Abstract
The aim of this study is to investigate the expression of ribosome-binding protein 1 (RRBP1) in invasive breast cancer and to analyze its relationship to clinical features and prognosis. RRBP1 expression was studied using real-time quantitative PCR and western blotting using pair-matched breast samples and immunohistochemical staining using a tissue microarray. Then the correlation between RRBP1 expression and clinicopathologic features was analyzed. RRBP1 mRNA and protein expression were significantly increased in breast cancer tissues compared with normal tissues. The protein level of RRBP1 is proved to be positively related to histological grade (P = 0.02), molecular subtype (P = 0.048) and status of Her-2 (P = 0.026) and P53 (P = 0.015). We performed a grade-stratified analysis of all patients according to the level of RRBP1 expression and found that RRBP1 overexpression highly affected overall survival in patients with early-stage (I and II) tumors (P = 0.042). Furthermore, Her-2 positive patients with negative RRBP1 expression had longer overall survival rates than those with positive RRBP1 expression (P = 0.031). Using multivariate analysis, it was determined that lymph node metastasis (LNM, P = 0.002) and RRBP1 expression (P = 0.005) were independent prognosis factors for overall survival. RRBP1 is a valuable prognostic factor in Her-2-positive breast cancer patients, indicating that RRBP1 is a potentially important target for the prediction of prognosis.
Keywords: Breast cancer, Her-2, histological grade, prognosis, RRBP1
Breast cancer is the leading cancer in women and represents the second leading cause of cancer death among all women.1 Her-2, one of the most well characterized breast cancer oncogenes, is found in approximately 25% of invasive breast cancers and is strongly associated with poor prognosis and a more aggressive phenotype.2,3 Overexpression of Her-2 has been demonstrated to promote breast cancer invasion and metastasis.4 In particular, tumors with Her-2 overexpression are known to be refractory to various types of chemotherapy and endocrine therapy and to be associated with shortened overall survival.5,6 Although breast cancer patients with Her-2 overexpression can benefit from a targeted therapy that uses the humanized monoclonal antibody trastuzumab or the Her-2 kinase inhibitor lapatinib, the prognosis of Her-2-positive patients is hard to predict.7,8 Thus, the identification of a new cancer biomarker, in addition to common clinicopathological risk factors to select Her-2-positive patients with good or bad outcomes, will assist the follow-up management of Her-2-positive breast cancer after surgery.
During the early stages of tumor development, cancer cells with high proliferation rates require the increased activity of endoplasmic reticulum protein folding, assembly, transport and subjected to endoplasmic reticulum stress (ERS). During tumorigenesis, an adaptive stress response promotes the unfolded protein response (UPR) and initiates the activation of survival cascade mechanisms. The activation of the UPR affects tumorigenesis and protects tumor cells from ERS.9 Ribosome-binding protein 1 (RRBP1), a membrane protein, was originally identified as a ribosome-binding protein on the rough endoplasmic reticulum. It is essential for the transportation and secretion of intracellular proteins in mammalian cells.10,11 RRBP1 has been studied in yeast, where it is a member of the ERS response and associated UPR.12 It has also been shown to interact with KIF5B, a motor protein highly expressed in several cancer cell lines.13 With regard to the possible role of RRBP1 in tumorigenesis and progression, RRBP1 overexpression has frequently occurred in colorectal cancer14 and lung cancer.15 However, thus far, there has been no evidence concerning the significance of RRBP1 mRNA and protein expression in breast cancer.
In the present study, we examine the expression of RRBP1 through real-time quantitative PCR (real-time qPCR) and western blotting using pair-matched breast samples and immunohistochemical staining using a tissue microarray (TMA), respectively. The correlations between RRBP1 expression and clinicopathologic features are analyzed.
Materials and Methods
Patients and clinical samples
This study used archival material from the Department of Pathology at Harbin Medical University Cancer Hospital, including tissue samples from 389 consecutive patients with histologically-confirmed breast cancer and 117 paired-normal tissue samples, all from 2006 (Table1). The patients received a minimum of four courses of anthracycline-based and/or taxane-based chemotherapy after surgery. Hormone treatment with tamoxifen or aromatase inhibitors was given to patients with hormone receptor positive (ER or PR, or both). Her-2-positive patients who agreed to receive anti-Her-2 targeted therapy were treated with adjuvant trastuzumab for 1 year at Harbin Medical University Cancer Hospital. Fresh cancer tissues and matched normal tissues from 48 patients were collected and stored at −80°C immediately after resection to extract protein and RNA. Cancer tissues and their paired-normal tissues were examined diagnostically by two pathologists. All of the patients had invasive breast cancer. The patients who presented with recurrent tumors, metastatic disease at presentation, bilateral tumors or other previous tumors, and those who had previously received neoadjuvant treatment were excluded. The tumor size at the largest diameter of the invasive cancer was measured in millimeters by the pathologist. Four-micron tissue sections were prepared from a formalin-fixed and paraffin-embedded sample. All breast cancer patients were routinely tested for ER(estrogen receptor), PR(progesterone receptor), Her-2, P53 and Ki67, assayed in paraffin-embedded, formation-fixed tissue using antibodies against the proteins ER, PR, Her-2, P53 and Ki67 (Zhong shan-Bio, Beijing, China). Immunohistochemical staining for ER and PR was performed using a conventional detection method and was considered positive if 1% or more of the nuclei in the invasive component of the tumor was stained.16 Positive staining for HER-2 was defined based on the percentage of tumor cells and the intensity of the membrane staining. A scored of 0 was given when no staining was observed or membrane staining of fewer than 10% of the tumor cells was observed. A faint or barely perceptible incomplete membrane staining detected in more than 10% of the tumor cells was scored as 1+. A weak to moderately complete membrane staining observed in more than 10% of the tumor cells was scored as 2+. A strong complete membrane staining observed in more than 10% of the tumor cells was scored as 3+. Scores of 0 to 1+ were regarded as negative, and 3+ were regarded as positive.17 We selected a Ki67 index of 14% or more Ki67-positive tumor nuclei as the best cut-point for human visual assessment.18 For P53, positive staining of more than 10% of the tumor cells was defined as positive tumor expression and staining of 10% or fewer of the cells as negative tumor expression.19 The present study was approved by the Ethical Committee of Harbin Medical University in Harbin, China. Informed consent was obtained from all the patients.
