistance in adults
Tuberculosis (TB) causes tremendous suffering worldwide, especially in low‐income and middle‐income countries. In 2012, 8.6 million people developed TB disease (active TB) for the first time and around 1.3 million people died. Most people with TB can be cured if the disease is diagnosed and properly treated. One of the problems in treating TB is that the bacteria become resistant to antibiotics. Detecting TB and TB drug resistance quickly is important for improving health, reducing deaths, and decreasing the spread of TB in communities.
Xpert® MTB/RIF is a new test that quickly detects TB and rifampicin resistance at the same time. Rifampicin is an important drug for treating people with TB. Since the test is automated, it does not require expert staff or an advanced laboratory.
Our objectives were to determine the diagnostic accuracy (sensitivity and specificity) for TB detection and rifampicin resistance detection. Sensitivity shows how often the test gives a positive result in people who really have TB. Specificity shows how often the test gives a negative result in people who do not have TB.
We included studies of adults with or without HIV infection thought to have pulmonary TB (TB in the lungs) or rifampicin resistance, and were most interested in the use of Xpert® MTB/RIF outside of the most advanced laboratories.
We also compared the sensitivity of Xpert® MTB/RIF to that of smear microscopy, the test commonly used for TB diagnosis in low‐ and middle‐income countries. Smear microscopy is low‐cost and fairly easy to do, but requires trained staff and is a hassle for patients, who must provide at least two sputum samples. Also, microscopy gives no information about drug resistance.
We searched for publications in any language up to 7 February 2013 and considered the study's risk of giving biased results.
What the results say
We included 27 studies involving around 9500 people. Most studies were performed in low‐ or middle‐income countries. We thought most studies had a low risk of bias.
The key findings were:
For TB detection, Xpert® MTB/RIF was accurate (it was highly sensitive (89%), detecting almost all cases; and specific (99%), that is, not registering positive in people who were actually negative).
For rifampicin resistance detection, Xpert® MTB/RIF was accurate that is sensitive (95%) and specific (98%).
Xpert® MTB/RIF appeared to have similar accuracy in people with and without HIV infection.
Applying the findings of the review to an imaginary group of 1000 people who go to their doctor with symptoms, but where only 100 of them (10%) actually have TB, Xpert® MTB/RIF would diagnose 88 cases and miss 12 cases, whereas smear microscopy would diagnose 65 cases and miss 35 cases.
To summarize, our review shows that Xpert® MTB/RIF is more accurate than smear microscopy for diagnosing TB and also accurate for detecting rifampicin resistance. Xpert® MTB/RIF may be useful in many countries, as it does not require advanced laboratory facilities or expert staff.
Summary of findings
Summary of findings 1.
Xpert MTB/RIF for TB detection
|
Review question: What is the diagnostic accuracy of Xpert MTB/RIF assay for detection of pulmonary TB? Patients/population: Adults with presumed pulmonary TB Role: Xpert MTB/RIF assay used as an initial test replacing microscopy and used as an add‐on test following a negative smear microscopy result Index test: Xpert MTB/RIF assay Reference standards: Solid or liquid culture Studies: Cross‐sectional Setting: Mainly intermediate level laboratories | ||||||
| Type of analysis | Effect (95% credible interval) | No. of participants (studies) | Test result | Number of results per 1000 patients tested (95% CrI) 1 | ||
| Prevalence 2.5% | Prevalence 5% | Prevalence 10% | ||||
| TB detection, Xpert MTB/RIF used as an initial test replacing smear microscopy | Median pooled sensitivity 89% (85, 92) and median pooled specificity 99% (98, 99) |
8998 (22) |
True Positives False Negatives False Positives True Negatives |
22 (21, 23) 3 (2, 4) 10 (10, 20) 965 (956, 965) |
45 (43, 46) 6 (4, 8) 10 (10,19) 941 (931, 941) |
89 (85, 92) 11 (8, 15) 9 (9, 18) 891 (882, 891) |
| Smear‐positive, culture‐positive | Median pooled sensitivity 98% (97, 99); specificity of Xpert MTB/RIF could not be estimated in these studies | 1936 (21) |
True Positives False Negatives False Positives True Negatives |
25 (24, 25) 1 (0, 1) *** *** |
49 (49, 50) 1 (1, 2) *** *** |
98 (97, 98) 2 (1, 3) *** *** |
| Smear‐negative, culture‐positive | Median pooled sensitivity 67% (60, 74) and median pooled specificity 99% (98, 99) | 7565 (21) |
True Positives False Negatives False Positives True Negatives |
17 (15, 19) 8 (7, 10) 10 (10, 20) 965 (956, 965) |
34 (31, 37) 16 (13, 20) 10 (10, 19) 941 (931, 941) |
68 (61, 74) 32 (26, 39) 9 (9, 18) 891 (882, 891) |
| HIV‐positive | Median pooled sensitivity 79% (70, 86) and median pooled specificity 98% (96, 99) | 1789 (7) |
True Positives False Negatives False Positives True Negatives |
20 (18, 22) 5 (4, 8) 20 (10, 39) 956 (936, 965) |
40 (35, 43) 11 (7,15) 19 (10,38) 931 (912, 941) |
79 (70, 86) 21 (14, 30) 18 (9, 36) 882 (864, 891) |
| HIV‐negative | Median pooled sensitivity 86% (76, 92) and median pooled specificity 99% (98, 100) | 1470 (7) |
True Positives False Negatives False Positives True Negatives |
22 (19, 23) 4 (2, 6) 10 (10, 20) 965 (956, 965) |
43 (38, 46) 7 (4,12) 10 (10,19) 941 (931, 941) |
86 (76, 92) 14 (8, 24) 9 (9, 18) 891 (882, 891) |
| TB detection, Xpert MTB/RIF used as an add‐on test following a negative smear microscopy result | Median pooled sensitivity 67% (60, 74) and median pooled specificity 99% (98, 99) | 7151 (23) |
True Positives False Negatives False Positives True Negatives |
17 (15, 19) 8 (7, 10) 10 (10, 20) 965 (956, 965) |
34 (30, 37) 17 (13, 20) 10 (10,19) 941 (931, 941) |
67 (60, 74) 33 (26, 40) 9 (9, 18) 891 (882, 891) |
1. The WHO suggested prevalence levels.
Summary of findings 2.
Incremental yield of Xpert MTB/RIF compared with smear microscopy
| Test result | Number of results per 1000 adults with presumed pulmonary TB (95% Crl) | |||||||
| Prevalence 2.5% | Prevalence 5% | Prevalence 10% | Prevalence 30% | |||||
| Smear Microscopy | Xpert MTB/RIF | Smear Microscopy | Xpert MTB/RIF | Smear Microscopy | Xpert MTB/RIF | Smear Microscopy | Xpert MTB/RIF | |
| True positives | 16 (14, 18) | 22 (21, 23) | 33 (29, 36) | 44 (42, 46) | 65 (57, 72) | 88 (84, 92) | 195 (171, 216) | 264 (252, 276) |
| Mean absolute difference in true positives | 6 more | 11 more | 23 more | 69 more | ||||
| False negatives | 9 (7, 11) | 3 (2, 4) | 18 (14, 22) | 6 (4, 8) | 35 (28, 43) | 12 (8, 16) |
105 (84, 129) | 36 (24, 48) |
| Mean absolute difference in false negatives | 6 less | 12 less | 23 less | 69 less | ||||
Summary of findings 3.
Xpert MTB/RIF for rifampicin resistance detection
|
Review question: What is the diagnostic accuracy of Xpert MTB/RIF assay for detection of rifampicin resistance? Patients/population: Adults with confirmed TB Role: Xpert MTB/RIF assay as an initial test replacing conventional phenotypic DST Index test: Xpert MTB/RIF assay Reference standards: Phenotypic culture‐based DST Studies: Cross‐sectional Setting: Mainly intermediate level laboratories | |||||
| Type of analysis |
Effect (95% credible interval) |
No. of participants (studies) | Test result | Number of results per 1000 patients tested (95% CrI) 1 | |
| Prevalence 5% | Prevalence 15% | ||||
| Rifampicin resistance detection, Xpert MTB/RIF used as an initial test replacing conventional DST |
Median pooled sensitivity 95% (90, 97) and median pooled specificity 98% (97, 99) | Median pooled sensitivity 555 (17) Median pooled specificity 2411 (24) |
True Positives False Negatives False Positives True Negatives |
48 (45, 49) 3 (2, 5) 19 (10, 29) 931 (922, 941) |
143 (135, 146) 8 (5, 15) 17 (9, 26) 833 (825, 842) |
1. The WHO suggested prevalence levels: 5% is considered equivalent to the upper limit for rifampicin resistance prevalence in new cases; 15% is considered equivalent to the lower limit for rifampicin resistance prevalence in previously treated cases.
Background
Tuberculosis (TB) is one of the world’s most important infectious causes of morbidity and mortality among adults. When TB is detected and effectively treated, the disease is largely curable. However, in 2012, 8.6 million people developed TB disease (active TB) for the first time (WHO Global Report 2013). Of the 8.6 million TB cases, 1.1 million, approximately 13%, occurred among people with HIV infection. In 2012, 1.3 million people died of TB, including 320,000 deaths (25%) among people who were HIV positive (WHO Global Report 2013).
Drug‐resistant TB, including multidrug‐resistant TB (MDR‐TB, defined as resistance to at least isoniazid and rifampicin, the two most important first‐line anti‐TB drugs) and extensively drug‐resistant TB (XDR‐TB, defined as MDR‐TB plus resistance to any fluoroquinolone, such as ofloxacin or moxifloxacin, and to at least one of three injectable second‐line drugs, amikacin, capreomycin, or kanamycin) has emerged as a serious threat to global health (Zumla 2012). In 2012, around 450,000 people developed MDR‐TB and an estimated 170,000 died from MDR‐TB (WHO Global Report 2013). Recently, the World Health Organization (WHO) reported the highest rates of MDR‐TB (greater than 65% in people who had previously received TB treatment) ever recorded in several areas of the former Soviet Union (Zignol 2012). Worldwide, for all forms of TB, a substantial percentage (˜35%) of patients are undiagnosed and a staggering percentage (˜75%) of patients with MDR‐TB remain undiagnosed (WHO Global Report 2013). Under 3% of people diagnosed with TB are tested to determine the pattern of drug resistance (Chaisson 2012). In addition to drug resistance, another major challenge is the accurate detection of smear‐negative disease, which disproportionately occurs in HIV‐positive people with TB (Harries 2004).
Accurate and rapid detection of TB, including smear‐negative TB and drug resistant‐TB, is critical for improving patient outcomes (increased cure and decreased mortality, additional drug resistance, treatment failure, and relapse) and decreasing TB transmission. Mycobacterial culture is generally considered the best available reference standard for TB diagnosis and is the first step in detecting drug resistance. However, culture is a relatively complex and slow procedure. Solid culture typically takes between four to eight weeks for results and liquid culture, though more rapid than solid culture, requires days and is more prone to contamination (WHO Policy Framework 2010). In addition, culture requires specialized laboratories and highly skilled staff. In early 2011, WHO endorsed a novel, rapid, automated, cartridge‐based nucleic acid amplification test (NAAT), the Xpert® MTB/RIF assay (Cepheid, Sunnyvale, USA) (hereafter referred to as Xpert MTB/RIF), that can simultaneously detect TB and rifampicin resistance (WHO Policy Xpert MTB/RIF 2011).
Target condition being diagnosed
Tuberculosis
TB is caused by the bacterium Mycobacterium tuberculosis and is spread from person to person through the air. TB most commonly affects the lungs (pulmonary TB), but may affect any organ or tissue, such as the brain or bones, outside of the lungs (extrapulmonary TB). Signs and symptoms of pulmonary TB include cough for at least two weeks, fever, chills, night sweats, weight loss, haemoptysis (coughing up blood), and fatigue. Signs and symptoms of extrapulmonary TB depend on the site of disease. TB treatment regimens must contain multiple drugs to which the organisms are sensitive to be effective. The treatment of MDR‐TB is complex, usually requiring two years or more of therapy and drugs that are less potent and more toxic than the drugs used to treat drug‐susceptible TB. The WHO issues international guidelines for TB treatment which are regularly updated.
Rifampicin resistance
Rifampicin inhibits bacterial DNA‐dependent RNA polymerase, encoded by the RNA polymerase gene (rpoB) (Hartmann 1967). Resistance to this drug has mainly been associated with mutations in a limited region of the rpoB gene (Telenti 1993). Rifampicin resistance may occur alone or in association with resistance to isoniazid and other drugs. In high MDR‐TB settings, the presence of rifampicin resistance alone may serve as a proxy for MDR‐TB (WHO Rapid Implementation 2011). Patients with drug‐resistant TB can transmit the infection to others.
Index test(s)
Xpert MTB/RIF is an automated polymerase chain reaction (PCR) test (molecular test) utilizing the GeneXpert® platform (Blakemore 2010Blakemore 2010; Cepheid 2009; Helb 2010). Xpert MTB/RIF is a single test that can detect both M. tuberculosis complex and rifampicin resistance within two hours after starting the test, with minimal hands‐on technical time. Unlike conventional nucleic acid amplification tests (NAATs), Xpert MTB/RIF is unique because sample processing and PCR amplification and detection are integrated into a single self‐enclosed test unit, the GeneXpert cartridge. Following sample loading, all steps in the assay are completely automated and self‐contained. In addition, the assay’s sample reagent, used to liquefy sputum, has potent tuberculocidal (the ability to kill TB bacteria) properties and so largely eliminates biosafety concerns during the test procedure (Banada 2010Banada 2010). These features allow the technology to be taken out of a reference laboratory and used nearer to the patient (Small 2011). Xpert MTB/RIF requires an uninterrupted and stable electrical power supply, temperature control, and yearly calibration of the cartridge modules (WHO Rapid Implementation 2011).
The test procedure may be used directly on clinical specimens, either raw sputum samples or sputum pellets (also called sputum sediment) created after decontaminating and concentrating the sputum (Blakemore 2010). In both cases, the test material is combined with the assay sample reagent, mixed by hand or vortex, and incubated at room temperature for 15 minutes. After the incubation step, 2 mL of the treated sample are transferred to the cartridge and the run is initiated (Helb 2010). According to the manufacturer, Xpert MTB/RIF may be used with fresh sputum samples, which may be either unprocessed sputum or processed sputum sediments. The sample reagent (sodium hydroxide and isopropanol):sample volume ratio is 2:1 for unprocessed sputum and 3:1 for sputum sediments. The manufacturer does not specifically mention the use of Xpert MTB/RIF with frozen specimens (Cepheid 2009).
The Xpert MTB/RIF limit of detection, "the lowest number of colony forming units per sample that can be reproducibly distinguished from negative samples with 95% confidence" (Cepheid 2009), is five genome copies of purified DNA per reaction or 131 colony forming units per mL in M. tuberculosis spiked sputum (Helb 2010). In comparison, identification of TB bacilli by microscopic examination requires at least 10,000 bacilli per mL of sputum (Toman 2004). Xpert MTB/RIF detects both live and dead bacteria (Miotto 2012).
Xpert MTB/RIF uses molecular beacon technology to detect rifampicin resistance. Molecular beacons are nucleic acid probes that recognize and report the presence or absence of the normal, rifampicin‐susceptible, 'wild type' sequence of the rpoB gene of TB. Five different coloured beacons are used, each covering a separate nucleic acid sequence within the amplified rpoB gene. When a beacon binds to the matching sequence, it fluoresces or 'lights up', which indicates the presence of one of the gene sequences that is characteristic of rifampicin‐susceptible TB. Failure of the beacon to bind or delayed binding to the matching sequence indicates potential rifampicin resistance. The number and timing of detection (when the fluorescent signal rises above a pre‐determined baseline cycle threshold) of positive beacons as well as results of sample processing controls allows the test to distinguish among the following results: 'No TB'; 'TB detected, rifampicin resistance detected'; 'TB detected, no rifampicin resistance detected'; and an 'invalid result' (Figure 1). A single Xpert MTB/RIF run will provide both detection of TB and detection of rifampicin resistance. One cannot deselect testing for rifampicin resistance and only run the assay for TB detection, although it is possible for the laboratory to omit results for rifampicin resistance when reporting to the healthcare provider.
Figure 1.

