Abstract
Kawasaki disease is an autoimmune disease found predominantly in children under the age of 5 years. Its incidence is higher in those who live in Asian countries or are of Asian descent. Kawasaki disease is characterized as an acute inflammation of the vasculature bed affecting mainly the skin, eyes, lymph nodes, and mucosal layers. Although the disease is usually self-limiting, patients may develop cardiac abnormalities that can lead to death. The exact cause of the disease is unknown; however, researchers hypothesize that an infectious agent is responsible for causing Kawasaki disease. Initial treatment options with intravenous immune globulin and aspirin are sufficient to cure most patients who acquire this disease. Unfortunately, in up to one-quarter of patients, the disease will be refractory to initial therapy and will require further management with corticosteroid, immunomodulatory, or cytotoxic agents. The lack of randomized, controlled trials makes treatment of refractory disease difficult to manage. Until larger randomized, controlled trials are published to give more guidance on therapy for this stage of disease, clinicians should use the data available from observational studies and case reports in conjunction with their clinical expertise to make treatment decisions.
INDEX TERMS: autoimmune disease, cyclophosphamide, cyclosporin, infliximab, methotrexate, methylprednisolone, mucocutaneous lymph node syndrome
INTRODUCTION
In 1967, a Japanese pediatrician, Dr. Tomisaku Kawasaki, discovered what is now known as Kawasaki disease. This autoimmune disease is described as an acute inflammation of the vasculature bed characterized by symptoms such as persistent fever, distinct erythema of the lips and oral mucosa, rash involving the trunk and extremities, bilateral conjunctivitis, and unilateral lymphadenopathy. Many times this disease is found to be acute and self-limiting; however, up to one-quarter of cases may develop cardiac sequelae such as coronary artery aneurysms, myocardial infarction or heart failure which can lead to death.1–2 Current acute treatment options are targeted at reducing inflammation and preventing thrombosis. In a number of patients, disease may be refractory to initial therapy and require further management with immunomodulatory and cytotoxic agents as well as corticosteroids.1–3 This article gives a brief overview of Kawasaki disease and discusses evidence-based treatment options for refractory disease that are more familiar to clinicians, with a focus on intravenous immunoglobulin (IVIG), corticosteroids, infliximab, cyclosporin, cyclophosphamide, and methotrexate therapy.
EPIDEMIOLOGY
Kawasaki disease is diagnosed predominantly in infants and young children under 5 years of age, with most cases occurring in infants.1,2 It is the leading cause of heart disease in this patient population. Its incidence is higher in males than in females (1.5:1 ratio) and those who live in Asian countries, particularly Japan, with 1 in 100 Japanese children developing Kawasaki disease by the age of 5 years.1–3
In the United States, the Centers for Disease Control and Prevention (CDC) reports a yearly average incidence of 20 cases per 100,000 children younger than 5 years of age who develop Kawasaki disease.4 The most common time of the year for this disease to occur is in winter and early spring. The incidence is, again, highest among Asians and Pacific Islanders, with approximately 30 occurrences per 100,000 children, and lowest in Caucasians, with approximately 12 cases per 100,000 children.4
Mortality is estimated to be about 0.17% in the United States and about 0.08% in Japan.3 The time frame for which patients may be at the highest risk for death is from 15 to 45 days after the onset of fever; however, death has been reported to occur years after the acquisition of this disease. Missed diagnoses and cardiac sequelae are the culprits attributing to mortality.3
ETIOLOGY
While the cause of Kawasaki disease remains unknown, researchers suggest a transmittable and infectious origin.1 This hypothesis is supported by data that have been gathered from symptoms, laboratory findings, a seasonal time frames, epidemic patterns of history, and reported cases. An infectious agent may trigger an immunologic response in individuals who are at high risk, but a single infectious agent has yet to be identified.1,2
A genetic predisposition may play a role in the pathogenesis of this disease due to its high incidence in a particular region of the world and trends in genetic polymorphisms. The incidence of Kawasaki disease in siblings of an index case is 10 times higher than in the general population. Also, children who are born from parents with Kawasaki disease have twice the risk of having this disease in their lifetime.5 Patients with a variant or polymorphism in the inositoltriphosphate 3-kinase C (ITPKC) gene or the Fc fragment of IgG low-affinity II-a receptor (FCGR2A) gene have increased susceptibility to Kawasaki disease.6 The ITPKC gene plays a role in inhibiting T-cell activation, and the FCGR2A gene plays a role in the function of IgG.6 Reduction in ITPKC expression leads to enhanced T-cell activation and thus increased interleukin-2 (IL-2) expression, which in turn plays a role in the development of Kawasaki disease.5 Patients with this genetic polymorphism are also found to be at higher risk of developing coronary artery lesion and unresponsiveness to IVIG therapy.7 In summary, these genetic predispositions may lead to inflammation and activation of the host's immune system, resulting in the clinical characteristics and vascular damages observed in Kawasaki disease.
PATHOPHYSIOLOGY
Inflammation is the process that contributes to the clinical manifestations of Kawasaki disease. The offending agent, likely infectious, is thought to interact with the endothelial cells in the arteries, leading to activation of adhesion molecules, proinflammatory cytokines, and vascular endothelial growth factors. Vascular permeability then occurs which eventually leads to scarring and stenosis of the arterial wall, contributing to events such as aneurysms and myocardial infarctions. It is important to remember that Kawasaki disease may involve blood vessels throughout the body, and this is the reason that non-cardiac symptoms, which include bilateral conjunctivitis, fissured lips, and peripheral erythema, may appear early in the disease state.1–3
BRIEF OVERVIEW OF ACUTE STAGE TREATMENT
Initial treatment of Kawasaki disease aims at reducing inflammation of the vasculature beds and includes high-dose aspirin in combination with a single infusion of IVIG. This combination reduces the length of fever and the incidence of coronary artery aneurysms.8 If given within 10 days of disease onset, this therapy reduces the risk of developing cardiac sequelae at 1 to 2 months from 20% to 25% to an incidence of 2% to 4%.9
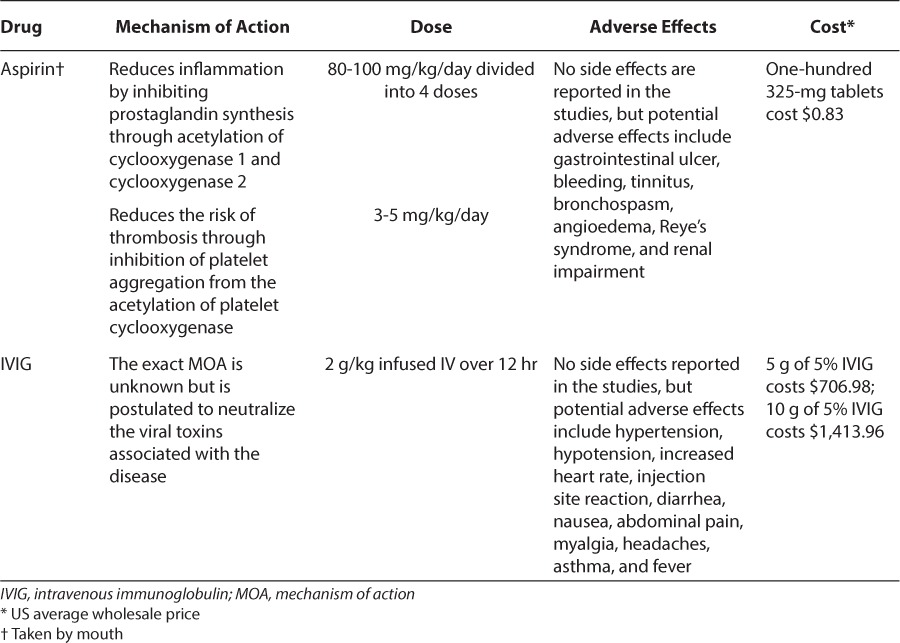
During the acute phase of the disease, large-dose aspirin (80–100 mg/kg/day) is used 4 times daily and is continued until the patient is afebrile for 48 to 72 hours or until day 14 of the illness. The dose is then reduced to 3 to 5 mg/kg/day during the convalescent stage and continued for 6 to 8 weeks, unless the patient has cardiac abnormalities, and then it will be continued indefinitely.8,10,11 Along with the initial aspirin dose, IVIG is given as a single infusion of 2 g/kg over 12 hours.12 Table 1 summarizes the data for initial therapies for Kawasaki disease, and Table 2 summarizes dosage and potential adverse effects of aspirin and IVIG.
