Abstract
The inflammasome is a caspase-1 containing complex that activates the proinflammatory cytokines IL-1β and IL-18 and results in the proinflammatory cell death known as pyroptosis. Numerous recent publications have highlighted the importance of inflammasome activation in the control of virus infection. Inflammasome activation during viral infection is dependent on a variety of upstream receptors including the NOD-Like receptor, RIG-I-Like receptor and AIM2-Like receptor families. Various receptors also function in inflammasome activation in different cellular compartments, including the cytoplasm and the nucleus. The effectiveness of inflammasomes at suppressing virus replication is highlighted by the prevalence and diversity of virus encoded inflammasome inhibitors. Also, the host has a myriad of regulatory mechanisms in place to prevent unwanted inflammasome activation and overt inflammation. Finally, recent reports begin to suggest that inflammasome activation and inflammasome modulation may have important clinical applications. Herein, we highlight recent advances and discuss potential future directions toward understanding the role of inflammasomes during virus infection.
Keywords: NLRP1, NLRP3, RIG-I, MAVS, AIM2, ASC, IFI16, Caspase-1, inflammasome, virus, IL-1β, IL-18, antiviral, cancer, oncogenic, tumor, immunopathology, inflammation
Introduction
Innate immunity to virus infection involves recognition of pathogen-associated molecular patterns (PAMPs), or danger associated molecular patterns (DAMPs), by pattern recognition receptors (PRRs). During virus infection, viral DNA and RNA are detected by a myriad of PRRs whose activation elicits antiviral responses and inflammation [1,2]. To mediate the innate immune response, binding of RNA or DNA to PRRs results in activation of NF-κB, IRFs, AP-1 and other transcription factors followed by pro-inflammatory gene expression. These include type I and type III interferons, interleukin (IL)-6, and TNF-α that immediately affect an antiviral state and initiate inflammation. In addition, pro-IL-1β and pro-IL-18 are also induced by PRR activation but are inactive. Pro-IL-1β and pro-IL-18 are processed into their functional forms and released from cells through multi-protein complexes known as inflammasomes. Inflammasomes come in many forms and are mainly activated by nucleotide binding and oligomerization domain (NOD)-like receptors (NLRs). Following recognition of PAMPs [3–5] or DAMPs [6–8], the active NLR oligomerizes and initiates formation of the inflammasome complex that usually consists of the adaptor apoptosis-associated speck-like protein containing a CARD (ASC) and the CARD domain containing pro-caspase-1. Inflammasome complex assembly results in caspase-1 activation and cleavage of pro-IL-1β and pro-IL-18, leading to their activation and release from cells.
In addition to NLRs, certain nucleic acid sensors are also capable of forming inflammasomes. The AIM2-like receptors, (ALRs, also referred to as the PYHIN family of proteins) are characterized by an N-terminal PYRIN domain (PYD) that is involved in homotypic protein-protein interactions and a C-terminal HIN-200 domain that binds to DNA [9]. Absent in melanoma 2 (AIM2) (in mice and humans) and gamma-interferon inducible-protein 16 (IFI16) in humans (p204 in mice) have demonstrated importance in inflammasome formation in response to DNA viruses [10–12]. Furthermore, the RNA sensor retinoic acid-inducible gene-I (RIG-I) appears to activate the inflammasome [13]. In this review, we highlight recent advances in the function of inflammasomes during viral infection, including viral evasion strategies and host regulatory mechanisms, and discuss the clinical relevance of these discoveries.
ALR inflammasomes
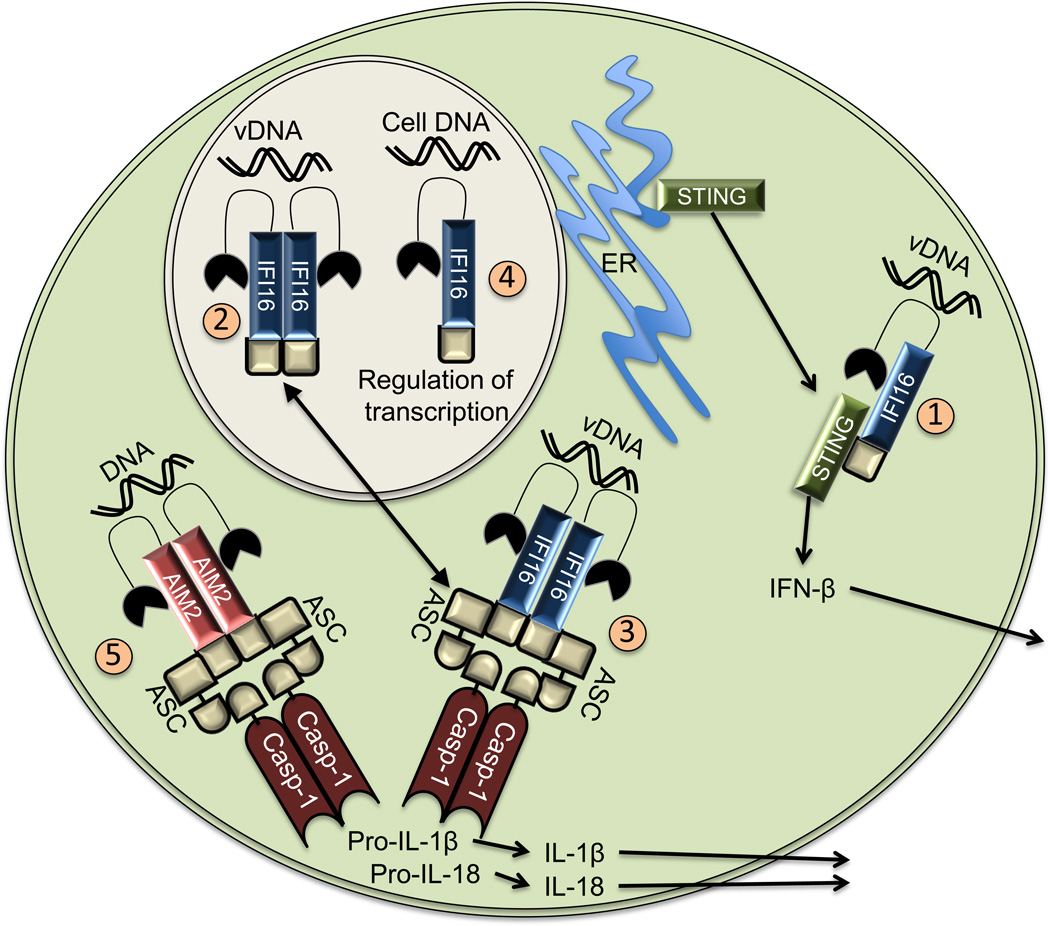
The ALR family consists of five members in humans and thirteen in mice and are induced in response to interferon signaling [14]. AIM2 detects DNA from a variety of sources including DNA virus infection in the cytoplasm [9,10,15,16] (Figure 1 and Table 1). In contrast, IFI16 can detect viral DNA in both the cytoplasm and the nucleus (Figure 1) [12,17,18]. Initial reports showed that infection with vaccinia virus, an orthopoxvirus that replicates in the cytoplasm, triggers inflammasome activation in an AIM2 dependent manner [10,11]. Murine cytomegalovirus (MCMV, also known as Murid herpesvirus 1, MuHV-1) is detected by AIM2 despite the fact that the genome is maintained and replicated in the nucleus. Infection of Aim2−/− mice with MCMV results in impaired IL-18 production, impaired IFN-γ production by NK cells, and higher virus titers in the blood [10]. In this same study, the authors also infected cells with herpes simplex virus-1 (HSV-1), but could not detect any role for AIM2. It is currently unclear why the betaherpesvirus MCMV is detected by AIM2 but not the alphaherpesvirus HSV-1 despite the fact that they both replicate their genomes in the nucleus of infected cells. Instead, HSV-1 activates the IFI16 dependent inflammasome, as do many other herpesvirus family members like bovine herpesvirus 1 (BoHV-1), Kaposi’s sarcoma-associated herpes virus (KSHV), and Epstein-Barr virus (EBV) [12,19–21] (Table 1). Also, activation of both the IFI16 and NLRP3 inflammasomes has been observed with HSV-1 [20,22] and AIM2, NLRP1 and NLRP3 for vaccinia virus [23,24], but the relative contribution or importance of multiple inflammasomes has not been addressed.
