Abstract
Distal-less genes (DLX) play important roles in regulating organism development. DLX2 is crucial for the differentiation and development of the primordium, which determines the subsequent development and phenotype of the maxillofacial skeletal patterns, and is the primary candidate gene that regulates the development of the first branchial arch. The aim of the present study was to investigate the effects of DLX2 overexpression on the osteogenic differentiation of MC3T3-E1 cells in vitro. A DLX2-expression retrovirus vector was constructed by subcloning with a murine stem cell virus (MSCV) and verified by sequencing. MC3T3-E1 cells were transfected with pMSCV-DLX2 and stable clones were selected with puromycin. The mRNA and protein expression levels of DLX2 were determined using quantitative polymerase chain reaction (PCR) and western blot analysis, respectively. In addition, the expression levels of the osteogenic biomarkers, alkaline phosphatase (ALP), osteocalcin (OCN), runt-related transcription factor (RUNX)2 and Msh homeobox (MSX)2, were assessed by quantitative PCR. ALP detection and Alizarin red staining were conducted to evaluate the effect of DLX2 overexpression on osteogenic differentiation. The data were analyzed by analysis of variance using the Student-Newman-Keuls method. Successful pMSCV-DLX2 construction, as verified by direct sequencing, enabled DLX2 overexpression in vitro. Enhanced ALP activity and Alizarin red staining were observed in the MC3T3-E1-DLX2 cells when compared with the control group. During osteogenic induction, DLX2 overexpression was demonstrated to upregulate ALP and MSX2 expression at the early stage and OCN expression at the late stage, while no statistically significant difference was observed in RUNX2 expression when compared with the control group. Therefore, DLX2 overexpression in vitro induced the osteogenic differentiation of MC3T3-E1 cells via upregulating bone formation-associated genes, such as ALP and MSX2.
Keywords: distal-less gene 2, osteogenic differentiation, MC3T3-E1 cells
Introduction
Distal-less genes (DLX) are divergent homeobox genes of the Hox gene family, including DLX1-6, which play important roles in regulating organism development (1,2). DLX2 overexpression occurs in the middle area of the first branchial arch, namely the maxillary process, between days 9.5 and 10.5 of mouse embryo development. This overexpression is crucial to the differentiation and development of the primordium, as demonstrated by experimental embryology, since it determines the subsequent development and phenotype of the maxillofacial skeletal patterns (3). Thereafter, DLX2 is the primary candidate gene that regulates the development of the first branchial arch (4). Craniofacial bone defects, cranial parietal bone ossification delay and cortical bone dysplasia in long bones have been observed in DLX2 knockout mouse models (5,6), indicating that the main function of DLX2 is to regulate the proper migration of ectomesenchymal cells and the normal osteogenic differentiation of preosteoblast cells. However, the function of DLX2 in regulating osteoblast differentiation and the underlying molecular mechanism remain controversial (7–9).
Therefore, the aim of the present study was to investigate the effect of DLX2 on osteogenic differentiation, and the possible underlying molecular mechanism, through in vitro transfection of a murine stem cell virus containing DLX2 (pMSCV-DLX2) into the preosteoblast cell line, MC3T3-E1.
Materials and methods
Materials
An MC3T3-E1 cell line was purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). Fetal calf serum (FCS) was obtained from the Beyotime Institute of Biotechnology (Haimen, China) and α-Mimimal Essential Medium (MEM) was purchased from Gibco Life Technologies (Carlsbad, CA, USA). α-MEM osteogenesis induction culture medium (α-MEM plus 1.0×10−7mol/l dexamethasone, 2.5×10−4 mol/l vitamin C and 1.0×10−2mol/l β-sodium glycerophosphate) and ρ-nitrophenylphosphate (PNPP) were purchased from Sigma-Aldrich (St. Louis, MO, USA). A 3180P INC Heraeus Hera Cell CO2 incubator was obtained from Heraeus Holding GmbH (Hanau, Germany) and a Leica DM IRB inverted phase contrast microscope was purchased from Leica Microsystems GmbH (Wetzlar, Germany). The Z2 Cell Counter was purchased from Beckman Coulter (Brea, CA, USA). A Prime Script™ RT Reagent Kit and SYBR Premix Ex Taq™ were purchased from Takara Biotechnology Co., Ltd. (Dalian, China). CAGGS/DLX2 was obtained from Dr Rubenstein (University of California, San Francisco, CA, USA).