Table 1.
Clinicopathological composition of tumor patients
| Characteristics | Number of cases |
|---|---|
| Age (years) | |
| Median | 49 |
| Range | 28–78 |
| Tumor size (cm) | |
| ≤2 | 128 |
| >2 | 261 |
| LNM | |
| Negative | 170 |
| Positive | 219 |
| TNM stage | |
| I, II | 259 |
| III | 130 |
| Histological grade | |
| I, II | 135 |
| III | 254 |
| ER | |
| Negative | 226 |
| Positive | 163 |
| PR | |
| Negative | 171 |
| Positive | 218 |
| Her-2 | |
| Negative | 247 |
| Positive | 84 |
| Ki67 | |
| Negative | 194 |
| Positive | 195 |
| P53 | |
| Negative | 77 |
| Positive | 312 |
| Subtype | |
| Luminal A | 74 |
| Luminal B | 131 |
| Her-2 | 47 |
| Basal-like | 79 |
ER, estrogen receptor; LNM, lymph node metastasis; PR, progesterone receptor.
Follow up
The clinical and pathological records of all patients in the study were reviewed periodically. Examinations were performed every 4–6 months for the first 5 years and every 12 months thereafter during the follow-up period. Patients were followed regularly for a minimum of 5 years of follow up at Harbin Medical University Cancer Hospital. The clinical records were obtained from the departments providing follow-up care. Survival was calculated in months from the date of diagnosis to whichever of the following occurred first: the date of death, the date last known to be alive, or 20 November 2012, which was the follow-up cut-off date used in our analysis. The median follow-up time was 72 months (range, 3–82 months).
RNA preparation and reverse transcription
Total RNA was extracted according to the protocol of TRIzol reagent (Invitrogen, Beijing, China) after ensuring that tumors contained more than 75% tumor cells and approved by pathologists in the pathology department. RNA quality and concentration were measured using a GeneQuant pro (GE Healthcare, Piscataway, NJ, USA). cDNA was synthesized from 1.0 μg of total RNA in a 20 μL reaction mixture using a PrimeScript RT reagent Kit with gDNA Eraser (Takara Bio, Otsu, Japan).
Real-time quantitative PCR
Real-time quantitative (qPCR) was performed using the ABI 7500 Fast Sequence Detection System (Applied Biosystems, Foster City, CA, USA) and Faststart Universal SYBR Green Master (ROX) reagent (Roche Applied Science, Mannheim, Germany) according to the manufacturer's instructions. The primers of RRBP1 were designed as follows: forward, 5′TGAATCCTCCAAAGACCACA3′; reverse, 5′CTTTCCCTCTCGCGTCTCT 3′. GAPDH was applied as the internal reference and its primers were as follows: 5′AACGACCCCTTCATTGAC3′; reverse, 5′TCCACGACATACTCAGCAC3′. Amplification was performed under the following conditions: 95°C for 10 min, followed by 40 cycles of 95° for 15 s and of 60°C for 60 s. The results of the Real-time qPCR experiments were calculated using the  method. Experiments were performed in triplicate in the same reaction.
method. Experiments were performed in triplicate in the same reaction.
Western blotting analysis
Frozen tissue samples were homogenized in RIPA buffer consisting of 1% protease inhibitor mixture. The mixture was centrifuged at 14 000 g for 15 min at 4°C and the supernatant was obtained. Total proteins were quantified using the Pierce BCA Protein Assay Kit (Thermo Scientific, Rockford, IL, USA), and 30 μg of protein per sample was separated onto a denaturing polyacrylamide gel containing SDS, and then transferred to a methanol-activated PVDF filter membrane (Bio-Rad, Hercules, CA, USA). Before immunodetection, membranes were blocked within 5% nonfat dry milk. Primary antibodies, anti-RRBP1 (1:1000; rabbit polyclonal; Abcam, Cambridge, MA, USA), were diluted in the buffer and incubated at 4°C overnight. After subsequent washing with TBST, membranes were incubated with secondary antibody (HRP-conjugated anti-rabbit) for 1 h at room temperature. The experiment was repeated in triplicate. The bands were detected by enhanced chemiluminescence detection reagents (Applygen Technologies, Beijing, China).