Figure 1. ALR signaling pathways.
AIM2-like receptors (ALRs) have roles in both inflammasome and inflammasome-independent signaling during virus infection. (1) In particular, IFI16 regulates interferon responses through the adaptor STING, which translocates from the endoplasmic reticulum (ER) to the cytosol in response to IFI16 activation. In the nucleus, IFI16 can bind viral DNA (vDNA) and activate the inflammasome in the nucleus (2) or peri-nuclear space (3). (4) IFI16 appears to both positively and negatively regulate gene expression through DNA binding and affecting histone modifications and chromatin packaging. (5) In the cytosol, AIM2 can recognize DNA of cellular or pathogen origin and activate the inflammasome. The only requirement for AIM2 recognition of DNA appears to be cytosolic localization. However, IFI16 binding of different forms of DNA must affect the function of IFI16 in an undefined fashion.
Table 1.
Virus recognition by ALRs.
| Virus | ALR | Biological Consequence | Reference(s) |
|---|---|---|---|
| HBV | AIM2 | Caspase-1 and IL-1β expression correlate with virus titers and renal damage in humans. | [26–28] |
| KSHV | IFI16 | Activates IFI16 dependent inflammasome in vitro. | [12,33] |
| EBV | IFI16, AIM2 | Inflammasome activation in vitro; IL-1β promoted tumor growth in vitro, but inflammasome activation promotes tumor regression in vivo via PMN infiltration. | [19,32] |
| HPV | IFI16, AIM2 | Increased expression of AIM2, IFI16 and IL-1β in infected tissue, associated with reduced tumor cell proliferation. | [35,36] |
| HSV-1/2 | IFI16 | IFI16 inflammasome activation in vitro; HSV-1 encoded ICP0 targets IFI16 for degradation; Proteasomal degradation of HSV1 capsid enhances IFI16 detection of viral DNA. | [18,20,77,81] |
| HCMV | IFI16 | IFI16 inflammasome activation in vitro; HCMV encoded pUL83 and pUL97 alter cellular localization of IFI16. | [78–81] |
| Vaccinia | AIM2 | AIM2 inflammasome activation and pyroptotic cell death in vitro. | [10,11] |
| MCMV | AIM2 | AIM2 inflammasome activation is necessary for control of virus infection in vivo and for NK cell responses. | [11] |
| BoHV-1 | IFI16 | IFI16 inflammasome activation detected in vitro but no effect on virus replication observed. | [21] |
| HIV | IFI16 | IFI16 inflammasome activation in CD4+ T cells results in T cell death and contributes to T cell depletion. | [53–55] |
Unlike TLR9 in the endosomal compartment or other cytoplasmic DNA sensors, ALRs bind to DNA in a sequence and AT nucleotide content-independent manner. Instead, DNA binding by ALRs is achieved by electrostatic interactions between positively charged residues in the HIN-200 domain and the negatively charged DNA sugar phosphate backbone [25]. Based on these reports, one of the biggest unanswered questions is how IFI16 can distinguish between host and viral DNA in the nucleus? It therefore seems likely that binding to different forms of DNA by IFI16 must be mediated by and also recruit different effector molecules to facilitate the observed responses. The exact identity of these molecules and the mechanisms of their recruitment remain to be discovered.
The clinical relevance of ALR inflammasomes activation during virus infection is just beginning to emerge. Expression of AIM2, IL-1β and IL-18 in hepatitis B virus (HBV) infected human PBMCs correlates with viral load, and AIM2 expression is lower in chronic as compared to acute cases [26]. AIM2 expression also correlates with renal damage during HBV infection, possibly indicating that AIM2 enhances immunopathology during HBV infection [27,28], but a definitive role remains to be determined.
Prior to their discovery as antiviral signaling molecules, AIM2 and IFI16 (p204) were recognized for their importance during cell growth and cancer development [29,30]. One emerging role for ALRs in the clinic is during oncogenic virus infection. Over 18% of all cancers are associated with persistent infections, and DNA viruses cause or are associate with a majority of these [31]. EBV causes oncogenesis in certain individuals resulting in Hodgkin's lymphoma, Burkitt's lymphoma and nasopharyngeal carcinoma. In tissue culture, inflammasome activation by EBV resulted in increased cell proliferation through IL-1β signaling [32]. However, in an in vivo mouse model, enhanced inflammasome activation resulted in increased neutrophil infiltration into EBV positive tumors and subsequent tumor regression [32]. In clinical samples, increased IL-1β and neutrophil infiltration into EBV associated nasopharyngeal carcinomas correlated with improved survival in human patients [32]. Based on these findings, inflammasome activation may hold potential therapeutic benefit during EBV associated cancer treatment.
Inflammasome activation may also be important during infection with other oncogenic viruses. KSHV is another gammaherpesvirus and is linked to the development of Kaposi Sarcoma. Although a direct relationship between inflammasome activation and cancer development has not been shown, two recent reports demonstrated that IFI16 associates with nuclear KSHV genomes and forms an inflammasome with ASC and caspase-1, resulting in activation of IL-1β and IL-18 [12,33]. Human papillomaviruses (HPV) HPV16 and HPV18 are associated with up to 70% of all cervical cancers [34]. A potential role for either IFI16 or AIM2 in the recognition of HPV in cervical cancer has been shown through the examination of human keratinocytes or cervical cancer biopsies where IL-1β, caspase-1, IFI16 and AIM2 gene or protein expression are increased in HPV infected tissue, but not in uninfected tissue [35,36]. High levels of IFI16 expression also correlated with reduced cell proliferation in HPV positive tumors, possibly indicating a protective role [36,37]. Transfection of primary keratinocytes with HPV DNA results in AIM2 inflammasome activation and the production of active IL-1β [35]. However, whether this is the case during natural infection is not known. Also, the HPV16 E6 protein targets pro-IL-1β for proteasomal degradation, thus inhibiting many of the potentially protective effects of inflammasome activation [38]. To clearly determine the role for ALR mediated inflammasome activation with HPV or KSHV, it will be important to examine the correlation between IL-1β levels and tumor development more directly in human clinical patients or develop an in vivo model where inflammasome function can be inhibited and tumor development examined.
NLRP3 inflammasome
Activation of the NLRP3 inflammasome by viruses is well established and the in vivo relevance for control of virus infection dependent on NLRP3 has been shown for many of them [39]. The list of viral pathogens that activate NLRP3 continues to grow and in the last two years seven new viruses have been reported to activate the NLRP3 inflammasome (Table 2). One common theme that has emerged is that NLRP3 senses the cellular stress and changes in ion concentration caused by virus encoded ion channels. This was initially reported for the M2 protein of influenza A virus [40]. More recently, several members of the picornaviridae family were shown to similarly activate NLRP3 through their 2B protein ion channel including encephalomyocarditis virus, poliovirus, enterovirus 71, and human rhinovirus [41,42]. The SH protein of human respiratory syncytial virus (RSV) also mediates NLRP3 inflammasome activation, as does the E protein of porcine reproductive and respiratory syndrome virus (PRRSV) [43,44]. Clearly, ion flux is a key feature for NLRP3 sensing of viral infection.
Table 2.