Establishment of pMSCV-puro-DLX2
Restriction enzyme identification with XhoI and EcoRI (Takara Biotechnology Co., Ltd.) was applied to pCAGGS/DLX2 and pMSCV-puro (GeneCopoeia, Rockville, MD, USA), respectively. The target fragments were collected through gel extraction according to the manufacturer's instructions (QIAquick Gel extraction kit, Qiagen, Hilden, Germany) and stored at 4°C overnight. pMSCV-puro and DLX2 were subsequently combined by T4 ligase (Takara Biotechnology Co. Ltd.) at 16°C overnight and sequenced.
293FT viral packaging
Transfection was performed when the cell density of the 293FT cell line reached 80–90%. The 293FT cell line was used to construct the retrovirus. Vesicular stomatitis virus glycoprotein (10 µg), GAG-pol (15 µg) and pMSCV (15 µg) were prepared for the retroviral packaging system. At 4–6 h after transfection, the culture medium in the T75 flask was replaced with 10 ml α-MEM containing 10% FCS, after which the virus was collected. This was done by collecting the pseudovirus-containing medium from the 293FT cell culture into sterile capped tubes for 48 h post-transfection. The tubes were then centrifuged at 550 × g for 10 min to remove cell debris and the supernatant was filtered through 45 µm polyethersulfone low protein-binding filters. The virus pellet was resuspended in phosphate-buffered saline (PBS) and stored at 4°C.
MC3T3-E1 transfection
The virus used for transfection was determined through dosage titration at 1:1, 10:1, 100:1 and 1,000:1. Polybrene (1/1,000; Sigma-Aldrich) was added to the medium, after which the plate was centrifuged for 3 min at 300 × g at 37°C. The culture medium was replaced 24 h after transfection. Following incubation in the culture medium for 48 h, 3 µg/ml puromycin was applied for growth selection. The cells that survived the selection were collected and passaged. Controls included pMSCV (dosage titration at 1:1, 10:1 and 100:1).
DLX2 expression detection with quantitative polymerase chain reaction (PCR) and western blot analysis
At 72 h after puromycin selection, the total RNA was extracted for quantitative PCR. This was performed using the SYBR Green system (Takara Bio, Inc, Shiga, Japan) with a total volume of 20 µl in a 96-well microwell plate. The reaction consisted of 10 µl SYBR Premix Ex Taq (Takara Biotechnology Co. Ltd.), 0.8 µl mixed primers, 1 µl cDNA template and 8.2 µl RNase-free distilled water. The PCR program was set at 95°C for 4 min followed by 40 cycles at 95°C for 20 sec, 60 °C for 15 sec and 72 °C for 20 sec. The threshold cycle values were calculated using the iQ5 software (Bio-Rad, Hercules, CA, USA). All the primers used in the experiments are shown in Table I. The analysis of the data was based on calculating the relative expression levels of these genes compared to the expression of the control (glyceraldehyde 3-phosphate dehydrogenase, a housekeeping gene).
Table I.
Primers for used for quantitative polymerase chain reaction.
| Gene | Forward sequence | Reverse sequence |
|---|---|---|
| DLX2 | 5′-CATGGGCTCCTACCAGTACCAC-3′ | 5′-TCGGATTTCAGGCTCAAGGTC-3′ |
| GAPDH | 5′-GGTGAAGGTCGGTGTGAACG-3′ | 5′-CTCGCTCCTGGAAGATGGTG-3′ |
| ALP | 5′-TGGGCATTGTGACTACCACTCGG-3′ | 5′-CCTCTGGTGGCATCTCGTTATCC-3′ |
| OCN | 5′-GGACCATCTTTCTGCTCACTCTG-3′ | 5′-GTTCACTACCTTATTGCCCTCCTG-3′ |
| RUNX2 | 5′-AACTTCCTGTGCTCCGTGCTG-3′ | 5′-TCGTTGAACCTGGCTACTTGG-3′ |
| MSX2 | 5′-GGAGCACCGTGGATACAGGA-3′ | 5′-AGGCTAGAAGCTGGGATGTGG-3′ |
DLX, distal-less gene; ALP, alkaline phosphatase; OCN, osteocalcin; RUNX2, runt-related transcription factor; MSX, Msh homeobox.