Tissue microarrays
Tissue microarrays (TMA) allowed the examination of a single biomarker in a high-throughput fashion to test a large number of normal and cancerous tissues simultaneously. TMA blocks were obtained by punching a tissue cylinder (core) with a diameter of 1.5 mm through a histological representative area of each “donor” tumor block, which was then inserted into an empty “recipient” TMA paraffin block using a manual tissue arrayer, as described previously.20 After the construction of the array block, all the tissue blocks were cut with a microtome to 4 μm and affixed to the slide. Blocks from 389 invasive breast cancer patients and their matched normal breast samples were arrayed as triplicate spots of 1.5 mm diameter on slides.
Immunohistochemistry staining
The tissue sections were dried at 70°C for 3 h. After deparaffinization and hydration, sections were washed in PBS (3 min × 3). The washed sections were treated with 3% H2O2 in the dark for 5–20 min. After washing in distilled water, sections were washed in PBS (5 min × 3). Antigen retrieval was performed in citrate buffer (pH 6.0) at 100°C for 10 min. Each section was then treated with RRBP1 rabbit polyclonal antibodies (Abcam, Cambridge, MA; at a dilution of 1:200 solution) at 4°C overnight. After washing in PBS (5 min × 3), each section was incubated with secondary antibody at room temperature for 30 min. After washing in PBS (5 min × 3), each section was treated with diaminobenzadine working solution at room temperature for 3–10 min, and the slides were counterstained with hematoxylin. For negative controls, the primary antibody was substituted with PBS. The positive controls were lung cancer tumors with positive expression of RRBP1.15
Evaluation of ribosome-binding protein 1 protein expression by immunohistochemistry
Semiquantitative expression levels were based on the intensity of staining in a series of randomly selected ten high-power fields, which was considered as representative of the average in a 400 × magnification field. Staining intensity was classified into four groups: level 0 (no staining), level 1 (0–20% of tumor cells stained), level 2 (20–50% of tumor cells stained) and level 3 (>50% of tumor cells stained).15 Overall expression was then graded as either negative expression (level 0) or positive expression (level 1–3).
Statistical analyses
All analyses were performed using statistical software (SPSS 17.0 for Windows; SPSS, Chicago, IL, USA). Associations between RRBP1 expression and patients’ clinicopathological features, including age, tumor size, lymph node metastasis (LNM), TNM stage, histological grade, molecular subtype, and status of ER, PR, Her-2, Ki67 and P53 were assessed using the χ2-test. The Kaplan–Meier method was used to estimate overall survival (OS). The influence of different variables on survival was assessed using Cox univariate and multivariate regression analyses. Risk ratios and their 95% confidence intervals (CI) were recorded for each marker. For continuous variables, student's t-test was performed. The level of significance was set at P < 0.05.
Results
Patients’ characteristics
Analyses for the immunoreactivity of RRBP1 were performed using specimens from 389 untreated female invasive breast cancer patients. The clinical characteristics of the patients are listed in Table1. The median age of the patients was 49 years old (range, 28–78). Of all the patients, LNM were present in 219 patients (56.3%), and absent in 170 patients (43.7%). A total of 259 (66.6%) patients were classified at stage I and II, and 130 (33.4%) were stage III. A total of 135 (34.7%) patients were classified as grade I and II, and the remaining patients were grade III (65.3%).
Ribosome-binding protein 1 mRNA and protein expression in breast cancer tissues
The analysis of real-time qPCR was used to confirm mRNA level. The mean expression value of RRBP1 mRNA in cancer tissue (24.10 ± 45.03, normalized by GAPDH gene expression) was significantly higher than the value in normal breast tissues (6.27 ± 12.18, P = 0.001). We then defined twofold changes as upregulation of RRBP1 mRNA between the cancer and corresponding normal tissues (Fig.1a). The results revealed that 83.3% (40/48) of breast cancer tissues expressed RRBP1 compared to the matched normal tissues.
Figure 1.
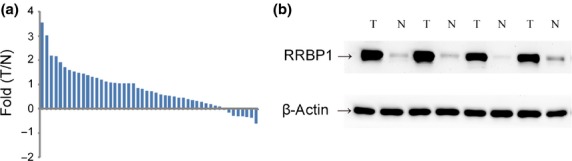
Ribosome-binding protein 1 (RRBP1)-elevated expression in fresh breast cancer tissues. (a) Histogram of RRBP1 mRNA expression in breast cancer. The RRBP1 mRNA expression was calculated using the  method and the relative expression in each patient was presented as a ratio of T (tumor tissue)/N (normal tissue). (b) Representative western blot analysis of RRBP1 expression in breast tissues. The levels of β-actin were used as an internal control.
method and the relative expression in each patient was presented as a ratio of T (tumor tissue)/N (normal tissue). (b) Representative western blot analysis of RRBP1 expression in breast tissues. The levels of β-actin were used as an internal control.