Recent discoveries or advances in NLRP3 activating viruses
| Virus | Biological Consequence | Reference(s) |
|---|---|---|
| IAV | Type-I interferon and RIG-I regulate NLRP3 activation in primary human lung epithelial cells. PB1-F2 viral protein from pathogenic viruses enhances NLRP3 inflammasome activation. | [66,68] |
| RSV | Inflammasome activation in response to cytosolic RNA is impaired in neonatal monocytes. | [74] |
| RVFV | Activates NLRP3 inflammasome in murine dendritic cells | [60] |
| Rhinovirus | 2B viroporin activates NLRP3 inflammasome in vitro | [42] |
| HCV | NLRP3 inflammasome activation occurs in monocytes and may result in both protective and destructive immune responses. | [45–49] |
| WNV | NLRP3 activation prevents virus replication and protects from neuronal damage. | [61,62] |
| DV | NLRP3 inflammasome activation in platelets is associated with increased vascular permeability and disease severity in human patients. | [65] |
| PRRS | NLRP3 activation in vitro and potentially in swine. | [43,44] |
| CHIKV | High NLRP3, caspase-1 and IL-1β levels are predictive of severe disease in humans | [63,64] |
| HIV-1 | NLRP3 inflammasome activation in vitro in both monocyte/macrophages and microglia; Activation may lead to HIV induced neuronal damage; Polymorphisms in NLRP3 associated with HIV infection in humans. | [47,50–52,58,59] |
| FIV | NLRP3 inflammasome activation associated with neuronal damage. | [52] |
| HTLV-1 | NLRP3 polymorphisms associated with HTLV-1 infection in humans. | [57] |
| FMLV | Virus mediated expression of IL- 1β enhances virus replication and inflammation in mice | [56] |
| HSV-1 | Activates both the NLRP3 and IFI16 inflammasomes in vitro. | [20] |
| Adenovirus | Persistence in endosomes enhances NLRP3 activation. | [83] |
Much progress has also been made on understanding the role for NLRP3 during hepatitis C virus (HCV) infection. Transfection of HCV RNA can induce NLRP3 inflammasome activation in a reactive oxygen species dependent manner, but the exact mechanism is not known [45,46]. Obtaining a better understanding of inflammasome activation may facilitate HCV treatment though, as the inflammasome is likely an important inflammatory pathway activated by HCV in monocytes, even when the interferon response is not [47]. It was further shown in vitro that IL-18 produced by the inflammasome in human monocytes helps to activate natural killer cells that facilitate the control of HCV infection [48]. Although this may be beneficial in the short term, chronic inflammation dependent on NLRP3 inflammasome activation has been suggested to result in the liver immunopathology observed during HCV infection [49]. Although progress has been made, there is still much about the clinical relevance of inflammasome activation during HCV infection that is unknown.
There have been a flurry of recent studies on HIV-1 activation of the NLRP3 inflammasome in monocytes [47,50,51] and NLRP3 activation appears to be especially important for neuronal damage resulting from HIV infection of microglial cells in the central nervous system [52]. The same authors also described a correlation between neuronal damage during infection with another retrovirus, feline immunodeficiency virus (FIV), that was associated with increased NLRP3, caspase-1 and IL-1β expression in vivo [52]. In addition to NLRP3, HIV-1 is further detected by the IFI16 inflammasome in CD4+ T cells [53] and pyroptotic cell death plays an important role in the depletion of CD4+ T cells that are not productively infected with HIV [53–55]. Inflammasome activation by retroviruses seems to be detrimental to the host in most instances and to have little effect on virus replication. This is perhaps highlighted by a study with another retrovirus, friend murine leukemia virus (FMLV), where the researcher inserted a modified IL-1β gene into the virus resulting in spontaneous production of functionally active IL-1β independent of inflammasome activation [56]. Surprisingly, this enhanced and prolonged FMLV replication in vivo and resulted in enhanced inflammation. Despite this, NLR containing inflammasome activation may play some protective role during retroviral infection in humans, as polymorphisms in NLRP1 and NLRP3 are more commonly found in patients infected with human T-lymphotropic virus (HTLV-1) or HIV-1 than in uninfected controls [57–59].
Finally, NLRP3 is now recognized to function in the detection of various highly pathogenic or hemorrhagic viruses such as Rift Valley fever virus (RVFV) [60] and NLRP3 inflammasome activation in neurons was essential for control of West Nile virus (WNV) replication and prevention of central nervous system damage in a mouse model of WNV infection [61,62]. On the other hand, increased expression of NLRP3 in human patients was observed during infection with chikungunya virus (CHIKV) and elevated caspase-1 expression and IL-1β levels were highly predictive markers of severe disease in humans [63,64]. Dengue virus (DV) also activates the NLRP3 inflammasome in human platelets that were experimentally infected or in platelets isolated from DV infected patients and high platelet derived IL-1β levels caused increased permeability in an in vitro assay and were associated with increased vascular permeability in clinical cases [65]. Infection of mice with influenza A virus strains that possess the PB1-F2 protein caused significantly more inflammasome activation and treatment of mice with purified PB1-F2 peptides from highly pathogenic strains resulted in increased inflammasome activation and lung inflammation than peptides from low pathogenic strains [66]. In all, the effects of NLRP3 inflammasome activation in vivo and in the clinical setting are very much virus specific and provide significant protection against some highly pathogenic viruses but contribute to the immunopathology of others.
RLR inflammasome
Previous reports suggested that RIG-I could form an inflammasome in response to certain viruses; however, some debate exists as to the exact role of RIG-I in inflammasome formation [13,67]. More recent publications indicate that the RIG-I-MAVS signaling axis may play several roles. Pothlichet et al. [68] reported that primary human bronchial epithelial cells (NHBEs) infected with influenza A virus resulted in RIG-I dependent priming of the NLRP3 inflammasome as well as direct RIG-I mediated inflammasome activation. Additionally, three separate reports demonstrated that MAVS associates with NLRP3 and either recruits NLRP3 to mitochondria or is essential in upregulating NLRP3 and pro-IL-1β, thus priming the inflammasome [60,69–71]. MAVS may further facilitate NLRP3 inflammasome activation through the formation of prion-like oligomers that result in membrane damage and subsequent NLRP3 inflammasome activation [72,73]. RIG-I priming or inflammasome activation may also be important for childhood diseases like RSV. Huang et al. [74] showed that in response to the RIG-I ligand 5’ppp-dsRNA, human neonatal monocytes collected from cord blood had weak inflammasome activation and no increase in IL-1β secretion compared to peripheral blood monocytes taken from adults. It appears that RIG-I can form its own inflammasome in response to some virus infections, but whether this is a major or minor function compared to its role in RIG-I-MAVS signaling and priming to facilitate NLRP3 inflammasome activation remains to be seen.
Viral and host regulation of inflammasomes
Viruses and their host are under constant evolutionary pressure from each other. As an example of pathogen-host co-evolution, ALR loci in different mammals are quite distinct and AIM2 and IFI16 appear to have evolved adaptively [75], suggesting a strong selective pressure [76]. Similarly, DNA viruses have evolved strategies to evade detection by ALRs. HSV-1 expresses an E3 ubiquitin ligase ICP0 that in human fibroblasts targets IFI16 for proteosomal degradation [77]. The HCMV tegument protein pUL83 suppresses IFI16 mediated detection of HCMV genomic DNA in the nucleus by binding to the IFI16 pyrin domain (PYD) through its own pyrin association domain (PAD) and preventing IFI16 oligomerization [78]. Interestingly, HCMV can hijack IFI16 through an interaction with the viral pUL83 protein, and HCMV utilizes IFI16 as a transcriptional activator for the initiation of immediate-early gene production [79]. IFI16 localization in the cell is dependent on its acetylation or phosphorylation by either cellular or virus encoded enzymes. HCMV encodes a viral kinase pUL97 that can phosphorylate IFI16, leading to IFI16 export from the nucleus, thereby preventing detection of HCMV DNA [80]. Acetylation, on the other had, appears to be carried out by the cellular p300 acetyltransferase [18]. Finally, DNA viruses encapsidate their DNA in an effort to mask it from detection by ALRs. However, during HSV-1 infection, the cellular ubiquitin proteasome machinery targets the HSV-1 capsid proteins for degradation, thus unmasking the viral DNA for IFI16 detection [81]. As with other PRRs, there is clearly an evolutionary arms race between DNA viruses and ALRs.