Table 1.
Initial Therapy for Kawasaki Disease9
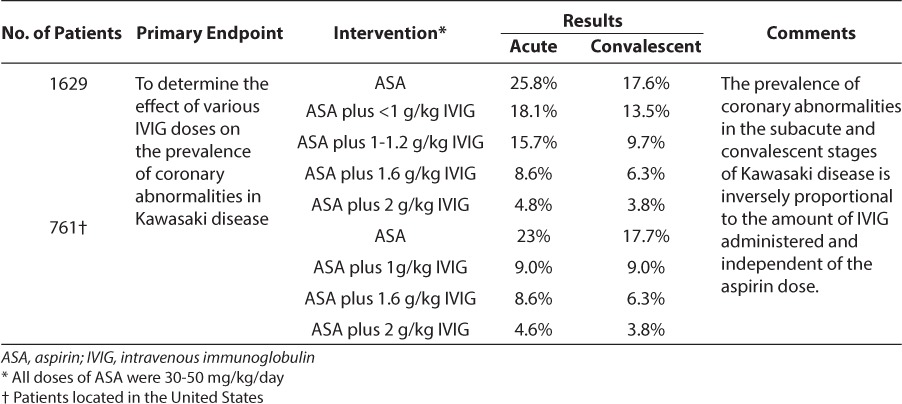
Table 2.
Initial Treatment Option for Kawasaki Disease8–12
REFRACTORY DISEASE AND TREATMENT OPTIONS
Refractory Kawasaki disease exists in 9.4 to 23% of patients treated with initial aspirin plus IVIG therapy.10 Patients are considered to have refractory disease if they develop or continue to have a fever within 36 hours of the IVIG infusion. These patients are at increased risk of developing cardiac abnormalities and will need additional treatment.11–13 The Egami scoring system has been developed to help predict whether a patient will develop refractory Kawasaki disease.14 This scoring system takes into account the patient's age, number of days the patient has been ill, platelet count, alanine transaminase level, and concentration of C-reactive protein (CRP). Although the Egami score works well in Japan, it has low sensitivity for predicting IVIG resistance when it is used in the United States.15 Multiple treatment options for refractory Kawasaki disease exist, including a second dose of IVIG or initiation of a new therapy such as corticosteroids, infliximab, cyclosporin, cyclophosphamide, or methotrexate.
Second Dose of Intravenous Immunoglobulin
According to the American Heart Association guidelines for Kawasaki disease, a second dose of IVIG 2 g/kg is recommended in patients for whom the initial dose has failed.1 Disease should be adequately controlled in up to 80% of patients who receive a second dose of IVIG.16 Burns et al17 evaluated the appropriate dosage of this second dose of IVIG, and the results indicated that the group of patients treated with 2 g/kg was less likely to develop coronary artery aneurysms than the group treated with 1 g/kg (5.9% vs. 57.1%, respectively; p = 0.014). No efficacy data exist today to support administering a third dose of IVIG therapy.
Corticosteroids
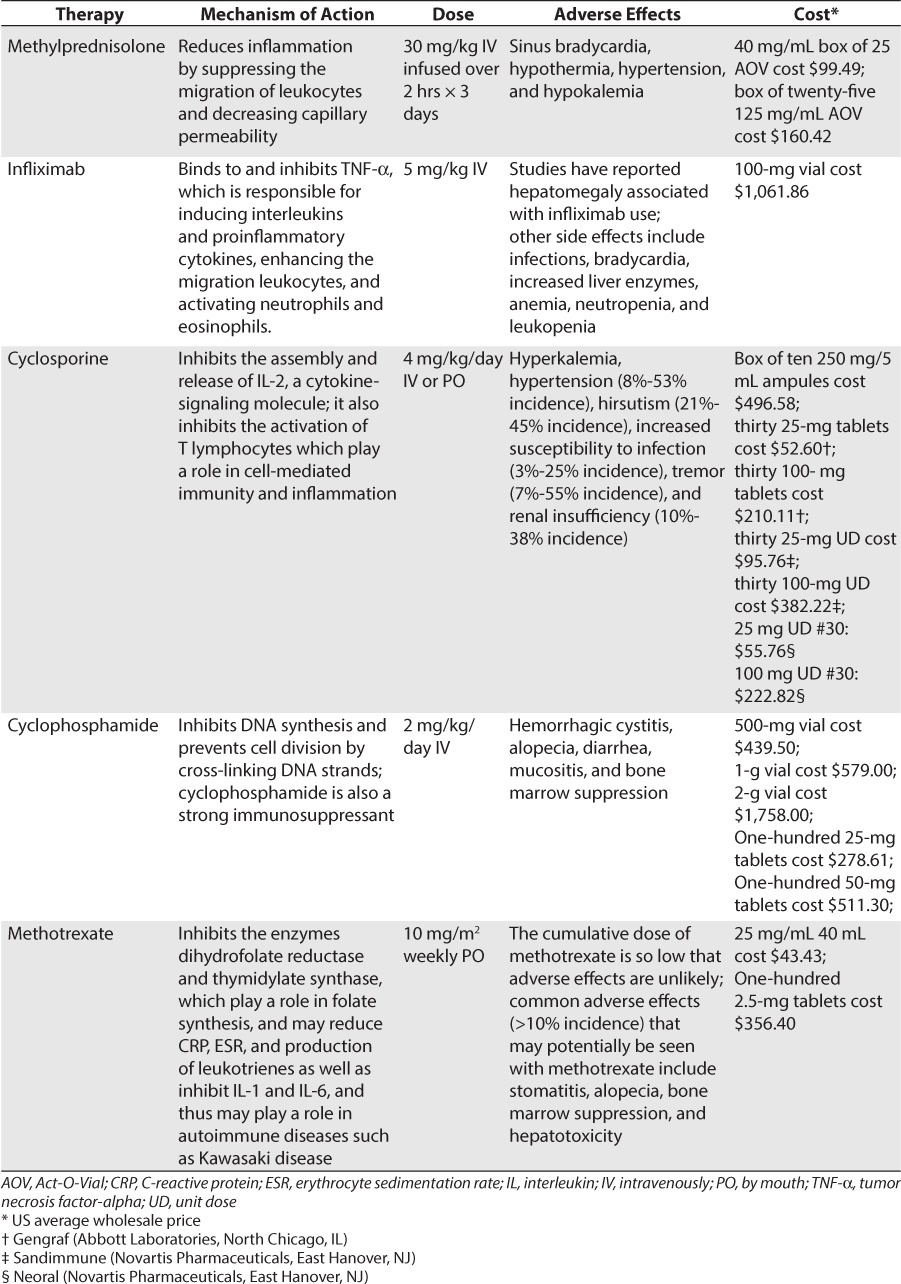
Corticosteroids help reduce the inflammation associated with Kawasaki disease through inhibition of the production of inflammatory mediators, suppressing the migration of leukocytes and decreasing capillary permeability. Corticosteroids inhibit phospholipase A2, an enzyme needed for the production of arachidonic acid and inflammatory mediators.13
Corticosteroid therapy is the treatment of choice for other states of vasculitis; however, their use in Kawasaki disease remains controversial, and they are reserved for those patients with disease that is refractory to IVIG therapy.10 In patients who have developed refractory Kawasaki disease, IV methylprednisolone therapy suppresses cytokine levels more rapidly than a second dose of IVIG.18 Even with this finding, the American Heart Association recommends using corticosteroids only in children for whom 2 or more trials of IVIG therapy have failed.1
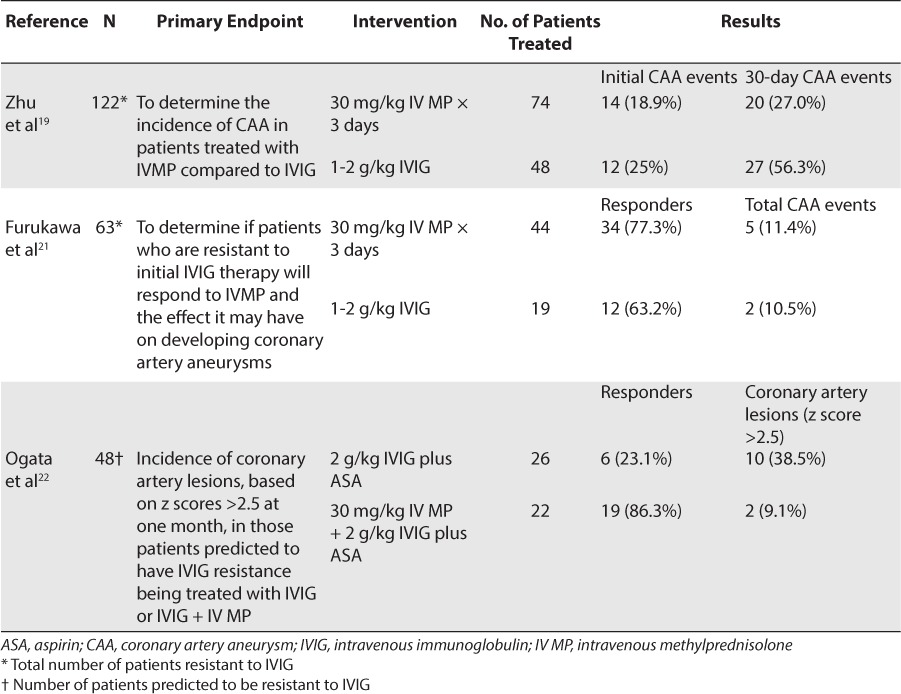
In a meta-analysis performed by Zhu et al,19 corticosteroid treatment improved the clinical course of refractory Kawasaki disease without increasing coronary artery lesions. Eleven studies were included in this meta-analysis, 7 of which investigated the addition of IV methylprednisolone to the initial treatment with large-dose IVIG plus aspirin, and the other 4 analyzed IV methylprednisolone used as a treatment in patients who had disease refractory to the initial large-dose IVIG therapy compared to a second dose of IVIG. In all 11 studies, the incidence of coronary artery aneurysms did not significantly increase in the patients treated with IV methylprednisolone compared to the control groups.19 These results indicated that IV methylprednisolone, without regard to the time of therapy, did not increase the risk of developing coronary artery aneurysms; and in those patients whose disease was refractory to IVIG therapy, the incidence of coronary artery aneurysms was actually reduced.