Cells were collected, washed 3 times with pre ice-cold PBS and lysed in SDS-lysis buffer containing protease inhibitors (Roche Diagnostics GmbH, Mannheim, Germany) for 20 min on ice. The lysates were centrifuged at 12,000 × g at 4°C for 30 min and the supernatants were boiled in sodium dodecyl sulfate sample buffer containing 0.5 M of mercaptoethanol. The samples were then separated on a 10% sodium dodecyl sulfate polyacrylamide gel and transferred to a polyvinylidene difluoride membrane using a semi-dry transfer apparatus (Bio-Rad, Hercules, CA, USA). The membrane was blocked with 5% milk for 1 h and then incubated with primary antibodies in modified D-PBS tween-20 buffer overnight. The primary antibodies were anti-DLX2 (1:800; Abcam, Cambridge, UK) and β-actin (1:1,000; Sigma-Aldrich). Following the primary antibody incubation, they were incubated with horse-radish peroxidase-conjugated secondary antibodies, including goat anti-rabbit and goat anti-mouse IgG (1:5,000; Beyotime Institute of Biotechnology, Haimen, China). The protein bands were visualised using an enhanced chemilluminescent western blot analysis kit (Pierce Biotechnology, Inc., Rockford, IL, USA).
Detection of DLX2 expression in the stable cell line following transfection
Following transfection and selection, the stable cell line was seeded into a six-well plate at a cell density of 1.5×104/cm2. α-MEM osteogenesis induction culture medium was added to the wells and the cells were cultured in conditions of 5% CO2 at 37°C. Cells were collected and protein samples were obtained on days 1, 4, 7 and 14 for western blot analysis.
Analysis of effect of DLX2 overexpression on osteogenesis-associated genes via quantitative PCR
Stable cell lines, following transfection and selection, were seeded into a six-well plate at a cell density of 1.5×104/cm2. The wells contained α-MEM osteogenesis induction culture medium and the cells were cultured in 5% CO2 at 37°C. Cells were collected and the total RNA was extracted on days 1, 4, 7 and 14 for quantitative PCR. The primers used for the PCR assays of ALP, osteocalcin (OCN), runt-related transcription factor (RUNX)2 and MSX2 are included in Table I.
ALP activity detection
Stable cell lines, following transfection and selection, were seeded into a 24-well plate at a cell density of 1.5×104/cm2. α-MEM osteogenesis induction culture medium was added to the wells and the cells were cultured in 5% CO2 at 37°C. ALP activity detection was performed on days 4, 7 and 14 using PNPP. The cells were washed with PBS and lysed with ALP buffer containing 0.2% Triton X-100, 42.74 mg MgCl2·6H2O, 20 µl HCl (10 mol/l), 19.4 ml diethylamine and 40 mg sodium azide dissolved in 200 ml distilled water. After 4 h in the incubator at 37°C, 190 µl supernatant was collected and added to a 96-well plate containing 190 µl substrate buffer per well. The optical density (OD) value was measured at 405 nm following incubation for 30 min at 37°C. A 10-µl sample of the supernatant was added to 200 µl protein analysis buffer and the OD value at 630 nm was detected after 5 min. The intracellular protein content was determined using a Micro BCA protein assay kit (Thermo Fisher Scientific, Waltham, MA, USA) and applied for the standard curve. The alkaline phosphatase activity was determined by measuring the optical density values for absorbance at 405 nm after incubation with p-nitrophenyl phosphate for 30 min at 37°C (Sigma-Aldrich).
Alizarin red staining
Alizarin red staining was used in a biochemical assay to determine, quantitatively by colorimetry, the presence of calcific deposition by cells of an osteogenetic lineage. Following transfection and selection, the stable cell lines were seeded into a six-well plate at a cell density of 1.5×104/cm2 with α-MEM osteogenesis induction culture medium. The cells were cultured in conditions of 5% CO2 at 37°C. Alizarin red staining (Sigma-Aldrich) was performed on days 3, 21 and 27, however, the stain was not that notable amongst the blank, pMSCV and pMSCV-Dl×2 groups. Each group was scanned for detection by a Canon LiDE 110 scanner (Canon, Beijing, China) following the staining.
Statistical analysis
Data were analyzed by analysis of variance using the Student-Newman-Keuls method. Statistical analysis was performed using SAS 6.04 software package (SAS Institute, Cary, NC, USA), where P<0.05 was considered to indicate a statistically significant difference.
Results
DLX2 expression in the stably transfected cell line
At 72 h after puromycin selection, mRNA expression of DLX2 was observed in the pMSCV-DLX2 group, but not in the pMSCV or non-transfection groups (Fig. 1). As shown in the western blot analysis, a 33-kDa band presented in the pMSCV-DLX2 group; however, this band was not observed in the pMSCV or non-transfection groups (Fig. 2). These observations indicated that the transfection was successful and expression of DLX2 had been induced.