Numerous viruses possess virulence factors that regulate the inflammasome through the direct inhibition of caspase-1 enzymatic function or blockade of ASC-Caspase-1 interaction [82]. Furthermore, the F1L protein of vaccinia virus is able to bind to and inhibit the function of NLRP1, thus suppressing inflammasome activation, and F1L mutant vaccinia virus infection in vivo resulted in early and enhanced inflammasome activation [24]. However, vaccinia virus has never been shown to activate the NLRP1 inflammasome directly, but it does activate both NLRP3 and AIM2 inflammasomes [10,11,23]. Therefore, the functional significance of this F1L-NLRP1 interaction is not completely clear, although the role for F1L inflammasome suppression is evident. NLRP3 inflammasome activation occurs primarily in response to pathogen induced damage or DAMPS and recent evidence suggests that the degree to which certain adenoviruses activate the NLRP3 inflammasome is due to their accumulation or rapid escape from endosomes prior to endosomal acidification with those escaping earlier causing less inflammasome activation [83]. Recent advances in the cellular pathways required for NLRP3 inflammasome activation include the requirement for mitofusin 2 for recruitment of NLRP3 to the mitochondria and inflammasome activation was dependent on maintenance of mitochondrial membrane potential and mitofusin 2-NLRP3 interaction [84]. The activation of DRP1, a GTPase that also regulates mitochondrial fusion, is regulated by a RIP3-RIP1 pathway in response to viral RNA and is necessary for the activation of NLRP3 due to the induction of mitochondrial damage [85]. Although both of these reports demonstrate the importance of the mitochondria during NLRP3 inflammasome activation, they also highlight the ongoing debate over the exact mechanisms involved, as mitochondrial damage reportedly suppressed NLRP3 activation dependent on mitofusin 2, but enhanced NLRP3 activation dependent on DRP1 [84,85].
Given the potent inflammatory stimulus provided by inflammasome activation, it is not surprising that host regulatory mechanisms have evolved to down-modulate signaling. AIM2 exists in the cytosol in an auto-inhibited state until binding to double stranded DNA of pathogen or host origin induces a conformational change resulting in self oligomerization and recruitment of ASC and caspase-1 [86]. To further ensure that ALRs are not unnecessarily activated, several suppression mechanisms appear to have evolved. Pyrin domain only protein 3 (POP3) is a recently described member of the PYHIN family in humans (although it is absent in mice) [87]. POP3 lacks a HIN domain and therefore does not bind to DNA, but it does have a PYD domain, which can bind to AIM2 and IFI16 and block ASC binding and subsequent inflammasome formation [87]. In mice, the PYHIN protein p202 (which is absent in humans) lacks a PYD but possess two HIN domains [88]. The p202 HIN1 domain is capable of binding to dsDNA, possibly sequestering it from AIM2. The HIN2 domain binds to the HIN domain of AIM2, spatially separating AIM2 PYDs and preventing ASC clustering. Although POP3 and p202 inhibit ALR activation through different mechanism, their roles in humans or mice respectively appear to be the same. Alternatively spliced isoforms of human AIM2 lacking either the PYD or the HIN domain have been also predicted, raising the possibility that these splice variants may inhibit AIM2 activation in a similar manner [89]. Promyelocytic Leukemia Protein sequesters ASC in the nucleus, thus preventing activation of both the AIM2 and NLRP3 inflammasomes [90]. Finally, removal of damaged mitochondria as a result of influenza A virus infection is dependent on NOD2 mediated autophagy of mitochondria and absence of NOD2 signaling during virus infection results in enhanced NLRP3 inflammasome activation and enhanced immunopathology in mice [91]. In all, inflammasome activation is a two edged sword that needs to be tightly controlled to restrain virus replication as well as prevent immunopathology.
Conclusions
Inflammasome activation is an essential part of the immune response to virus infection and holds the promise of many potential therapeutic drug- or vaccine-based interventions for either the control of virus replication or resolution of excessive inflammation and immunopathology. In the clinic, inhibition of NLRP3 or IL-1β is already used for several autoinflammatory or autoimmune diseases, but there has been little clinical research directed at using such treatments during viral infection. Although the range of viruses detected by NLRP3 has grown immensely in the last few years, the overall range of viruses detected by AIM2 or IFI16 remains to be determined. Also, there is little reported on the clinical role for ALRs so that it is difficult to make any conclusions as to the clinical importance of ALRs during virus infection except perhaps with regard to viral infections associated with cancer. In addition, IFI16 and AIM2 are members of a diverse set of DNA binding proteins that perform several antiviral and proinflammatory roles during virus infection. The ability of IFI16 to perform numerous functions independent of any known specificity for DNA binding (Figure 1) suggests that there is much about the function of this and other ALRs that we do not understand and which needs to be understood before targeting this protein for therapeutic purposes. In the case of NLRP3, the exact mechanisms for damage sensing are still debated, but the current body of literature suggests that ion flux in a variety of forms may be a common mechanism for sensing damage. Also, NLRP3 is frequently activated in conjunction with other inflammasomes such as AIM2 and IFI16, but the relative contributions need further examination. In conclusion, the realization of the clinical potential for inflammasomes during virus infection will require additional investigation but great strides have been made and reports regarding host- or virus-encoded inhibitors of inflammasome activation have also provided us with potential tools for the development of such treatments.
Highlights.
Advances in ALR mediated inflammasome activation in response to viral DNA.
Viral virulence factors that impair inflammasome activation.
Host regulation of inflammasomes to prevent unwanted inflammation.
Recent advances in understanding NLRP3 inflammasome activation during virus infection.
Potential clinical roles for inflammasome activation.