In the 4 studies of 122 patients who had disease refractory to the initial large-dose IVIG therapy, 74 patients were treated with IV methylprednisolone, and 48 patients were treated with a second dose of IVIG.19 Differences between development of coronary artery aneurysms in methylprednisolone-treated patients and that in controls were not significant (odds ratio [OR] = 0.87; confidence interval [CI] = 0.32–2.36; p = 0.78), and 1 study actually showed a decrease in the occurrence of coronary artery lesions compared to that in the control group.20 The incidence of coronary artery aneurysms at 1 month after treatment also did not show a significant increase in cardiac abnormalities, with 20 events in the patients treated with IV methylprednisolone compared to 27 events in the patients treated with a second dose of IVIG (p = 0.23). Those patients treated with IV methylprednisolone had a significant decrease in the duration of fever, and methylprednisolone reduced the number of patients needing additional treatment. The results of that study showed a potential benefit in adding IV methylprednisolone in patients with disease refractory to initial treatment or in those patients whose disease was predicted to be refractory to initial high-dose IVIG, with no additional risk of developing coronary abnormalities.19
In a study performed by Furukawa et al,21 a second dose of IVIG was compared to IV methylprednisolone in 63 IVIG-resistant patients. Of the 63 patients, 44 were treated with IV methylprednisolone, 30 mg/kg/day for 3 days, and 19 were treated with a second dose of IVIG at 1–2 g/kg. To determine the effect of the treatments, echocardiograms were performed 1 to 2 times a week. Of the patients who received IV methylprednisolone, 34 (77%) responded successfully compared to 12 (63%) patients who received a second dose of IVIG. The patients treated with IV methylprednisolone had a faster initial resolution of fever. All 44 methylprednisolone-treated patients became afebrile within 1 day of therapy, whereas only 12 of the 19 patients treated with a second dose of IVIG (63%) became afebrile. All of the patients who responded to the IV methylprednisolone or the second dose of IVIG did not develop coronary artery aneurysms; however, 5 of the 10 patients who did not respond to IV methylprednisolone developed coronary abnormalities, and 2 of the 7 patients who did not respond to a second dose of IVIG developed coronary abnormalities. These results suggested that IV methylprednisolone is a viable treatment option for patients who are refractory to initial IVIG therapy while not increasing the incidence of developing coronary abnormalities.21
If a patient is predicted to have resistance to IVIG, IVIG plus IV methylprednisolone therapy can be used. In a randomized controlled trial, 48 patients who were predicted to have IVIG resistance based on their Egami scores were assigned to receive either IVIG (n = 26) or IVIG plus methylprednisolone therapy (n = 22).22 Echocardiograms were performed on all patients before the initiation of treatment, at 36 hours, and again at 30 days. All patients received IVIG at 2 g/kg over 24 hours and aspirin, 30 mg/kg every 8 hours, until they were afebrile for 36 hours, and the aspirin dose was then reduced to 5 mg/kg/day. Those patients who received additional IV methylprednisolone with IVIG were given methylprednisolone, 30 mg/kg, as a 1-time dose administered over 2 hours prior to receiving their IVIG therapy. Of those patients treated with IVIG plus methylprednisolone, 86.4% had prompt resolution of fever compared to 23.1% in the IVIG group. Cardiac artery dimension z scores did not differ significantly between the 2 groups prior to treatment. The z scores, however, were significantly smaller at 36 hours in the patients treated with IVIG plus aspirin plus IV methylprednisolone than those in patients treated with IVIG and aspirin alone (p = 0.016). Echocardiograms at 30 days showed that the left main artery z score was also significantly smaller in the patients treated with methylprednisolone (p = 0.043). In addition, the incidence of coronary artery lesions at 30 days after treatment was significantly reduced in the patients treated with IV methylprednisolone compared to those treated with IVIG alone (9.1% vs 38.5%, respectively, p = 0.04). These results indicated that adding a corticosteroid to IVIG therapy was safe and effective in those patients predicted to have IVIG resistance.22 Table 3 summarizes the data for corticosteroid use in refractory Kawasaki disease.