Figure 1.
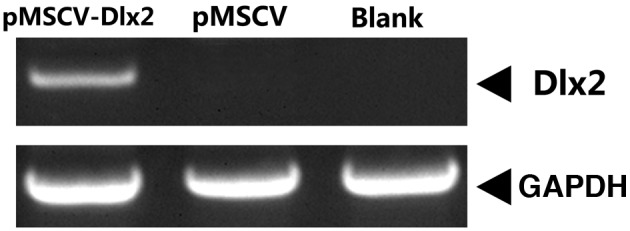
Analysis of DLX2 mRNA expression levels in the MC3T3-E1 cell line using quantitative polymerase chain reaction. DLX, distal-less gene; MSCV, murine stem cell virus. GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Figure 2.
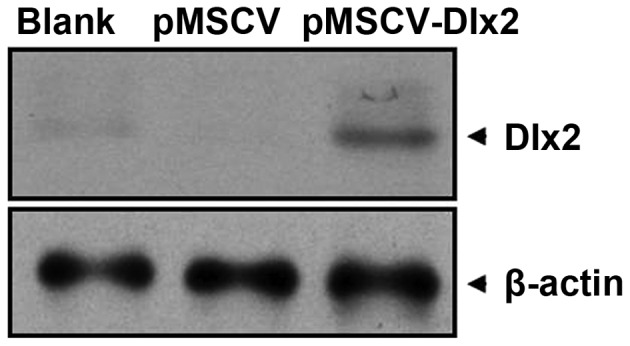
Analysis of DLX2 protein expression levels in the MC3T3-E1 cell line using western blot analysis. DLX, distal-less gene; MSCV, murine stem cell virus.
DLX2 expression in the stable clones selected with osteogenesis induction
On day 1 following osteogenesis induction, DLX2 expression was detected in the pMSCV-DLX2 group, which was shown to remain stable until day 14 post-transfection. Increasing endogenic DLX2 expression was observed in the pMSCV group (Fig. 3).
Figure 3.
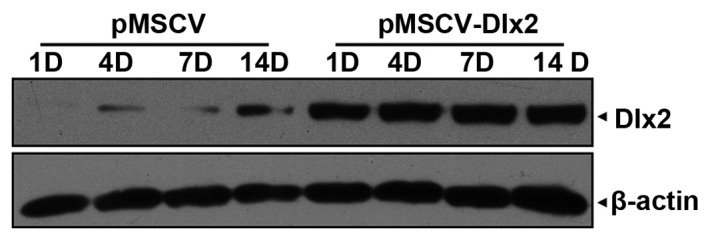
Western blot analysis showing DLX2 protein expression levels following osteogenic induction in pMSCV-DLX2 stably transfected cells. DLX, distal-less gene; MSCV, murine stem cell virus; D, day.
Effect of DLX2 overexpression on the expression levels of osteogenesis-associated genes
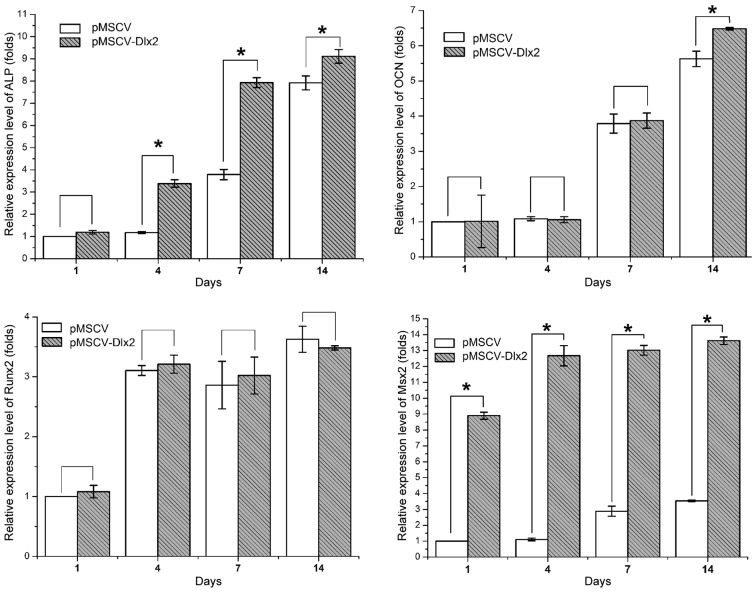
Expression levels of osteogenesis-associated genes are shown in Fig. 4. The relative expression levels were calculated, as compared with the control group on day 1, and are outlined in Table II. Statistically significant differences between the control and experimental cells were observed in the expression of ALP (P<0.01), OCN (P<0.05, on day 14) and MSX2 (P<0.01), but not in the expression of RUNX2 (P>0.05).