The difference in RRBP1 expression between cancer and normal tissues reflected at protein level was investigated using western blotting and immunohistochemistry. Overall, breast cancer tissues exhibited dramatically higher levels of RRBP1 protein expression compared with normal tissues. Significantly greater expression of RRBP1 protein was found in cancer tissues than in matched normal tissues during western blotting analysis (Fig.1b). Immunoreactivity for RRBP1 protein is shown in brown color and presented diffusely in the cytosolic regions of invasive breast cancer cells (Fig.2). In 17 of 117 paired-normal tissues, we demonstrated positive RRBP1 expression (i.e. 14.5% of the normal tissues). Of the 389 breast cancer specimens, we found positive expression in 164 (42.2%) samples (P < 0.001).
Figure 2.
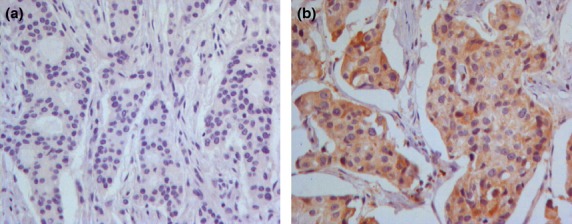
Immunohistochemical staining of ribosome-binding protein 1 (RRBP1) in breast tissues. (a) Negative staining in normal breast tissue. (b) RRBP1 positive expression in breast cancer tissue.
Relationship between immunoreactivity of ribosome-binding protein 1 and clinicopathological features
We analyzed the associations between levels of RRBP1 expression and a series of clinicopathological characteristics, including age, tumor size, LNM, TNM stage, histological grade molecular subtypes, and status of ER, PR, Her-2, Ki67 and P53 in breast cancer patients (Table2). RRBP1 was significantly correlated with histological grade, Her-2, P53 and molecular subtypes. We detected RRBP1 expression in 73 of the 135 (49.7%) grade I and II patients, and in 91 of 254 (37.6%) grade III patients (P = 0.020). The data demonstrated that the RRBP1 expression was much greater in early-stage than in late-stage invasive breast cancer. A total of 45 of 84 patients (53.6%) who were Her-2 positive had significantly higher incidence of RRBP1 expression than those patients who were Her-2 negative (39.7%, 98 of 247, P = 0.026). RRBP1 was present in 29.9% (23 of 77) and 45.2% (141 of 312) of patients in the P53 negative group and P53 positive group, respectively (P = 0.015). Meanwhile, expression of RRBP1 protein was associated with subtypes of breast cancer; the positive rate was higher in the Her-2-positive subtype (P = 0.048). We found that there were no significant correlations between RRBP1 and clinicopathological parameters, including the patients’ age (P = 0.521), tumor size (P = 0.656), LNM (P = 0.630), TNM stage (P = 0.967) and status of ER (P = 0.272), PR (P = 0.860) and Ki67 (P = 0.325).
Table 2.
Correlation between ribosome-binding protein 1 expression and clinicopathological features
| Characteristics | Cases | Negative | Positive | P-value |
|---|---|---|---|---|
| Age | ||||
| >50 | 209 | 124 (59.3) | 85 (40.7) | 0.521 |
| ≥50 | 180 | 101 (56.1) | 79 (43.9) | |
| Tumor size | ||||
| ≤2 | 128 | 72 (56.2) | 56 (43.8) | 0.656 |
| >2 | 261 | 153 (58.6) | 108 (41.4) | |
| LNM | ||||
| Negative | 170 | 96 (56.5) | 74 (43.5) | 0.630 |
| Positive | 219 | 129 (58.9) | 90 (41.1) | |
| TNM stage | ||||
| I, II | 259 | 150 (57.9) | 109 (42.1) | 0.967 |
| III | 130 | 75 (57.7) | 55 (42.3) | |
| Histological grade | ||||
| I, II | 135 | 74 (50.3) | 73 (49.7) | 0.020 |
| III | 254 | 151 (62.4) | 91 (37.6) | |
| ER status | ||||
| Negative | 226 | 136 (60.2) | 90 (39.8) | 0.272 |
| Positive | 163 | 89 (54.6) | 74 (45.4) | |
| PR status | ||||
| Negative | 171 | 103 (60.2) | 68 (39.8) | 0.860 |
| Positive | 218 | 122 (56.0) | 96 (44.0) | |
| Her-2 status | ||||
| Negative | 247 | 149 (60.3) | 98 (39.7) | 0.026 |
| Positive | 84 | 39 (46.4) | 45 (53.6) | |
| Ki67 status | ||||
| Negative | 194 | 117 (60.3) | 77 (39.7) | 0.325 |
| Positive | 195 | 108 (55.4) | 87 (44.6) | |
| P53 status | ||||
| Negative | 77 | 54 (70.1) | 23 (29.9) | 0.015 |
| Positive | 312 | 171 (54.8) | 141 (45.2) | |
| Subtype | ||||
| Luminal A | 74 | 41 (55.4) | 33 (44.6) | 0.048 |
| Luminal B | 131 | 76 (58.0) | 55 (42.0) | |
| Her-2 | 47 | 19 (40.4) | 28 (59.6) | |
| Basal-like | 79 | 52 (65.8) | 27 (34.2) | |
ER, estrogen receptor; LNM, lymph node metastasis; PR, progesterone receptor.