Readout of Xpert® MTB/RIF assay for a TB positive, rifampicin‐susceptible specimen. Courtesy: Karin Weyer, the Global TB Programme, WHO
Since Xpert MTB/RIF was released, there have been four generations (G1, G2, G3, and G4) of the test involving different software and cartridge combinations. G4 is the only Xpert MTB/RIF software and cartridge combination in current use. G4 contains modifications that improved determination of rifampicin resistance detection as previous Xpert MTB/RIF versions had found that some rifampicin susceptibility results were falsely resistant. Studies using all Xpert MTB/RIF generations are included in this updated Cochrane Review.
Clinical pathway
Patients with presumed TB or MDR‐TB would undergo testing with Xpert MTB/RIF. Xpert MTB/RIF could be performed as an initial test or as an add‐on test after prior testing with microscopy (WHO Policy Xpert MTB/RIF 2011). Following an Xpert MTB/RIF test, subsequent culture and drug susceptibility testing (DST) are recommended to monitor treatment progress and to detect resistance to drugs other than rifampicin (WHO Rapid Implementation 2011).
Settings of interest
We defined the settings of interest as intermediate‐level and peripheral‐level laboratories. The latter may be associated with primary health care facilities. We acknowledge that not all peripheral‐level laboratories will be able to satisfy the operational requirements recommended for Xpert MTB/RIF, namely an uninterrupted and stable electrical power supply, temperature control, and yearly calibration of the instrument modules. However, Xpert MTB/RIF is most likely to have an impact on patient health when it is used in a setting, such as a primary health care facility, where treatment can be started as soon as possible. The level of laboratory services is not to be confused with the setting where the patient received treatment. The Global Laboratory Initiative Roadmap presents a tiered system to describe laboratory service levels: peripheral, intermediate, and central, each level with its own set of responsibilities (Global Laboratory Initiative 2010). Although three levels are described, the Roadmap recognizes that responsibilities at a given level may vary, depending on the needs of countries and diagnostic strategies. Intermediate‐level laboratories typically perform tests such as microscopy, rapid molecular tests, culture, and DST. Peripheral‐level laboratories typically perform only smear‐microscopy and refer samples or patients in need of further tests, such as rapid molecular testing, culture, or DST, to a higher level laboratory (Global Laboratory Initiative 2010).
It should be noted that in the original Cochrane Review, we described the setting of interest as peripheral‐level laboratories based on a different classification system previously in use (WHO Policy Framework 2010).
Role of index test(s)
We were interested in the following purposes for testing:
I. Xpert MTB/RIF for TB detection
A. Xpert MTB/RIF used as an initial test replacing smear microscopy in a population unselected by smear status
B. Xpert MTB/RIF used as an add‐on test following a negative smear microscopy result
II. Xpert MTB/RIF for rifampicin resistance detection
A. Xpert MTB/RIF used as an initial test for rifampicin resistance replacing conventional phenotypic DST as the initial test
As mentioned, Xpert MTB/RIF does not eliminate the need for subsequent culture and phenotypic DST, which are required to monitor treatment progress and to detect resistance to drugs other than rifampicin.
Alternative test(s)
In this section, we describe selected alternative tests for detection of TB and rifampicin resistance. For a comprehensive review of these tests, we refer the reader to several excellent resources (Drobniewski 2012; Nahid 2012; UNITAID 2013).
Smear microscopy, which involves the direct examination of sputum smears with Ziehl‐Neelsen staining for acid‐fast bacilli (M. tuberculosis bacteria), is the most commonly used test for TB detection in resource‐limited settings (International Standards 2009). Advantages of smear microscopy include its simplicity, low cost, speed, and high specificity in high TB burden areas. In addition, smear microscopy identifies the most infectious TB patients. Smear microscopy can be performed in basic laboratories. Drawbacks of smear microscopy include the need for specialized training and its relatively low sensitivity, 50% to 60% on average for a direct smear (Steingart 2006a). Although, the sensitivity of microscopy can be improved by approximately 10% with fluorescence (Steingart 2006), a large number of TB cases still go undiagnosed. Smear‐negative TB is disproportionately higher in HIV‐positive than HIV‐negative individuals, accounting for 24% to 61% of all pulmonary cases in people living with HIV (Getahun 2007; Perkins 2007). Microscopy cannot distinguish between drug‐susceptible TB and drug‐resistant TB.
Nucleic acid amplification tests (NAATs) are molecular systems that can detect small quantities of genetic material (DNA or RNA) from microorganisms, such as M. tuberculosis. A variety of molecular amplification methods are available, of which PCR is the most common. NAATs are available as commercial kits and in‐house tests (based on a protocol developed in a non‐commercial laboratory) and are used routinely in high‐income countries for TB detection. In‐house PCR is widely used in developing countries because these tests are less expensive than commercial kits. However, in‐house PCR is known to produce highly inconsistent results (Flores 2005). The use of NAATs has recently been recommended as standard practice in the United States (CDC 2009). The main advantage of NAATs is that they can provide results several weeks earlier than culture (CDC 2009). Drawbacks are that these tests are often too expensive and complex for routine use by TB programmes in resource‐limited settings. In addition, although the specificity of NAATs is high, some NAATs have shown variable and low sensitivity, especially in sputum smear‐negative patients (Flores 2005; Greco 2006; Ling 2008a).
Alternative molecular methods for DST include the commercial line probe assays, INNO‐LiPA Rif.TB (Innogenetics, Ghent, Belgium) and GenoType® MTBDRplus assay (Hain LifeScience GmbH, Nehren, Germany). The INNO‐LiPA Rif.TB assay targets common mutations in the rpoB gene associated with rifampicin resistance, while the GenoType® MTBDRplus assay also targets the common mutations in katG and inhA genes associated with isoniazid resistance in addition to the mutations in the rpoB gene (UNITAID 2013). Advantages of line probe assays are that they can provide a result for detection of TB and drug resistance in one to two days. Also, they have both high sensitivity (greater than 97%) and high specificity (greater than 99%) for the detection of rifampicin resistance alone, or in combination with isoniazid (sensitivity greater than 90%; specificity greater than 99%), on TB isolates and smear‐positive sputum specimens (Ling 2008). Drawbacks are that line probe assays are expensive and must be used in reference laboratories (Nahid 2012). These tests have been endorsed by WHO (WHO Policy Line Probe Assays 2008).
Rationale
Xpert MTB/RIF, if accurate, would provide obvious benefits for patients (earlier diagnosis and the opportunity to begin earlier, appropriate treatment) and for public health (opportunities to interrupt TB transmission), especially in developing countries. To our knowledge, at the time of writing, one non‐Cochrane systematic review on the diagnostic accuracy of Xpert MTB/RIF has been published (Chang 2012). However, the authors performed literature searching to 1 October 2011 and used statistical methods for meta‐analysis other than the currently recommended bivariate random‐effects models (Macaskill 2010).
WHO issued initial recommendations on the use of Xpert MTB/RIF in early 2011 (WHO Policy Xpert MTB/RIF 2011). We performed this updated Cochrane Review as part of a WHO process to develop updated guidelines on the use of the test (WHO Xpert MTB/RIF Policy Update 2013).
Objectives
Primary objectives
Since Xpert MTB/RIF can detect both TB and rifampicin resistance, we had two review questions with the following primary objectives:
Xpert MTB/RIF for TB detection
To determine summary estimates of the diagnostic accuracy of Xpert MTB/RIF for the diagnosis of pulmonary TB in adults
Xpert MTB/RIF for rifampicin resistance detection
To determine summary estimates of the diagnostic accuracy of Xpert MTB/RIF for detection of rifampicin resistance in adults
Secondary objectives
Our secondary objective was to investigate heterogeneity in Xpert MTB/RIF sensitivity and specificity in relation to covariates. For TB detection, the covariates of interest were smear status; HIV status; condition of the specimens (fresh versus frozen); preparation of the specimens (unprocessed versus processed); country income status; proportion of TB cases in the study; and type of setting for running Xpert MTB/RIF (clinical or laboratory). For rifampicin resistance detection, the covariates of interest were Xpert MTB/RIF assay version and proportion of rifampicin resistant samples in the study (prevalence of rifampicin resistance in the study population).
Methods
Criteria for considering studies for this review
Types of studies
We included primary studies that assessed the diagnostic accuracy of Xpert MTB/RIF for both pulmonary TB and rifampicin resistance, or pulmonary TB alone. Diagnostic accuracy studies are typically cross‐sectional in design. However, we also searched for randomized controlled trials (RCTs) and cohort studies. We only included studies that reported data comparing Xpert MTB/RIF to an acceptable reference standard from which we could extract true positive (TP), true negative (TN), false positive (FP), and false negative (FN) values. Xpert MTB/RIF could be assessed alone or together with other tests.
We excluded studies with a case‐control design because these types of studies are prone to bias, in particular, studies enrolling patients with severe disease and healthy participants without disease. We also excluded studies reported only in abstracts.
Participants
We included studies that recruited adult or predominantly adult patients, aged 15 years or older, presumed to have pulmonary TB or MDR‐TB, with or without HIV infection. Also, we included studies that assessed the diagnostic accuracy of Xpert MTB/RIF using sputum and other respiratory specimens (such as fluid obtained from bronchial alveolar lavage and tracheal aspiration) consistent with the intended use of the manufacturer (Cepheid 2009), and studies from all types of health facilities and all laboratory levels (peripheral, intermediate, and central) from all countries. The majority of included studies provided data on the age of study participants. We considered it highly likely that studies that did not report age data involved all or mostly adults for the following reasons: the vast majority of specimens evaluated with Xpert MTB/RIF were sputum specimens and children have difficulty producing sputum; we excluded data on specimens obtained by gastric aspiration, as this specimen collection method is used mostly for investigating TB in children; we excluded studies that specifically evaluated the use of Xpert MTB/RIF in children; and we performed a sensitivity analysis by dropping studies that did not report age data to check whether the accuracy results changed Sensitivity analyses.
Index tests
Xpert MTB/RIF was the index test under evaluation.
We also compared Xpert MTB/RIF with smear microscopy, either Ziehl‐Neelsen microscopy, fluorescence microscopy, or both microscopy methods.
Target conditions
The target conditions were active pulmonary TB and rifampicin resistance.
Reference standards
For TB, acceptable reference standards used solid media (Löwenstein‐Jensen, Middlebrook 7H10 or 7H11, or Ogawa media) or a commercial liquid culture system, (such as BACTEC™ 460TB System or BACTEC™ MGIT™ 960 Mycobacterial Detection System, BD, USA; BacT/ALERT® System, bioMérieux, France; or VersaTREK® Mycobacteria Detection & Susceptibility, Thermo Fisher Scientific, USA).
For rifampicin resistance, the reference standards were phenotypic culture‐based DST methods recommended by WHO (WHO Policy DST 2008). Acceptable methods were the proportion method performed on solid media (such as Löwenstein‐Jensen, Middlebrook 7H10 or 7H11, or Ogawa media), use of a commercial liquid culture system (such as BACTEC™ 460TB System or BACTEC™ MGIT™ 960 Mycobacterial Detection System, BD, USA; BacT/ALERT® System, bioMérieux, France; or VersaTREK® Mycobacteria Detection & Susceptibility, Thermo Fisher Scientific, USA), or both.
Search methods for identification of studies
We attempted to identify all relevant studies regardless of language or publication status (published, unpublished, in press, and ongoing).
Electronic searches
Vittoria Lutje, (VL) the Information Specialist for the Cochrane Infectious Diseases Group, performed searches on three occasions, 25 September 2011, 15 December 2011, and 7 February 2013. Using the strategy described in Appendix 1, she searched the following databases: Cochrane Infectious Diseases Group Specialized Register; MEDLINE; EMBASE; ISI Web of Knowledge; MEDION; LILACS; BIOSIS; and SCOPUS. She also searched the metaRegister of Controlled Trials (mRCT) and the search portal of the WHO International Clinical Trials Registry Platform, to identify ongoing trials. We limited all searches to 2007 onward because the development of Xpert MTB/RIF was completed in 2009 and the first paper describing its clinical use was published electronically in 2009 (Helb 2010). VL performed the searches without language restriction.
Searching other resources
To identify additional published, unpublished, and ongoing studies, we performed the following tasks:
reviewed reference lists of included articles and review articles identified through the above methods;
contacted Cepheid, the test manufacturer;
handsearched WHO reports on Xpert MTB/RIF;
contacted researchers at the Foundation for Innovative New Diagnostics (FIND), members of the Stop TB Partnership's New Diagnostics Working Group, and other experts in the field of TB diagnosis.
Data collection and analysis
Selection of studies
Two review authors (KRS and DJH) independently scrutinized titles and abstracts identified from electronic literature searches to identify potentially eligible studies. We retrieved the article of any citation identified by either review author for full‐text review. KRS and DJH independently assessed articles for inclusion using predefined inclusion and exclusion criteria, and resolved any discrepancies by discussion between the review authors. We listed the excluded studies and the reasons for their exclusion.
We named studies according to the surname of the first author and year of publication. For multicentre studies, the study‐naming scheme uniquely identified multiple study centres from within each study (for example, Boehme 2010a; Boehme 2010b), each of which reported data separately for a distinct population at a given study site. Hence, the number of study centres exceeds the number of studies.
Data extraction and management
We extracted data on the following characteristics:
author, publication year, study design, case country of residence, country income status classified by the World Bank List of Economies (World Bank 2012), level of laboratory services, type of setting for running Xpert MTB/RIF (clinical or laboratory);
population, age, gender, HIV status, smear status, and follow‐up;
reference standard;
Xpert MTB/RIF assay version;
specimen collection (such as expectorated sputum, induced sputum);
condition of the specimen (fresh or frozen);
preparation of the specimen (processed or unprocessed);
QUADAS‐2 items (Whiting 2011);
data for two‐by‐two tables for Xpert MTB/RIF, including results reported as uninterpretable (results reported as indeterminate, invalid, error, or no result);
time to diagnosis (time from specimen collection until there is an available TB result in laboratory or clinic);
time to treatment initiation (time from specimen collection until time patient starts treatment).
Whenever possible, we extracted TP, FP, FN, and TN values based on one Xpert MTB/RIF result for one specimen provided by one patient. However, in some of the studies, the number of specimens (and Xpert MTB/RIF results) exceeded the number of patients, suggesting that a single patient may have provided multiple specimens. We therefore compared pooled sensitivity and specificity for TB detection in all studies with pooled sensitivity and specificity in the subset of studies that provided one Xpert MTB/RIF result based on one specimen provided by one patient (see Sensitivity analyses).
Concerning the condition of the specimen, although the manufacturer recommends use of fresh specimens, we were aware that studies had been conducted using frozen specimens so we extracted this information as well.
Concerning the definition of smear positivity, as the vast majority of included studies performed Xpert MTB/RIF in intermediate‐level or central‐level laboratories, we assumed these studies adhered to the revised definition of a new sputum smear‐positive pulmonary TB case based on the presence of at least one acid‐fast bacillus in at least one sputum sample in countries with a well‐functioning external quality assurance system (WHO Policy Smear‐positive TB Case 2007).
We developed a standardized data extraction form and piloted the form with four studies. Based upon the pilot, we finalized the form. Two review authors (KRS and DH) independently extracted data from each study using the final form. We contacted study authors for missing data and clarifications and entered all data into Microsoft® Excel. The final data extraction form is in Appendix 2.
Assessment of methodological quality
We appraised the quality of included studies with the Quality Assessment of Diagnostic Accuracy Studies (QUADAS‐2) tool (Whiting 2011). QUADAS‐2 consists of four domains: patient selection, index test, reference standard, and flow and timing. We assessed all domains for the potential for risk of bias and the first three domains for concerns regarding applicability. We used questions, called signalling questions, for each domain to form judgments about the risk of bias. As recommended, we first developed guidance on how to appraise each signalling question and interpret this information tailored to this review. Then, one review author (KRS) piloted the tool with four of the included studies. Based on experience gained from the pilot, we finalized the tool. Two review authors (KRS and DH) independently assessed the methodological quality of the included studies with the finalized tool. We presented results in the text, graphs, and a table. We did not generate a summary "quality score" because of problems associated with such numeric scores (Juni 1999; Whiting 2005). We explained definitions for using QUADAS‐2 in Appendix 3.
Statistical analysis and data synthesis
We performed descriptive analyses for the results of the included studies using Stata 12 (Stata) and presented key study characteristics in Characteristics of included studies. We used data reported in the two‐by‐two tables to calculate sensitivity and specificity estimates and 95% confidence intervals (CI) for individual studies and to generate forest plots using Review Manager 5. Whenever possible, we included NTM as non‐TB for specificity determinations. We chose to use data that were not subject to discrepant analyses (unresolved data), since resolved data after discrepant analyses are a potential for risk of bias (Hadgu 2005).
We carried out meta‐analyses to estimate the pooled sensitivity and specificity of Xpert MTB/RIF separately for TB detection (I. A. and I. B.) and rifampicin resistance detection (II. A.). When possible, we determined pooled estimates using an adaptation of the bivariate random‐effects model (Reitsma 2005) to allow for a hierarchical structure for the two multicentre studies (Boehme 2010; Boehme 2011). The bivariate random‐effects approach allowed us to calculate the pooled estimates of sensitivity and specificity while dealing with potential sources of variation caused by (1) imprecision of sensitivity and specificity estimates within individual studies; (2) correlation between sensitivity and specificity across studies; and (3) variation in sensitivity and specificity between studies. In a few cases, namely TB detection among smear‐positive individuals and rifampicin resistance detection (described below), where data were insufficient for bivariate analyses, we performed univariate analyses.
To compare the relative value of Xpert MTB/RIF and smear microscopy, we estimated the difference between their pooled sensitivities and pooled specificities. For this analysis, the specificity of smear was assumed to be 100% (Toman 2004a; Steingart 2006a). We also presented the data in a descriptive plot showing the estimates of sensitivity and specificity of Xpert MTB/RIF compared with those of smear microscopy in studies that reported on both tests.
To determine the value of Xpert MTB/RIF as a replacement test for smear (I. A.), we included studies with unselected individuals presumed to have TB to estimate pooled sensitivity and specificity. To determine the value of Xpert MTB/RIF as an add‐on test (I. B.), we estimated its sensitivity and specificity among smear‐negative individuals presumed to have TB. We did this by including individual studies that enrolled individuals preselected to be predominantly smear negative as well as studies providing results for unselected smear‐negative individuals.
For rifampicin resistance detection, we performed univariate meta‐analyses (using all available data) to determine sensitivity and specificity estimates separately. We did this because, in several studies, all patients were rifampicin susceptible (rifampicin resistance negatives), thus contributing data for specificity but not for sensitivity. We also performed a sensitivity analysis using the bivariate random‐effects model for the subset of studies that provided data for both sensitivity and specificity.
We estimated all models using a Bayesian approach with non‐subjective prior distributions and implemented using WinBUGS (Version 1.4.3) (Lunn 2000). Under the Bayesian approach, all unknown parameters must be provided a prior distribution that defines the range of possible values of the parameter and the likelihood of each of those values based on information external to the data. In order to let the observed data determine the final results, we chose to use low‐information prior distributions over the pooled sensitivity and specificity parameters and their between‐study standard deviation parameters. The model we used is summarized in the Statistical Appendix together with the WinBUGS program used to implement it (Appendix 4). Information from the prior distribution is combined with the likelihood of the observed data in accordance with Bayes theorem to obtain a posterior distribution for each unknown parameter.
Using a sample from the posterior distribution, we can obtain various descriptive statistics of interest. We estimated the median pooled sensitivity and specificity and their 95% credible intervals (CrI).The median or the 50% quantile is the value below which 50% of the posterior sample lies. We reported the median because the posterior distributions of some parameters may be skewed and the median would be considered a better point estimate of the unknown parameter than the mean in such cases. The 95% CrI is the Bayesian equivalent of the classical (frequentist) 95% CI. (We have indicated 95% CI for individual study estimates and 95% CrI for pooled study estimates as appropriate). The 95% CrI may be interpreted as an interval that has a 95% probability of capturing the true value of the unknown parameter given the observed data and the prior information.
We also estimated the 'predicted' sensitivity and specificity in a future study together with their 95% CrIs. The predicted estimate is our best guess for the estimate in a future study and is the same as the pooled estimate. The CrIs, however, may be different. These values are derived from the predicted region typically reported in a bivariate meta‐analysis plot. If there is no heterogeneity at all between studies, the CI (or CrI) around the predicted estimate will be the same as the CI around the pooled estimate. On the other hand, if there is considerable heterogeneity between studies, the CI around the predicted estimate will be much wider than the CI around the pooled estimate. We generated the plots using R (version 2.15.1) (R 2008).
Approach to uninterpretable Xpert MTB/RIF results
We excluded uninterpretable test results from the analyses for determination of sensitivity and specificity for both TB detection and rifampicin resistance detection. We used a hierarchical model for a single proportion to estimate the pooled proportion of uninterpretable Xpert MTB/RIF results.
Investigations of heterogeneity
I. Xpert MTB/RIF for TB detection
Effect of smear status and HIV status
We investigated heterogeneity by performing subgroup analyses to determine sensitivity and specificity estimates for patients classified by smear or HIV status. Within subgroups, we analyzed the data in two ways: 1) we performed descriptive analyses where we included all studies that provided relevant data and displayed these data in forest plots; and 2) we performed meta‐analyses where we included only studies that provided data for both subgroups (for example, smear‐positive and smear‐negative subgroups) within the same study. In the latter head‐to‐head comparison, we hoped to achieve a similar distribution of other patient characteristics and manner of test execution in the subgroups. For meta‐analyses, we presented pooled accuracy results in tables.
Effect of other covariates
To study the impact of additional covariates of interest, we performed meta‐regression with the following covariates: condition of the specimen (fresh versus frozen), preparation of the specimen (unprocessed versus processed), proportion of TB cases in the study (≤ 30% versus > 30%, proportion based on the median value in the included studies), and country income status (low‐ or middle‐income versus high‐income). We fit these models separately among smear‐positive and smear‐negative patients in an effort to adjust for smear status. All the aforementioned covariates were categorical, study‐level covariates. We did not consider type of setting (clinical versus laboratory) due to insufficient data.
II. Xpert MTB/RIF for rifampicin resistance detection
Effect of the Xpert MTB/RIF assay version
A major source of heterogeneity in systematic reviews of diagnostic test accuracy is the difference in values used to define a positive test between studies. In the Xpert system, the basis for rifampicin resistance detection is the difference between the first (early cycle threshold) and the last (late cycle threshold) M. tuberculosis‐specific beacon (Lawn 2011a). The original Xpert MTB/RIF system configuration reported rifampicin resistance when the difference in the cycle threshold was >3.5 cycles and rifampicin sensitive when the difference in the cycle threshold was ≤3.5 cycles (Xpert MTB/RIF G1 assay). After May 2010, the manufacturer modified the difference in the cycle threshold cut‐off to improve Xpert MTB/RIF specificity for rifampicin resistance detection. This change affected the Xpert MTB/RIF G2 and G3 assays. Another modification was implemented in late 2011 affecting the Xpert MTB/RIF G4 assay. Therefore, we explored the effect of the Xpert MTB/RIF assay version on the sensitivity and specificity estimates for rifampicin resistance detection.
Effect of proportion rifampicin resistance in the study
We also explored the influence of the proportion rifampicin‐resistant samples on the pooled sensitivity and specificity estimates by including a covariate, proportion rifampicin resistance ≤ 15 and > 15%, in the regression model.
Sensitivity analyses
We performed sensitivity analyses by limiting inclusion in the meta‐analysis to: 1) studies that provided data by age that explicitly met the age criterion for participants; 2) studies where consecutive patients were selected; 3) studies where a single specimen yielded a single Xpert MTB/RIF result for a given patient; and 4) studies that explicitly represented the use of Xpert MTB/RIF for the diagnosis of individuals thought to have TB. In order to assess the influence of two large multicentre manufacturer‐supported studies on the summary estimates, we performed an analysis excluding these studies (Boehme 2010; Boehme 2011).
Assessment of reporting bias
We chose not to carry out formal assessment of publication bias using methods such as funnel plots or regression tests because such techniques have not been helpful for diagnostic test accuracy studies (Macaskill 2010). However, Xpert MTB/RIF is produced by only one manufacturer and, as a new test for which there has been considerable attention and scrutiny, we believe reporting bias was minimal.
Other analyses
NTM
NTM, such as M. avium complex and M. intracellulare, comprise a multi‐species group of human pathogens that are ubiquitous in water and soil. NTM can cause severe pulmonary and other diseases that share clinical signs with TB but are treated differently. People infected with HIV with severe immunosuppression are particularly vulnerable to infections caused by NTM (Gopinath 2010). We summarized separately data for NTM by determining the percent of false‐positive Xpert MTB/RIF results in samples that grew NTMs (see Other analyses: NTM).
Results
Results of the search
We identified 27 unique studies, integrating nine new studies since publication of the original Cochrane Review (Steingart 2013). Of the 27 studies, two were international multicentre studies (Boehme 2010; Boehme 2011) carried out at five and six study centres, respectively. The two studies by Boehme involved different patients. We presented descriptive characteristics and the methodological quality assessment at both study and study centre levels, and meta‐analysis results at study level. One other study, conducted at three sites, reported accuracy data for the three sites combined; we considered this to be a single study and a single study centre (Marlowe 2011). Hence there were 27 studies representing 36 study centres.Figure 2 shows the flow of studies in the updated literature search. Characteristics of excluded studies lists studies excluded in this update and the original Cochrane Review.
Figure 2.

Flow diagram of studies in the review
Methodological quality of included studies
Figure 3 shows the overall risk of bias and applicability concerns for the 36 study centres. Figure 4 presents the quality assessment results for the individual study centres. In the patient selection domain, 27 study centres (75%) were at low risk of bias because the centre enrolled participants consecutively and avoided inappropriate exclusions. The remaining study centres were at (1) high risk of bias because either the manner of patient selection was by convenience (Bowles 2011; Hanif 2011; Ioannidis 2011; Malbruny 2011; Marlowe 2011; Miller 2011) or the study preselected smear‐positive patients (Friedrich 2011; Williamson 2012), or (2) unclear risk of bias because the manner of patient selection was not stated (Ciftci 2011). With regard to applicability (patient selection domain), 24 of the 36 study centres (67%), corresponding to 16 of the 27 studies (59%), were of low concern because these study centres evaluated sputum specimens and ran Xpert MTB/RIF in intermediate‐level or peripheral‐level laboratories associated with primary care clinics (Hanrahan 2013; Van Rie 2013). We judged the remaining study centres as follows: high concern, two that mainly evaluated bronchial aspirates (Al‐Ateah 2012; Marlowe 2011) and one that ran Xpert MTB/RIF as a screening test (Lawn 2011), or unclear concern, nine that ran Xpert MTB/RIF in a central‐level laboratory (Boehme 2010a; Boehme 2010d; Bowles 2011; Hanif 2011; Ioannidis 2011; Kurbatova 2013; Marlowe 2011; Rachow 2011; Teo 2011). In the index test (Xpert MTB/RIF) domain, we considered all study centres to be at low risk of bias and low concern regarding applicability. In the reference standard domain, we judged 33 study centres (92%) to be at low risk of bias for TB and 34 study centres (94%) to be at low risk of bias for rifampicin resistance because the reference standard results were interpreted without knowledge of the results of the Xpert MTB/RIF assay. Applicability was of low concern for all studies in the reference standard domain. In the flow and timing domain, 31 study centres (86%) were at low risk of bias because all patients were accounted for in the analysis and information about uninterpretable results was provided. We had nearly complete information for all study centres.
Figure 3.

Risk of bias and applicability concerns graph: review authors' judgements about each domain presented as percentages across the 36 included study centres (27 studies). The reference standard domain pertains to TB as the target condition. See text for the reference standard relating to rifampicin resistance.
Figure 4.

Risk of bias and applicability concerns summary: review authors' judgements about each domain for each included study centre.
Findings
For TB detection, the 27 studies included 9557 participants. The median number of participants in the studies was 145 (Interquartile range (IQR) 99 to 211). The proportion of TB cases in the studies ranged from 4.0% (Hanif 2011) to 100% (Friedrich 2011), median 34.4% (IQR 23.1 to 57.4).
Of the 27 studies, 24 studies (33 study centres) including 2966 participants provided data for rifampicin resistance detection. Of the three studies that were not included, one study presented combined results for pulmonary and extrapulmonary specimens (Moure 2011); one study did not report information on rifampicin resistance (Helb 2010); and one study did not use the defined reference standard (Barnard 2012). Seven studies detected no rifampicin resistance with the reference standard (Al‐Ateah 2012; Ciftci 2011; Hanif 2011; Marlowe 2011; Rachow 2011; Safianowska 2012; Van Rie 2013). The proportion of rifampicin resistant samples in the studies ranged from 0.0% to 56.6% (Kurbatova 2013), median 3.2% (IQR 0.0 to 13.2).
Characteristics of included studies presents key characteristics for the 27 studies. All 27 studies used a cross‐sectional study design for determining the diagnostic accuracy of Xpert MTB/RIF. The majority of studies evaluated expectorated sputum. Sixteen studies (59%), corresponding to 25 study centres (69%), were located in low‐income or middle‐income countries. In the countries represented by the 36 study centres, TB incidence rates per 100,000 population ranged from 3.9 (USA) to 993 (South Africa). Percent MDR‐TB among new TB cases ranged from 0% (Kuwait) to 22% (Azerbaijan) and among retreatment cases ranged from 0% (Singapore, Tanzania) to 56% (Azerbaijan) (WHO Drug Resistance 2008; WHO M/XDR‐TB 2010; Wright 2009; Zignol 2012).
I. Xpert MTB/RIF for TB detection
A. Xpert MTB/RIF used as an initial test replacing smear microscopy in a population unselected by smear status
We have presented forest plots of Xpert MTB/RIF sensitivity and specificity for TB detection for the 27 studies (36 study centres) in Figure 5. Sensitivity estimates ranged from 58% to 100% and specificity estimates ranged from 86% to 100%.
Figure 5.

Forest plots of Xpert MTB/RIF sensitivity and specificity for TB detection, Xpert MTB/RIF used as an initial test replacing smear microscopy. The individual studies are ordered by decreasing sensitivity. TP = True Positive; FP = False Positive; FN = False Negative; TN = True Negative. Between brackets are the 95% CI of sensitivity and specificity. The figure shows the estimated sensitivity and specificity of the study (blue square) and its 95% CI (black horizontal line). Xpert MTB/RIF specificity could not be estimated in one study.
We included 22 of the total 27 studies (8998 participants) in this meta‐analysis. We excluded five studies that enrolled primarily only smear‐positive or smear‐negative patients (Friedrich 2011; Ioannidis 2011; Moure 2011; Van Rie 2013; Williamson 2012). For TB detection, Xpert MTB/RIF pooled sensitivity and specificity were 89% (95% CrI 85% to 92%) and 99% (95% CrI 98% to 99%), respectively (Table 5). The predicted sensitivity and specificity of Xpert MTB/RIF for TB detection were 89% (95% CrI 63% to 97%) and 99% (95% CrI 90% to 100%), respectively. In relation to the pooled values, the wider 95% CrIs around the predicted values suggested some variability between studies, particularly in sensitivity (Table 5). Figure 6 presents the pooled and predicted sensitivity and specificity estimates together with the credible and prediction regions for Xpert MTB/RIF for TB detection. The summary point appears close to the upper left‐hand corner of the plot, suggesting high accuracy of Xpert MTB/RIF for TB detection. The 95% credible region around the summary (pooled) value of sensitivity and specificity, the region that contains likely combinations of the pooled sensitivity and specificity, is relatively narrow. The 95% prediction region is wider, displaying more uncertainty as to where the likely values of sensitivity and specificity might occur in a future study.
Table 1.
Xpert MTB/RIF assay for detection of TB and rifampicin resistance
| Type of analysis (Number of studies, participants) |
Median pooledsensitivity (95% credible interval) |
Median pooledspecificity (95% credible interval) |
Median predicted sensitivity (95% credible interval) |
Median predicted specificity (95% credible interval) |
| Xpert MTB/RIF used as an initial test for TB detection replacing microscopy (22, 8998) | 89% (85, 92) | 99% (98, 99) | 89% (63, 97) | 99% (90, 100) |
| Xpert MTB/RIF used as an add‐on test for TB detection following a negative smear microscopy result (21, 6950) | 67% (60, 74) | 99% (98, 99) | 67% (42, 85) | 99% (89, 100) |
| Xpert MTB/RIF used as an initial test for rifampicin resistance detection replacing conventional DST as the initial test * | 95% (90, 97) | 98% (97, 99) | 95% (80, 99) | 98% (94, 100) |
* For rifampicin resistance detection, pooled sensitivity and specificity estimates were determined separately by univariate analyses. Pooled sensitivity, number of studies = 17 (555 participants); pooled specificity, number of studies = 24 (2411 participants).
Figure 6.
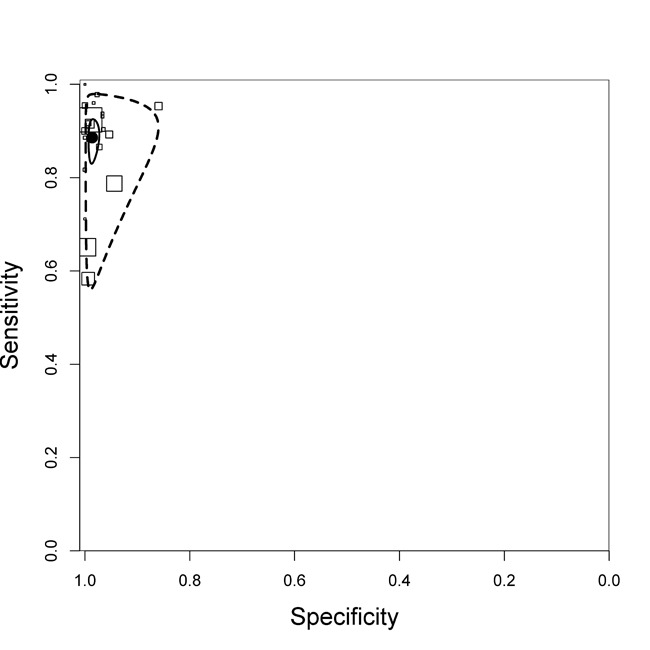
Summary plots of Xpert MTB/RIF sensitivity and specificity for TB detection, Xpert MTB/RIF used as an initial test replacing smear microscopy. Each individual study is represented by an empty square. The size of the square is proportional to the sample size of the study such that larger studies are represented by larger squares. The filled circle is the median pooled estimate for sensitivity and specificity. The solid curves represent the 95% credible region around the summary estimate; the dashed curves represent the 95% prediction region.
TB detection, Xpert MTB/RIF compared with smear microscopy
Twenty‐one studies (8880 participants) provided data from which to compare the sensitivity of Xpert MTB/RIF and smear microscopy. Figure 7 displays results of smear microscopy versus Xpert MTB/RIF for the individual studies. In the meta‐analysis, the sensitivity estimate for Xpert MTB/RIF was the same as the estimate in the meta‐analysis in I. A., the difference in the number of studies and participants being due to use of the subset of studies that also reported results by smear status. For smear microscopy, the pooled sensitivity was 65% (95% CrI 57% to 72%). For Xpert MTB/RIF, the pooled sensitivity was 88% (95% CrI 84% to 92%). Therefore, in comparison with smear microscopy, Xpert MTB/RIF increased TB detection among culture‐confirmed cases by 23% (95% CrI 15% to 32%).
Figure 7.

Study results of smear microscopy (green circle) versus Xpert MTB/RIF (red circle) plotted in ROC space. The specificity of smear was assumed to be 100%.
B. Xpert MTB/RIF used as an add‐on test following a negative smear microscopy result
Three studies performed microscopy and, for those patients found to be smear‐negative, subsequently ran Xpert MTB/RIF (Ioannidis 2011; Moure 2011; Van Rie 2013). Two of these studies were laboratory‐based assessments performed in high‐income countries (Ioannidis 2011; Moure 2011). One study performed Xpert MTB/RIF at a primary care clinic in a low‐income country (Van Rie 2013). For the three studies, sensitivities ranged from 64% to 83% and specificities from 94% to 100% (Figure 8).
Figure 8.