Table 3.
Corticosteroid Use in Refractory Kawasaki Disease
Common adverse effects of IV methylprednisolone that were reported in the studies include sinus bradycardia, hypothermia, hypertension, and hypokalemia. In the study by Zhu et al,19 4 patients treated with IV methylprednisolone experienced serious adverse effects including sensorineural hearing loss, shock, and respiratory failure. One patient treated with IV methylprednisolone developed idiopathic thrombocytopenic purpura and subsequently died; however, the death was believed not to be related to the Kawasaki disease or the methylprednisolone treatment.19 In the study by Furukawa et al,21 5 patients developed hypertension, 3 developed hypothermia, and 3 developed sinus bradycardia. One young patient developed transient fibular nerve paralysis 6 days after the administration of methylprednisolone (disease day 8), but the association with corticosteroid use was not certain. Two patients who received IVIG plus IV methylprednisolone developed a fever within 24 hours of receiving the dose, but they became afebrile within 6 hours after the onset. Patients receiving the combination therapy also had a higher incidence of hypothermia, bradycardia, and hypertension; however, these adverse effects were transient and resolved by 36 hours.22 Overall, treatment with methylprednisolone appears to be safe and tolerable.
Infliximab
Infliximab is a monoclonal antibody that binds to and inhibits tumor necrosis factor-alpha (TNF-α). TNF-α is responsible for inducing interleukins and proinflammatory cytokines, enhancing the migration of leukocytes, and activating neutrophils and eosinophils. TNF-α inhibitors can reduce the inflammation associated with Kawasaki disease. TNF-α plasma concentrations and soluble TNF receptor concentrations are increased during the acute phase of Kawasaki disease and are associated with the development of coronary artery aneurysms.23
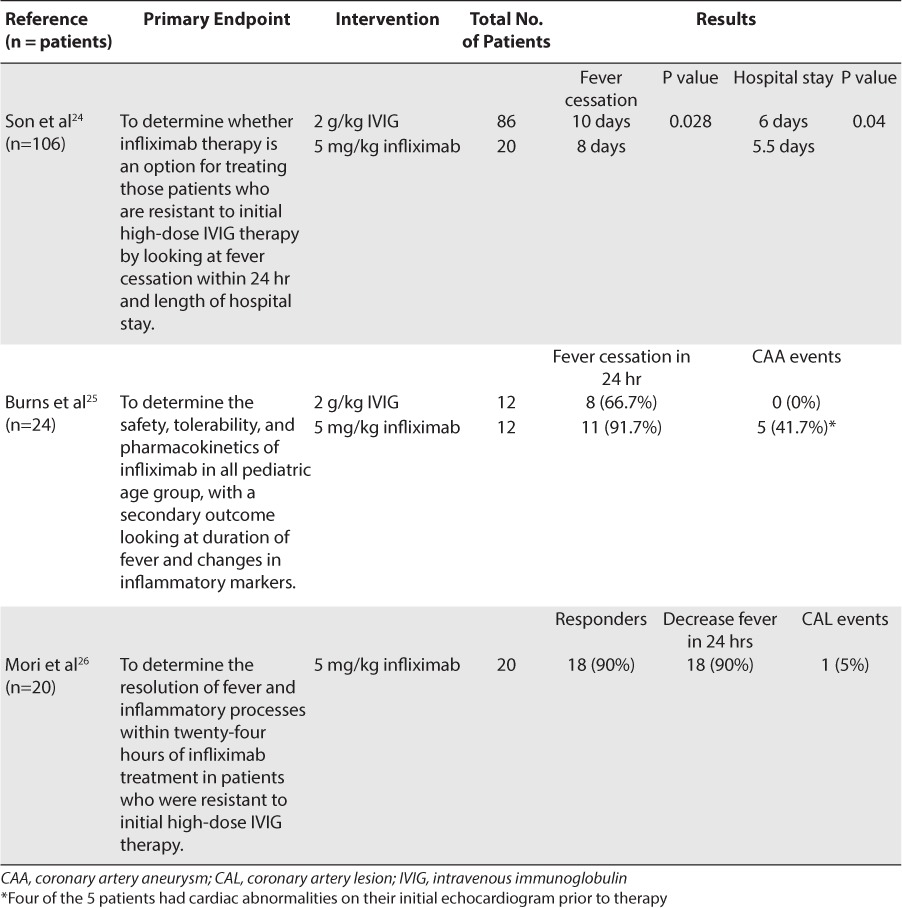
In a retrospective study of 106 patients who were resistant to initial IVIG therapy, the use of infliximab was compared to the use of a second dose of IVIG.24 Twenty patients were treated with infliximab, 5 mg/kg, and 86 patients were treated with a second dose of IVIG, 2 g/kg. The patients treated with infliximab had a significant reduction in the days of continued fever (8 days vs 10 days, respectively; p = 0.028) and a significantly shorter hospital stay (5.5 days vs 6 days, respectively; p = 0.04)]. The majority of the patients responded to the study drug and did not require retreatment (85% infliximab vs 76% second dose of IVIG; p = 0.55).
Burns et al25 published a prospective randomized trial in 24 patients with disease refractory to initial IVIG treatment. The study compared the safety, tolerability, and pharmacokinetics of infliximab with those of a second infusion of IVIG. Twelve patients received infliximab, 5 mg/kg infusion, whereas the other 12 patients received a second dose of IVIG, 2 g/kg. Secondary outcomes of the study included fever cessation within 24 hours of treatment and coronary dimensions based on z scores, with an aneurysm defined as z ≥ 4.25 Echocardiograms were performed on the subjects within 48 hours of randomization, again at 2 weeks, and again at 4 weeks. Eleven of the 12 patients who received infliximab had fever cessation compared to 8 of the 12 patients in the second IVIG-infusion group. Five of the subjects who received infliximab had evidence of coronary artery aneurysm during the 4-week study period; however, 4 of these 5 patients already had evidence of coronary abnormalities on their baseline echocardiograms. There were no significant difference in baseline characteristics between the 2 study groups, and both of the groups tolerated the treatment options well. That was the first study using infliximab as a treatment option in patients less than 12 months of age, and the results showed that the pharmacokinetics of the drug did not differ based on age. Burns et al25 demonstrated that infliximab is another treatment option for patients with initial IVIG therapy resistance.