Acknowledgements
We thank Si Ming Man for helpful discussion in the preparation of this manuscript. This work was supported by grants to T.-D.K. from the US National Institutes of Health through the National Institute of Arthritis and Musculoskeletal and Skin Diseases (Award Number AR056296), the National Institute of Allergy and Infectious Disease (Award Number AI101935) and the National Cancer Institute (Award Number CA163507). This work was also funded by the American Lebanese Syrian Associated Charities to T.-D.K. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Unterholzner L. The interferon response to intracellular DNA: why so many receptors? Immunobiology. 2013;218:1312–1321. doi: 10.1016/j.imbio.2013.07.007. [DOI] [PubMed] [Google Scholar]
- 2.Patel JR, Garcia-Sastre A. Activation and regulation of pathogen sensor RIG-I. Cytokine Growth Factor Rev. 2014 doi: 10.1016/j.cytogfr.2014.08.005. [DOI] [PubMed] [Google Scholar]
- 3.Boyden ED, Dietrich WF. Nalp1b controls mouse macrophage susceptibility to anthrax lethal toxin. Nat Genet. 2006;38:240–244. doi: 10.1038/ng1724. [DOI] [PubMed] [Google Scholar]
- 4.Mariathasan S, Newton K, Monack DM, Vucic D, French DM, Lee WP, Roose-Girma M, Erickson S, Dixit VM. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature. 2004;430:213–218. doi: 10.1038/nature02664. [DOI] [PubMed] [Google Scholar]
- 5.Martinon F, Burns K, Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol Cell. 2002;10:417–426. doi: 10.1016/s1097-2765(02)00599-3. [DOI] [PubMed] [Google Scholar]
- 6.Mariathasan S, Weiss DS, Newton K, McBride J, O'Rourke K, Roose-Girma M, Lee WP, Weinrauch Y, Monack DM, Dixit VM. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature. 2006;440:228–232. doi: 10.1038/nature04515. [DOI] [PubMed] [Google Scholar]
- 7.Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006;440:237–241. doi: 10.1038/nature04516. [DOI] [PubMed] [Google Scholar]
- 8.Kanneganti TD, Lamkanfi M, Kim YG, Chen G, Park JH, Franchi L, Vandenabeele P, Nunez G. Pannexin-1-mediated recognition of bacterial molecules activates the cryopyrin inflammasome independent of Toll-like receptor signaling. Immunity. 2007;26:433–443. doi: 10.1016/j.immuni.2007.03.008. [DOI] [PubMed] [Google Scholar]
- 9.Fernandes-Alnemri T, Yu JW, Datta P, Wu J, Alnemri ES. AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature. 2009;458:509–513. doi: 10.1038/nature07710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hornung V, Ablasser A, Charrel-Dennis M, Bauernfeind F, Horvath G, Caffrey DR, Latz E, Fitzgerald KA. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature. 2009;458:514–518. doi: 10.1038/nature07725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rathinam VA, Jiang Z, Waggoner SN, Sharma S, Cole LE, Waggoner L, Vanaja SK, Monks BG, Ganesan S, Latz E, et al. The AIM2 inflammasome is essential for host defense against cytosolic bacteria and DNA viruses. Nat Immunol. 2010;11:395–402. doi: 10.1038/ni.1864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kerur N, Veettil MV, Sharma-Walia N, Bottero V, Sadagopan S, Otageri P, Chandran B. IFI16 acts as a nuclear pathogen sensor to induce the inflammasome in response to Kaposi Sarcoma-associated herpesvirus infection. Cell Host Microbe. 2011;9:363–375. doi: 10.1016/j.chom.2011.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Poeck H, Bscheider M, Gross O, Finger K, Roth S, Rebsamen M, Hannesschlager N, Schlee M, Rothenfusser S, Barchet W, et al. Recognition of RNA virus by RIG-I results in activation of CARD9 and inflammasome signaling for interleukin 1 beta production. Nat Immunol. 2010;11:63–69. doi: 10.1038/ni.1824. [DOI] [PubMed] [Google Scholar]
- 14.Connolly DJ, Bowie AG. The emerging role of human PYHIN proteins in innate immunity: Implications for health and disease. Biochem Pharmacol. 2014 doi: 10.1016/j.bcp.2014.08.031. [DOI] [PubMed] [Google Scholar]
- 15.Burckstummer T, Baumann C, Bluml S, Dixit E, Durnberger G, Jahn H, Planyavsky M, Bilban M, Colinge J, Bennett KL, et al. An orthogonal proteomic-genomic screen identifies AIM2 as a cytoplasmic DNA sensor for the inflammasome. Nat Immunol. 2009;10:266–272. doi: 10.1038/ni.1702. [DOI] [PubMed] [Google Scholar]
- 16.Roberts TL, Idris A, Dunn JA, Kelly GM, Burnton CM, Hodgson S, Hardy LL, Garceau V, Sweet MJ, Ross IL, et al. HIN-200 proteins regulate caspase activation in response to foreign cytoplasmic DNA. Science. 2009;323:1057–1060. doi: 10.1126/science.1169841. [DOI] [PubMed] [Google Scholar]
- 17.Unterholzner L, Keating SE, Baran M, Horan KA, Jensen SB, Sharma S, Sirois CM, Jin T, Latz E, Xiao TS, et al. IFI16 is an innate immune sensor for intracellular DNA. Nat Immunol. 2010;11:997–1004. doi: 10.1038/ni.1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li T, Diner BA, Chen J, Cristea IM. Acetylation modulates cellular distribution and DNA sensing ability of interferon-inducible protein IFI16. Proc Natl Acad Sci U S A. 2012;109:10558–10563. doi: 10.1073/pnas.1203447109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ansari MA, Singh VV, Dutta S, Veettil MV, Dutta D, Chikoti L, Lu J, Everly D, Chandran B. Constitutive interferon-inducible protein 16-inflammasome activation during Epstein-Barr virus latency I, II, and III in B and epithelial cells. J Virol. 2013;87:8606–8623. doi: 10.1128/JVI.00805-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Johnson KE, Chikoti L, Chandran B. Herpes simplex virus 1 infection induces activation and subsequent inhibition of the IFI16 and NLRP3 inflammasomes. J Virol. 2013;87:5005–5018. doi: 10.1128/JVI.00082-13. Highlights the ability of viruses to activate multiple inflammasomes simultaneously and raises the question of individual importance/ redundancy
- 21.Wang J, Alexander J, Wiebe M, Jones C. Bovine herpesvirus 1 productive infection stimulates inflammasome formation and caspase 1 activity. Virus Res. 2014;185:72–76. doi: 10.1016/j.virusres.2014.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang M, Covar J, Zhang NY, Chen W, Marshall B, Mo J, Atherton SS. Virus spread and immune response following anterior chamber inoculation of HSV-1 lacking the Beclin-binding domain (BBD) J Neuroimmunol. 2013;260:82–91. doi: 10.1016/j.jneuroim.2013.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Delaloye J, Roger T, Steiner-Tardivel QG, Le Roy D, Knaup Reymond M, Akira S, Petrilli V, Gomez CE, Perdiguero B, Tschopp J, et al. Innate immune sensing of modified vaccinia virus Ankara (MVA) is mediated by TLR2-TLR6, MDA-5 and the NALP3 inflammasome. PLoS Pathog. 2009;5:e1000480. doi: 10.1371/journal.ppat.1000480. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 24.Gerlic M, Faustin B, Postigo A, Yu EC, Proell M, Gombosuren N, Krajewska M, Flynn R, Croft M, Way M, et al. Vaccinia virus F1L protein promotes virulence by inhibiting inflammasome activation. Proc Natl Acad Sci U S A. 2013;110:7808–7813. doi: 10.1073/pnas.1215995110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jin T, Perry A, Jiang J, Smith P, Curry JA, Unterholzner L, Jiang Z, Horvath G, Rathinam VA, Johnstone RW, et al. Structures of the HIN domain:DNA complexes reveal ligand binding and activation mechanisms of the AIM2 inflammasome and IFI16 receptor. Immunity. 2012;36:561–571. doi: 10.1016/j.immuni.2012.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu DL, Xu GH, Lu SM, Ma BL, Miao NZ, Liu XB, Cheng YP, Feng JH, Liu ZG, Feng D, et al. Correlation of AIM2 expression in peripheral blood mononuclear cells from humans with acute and chronic hepatitis B. Hum Immunol. 2013;74:514–521. doi: 10.1016/j.humimm.2013.01.022. [DOI] [PubMed] [Google Scholar]
- 27.Du W, Zhen J, Zheng Z, Ma S, Chen S. Expression of AIM2 is high and correlated with inflammation in hepatitis B virus associated glomerulonephritis. J Inflamm (Lond) 2013;10:37. doi: 10.1186/1476-9255-10-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhen J, Zhang L, Pan J, Ma S, Yu X, Li X, Chen S, Du W. AIM2 mediates inflammation-associated renal damage in hepatitis B virus-associated glomerulonephritis by regulating caspase-1, IL-1beta, and IL-18. Mediators Inflamm. 2014;2014:190–860. doi: 10.1155/2014/190860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.DeYoung KL, Ray ME, Su YA, Anzick SL, Johnstone RW, Trapani JA, Meltzer PS, Trent JM. Cloning a novel member of the human interferon-inducible gene family associated with control of tumorigenicity in a model of human melanoma. Oncogene. 1997;15:453–457. doi: 10.1038/sj.onc.1201206. [DOI] [PubMed] [Google Scholar]
- 30.Lembo M, Sacchi C, Zappador C, Bellomo G, Gaboli M, Pandolfi PP, Gariglio M, Landolfo S. Inhibition of cell proliferation by the interferon-inducible 204 gene, a member of the Ifi 200 cluster. Oncogene. 1998;16:1543–1551. doi: 10.1038/sj.onc.1201677. [DOI] [PubMed] [Google Scholar]
- 31.Parkin DM. The global health burden of infection-associated cancers in the year 2002. International journal of cancer. 2006;118:3030–3044. doi: 10.1002/ijc.21731. [DOI] [PubMed] [Google Scholar]
- 32. Chen LC, Wang LJ, Tsang NM, Ojcius DM, Chen CC, Ouyang CN, Hsueh C, Liang Y, Chang KP, Chen CC, et al. Tumour inflammasome-derived IL-1beta recruits neutrophils and improves local recurrence-free survival in EBV-induced nasopharyngeal carcinoma. EMBO Mol Med. 2012;4:1276–1293. doi: 10.1002/emmm.201201569. A seminal report describing the protective effect of inflammasome activation during virus induced cancer in an in vivo mouse model and in clinical samples.