Figure 4.
Quantitative polymerase chain reaction analysis showing the relative mRNA expression levels of the bone formation-associated genes, ALP, OCN, RUNX2 and MSX2. Values were normalized against the mRNA expression level of the control group on day 1. *P<0.05, pMSCV-DLX2 group vs. pMSCV group (n=3). DLX, distal-less gene; ALP, alkaline phosphatase; OCN, osteocalcin; RUNX2, runt-related transcription factor; MSX, Msh homeobox; MSCV, murine stem cell virus.
Table II.
Quantitative polymerase chain reaction analysis of the relative expression levels of bone formation-associated genes.
| Gene | Day 1 | Day 4 | Day 7 | Day 14 |
|---|---|---|---|---|
| ALP | ||||
| pMSCV | 1 | 1.18±0.045 | 3.719±0.23 | 7.971±0.32 |
| pMSCV-DLX2 | 1.193±0.076 | 3.387±0.172 | 7.93±0.223 | 9.117±0.305 |
| OCN | ||||
| pMSCV | 1 | 1.084±0.058 | 3.79±0.271 | 5.56±0.217 |
| pMSCV-DLX2 | 1.013±0.075 | 1.057±0.087 | 3.873±0.215 | 6.482±0.035 |
| RUNX2 | ||||
| pMSCV | 1 | 3.104±0.083 | 2.86±0.395 | 3.626±0.217 |
| pMSCV-DLX2 | 1.081±0.104 | 3.209±0.149 | 3.02±0.308 | 3.481±0.03 |
| MSX2 | ||||
| pMSCV | 1 | 1.104±0.083 | 2.882±0.313 | 3.54±0.046 |
| pMSCV-DLX2 | 8.901±0.223 | 12.676±0.641 | 13.02±0.308 | 13.615±0.236 |
Statistically significant differences between the control and experimental groups were observed in the expression of ALP (P<0.01, on days 4, 7, 14), OCN,(P<0.05, on day 14) and MSX2 (P<0.01, on day 1, 4, 7, 14), but not in the expression of RUNX2 (P>0.05). DLX, distal-less gene; ALP, alkaline phosphatase; OCN, osteocalcin; RUNX2, runt-related transcription factor; MSX, Msh homeobox; MSCV, murine stem cell virus.
ALP activity
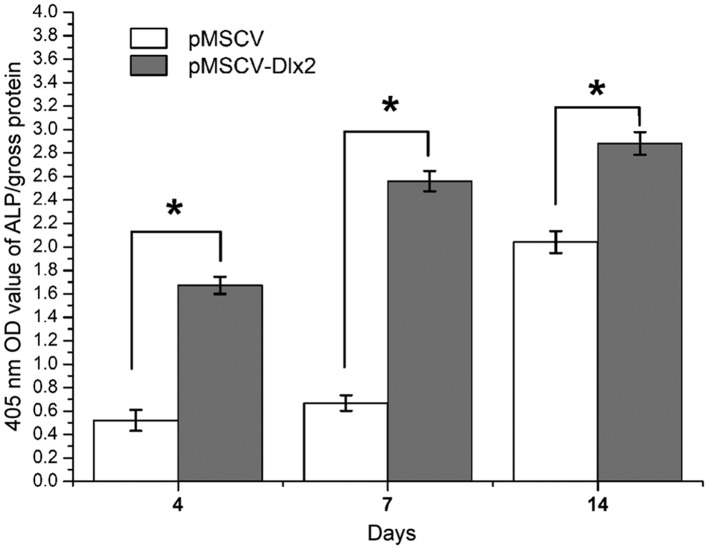
Higher ALP activity levels were observed on days 4, 7 and 14 after osteogenesis induction in the transfection group (pMSCV-DLX2), and statistically significant differences were observed when compared with the control group (P<0.01). The highest activity was observed on day 14, as shown in Fig. 5. The ALP activity levels are listed in Table III.
Figure 5.