In an open-label case series report, 20 patients resistant to large-dose IVIG therapy, up to 4 g/kg, were treated with 5 mg/kg of infliximab.26 They were evaluated 48 hours after the infusion for signs and symptoms of improvement and again at 30 days with an echocardiogram.26 All 20 patients showed increased echogenicity or mild dilation in their coronary arteries on the baseline echocardiogram. After the infliximab infusion, 18 of the 20 patients had a rapid decrease in their temperature within 24 hours and a gradual decrease in other symptoms within 3 days. The infliximab infusion also provided antiinflam-matory effects in these patients over the 3 days after the infusion. At the 30-day examination, 1 of the patients had a coronary artery lesion but had complete regression at 1 year.26 No adverse effects were reported in the patients treated with infliximab. This open-label case series showed that the majority of patients with refractory Kawasaki disease had rapid improvement in their inflammatory markers after the infliximab infusion.26 Only 2 patients were defined as resistant to infliximab and needed plasma exchange therapy.26 Table 4 summarizes the data for infliximab use in refractory Kawasaki disease.
Table 4.
Infliximab in Refractory Kawasaki Disease
In the study performed by Son et al,24 patients treated with infliximab had a higher prevalence of transient hepatomegaly than those who received IVIG (6 [19%] vs 1 [1.5%], respectively; p = 0.004); however, hepatomegaly was reported in patients who had Kawasaki disease due to the inflammatory nature of the disease. Similarly, in the randomized trial performed by Burns et al,25 the only adverse effect seen in the infliximab group was transient hepatomegaly that resolved within the 4-week study period. The cause of hepatomegaly is not known; it is believed to be either venous congestion due to cardiac dysfunction or acute inflammation of the liver and hydrops of the gallbladder.25
Although no serious adverse effects were reported in the trials and case reports using infliximab, infections, bradycardia, anemia, neutropenia, and leukopenia may develop. Even though these studies were not powered to determine the potential risk of infections or malignancy, it should be considered when treating children with this type of medication. Further investigation is required in larger study populations to determine the risk of malignancy associated with a one-time dose of 5 mg/kg infliximab in the pediatric population.27 With the current data available, the benefits of using infliximab for refractory Kawasaki disease appear to outweigh the potential risk of this medication.
Cyclosporine
Cyclosporine is postulated to reduce inflammation in patients with Kawasaki disease. It is included in a class of medications known as calcineurin inhibitors. In normal physiology, calcineurin is required for cytokine synthesis. Cyclosporine inhibits the assembly and release of IL-2, a cytokine-signaling molecule. It also inhibits the activation of T lymphocytes, which play a role in cell-mediated immunity and inflammation.27
Case reports and trials present controversial data in the efficacy of cyclosporine for the treatment of refractory Kawasaki disease. Kuijpers et al28 reported a case of a previously healthy 16-month-old male who was readmitted with refractory Kawasaki disease after initial standard therapy of aspirin and IVIG and 3 days of IV methylprednisolone failed. Cyclosporine 5 mg/kg IV was administered for 4 days before being switched to the oral formulation and was continued for a total of 7 weeks. Complete recovery was achieved within 2 weeks of starting cyclosporine; however, the patient's symptoms returned 20 days after discontinuation of therapy. Cyclosporine therapy was restarted with amelioration of symptoms. The patient was admitted for further observation during which he experienced cardiac arrest with sudden death attributable to myocardial infarction. The authors concluded that cyclosporine might not have been able to impact the coronary inflammation and proliferation as seen in this case due to the exaggerated inflammatory response of the vascular smooth muscle.28
Suzuki et al29 published a pilot study evaluating the use of cyclosporine in patients with refractory Kawasaki disease. Of the 329 Kawasaki disease patients who were admitted, 28 had refractory disease and were treated with oral cyclosporine at an initial dose of 4 mg/kg/day divided into 2 equal daily doses. The doses were adjusted to maintain a trough concentration of 60 to 200 ng/mL. Twenty-two patients (78.6%) became afebrile and had reduction in their CRP concentration within 5 days. The remaining 6 patients (21.4%) remained febrile after finishing their treatment with cyclosporine, and 4 of the 6 patients (1.2%) developed coronary arterial lesions either prior to or after the start of therapy. The only adverse effect reported was hyperkalemia without any occurrence of arrhythmias. The authors suggested that specific subgroups of patients might be resistant to cyclosporine due to reasons such as late timing of administration or insufficient suppressive effects for the severity of inflammation despite adequate serum concentrations.29
Another small interventional trial was conducted by Minami et al30 in which a continuous infusion of cyclosporine was administered in IVIG-resistant patients. Four patients between 9 months and 5 years of age were enrolled in the study. Two patients received cyclosporine 1.5 mg/kg/day, and the other 2 received 3 mg/kg/day. Cyclosporine serum concentrations (goal = 300–500 ng/mL) were monitored, and the patients were reexamined at 1 month. The patients who initially received the higher dose of cyclosporine became afebrile within 24 hours, whereas the patients in the low-dose treatment arm did not. Coronary artery lesions were not observed 1 month after the onset of disease. The authors concluded that a higher therapeutic serum concentration, and thus a higher initial dose, might be necessary to rapidly improve fever and decrease inflammation. No adverse effects were reported. Prospective studies are required with a larger sample size to determine the effectiveness of this cyclosporine dose.30
Hamada et al31 analyzed drug serum concentrations and inflammatory markers in 19 patients treated with oral cyclosporine 4 mg/kg/day for 14 days. Serum samples were collected on days 0, 7, and 14 of treatment. Cyclosporine trough concentrations were maintained between 60 and 200 ng/mL. Fourteen patients became afebrile within 5 days of cyclosporine treatment, and 5 patients remained febrile. One of the 14 responders and 2 of the 5 non-responders developed coronary arterial lesions. Reports from other researchers indicated that treatments with higher doses of oral cyclosporine (i.e., 10 mg/kg/day) had provided beneficial results and that higher doses might be required in patients who did not initially respond to 4 mg/kg/day.6 Nonetheless, further studies need to be conducted to determine the cytokines that are predominantly responsible for the formation of coronary arterial lesions so appropriate therapy can be developed to target these markers.28
A recent study by Tremoulet et al6 described 9 Japanese patients between 2 months and 11 years of age with Kawasaki disease, who were successfully treated with cyclosporine after not responding to multiple therapies. All 9 patients initially received standard therapy with daily aspirin and 2 doses of IVIG before receiving further management with either IV methylprednisolone or IV infliximab or participating in a clinical trial. Coronary artery aneurysms developed in 4 patients before the initiation of a calcineurin inhibitor but subsided soon afterward. Cyclosporine was initially started at doses ranging from 3 to 5.1 mg/kg/day IV, divided every 12 hours or 9.9 to 11.7 mg/kg/day by mouth, divided every 12 hours in order to determine a dose that would reach target serum trough levels of 50 to 150 ng/mL after 3 doses or a peak serum concentration range of 300 to 600 ng/mL. Six of the 7 patients treated with cyclosporine for persistent fevers became afebrile within 24 hours and the other within 72 hours. One patient was treated with cyclosporine due to steroid intolerance and therapy was able to be tapered once cyclosporine was initiated. One other patient received cyclosporine for persistent rash and arthritis, which resolved within 48 hours. The duration of therapy lasted from 2 weeks to 3 months in all patients treated with cyclosporine. The authors concluded that administration of IV cyclosporine until the cessation of fever followed by oral cyclosporine would yield rapid therapeutic levels and clinical response. A standardized treatment protocol was developed at the University of California at San Diego based on experience with this patient population. The IV cyclosporine regimen in this protocol is initiated at 3 mg/kg/day twice daily, followed by 10 mg/kg/day by mouth twice daily once the patients have been afebrile for greater than 24 hours. A tapered regimen (10% every 3 days over 1 month) is then started after 2 weeks of treatment or once the patient is afebrile or CRP concentration is ≤1 mg/dL and clinically improving, whichever is longer.6 Table 5 summarizes the data for cyclosporine use in refractory Kawasaki disease.