Forest plots of Xpert MTB/RIF for TB detection in studies reporting data for smear‐negative patients. We also used these data as a proxy for the accuracy of Xpert MTB/RIF used as an add‐on test following a negative smear microscopy result. TP = True Positive; FP = False Positive; FN = False Negative; TN = True Negative. Between brackets the 95% CI of sensitivity and specificity. The figure shows the estimated sensitivity and specificity of the study (blue square) and its 95% CI (black horizontal line).
In the meta‐analysis, the pooled sensitivity was 67% (95% CrI 60% to 74%) and the pooled specificity was 99% (95% CrI 98% to 99%; 21 studies, 6950 participants; Table 5). Therefore, 67% of smear‐negative culture‐confirmed TB cases were detected using Xpert MTB/RIF following smear microscopy, increasing case detection by 67% (95% CrI, 60% to 74%) in this group.
Figure 9 presents the pooled and predicted sensitivity and specificity estimates together with the credible and prediction regions for this analysis. The summary point is relatively far from the upper left‐hand corner of the plot, suggesting lower accuracy of Xpert MTB/RIF when used as an add‐on test than as a replacement test. The 95% credible region around the summary value of sensitivity and specificity is relatively wide.
Figure 9.

Summary plots of Xpert MTB/RIF sensitivity and specificity for TB detection, Xpert MTB/RIF used as an add‐on test following a negative smear microscopy result. Each individual study is represented by an empty square. The size of the square is proportional to the sample size of the study such that larger studies are represented by larger squares. The filled circle is the median pooled estimate for sensitivity and specificity. The solid curve represents the 95% credible region around the summary estimate; the dashed curves represent the 95% prediction region.
Uninterpretable results
Of the total 27 studies, seven studies (Al‐Ateah 2012; Hanif 2011; Hanrahan 2013; Miller 2011; Moure 2011; Williamson 2012; Zeka 2011) reported zero uninterpretable results and four studies (Bowles 2011; Ciftci 2011; Helb 2010; Rachow 2011) did not provide information about uninterpretable results. Of 11,408 tests performed, the pooled proportion of uninterpretable test results was very low (1.0%, 95% CrI 0.05% to 2.0%).
Investigations of heterogeneity, TB detection
It is possible that the accuracy of Xpert MTB/RIF in clinical subgroups of patients could differ causing heterogeneity in Xpert MTB/RIF performance. We therefore determined sensitivity and specificity estimates for patients grouped by smear or HIV status.
TB detection in smear‐positive and smear‐negative individuals presumed to have TB
Smear‐positive TB
Figure 10 displays the forest plots for studies reporting data for smear‐positive patients (24 studies, 33 study centres, 2020 participants). There was little heterogeneity in the sensitivity estimates (range 95% to 100%). In the meta‐analysis, the pooled sensitivity for smear‐positive, culture‐positive TB was 98% (95% CrI 97% to 99%; 21 studies, 1936 participants; Table 6). We did not include Van Rie 2013 in the meta‐analysis as this study preselected smear‐negative patients, though it did report a sensitivity estimate for Xpert MTB/RIF of 75% among four smear‐positive patients. We did not estimate Xpert MTB/RIF pooled specificity in the studies in the smear‐positive subgroup because almost all participants were considered to be true TB positive.
Table 2.
Impact of covariates on heterogeneity of Xpert MTB/RIF sensitivity and specificity, TB detection
| Covariate (Number of studies) |
Median pooled sensitivity (95% credible interval) |
Median pooled specificity (95% credible interval) |
|
| Smear status | |||
| Smear + (21) | 98% (97, 99) | *** | |
| Smear ‐ (21) | 67% (60, 74) | 99% (98, 99) | |
| Difference (Smear+ minus Smear‐) | 31% (24, 38) | *** | |
| P (Smear+ > Smear‐) | 1.00 | *** | |
| HIV status | |||
| HIV‐ (7) | 86% (76, 92) | 99% (98, 100) | |
| HIV+ (7) | 79% (70, 86) | 98% (96, 99) | |
| Difference (HIV‐ minus HIV+) | 7% (‐5, 18) | 1% (‐1, 3) | |
| P (HIV‐ > HIV+) | 0.90 | 0.85 | |
| Covariate (number of studies) | Within smear positive | Within smear negative | |
|
Median pooled sensitivity (95% credible interval) |
Median pooled sensitivity (95% credible interval) |
Median pooled specificity (95% credible interval) |
|
| HIV status | |||
| HIV‐ | *** | *** | *** |
| HIV+ (4) | 97% (90, 99)** | 61% (40, 81)** | 99% (97, 100)# |
| Difference (HIV‐ minus HIV+) | *** | *** | *** |
| P (HIV‐ > HIV+) | *** | *** | *** |
| Condition of specimen | |||
| Fresh (12) | 99% (98, 100) | 67% (58, 76) | 99% (98, 100) |
| Frozen (6) | 97% (95, 99) | 61% (48, 73) | 98% (95, 99) |
| Difference (Fresh minus Frozen) | 1% (‐0.4, 4) | 6% (‐9, 22) | 1% (‐0.4, 4) |
| P (Fresh > Frozen) | 0.92 | 0.79 | 0.92 |
| Specimen preparation | |||
| Unprocessed (10) | 98% (97, 99) | 69% (60, 78) | 98% (97, 99) |
| Processed (11) | 99% (97, 99) | 64% (54, 75) | 99% (98, 100) |
| Difference (Unprocessed minus Processed) | ‐0.1% (‐2, 2) | 5% (‐9, 18) | ‐1% (‐2, 1) |
| P (Unprocessed > Processed) | 0.45 | 0.76 | 0.20 |
| Proportion TB cases in the study | |||
| > 30% (12)* | 99% (97, 99) | 70% (62, 78) | 98% (96, 99) |
| ≤ 30% (9)* | 98% (96, 99) | 61% (50, 73) | 99% (98, 100) |
| Difference (> 30% minus ≤ 30%) | 0.5% (‐1, 2) | 9% (‐5, 22) | ‐1% (‐3, 0.2) |
| P (> 30% minus ≤ 30%) | 0.74 | 0.90 | 0.05 |
| Country income level | |||
| High‐income (8) | 99% (98, 100) | 73% (62, 83) | 99% (97, 100) |
| Low‐income and middle‐income (13) | 98% (97, 99) | 64% (56, 73) | 99% (97, 99) |
| Difference (High‐income minus Low‐ and middle‐income) | 1% (‐1, 2) | 9% (‐5, 22) | 0.3% (‐1, 2) |
| P (High‐income > Low‐ and middle‐income) | 0.88 | 0.90 | 0.69 |
* We selected 30% as a cut‐off based on the median proportion of TB cases in the included studies
**Results are from a univariate analysis
***Values could not be determined
#Results are from a univariate analysis based on three studies
Figure 10.

Forest plot of Xpert MTB/RIF sensitivity for TB detection in studies reporting data for smear‐positive patients. The squares represent the sensitivity and specificity of one study, the black line its CI. TP = true positive; FP = false positive; FN = false negative; TN = true negative. Xpert MTB/RIF specificity could not be estimated in these studies.
Smear‐negative TB
Figure 8 displays the forest plots for studies reporting data for smear‐negative patients (24 studies, 33 study centres, 7264 participants). There was considerable variability in sensitivity estimates (range 43% to 100%). Specificity estimates showed less variability (range 86% to 100%). Lawn 2011 yielded the lowest sensitivity. This study used Xpert MTB/RIF as a TB screening test in HIV‐infected patients with advanced immunodeficiency enrolling in antiretroviral therapy services. The meta‐analysis included 21 studies. The pooled sensitivity estimate for smear‐negative, culture‐positive TB was 67% (95% CrI 60% to 74%), considerably lower than the pooled sensitivity estimate for smear‐positive, culture‐positive TB which was 98% (95% CrI 97% to 99%; Table 6).
TB detection in HIV‐negative and HIV‐positive individuals presumed to have TB
Figure 11 displays the forest plots for studies reporting data for HIV‐negative individuals (nine studies, 18 study centres, 2555 participants) and HIV‐positive individuals (10 studies, 16 study centres, 2474 participants). Sensitivity was variable in both the HIV‐negative subgroup (56% to 100%) and HIV‐positive subgroup (0% to 100%). The small number of participants in several studies may have contributed to some of this variability. Specificity varied less than sensitivity in both subgroups: 96% to 100% in the HIV‐negative subgroup and 92% to 100% in the HIV‐positive subgroup.
Figure 11.

Forest plots of Xpert MTB/RIF sensitivity and specificity for TB detection in HIV‐positive and HIV‐negative subgroups. The squares represent the sensitivity and specificity of one study and the black line represent its CI. TP = true positive; FP = false positive; FN = false negative; TN = true negative.
The meta‐analysis included seven studies that provided data for both HIV‐negative (1470 participants) and HIV‐positive (1789 participants) individuals (Boehme 2010; Boehme 2011; Hanrahan 2013; Rachow 2011; Scott 2011; Theron 2011; Van Rie 2013). The pooled sensitivity was 86% (95% CrI 76% to 92%) in the HIV‐negative subgroup and 79% (95% CrI 70% to 86%) in the HIV‐positive subgroup (Table 6). Corresponding pooled specificities were similar: 99% (95% CrI 98% to 100%) and 98% (95% CrI 96% to 99%), respectively (Table 6). When adjusting for the percentage of smear‐positive patients in each study, the impact of HIV decreased suggesting that some of the differences between the HIV‐positive and HIV‐negative subgroups could be attributed to differences in smear status (Table 6).
TB detection among HIV‐positive individuals by smear status
Four studies reported data from which to assess the accuracy of Xpert MTB/RIF in HIV‐positive individuals by smear status (Balcells 2012; Carriquiry 2012; Lawn 2011). Among people with HIV, Xpert MTB/RIF pooled sensitivity was 61% (95% CrI 40% to 81%) for smear‐negative, culture‐positive TB compared with 97% (95% CrI 90% to 99%) for smear‐positive, culture‐positive TB, a statistically significant result (Table 6). Hence, among people with HIV‐TB coinfection, people with HIV infection and smear‐positive disease were more likely to be diagnosed with TB using Xpert MTB/RIF than those with HIV infection and smear‐negative disease.
Effect of condition of the specimen
As mentioned above, although the manufacturer recommends use of fresh specimens, we were aware that studies had been conducted using frozen specimens; therefore we explored the effect of the condition of the specimen on Xpert MTB/RIF performance. The pooled sensitivities and specificities were slightly higher for fresh specimens compared with frozen specimens within both the smear‐positive and smear‐negative groups, however there was considerable overlap in the CrIs around these estimates (Table 6).
Effect of specimen preparation
The pooled sensitivity was higher for unprocessed specimens compared with processed specimens in smear‐negative patients, though there was considerable overlap in the CrIs around these estimates (Table 6).
Effect of the proportion of culture‐confirmed TB cases in the study
For this analysis, we used a cutoff of 30% TB cases because 30% was around the median proportion of TB cases in the included studies. Within smear‐negatives, the pooled sensitivity was higher for studies with a higher proportion of TB cases; however there was considerable overlap in the CrIs around these estimates (Table 6).
Effect of country income status
There did not appear to be an important difference in the pooled sensitivity of Xpert MTB/RIF according to country income status after adjusting for smear status (Table 6).
II. Rifampicin resistance detection
A. Xpert MTB/RIF used as an initial test replacing conventional DST
The 24 studies (33 study centres) in this analysis included 555 rifampicin‐resistant specimens, median two specimens (range 1 to 250). Two studies accounted for the majority (82%, 455/555) of the rifampicin‐resistant specimens (Boehme 2010; Boehme 2011). Seven studies contributed only specificity data (presence of rifampicin‐susceptible TB) (Al‐Ateah 2012; Ciftci 2011; Hanif 2011; Marlowe 2011; Rachow 2011; Safianowska 2012; Van Rie 2013) but not sensitivity data (presence of rifampicin‐resistant TB). Figure 12 shows the forest plots of sensitivity and specificity for this analysis. The individual study centres in the plots are ordered by decreasing sensitivity and decreasing number of true positive results. Although, there was heterogeneity in sensitivity estimates (ranging from 33% to 100%), in general there was less variability among study centres with a higher number of rifampicin‐resistant specimens. Specificity showed less variability than sensitivity, ranging from 83% to 100%. The pooled sensitivity and specificity by univariate analysis were 95% (95% CrI 90% to 97%) and 98% (95% CrI 97% to 99%), respectively (Table 5).
Figure 12.