Prognostic significance of ribosome-binding protein 1 expression in different grades of breast cancer patients
To eliminate the effect of histological grade on prognosis, we performed a grade-stratified analysis of all patients according to the level of RRBP1 expression and found that RRBP1 overexpression highly affected OS in patients with early-stage (Ι and II) tumors (P = 0.042). However, no such association was observed in the breast cancer patients with late-stage tumors (P = 0.584). The survival curves for the breast cancer patients in the two groups stratified by histological stage are illustrated in Figure3.
Figure 3.
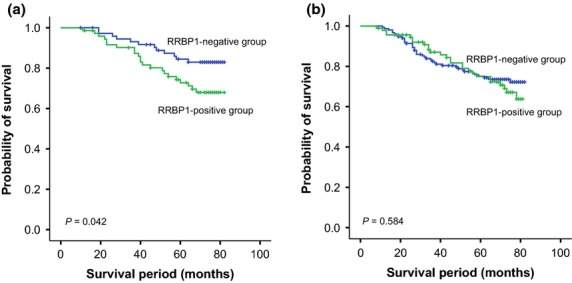
A stage-stratified analysis of ribosome-binding protein 1 (RRBP1) expression in patients with breast cancers. Survival curves showed the correlation of RRBP1 with overall survival (OS) in breast cancer patients in different histological grades: (a) OS in histological grade I and II (P = 0.042); and (b) OS in histological grade III (P = 0.584).
Predictive significance of ribosome-binding protein 1 expression in Her-2 positive invasive breast cancer patients
The Kaplan–Meier 5-year survival curve stratified for RRBP1 expression in Her-2-positive patients is shown in Figure4. Among the 84 Her-2-positive patients, RRBP1 expression showed significant effects on OS (P = 0.031). These data indicate that RRBP1 expression is associated with worse OS in Her-2-positive breast cancer.
Figure 4.
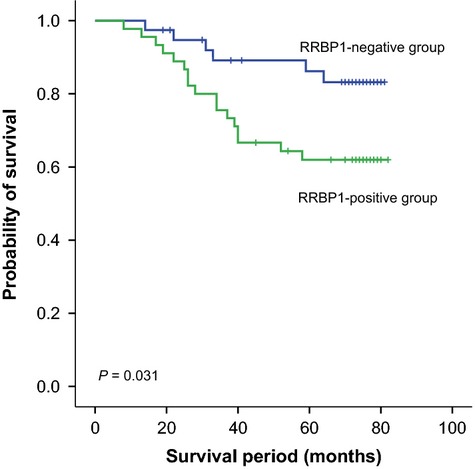
Kaplan–Meier analysis for overall survival (OS) based on ribosome-binding protein 1 (RRBP1) expression in Her-2-positive breast cancer patients.
Both univariate and multivariate survival analyses were used to evaluate the effects of RRBP1 expression and clinicopathological characteristics on prognosis in Her-2-positive patients. Univariate analyses of OS using Cox regression analysis identified LNM (P = 0.012), TNM stage (P = 0.007) and RRBP1 expression (P = 0.039) as significant prognostic predictors. Other features had no prognostic value. Using multivariate analysis, we found that LNM (P = 0.002) and RRBP1 expression (P = 0.005) were independent prognostic factors (Table3).
Table 3.
Prognostic factors in the Cox proportional hazards model
| Variables | Risk ratio | Univariate 95% confidence interval | P-value | Risk ratio | Multivariate 95% confidence interval | P-value |
|---|---|---|---|---|---|---|
| Age (years) | ||||||
| ≥50/<50 | 1.339 | 0.587–3.055 | 0.487 | |||
| Tumor size (cm) | ||||||
| ≤2.0/>2.0 | 0.986 | 0.366–2.656 | 0.977 | |||
| LNM | ||||||
| Positive/negative | 4.004 | 1.359–11.795 | 0.012 | 5.581 | 1.859–16.751 | 0.002 |
| TNM stage | ||||||
| I and II/III | 3.244 | 1.371–7.672 | 0.007 | |||
| Histological grade | ||||||
| I and II/III | 1.461 | 0.632–3.378 | 0.375 | |||
| ER status | ||||||
| Positive/negative | 0.244 | 0.057–1.041 | 0.057 | |||
| PR status | ||||||
| Positive/negative | 0.732 | 0.289–1.858 | 0.512 | |||
| Ki67 status | ||||||
| Positive/negative | 2.790 | 0.828–9.395 | 0.098 | |||
| P53 status | ||||||
| Positive/negative | 0.514 | 0.202–1.306 | 0.162 | |||
| Adjuvant trastuzumab | ||||||
| Received/not received | 0.414 | 0.176–0.978 | 0.044 | |||
| RRBP1 expression | ||||||
| Positive/negative | 2.667 | 1.051–6.769 | 0.039 | 3.874 | 1.501–9.997 | 0.005 |
ER, estrogen receptor; LNM, lymph node metastasis; PR, progesterone receptor.