- 33.Singh VV, Kerur N, Bottero V, Dutta S, Chakraborty S, Ansari MA, Paudel N, Chikoti L, Chandran B. Kaposi's sarcoma-associated herpesvirus latency in endothelial and B cells activates gamma interferon-inducible protein 16-mediated inflammasomes. J Virol. 2013;87:4417–4431. doi: 10.1128/JVI.03282-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Durst M, Gissmann L, Ikenberg H, zur Hausen H. A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions. Proc Natl Acad Sci U S A. 1983;80:3812–3815. doi: 10.1073/pnas.80.12.3812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Reinholz M, Kawakami Y, Salzer S, Kreuter A, Dombrowski Y, Koglin S, Kresse S, Ruzicka T, Schauber J. HPV16 activates the AIM2 inflammasome in keratinocytes. Arch Dermatol Res. 2013;305:723–732. doi: 10.1007/s00403-013-1375-0. [DOI] [PubMed] [Google Scholar]
- 36.Mazibrada J, Longo L, Vatrano S, Cappia S, Giorcelli J, Pentenero M, Gandolfo S, Volante M, dell'Oste V, Lo Cigno I, et al. Differential expression of HER2, STAT3, SOX2, IFI16 and cell cycle markers during HPV-related head and neck carcinogenesis. New Microbiol. 2014;37:129–143. [PubMed] [Google Scholar]
- 37.Azzimonti B, Pagano M, Mondini M, De Andrea M, Valente G, Monga G, Tommasino M, Aluffi P, Landolfo S, Gariglio M. Altered patterns of the interferon-inducible gene IFI16 expression in head and neck squamous cell carcinoma: immunohistochemical study including correlation with retinoblastoma protein, human papillomavirus infection and proliferation index. Histopathology. 2004;45:560–572. doi: 10.1111/j.1365-2559.2004.02000.x. [DOI] [PubMed] [Google Scholar]
- 38. Niebler M, Qian X, Hofler D, Kogosov V, Kaewprag J, Kaufmann AM, Ly R, Bohmer G, Zawatzky R, Rosl F, et al. Post-translational control of IL-1beta via the human papillomavirus type 16 E6 oncoprotein: a novel mechanism of innate immune escape mediated by the E3-ubiquitin ligase E6-AP and p53. PLoS Pathog. 2013;9:e1003536. doi: 10.1371/journal.ppat.1003536. Although ubiquitination and proteasome degradation have been shown to regulate NLRP3 activation, this is the first report that a virus can directly degrade IL-1².
- 39.Lupfer C, Kanneganti TD. The expanding role of NLRs in antiviral immunity. Immunol Rev. 2013;255:13–24. doi: 10.1111/imr.12089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ichinohe T, Pang IK, Iwasaki A. Influenza virus activates inflammasomes via its intracellular M2 ion channel. Nat Immunol. 2010;11:404–410. doi: 10.1038/ni.1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ito M, Yanagi Y, Ichinohe T. Encephalomyocarditis virus viroporin 2B activates NLRP3 inflammasome. PLoS Pathog. 2012;8:e1002857. doi: 10.1371/journal.ppat.1002857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Triantafilou K, Kar S, van Kuppeveld FJ, Triantafilou M. Rhinovirus-induced calcium flux triggers NLRP3 and NLRC5 activation in bronchial cells. Am J Respir Cell Mol Biol. 2013;49:923–934. doi: 10.1165/rcmb.2013-0032OC. [DOI] [PubMed] [Google Scholar]
- 43.Bi J, Song S, Fang L, Wang D, Jing H, Gao L, Cai Y, Luo R, Chen H, Xiao S. Porcine reproductive and respiratory syndrome virus induces IL-1beta production depending on TLR4/MyD88 pathway and NLRP3 inflammasome in primary porcine alveolar macrophages. Mediators Inflamm. 2014;2014:403–515. doi: 10.1155/2014/403515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang K, Hou Q, Zhong Z, Li X, Chen H, Li W, Wen J, Wang L, Liu W, Zhong F. Porcine reproductive and respiratory syndrome virus activates inflammasomes of porcine alveolar macrophages via its small envelope protein E. Virology. 2013;442:156–162. doi: 10.1016/j.virol.2013.04.007. [DOI] [PubMed] [Google Scholar]
- 45.Burdette D, Haskett A, Presser L, McRae S, Iqbal J, Waris G. Hepatitis C virus activates interleukin-1beta via caspase-1-inflammasome complex. J Gen Virol. 2012;93:235–246. doi: 10.1099/vir.0.034033-0. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 46.Chen W, Xu Y, Li H, Tao W, Xiang Y, Huang B, Niu J, Zhong J, Meng G. HCV genomic RNA activates the NLRP3 inflammasome in human myeloid cells. PLoS One. 2014;9:e84953. doi: 10.1371/journal.pone.0084953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chattergoon MA, Latanich R, Quinn J, Winter ME, Buckheit RW, 3rd, Blankson JN, Pardoll D, Cox AL. HIV and HCV activate the inflammasome in monocytes and macrophages via endosomal Toll-like receptors without induction of type 1 interferon. PLoS Pathog. 2014;10:e1004082. doi: 10.1371/journal.ppat.1004082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Serti E, Werner JM, Chattergoon M, Cox AL, Lohmann V, Rehermann B. Monocytes activate natural killer cells via inflammasome-induced interleukin 18 in response to hepatitis C virus replication. Gastroenterology. 2014;147:209–220. e203. doi: 10.1053/j.gastro.2014.03.046. These two papers examine the potential protective and detrimental roles for the NLRP3 inflammasome during HCV infection.
- 49. Negash AA, Ramos HJ, Crochet N, Lau DT, Doehle B, Papic N, Delker DA, Jo J, Bertoletti A, Hagedorn CH, et al. IL-1beta production through the NLRP3 inflammasome by hepatic macrophages links hepatitis C virus infection with liver inflammation and disease. PLoS Pathog. 2013;9:e1003330. doi: 10.1371/journal.ppat.1003330. These two papers examine the potential protective and detrimental roles for the NLRP3 inflammasome during HCV infection.