Forest plots of Xpert MTB/RIF sensitivity and specificity for detection of rifampicin resistance, Xpert used as an initial test replacing conventional DST as the initial test. The individual studies are ordered by decreasing sensitivity and decreasing number of true positives. The squares represent the sensitivity and specificity of one study, the black line its CI. TP = true positive; FP = false positive; FN = false negative; TN = true negative.
Investigations of heterogeneity, rifampicin resistance detection
Effect of the Xpert MTB/RIF assay version
A major source of heterogeneity in systematic reviews of diagnostic test accuracy is the difference in values used to define a positive test between studies. The basis for rifampicin resistance detection in the Xpert MTB/RIF system is the difference between the first (early cycle threshold) and the last (late cycle threshold) M. tuberculosis‐specific beacon (Lawn 2011a). This difference is referred to as the delta cycle threshold. The original Xpert MTB/RIF system configuration reported rifampicin resistance when the delta cycle threshold was > 3.5 cycles and rifampicin sensitive when the delta cycle threshold was ≤ 3.5 cycles (Xpert MTB/RIF G1 assay). After May 2010, the manufacturer modified the delta cycle threshold cut‐off to improve Xpert MTB/RIF specificity for rifampicin resistance detection (Xpert MTB/RIF G2 and G3 assays). Another modification was implemented in late 2011 (Xpert MTB/RIF G4 assay). G4 is now the only cartridge available for use.
Therefore, we explored the effect of the Xpert MTB/RIF version on the sensitivity and specificity estimates for rifampicin resistance detection. The pooled sensitivity was 93% (95% CrI 87% to 97%) for studies using Xpert MTB/RIF G2, G3, or G4 assays (13 studies) and 97% (95% CrI 91% to 99%) for studies using Xpert MTB/RIF G1 assay (four studies) (Table 7). The corresponding pooled specificities were 98% (95% CrI 96% to 99%; 16 studies) and 99% (95% CrI 98% to 100%; seven studies) (Table 7). Thus, when G1 alone was compared with a set containing the later Xpert MTB/RIF versions, there was considerable overlap between the accuracy estimates for the different Xpert MTB/RIF versions and no apparent difference between them.
Table 3.
Impact of covariates on heterogeneity of Xpert MTB/RIF sensitivity and specificity, rifampicin resistance detection
| Covariate |
Median pooled sensitivity (95% credible interval) |
Median pooled specificity (95% credible interval) |
| Xpert MTB/RIF assay version* | ||
| G2, G3, G4 | 93% (87, 97) | 98% (96, 99) |
| G1 | 97% (91, 99) | 99% (98, 100) |
| Difference (G2, G3, G4 minus G1) | ‐4% (‐10, 3) | ‐1% (‐3, ‐0.2) |
| P (G2, G3, G4 > G1) | 0.09 | 0.01 |
| Proportion rifampicin resistance in the study** | ||
| > 15% | 96% (91, 98) | 97% (94, 99) |
| ≤ 15% | 91% (79, 97) | 99% (98, 99) |
| Difference (> 15% minus ≤ 15%) | 4% (‐3, 16) | ‐2% (‐4, 0.1) |
| P (> 15% greater than ≤ 15%) | 0.87 | 0.03 |
Pooled sensitivity and specificity estimates were determined separately by univariate analyses.
*Xpert MTB/RIF assay version G2, G3, G4: pooled sensitivity (13 studies) and pooled specificity (16 studies); Xpert MTB/RIF assay version G1: pooled sensitivity (four studies) and pooled specificity (seven studies).
** Proportion rifampicin resistance > 15%: pooled sensitivity (six studies) and pooled specificity (six studies); proportion rifampicin resistance ≤ 15%: pooled sensitivity (11 studies) and pooled specificity (18 studies).
Xpert MTB/RIF G4 assay effect on specificity
One study used Xpert MTB/RIF G4 assay and provided data for specificity determinations (Kurbatova 2013). Kurbatova 2013 found a specificity of 95% (42/44; 95% CI 85% to 99%) (Figure 12).
Effect of proportion rifampicin resistant samples in the study
For this analysis, we used a cutoff of 15% for the proportion of rifampicin resistant samples in the study. The pooled sensitivity was 96% (95% CrI 91% to 98%) for studies with proportion rifampicin resistance > 15%, higher than the pooled sensitivity of 91% (95% CrI 79% to 97%) for studies with proportion rifampicin resistance ≤ 15%. The corresponding pooled specificities were 97% (95% CrI 94% to 99%) and 99% (95% CrI 98% to 99%) (Table 7). The differences in Xpert MTB/RIF sensitivity and specificity were not significantly different from 0 (Table 7).
Sensitivity analyses
For TB detection, we undertook sensitivity analyses by limiting inclusion in the meta‐analysis to: 1) studies that provided age data that met our inclusion criterion for adults; 2) studies that used consecutive sampling; 3) studies where a single specimen yielded a single Xpert MTB/RIF result for a given individual; and 4) studies that explicitly tested individuals with presumed TB. We also performed a sensitivity analysis by excluding from the meta‐analysis the two large multicentre studies (Boehme 2010; Boehme 2011). These sensitivity analyses made no difference to any of the findings (Table 8).
Table 4.
Sensitivity analyses
| Type of analysis (Number of studies, participants) |
Median pooled sensitivity (95% credible interval) |
Median pooled specificity (95% credible interval) |
Median predicted sensitivity (95% credible interval) |
Median predicted specificity (95% credible interval) |
| TB detection, without Boehme 2010 and Boehme 2011 (20, 3748) | 88% (83, 92) | 98% (97, 99) | 88% (62, 97) | 98% (89, 100) |
| TB detection, studies that provided data by age that met the criterion for adults (14, 7880) | 87% (81, 92) | 99% (98, 99) | 87% (58, 97) | 99% (95, 100) |
| TB detection, studies where consecutive patients were selected (17, 8465) | 87% (82, 91) | 99% (98, 99) | 87% (59, 97) | 99% (90, 100) |
| TB detection, studies where a single specimen yielded a single Xpert MTB/RIF result for a given patient (14, 7912) | 85% (79, 91) | 99% (98, 99) | 85% (54, 97) | 99% (95, 100) |
| TB detection, studies that clearly represented the use of the test for diagnosis of patients with presumed TB (16, 7974) | 89% (85, 93) | 99% (97, 99) | 89% (69, 97) | 99% (89, 100) |
| Rifampicin resistance detection by bivariate analyses (17, 2621) | 95% (90, 97) | 98% (97, 99) | 95% (80, 99) | 98% (93, 100) |
Other analyses
NTM
Fourteen studies (2626 participants) provided data on a variety of NTM that grew from the specimens tested to look for evidence of cross‐reactivity: one NTM (Al‐Ateah 2012); four NTM (Barnard 2012); six NTM (Bowles 2011); one NTM (Ioannidis 2011); one NTM (Kurbatova 2013); 41 NTM (Marlowe 2011); 20 NTM (Moure 2011); 45 NTM (Rachow 2011); seven NTM (Safianowska 2012); five NTM (Scott 2011); 13 NTM (Teo 2011); eight NTM (Theron 2011); three NTM (Van Rie 2013); and 22 NTM (Williamson 2012). Among these 14 studies comprising 180 NTM, Xpert MTB/RIF was positive in only one (0.6%) specimen that grew NTM (Rachow 2011).
Discussion
This updated Cochrane Review on the diagnostic accuracy of Xpert MTB/RIF for TB detection and rifampicin resistance detection in adults summarizes the current literature and integrates nine new studies (33% of the included papers) identified since the original Cochrane Review (Steingart 2013). The findings in this update are consistent with those reported previously.
In adults presumed to have TB, with or without HIV infection, we found Xpert MTB/RIF to be sensitive and specific for TB detection. Xpert MTB/RIF may also be valuable as an add‐on test following smear microscopy (sensitivity 67%). When used as an initial test replacing phenotypic culture‐based DST, we found Xpert MTB/RIF provides accurate results for rifampicin resistance detection.
Sensitivity and specificity depend on the performance of a test in a particular situation, defined by the population, the setting, and prior testing. In a different population or setting or with a different testing strategy, the sensitivity and specificity are likely to change (Bossuyt 2008). Overall, we had low concern about applicability. Of note, a recent RCT found that Xpert MTB/RIF run by nurses at point‐of‐care in primary care clinics obtained similar sensitivity 83.3% (95% CI 77.2% to 88.0%) to that achieved when Xpert MTB/RIF was performed by laboratory technicians in central level laboratories 83.2% (95% CI 79.0% to 86.8%) (Theron 2013).
We found Xpert MTB/RIF sensitivity for TB detection to be higher in fresh specimens than in frozen specimens. Although we did not find conclusive evidence, one possible explanation for this observation is that investigators may have used an increased amount of buffer volume to resuspend frozen sputum specimens causing a dilution effect (Scott 2011). We also found that, in comparison with processed specimens, unprocessed specimens had slightly higher sensitivity in smear‐negative patients. In addition, we found higher pooled accuracy of Xpert MTB/RIF in studies performed in high‐income countries than in low‐ or middle‐income countries. However, after adjustment for smear status, the strength of these associations decreased. Therefore, there was no conclusive evidence supporting the impact of either specimen preparation or country income on Xpert MTB/RIF sensitivity for TB detection.
We included only two studies that used Xpert MTB/RIF G4, the version in current use. We excluded one study of Xpert MTB/RIF G4 (based on concern about the use of duplicate data and discrepant analyses); however, we feel the findings from this study for rifampicin resistance detection are worth mentioning. The Foundation for Innovative Diagnostics (FIND) evaluated the performance of Xpert MTB/RIF G4 using archived sputum specimens from individuals with presumed TB from Germany, Peru, Azerbaijan, Uganda, Cape Town, and South Africa (FIND 2011). Conventional DST for rifampicin resistance was mainly performed by the Lowenstein‐Jensen proportion method or MGIT. Genetic sequencing was performed on samples with discordant Xpert MTB/RIF/conventional DST results and these results were used for determination of sensitivity and specificity. The overall sensitivity (rifampicin resistant) was 98.9% (87/88) (95% CI 93.8% to 99.8%) and the overall specificity (rifampicin sensitive) was 99.8% (433/434) (95% CI 98.7% to 100.0%). For Xpert MTB/RIF rifampicin‐sensitive/DST‐resistant discordants, sequencing of the rpoB region was performed in four cases and discordant results resolved in three of these cases; for Xpert MTB/RIF rifampicin‐resistant/DST‐sensitive discordants, sequencing of the rpoB region was performed in nine cases and discordant results resolved in eight of these cases (FIND 2011). In light of these findings, we would expect G4 to have comparable or increased accuracy for rifampicin resistance detection compared with earlier Xpert MTB/RIF versions.
We acknowledge that patient outcomes are clearly important to patients, decision makers, and the wider TB community. Outcomes in addition to diagnostic accuracy, however, could not be systematically addressed, as they would have required a different methodology. Nonetheless, we looked for and summarized two 'time to event' outcomes (time to result and time to treatment initiation) when data were provided in the included studies (Table 9). Xpert MTB/RIF results for TB detection were usually reported within two hours or on the same day, compared with liquid culture results reported in around 16 to 20 days. Studies reporting on time to detection of rifampicin resistance found that, compared with conventional methods, Xpert MTB/RIF greatly decreased the time to diagnosis. However, early detection of rifampicin resistance may not lead to improved patient outcomes if the result is not linked to appropriate treatment, services, and supervision (WHO Xpert MTB/RIF Checklists 2011). Two studies provided information about time to treatment initiation. In Boehme 2011, for smear‐negative, culture‐positive TB, the median delay in beginning treatment was 56 days (IQR 39 to 81) before Xpert MTB/RIF was introduced, compared with five days (IQR 2 to 8) after Xpert MTB/RIF was introduced. In Van Rie 2013, for smear‐negative, culture‐positive TB patients with Xpert MTB/RIF positive results, treatment was begun on the same day compared with 13 days for patients diagnosed by other methods. Data regarding delays in switching from the standard regimen for drug‐susceptible TB to an appropriate regimen for MDR‐TB would also be useful because of the potential harms to patients being treated with the wrong drug regimen. We are aware of several recently completed RCTs in South Africa, Brazil, and India that will give insights on user acceptability, operational performance, and patient‐important outcomes (Durovni 2013; Raizada 2013; Theron 2013). These and other studies on patient outcomes will need to be systematically reviewed.
Table 5.
Selected patient‐important outcomes as reported in the included studies
| Study | Time to TB detection | Time to rifampicin resistance detection | Time to treatment initiation |
| Balcells 2012 | Median (range) Xpert MTB/RIF: 0 days Liquid culture 10 days (5 to 22 days) 8 days for smear‐positive, 15 days for smear‐negative cases |
||
| Boehme 2011 | Median (IQR) Xpert MTB/RIF: 0 days (0, 1) Smear: 1 day (0, 1) Solid culture: 30 days (23, 43) Liquid culture: 16 days (13, 21) |
Median (IQR) Xpert MTB/RIF: 1 day (0, 1) Line probe assay (direct testing): 20 days (10, 16) Phenotypic DST: 106 days (30, 124) |
Median (IQR) Smear‐, culture+ TB Before Xpert MTB/RIF introduced: 56 days (39, 81) After Xpert MTB/RIF introduced: 5 days (2, 8) |
| Helb 2010 | Xpert MTB/RIF (1 sample): 1 hour 55 minutes Xpert MTB/RIF (8 samples processed together): 2 hours |
||
| Lawn 2011 | Median* (IQR) Xpert MTB/RIF: 4 days (3, 6) Smear: 3 days (2, 5) Liquid culture (smear+): 12 days (10,14) Liquid culture (smear‐): 20 days (17, 27) |
Xpert MTB/RIF: mean 2 days MTBDRplus assay (with positive culture isolate): mean 21 days Phenotypic DST (liquid culture): mean 40 days |
|
| Marlowe 2011 | Xpert MTB/RIF: hands‐on time was 5 minutes; run time was less than 2 hours | ||
| Miller 2011 | Xpert MTB/RIF: hands‐on time was 15 minutes: run time was 113 minutes | ||
| Moure 2011 | Xpert MTB/RIF: total time of 2 hours | ||
| Rachow 2011 | Xpert MTB/RIF: within two hours | ||
| Van Rie 2013 | Xpert MTB/RIF: results were available the same day | Xpert MTB/RIF positive patients: 0 days (0,0) Patients diagnosed by other methods: 13 days (10, 20) |
|
| Zeka 2011** | Xpert MTB/RIF (routine practice): 3 to 24 hours Liquid culture: 19 days mean (range 3 to 42 days) |
*Median delay between sputum collection and results being available to the clinic.
**Times provided for both pulmonary and extrapulmonary specimens jointly.
Abbreviations: DST, drug susceptibility testing; IQR, interquartile range.
A recent analysis of cost and affordability found that, globally, the use of Xpert MTB/RIF to diagnose MDR‐TB would cost less (US$70 to 90 million per year) than what it would cost to use a combination of conventional diagnostics (US$123 to 191 million per year). Conventional diagnostics may include smear microscopy, chest radiography, culture, and culture‐based DST following WHO‐recommended algorithms. In addition, using Xpert MTB/RIF to diagnose TB in people living with HIV would also cost less than conventional diagnostics, both globally and in the vast majority of high TB burden countries. However, for almost all countries, the deployment of Xpert MTB/RIF to diagnose TB in all individuals with signs and symptoms of TB would cost more than the use of conventional diagnostics (which may include smear microscopy and follow‐up chest radiography for those with smear‐negative results) (Pantoja 2013). Several additional cost‐effectiveness studies have been published (Abimbola 2012; Andrews 2012; Dowdy 2011; Meyer‐Rath 2012; Schnippel 2012; Vassall 2011).
Xpert MTB/RIF has now begun to be rolled out in over 20 countries via UNITAID, with a price drop from $16.86 to $9.98 (US) per cartridge, a price that will remain in effect until 2022 (The Gates Foundation 2012; UNITAID 2012). UNITAID is a global health initiative working to increase access for tests and medicines for HIV/AIDS, TB, and malaria. Since WHO endorsed the use of Xpert MTB/RIF, country‐level policy makers have been making decisions about adoption and scale‐up. The uptake has been much faster than for any other TB technology recommended by WHO over the last 10 years.
This review represents the most comprehensive review on the diagnostic accuracy of Xpert MTB/RIF and provides evidence that may help countries make decisions about scaling up Xpert MTB/RIF for programmatic management of TB and drug‐resistant TB. Although the information in this review will help to inform, other factors such as level of deployment in the health system, cost, and operational considerations (including the ability to maintain an uninterrupted and stable electrical power supply, temperature control, and maintenance of the cartridge modules) will also influence those decisions, as discussed in recent publications (Trébucq 2011; Denkinger 2013).
Summary of main results
We have summarized the main results in the Summary of Findings tables (Table 1;Table 2; and Table 3).
When used as an initial test replacing smear microscopy, Xpert MTB/RIF detected 89% of TB cases with high specificity (99%).
When used as an add‐on test following smear microscopy, Xpert MTB/RIF detected 67% of TB cases with high specificity (99%).
Xpert MTB/RIF sensitivity for smear‐positive, culture‐positive TB was 98%.
In comparison with smear microscopy, Xpert MTB/RIF increased TB detection among culture‐confirmed cases by 23%.
Xpert MTB/RIF detected 79% of pulmonary TB cases in people infected with HIV and 86% of pulmonary TB cases in people without HIV. However, after adjustment for smear status, there was no evidence of a difference between the HIV‐positive and HIV‐negative subgroups.
When used as an initial test replacing phenotypic culture‐based DST, Xpert MTB/RIF detected 95% of rifampicin‐resistant TB cases with a specificity of 98%.
The pooled proportion of Xpert MTB/RIF uninterpretable results was very low.
Application of the meta‐analysis to a hypothetical cohort
The Summary of findings tables summarize the findings of the review by applying the results to a hypothetical cohort of 1000 individuals thought to have TB or MDR‐TB. We present several different scenarios: for Xpert MTB/RIF used as an initial test for TB detection or as an add‐on test following microscopy, the prevalence of TB in the setting or patient subgroup varies from 2.5% to 5% to 10%; and for Xpert MTB/RIF for rifampicin resistance detection, the prevalence of rifampicin resistance in the setting varies from 5% to 15% (5% is estimated to be equivalent to the upper limit for rifampicin resistance prevalence in new cases; 15% is estimated to be the lower limit for rifampicin resistance prevalence among previously treated cases). The consequences of false positive results are likely patient anxiety, morbidity from additional testing and unnecessary treatment, and possible delay in further diagnostic evaluation. The consequences of false negative results are an increased risk of patient morbidity and mortality, and continued risk of community transmission of TB.
I. Xpert MTB/RIF for TB detection
A. Xpert MTB/RIF used as an initial test replacing smear microscopy in a population unselected by smear status
TB prevalence of 2.5%: if the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 patients thought to have TB, where 25 patients actually do have TB, then Xpert MTB/RIF would be expected to miss three cases and falsely diagnose 10 cases (Table 1).
TB prevalence of 5%: if the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 patients thought to have TB, where 50 patients actually do have TB, then Xpert MTB/RIF would be expected to miss six cases and falsely diagnose 10 cases (Table 1).
TB prevalence of 10%; if the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 patients thought to have TB, where 100 patients actually do have TB, then Xpert MTB/RIF would be expected to miss 11 cases and falsely diagnose nine cases (Table 1).
If the pooled estimates for Xpert MTB/RIF and smear microscopy are applied to a hypothetical cohort of 1000 patients where 10% of those presenting with symptoms have TB, Xpert MTB/RIF will diagnose 88 cases and miss 12 cases, whereas sputum microscopy will diagnose 65 cases and miss 35 cases (Table 2)
B. Xpert MTB/RIF used as an add‐on test following a negative smear microscopy result
TB prevalence of 2.5%; if the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 patients thought to have TB, where 25 patients actually do have TB, then Xpert MTB/RIF would be expected to miss eight cases and falsely diagnose 10 cases (Table 1).
TB prevalence of 5%: if the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 patients thought to have TB, where 50 patients actually do have TB, then Xpert MTB/RIF would be expected to miss 17 cases and falsely diagnose 10 cases (Table 1).
TB prevalence of 10%: if the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 patients thought to have TB, where 100 patients actually do have TB, then Xpert MTB/RIF would be expected to miss 33 cases and falsely diagnose nine cases (Table 1).
II. Xpert MTB/RIF for rifampicin resistance detection
A. Xpert MTB/RIF used as an initial test for rifampicin resistance replacing conventional DST as the initial test
If the pooled estimates for Xpert MTB/RIF are applied to a hypothetical cohort of 1000 individuals where 15% of those presenting with symptoms are rifampicin resistant, Xpert MTB/RIF would correctly identify 143 individuals as rifampicin resistant and miss eight cases and 833 individuals as rifampicin susceptible and wrongly identify 17 individuals as resistant. In comparison, where 5% of those presenting with symptoms are rifampicin resistant, Xpert MTB/RIF would correctly identify 48 individuals as rifampicin resistant and miss three cases and correctly identify 931 individuals as rifampicin susceptible and wrongly identify 19 individuals as resistant (Table 3).
Strengths and weaknesses of the review
The findings in this review are based on comprehensive searching, strict inclusion criteria, and standardized data extraction. The strength of our review is that it enables an assessment of the diagnostic accuracy of Xpert MTB/RIF for detection of TB when Xpert MTB/RIF is used as a replacement test for smear microscopy or as an add‐on test following smear microscopy. In addition, the review allows a determination of the accuracy of Xpert MTB/RIF for detection of rifampicin resistance when Xpert MTB/RIF is used as an initial test replacing conventional DST.
Completeness of evidence
This data set involved comprehensive searching and correspondence with experts in the field and the test manufacturer to identify additional studies, as well as repeated correspondence with study authors to obtain additional data and information that was missing from the papers. The search strategy included studies published in all languages. However, we acknowledge that we may have missed some studies despite the comprehensive search. Lastly, the evidence in this review is mostly derived from high TB incidence countries and should be carefully extrapolated to low incidence settings.
Accuracy of the reference standards used
Culture is regarded as the best available reference standard for active TB disease and was the reference standard for TB in this review. Phenotypic culture‐based DST methods using WHO‐recommended critical concentrations were the reference standards for rifampicin resistance (WHO Policy DST 2008). Concerning the latter, several studies have raised concerns about rapid DST methods, in particular automated MGIT 960, for rifampicin using the recommended critical concentrations. Van Deun 2009 reported that BACTEC 460 and MGIT 960 missed certain strains associated with low‐level rifampicin resistance. Furthermore, using Xpert MTB/RIF and gene sequencing, Williamson 2012a identified four patients (three with clinical information available) whose TB isolates contained mutations to the rpoB gene but appeared to be rifampicin susceptible using MGIT 960 (36). In this study, 2/49 (4.1%) patients whose isolates did not have apparent rpoB gene mutations, experienced treatment failure compared with 3/3 (100%) patients whose isolates did have rpoB gene mutations and were deemed rifampicin susceptible with phenotypic methods (Williamson 2012a). Recently, in a study involving retreatment patients, Van Deun and colleagues found that disputed rpoB mutations conferring low‐grade resistance were often missed by rapid phenotypic DST, particularly with the MGIT 960 system, but to a minor extent also by conventional slow DST. The authors suggested this may be the reason for the perceived insufficient specificity of molecular DST for rifampicin (Van Deun 2013). In light of these findings, it is unclear whether and to what extent Xpert MTB/RIF might out‐perform phenotypic DST methods for rifampicin resistance. Specifically, the determination of the specificity of a molecular DST method based on phenotypic DST alone may underestimate the specificity of a molecular DST.
Quality and quality of reporting of the included studies
The majority of studies used consecutive selection of participants and interpreted the reference standard results without knowledge of Xpert MTB/RIF results. Xpert MTB/RIF results are generated automatically, without requiring subjective interpretation. In general, studies were fairly well reported, though we corresponded with almost all authors for additional data and missing information. We encourage authors of future studies to follow the recommendations in the STARD statement to improve the quality of reporting (Bossuyt 2003).
Completeness and relevance of the review
We noted that most studies performed Xpert MTB/RIF in intermediate‐level or peripheral‐level laboratories, which are settings that matched the review question. This review included studies using all four generations of Xpert MTB/RIF (G1, G2, G3, G4 cartridges). G4, which is used with software version 4.0 or higher, is now included in all Xpert MTB/RIF kits. This review did not address the use of Xpert MTB/RIF in children or in non‐respiratory specimens for the diagnosis of extrapulmonary TB.
Applicability of findings to the review question
We found Xpert MTB/RIF to be sensitive and specific for TB detection and rifampicin resistance detection with relatively few false‐positive and false‐negative results. The consequences of false‐positive results are likely patient anxiety, morbidity from additional and unnecessary testing and, particularly in the case of second‐line anti‐TB drugs, costly treatment and possible delay in further diagnostic evaluation. The consequences of false‐negative results are an increased risk of patient morbidity and mortality, and continued risk of community transmission of TB. The majority of studies evaluated the accuracy of Xpert MTB/RIF in sputum specimens submitted by patients thought to have TB, and conducted the test outside of central laboratories. Although the patient characteristics and settings matched our review question, as studies were carried out under research conditions, it is possible that the accuracy of Xpert MTB/RIF may be lower in routine practice settings. In studies assessing the impact of Xpert MTB/RIF on patient outcomes, Xpert MTB/RIF results for TB detection were reported more rapidly than liquid culture results, and Xpert MTB/RIF results for rifampicin resistance detection were reported much faster than culture‐based methods.
Authors' conclusions
In adults thought to have TB, with or without HIV infection, Xpert MTB/RIF is sensitive and specific. In comparison with smear microscopy, this test substantially increases TB detection among culture‐confirmed cases. Xpert MTB/RIF has higher sensitivity for TB detection in smear‐positive patients than smear‐negative patients. Nonetheless, it may also be valuable as an add‐on test following smear microscopy in patients who have previously been found to be smear‐negative. For detection of rifampicin resistance, in adults thought to have MDR‐TB, Xpert MTB/RIF provides accurate results and can allow rapid initiation of MDR‐TB treatment, pending results from conventional culture and DST. The ongoing use of Xpert MTB/RIF in TB programmes in high TB burden settings, as well as its use in primary care clinics where the test provides the opportunity to begin treatment promptly, will contribute evidence on whether its use leads to improvements in patient health.
Future studies should assess the diagnostic accuracy of Xpert MTB/RIF in peripheral‐level laboratories and clinical settings, such as primary health facilities, TB screening centres, and antiretroviral clinics, especially settings where the test is performed at the point of care. Systematic reviews have been performed on Xpert MTB/RIF for the diagnosis of extrapulmonary TB and paediatric TB and the findings described in the updated WHO policy statement on the use of Xpert MTB/RIF (WHO Xpert MTB/RIF Policy Update 2013). Future systematic reviews should summarize the growing body of evidence on patient and public health, cost, and cost effectiveness.
Feedback
Boyles, 7 October 2014
Summary
Name: Tom Boyles
Affiliation: University of Cape Town
I certify that I have no affiliations with or involvement in any organization or entity with a financial interest in the subject matter of my feedback.
In the initial version of Steingart et al's systematic review of the Xpert® MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults (Steingart 2013) includes 15 studies where Xpert was used as an initial test replacing smear microscopy, with the majority of patients being drawn from two major studies (Boehme 2010a, Boehme 2011a). My comment relates to the appropriate reference standard for tuberculosis is these studies. The systematic review appraised the quality of included studies with the Quality Assessment of Diagnostic Accuracy Studies (QUADAS‐2) (Whiting 2011) tool which states that estimates of test accuracy are based on the assumption that the reference standard is 100% sensitive and that specific disagreements between the reference standard and index test result from incorrect classification by the index test.
For each of the studies in question the reference standard for tuberculosis is listed as “Löwenstein‐Jensen culture and MGIT 960” and the review considers that the reference standard is likely to correctly classify the target condition. There is considered to be low risk of bias or applicability concerns relating to the reference test.
However, in Boehme et al 2010 there were 105 patients with ‘clinical tuberculosis’ who were excluded from the analysis. These patients were negative by the reference standard of Löwenstein‐Jensen culture and MGIT 960 and should have been included in the ‘no tuberculosis’ group. In Boehme et al 2011 there were 153 similar patients who were excluded from the analysis.
Neither paper gives justification for the exclusion of these patients who according to QUADAS‐2 were negative by the reference standard and should be included in the ‘no tuberculosis’ group. Ideally the systematic review should be amended to include these patients but if the data is unavailable the risk of bias should be acknowledged.
Note from the Editors: In addition to the above feedback, Boyles et al. published a case study in The International Journal of Tuberculosis and Lung Disease which outlined the above arguments, and illustrates this with a case study (Boyles 2014); which the Cochrane authors respond to, in the same journal (see below).
Reply
The review authors thank Boyles et al. for this comment. They raise important points about the selective exclusion of culture negative clinical TB cases in the Boehme studies.
We considered the published case study (Boyles 2014) in detail, and in response we carried out additional analyses to determine whether the Boehme studies unduly influenced the overall findings of this Cochrane review. One way we did this was by repeating the meta‐analysis with studies for which we could extract data for all enrolled participants, including patients classified as ‘clinical tuberculosis’ with negative sputum culture. We considered these participants as not having TB. In the new analysis, we found pooled sensitivity and specificity estimates to be similar to those we previously reported.
We published our findings as a response to Boyles et al. in The International Journal of Tuberculosis and Lung Disease (Steingart 2015).
Acknowledgements
The editorial base of the Cochrane Infectious Diseases Group (CIDG) is funded by UKaid from the UK Government for the benefit of developing countries. We thank all authors of the included studies for answering our questions and providing additional data. We are grateful to Vittoria Lutje, Information Retrieval Specialist with the CIDG, for help with the search strategy. We thank Hojoon Sohn and Lorie Kloda who contributed to the original review version. Also, we acknowledge Edward Desmond, California State Department of Health, for his comments on alternative TB tests, and Ellen Jo Baron, Cepheid, for her comments on the index test. In addition, we thank Chris Gilpin for assisting with the laboratory classification scheme and Matteo Zignol for providing the prevalence rates for MDR‐TB, both experts are with the Global TB Programme, World Health Organization, Geneva; and Selcan Alptekin, Rothamsted Research, for translation assistance.
Appendices
Appendix 1. Detailed search strategies
Search strategy: Medline (OVID) and Embase (OVID)
1. (tuberculosis or TB).tw
limit 1 to yr="2007 ‐Current"
2. Mycobacterium tuberculosis/
limit 2 to yr="2007 ‐Current"
3. Tuberculosis, Multidrug‐Resistant/ or Tuberculosis/ or Tuberculosis, Pulmonary/
limit 3 to yr="2007 ‐Current"
4. 1 or 2 or 3
5. (Xpert or GeneXpert or cepheid or( near* patient)). tw.
limit 4 to yr="2007 ‐Current"
4 and 5
Search strategy: Web of Knowledge (SCI‐expanded, SSCI, Conference Proceedings science, BIOSIS previews)
(tuberculosis OR TB OR mycobacterium) (topic) AND (Xpert OR Genexpert OR cepheid) (topic)
Search strategy: LILACS
(tuberculosis OR TB OR mycobacterium) (Words) AND (xpert OR Genexpert OR Cepheid) (Words)
Search strategy: SCOPUS
(tuberculosis OR TB OR mycobacterium) (title, abstract, keywords) AND (xpert OR Genexpert OR Cepheid) (title, abstract, keywords)
Appendix 2. Data extraction form
| ID | |
| ID substudy (for study centres: a, b, c, etc) | |
| First author | |
| Corresponding author and email address | |
| Was author contacted? | 1 – Yes 2 – No If yes, dates(s) |
| Title | |
| Year (of publication) | |
| Year (study start date) | |
| Language | 1 – English 2 – Other If other, specify: |
| For TB detection, what reference standard(s) was used? | 1 – Solid culture (specify 1a) 2 – Liquid culture (specify 2a) 3 – Both solid and liquid culture (specify 1a and 2a) 9 – Unknown/not reported 1a ‐ Solid culture LJ 7H10 7H11 Other 2a – Liquid culture MGIT 960 Bactec 460 Other |
|
For rifampicin resistance detection, what reference standard(s) was used? |
1 – Solid culture (specify 1a) 2 – Liquid culture (specify 2a) 3 – Both solid and liquid culture (specify 1a and 2a) 9 – Unknown/not reported 1a ‐ Solid culture LJ 7H10 7H11 Other Specify method, for example, proportion Critical concentration for RIF per WHO? 2a – Liquid culture MGIT 960 Bactec 460 Other |
| Clinical setting; describe as written in the paper | 1 – Outpatient 2 ‐ Inpatient 3 – Both out‐ and in‐patient 4 – Other, specify 5 – Laboratory 9 – Unknown/not reported Describe as in paper: |
|
Laboratory services level State name of laboratory |
1 ‐ Central 2 ‐ Intermediate 3 ‐ Peripheral 4 ‐ Other, specify |
| Was Xpert run outside a laboratory, for example, clinic? Describe | 1 ‐ Yes 2 ‐ No |
| Indicate the purpose of testing as described in the study | |
| Country where study was conducted | |
| Country World Bank Classification | 1 – Middle/low 2 – High 3 – Both middle/low and high |
| Study design | 1 – RCT 2 – Cross‐sectional 3 – Cohort 4 – Other, specify 9 – Unknown/not reported If other, specify: |
| Participant selection | 1 – Consecutive 2 – Random 3 – Convenience 7 – Other 9 – Unknown/not reported |
| Direction of study data collection | 1 – Prospective 2 – Retrospective 9 – Unknown/not reported |
| Number after screening by inclusion and exclusion criteria | _____ 9 – Unknown/not reported |
| Number included in analysis (# screened ‐ # withdrawals) | _____ 9 – Unknown/not reported |
| Unit of analysis | 1 – One specimen per patient 2 – Multiple specimens per patient 3 ‐ Unknown number of specimens per patient 9 – Unknown/not reported Describe as in paper, if unclear: |
| Prior testing by microscopy | 1 – Yes 2 – No 9 – Unknown/not reported |
|
Was the index test result interpreted without knowledge of the result of the reference standard result? |
1 ‐ Yes |
|
TB detection: Was the reference standard result interpreted without knowledge of the index test result of the result? |
1 – Yes 2 – No 9 – Unknown/not reported |
|
Rifampicin resistance detection: Was the reference standard result interpreted without knowledge of the index test result of the result? |
1 – Yes 2 – No 9 – Unknown/not reported |
| Comments about study design | |
| Patient characteristics and setting | |
| Age (range, mean (SD), median (IQR) | |
| % female | |
| Did the study include patients with previous TB history? | 1 – Yes 2 – No 9 – Unknown/not reported |
| If so, what is the percentage? | % Specify numerator/denominator |
| HIV status of participants | 0 – HIV ‐ 1 – HIV + 2 – Both HIV+/‐ 9 – Unknown/not reported |
| If HIV‐positive participants included, what is the percentage? | % Specify numerator/denominator |
|
Type of specimen (may include expectorated, induced, bronchial alveolar lavage (BAL), tracheal aspirates) |
1 – All expectorated 2 – All induced 3 – All BAL 4 – Multiple types 5 – Other 9 – Unknown/not reported If 4 or 5, describe types and record numbers: |
| Were Xpert sample and culture obtained from same specimen? | 1 – Yes 2 – No 9 – Unknown/not reported |
| Number of cultures used to exclude TB | 1 – One 2 – Two 3 – Three 4 – Four 5 – Other, specify 9 – Unknown/not reported Specify, if > 4: NOTES: |
| Pre‐treatment processing procedure for Xpert | 1 – None 2 – NALC‐NaOH 3 – NaOH (Petroff) 4 – Other 9 – Unknown/not reported |
| Was microscopy used | 1 – Yes 2 – No 9 – Unknown/not reported |
| Type of microscopy used | 1 – Ziehl‐Neelsen 2 – FM 9 – Unknown/not reported |
| Smear type | 1 – Direct 2 – Concentrated (processed) 9 – Unknown/not reported |
| Minimum number of sputum specimens used to determine smear positivity | 1 – One 2 – Two 3 – Three 4 – > 3 9 – Unknown/not reported |
| How was a positive smear defined? (if guideline referenced, look up guideline) | ≥____bacilli per____ high power fields 9 – Unknown/not reported * Complete both fields |
| For Xpert specimen, what was the condition of the specimen when tested? | 1 – Fresh 2 – Frozen 9 – Unknown/not reported |
| If fresh, specify: | 1 – Tested after storage at room temperature or if refrigerated within 48 hours of collection 2 – Tested after storage at room temperature or if refrigerated > 48 hours after collection 9 – Unknown/not reported |
| If frozen, specify: | 1 – Tested after frozen < 1 year of storage 2 – Tested frozen ≥ 1 year of storage 9 – Unknown/not reported |
| Version of software for test interpretation | 1 ‐ Version 1 2 ‐ Version 2 3 ‐ Version 3 4 ‐ Version 4 9 – Unknown/not reported |
|
Enter percentage contaminated cultures, if provided: Number contaminated culture results/ Total number cultures performed |
_____________ 9 – Unknown/not reported |
| Were uninterpretable results reported for Xpert for TB detection? | 1 – Yes 2 – No 9 – Unknown/not reported |
| Were uninterpretable results reported for Xpert for rifampicin resistance detection? | 1 – Yes 2 – No 9 – Unknown/not reported |
| Were patient important outcomes evaluated? | 1 – Yes 2 – No 9 – Unknown/not reported |
| Time to diagnosis | Xpert: Culture: 9 – Unknown/not reported |
| Time to treatment initiation | Xpert: Culture: 9 – Unknown/not reported |
| Other patient outcomes | Specify: |
| Number NTM/Number of specimens tested; provide Xpert results |
TABLES
| TB detection, all studies | Confirmed TB | |||
| Yes | No | Total | ||
| Xpert result | Positive | |||
| Negative | ||||
| Total | ||||
| Uninterpretable | ||||
| TB detection, smear positive | Confirmed TB | |||
| Yes | No | Total | ||
| Xpert result | Positive | |||
| Negative | ||||
| Total | ||||
| Uninterpretable | ||||
| TB detection, smear negative | Confirmed TB | |||
| Yes | No | Total | ||
| Xpert result | Positive | |||
| Negative | ||||
| Total | ||||
| Uninterpretable | ||||
| TB detection, HIV‐positive | Confirmed TB | |||
| Yes | No | Total | ||
| Xpert result | Positive | |||
| Negative | ||||
| Total | ||||
| Uninterpretable | ||||
| TB detection, HIV‐negative | Confirmed TB | |||
| Yes | No | Total | ||
| Xpert result | Positive | |||
| Negative | ||||
| Total | ||||
| Uninterpretable | ||||
| RIF resistance detection | Confirmed rifampicin resistance | |||
| Yes | No | Total | ||
| Xpert result | Yes (resistant) | |||
| No (susceptible) | ||||
| Total | ||||
| Uninterpretable | ||||
|
Smear microscopy |
Confirmed TB | |||
| Yes | No | Total | ||
| Smear result | Positive | |||
| Negative | ||||
| Total | ||||
Appendix 3. Rules for QUADAS‐2
Domain 1: Patient selection
Risk of bias: Could the selection of patients have introduced bias?
Signalling question 1: Was a consecutive or random sample of patients enrolled? We scored 'yes' if the study enrolled a consecutive or random sample of eligible patients; 'no' if the study selected patients by convenience; and 'unclear' if the study did not report the manner of patient selection or we could not tell.
Signalling question 2: Was a case‐control design avoided? Studies using a case‐control design were not included in the review because this study design, especially when used to compare results in severely ill patients with those in relatively healthy individuals, may lead to overestimation of accuracy in diagnostic studies. We scored 'yes' for all studies.
Signalling question 3: Did the study avoid inappropriate exclusions? We scored 'yes' if the study included both smear‐positive and smear‐negative individuals or only smear‐negative individuals; 'no' if the study included only smear‐positive individuals; and 'unclear' if we could not tell.
Applicability: Are there concerns that the included patients and setting do not match the review question?
We were interested in how Xpert MTB/RIF performed in patients presumed to have pulmonary TB or MDR‐TB whose specimens (predominantly sputum specimens) were evaluated as they would be in routine practice, in intermediate‐level laboratories or primary health care facilities. We judged 'unclear' concern if Xpert MTB/RIF was run in a central‐level laboratory. We assumed a central‐level laboratory used highly trained staff. However, we acknowledge, that for some studies, the reason Xpert MTB/RIF was performed in the central‐level laboratory was the requirement for a sophisticated laboratory infrastructure to perform culture (the reference standard) not to perform Xpert MTB/RIF. We judged 'high concern' if the majority of specimens were respiratory specimens other than sputum.
Domain 2: Index test
Risk of bias: Could the conduct or interpretation of the index test have introduced bias?
Signallingquestion 1: Were the index test results interpreted without knowledge of the results of the reference standard? We answered this question 'yes' for all studies because Xpert test results were automatically generated and the user was provided with printable test results. Thus, there is no room for subjective interpretation of test results.
Signallingquestion 2: If a threshold was used, was it prespecified? The threshold was prespecified in all versions of Xpert. We answered this question 'yes' for all studies.
For risk of bias, we scored 'low concern' for all studies.
Applicability: Are there concerns that the index test, its conduct, or its interpretation differ from the review question? Variations in test technology, execution, or interpretation may affect estimates of the diagnostic accuracy of a test. However, we judged these issues to be of ‘low concern’ for all studies in this review.
Domain 3: Reference standard
Risk of bias: Could the reference standard, its conduct, or its interpretation have introduced bias?
We considered this domain separately for the reference standard for TB detection and the reference standard for rifampicin resistance.
Signallingquestion 1: Is the reference standard likely to correctly classify the target condition? For pulmonary TB: although culture is not 100% accurate, it is considered to be the gold standard for TB diagnosis. For rifampicin resistance: similarly, although DST by conventional phenotypic methods is not 100% accurate, it is considered to be the gold standard. We answered this question ‘yes’ for all studies.
Signallingquestion 2: (TB) Were the reference standard results interpreted without knowledge of the results of the index test? We scored 'yes' if the reference test provided an automated result (for example, MGIT 960), blinding was explicitly stated, or it was clear that the reference standard was performed at a separate laboratory and/or performed by different people. We scored 'no' if the study stated that the reference standard result was interpreted with knowledge of the Xpert test result. We scored 'unclear' if we could not tell.
Signallingquestion 3: (Rifampicin resistance) We added a signalling question for rifampicin resistance because judgments might differ for TB and for rifampicin resistance, the two target conditions. Were the reference standard results interpreted without knowledge of the results of the index test? We scored 'yes' if the reference test provided an automated result (for example, MGIT 960), blinding was explicitly stated, or it was clear that the reference standard was performed at a separate laboratory and/or performed by different people. We scored 'no' if the study stated that the reference standard result was interpreted with knowledge of the Xpert test result. We scored 'unclear' if we could not tell.
Applicability: Are there concerns that the target condition as defined by the reference standard does not match the question? We judged applicability to be of 'low concern' for all studies for both pulmonary TB and rifampicin resistance.
Domain 4: Flow and timing
Risk of bias: Could the patient flow have introduced bias?
Signallingquestion 1: Was there an appropriate interval between the index test and reference standard? In the majority of included studies, we expected specimens for Xpert and culture to be obtained at the same time when patients were presumed to have TB. However, even if there were a delay of several days or weeks between index test and reference standard, TB is a chronic disease and we considered misclassification of disease status to be unlikely. We answered this question 'yes' for all studies.
Signallingquestion 2: Did all patients receive the same reference standard? We answered this question 'yes' for all studies as an acceptable reference standard (either solid or liquid culture) was specified as a criterion for inclusion in the review. However, we acknowledge that it is possible that some specimens could undergo solid culture and others liquid culture. This could potentially result in variations in accuracy, but we thought the variation would be minimal.
Signallingquestion 3: Were all patients included in the analysis? We determined the answer to this question by comparing the number of patients enrolled with the number of patients included in the two‐by‐two tables.
Appendix 4. Statistical appendix
Bayesian bivariate hierarchical model
The Bayesian bivariate hierarchical model used for the meta‐analyses is summarized below. The hierarchical framework took into account heterogeneity between studies and also between centres within two of the largest studies. The model was derived as an extension of previously described models (Chu 2009; Reitsma 2005). A WinBUGS program to fit this model is provided below. Three independent, dispersed sets of starting values were used to run separate chains. The Gelman‐Rubin statistic within the WinBUGS program was used to assess convergence. No convergence problems were observed. The first 3000 iterations were treated as burn‐in iterations and dropped. Summary statistics were obtained based on a total of 15,000 iterations resulting from the three separate chains.
Notation: From the jth centre in the ith study we extracted the cross‐tabulation between the index and reference tests TPij, FPij, TNij, FNij. The sensitivity in ijth study is denoted by Sij and the specificity by SPij. We denote the Binomial probability distribution with sample size N and probability p as Binomial(p,N), the Bivariate Normal probability distribution with mean vector μ and variance‐covariance matrix Σ as BVN(μ, Σ), the univariate Normal distribution with mean m and variance s by N(m, s) and the Uniform probability distribution between a and b by Uniform(a,b).
Likelihood Figure 13:
Figure 13.