Table 5.
Cyclosporine Use in Refractory Kawasaki Disease
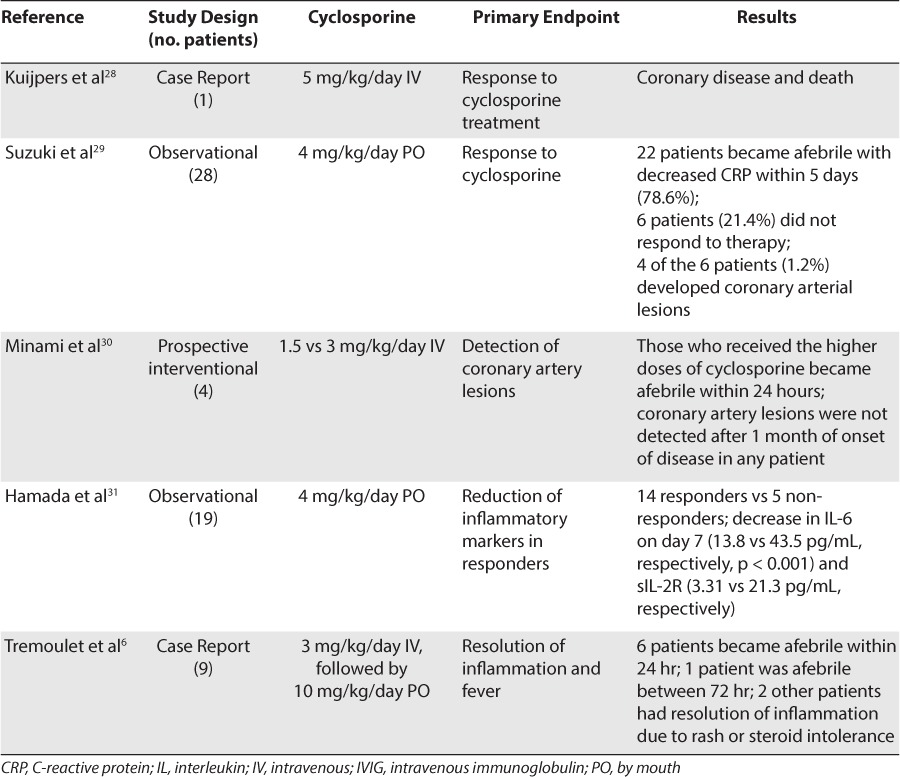
The adverse effects reported in the aforementioned studies were minimal. Suzuki et al29 reported hyperkalemia without any occurrence of further complications. Tremoulet et al6 reported hypomagnesemia in 2 patients, hirsutism in 2 patients, and acute otitis media in 1 patient. Generally, major adverse effects to consider include hypertension (8%–53%), hirsutism (21%–45%), increased susceptibility to infection (3%–25%), tremor (7%–55%), and renal insufficiency (10%–38%).6,27,29 Clinicians need to monitor serum trough concentrations of cyclosporine as well as the patients' blood pressure, complete blood count, and basic metabolic panel while the patients are receiving this therapy.
Cyclophosphamide
Cyclophosphamide is another immunosuppressant thought to decrease inflammation in Kawasaki disease. It is a prodrug in a class of medications known as alkylating agents. These agents inhibit DNA synthesis and prevent cell division by cross-linking DNA strands. Preventing cell division leads to cell death, and cytokines are some of the immune cells involved in this process. When immune cells are suppressed, inflammation is then reduced.27
Data for use of cyclophosphamide in Kawasaki disease are even scarcer than those for cyclosporine. Wallace et al32 reported the outcomes of pulse IV methylprednisolone and cyclophosphamide in patients with refractory Kawasaki disease in a 3-year retrospective analysis. Sixty-five patients were evaluated of whom 5 had persistent disease after 2 doses of IVIG (4 patients developed coronary artery thrombosis and 3 developed coronary aneurysm). Four of the 5 patients were then treated with IV methylprednisolone, 30 mg/kg/day, and 2 of these 5 were also treated with IV cyclophosphamide, 2 mg/kg/day. Methylprednisolone therapy in these 2 patients was unable to be tapered without reoccurrence of symptoms; however, the addition of cyclophosphamide allowed tapering. Cyclophosphamide and methylprednisolone were continued at discharge and tapered over 1.5 to 7 months without mentioning a dosage regimen. None of these patients experienced any further coronary aneurysms or death after finishing the therapy.32
Wallace et al32 did not report any adverse effects that might have potentially been experienced by the patients who were given cyclophosphamide in the study. Potential common adverse effects of cyclophosphamide physicians should be familiar with include hemorrhagic cystitis, alopecia, diarrhea, mucositis, and bone marrow suppression (frequency not defined).27 Clinicians need to monitor the patients' complete blood count, serum electrolytes, and serum creatinine while the patients are taking this therapy.
Methotrexate
Methotrexate, initially used as an anticancer agent in the 1950s, is an antimetabolite that inhibits the synthesis, replication, and repair of DNA. It inhibits the enzymes dihydrofolate reductase and thymidylate synthase, which play a role in folate synthesis. This process is necessary for the production of purines that are the building blocks of DNA. Methotrexate has also been thought to reduce CRP, erythrocyte sedimentation rate, and the production of leukotrienes as well as to inhibit IL-1 and IL-6. Thus, methotrexate may play a role in autoimmune diseases such as Kawasaki disease.27
In 2002, Lee et al33 reported the case of a 6-year-old male with refractory Kawasaki disease who was treated with both methotrexate, 10 mg/m2 weekly, and dexamethasone, 0.6 mg/kg/day, starting on day 38 from the onset of illness. The dosage forms of methotrexate and dexamethasone were not stated. Up until that point, the patient had already received several courses of IVIG without clinical improvement and had developed dilation of the right coronary artery. Corticosteroids were then started, with a transient improvement before methotrexate was added to the regimen. After the administration of methotrexate, the patient remained afebrile and was discharged with a regimen of weekly methotrexate and aspirin. Intended duration of therapy or adverse effects were not reported.33
Ahn et al34 evaluated the effect of small-dose methotrexate in 4 cases of refractory Kawasaki disease in patients between 8 months and 8 years of age. All 4 patients had been treated with aspirin, IVIG, and IV dexamethasone with recurrent symptoms or no response. After treatment with weekly PO methotrexate, 10 mg/m2, clinical and laboratory improvements were seen without any progression of cardiac abnormalities. The duration of treatment varied from 3 weeks to 7 months depending on the clinical improvement of each patient. The patients did not experience any adverse effects associated with the methotrexate therapy.34
Lee et al35 reported similar cases in which 17 patients with persistent fevers after initial therapy were treated with methotrexate at a low dose of 10 mg/m2 PO weekly. Fevers were reduced within 24 hours of methotrexate therapy (38.6°C vs 37.0°C, respectively; p < 0.001), and average CRP concentration was remarkably reduced after 1 week (8.9 mg/dL vs 1.2 mg/dL, respectively; p < 0.001). Methotrexate was found to rapidly reduce fever and inflammatory markers without provoking any adverse effects; however, the authors suggested that methotrexate should be further assessed for the improvement of coronary artery outcome.35 Table 6 summarizes the data for methotrexate use in refractory Kawasaki disease.