- 50.Hernandez JC, Latz E, Urcuqui-Inchima S. HIV-1 induces the first signal to activate the NLRP3 inflammasome in monocyte-derived macrophages. Intervirology. 2014;57:36–42. doi: 10.1159/000353902. [DOI] [PubMed] [Google Scholar]
- 51.Guo H, Gao J, Taxman DJ, Ting JP, Su L. HIV-1 infection induces interleukin-1beta production via TLR8 protein-dependent and NLRP3 inflammasome mechanisms in human monocytes. J Biol Chem. 2014;289:21716–21726. doi: 10.1074/jbc.M114.566620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Walsh JG, Reinke SN, Mamik MK, McKenzie BA, Maingat F, Branton WG, Broadhurst DI, Power C. Rapid inflammasome activation in microglia contributes to brain disease in HIV/AIDS. Retrovirology. 2014;11:35. doi: 10.1186/1742-4690-11-35. HIV induced dementia is an important clinical manifestation and Walsh et al. indicate that this may result from HIV infection of microglia and subsequent immune mediated cell death of neurons.
- 53. Monroe KM, Yang Z, Johnson JR, Geng X, Doitsh G, Krogan NJ, Greene WC. IFI16 DNA sensor is required for death of lymphoid CD4 T cells abortively infected with HIV. Science. 2014;343:428–432. doi: 10.1126/science.1243640. This group of publications elucidates the mechanism by which CD4 T cells that are abortively infected by HIV still undergo IFI16 inflammasome mediated cell death, thus further reducing T cell numbers in AIDS patients.
- 54. Doitsh G, Galloway NL, Geng X, Yang Z, Monroe KM, Zepeda O, Hunt PW, Hatano H, Sowinski S, Munoz-Arias I, et al. Cell death by pyroptosis drives CD4 T-cell depletion in HIV-1 infection. Nature. 2014;505:509–514. doi: 10.1038/nature12940. This group of publications elucidates the mechanism by which CD4 T cells that are abortively infected by HIV still undergo IFI16 inflammasome mediated cell death, thus further reducing T cell numbers in AIDS patients.
- 55. Doitsh G, Cavrois M, Lassen KG, Zepeda O, Yang Z, Santiago ML, Hebbeler AM, Greene WC. Abortive HIV infection mediates CD4 T cell depletion and inflammation in human lymphoid tissue. Cell. 2010;143:789–801. doi: 10.1016/j.cell.2010.11.001. This group of publications elucidates the mechanism by which CD4 T cells that are abortively infected by HIV still undergo IFI16 inflammasome mediated cell death, thus further reducing T cell numbers in AIDS patients.
- 56.Browne EP. An interleukin-1 Beta-encoding retrovirus exhibits enhanced replication in vivo. J Virol. 2015;89:155–164. doi: 10.1128/JVI.02314-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kamada AJ, Pontillo A, Guimaraes RL, Loureiro P, Crovella S, Brandao LA. NLRP3 polymorphism is associated with protection against human T-lymphotropic virus 1 infection. Mem Inst Oswaldo Cruz. 2014;109:960–963. [PubMed] [Google Scholar]
- 58.Pontillo A, Oshiro TM, Girardelli M, Kamada AJ, Crovella S, Duarte AJ. Polymorphisms in inflammasome' genes and susceptibility to HIV-1 infection. J Acquir Immune Defic Syndr. 2012;59:121–125. doi: 10.1097/QAI.0b013e3182392ebe. [DOI] [PubMed] [Google Scholar]
- 59.Pontillo A, Brandao LA, Guimaraes RL, Segat L, Athanasakis E, Crovella S. A 3'UTR SNP in NLRP3 gene is associated with susceptibility to HIV-1 infection. J Acquir Immune Defic Syndr. 2010;54:236–240. doi: 10.1097/QAI.0b013e3181dd17d4. [DOI] [PubMed] [Google Scholar]
- 60.Ermler ME, Traylor Z, Patel K, Schattgen SA, Vanaja SK, Fitzgerald KA, Hise AG. Rift Valley fever virus infection induces activation of the NLRP3 inflammasome. Virology. 2014;449:174–180. doi: 10.1016/j.virol.2013.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kumar M, Roe K, Orillo B, Muruve DA, Nerurkar VR, Gale M, Jr, Verma S. Inflammasome adaptor protein apoptosis-associated speck-like protein containing CARD (ASC) is critical for the immune response and survival in west Nile virus encephalitis. J Virol. 2013;87:3655–3667. doi: 10.1128/JVI.02667-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ramos HJ, Lanteri MC, Blahnik G, Negash A, Suthar MS, Brassil MM, Sodhi K, Treuting PM, Busch MP, Norris PJ, et al. IL-1beta signaling promotes CNS-intrinsic immune control of West Nile virus infection. PLoS Pathog. 2012;8:e1003039. doi: 10.1371/journal.ppat.1003039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ng LF, Chow A, Sun YJ, Kwek DJ, Lim PL, Dimatatac F, Ng LC, Ooi EE, Choo KH, Her Z, et al. IL-1beta, IL-6, and RANTES as biomarkers of Chikungunya severity. PLoS One. 2009;4:e4261. doi: 10.1371/journal.pone.0004261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wikan N, Khongwichit S, Phuklia W, Ubol S, Thonsakulprasert T, Thannagith M, Tanramluk D, Paemanee A, Kittisenachai S, Roytrakul S, et al. Comprehensive proteomic analysis of white blood cells from chikungunya fever patients of different severities. J Transl Med. 2014;12:96. doi: 10.1186/1479-5876-12-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Hottz ED, Lopes JF, Freitas C, Valls-de-Souza R, Oliveira MF, Bozza MT, Da Poian AT, Weyrich AS, Zimmerman GA, Bozza FA, et al. Platelets mediate increased endothelium permeability in dengue through NLRP3-inflammasome activation. Blood. 2013;122:3405–3414. doi: 10.1182/blood-2013-05-504449. NLRP3 inflammasome activation is demonstrated for the first time in platelets and this correlates with hemorrhaging and disease severity during dengue virus infection in human patients.
- 66.McAuley JL, Tate MD, MacKenzie-Kludas CJ, Pinar A, Zeng W, Stutz A, Latz E, Brown LE, Mansell A. Activation of the NLRP3 inflammasome by IAV virulence protein PB1-F2 contributes to severe pathophysiology and disease. PLoS Pathog. 2013;9:e1003392. doi: 10.1371/journal.ppat.1003392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Rajan JV, Rodriguez D, Miao EA, Aderem A. The NLRP3 inflammasome detects encephalomyocarditis virus and vesicular stomatitis virus infection. J Virol. 2011;85:4167–4172. doi: 10.1128/JVI.01687-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Pothlichet J, Meunier I, Davis BK, Ting JP, Skamene E, von Messling V, Vidal SM. Type I IFN Triggers RIG-I/TLR3/NLRP3-dependent Inflammasome Activation in Influenza A Virus Infected Cells. PLoS Pathog. 2013;9:e1003256. doi: 10.1371/journal.ppat.1003256. This report provides evidence for a MAVS independent, RIG-I dependent inflammasome in human primary lung epithelial cells.
- 69.Lawrence TM, Hudacek AW, de Zoete MR, Flavell RA, Schnell MJ. Rabies Virus is Recognized by the NLRP3 Inflammasome and Activates IL-1beta Release in Murine Dendritic Cells. J Virol. 2013 doi: 10.1128/JVI.00203-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Subramanian N, Natarajan K, Clatworthy MR, Wang Z, Germain RN. The adaptor MAVS promotes NLRP3 mitochondrial localization and inflammasome activation. Cell. 2013;153:348–361. doi: 10.1016/j.cell.2013.02.054. This set of papers validates the role for MAVS in NLRP3 inflammasome activation only during virus infection, either through recruitment to the mitochondria or through cell damage.
- 71. Park S, Juliana C, Hong S, Datta P, Hwang I, Fernandes-Alnemri T, Yu JW, Alnemri ES. The mitochondrial antiviral protein MAVS associates with NLRP3 and regulates its inflammasome activity. J Immunol. 2013;191:4358–4366. doi: 10.4049/jimmunol.1301170. This set of papers validates the role for MAVS in NLRP3 inflammasome activation only during virus infection, either through recruitment to the mitochondria or through cell damage.