Bayesian bivariate hierarchical model, likelihood.
The pooled sensitivity is given by 1/1+exp (‐μ1) and pooled specificity as 1/1+exp (μ2).
Prior distributions Figure 14:
Figure 14.

Bayesian bivariate hierarchical model, prior distributions.
Meta‐regression models:
To examine the impact of a dichotomous covariate (Z) on the pooled sensitivity and specificity parameters, we expressed the logit(sensitivity) and logit(specificity) as linear functions of Z as follows:
μ1 = a1+ b1Z and μ2 = a2+ b2Z
Prior distributions were placed over the coefficients in the linear function: a1 and a2˜ N(0,4) and b1 and b2˜ N(0,1.39) (Buzoianu 2008).
‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐
# WinBUGS PROGRAM FOR ESTIMATING A BIVARIATE HIERARCHICAL META‐ANALYSIS MODEL
# FOR SENSITIVITY AND SPECIFICITY ALLOWING FOR HETEROGENEITY BETWEEN STUDIES
# AND HETEROGENEITY BETWEEN CENTRES WITHIN TWO OF THE STUDIES (BOEHME 2010 and 2011)
model {
############################# BOEHME 2010 #############################
for(j in 1:5) {
logit(se.q[j])<‐q1[j,1]
logit(sp.q[j])<‐q1[j,2]
q1[j,1:2]˜ dmnorm(l[1,1:2], T1[1:2,1:2])
pos1[j]<‐TP1[j]+FN1[j]
neg1[j]<‐TN1[j]+FP1[j]
TP1[j] ˜ dbin(se.q[j],pos1[j])
FP1[j] ˜ dbin(sp.q[j],neg1[j])
}
T1[1:2,1:2]<‐inverse(SIGMA1[1:2,1:2])
# Between‐centre variance‐covariance matrix for Boehme 2010
SIGMA1[1,1] <‐ sigma1[1]*sigma1[1]
SIGMA1[2,2] <‐ sigma1[2]*sigma1[2]
SIGMA1[1,2] <‐ k1*sigma1[1]*sigma1[2]
SIGMA1[2,1] <‐ k1*sigma1[1]*sigma1[2]
prec1[1] ˜ dgamma(2,0.5)
prec1[2] ˜ dgamma(2,0.5)
k1 ˜ dunif(‐1,1)
sigma1[1]<‐pow(prec1[1],‐0.5)
sigma1[2]<‐pow(prec1[2],‐0.5)
# Overall sens/spec across centres in Boehme 2010
se[1]<‐1/(1+exp(‐l[1,1]))
sp[1]<‐1/(1+exp(l[1,2]))
l[1,1:2] ˜ dmnorm(mu[1:2], T[1:2,1:2])
############################# BOEHME 2011 #############################
for(j in 1:6) {
logit(se.r[j])<‐ r1[j,1]
logit(sp.r[j])<‐ r1[j,2]
r1[j,1:2]˜ dmnorm(l[2,1:2], T2[1:2,1:2])
pos2[j]<‐TP2[j]+FN2[j]
neg2[j]<‐TN2[j]+FP2[j]
TP2[j] ˜ dbin(se.r[j],pos2[j])
FP2[j] ˜ dbin(sp.r[j],neg2[j])
}
T2[1:2,1:2]<‐inverse(SIGMA2[1:2,1:2])
# Between‐centre variance‐covariance matrix for Boehme 2011
SIGMA2[1,1] <‐ sigma2[1]*sigma2[1]
SIGMA2[2,2] <‐ sigma2[2]*sigma2[2]
SIGMA2[1,2] <‐ k2*sigma2[1]*sigma2[2]
SIGMA2[2,1] <‐ k2*sigma2[1]*sigma2[2]
prec2[1] ˜ dgamma(2,0.5)
prec2[2] ˜ dgamma(2,0.5)
k2 ˜ dunif(‐1,1)
sigma2[1]<‐pow(prec2[1],‐0.5)
sigma2[2]<‐pow(prec2[2],‐0.5)
# Overall sens/spec across centres in Boheme 2011
se[2]<‐1/(1+exp(‐l[2,1]))
sp[2]<‐1/(1+exp(l[2,2]))
l[2,1:2] ˜ dmnorm(mu[1:2], T[1:2,1:2])
############################# SINGLE CENTRE STUDIES #############################
for(i in 3:22) {
logit(se[i]) <‐ l[i,1]
logit(sp[i]) <‐ l[i,2]
pos[i]<‐TP[i]+FN[i]
neg[i]<‐TN[i]+FP[i]
TP[i] ˜ dbin(se[i],pos[i])
FP[i] ˜ dbin(sp[i],neg[i])
l[i,1:2] ˜ dmnorm(mu[1:2], T[1:2,1:2])
}
############################# HYPER PRIOR DISTRIBUTIONS #############################
mu[1] ˜ dnorm(0,0.25)
mu[2] ˜ dnorm(0,0.25)
T[1:2,1:2]<‐inverse(TAU[1:2,1:2])
# Between‐study variance‐covariance matrix
TAU[1,1] <‐ tau[1]*tau[1]
TAU[2,2] <‐ tau[2]*tau[2]
TAU[1,2] <‐ rho*tau[1]*tau[2]
TAU[2,1] <‐ rho*tau[1]*tau[2]
tau[1]<‐pow(prec[1],‐0.5)
tau[2]<‐pow(prec[2],‐0.5)
# prec is the between‐study precision in the logit(sensitivity) and logit(specificity)
prec[1] ˜ dgamma(2,0.5)
prec[2] ˜ dgamma(2,0.5)
rho ˜ dunif(‐1,1)
# Pooled sensitivity and specificity
Pooled_S<‐1/(1+exp(‐mu[1]))
Pooled_C<‐1/(1+exp(mu[2]))
# Predicted sensitivity and specificity in a new study
l.new[1:2] ˜ dmnorm(mu[],T[,])
sens.new <‐ 1/(1+exp(‐l.new[1]))
spec.new <‐ 1/(1+exp(l.new[2]))
}
############################## DATA #####################################
# DATA WAS READ FROM THREE SEPARATE FILES
# DATA 1 ‐ BOEHME 2010
TP1[] FP1[] FN1[] TN1[]
123 1 24 68
201 0 8 101
136 1 10 185
36 3 7 215
179 0 8 35
END
#row 1 : Azerbaijan
#row 2 : Peru
#row 3 : South Africa, Cape Town
#row 4 : South Africa, Durban
#row 5 : India
############################################################################
# DATA 2 ‐ FROM BOEHME 2011
TP2[] FP2[] FN2[] TN2[]
203 4 26 303
171 3 6 825
201 2 32 669
121 0 24 144
101 16 0 671
136 5 12 234
END
#Boheme 2011
#row 1 : Azerbaijan
#row 2 : Peru
#row 3 : South Africa
#row 4 : Uganda
#row 5 : India
#row 6 : The Philippines
############################################################################
# DATA 3 ‐ FROM BOEHME 2011
TP[] FP[] FN[] TN[]
NA NA NA NA
NA NA NA NA
42 0 2 128
11 1 1 147
37 0 15 16
60 1 4 29
44 2 1 84
24 1 1 59
54 0 6 146
41 2 22 479
67 0 15 25
102 17 5 104
42 2 30 320
12 0 0 46
116 4 14 82
27 2 2 58
61 1 8 102
15 1 2 127
58 3 9 107
56 2 6 55
111 19 30 320
31 0 4 68
END
#row 1 : Boheme 2010
#row 2 : Boheme 2011
#row 3 : Al‐Ateah 2012
#row 4 : Balcells 2012
#row 5 : Barnard 2012
#row 6 : Bowles 2011
#row 7 : Carriquiry 2012
#row 8 : Cifci 2011
#row 9 : Hanif 2011
#row 10 : Hanrahan 2013
#row 11 : Helb 2010
#row 12 : Kurbatova 2012
#row 13 : Lawn 2011
#row 14 : Malbruny 2011
#row 15 : Marlowe 2011
#row 16 : Miller 2011
#row 17 : Rachow 2011
#row 18 : Safianowska 2012
#row 19 : Scott 2011
#row 20 : Teo 2011
#row 21 : Theron 2011
#row 22 : Zeka 2011
############################################################################
Data
Presented below are all the data for all of the tests entered into the review.
Tests.
Data tables by test
| Test | No. of studies | No. of participants |
|---|---|---|
| 1 TB detection, all studies | 36 | 9557 |
| 2 Add on | 33 | 7264 |
| 3 Smear positive | 33 | 2020 |
| 4 Smear negative | 33 | 7264 |
| 5 HIV positive | 15 | 2474 |
| 6 HIV negative | 18 | 2555 |
| 7 TB detection, condition of specimen | 36 | 9557 |
| 8 TB detection, specimen preparation | 36 | 9557 |
| 9 Proportion TB cases | 36 | 9557 |
| 10 Income status | 36 | 9557 |
| 11 RIF resistance detect | 33 | 2966 |
| 12 Xpert version | 33 | 2966 |
| 13 Proportion RIF resistance | 33 | 2966 |
Test 1.

TB detection, all studies.
Test 2.

Add on.
Test 3.

Smear positive.
Test 4.

Smear negative.
Test 5.

HIV positive.
Test 6.

HIV negative.
Test 7.

TB detection, condition of specimen.
Test 8.

TB detection, specimen preparation.
Test 9.

Proportion TB cases.
Test 10.
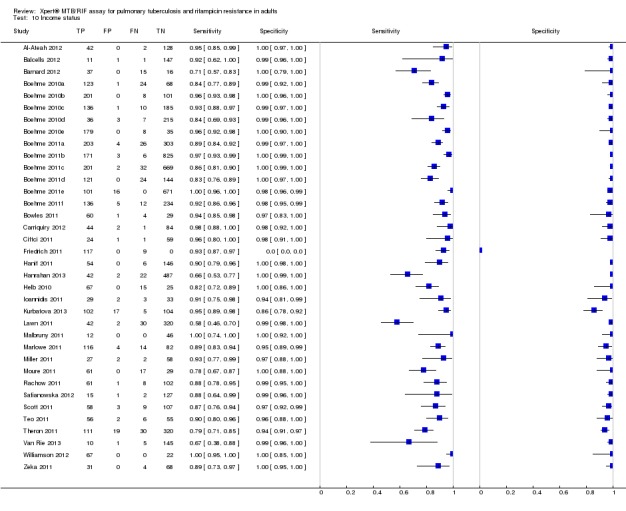
Income status.
Test 11.

RIF resistance detect.
Test 12.

Xpert version.
Test 13.