Discussion
Ribosome-binding protein 1 is an endoplasmic reticulum membrane protein10,21,22 that is essential for ribosome binding and for the translocation of nascent proteins across the membrane of the rough endoplasmic reticulum.23 It has been shown to play an important role in procollagen biosynthesis in secretory tissues and in the terminal differentiation of secretory tissues.10,21–26 Recent studies reveal that the RRBP1 level affects the development of tumorigenesis.14,15 RRBP1 was highly expressed in lung cancer tissue compared with adjacent normal tissues, as assessed by immunohistochemistry using lung cancer TMA and real-time qPCR using paired normal cancer tissues.15 Elevated levels of RRBP1 proteins have been detected in colorectal cancer tissues.14 Although several studies on the expression of RRBP1 in cancers have supported the hypothesis that overexpression of RRBP1 is a stage in tumor development, until now there has been no report of RRBP1 being involved in breast cancer development. In the present study, we detected that the expression of RRBP1 is upregulated at both mRNA and protein levels in breast cancer. Our results concur with the data analysis from the UCSC cancer browser (https://genome-cancer.ucsc.edu, TCGA), which shows higher RRBP1 expression levels in breast cancer tissues than in normal breast tissues (P < 0.001). Our results are consistent with published data and associate higher RRBP1 expression in human breast cancer tissues with increasing propensity for tumor formation.
We first investigated the elevated expression of RRBP1 in human invasive breast cancer at the level of mRNA and protein. We then attempted to correlate RRBP1 expression with clinicopathological features and survival. Overall, positive correlations of RRBP1 with histological grade (P = 0.020), Her-2 status (P = 0.026), P53 status (P = 0.015) and molecular subtypes (P = 0.048) have been demonstrated in the current study.
The relationships of many clinical, pathological and molecular factors to patient survival in invasive breast cancer have been investigated. Although prognosis in general correlates with clinical variables, such as tumor stage, it is currently difficult to predict the clinical outcome for individual patients with early-stage tumors. The endoplasmic reticulum is a key cellular organelle that is involved in protein homeostasis. When the normal mechanisms within the endoplasmic reticulum that ensure fidelity of protein folding are disturbed, unfolded proteins accumulate in the endoplasmic reticulum. Many known triggers for endoplasmic reticulum dysfunction lead to disturbances in protein folding in the endoplasmic reticulum.27–30 The initial response is to correct this through inhibition of protein synthesis and increased degradation of unfolded protein by way of proteasomes. Simultaneously, adaptive genes are activated which improve protein folding and help the cell adapt to the trigger for the UPR.31 During the early stage of tumor development, the activation of the UPR affects tumorigenesis and protects tumor cells from ERS.32 In our research, a statistically significant correlation was found between earlier grade and positive RRBP1 expression (P = 0.020), which suggested that RRBP1 overexpression might be an early event in the progression of invasive breast cancer. Moreover, previous studies have shown that RRBP1 is associated with the UPR.12,15 Therefore, RRBP1 could be a key player in the initial maintenance of breast cancer cell survival. Furthermore, by performing a grade-stratified analysis of all patients according to the level of RRBP1 expression, we found that RRBP1 overexpression highly affected OS in patients with early-grade (grade I and II) tumors (P = 0.042). We demonstrated that RRBP1 overexpression might be a prognostic indicator for early-stage invasive breast cancer.
Amplification of Her-2 gene is observed in approximately 25% of invasive breast cancers.2,3 In addition, Her-2 is also overexpressed in a variety of other human tumors, including ovarian,2 lung,33 gastric34,35 and oral36 cancers. Breast cancer tumors overexpressing Her-2 oncoprotein are associated with a more aggressive clinical behavior.37,38 The current standard of care for Her-2-positive breast cancer is therapy with the humanized monoclonal antibody tratuzumab,39 which targets the C-terminal portion of Her-2.40 Previous study has shown that RRBP1 is associated with the UPR.12,15 In addition, experiments have revealed that the UPR is a possible means of overriding the effect of trastuzumab in Her-2-positive cancer cells.41 In this study, correlation between Her-2 and RRBP1 was also observed (P = 0.026). Statistical analysis of the UCSC cancer browser database also revealed that RRBP1 expression is highly correlated with Her-2 expression in human breast cancer (P = 0.016). Moreover, high RRBP1 expression correlates with poor OS in Her-2-positive patients (P = 0.031). RRBP1 expression is an independent predictor for survival in Her-2 positive patients. Thus, we hypothesized that UPR-caused trastuzumab resistance might be the reason for poor prognosis in patients with RRBP1 expression. However, further research is required to pinpoint its molecular mechanisms.
In summary, we show a significant correlation between expression of RRBP1 and histological grade, Her-2, P53 and molecular subtypes in breast cancer. Our data also imply that RRBP1 is a valuable prognostic factor in Her-2-positive breast cancer patients, indicating that RRBP1 is a potentially important target for the prediction of prognosis. The roles of RRBP1 in breast cancer need to be tested in vivo. Therefore, further studies are required to identify its function and its role in the progression and poor prognosis of Her-2-positive patients. In addition, more clinical trails should be applied to confirm whether RRBP1 could be used as a novel prognostic indicator.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (81172498/H1622, 81101997/H1622) and Heilongjiang Special Funds for outstanding youth (No. YJSCX2012-239HLJ).
Disclosure Statement
The authors have no conflict of interest to declare.