- 72. Cai X, Chen J, Xu H, Liu S, Jiang QX, Halfmann R, Chen ZJ. Prion-like polymerization underlies signal transduction in antiviral immune defense and inflammasome activation. Cell. 2014;156:1207–1222. doi: 10.1016/j.cell.2014.01.063. This set of papers validates the role for MAVS in NLRP3 inflammasome activation only during virus infection, either through recruitment to the mitochondria or through cell damage.
- 73. Franchi L, Eigenbrod T, Munoz-Planillo R, Ozkurede U, Kim YG, Chakrabarti A, Gale M, Jr, Silverman RH, Colonna M, Akira S, et al. Cytosolic double-stranded RNA activates the NLRP3 inflammasome via MAVS-induced membrane permeabilization and K+ efflux. J Immunol. 2014;193:4214–4222. doi: 10.4049/jimmunol.1400582. This set of papers validates the role for MAVS in NLRP3 inflammasome activation only during virus infection, either through recruitment to the mitochondria or through cell damage.
- 74.Huang H, Saravia J, You D, Shaw AJ, Cormier SA. Impaired gamma delta T cell-derived IL-17A and inflammasome activation during early respiratory syncytial virus infection in infants. Immunol Cell Biol. 2014 doi: 10.1038/icb.2014.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cagliani R, Forni D, Biasin M, Comabella M, Guerini FR, Riva S, Pozzoli U, Agliardi C, Caputo D, Malhotra S, et al. Ancient and Recent Selective Pressures Shaped Genetic Diversity at AIM2-Like Nucleic Acid Sensors. Genome Biology and Evolution. 2014;6:830–845. doi: 10.1093/gbe/evu066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Brunette RL, Young JM, Whitley DG, Brodsky IE, Malik HS, Stetson DB. Extensive evolutionary and functional diversity among mammalian AIM2-like receptors. The Journal of Experimental Medicine. 2012;209:1969–1983. doi: 10.1084/jem.20121960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Orzalli M, DeLuca N, Knipe D. HSV-1 ICP0 redistributes the nuclear IFI16 pathogen sensor and promotes its degradation. PNAS. 2012 [Google Scholar]
- 78. Li T, Chen J, Cristea IM. Human cytomegalovirus tegument protein pUL83 inhibits IFI16-mediated DNA sensing for immune evasion. Cell Host Microbe. 2013;14:591–599. doi: 10.1016/j.chom.2013.10.007. These two articles demonstrate the evolutionary adaptation or acquisition of a viral protein (pUL83) that mimics the host pyrin interaction domain to prevent IFI16 activation of host responses and the further adaptation of using IFI16 as a transcriptional activator for the virus.
- 79. Cristea IM, Moorman NJ, Terhune SS, Cuevas CD, O'Keefe ES, Rout MP, Chait BT, Shenk T. Human cytomegalovirus pUL83 stimulates activity of the viral immediate-early promoter through its interaction with the cellular IFI16 protein. J Virol. 2010;84:7803–7814. doi: 10.1128/JVI.00139-10. These two articles demonstrate the evolutionary adaptation or acquisition of a viral protein (pUL83) that mimics the host pyrin interaction domain to prevent IFI16 activation of host responses and the further adaptation of using IFI16 as a transcriptional activator for the virus.
- 80.Dell'Oste V, Gatti D, Gugliesi F, De Andrea M, Bawadekar M, Lo Cigno I, Biolatti M, Vallino M, Marschall M, Gariglio M, et al. Innate nuclear sensor IFI16 translocates into the cytoplasm during the early stage of in vitro human cytomegalovirus infection and is entrapped in the egressing virions during the late stage. J Virol. 2014;88:6970–6982. doi: 10.1128/JVI.00384-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Horan KA, Hansen K, Jakobsen MR, Holm CK, Soby S, Unterholzner L, Thompson M, West JA, Iversen MB, Rasmussen SB, et al. Proteasomal degradation of herpes simplex virus capsids in macrophages releases DNA to the cytosol for recognition by DNA sensors. J Immunol. 2013;190:2311–2319. doi: 10.4049/jimmunol.1202749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lupfer CR, Kanneganti TD. The role of inflammasome modulation in virulence. Virulence. 2012;3:262–270. doi: 10.4161/viru.20266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Teigler JE, Kagan JC, Barouch DH. Late endosomal trafficking of alternative serotype adenovirus vaccine vectors augments antiviral innate immunity. J Virol. 2014;88:10354–10363. doi: 10.1128/JVI.00936-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ichinohe T, Yamazaki T, Koshiba T, Yanagi Y. Mitochondrial protein mitofusin 2 is required for NLRP3 inflammasome activation after RNA virus infection. Proc Natl Acad Sci U S A. 2013;110:17963–17968. doi: 10.1073/pnas.1312571110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Wang X, Jiang W, Yan Y, Gong T, Han J, Tian Z, Zhou R. RNA viruses promote activation of the NLRP3 inflammasome through a RIP1-RIP3-DRP1 signaling pathway. Nat Immunol. 2014;15:1126–1133. doi: 10.1038/ni.3015. The RIP1–RIP3 pathway is traditionally associated with programmed necrosis, but this report shows that in response to viral RNA, NLRP3 is activated due to RIP1-RIP3-DRP1 induced mitochondrial damage.
- 86.Jin T, Perry A, Smith P, Jiang J, Xiao TS. Structure of the absent in melanoma 2 (AIM2) pyrin domain provides insights into the mechanisms of AIM2 autoinhibition and inflammasome assembly. J Biol Chem. 2013;288:13225–13235. doi: 10.1074/jbc.M113.468033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Khare S, Ratsimandresy RA, de Almeida L, Cuda CM, Rellick SL, Misharin AV, Wallin MC, Gangopadhyay A, Forte E, Gottwein E, et al. The PYRIN domain-only protein POP3 inhibits ALR inflammasomes and regulates responses to infection with DNA viruses. Nat Immunol. 2014;15:343–353. doi: 10.1038/ni.2829. The necessity of regulating ALR inflammasomes is highlighted by the separate evolution of POP3 in humans and p202 in mice for the control of ALR mediated inflammasome activation.
- 88. Yin Q, Sester DP, Tian Y, Hsiao YS, Lu A, Cridland JA, Sagulenko V, Thygesen SJ, Choubey D, Hornung V, et al. Molecular mechanism for p202-mediated specific inhibition of aim2 inflammasome activation. Cell Reports. 2013;4:327–339. doi: 10.1016/j.celrep.2013.06.024. The necessity of regulating ALR inflammasomes is highlighted by the separate evolution of POP3 in humans and p202 in mice for the control of ALR mediated inflammasome activation.
- 89.Choubey D, Duan X, Dickerson E, Ponomareva L, Panchanathan R, Shen H, Srivastava R. Interferon-inducible p200-family proteins as novel sensors of cytoplasmic DNA: Role in inflammation and autoimmunity. Journal of Interferon and Cytokine Research. 2010;30:371–380. doi: 10.1089/jir.2009.0096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Dowling JK, Becker CE, Bourke NM, Corr SC, Connolly DJ, Quinn SR, Pandolfi PP, Mansell A, O'Neill LA. Promyelocytic leukemia protein interacts with the apoptosis-associated speck-like protein to limit inflammasome activation. J Biol Chem. 2014;289:6429–6437. doi: 10.1074/jbc.M113.539692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lupfer C, Thomas PG, Anand PK, Vogel P, Milasta S, Martinez J, Huang G, Green M, Kundu M, Chi H, et al. Receptor interacting protein kinase 2-mediated mitophagy regulates inflammasome activation during virus infection. Nat Immunol. 2013;14:480–488. doi: 10.1038/ni.2563. [DOI] [PMC free article] [PubMed] [Google Scholar]