Proportion RIF resistance.
What's new
| Date | Event | Description |
|---|---|---|
| 16 March 2015 | Feedback has been incorporated | Feedback from Dr Tom Boyles at University of Cape Town has been incorporated and responded to. |
History
Protocol first published: Issue 1, 2012 Review first published: Issue 1, 2013
| Date | Event | Description |
|---|---|---|
| 6 May 2014 | Amended | Following information from one of the trial authors, details of the version of Xpert MTB/RIF used in Balcells 2012 have been corrected. |
| 13 February 2014 | Amended | Sentence moved in abstract; corrected 'pooled median sensitivity' to 'median pooled sensitivity' throughout. |
| 30 November 2013 | New search has been performed | 1. We performed an updated literature search on 7 February 2013. 2. For smear microscopy as a comparator test, we added a descriptive plot showing the estimates of sensitivity and specificity of Xpert compared with those of smear microscopy in studies that reported on both tests. 3. We included studies using Xpert version G4 (two studies) and studies evaluating Xpert in primary care clinics (two studies). These studies did not change the overall findings. 4. We improved the QUADAS‐2 assessment concerning applicability. 5. For TB detection, we repeated our earlier meta‐regression analyses within subgroups defined by smear status. 6. For rifampicin resistance detection, we performed univariate meta‐analyses for sensitivity and specificity separately in order to include studies in which no rifampicin resistance was detected. We also performed a sensitivity analysis using the bivariate random‐effects model for the subset of studies that provided data for both sensitivity and specificity. 7. We revised the summary of findings table to include clinical scenarios with prevalence levels recommended by the World Health Organization. 8. In the Background, we shortened the section on alternative tests to include only those tests most relevant to the review. 9. We added health economic considerations to the Discussion. 10. We added updated TB surveillance information. |
| 30 November 2013 | New citation required but conclusions have not changed | We conducted a new search and revised the review as described. |
| 17 January 2013 | Amended | We made some minor edits to the text to correct typographical errors. In addition, we replaced Figures 6, 8, 11, and 13 with new figures with minor modifications to the prediction regions. |
Differences between protocol and review
In the protocol we stated that we would extract data on industry sponsorship. However, we became aware that the Foundation for Innovative New Diagnostics (FIND) had negotiated a special price for the assay for TB endemic countries. As the majority of the included study centres were located in TB endemic countries, we assumed Xpert MTB/RIF had been purchased at the negotiated price. Therefore, we did not consider the included studies to be sponsored by industry.
We compared the accuracy of Xpert MTB/RIF for TB detection in high‐income versus low‐ and middle‐income countries. This comparison was not mentioned in the protocol. NTM were not mentioned in the protocol. In the update, NTM were considered non‐TB. We summarized separately data for NTM by determining the percent of false‐positive Xpert MTB/RIF results in samples that grew NTM.
We stated we would discuss the consequences when an uninterpretable test result was considered to be a (false) true negative result (may lead to missed/delayed diagnosis, with potential for increased morbidity, mortality, and TB transmission), or considered to be a (false) true positive result (may lead to unnecessary treatment with adverse events and increased anxiety). Since the rate of uninterpretable results was very low, we did not discuss these consequences.
Exploration of different reference standards, culture and clinical, while an interesting and important question, was beyond what we could carryout in an already complex review, with two review questions and multiple factors (including condition of specimen, income status, clinical subgroups) that could affect the summary estimates.
We performed additional sensitivity analyses for studies that did not clearly report the reason for testing and clinical information about patients and for studies that did not explicitly report patient age.
We initially used QUADAS, as mentioned in the protocol, but switched to QUADAS‐2 for the original review and updated review.
For smear microscopy as a comparator test, we added a descriptive plot showing the estimates of sensitivity and specificity of Xpert MTB/RIF compared to estimates of sensitivity of smear microscopy in studies that reported on both tests. We assumed smear specificity was 100%.
For TB detection, we repeated our earlier meta‐regression analyses within subgroups defined by smear status.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Al‐Ateah 2012
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of participants, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Not stated Age: Not stated Sex, female: 46.2% HIV infection: 0.6% History of TB: Not stated Sample size: 172 Clinical setting: Laboratory‐based evaluation of respiratory specimens Laboratory level: Intermediate Country: Saudi Arabia World Bank Income Classification: High‐income TB incidence rate: 17 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.8% (Source: Nationwide survey 2010) and among retreatment cases = 16% (Source: Nationwide survey 2010) Proportion of TB cases in the study: 25.6% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Processed Xpert MTB/RIF version: Not stated |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | The majority of specimens were obtained from bronchoalveolar lavage | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | High | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Balcells 2012
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Patients who fulfilled at least one of the following criteria: cough (> 10 days), bloody sputum, pneumonia unresponsive to previous antibiotics, fever (> 10 days), abnormal CXR or weight loss Age: Mean 37.4 years (range 19 to 65) Sex, female: 20.6% HIV infection: 100% History of TB: 11.8% Sample size: 160 Clinical setting: Five hospitals and their respective HIV clinics Laboratory level: Intermediate Country: Chile World Bank Income Classification: Middle‐income TB incidence rate: 18 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.7% (Source: Nationwide survey 2001) and among retreatment cases = 3.2% (Source: Nationwide surveillance 2011) Proportion of TB cases in the study: 7.5% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Processed Xpert MTB/RIF version: 2 and 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | |||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Barnard 2012
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Not stated Age: Predominantly adult, median age 41 Sex, female: 43.6% HIV infection: Not stated History of TB: 100% Sample size: 68 Clinical setting: Laboratory‐based evaluation of clinical specimens from previously treated patients Laboratory level: Intermediate Country: South Africa, Cape Town World Bank Income Classification: Middle‐income TB incidence rate: per 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Western Cape Province, 2002) and among retreatment cases = 4.0% (Source: Survey in Western Cape Province, 2002) Proportion of TB cases in the study: 76.5% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Processed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Fifteen patients submitted specimens for treatment monitoring, not diagnosis; three patients were 5, 7, and 10 years of age; all other patients were 16 years of age or older; according to GenoType MTBDRplus (v1.0) assay as the reference standard, Xpert MTB/RIF sensitivity and specificity for rifampicin resistance detection were 100% (based on three rifampicin resistant samples; 33 rifampicin susceptible samples). | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2010a
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Persistent productive cough for ≥ two weeks Age: Median 37 years; range 20 to 69 years Sex, female: 0% HIV infection: 4.7% History of TB: 54.6% Sample size: 216 Clinical setting: Special treatment facility for prisoners, high MDR‐TB setting Laboratory level: Central Country: Azerbaijan World Bank Income Classification: Middle‐income TB incidence rate: 113 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 22% (Source: survey in Baku City, 2007) and among retreatment cases = 56% (Source: survey in Baku City, 2007) Proportion of TB cases in study centre: 68.1% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Women were not included, but otherwise considered representative spectrum Data for one specimen per patient were provided by the study author |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | No | ||
| Low | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2010b
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Persistent productive cough for ≥ two weeks Age: Median 31 years; range 18 to 79 years Sex, female: 43.3% HIV infection: 1.7% History of TB: 23.7% Sample size: 310 Clinical setting: Primary health care DOTS (directly observed treatment, short‐course) centres in shanty towns Laboratory level: Intermediate Country: Peru World Bank Income Classification: Middle‐income TB incidence rate: 101 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 5.3% (Source: Nationwide survey 2006) and among retreatment cases = 24% (Source: Nationwide survey 2006) Proportion of TB cases in study centre: 67.4% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Data for one specimen per patient were provided by the study author | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2010c
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Persistent productive cough for ≥ two weeks Age: Median 36 years; range 18 to 80 years Sex, female: 34.1% HIV infection: 76.1% History of TB: 43.0% Sample size: 332 Clinical setting: Clinic, high HIV setting Laboratory level: Intermediate Country: South Africa, Cape Town World Bank Income Classification: Middle‐income TB incidence rate: per 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Western Cape Province, 2002) and among retreatment cases = 4.0% (Source: Survey in Western Cape Province, 2002) Proportion of TB cases in study centre: 44.0% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rfiampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Data for one specimen per patient were provided by the study author | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2010d
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Persistent productive cough for ≥ two weeks Age: Median 32 years; range 18 to 68 years Sex, female: 59.4% HIV infection: 71.4% History of TB: 45.1% Sample size: 261 Clinical setting: TB clinics, high HIV setting Laboratory level: Central Country: South Africa, Durban World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.7% (Source: Survey in Kwazulu‐Natal Province, 2002) and among retreatment cases = 7.7% (Source: Survey in Kwazulu‐Natal Province, 2002) Proportion of TB cases in study centre: 16.5% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Middlebrook 7H11 culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Data for one specimen per patient were provided by the study author | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | No | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2010e
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Persistent productive cough for ≥ two weeks Age: Median 30 years; range 17 to 88 years Sex, female: 39.1% HIV infection: 4.4% History of TB: 75.2% Sample size: 222 Clinical setting: Tertiary hospital, high MDR‐TB setting Laboratory level: Intermediate Country: India World Bank Income Classification: Middle‐income TB incidence rate: 181 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 2.1% (Source: Survey in Andhra Pradesh, 2009) and among retreatment cases = 12% (Source: Survey in Andhra Pradesh, 2009) Proportion of TB cases in study centre: 84.2% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Data for one specimen per patient were provided by the study author | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | No | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2011a
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 36 years; IQR 30 to 44 years Sex, female: < 1% HIV infection: < 1% History of TB: Not stated Sample size: 536 for detection of MTB; 211 for detection of rifampicin resistance Clinical setting: MDR‐TB screening facility Laboratory level: Intermediate Country: Azerbaijan World Bank Income Classification: Middle‐income TB incidence rate: 113 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 22% (Source: survey in Baku City, 2007) and among retreatment cases = 56% (Source: survey in Baku City, 2007) Proportion of TB cases in study centre: 42.7% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Follow‐up reported for all sites combined: 24/153 patients with culture‐negative, clinically diagnosed TB had positive results on MTB/RIF testing. 20/24 patients had follow‐up, and all 20 improved on TB treatment | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2011b
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 37 years; IQR 26 to 53 years Sex, female: 49% HIV infection: < 1% History of TB: Not stated Sample size: 1005 for detection of TB; 185 for detection of rifampicin resistance Clinical setting: Two health centres and one district hospital Laboratory level: Intermediate Country: Peru World Bank Income Classification: Middle‐income TB incidence rate: 101 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 5.3% and among retreatment cases = 23.6% (Source: Nationwide survey, 2006) Proportion of TB cases in study centre: 17.6% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Follow‐up was reported for all sites combined, see Boehme 2011a | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2011c
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 36 years; IQR 29 to 46 years Sex, female: 49% HIV infection: 38% History of TB: Not stated Sample size: 904 for detection of TB; 188 for detection of rifampicin resistance Clinical setting: One health centre and one provincial hospital Laboratory level: Intermediate Country: South Africa, Cape Town World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% and among retreatment cases = 4.0% (Source: Survey in Western Cape Province, 2002) Proportion of TB cases in study centre: 25.8% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 and MTBDRplus |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Follow‐up was reported for all sites combined, see Boehme 2011a MTBDRplus was done on culture isolates for smear‐negative sputum |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | No | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2011d
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 32 years; IQR 26 to 38 years Sex, female: < 46% HIV infection: < 68% History of TB: Not stated Sample size: 289 for detection of TB; 116 for detection of rifampicin resistance Clinical setting: Emergency unit of referral hospital Laboratory level: Intermediate Country: Uganda World Bank Income Classification: Low‐income TB incidence rate: 193 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.4% (Source: Nationwide survey, 2011) and among retreatment cases = 12% (Source: Nationwide survey, 2011) Proportion of TB cases in the study centre: 50.2% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media and line probe assay |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Follow‐up was reported for all sites combined, see Boehme 2011a Line‐probe assay and, for 10% of culture positive patients (every tenth patient), Löwenstein–Jensen proportion was performed on MGIT isolates (except when only positive on Löwenstein–Jensen). |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | No | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2011e
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 45 years; IQR 32 to 58 years Sex, female: 30% HIV infection: 4% History of TB: Not stated Sample size: 788 for detection of TB; 103 for detection of rifampicin resistance Clinical setting: Health centre Laboratory level: Intermediate Country: India World Bank Income Classification: Middle‐income TB incidence rate: 181 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 2.1% (Source: Survey in Andhra Pradesh, 2009) and among retreatment cases = 12% (Source: Survey in Andhra Pradesh, 2009) Proportion of TB cases in the study centre: 12.8% |
||
| Index tests | Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Follow‐up was reported for all sites combined, see Boehme 2011a | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Boehme 2011f
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants; site in a multicentre study | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 47 years; IQR 34 to 58 years Sex, female: 36% HIV infection: < 1% History of TB: Not stated Sample size: 387 for detection of TB; 257 for detection of rifampicin resistance Clinical setting: MDR‐TB screening facility Laboratory level: Intermediate Country: Philippines World Bank Income Classification: Middle‐income TB incidence rate: 270 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 4% (Source: Nationwide survey, 2004) and among retreatment cases = 21% (Source: Nationwide survey, 2004) Proportion of TB cases in the study centre: 38.2% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Ogawa culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Follow‐up was reported for all sites combined, see Boehme 2011a | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Unclear | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Bowles 2011
| Study characteristics | |||
| Patient sampling | Prospective and retrospective study with enrolment of participants by convenience | ||
| Patient characteristics and setting | Presenting signs and symptoms: Not reported Age: Not stated Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 89 Clinical setting: Laboratory‐based evaluation of respiratory specimens (predominantly sputum specimens) from a TB reference clinic Laboratory level: Central Country: Netherlands World Bank Income Classification: High‐income TB incidence rate: 6.8 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.7% and among retreatment cases = 4.5% (Source: Nationwide surveillance, 2011) Proportion of TB cases in the study: 71.9% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: 26 fresh and 63 frozen (previously stored) samples Specimen Preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Sample included two extrapulmonary specimens (one pleural fluid and one gastric aspirate) One patient whose sample was smear and culture‐negative was culture‐positive on a sample 11 days later |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | No | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| High | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Unclear | ||
| Unclear | |||
Carriquiry 2012
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough for greater than 10 days with abnormal chest X‐ray and at least one of the following symptoms: fever, fatigue, night sweats, haemoptysis, chest pain, or weight loss Age: Median 35 years (IQR 29 to 42) Sex, female: 27.5% HIV infection: 100% History of TB: 57.3% Sample size: 131 Clinical setting: Two tertiary hospitals Laboratory level: Intermediate Country: Peru World Bank Income Classification: Middle‐income TB incidence rate: 101 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 5.3% (Source: Nationwide survey 2006) and among retreatment cases = 24% (Source: Nationwide survey 2006) Proportion of TB cases in the study: 34.4% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 2 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | |||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Ciftci 2011
| Study characteristics | |||
| Patient sampling | Prospective study; the sampling method was unclear | ||
| Patient characteristics and setting | Presenting signs and symptoms: Symptoms suggestive of TB Age: Not stated Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 85 Clinical setting: Laboratory‐based evaluation of respiratory specimens (predominantly sputum) at a university hospital Laboratory level: Intermediate Country: Turkey World Bank Income Classification: Middle‐income TB incidence rate: 24 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Ankara City 2011) and among retreatment cases = 38% (Source: Survey in Ankara City 2011) Proportion of TB cases in the study: 29.4% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh Specimen Preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: BACTEC 460 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: BACTEC 460 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Paper was written in Turkish: sample included 10 extrapulmonary specimens (five pleural fluid and five urine samples); no patients were found to have rifampicin resistance | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Unclear | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Unclear | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Unclear | ||
| Unclear | |||
Friedrich 2011
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Cross‐sectional design with consecutive enrolment of participants, prospective data collection Presenting signs and symptoms: Patients recently diagnosed with smear‐positive first time TB, untreated Age: Eligible aged 18 to 65 years Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 126 Clinical setting: Two medical centres Laboratory level: Intermediate Country: South Africa, Cape Town World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Western Cape Province, 2002) and among retreatment cases = 4.0% (Source: Survey in Western Cape Province, 2002) Proportion of TB cases in the study: 100.0% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh Specimen Preparation: Processed Xpert MTB/RIF version: 2 and 4 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | The aim of this study was to assess NAATs for selecting patients for clinical trials of anti‐TB medication. Patients with severe co‐morbidities were excluded. This study was used only for determination of sensitivity because all enrolled patients were predetermined to have TB disease. | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | No | ||
| High | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Hanif 2011
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: Presumed TB based on presence of cough and radiographic findings Age: Range 20 to 57 years old Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 206 Clinical setting: Laboratory‐based evaluation of respiratory specimens (predominantly sputum) at a university hospital Laboratory level: Central Country: Kuwait World Bank Income Classification: High‐income TB incidence rate: 36 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0% and among retreatment cases = 12% (Source: Nationwide surveillance, 2011) Proportion of TB cases in the study: 29.1% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh Specimen Preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: BACTEC 460 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | No patients were found to have rifampicin resistance | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | No | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| High | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | No | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Hanrahan 2013
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Prolonged (> two weeks) cough and/or other TB symptoms Age: 18 and older Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 551 Clinical setting: Primary care clinic Laboratory level: Peripheral Country: South Africa, Johannesburg World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.4% (Source: Survey in Gauteng province, 2002) and among retreatment cases = 5.5% (Source: Survey in Gauteng province, 2002) Proportion of TB cases in the study: 11.6% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | |||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Helb 2010
| Study characteristics | |||
| Patient sampling | Retrospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: Cough lasting at least two weeks Age: Median 34 years; range 18 to 76 years Sex, female: 30.8% HIV infection: 0.9% History of TB: 1.9% Sample size: 107 Clinical setting: TB hospital Laboratory level: Intermediate Country: Vietnam World Bank Income Classification: Middle‐income TB incidence rate: 199 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 2.7% (Source: Nationwide survey, 2006) and among retreatment cases = 19% (Source: Nationwide survey, 2006) Proportion of TB cases in the study: 76.6% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Frozen Specimen Preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard: Löwenstein‐Jensen culture and MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Rifampicin resistance data were not reported | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Unclear | ||
| Unclear | |||
Ioannidis 2011
| Study characteristics | |||
| Patient sampling | Prospective and retrospective study with enrolment of participants by convenience | ||
| Patient characteristics and setting | Presenting signs and symptoms: High suspicion of TB in patients found to be predominantly smear negative by microscopy examination Age: Not stated Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 66 Clinical setting: Laboratory‐based evaluation in routine hospital setting Laboratory level: Central Country: Greece World Bank Income Classification: High‐income TB incidence rate: 3.8 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Nationwide surveillance, 2010) and among retreatment cases = 6.7% (Source: Nationwide surveillance, 2010) Proportion of TB cases in the study: 48.0% |
||
| Index tests | Index: Xpert MTB/RIF assay Condition: Fresh Preparation: Processed Xpert MTB/RIF version: 2 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on Löwenstein‐Jensen media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Specimens were predominantly smear‐negative | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | No | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| High | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Kurbatova 2013
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Presumptive or recently diagnosed TB Age: Not stated Sex, female: Not stated HIV infection: estimated < 5 % History of TB: Not stated Sample size: 238 Clinical setting: Laboratory‐based evaluation Laboratory level: Central and intermediate Country: Russia World Bank Income Classification: Middle‐income TB incidence rate: 97 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 20% (Source: Surveillance in 20 Oblasts 2010) and among retreatment cases = 46% (Source: Surveillance in 20 Oblasts 2008) Proportion of TB cases in the study: 46.9% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 4 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Fresh, unconcentrated sputum was initially homogenized using a vortex with glass beads | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Lawn 2011
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: HIV‐infected patients with advanced immunodeficiency; the majority of patients had one or more of the following TB symptoms: current cough, fever, night sweats, or weight loss Age: Median 34 years; IQR 28 to 41 years Sex, female: 65.4% HIV infection: 100% History of TB: 26.5% Sample size: 394 Clinical setting: HIV anti‐retroviral clinic; all patients were screened for TB Laboratory level: Intermediate Country: South Africa, Cape Town World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Western Cape Province, 2002) and among retreatment cases = 4.0% (Source: Survey in Western Cape Province, 2002) Proportion of TB cases in the study: 18.3% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh Specimen Preparation: Processed Xpert MTB/RIF version: Not stated |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | This study evaluated the use of Xpert to screen HIV‐infected patients with advanced immunodeficiency enrolling in antiretroviral therapy services regardless of symptoms, although the majority of patients in the study had TB symptoms. Of three patients with apparent false‐positive Xpert MTB/RIF results, on follow‐up, two patients had overt pulmonary and systemic symptoms suggestive of TB and improved on anti‐TB treatment. The third patient was lost to follow‐up. Median CD4 cell count, 171 cells/ml; IQR 102 to 236 |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | High | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Malbruny 2011
| Study characteristics | |||
| Patient sampling | Prospective and retrospective study with enrolment of participants by convenience | ||
| Patient characteristics and setting | Presenting signs and symptoms: Clinical symptoms suggestive of TB Age: Median 52 years Sex, female: 40.2% HIV infection: Not stated History of TB: Not stated Sample size: 58 Clinical setting: Laboratory‐based evaluation of respiratory specimens (predominantly bronchial aspirates) at a university hospital Laboratory level: Intermediate Country: France World Bank Income Classification: High‐income TB incidence rate: 4.3 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.0% (Source: Nationwide surveillance, 2009) and among retreatment cases = 13% (Source: Nationwide surveillance, 2009) Proportion of TB cases in the study: 20.7% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh and frozen Specimen Preparation: Processed Xpert MTB/RIF version: 1 and 2 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Solid culture, type unspecified, and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | 31/58 (53.4%) of samples were bronchial aspirates One rifampicin‐resistant isolate was identified |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | No | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| High | High | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Marlowe 2011
| Study characteristics | |||
| Patient sampling | Prospective and retrospective study with selection of specimens by convenience at two sites and consecutive selection of smear‐positive specimens at one site | ||
| Patient characteristics and setting | Presenting signs and symptoms: Not reported Age: Not stated Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 216 Clinical setting: Laboratory‐based evaluation of respiratory samples Laboratory level: Central (one laboratory) and intermediate (two laboratories) Country: USA World Bank Income Classification: High income TB incidence rate: 3.9 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.4% (Source: Nationwide surveillance, 2011) and among retreatment cases = 7.6% (Source: Nationwide surveillance, 2011) Proportion of TB cases in the study: 60.2% |
||
| Index tests | Index: Xpert MTB/RIF assay Condition: Fresh and frozen Preparation: Processed Xpert MTB/RIF version: Not stated |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture, Middlebrook 7H11 culture, and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: phenotypic drug susceptibility testing with agar‐based solid media and MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Unit of analysis was specimen Different reference standards were used at each of the three sites |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | No | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| High | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | No | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Miller 2011
| Study characteristics | |||
| Patient sampling | Retrospective study; with enrolment of participants by convenience | ||
| Patient characteristics and setting | Presenting signs and symptoms: Not reported Age: Data provided for patients with pulmonary and extrapulmonary combined; 95% of patients were 15 years and older Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 89 pulmonary specimens (in addition, study included 23 extrapulmonary specimens) Clinical setting: Laboratory‐based evaluation of clinical specimens at a university hospital Laboratory level: Intermediate Country: USA World Bank Income Classification: High income TB incidence rate: 3.9 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.4% (Source: Nationwide surveillance, 2011) and among retreatment cases = 7.6% (Source: Nationwide surveillance, 2011) Proportion of TB cases in the study: 32.6% |