Table 6.
Methotrexate Use in Refractory Kawasaki Disease
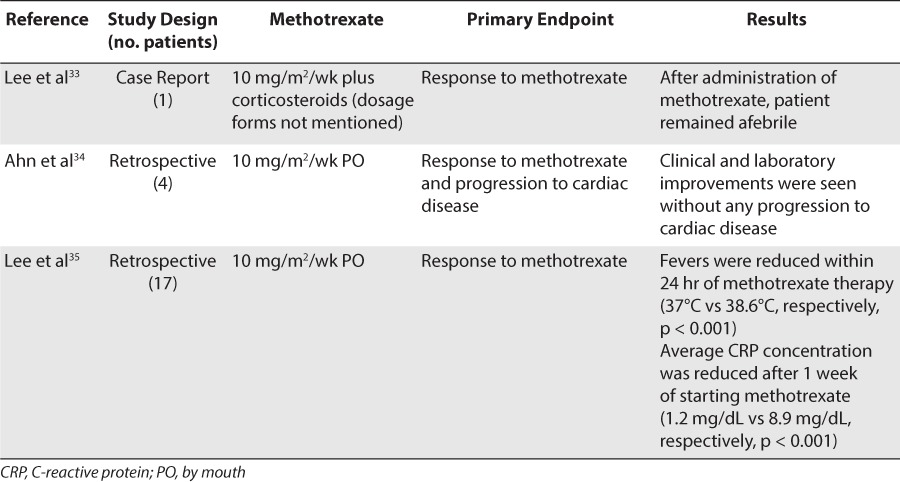
Clinical studies and case reports regarding methotrexate therapy for refractory Kawasaki disease have shown either minimal or no adverse effects. Lee et al35 suggested that the cumulative dose of methotrexate is so low in this setting that adverse effects are unlikely to be observed.35 Common adverse effects (>10%) that may potentially be seen with methotrexate include stomatitis, alopecia, bone marrow suppression, and hepatotoxicity.34 Clinicians should monitor patient's complete blood counts and liver function tests while they are taking this therapy.
CLINICAL APPLICATION
Refractory Disease
The evidence supporting treatment of refractory Kawasaki disease is scarce, consisting mainly of observational studies and case series. For this reason, this topic of interest is very controversial. There are many agents that may be considered for refractory disease. A repeated dose of IVIG seems to be the common practice in many reported cases, before other treatment options are considered; however, patients who do not respond to a repeated dose of IVIG have further delay of adequate therapy and are at increased risk of developing cardiac sequelae and death. Identifying and treating those patients who may have refractory Kawasaki disease is of utmost importance.
Although definite conclusions cannot be drawn from the small pool of evidence, it may be worthwhile to consider initiating corticosteroids after the first treatment failure of IVIG instead of waiting until after the second treatment failure. Patients who are predicted to fail initial IVIG therapy based on an Egami score of ≥3 have benefitted from initial treatment with IVIG in combination with corticosteroids.22 Few cases reports have noted recurrent fever and clinical symptoms as corticosteroid therapy is tapered;32 therefore, an additional agent for refractory disease such as cyclophosphamide or methotrexate may be added to allow for this taper to occur. Corticosteroids may exacerbate symptoms in patients with a history of uncontrolled diabetes or venous thromboembolism; patients with these clinical outcomes will likely benefit from other treatment options. Patients with a history of seizures or uncontrolled hypertension should also explore treatment options other than cyclosporine due to its potential adverse effects. Cyclophosphamide and methotrexate may cause myelosuppression and should not be used in patients who may be taking concurrent myelosuppressive medications for other disease states. It is also important to be vigilant in all patients who have renal or hepatic dysfunction when initiating these medications.
Beyond the use of corticosteroids, it is difficult to differentiate among the efficacies of other agents in refractory disease. As mentioned before, specific patient characteristics, adverse effect profiles of the drug, and possible contraindications for the patient should be taken into account when choosing the therapy. Table 7 summarizes the major treatment options and their proposed dosages for refractory Kawasaki disease.
Table 7.
CONCLUSIONS
About 20% of patients with Kawasaki disease will be refractory to initial therapy. The lack of randomized, controlled studies makes treatment of refractory disease difficult. Delayed control of the symptoms may increase morbidity and mortality. Current treatment options for refractory disease are based mainly on observational studies and case reports. Until larger randomized, controlled trials become available to give more guidance on therapy for this stage of disease, clinicians should use the available data in conjunction with their clinical expertise and the adverse effect profile of the available drugs to make treatment decisions.
ABBREVIATIONS
- CRP
C-reactive protein
- IgG
immunoglobulin G
- IL
interleukin
- IV
intravenous
- IVIG
intravenous immunoglobulin
- TNF-α
tumor necrosis factor-alpha
Footnotes
Disclosures The authors declare no conflicts or financial interest in any product or service mentioned in the manuscript, including grants, equipment, medications, employment, gifts, and honoraria.
REFERENCES
- 1.Newburger JW, Takahashi M, Gerber MA et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the committee on rheumatic fever, endocarditis and Kawasaki disease, council on cardiovascular disease in the young. American Heart Association. Circulation. 2004;110(17):2747–2771. doi: 10.1161/01.CIR.0000145143.19711.78. [DOI] [PubMed] [Google Scholar]
- 2.Dajani AS, Taubert KA, Gerber MA et al. Diagnosis and therapy of Kawasaki disease in children. Circulation. 1993;87(5):1776–1780. doi: 10.1161/01.cir.87.5.1776. [DOI] [PubMed] [Google Scholar]
- 3.Senzaki H. Long-term outcome of Kawasaki disease. Circulation. 2008;118(25):2763–2772. doi: 10.1161/CIRCULATIONAHA.107.749515. [DOI] [PubMed] [Google Scholar]
- 4.US Centers for Disease Control and Prevention. Kawasaki syndrome. http://www.cdc.gov/kawasaki/. Accessed June 11, 2014.