References
- Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9–29. doi: 10.3322/caac.21208. [DOI] [PubMed] [Google Scholar]
- Slamon DJ, Godolphin W, Jones LA, et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244:707–12. doi: 10.1126/science.2470152. [DOI] [PubMed] [Google Scholar]
- Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235:177–82. doi: 10.1126/science.3798106. [DOI] [PubMed] [Google Scholar]
- Yu D, Hung MC. Overexpression of ErbB2 in cancer and ErbB2-targeting strategies. Oncogene. 2000;19:6115–21. doi: 10.1038/sj.onc.1203972. [DOI] [PubMed] [Google Scholar]
- Pietras RJ, Arboleda J, Reese DM, et al. HER-2 tyrosine kinase pathway targets estrogen receptor and promotes hormone-independent growth in human breast cancer cells. Oncogene. 1995;10:2435–46. [PubMed] [Google Scholar]
- Alaoui-Jamali MA, Paterson J, Al Moustafa AE, Yen L. The role of ErbB-2 tyrosine kinase receptor in cellular intrinsic chemoresistance: mechanisms and implications. Biochem Cell Biol. 1997;75:315–25. [PubMed] [Google Scholar]
- Konecny GE, Pegram MD, Venkatesan N, et al. Activity of the dual kinase inhibitor lapatinib (GW572016) against HER-2-overexpressing and trastuzumab-treated breast cancer cells. Cancer Res. 2006;66:1630–9. doi: 10.1158/0008-5472.CAN-05-1182. [DOI] [PubMed] [Google Scholar]
- Shepard HM, Jin P, Slamon DJ, Pirot Z, Maneval DC. Herceptin. Handb Exp Pharmacol. 2008;181:183–219. doi: 10.1007/978-3-540-73259-4_9. [DOI] [PubMed] [Google Scholar]
- Fulda S, Debatin KM. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene. 2006;25:4798–811. doi: 10.1038/sj.onc.1209608. [DOI] [PubMed] [Google Scholar]
- Benyamini P, Webster P, Meyer DI. Knockdown of p180 eliminates the terminal differentiation of a secretory cell line. Mol Biol Cell. 2009;20:732–44. doi: 10.1091/mbc.E08-07-0682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savitz AJ, Meyer DI. Identification of a ribosome receptor in the rough endoplasmic reticulum. Nature. 1990;346:540–4. doi: 10.1038/346540a0. [DOI] [PubMed] [Google Scholar]
- Hyde M, Block-Alper L, Felix J, Webster P, Meyer DI. Induction of secretory pathway components in yeast is associated with increased stability of their mRNA. J Cell Biol. 2002;156:993–1001. doi: 10.1083/jcb.200112008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardoso CM, Groth-Pedersen L, Hoyer-Hansen M, et al. Depletion of kinesin 5B affects lysosomal distribution and stability and induces peri-nuclear accumulation of autophagosomes in cancer cells. PLoS One. 2009;4:e4424. doi: 10.1371/journal.pone.0004424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krasnov GS, Oparina N, Khankin SL, et al. Colorectal cancer 2D-proteomics: identification of altered protein expression. Mol Biol. 2009;43:348–56. doi: 10.1134/s0026893309020186. [DOI] [PubMed] [Google Scholar]
- Tsai HY, Yang YF, Wu AT, et al. Endoplasmic reticulum ribosome-binding protein 1 (RRBP1) overexpression is frequently found in lung cancer patients and alleviates intracellular stress-induced apoptosis through the enhancement of GRP78. Oncogene. 2013;32:4921–31. doi: 10.1038/onc.2012.514. [DOI] [PubMed] [Google Scholar]
- Hammond ME, Hayes DF, Dowsett M, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010;28:2784–95. doi: 10.1200/JCO.2009.25.6529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu CY, Ho DM, Yang CF, Lai CR, Yu IT, Chiang H. Interobserver reproducibility of Her-2/neu protein overexpression in invasive breast carcinoma using the DAKO HercepTest. Am J Clin Pathol. 2002;118:693–8. doi: 10.1309/6ANB-QXCF-EHKC-7UC7. [DOI] [PubMed] [Google Scholar]
- Cheang MC, Chia SK, Voduc D, et al. Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst. 2009;101:736–50. doi: 10.1093/jnci/djp082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millar EK, Graham PH, McNeil CM, et al. Prediction of outcome of early ER+ breast cancer is improved using a biomarker panel, which includes Ki-67 and p53. Br J Cancer. 2011;105:272–80. doi: 10.1038/bjc.2011.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker RL, Huntsman DG, Lesack DW, et al. Assessment of interlaboratory variation in the immunohistochemical determination of estrogen receptor status using a breast cancer tissue microarray. Am J Clin Pathol. 2002;117:723–8. doi: 10.1309/PEF8-GL6F-YWMC-AG56. [DOI] [PubMed] [Google Scholar]
- Ogawa-Goto K, Tanaka K, Ueno T, et al. p180 is involved in the interaction between the endoplasmic reticulum and microtubules through a novel microtubule-binding and bundling domain. Mol Biol Cell. 2007;18:3741–51. doi: 10.1091/mbc.E06-12-1125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbe L, Lundberg E, Oksvold P, et al. Toward a confocal subcellular atlas of the human proteome. Mol Cell Proteomics. 2008;7:499–508. doi: 10.1074/mcp.M700325-MCP200. [DOI] [PubMed] [Google Scholar]