||
| Index tests | Index: Xpert MTB/RIF assay Condition: Frozen Preparation: Processed Xpert MTB/RIF version: Not stated |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Of specimens tested, four were positive by Xpert MTB/RIF for rifampicin resistance; three were positive by the reference standard | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | No | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| High | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Moure 2011
| Study characteristics | |||
| Patient sampling | Retrospective study with enrolment of participants by convenience | ||
| Patient characteristics and setting | Presenting signs and symptoms: Patients found to be smear negative by microscopy examination Age: All patients were 15 years of age or older Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 107 Clinical setting: Laboratory‐based evaluation of clinical specimens at a university hospital Laboratory level: Intermediate Country: Spain World Bank Income Classification: High income TB incidence rate: 15 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.2% (Source: Survey in Galicia region, 2005) and among retreatment cases = 1.5% (Source: Survey in Galicia region, 2005) Proportion of TB cases in the study: 72.9% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Frozen Specimen Preparation: Processed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Lowenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Bactec 460 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Sample set included one pulmonary biopsy specimen Of 85 pulmonary and extrapulmonary specimens tested, six were positive by Xpert MTB/RIF for rifampicin resistance, and seven specimens were positive by the reference standard |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | No | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| High | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Rachow 2011
| Study characteristics | |||
| Patient sampling | Retrospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: Presumed pulmonary TB based on clinical and radiographic findings Age: Mean 39 years (SD = 13.8) Sex, female: 51.7% HIV infection: 58.9% History of TB: Not stated Sample size: 172 Clinical setting: Referral hospital, high HIV setting Laboratory level: Central Country: United Republic of Tanzania World Bank Income Classification: Low‐income TB incidence rate: 169 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.1% (Source: Nationwide survey, 2007) and among retreatment cases = 0% (Source: Nationwide survey, 2007) Proportion of TB cases in the study: 40.1% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Frozen Specimen Preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Patients were followed for a period of 56 days. Among 77 patients classified as smear negative, culture negative 'clinical TB', Xpert MTB/RIF was positive in seven (9.1%) patients No patients were found to have rifampicin resistance |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Unclear | ||
| Unclear | |||
Safianowska 2012
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Patients presumed to have TB Age: Mean 61 years; range 20 to 97 years Sex, female: 36.6% HIV infection: 0% History of TB: Not stated Sample size: 145 Clinical setting: Laboratory‐based evaluation Laboratory level: Intermediate Country: Poland World Bank Income Classification: High‐income TB incidence rate: 23 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.5% (Source: Nationwide surveillance, 2011) and among retreatment cases = 3.5% (Source: Nationwide surveillance, 2011) Proportion of TB cases in the study: 11.8% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh Specimen Preparation: Processed Xpert MTB/RIF version: 1 and 2 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Löwenstein‐Jensen media, method not specified |
||
| Flow and timing | |||
| Comparative | |||
| Notes | |||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Scott 2011
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: Patients presumed to have TB, presenting with cough, fever, night sweats, and/or weight loss Age: Mean 32 years; range 19 to 75 years Sex, female: 41.1% HIV infection: 69.0% History of TB: Not stated Sample size: 177 Clinical setting: Primary care clinic Laboratory level: Intermediate Country: South Africa, Johannesburg World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.4% (Source: Survey in Gauteng province, 2002) and among retreatment cases = 5.5% (Source: Survey in Gauteng province, 2002) Proportion of TB cases in the study: 37.9% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Frozen Specimen Preparation: Processed Xpert MTB/RIF version: 1 and 2 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | One follow‐up visit was performed approximately 60 days after enrolment Xpert MTB/RIF was performed on frozen specimens while MGIT culture and smear microscopy were performed on fresh specimens |
||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Teo 2011
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: Patients thought to have TB based on symptoms and radiographic findings Age: Not stated Sex, female: Not stated HIV infection: Not stated History of TB: Not stated Sample size: 106 Clinical setting: University hospital Laboratory level: Central Country: Singapore World Bank Income Classification: High‐income TB incidence rate: 37 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.6% (Source: Nationwide surveillance, 2011) and among retreatment cases = 0% (Source: Nationwide surveillance, 2011) Proportion of TB cases in the study: 58.5% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Fresh Specimen Preparation: Processed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Gene sequencing |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Respiratory specimens (predominantly sputum) submitted for routine testing; only one rifampicin‐resistant isolate was identified | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Unclear | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | No | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | No | ||
| High | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Theron 2011
| Study characteristics | |||
| Patient sampling | Retrospective study with consecutive enrolment of participants | ||
| Patient characteristics and setting | Presenting signs and symptoms: Patients presumed to have TB based on compatible signs and symptoms Age: Median 36 years; range 18 to 83 years Sex, female: 32.3% HIV infection: 31.3% History of TB: 34.3% Sample size: 480 Clinical setting: Two primary care clinics in a high HIV prevalence area Laboratory level: Intermediate Country: South Africa, Cape Town World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Western Cape Province, 2002) and among retreatment cases = 4.0% (Source: Survey in Western Cape Province, 2002) Proportion of TB cases in the study: 29.4% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Frozen Specimen Preparation: Unprocessed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Short‐term follow‐up cultures were obtained; 16 of 19 Xpert MTB/RIF‐positive culture‐negative patients were considered likely to be TB cases based on follow‐up cultures, gene sequencing, and the presence of characteristic radiographic features using a standardised scoring system | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Van Rie 2013
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Prolonged (> two weeks) cough or other TB symptoms, or both, and had two prior‐negative smear by fluorescence microscopy Age: Median 36 years (IQR 30 to 34) Sex, female: 56.8% HIV infection: 72.4% History of TB: 17.6% Sample size: 199 Clinical setting: Primary care clinic Laboratory level: Peripheral Country: South Africa, Johannesburg World Bank Income Classification: Middle‐income TB incidence rate: 993 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 1.4% (Source: Survey in Gauteng province, 2002) and among retreatment cases = 5.5% (Source: Survey in Gauteng province, 2002) Proportion of TB cases in the study: 9.3% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Unprocessed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Only those patients presumed to have TB who returned for results of the initial smear microscopy examinations were enrolled | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | No | ||
| High | |||
Williamson 2012
| Study characteristics | |||
| Patient sampling | Cross‐sectional design with consecutive enrolment of smear‐positive patients, prospective data collection | ||
| Patient characteristics and setting | Presenting signs and symptoms: Clinical symptoms not reported: smear‐positive specimens Age: > 15 years Sex, female: Not stated HIV infection: estimated < 1% History of TB: Not stated Sample size: 89 Clinical setting: Laboratory‐based evaluation Laboratory level: Intermediate Country: New Zealand World Bank Income Classification: High‐income TB incidence rate: 7.6 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 2.5% (Source: Nationwide surveillance 2009) and among retreatment cases = 13% (Source: Nationwide surveillance 2009) Proportion of TB cases in the study: 75.3% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen condition: Fresh Specimen preparation: Processed Xpert MTB/RIF version: 3 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: MGIT 960 Target condition: Rifampicin resistance Reference standard for rifampicin resistance: MGIT 960 |
||
| Flow and timing | |||
| Comparative | |||
| Notes | |||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | No | ||
| High | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | Yes | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | Yes | ||
| Low | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Zeka 2011
| Study characteristics | |||
| Patient sampling | Prospective study with consecutive enrolment of patients | ||
| Patient characteristics and setting | Presenting signs and symptoms: Clinical findings of possible TB Age: Median 48 years; range 25 to 70 years Sex, female: 42.4% HIV infection: Not stated History of TB: Not stated Sample size: 103 Clinical setting: Laboratory‐based evaluation of routine sputum specimens at a university hospital Laboratory level: Intermediate Country: Turkey World Bank Income Classification: Middle‐income TB incidence rate: 24 per 100,000 MDR‐TB prevalence: Percent MDR‐TB among new TB cases = 0.9% (Source: Survey in Ankara City 2011) and among retreatment cases = 38% (Source: Survey in Ankara City 2011) Proportion of TB cases in the study: 34.0% |
||
| Index tests | Index: Xpert MTB/RIF assay Specimen Condition: Frozen Specimen Preparation: Processed Xpert MTB/RIF version: 1 |
||
| Target condition and reference standard(s) | Target condition: Pulmonary TB Reference standard for pulmonary TB: Löwenstein‐Jensen culture and MB/MBacT liquid medium Target condition: Rifampicin resistance Reference standard for rifampicin resistance: Proportion method on 7H10 media |
||
| Flow and timing | |||
| Comparative | |||
| Notes | Only one rifampicin resistant isolate was identified. Data for sputum specimens were provided by the study author | ||
| Methodological quality | |||
| Item | Authors' judgement | Risk of bias | Applicability concerns |
| DOMAIN 1: Patient Selection | |||
| Was a consecutive or random sample of patients enrolled? | Yes | ||
| Was a case‐control design avoided? | Yes | ||
| Did the study avoid inappropriate exclusions? | Yes | ||
| Low | Low | ||
| DOMAIN 2: Index Test All tests | |||
| Were the index test results interpreted without knowledge of the results of the reference standard? | Yes | ||
| If a threshold was used, was it pre‐specified? | Yes | ||
| Low | Low | ||
| DOMAIN 3: Reference Standard | |||
| Is the reference standards likely to correctly classify the target condition? | Yes | ||
| Were the reference standard results for TB detection interpreted without knowledge of the results of the index test? | No | ||
| Were the reference standard results for rifampicin resistance detection interpreted without knowledge of the results of the index test? | No | ||
| High | Low | ||
| DOMAIN 4: Flow and Timing | |||
| Was there an appropriate interval between index test and reference standard? | Yes | ||
| Did all patients receive the same reference standard? | Yes | ||
| Were all patients included in the analysis? | Yes | ||
| Low | |||
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Alvarez‐Uria 2012 | Reference standard not satisfied. |
| Andersen 2011 | Editorial and comment. |
| Armand 2011 | This was a case‐control study that compared Xpert MTB/RIF with an in‐house IS6110‐based real‐time PCR using TaqMan probes (IS6110‐TaqMan assay) for TB detection. |
| Banada 2010 | Technical paper. |
| Bates 2013 | This study evaluated Xpert MTB/RIF for the diagnosis of TB in children. |
| Blakemore 2010 | Technical paper. |
| Blakemore 2011 | This was a technical paper that compared bacterial load quantitation determined by Xpert MTB/RIF with the load determined by conventional quantitative methods. |
| Causse 2011 | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Chegou 2011 | Narrative review. |
| Clouse 2012 | Study on patient impact. |
| Cuevas 2011 | Narrative review. |
| Dorjee 2012 | Case report. |
| Dorman 2012 | Prevalence survey. |
| Dowdy 2011 | Cost‐effectiveness study. |
| Evans 2011 | Editorial and comment. |
| Farga 2011 | Narrative review. |
| Fenner 2011 | Editorial and comment. |
| Ferrara 2011 | Editorial and comment. |
| FIND 2011 | This study compared Xpert MTB/RIF G3 and G4. We excluded it because of concern about duplicate data. In addtion, the crieria for the reference standard for rifampicin resistance detection were not satisfied. |
| Friedrich 2011a | This study evaluated Xpert MTB/RIF for the diagnosis of pleural TB. |
| Gotuzzo 2011 | Editorial and comment. |
| Hesseling 2011 | Editorial and comment. |
| Hillemann 2011 | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Hoek 2011 | Narrative review. |
| Ioannidis 2010 | We could not obtain this article. |
| Kim 2012 | Case‐control study. |
| Kirwan 2012 | Editorial and comment. |
| Lawn 2011a | This was a narrative review that covered the development, technical details, and diagnostic accuracy of Xpert MTB/RIF in adults and children. |
| Lawn 2011b | Editorial and comment. |
| Lawn 2012 | Study on patient impact. |
| Lawn 2012a | Data insufficient for 2 x 2 table. |
| Lawn 2012b | Correspondence. |
| Lawn 2012c | Primarily a lipoarabinomannan detection study. |
| Lawn 2012d | Duplicate data. |
| Ligthelm 2011 | This study evaluated Xpert MTB/RIF for the diagnosis of TB lymphadenitis. |
| Melzer 2011 | Editorial and comment. |
| Miotto 2012 | Treatment monitoring. |
| Morris 2010 | Editorial and comment. |
| Morris 2011 | Editorial and comment. |
| Moure 2012 | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Muñoz 2013 | Study on patient impact. |
| Narasimooloo 2012 | Study on patient impact. |
| Nhu 2013 | This study evaluated Xpert MTB/RIF for the diagnosis of TB in children. |
| Nicol 2011 | This study evaluated Xpert for the diagnosis of TB in children. |
| Ntinginya 2012 | This study included both adults and children. The study used an active case finding strategy involving previously known TB cases and identified five additional culture‐confirmed TB cases (5/219). Xpert MTB/RIF showed a positive result in all five culture‐confirmed TB cases (sensitivity = 100%). We considered the study design to be different from a diagnostic test accuracy study and therefore did not include this study in the review. |
| O'Grady 2012 | This study evaluated Xpert MTB/RIF in patients able to produce sputum, irrespective of admission diagnosis, not presumed TB patients. |
| Perkins 2011 | Correspondence. |
| Peter 2012a | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Peters 2012 | Correspondence. |
| Rachow 2012 | This study evaluated Xpert for the diagnosis of TB in children. |
| Salvo 2011 | Editorial and comment. |
| Scott 2012 | Technical paper. |
| Swaminathan 2012 | Relevance. This study disucssed lipoarabinomannan. |
| Tansey 2009 | Editorial and comment. |
| Taylor 2012 | This study evaluated Xpert for the diagnosis of extrapulmonary TB. |
| Theron 2011a | Editorial and comment. |
| Theron 2012 | Treatment monitoring. |
| Theron 2012a | Duplicate data. |
| Theron 2012b | Duplicate data. |
| Tortoli 2012 | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Trebucq 2012 | Editorial and comment. |
| Trebucq 2012a | Correspondence. |
| Vadwai 2011 | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Vadwai 2012 | Correspondence. |
| Van Rie 2010 | This was a review that covered technical details of Xpert MTB/RIF and the test's potential value as a point‐of‐care test. |
| Van Rie 2011 | Case report. |
| van Zyl‐Smit 2011 | Technical paper. |
| Walters 2012 | This study evaluated Xpert MTB/RIF for the diagnosis of TB in children. |
| Williamson 2012a | Case‐control study. |
| Wood 2012 | This study evaluated Xpert MTB/RIF for the diagnosis of extrapulmonary TB. |
| Yoon 2012 | Duplicate data. |
| Zar 2012 | This study evaluated Xpert MTB/RIF for the diagnosis of TB in children. |
Characteristics of ongoing studies [ordered by study ID]
| Trial name or title | GeneXpert MTB/RIF, a new tool for the diagnosis of pulmonary tuberculosis in two municipalities in Brazil |
| Target condition and reference standard(s) | TB cases bacteriologically confirmed |
| Index and comparator tests | Xpert MTB/RIF assay and smear microscopy |
| Starting date | January 2012 |
| Contact information | Betina Durovni bdurovni@saude.rio.rj.br |
| Notes | Group‐randomized pragmatic trial following a stepped‐wedge design. Identifier: NCT01363765 |
| Trial name or title | Evaluation of Xpert MTB/RIF assay for the rapid identification of TB and TB rifampin resistance in HIV‐infected and HIV‐uninfected patients with presumed pulmonary tuberculosis |
| Target condition and reference standard(s) | TB: reference standard: MGIT culture |
| Index and comparator tests | Xpert MTB/RIF assay |
| Starting date | 24 April 2012 |
| Contact information | Jay (John) Dwyer jdwyer@php.ucsf.edu |
| Notes | Cohort study of diagnostic accuracy of Xpert MTB/RIF in HIV‐infected and HIV‐uninfected patients presumed to have pulmonary TB. Identifier: NCT01587469 |
| Trial name or title | A randomised control trial of sputum induction, and new and emerging technologies in a high HIV prevalence primary care setting |
| Target condition and reference standard(s) | TB: liquid culture |
| Index and comparator tests | Xpert MTB/RIF assay |
| Starting date | August 2009 |
| Contact information | Jonny Peter Jonny.Peter@uct.ac.za |
| Notes | RCT to evaluate sputum induction for TB diagnosis in a primary care clinic for adults presumed to have TB. Identifier: NCT01545661 |
Contributions of authors
MP conceived the original idea for the review. KRS, MP, and ND wrote the protocol. LAK drafted the search strategy. For this updated Cochrane Review, KRS and DH reviewed articles for inclusion and extracted data. KRS, IS, and ND analysed the data. KRS, MP, and ND interpreted the analyses. KRS drafted the manuscript. ND drafted the statistical analysis section and Appendix 4. KRS, CCB, MP, and ND provided critical revisions to the manuscript. All authors read and approved the final manuscript draft.
Sources of support
Internal sources
Liverpool School of Tropical Medicine, UK.
External sources
-
United States Agency for International Development (USAID), USA.
Development and publication of this manuscript was in part made possible with financial support from the United States Agency for International Development (USAID)
Declarations of interest
The CIDG provided funding in part for this review. KRS serves as Co‐ordinator of the Evidence Synthesis and Policy Subgroup of Stop TB Partnership's New Diagnostics Working Group. KRS received funding to carry out the original Cochrane Review from CIDG and McGill University and the updated Cochrane Review from the United States Agency for International Development (USAID), USA. MP is a recipient of a New Investigator Award from the Canadian Institutes of Health Research (CIHR) and a salary award from Fonds de recherche du Québec ‐ Santé. MP serves as an external consultant for the Bill & Melinda Gates Foundation. CCB is employed by the Foundation for Innovative New Diagnostics (FIND) and has conducted studies and published on Xpert MTB/RIF as part of a collaborative project between FIND, a Swiss non‐profit, Cepheid, a US company, and academic partners. The product developed through this partnership was developed under a contract that obligated FIND to pay for development costs and trial costs and Cepheid to make the test available at specified preferential pricing to the public sector in developing countries. The authors have no financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the review apart from those disclosed. IS received funding to carry out the updated Cochrane Review from CIDG and USAID.
Unchanged, comment added to review
References
References to studies included in this review
- Al‐Ateah SM, Al‐Dowaidi MM, El‐Khizzi NA. Evaluation of direct detection of Mycobacterium tuberculosis complex in respiratory and non‐respiratory clinical specimens using the Cepheid Gene Xpert® system. Saudi Medical Journal 2012;33(10):1100‐5. [PubMed] [Google Scholar]
- Balcells ME, García P, Chanqueo L, Bahamondes L, Lasso M, Gallardo AM, et al. Rapid molecular detection of pulmonary tuberculosis in HIV‐infected patients in Santiago, Chile. International Journal of Tuberculosis and Lung Disease 2012;16(10):1349‐53. [DOI] [PubMed] [Google Scholar]
- Barnard M, Gey van Pittius NC, Helden PD, Bosman M, Coetzee G, Warren RM. The diagnostic performance of the GenoType MTBDRplus version 2 line probe assay is equivalent to that of the Xpert MTB/RIF assay. Journal of Clinical Microbiology 2012;50(11):3712‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nabeta P, Hillemann D, Nicol MP, Shenai S, Krapp F, et al. Rapid molecular detection of tuberculosis and rifampin resistance. New England Journal of Medicine 2010;363(11):1005‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nabeta P, Hillemann D, Nicol MP, Shenai S, Krapp F, et al. Rapid molecular detection of tuberculosis and rifampin resistance. New England Journal of Medicine 2010;363(11):1005‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nabeta P, Hillemann D, Nicol MP, Shenai S, Krapp F, et al. Rapid molecular detection of tuberculosis and rifampin resistance. New England Journal of Medicine 2010;363(11):1005‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nabeta P, Hillemann D, Nicol MP, Shenai S, Krapp F, et al. Rapid molecular detection of tuberculosis and rifampin resistance. New England Journal of Medicine 2010;363(11):1005‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nabeta P, Hillemann D, Nicol MP, Shenai S, Krapp F, et al. Rapid molecular detection of tuberculosis and rifampin resistance. New England Journal of Medicine 2010;363(11):1005‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowles EC, Freyée B, Ingen J, Mulder B, Boeree M J, Soolingen D. Xpert MTB/RIF®, a novel automated polymerase chain reaction‐based tool for the diagnosis of tuberculosis. International Journal of Tuberculosis and Lung Disease 2011;15(7):988‐9. [DOI] [PubMed] [Google Scholar]
- Carriquiry G, Otero L, González‐Lagos E, Zamudio C, Sánchez E, Nabeta P, et al. A diagnostic accuracy study of Xpert®MTB/RIF in HIV‐positive patients with high clinical suspicion of pulmonary tuberculosis in Lima, Peru. PLoS One 2012;7(9):e44626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciftçi IH, Aslan MH, Aşik G. [Evaluation of Xpert MTB/RIF results for the detection of Mycobacterium tuberculosis in clinical samples]. Mikrobiyoloji bülteni 2011;45(1):43‐7. [PubMed] [Google Scholar]
- Friedrich SO, Venter A, Kayigire XA, Dawson R, Donald PR, Diacon AH. Suitability of Xpert MTB/RIF and genotype MTBDRplus for patient selection for a tuberculosis clinical trial. Journal of Clinical Microbiology 2011;49(8):2827‐31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanif SN, Eldeen HS, Ahmad S, Mokaddas E. GeneXpert® MTB/RIF for rapid detection of Mycobacterium tuberculosis in pulmonary and extra‐pulmonary samples. International Journal of Tuberculosis and Lung Disease 2011;15(9):1274‐5. [DOI] [PubMed] [Google Scholar]
- Hanrahan CF, Selibas K, Deery CB, Dansey H, Clouse K, et al. Time to treatment and patient outcomes among TB suspects screened by a single point‐of‐Care Xpert MTB/RIF at a primary care clinic in Johannesburg, South Africa. PLoS ONE 2013;8(6):e65421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helb D, Jones M, Story E, Boehme C, Wallace E, Ho K, et al. Rapid detection of Mycobacterium tuberculosis and rifampin resistance by use of on‐demand, near‐patient technology. Journal of Clinical Microbiology 2010;48(1):229‐37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ioannidis P, Papaventsis D, Karabela S, Nikolaou S, Panagi M, Raftopoulou E, et al. Cepheid GeneXpert MTB/RIF assay for Mycobacterium tuberculosis detection and rifampin resistance identification in patients with substantial clinical indications of tuberculosis and smear‐negative microscopy results. Journal of Clinical Microbiology 2011;49(8):3068‐70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurbatova EV, Kaminski DA, Erokhin VV, Volchenkov GV, Andreevskaya SN, Chernousova LN, et al. Performance of Cepheid® Xpert MTB/RIF® and TB‐Biochip® MDR in two regions of Russia with a high prevalence of drug‐resistant tuberculosis. European Journal of Clinical Microbiology and Infectious Disease 2013;32(6):735‐43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawn SD, Brooks SV, Kranzer K, Nicol MP, Whitelaw A, Vogt M, et al. Screening for HIV‐associated tuberculosis and rifampicin resistance before antiretroviral therapy using the Xpert MTB/RIF assay: a prospective study. PLoS Medicine 2011;8(7):e1001067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malbruny B, Marrec G, Courageux K, Leclercq R, Cattoir V. Rapid and efficient detection of Mycobacterium tuberculosis in respiratory and non‐respiratory samples. International Journal of Tuberculosis and Lung Disease 2011;15(4):553‐5. [DOI] [PubMed] [Google Scholar]
- Marlowe EM, Novak‐Weekley SM, Cumpio J, Sharp SE, Momeny MA, Babst A, et al. Evaluation of the Cepheid Xpert MTB/RIF assay for direct detection of Mycobacterium tuberculosis complex in respiratory specimens. Journal of Clinical Microbiology 2011;49(4):1621‐3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller MB, Popowitch EB, Backlund MG, Ager EP. Performance of Xpert MTB/RIF RUO Assay and IS6110 Real‐Time PCR for Mycobacterium tuberculosis detection in clinical samples. Journal of Clinical Microbiology 2011;49(10):3458‐62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moure R, Muñoz L, Torres M, Santin M, Martín R, Alcaide F. Rapid detection of Mycobacterium tuberculosis complex and rifampin resistance in smear‐negative clinical samples by use of an integrated real‐time PCR method. Journal of Clinical Microbiology 2011;49(3):1137‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rachow A, Zumla A, Heinrich N, Rojas‐Ponce G, Mtafya B, Reither K, et al. Rapid and accurate detection of Mycobacterium tuberculosis in sputum samples by Cepheid Xpert MTB/RIF assay‐‐a clinical validation study. PLoS One 2011;6(6):e20458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Safianowska A, Walkiewicz R, Nejman‐Gryz P, Grubek‐Jaworska H. [Two selected commercially based nucleic acid amplification tests for the diagnosis of tuberculosis]. Pneumonologia Alergologia Polska 2012;80(1):6‐12. [PubMed] [Google Scholar]
- Scott LE, McCarthy K, Gous N, Nduna M, Rie A, Sanne I, et al. Comparison of Xpert MTB/RIF with other nucleic acid technologies for diagnosing pulmonary tuberculosis in a high HIV prevalence setting: a prospective study. PLoS Medicine 2011;8(7):e1001061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teo J, Jureen R, Chiang D, Chan D, Lin R. Comparison of two nucleic acid amplification assays, the Xpert MTB/RIF and the amplified Mycobacterium tuberculosis Direct assay, for the detection of Mycobacterium tuberculosis in respiratory and non‐respiratory specimens. Journal of Clinical Microbiology 2011;49(10):3659‐62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theron G, Peter J, Zyl‐Smit R, Mishra H, Streicher E, Murray S, et al. Evaluation of the Xpert MTB/RIF assay for the diagnosis of pulmonary tuberculosis in a high HIV prevalence setting. American Journal of Respiratory Critical Care Medicine 2011;184(1):132‐40. [DOI] [PubMed] [Google Scholar]
- Rie A, Page‐Shipp L, Hanrahan CF, Schnippel K, Dansey H, Bassett J, et al. Point‐of‐care Xpert® MTB/RIF for smear‐negative tuberculosis suspects at a primary care clinic in South Africa. International Journal of Tuberculosis and Lung Disease 2013;17(3):368‐72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson DA, Basu I, Bower J, Freeman JT, Henderson G, Roberts SA. An evaluation of the Xpert MTB/RIF assay and detection of false‐positive rifampicin resistance in Mycobacterium tuberculosis. Diagnostic Microbiology and Infectious Disease 2012;74(2):207‐9. [DOI] [PubMed] [Google Scholar]
- Zeka AN, Tasbakan S, Cavusoglu C. Evaluation of the GeneXpert MTB/RIF assay for the rapid diagnosis of tuberculosis and detection of RIF‐resistance in pulmonary and extrapulmonary specimens. Journal of Clinical Micobiology 2011;49(12):4138‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies excluded from this review
- Alvarez‐Uria G, Azcona J M, Midde M, Naik PK, Reddy S, Reddy R. Rapid diagnosis of pulmonary and extrapulmonary tuberculosis in HIV‐infected patients. Comparison of LED fluorescent microscopy and the GeneXpert MTB/RIF assay in a district hospital in India. Tuberculosis Research and Treatment 2012;Article ID: 932862:1‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen AB, Lillebæk T, Bang D, Prahl J. [Treatment and diagnostics of tuberculosis: moving slowly forward]. Ugeskrift for Laeger 2011;173(12):897‐9. [PubMed] [Google Scholar]
- Armand S, Vanhuls P, Delcroix G, Courcol R, Lemaître N. Comparison of the Xpert MTB/RIF test with an IS6110‐TaqMan real‐time PCR assay for direct detection of Mycobacterium tuberculosis in respiratory and nonrespiratory specimens. Journal of Clinical Microbiology 2011;49(5):1772‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banada PP, Sivasubramani SK, Blakemore R, Boehme C, Perkins MD, Fennelly K, et al. Containment of bioaerosol infection risk by the Xpert MTB/RIF assay and its applicability to point‐of‐care settings. Journal of Clinical Microbiology 2010;48(10):3551‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bates M, O'Grady J, Maeurer M, Tembo J, Chilukutu L, Chabala C, et al. Assessment of the Xpert MTB/RIF assay for diagnosis of tuberculosis with gastric lavage aspirates in children in sub‐Saharan Africa: a prospective descriptive study. Lancet Infectious Diseases 2013;13(1):36‐42. [DOI] [PubMed] [Google Scholar]
- Blakemore R, Story E, Helb D, Kop J, Banada P, Owens MR, et al. Evaluation of the analytical performance of the Xpert MTB/RIF assay. Journal of Clinical Microbiology 2010;48(7):2495‐501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blakemore R, Nabeta P, Davidow A L, Vadwai V, Tahirli R, Munsamy V, et al. A multi‐site assessment of the quantitative capabilities of the Xpert® MTB/RIF assay. American Journal of Respiratory Critical Care Medicine 2011;184(9):1076‐84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Causse M, Ruiz P, Gutiérrez‐Aroca JB, Casal M. Comparison of two molecular methods for rapid diagnosis of extrapulmonary tuberculosis. Journal of Clinical Microbiology 2011;49(8):3065‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chegou NN, Hoek KG, Kriel M, Warren RM, Victor TC, Walzl G. Tuberculosis assays: Past, present and future. Expert Review of Anti‐Infective Therapy 2011;9(4):457‐69. [DOI] [PubMed] [Google Scholar]
- Clouse K, Page‐Shipp L, Dansey H, Moatlhodi B, Scott L, Bassett J, et al. Implementation of Xpert MTB/RIF for routine point‐of‐care diagnosis of tuberculosis at the primary care level. South African Medical Journal 2012;102(10):805‐7. [DOI] [PubMed] [Google Scholar]