- 5.Hata A, Onouchi Y. Susceptibility genes for Kawasaki disease: toward implementation of personalized medicine. J Hum Genet. 2009;54(2):67–73. doi: 10.1038/jhg.2008.9. [DOI] [PubMed] [Google Scholar]
- 6.Tremoulet AH, Pancoast P, Franco A et al. Calcineurin inhibitor treatment of intravenous immunoglobulin-resistant Kawasaki disease. J Pediatr. 2012;161(3):506–512. doi: 10.1016/j.jpeds.2012.02.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Onouchi Y, Gunji T, Burns J et al. ITPKC functional polymorphism associated with Kawasaki disease susceptibility and formation of coronary artery aneurysms. Nat Genet. 2008;40(1):35–42. doi: 10.1038/ng.2007.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Saulsbury FT. Comparison of high-dose and low-dose aspirin plus intravenous immunoglobulin in the treatment of Kawasaki syndrome. Clin Pediatr (Phila) 2002;41(8):597–601. doi: 10.1177/000992280204100807. [DOI] [PubMed] [Google Scholar]
- 9.Terai M, Shulman ST. Prevalence of coronary artery abnormalities in Kawasaki disease is highly dependent on gamma globulin dose but independent of salicylate dose. J Pediatr. 1997;131(6):888–893. doi: 10.1016/s0022-3476(97)70038-6. [DOI] [PubMed] [Google Scholar]
- 10.Dimitriades VR, Brown AG, Gedalia A. Kawasaki disease: pathophysiology, clinical manifestations, and management. Curr Rheumatol Rep. 2014;16(6):423. doi: 10.1007/s11926-014-0423-x. [DOI] [PubMed] [Google Scholar]
- 11.Newburger JW, Takahashi M, Gerber MA et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the committee on rheumatic fever, endocarditis, and Kawasaki disease, council on cardiovascular disease in the young. American Heart Association. Pediatrics. 2004;114(6):1708–1733. doi: 10.1542/peds.2004-2182. [DOI] [PubMed] [Google Scholar]
- 12.Kuo HC, Yang KD, Chang WC et al. Kawasaki disease: an update on diagnosis and treatment. Pediatr Neonatol. 2012;53(1):4–11. doi: 10.1016/j.pedneo.2011.11.003. [DOI] [PubMed] [Google Scholar]
- 13.Freeman AF, Shulman ST. Refractory Kawasaki disease. Pediatr Infect Dis J. 2004;23(5):463–464. doi: 10.1097/01.inf.0000125893.66941.e0. [DOI] [PubMed] [Google Scholar]
- 14.Egami K, Muta H, Ishii M et al. Prediction of resistance to intravenous immunoglobulin treatment in patients with Kawasaki disease. J Pediatr. 2006;149(2):237–240. doi: 10.1016/j.jpeds.2006.03.050. [DOI] [PubMed] [Google Scholar]
- 15.Sleeper LA, Minich LL, McCrindle BM et al. Evaluation of Kawasaki disease risk scoring systems for intravenous immunoglobulin resistance. J Pediatr. 2011;158(5):831–835.e3. doi: 10.1016/j.jpeds.2010.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Han RK, Silverman ED, Newman A et al. Management and outcome of persistent or recurrent fever after initial intravenous [gamma] globulin therapy in acute Kawasaki disease. Arch Pediatr Adolesc Med. 2000;154(7):694–699. doi: 10.1001/archpedi.154.7.694. [DOI] [PubMed] [Google Scholar]
- 17.Burns JC, Capparelli EV, Brown JA et al. Intravenous [gamma]-globulin treatment and retreatment in Kawasaki disease. US/Canadian Kawasaki Syndrome Study Group. Pediatr Infect Dis J. 1998;17(12):1144–1148. doi: 10.1097/00006454-199812000-00009. [DOI] [PubMed] [Google Scholar]
- 18.Miura M, Kohno K, Ohki H et al. Effects of methylprednisolone pulse on cytokine levels in Kawasaki disease patients unresponsive to intravenous immunoglobulin. Eur J Pediatr. 2008;167(10):1119–1123. doi: 10.1007/s00431-007-0642-5. [DOI] [PubMed] [Google Scholar]
- 19.Zhu B, Lv H, Sun L et al. A meta-analysis on the effect of corticosteroid therapy in Kawasaki disease. Eur J Pediatr. 2012;171(3):571–578. doi: 10.1007/s00431-011-1585-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ogata S, Bando Y, Kimura S et al. The strategy of immune globulin resistant Kawasaki disease: a comparative study of additional immune globulin and steroid pulse therapy. J Cardiol. 2009;53(1):15–19. doi: 10.1016/j.jjcc.2008.08.002. [DOI] [PubMed] [Google Scholar]
- 21.Furukawa T, Kishiro M, Akimoto K et al. Effects of steroid pulse therapy on immunoglobulin-resistant Kawasaki disease. Arch Dis Child. 2008;93(2):142–146. doi: 10.1136/adc.2007.126144. [DOI] [PubMed] [Google Scholar]
- 22.Ogata S, Ogihara Y, Honda T et al. Corticosteroid pulse combination therapy for refractory Kawasaki disease: a randomized trial. Pediatrics. 2012;129(1):e17–23. doi: 10.1542/peds.2011-0148. [DOI] [PubMed] [Google Scholar]
- 23.Levin M, Burgner D. Treatment of Kawasaki disease with anti-TNF antibodies. Lancet. 2014;383(9930):1700–1703. doi: 10.1016/S0140-6736(14)60131-8. [DOI] [PubMed] [Google Scholar]
- 24.Son MB, Gauvreau K, Burns JC et al. Infliximab for intravenous immunoglobulin resistance in Kawasaki disease: a retrospective study. J Pediatr. 2011;158(4):644–649. doi: 10.1016/j.jpeds.2010.10.012. [DOI] [PubMed] [Google Scholar]
- 25.Burns JC, Best BM, Mejas A et al. Infliximab treatment of intravenous immunoglobulin-resistant Kawasaki disease. J Pediatr. 2008;153(6):833–838. doi: 10.1016/j.jpeds.2008.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mori M, Imagawa T, Hara R et al. Efficacy and limitation of infliximab treatment for children with Kawasaki disease intractable to intravenous immunoglobulin therapy: report of an open-label case series. J Rheumatol. 2012;39(4):864–867. doi: 10.3899/jrheum.110877. [DOI] [PubMed] [Google Scholar]
- 27.Lexicomp, Inc. Lexi-Drugs, version 2.0.1(165) 2014. http://www.lexi.com/. Accessed April 18, 2015.
- 28.Kuijpers TW, Biezeveld M, Achterhuis A et al. Longstanding obliterative panarteritis in Kawasaki disease: lack of cyclosporine A effect. Pediatrics. 2003;112(4):986–992. doi: 10.1542/peds.112.4.986. [DOI] [PubMed] [Google Scholar]
- 29.Suzuki H, Terai M, Hamada H et al. Cyclosporin A treatment for Kawasaki disease refractory to initial and additional intravenous immunoglobulin. Pediatr Infect Dis J. 2011;30(10):871–876. doi: 10.1097/INF.0b013e318220c3cf. [DOI] [PubMed] [Google Scholar]
- 30.Minami T, Shiraishi H, Oka K et al. Continuous infusion of cyclosporin A in intravenous immunoglobulin resistant Kawasaki disease patients. Jichi Med University J. 2011;34:109–114. [Google Scholar]
- 31.Hamada H, Suzuki H, Abe J et al. Inflammatory cytokine profiles during cyclosporin treatment for immunoglobulin-resistant Kawasaki disease. Cytokine. 2012;60(3):681–685. doi: 10.1016/j.cyto.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 32.Wallace CA, French JW, Kahn SJ et al. Initial intravenous gammaglobulin treatment failure in Kawasaki disease. Pediatrics. 2000;105(6):e78. doi: 10.1542/peds.105.6.e78. [DOI] [PubMed] [Google Scholar]
- 33.Lee MS, An SY, Jang GC et al. A Case of intravenous immunoglobulin-resistant Kawasaki disease treated with methotrexate. Yonsei Med J. 2002;43(4):527–532. doi: 10.3349/ymj.2002.43.4.527. [DOI] [PubMed] [Google Scholar]
- 34.Ahn SY, Kim DS. Treatment of intravenous immunoglobulin-resistant Kawasaki disease with methotrexate. Scand J Rheumatol. 2005;34(2):136–139. doi: 10.1080/03009740510026328. [DOI] [PubMed] [Google Scholar]
- 35.Lee TJ, Kim KH, Chun JK et al. Low-dose methotrexate therapy for intravenous immunoglobulin-resistant Kawasaki disease. Yonsei Med J. 2008;49(5):714–718. doi: 10.3349/ymj.2008.49.5.714. [DOI] [PMC free article] [PubMed] [Google Scholar]