- Savitz AJ, Meyer DI. 180-kD ribosome receptor is essential for both ribosome binding and protein translocation. J Cell Biol. 1993;120:853–63. doi: 10.1083/jcb.120.4.853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen JV, Blagoev B, Gnad F, et al. Global, in vivo, and site-specific phosphorylation dynamics in signaling networks. Cell. 2006;127:635–48. doi: 10.1016/j.cell.2006.09.026. [DOI] [PubMed] [Google Scholar]
- Wanker EE, Sun Y, Savitz AJ, Meyer DI. Functional characterization of the 180-kD ribosome receptor in vivo. J Cell Biol. 1995;130:29–39. doi: 10.1083/jcb.130.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueno T, Tanaka K, Kaneko K, et al. Enhancement of procollagen biosynthesis by p180 through augmented ribosome association on the endoplasmic reticulum in response to stimulated secretion. J Biol Chem. 2010;285:29941–50. doi: 10.1074/jbc.M109.094607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brostrom MA, Prostko CR, Gmitter D, Brostrom CO. Independent signaling of grp78 gene transcription and phosphorylation of eukaryotic initiator factor 2 alpha by the stressed endoplasmic reticulum. J Biol Chem. 1995;270:4127–32. doi: 10.1074/jbc.270.8.4127. [DOI] [PubMed] [Google Scholar]
- Feng B, Yao PM, Li Y, et al. The endoplasmic reticulum is the site of cholesterol-induced cytotoxicity in macrophages. Nat Cell Biol. 2003;5:781–92. doi: 10.1038/ncb1035. [DOI] [PubMed] [Google Scholar]
- Gomez E, Powell ML, Bevington A, Herbert TP. A decrease in cellular energy status stimulates PERK-dependent eIF2alpha phosphorylation and regulates protein synthesis in pancreatic beta-cells. Biochem J. 2008;410:485–93. doi: 10.1042/BJ20071367. [DOI] [PubMed] [Google Scholar]
- Su HL, Liao CL, Lin YL. Japanese encephalitis virus infection initiates endoplasmic reticulum stress and an unfolded protein response. J Virol. 2002;76:4162–71. doi: 10.1128/JVI.76.9.4162-4171.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertolotti A, Zhang Y, Hendershot LM, Harding HP, Ron D. Dynamic interaction of BiP and ER stress transducers in the unfolded-protein response. Nat Cell Biol. 2000;2:326–32. doi: 10.1038/35014014. [DOI] [PubMed] [Google Scholar]
- Clarke R, Cook KL, Hu R, et al. Endoplasmic reticulum stress, the unfolded protein response, autophagy, and the integrated regulation of breast cancer cell fate. Cancer Res. 2012;72:1321–31. doi: 10.1158/0008-5472.CAN-11-3213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tateishi M, Ishida T, Mitsudomi T, Kaneko S, Sugimachi K. Prognostic value of c-erbB-2 protein expression in human lung adenocarcinoma and squamous cell carcinoma. Eur J Cancer. 1991;27:1372–5. doi: 10.1016/0277-5379(91)90012-3. [DOI] [PubMed] [Google Scholar]
- Lemoine NR, Jain S, Silvestre F, et al. Amplification and overexpression of the EGF receptor and c-erbB-2 proto-oncogenes in human stomach cancer. Br J Cancer. 1991;64:79–83. doi: 10.1038/bjc.1991.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Vita F, Giuliani F, Silvestris N, Catalano G, Ciardiello F, Orditura M. Human epidermal growth factor receptor 2 (HER2) in gastric cancer: a new therapeutic target. Cancer Treat Rev. 2010;36(Suppl 3):S11–5. doi: 10.1016/S0305-7372(10)70014-1. [DOI] [PubMed] [Google Scholar]
- Citri A, Skaria KB, Yarden Y. The deaf and the dumb: the biology of ErbB-2 and ErbB-3. Exp Cell Res. 2003;284:54–65. doi: 10.1016/s0014-4827(02)00101-5. [DOI] [PubMed] [Google Scholar]
- Yarden Y. Biology of HER2 and its importance in breast cancer. Oncology. 2001;61(Suppl 2):1–13. doi: 10.1159/000055396. [DOI] [PubMed] [Google Scholar]
- Harari D, Yarden Y. Molecular mechanisms underlying ErbB2/HER2 action in breast cancer. Oncogene. 2000;19:6102–14. doi: 10.1038/sj.onc.1203973. [DOI] [PubMed] [Google Scholar]
- Fendly BM, Winget M, Hudziak RM, Lipari MT, Napier MA, Ullrich A. Characterization of murine monoclonal antibodies reactive to either the human epidermal growth factor receptor or HER2/neu gene product. Cancer Res. 1990;50:1550–8. [PubMed] [Google Scholar]
- Cho HS, Mason K, Ramyar KX, et al. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature. 2003;421:756–60. doi: 10.1038/nature01392. [DOI] [PubMed] [Google Scholar]
- Kumandan S, Mahadevan NR, Chiu K, DeLaney A, Zanetti M. Activation of the unfolded protein response bypasses trastuzumab-mediated inhibition of the PI-3K pathway. Cancer Lett. 2013;329:236–42. doi: 10.1016/j.canlet.2012.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]