- Cuevas LE. The urgent need for new diagnostics for symptomatic tuberculosis in children. Indian Journal of Pediatrics 2011;78(4):449‐55. [DOI] [PubMed] [Google Scholar]
- Dorjee K, Salvo F, Dierberg KL. Xpert® MTB/RIF diagnosed disseminated smear‐negative MDR‐TB in a sub‐district hospital in India. International Journal of Tuberculosis and Lung Disease 2012;16(11):1560‐1. [DOI] [PubMed] [Google Scholar]
- Dorman SE, Chihota VN, Lewis JJ, Shah M, Clark D, Grant AD, et al. Performance characteristics of the Cepheid Xpert MTB/RIF test in a tuberculosis prevalence survey. PLoS One 2012;7(8):e43307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dowdy DW, Cattamanchi A, Steingart KR, Pai M. Is scale‐up worth it? Challenges in economic analysis of diagnostic tests for tuberculosis. PLoS Medicine 2011;8(7):e1001063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans CA. GeneXpert‐‐a game‐changer for tuberculosis control?. PLoS Medicine 2011;8(7):e1001064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farga Cuesta, V. [New challenges in tuberculosis] [Nuevos desafíos en tuberculosis]. Revista chilena de enfermedades respiratorias 2011;27(2):161‐8. [Google Scholar]
- Fenner L, Boulle A, Egger M. In reply to 'Pre‐screening with GeneXpert® MTB/RIF may increase use of isoniazid preventive therapy in antiretroviral programmes'. International Journal of Tuberculosis and Lung Disease 2011;15(9):1273‐4. [DOI] [PubMed] [Google Scholar]
- Ferrara G, O'Grady J, Zumla A, Maeurer M. Xpert MTB/RIF test for tuberculosis. Lancet 2011;378(9790):482. [DOI] [PubMed] [Google Scholar]
- Foundation for Innovative Diagnostics. Performance of Xpert MTB/RIF Version G4 assay, Version and date: 1.0/30 Nov 2011, Project: 7210. Accessed at http://www.stoptb.org/wg/gli/assets/documents/map/findg4cartridge.pdf2011:1‐8.
- Friedrich SO, Groote‐Bidlingmaier F, Diacon AH. Xpert MTB/RIF assay for the diagnosis of pleural tuberculosis. Journal of Clinical Microbiology 2011;49(12):4341‐2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotuzzo E. Xpert MTB/RIF for diagnosis of pulmonary tuberculosis. Lancet Infectious Diseases 2011;11(11):802‐3. [DOI] [PubMed] [Google Scholar]
- Hesseling AC, Graham SM, Cuevas LE. Rapid molecular detection of tuberculosis. New England Journal of Medicine 2011;364(2):183‐4. [DOI] [PubMed] [Google Scholar]
- Hillemann D, Rüsch‐Gerdes S, Boehme C, Richter E. Rapid molecular detection of extrapulmonary tuberculosis by the automated GeneXpert MTB/RIF system. Journal of Clinical Microbiology 2011;49(4):1202‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoek KG, Rie A, Helden PD, Warren RM, Victor TC. Detecting drug‐resistant tuberculosis: the importance of rapid testing. Molecular Diagnosis & Therapy 2011;15(4):189‐94. [DOI] [PubMed] [Google Scholar]
- Ioannidis P, Papaventsis D, Nikolaou S, Karabela S, Konstantinidou E, Marinou I, et al. Tuberculosis resistance detection rate to the two main anti‐TB drugs, isoniazid and rifampicin, using molecular techniques: Experience of the Hellenic National Reference Center for Mycobacteria. Acta Microbiologica Hellenica 2010;55:175‐82. [Google Scholar]
- Kim SY, Kim H, Kim SY, Ra EK, Joo SI, Shin S, et al. The Xpert® MTB/RIF assay evaluation in South Korea, a country with an intermediate tuberculosis burden. International Journal of Tuberculosis and Lung Disease 2012;16(11):1471‐6. [DOI] [PubMed] [Google Scholar]
- Kirwan DE, Cárdenas MK, Gilman RH. Rapid implementation of new TB diagnostic tests: is it too soon for a global roll‐out of Xpert MTB/RIF?. American Journal of Tropical Medicine and Hygiene 2012;87(2):197‐201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawn SD, Nicol MP. Xpert® MTB/RIF assay: development, evaluation and implementation of a new rapid molecular diagnostic for tuberculosis and rifampicin resistance. Future Microbiology 2011;6(9):1067‐82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawn SD. Pre‐screening with GeneXpert® MTB/RIF may increase use of isoniazid preventive therapy in antiretroviral programmes. International Journal of Tuberculosis and Lung Disease 2011;15(9):1272‐3. [DOI] [PubMed] [Google Scholar]
- Lawn SD, Kerkhoff AD, Vogt M, Ghebrekristos Y, Whitelaw A, Wood R. Characteristics and early outcomes of patients with Xpert MTB/RIF‐negative pulmonary tuberculosis diagnosed during screening before antiretroviral therapy. Clinical Infectious Diseases 2012;54(8):1071‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawn SD, Kerkhoff AD, Vogt, M, Wood R. High diagnostic yield of tuberculosis from screening urine samples from HIV‐infected patients with advanced immunodeficiency using the Xpert MTB/RIF assay. Journal of Acquired Immune Deficiency Syndromes 2012;60(3):289‐94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawn SD, Kerkhoff, AD, Wood R. Reply to Theron et al "Characteristics of Xpert MTB/RIF‐negative patients with pulmonary tuberculosis". Clinical Infectious Diseases 2012;55(3):473‐4. [DOI] [PubMed] [Google Scholar]
- Lawn SD, Kerkhoff AD, Vogt M, Wood R. Clinical significance of lipoarabinomannan detection in urine using a low‐cost point‐of‐care diagnostic assay for HIV‐associated tuberculosis. AIDS 2012;26(13):1635‐43. [DOI] [PubMed] [Google Scholar]
- Lawn SD, Kerkhoff AD, Vogt M, Wood R. Diagnostic accuracy of a low‐cost, urine antigen, point‐of‐care screening assay for HIV‐associated pulmonary tuberculosis before antiretroviral therapy: a descriptive study. Lancet Infect Diseases 2012;12(3):201‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ligthelm LJ, Nicol MP, Hoek KG, Jacobson R, Helden PD, Marais BJ, et al. Xpert MTB/RIF for rapid diagnosis of tuberculous lymphadenitis from fine‐needle‐aspiration biopsy specimens. Journal of Clinical Microbiology 2011;49(11):3967‐70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melzer M. An automated molecular test for Mycobacterium tuberculosis and resistance to rifampin (Xpert MTB/RIF) is sensitive and can be carried out in less than 2 h. Evidence Based Medicine 2011;16(1):19. [DOI] [PubMed] [Google Scholar]
- Miotto P, Bigoni S, Migliori GB, Matteelli A, Cirillo DM. Early tuberculosis treatment monitoring by Xpert(R) MTB/RIF. European Respiratory Journal2012; Vol. 39, issue 5:1269‐71. [DOI] [PubMed]
- Morris K. Xpert TB diagnostic highlights gap in point‐of‐care pipeline. Lancet Infectious Diseases 2010;10(11):742‐3. [DOI] [PubMed] [Google Scholar]
- Morris K. The new face of tuberculosis diagnosis. Lancet Infectious Diseases 2011;11(10):736‐7. [Google Scholar]
- Moure R, Martin, R, Alcaide F. Effectiveness of an integrated real‐time PCR method for detection of the Mycobacterium tuberculosis complex in smear‐negative extrapulmonary samples in an area of low tuberculosis prevalence. Journal of Clinical Microbiology 2012;50(2):513‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muñoz L, Moure R, Porta N, Gonzalez L, Guerra R, Alcaide F, et al. GeneXpert® for smear‐negative pulmonary tuberculosis: does it play a role in low‐burden countries?. Diagnostic Microbiology Infectious Disease 2013;75(3):325‐6. [DOI] [PubMed] [Google Scholar]
- Narasimooloo R, Ross A. Delay in commencing treatment for MDR TB at a specialised TB treatment centre in KwaZulu‐Natal. South African Medical Journal 2012;102(6 Pt 2):360‐2. [DOI] [PubMed] [Google Scholar]
- Nhu NT, Ha DT, Anh ND, Thu DD, Duong TN, Quang ND, et al. Evaluation of Xpert MTB/RIF and MODS assay for the diagnosis of pediatric tuberculosis. BMC Infectious Diseases 2013;13:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicol MP, Workman L, Isaacs W, Munro J, Black F, Eley B, et al. Accuracy of the Xpert MTB/RIF test for the diagnosis of pulmonary tuberculosis in children admitted to hospital in Cape Town, South Africa: a descriptive study. Lancet Infectious Diseases 2011;11(11):819‐24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ntinginya EN. Squire SB, Millington KA, Mtafya B, Saathoff E, Heinrich N, et al. Performance of the Xpert® MTB/RIF assay in an active case‐finding strategy: a pilot study from Tanzania. International Journal of Tuberculosis and Lung Disease 2012;16(11):1468‐70. [DOI] [PubMed] [Google Scholar]
- O'Grady J, Bates M, Chilukutu L, Mzyece J, Cheelo B, Chilufya M, et al. Evaluation of the Xpert MTB/RIF assay at a tertiary care referral hospital in a setting where tuberculosis and HIV infection are highly endemic. Clinical Infectious Diseases 2012;55(9):1171‐8. [DOI] [PubMed] [Google Scholar]
- Perkins MD, Alland D, Nicol MP, Nabeta P, Boehme CC. Xpert MTB/RIF test for tuberculosis. Authors' reply. Lancet 2011;378(9790):482‐3. 21821179 [Google Scholar]
- Peter JG, Theron G, Muchinga TE, Govender U, Dheda K. The diagnostic accuracy of urine‐based Xpert MTB/RIF in HIV‐infected hospitalized patients who are smear‐negative or sputum scarce. PLoS One 2012;7(7):e39966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters D, Theron G, Peter J, Dheda K. Should Xpert® MTB/RIF be rolled out in low‐income countries?. International Journal of Tuberculosis and Lung Disease 2012;16(5):702‐3. [DOI] [PubMed] [Google Scholar]
- Rachow A, Clowes P, Saathoff E, Mtafya B, Michael E, Ntinginya EN, et al. Increased and expedited case detection by Xpert MTB/RIF assay in childhood tuberculosis: a prospective cohort study. Clinical Infectious Diseases 2012;54(10):1388‐96. [DOI] [PubMed] [Google Scholar]
- Salvo F, Sadutshang TD, Migliori GB, Zumla A, Cirillo DM. Xpert MTB/RIF test for tuberculosis. Lancet 2011;378(9790):481‐2. [DOI] [PubMed] [Google Scholar]
- Scott LE, Gous N, Cunningham BE, Kana BD, Perovic O, Erasmus L, et al. Dried culture spots for Xpert MTB/RIF external quality assessment: results of a phase 1 pilot study in South Africa. Journal of Clinical Microbiology 2012;49(12):4356‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swaminathan S, Rekha VV. Antigen detection as a point‐of‐care test for TB: the case of lipoarabinomannan. Future Microbiology 2012;7(5):559‐64. [DOI] [PubMed] [Google Scholar]
- Tansey B. Cepheid unveils fast TB test to aid developing countries. AIDS Reader 2009;19(6):229. [PubMed] [Google Scholar]
- Taylor N, Gaur RL, Baron EJ, Banaei N. Can a simple flotation method lower the limit of detection of Mycobacterium tuberculosis in extrapulmonary samples analyzed by the GeneXpert MTB/RIF assay?. Journal of Clinical Microbiology 2012;50(7):2272‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theron G, Peter J, Dheda K. Xpert MTB/RIF test for tuberculosis. Lancet 2011;378(9790):481. [DOI] [PubMed] [Google Scholar]
- Theron G, Peter J, Lenders L, Zyl‐Smit R, Meldau R, Govender U, et al. Correlation of mycobacterium tuberculosis specific and non‐specific quantitative Th1 T‐cell responses with bacillary load in a high burden setting. PLoS One 2012;7(5):e37436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theron G, Pinto L, Peter J, Mishra HK, Mishra HK, Zyl‐Smit R, et al. The use of an automated quantitative polymerase chain reaction (Xpert MTB/RIF) to predict the sputum smear status of tuberculosis patients. Clinical Infectious Diseases 2012;54(3):384‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theron G, Pooran A. Peter J, Zyl‐Smit R, Kumar Mishra H, Meldau R, et al. Do adjunct tuberculosis tests, when combined with Xpert MTB/RIF, improve accuracy and the cost of diagnosis in a resource‐poor setting?. European Respiratory Journal 2012;40(1):161‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tortoli E, Russo C, Piersimoni C, Mazzola E, Dal Monte P, Pascarella M, et al. Clinical validation of Xpert MTB/RIF for the diagnosis of extrapulmonary tuberculosis. European Respiratory Journal 2012;40(2):442‐7. [DOI] [PubMed] [Google Scholar]
- Trébucq A, Enarson DA, Chiang CY, Deun A, Harries AD, Boillot F, et al. Xpert® MTB/RIF for national tuberculosis programmes in low‐income countries: when, where and how?. International Journal of Tuberculosis and Lung Disease 2012;15(12):1567‐72. [DOI] [PubMed] [Google Scholar]
- Trebucq A, Harries AD, Rieder HL. Author reply: Should Xpert MTB/RIF be rolled out in low‐income countries?. International Journal of Tuberculosis and Lung Disease 2012;16(5):703‐4. [DOI] [PubMed] [Google Scholar]
- Vadwai V, Boehme C, Nabeta P, Shetty A, Alland D, Rodrigues C. Xpert MTB/RIF: a new pillar in diagnosis of extrapulmonary tuberculosis?. Journal of Clinical Microbiology 2011;49(7):2540‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vadwai V, Boehme C, Nabeta P, Shetty A, Rodrigues C. Need to confirm isoniazid susceptibility in Xpert MTB/RIF rifampin susceptible cases. Indian Journal of Medical Research 2012;135(4):560‐1. [PMC free article] [PubMed] [Google Scholar]
- Rie A, Page‐Shipp L, Scott L, Sanne I, Stevens W. Xpert(®) MTB/RIF for point‐of‐care diagnosis of TB in high‐HIV burden, resource‐limited countries: hype or hope?. Expert Review of Molecular Diagnostics 2010;10(7):937‐46. [DOI] [PubMed] [Google Scholar]
- Rie A. A single Xpert MTB/RIF test of sputum for diagnosis of tuberculosis and multidrug resistance shows high sensitivity and specificity and reduces diagnosis and treatment delays. Evidence Based Medicine 2011;16(6):174‐5. [DOI] [PubMed] [Google Scholar]
- Zyl‐Smit RN, Binder A, Meldau R, Mishra H, Semple PL, Theron G, et al. Comparison of quantitative techniques including Xpert MTB/RIF to evaluate mycobacterial burden. PLoS One 2011;6(12):e28815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walters E, Gie RP, Hesseling AC, Friedrich SO, Diacon AH. Rapid diagnosis of pediatric intrathoracic tuberculosis from stool samples using the Xpert MTB/RIF Assay: a pilot study. Pediatric Infectious Disease Journal 2012;31(12):1316. [DOI] [PubMed] [Google Scholar]
- Williamson DA, Roberts SA, Bower JE, Vaughan R, Newton S, Lowe O, et al. Clinical failures associated with rpoB mutations in phenotypically occult multidrug‐resistant Mycobacterium tuberculosis. International Journal of Tuberculosis and Lung Disease 2012;16(2):216‐20. [DOI] [PubMed] [Google Scholar]
- Wood R, Racow K, Bekker LG, Middelkoop K, Vogt M, Kreiswirth BN, et al. Lipoarabinomannan in urine during tuberculosis treatment: association with host and pathogen factors and mycobacteriuria. BMC Infectious Diseases 2012;12:47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon C, Cattamanchi A, Davis JL, Worodria W, Boon S, Kalema N, et al. Impact of Xpert MTB/RIF testing on tuberculosis management and outcomes in hospitalized patients in Uganda. PLoS One 2012;7(11):e48599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zar HJ, Workman L, Isaacs W, Munro J, Black F, Eley B, et al. Rapid molecular diagnosis of pulmonary tuberculosis in children using nasopharyngeal specimens. Clinical Infectious Diseases 2012;55(8):1088‐95. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to ongoing studies
- GeneXpert MTB/RIF, a new tool for the diagnosis of pulmonary tuberculosis in two municipalities in Brazil. Ongoing studyJanuary 2012.
- Evaluation of Xpert MTB/RIF assay for the rapid identification of TB and TB rifampin resistance in HIV‐infected and HIV‐uninfected patients with presumed pulmonary tuberculosis. Ongoing study 24 April 2012.
- A randomised control trial of sputum induction, and new and emerging technologies in a high HIV prevalence primary care setting. Ongoing studyAugust 2009.
Additional references
- Abimbola TO, Marston BJ, Date AA, Blandford JM, Sangrujee N, Wiktor SZ. Cost‐effectiveness of tuberculosis diagnostic strategies to reduce early mortality among persons with advanced HIV infection initiating antiretroviral therapy. Journal of Acquired Immune Deficiency Syndromes 2012;60(1):e1‐7. [DOI] [PubMed] [Google Scholar]
- Andrews JR, Lawn SD, Rusu C, Wood R, Noubary F, Bender MA, et al. The cost‐effectiveness of routine tuberculosis screening with Xpert MTB/RIF prior to initiation of antiretroviral therapy: a model‐based analysis. AIDS 2012;26(8):987‐95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nabeta P, Hillemann D, Nicol MP, Shenai S, Krapp F, et al. Rapid molecular detection of tuberculosis and rifampin resistance. New England Journal of Medicine 2010;363(11):1005‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehme CC, Nicol MP, Nabeta P, Michael JS, Gotuzzo E, Tahirli R, et al. Feasibility, diagnostic accuracy, and effectiveness of decentralised use of the Xpert MTB/RIF test for diagnosis of tuberculosis and multidrug resistance: a multicentre implementation study. Lancet 2011;377(9776):1495‐505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bossuyt PM, Reitsma JB, Bruns DE, Gatsonis CA, Glasziou PP, Irwig LM, et al. The STARD statement for reporting studies of diagnostic accuracy: explanation and elaboration. Clinical Chemistry 2003;49(1):7‐18. [DOI] [PubMed] [Google Scholar]
- Bossuyt PM, Leeflang MM. Chapter 6: Developing Criteria for Including Studies. Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy Version 0.4 [updated September 2008]. The Cochrane Collaboration, 2008. [Google Scholar]
- Boyles TH, Hughes J, Cox V, Burton R, Meintjes G, Mendelson M. False‐positive Xpert® MTB/RIF assays in previously treated patients: need for caution in interpreting results. The International Journal of Tuberculosis and Lung Disease 18(7):876‐8. [DOI] [PubMed] [Google Scholar]
- Buzoianu M, Kadane JB. Adjusting for verification bias in diagnostic test evaluation: a Bayesian approach. Statististics in Medicine 2008;27(13):2453‐73. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Updated guidelines for the use of nucleic acid amplification tests in the diagnosis of tuberculosis. Morbidity and Mortality Weekly Report 2009;58(1):7‐10. [PubMed] [Google Scholar]
- Cepheid. Brochure: Xpert®MTB/RIF. Two‐hour detection of MTB and resistance to rifampicin. http://www.cepheid.com/media/files/eu/brochures/XpertMTB_Broch_R9_EU.pdf. Sunnyvale, Accessed 17 June 2012:1‐4.
- Chaisson RE, Nuermberger EL. Confronting multidrug‐resistant tuberculosis. New England Journal of Medicine 2012;366(23):2223‐4. [DOI] [PubMed] [Google Scholar]
- Chang K, Lu W, Wang J, Zhang K, Jia S, Li F, et al. Rapid and effective diagnosis of tuberculosis and rifampicin resistance with Xpert MTB/RIF assay: a meta‐analysis. Journal of Infection 2012;64(6):580‐8. [DOI] [PubMed] [Google Scholar]
- Chu H, Chen S, Louis TA. Random effects models in a meta‐analysis of the accuracy of two diagnostic tests without a gold standard. Journal of the American Statistical Association 2009;104(486):512‐23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denkinger CM, Nicolau I, Ramsay A, Chedore P, Pai M. Are peripheral microscopy centres ready for next generation molecular tuberculosis diagnostics?. European Respiratory Journal 2013;42(2):544‐7. [DOI] [PubMed] [Google Scholar]
- Drobniewski F, Nikolayevskyy V, Balabanova Y, Bang D, Papaventsis D. Diagnosis of tuberculosis and drug resistance: what can new tools bring us?. International Journal of Tuberculosis and Lung Disease 2012;16(7):860‐70. [DOI] [PubMed] [Google Scholar]
- Flores LL, Pai M, Colford JM Jr, Riley LW. In‐house nucleic acid amplification tests for the detection of Mycobacterium tuberculosis in sputum specimens: meta‐analysis and meta‐regression. BMC Microbiology 2005;5:55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Getahun H, Harrington M, O'Brien R, Nunn P. Diagnosis of smear‐negative pulmonary tuberculosis in people with HIV infection or AIDS in resource‐constrained settings: informing urgent policy changes. Lancet 2007;369(9578):2042‐9. [DOI] [PubMed] [Google Scholar]
- Global Laboratory Initiative. A roadmap for ensuring quality tuberculosis diagnostics services within national laboratory strategic plans. World Health Organization. Geneva: World Health Organization, 2010.
- Gopinath K, Singh S. Non‐tuberculous mycobacteria in TB‐endemic countries: Are we neglecting the danger?. PLoS Neglected Tropical Diseases 2010;4(4):e615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greco S, Girardi E, Navarra A, Saltini C. Current evidence on diagnostic accuracy of commercially based nucleic acid amplification tests for the diagnosis of pulmonary tuberculosis. Thorax 2006;61(9):783‐90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadgu A, Dendukuri N, Hilden J. Evaluation of nucleic acid amplification tests in the absence of a perfect gold‐standard test: a review of the statistical and epidemiologic issues. Epidemiology 2005;16(5):604–12. [DOI] [PubMed] [Google Scholar]
- Harries A. How does the diagnosis of tuberculosis in persons infected with HIV differ from diagnosis in persons not infected with HIV?. In: Frieden T editor(s). Toman’s tuberculosis: case detection, treatment, and monitoring – questions and answers. WHO/HTM/TB/2004.334. Geneva: World Health Organization, 2004:80‐3. [Google Scholar]
- Hartmann G, Honikel KO, Knüsel F, Nüesch J. The specific inhibition of the DNA‐directed RNA synthesis by rifamycin. Biochimica et Biophysica Acta 1967;145(3):843‐4. [DOI] [PubMed] [Google Scholar]
- Tuberculosis coalition for technical assistance. International Standards for Tuberculosis Care (ISTC). 2nd Edition. The Hague: Tuberculosis coalition for technical assistance, 2009. [Google Scholar]
- Juni P, Witschi A, Bloch R, Egger M. The hazards of scoring the quality of clinical trials for meta‐analysis. Journal of the American Medical Association 1999;282(11):1054‐60. [DOI] [PubMed] [Google Scholar]
- Ling DI, Zwerling AA, Pai M. GenoType MTBDR assays for the diagnosis of multidrug‐resistant tuberculosis: a meta‐analysis. European Respiratory Journal 2008;32(5):1165‐74. [DOI] [PubMed] [Google Scholar]
- Ling DI, Flores LL, Riley LW, Pai M. Commercial nucleic‐acid amplification tests for diagnosis of pulmonary tuberculosis in respiratory specimens: meta‐analysis and meta‐regression. PLoS One 2008;3(2):e1536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lunn DJ, Thomas A, Best N, Spiegelhalter D. WinBUGS ‐ a Bayesian modelling framework: concepts, structure, and extensibility. Statistics and Computing 2000;10:325‐37. [Google Scholar]
- Macaskill P, Gatsonis C, Deeks JJ, Harbord RM, Takwoingi Y. Analysing and Presenting Results. In: Deeks JJ, Bossuyt PM, C Gatsonis editor(s). Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy. Version 0.9.0. The Cochrane Collaboration, 2010. [Google Scholar]
- Meyer‐Rath G, Schnippel K, Long L, MacLeod W, Sanne I, Stevens W, et al. The impact and cost of scaling up GeneXpert MTB/RIF in South Africa. PLoS One 2012;7(5):e36966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nahid P, Kim PS, Evans CA, Alland D, Barer M, Diefenbach J, et al. Clinical research and development of tuberculosis diagnostics: moving from silos to synergy. Journal of Infectious Disease 2012;205(Suppl 2):S159‐68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantoja A, Fitzpatrick C, Vassall A, Weyer K, Floyd K. Xpert MTB/RIF for diagnosis of tuberculosis and drug‐resistant tuberculosis: a cost and affordability analysis. European Respiratory Journal 2013;42(3):708‐20. [DOI] [PubMed] [Google Scholar]
- Perkins MD, Cunningham J. Facing the crisis: improving the diagnosis of tuberculosis in the HIV era. Journal of Infectious Diseases 2007;196(Suppl 1):S15‐27. [DOI] [PubMed] [Google Scholar]
- R Development Core Team. R: A language and environment for statistical computing. Version Version 2.15.1. Vienna: R Foundation for Statistical Computing, 2008.
- Raizada N, Sachdeva KS, Sreenivas A, Vadera B, Parmar M, Kulsange S, et al. Feasibility of decentralised deployment of Xpert MTB/RIF test In resource constraint settings in India data (as supplied 11 October 2013). Data on file.
- Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH. Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. Journal of Clinical Epidemiology 2005;58(10):982‐90. [DOI] [PubMed] [Google Scholar]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.0. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2008.
- Schnippel K, Meyer‐Rath G, Long L, MacLeod W, Sanne I, et al. Scaling up Xpert MTB/RIF technology: the costs of laboratory‐ vs. clinic‐based roll‐out in South Africa. Tropical Medicine and International Health 2012;17(9):1142‐51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Small PM, Pai M. Tuberculosis diagnosis ‐ time for a game change. New England Journal of Medicine 2011;363(111):1070‐1. [DOI] [PubMed] [Google Scholar]
- StataCorp. StataCorp LP. Stata Statistical Software. Version 12. College Station, Texas: StataCorp. StataCorp LP, 2011.
- Steingart KR, Henry M, Ng V, Hopewell PC, Ramsay A, Cunningham J, et al. Fluorescence versus conventional sputum smear microscopy for tuberculosis: a systematic review. Lancet Infectious Diseases 2006;6(9):570‐81. [DOI] [PubMed] [Google Scholar]
- Steingart KR, Ng V, Henry M, Hopewell PC, Ramsay A, Cunningham J, et al. Sputum processing methods to improve the sensitivity of smear microscopy for tuberculosis: a systematic review. Lancet Infectious Diseases 2006;6(10):664‐74. [DOI] [PubMed] [Google Scholar]
- Steingart KR, Schiller I, Dendukuri N, Lalli M, Houben R, Churchyard G, et al. In reply to 'False‐positive Xpert(®) MTB/RIF assays in previously treated patients'. The International Journal of Tuberculosis and Lung Disease 19(3):366‐7. [DOI] [PubMed] [Google Scholar]
- Telenti A, Imboden P, Marchesi F, Lowrie D, Cole S, Colston MJ, et al. Detection of rifampicin‐resistance mutations in Mycobacterium tuberculosis. Lancet 1993;341(8846):647‐50. [DOI] [PubMed] [Google Scholar]
- The Gates Foundation. Public‐private partnership announces immediate 40 percent cost reduction for rapid TB test. http://www.gatesfoundation.org/press‐releases/Pages/public‐private‐partnership‐40‐percent‐reduction‐TB‐test.aspx 6 August 2012.
- Theron G, Zijenah L, Chanda D, Clowes P, Andrea Rachow A, Lesosky M, et al. Feasibility, accuracy, and clinical impact of point‐of‐care Xpert MTB/RIF testing for tuberculosis in primary‐care settings in Africa: a multicentre, randomised controlled trial. Lancet 2013 October 25 [Epub ahead of print]. [DOI: 10.1016/S0140-6736(13)62073-5] [DOI] [PubMed] [Google Scholar]
- Toman K. How many bacilli are present in a sputum specimen found positive by smear microscopy?. In: Frieden T editor(s). Toman’s tuberculosis: case detection, treatment, and monitoring – questions and answers. WHO/HTM/TB/2004.334. 2nd Edition. Geneva: World Health Organization, 2004:11‐13. [Google Scholar]
- Toman K. How reliable is smear microscopy?. In: Frieden T editor(s). Toman’s tuberculosis: case detection, treatment, and monitoring – questions and answers. WHO/HTM/TB/2004.334. 2nd Edition. Geneva: World Health Organization, 2004:14‐22. [Google Scholar]
- Trébucq A, Enarson DA, Chiang CY, Deun A, Harries AD, Boillot F, et al. Xpert® MTB/RIF for national tuberculosis programmes in low‐income countries: when, where and how?. International Journal of Tuberculosis and Lung Disease 2011;15(12):1567‐72. [DOI] [PubMed] [Google Scholar]
- UNITAID. UNITAID approves US$30 million for innovative project to roll out ground‐breaking tuberculosis test at reduced cost. http://www.unitaid.eu/resources/news/releases/943‐unitaid‐approves‐us‐30‐million‐for‐innovative‐project‐to‐roll‐out‐ground‐breaking‐tuberculosis‐test‐at‐reduced‐cost 13 June 2012.
- Boyle D, Pai M. UNITAID: Tuberculosis diagnostics technology and market landscape, second edition. Geneva: WHO, 2013. [Google Scholar]
- Deun A, Barrera L, Bastian I, Fattorini L, Hoffmann H, Kam KM, et al. Mycobacterium tuberculosis strains with highly discordant rifampin susceptibility test results. Journal of Clinical Microbiology 2009;47(11):3501‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deun A, Aung KJ, Bola V, Lebeke R, Hossain MA, Rijk WB, et al. Rifampin drug resistance tests for tuberculosis: challenging the gold standard. Journal of Clinical Microbiology 2013;51(8):2633‐40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vassall A, Kampen S, Sohn H, Michael JS, John KR, Boon S, et al. Rapid diagnosis of tuberculosis with the Xpert MTB/RIF assay in high burden countries: a cost‐effectiveness analysis. PLoS Medicine 2011;8(11):e1001120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whiting P, Harbord R, Kleijnen J. No role for quality scores in systematic reviews of diagnostic accuracy studies. BMC Medical Research Methodology 2005;5:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, et al. QUADAS‐2: a revised tool for the quality assessment of diagnostic accuracy studies. Annals of Internal Medicine 2011;155(8):529‐36. [DOI] [PubMed] [Google Scholar]
- World Health Organization. Anti‐tuberculosis drug resistance in the world: fourth global report. WHO/HTM/TB/2008.394. Geneva: World Health Organization, 2008. [Google Scholar]
- World Health Organization. Global tuberculosis report 2013. WHO/HTM/TB/2013.11. Geneva: World Health Organization, 2013. [Google Scholar]
- World Health Organization. Multidrug and extensively drug‐resistant TB (M/XDR‐TB): 2010 global report on surveillance and response. WHO/HTM/TB/2010.3. Geneva: World Health Organization, 2010. [Google Scholar]
- World Health Organization. Policy guidance on drug‐susceptibility testing (DST) of second‐line antituberculosis drugs. WHO/HTM/TB/2008.392. Geneva: World Health Organization, 2008. [PubMed] [Google Scholar]
- World Health Organization. Policy framework for implementing new tuberculosis diagnostics. Geneva: World Health Organization, 2011. [Google Scholar]
- World Health Organization. Molecular line probe assays for rapid screening of patients at risk of multidrug‐resistant tuberculosis (MDR‐TB). Geneva: World Health Organization, 2008:1‐9. [Google Scholar]
- World Health Organization. Definition of a new sputum smear‐positive TB case. http://www.who.int/tb/laboratory/policy_sputum_smearpositive_tb_case/en/index.html2007.
- World Health Organization. Policy statement: automated real‐time nucleic acid amplification technology for rapid and simultaneous detection of tuberculosis and rifampicin resistance: Xpert MTB/RIF system. WHO/HTM/TB/2011.4. Geneva: World Health Organization, 2011. [PubMed] [Google Scholar]
- World Health Organization. Rapid implementation of the Xpert MTB/RIF diagnostic test. Technical and operational ‘How‐to’. Practical considerations. WHO/HTM/TB/2011.2. Geneva: World Health Organization, 2011. [Google Scholar]
- World Health Organization. Checklist of prerequisites to country implementation of Xpert MTB/RIF and key action points at country level. WHO/HTM/TB/2011.12. Geneva: World Health Organization, 2011. [Google Scholar]
- World Health Organization. Automated real‐time nucleic acid amplification technology for rapid and simultaneousdetection of tuberculosis and rifampicin resistance: Xpert MTB/RIF system for the diagnosis of pulmonary and extrapulmonary TB in adults and children: policy update WHO/HTM/TB/2013.14. Automated real‐time nucleic acid amplification technology for rapid and simultaneous detection of tuberculosis and rifampicin resistance: Xpert MTB/RIF system for the diagnosis of pulmonary and extrapulmonary TB in adults and children: policy update. WHO/HTM/TB/2013.14. Geneva: World Health Organization, 2013. [PubMed] [Google Scholar]
- World Bank. World Bank List of Economies http://www.healthsystemsglobal.org/Portals/0/files/World_bank_list_july2012.pdf. Washington (District of Columbia): World Bank, 2012. [Google Scholar]
- Wright A, Zignol M, Deun A, Falzon D, Gerdes SR, Feldman K, et al. Epidemiology of antituberculosis drug resistance 2002‐07: an updated analysis of the Global Project on Anti‐Tuberculosis Drug Resistance Surveillance. Lancet 2009;373(9678):1861‐73. [DOI] [PubMed] [Google Scholar]
- Zignol M, Gemert W, Falzon D, Sismanidis C, Glaziou P, Floyd K, et al. Surveillance of anti‐tuberculosis drug resistance in the world: an updated analysis, 2007‐2010. Bulletin of the World Health Organization 2012;90(2):111‐119D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zumla A, Abubakar I, Raviglione M, Hoelscher M, Ditiu L, McHugh TD, et al. Drug‐resistant tuberculosis‐‐current dilemmas, unanswered questions, challenges, and priority needs. Journal of Infectious Diseases 2012;205(Suppl 2):S228‐40. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
- Steingart KR, Sohn H, Schiller I, Kloda LA, Boehme CC, Pai M, Dendukuri N. Xpert® MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database of Systematic Reviews 2013, Issue 1. [DOI: 10.1002/14651858.CD009593.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]