Semi-quantitative analysis of ALP activity. *P<0.05, pMSCV-DLX2 group vs. pMSCV group (n=3). ALP, alkaline phosphatase; MSCV, murine stem cell virus; OD, optical density; DLX, distal-less gene.
Table III.
Semi-quantitative analysis of ALP activity.
| Group | Day 4 | Day 7 | Day 14 |
|---|---|---|---|
| pMSCV | 0.521±0.089 | 0.668±0.066 | 2.041±0.094 |
| pMSCV-DLX2 | 1.673±0.073 | 2.561±0.087 | 2.881±0.097 |
A statistically significant difference was observed when compared with the control group (P<0.01). DLX, distal-less gene; ALP, alkaline phosphatase; MSCV, murine stem cell virus.
Alizarin red staining
Positive results from the Alizarin red staining were observed on day 21 following osteogenesis induction, as shown in Fig. 6. The results for staining at day 27 were almost the same to those on day 21 and are therefore not discussed. In the DLX2 transfection group, the alizarin red staining was used to show the calcium nodule which indicated that DLX2 promoted the osteogenic differentiation of MC3T3-E1 cells.
Figure 6.

Alizarin red staining of MC3T3-E1 cells on day 21. DLX, distal-less gene; MSCV, murine stem cell virus.
Discussion
DLX are divergent homeobox genes that are part of the Hox gene family, which are involved in the regulation of maxillofacial bone development. However, the function of DLX remains controversial and the regulatory mechanisms are yet to be elucidated. In two previous studies, Qiu et al (5,6) created a DLX2−/− mouse model through gene knockout technology. Newborn DLX2−/− mice were found to die after birth with structural abnormalities observed in the bones originating from the first branchial arch maxillary process, such as the basisphenoid, greater wing of the sphenoid bone and pterygoid lamina, which was accompanied by a cleft palate, cranial parietal bone ossification delay and cortical bone dysplasia in long bones. These observations indicated that DLX2 was crucial in regulating craniofacial bone development and differentiation. In addition, gene expression microarray analysis revealed that DLX2 is an early response gene in the regulation of bone morphogenetic protein (BMP)2-mediated osteogenic differentiation (10). Therefore, the DLX knockout mouse models and DLX overexpression experiments indicated that DLX promotes skeleton formation; however, the effect of DLX on osteogenic differentiation remains unknown. Lee et al (11) found that DLX5, which is closely associated to DLX2, suppressed the expression of OCN, in contrast to DLX2. In addition, overexpression of DLX5 was reported to result in bone formation disorder in immunodeficient mice and mineralized matrix deposition of cells cultured in vitro. In vitro, overexpression of DLX5 was shown to have no effect on chondroplast differentiation (12). In the present study, MC3T3-E1 cells were transfected with pMSCV-DLX2 and stable clones were selected with puromycin. Subsequently, the mRNA and protein expression levels of DLX2 were determined by quantitative PCR and western blot analysis, respectively. This stable transfection cell line established the basis for investigating the effect of DLX2 overexpression on osteogenic differentiation and the possible underlying mechanisms.
ALP is hypothesized to be an indicator of early osteogenesis (13), and is known to hydrolyze numerous types of phosphates under alkaline conditions to promote cell maturation and calcification (14). ALP has been shown to be highly expressed during early osteogenic differentiation (day 7) through induction by morphogenetic proteins (15,16). In the present study, ALP activity in the MC3T3-E1 cell line was upregulated in the early stage of osteogenesis between days 7 and 14, as induced by the osteogenesis culture medium. As indicated in the quantitative PCR and ALP activity experiments, DLX2 upregulated ALP activity as early as day 4. On day 14, no statistically significant difference was observed between the groups, indicating that DLX2 primarily functions in the early stages of osteogenic differentiation, which is different from that of BMPs (15). OCN is the most abundant non-procollagen protein in the bone tissue, and can promote osteogenic differentiation by combining with minerals (17). In osteoblasts cultured in vitro, OCN upregulation has been observed during the mineralization stage; however, OCN expression has subsequently decreased until the end of osteogenic differentiation (13,18,19). The results of the present study demonstrated that low expression levels of OCN were observed in each group during the early stage of osteogenesis on days 1, 4 and 7, and no statistically significant differences were observed among the groups. However, on day 14, higher expression levels of OCN were observed in the transfection group (P<0.05). In addition, positive Alizarin red staining indicated that DLX2 promoted the expression of OCN and osteogenesis at the later stage.
RUNX2 belongs to the Runt domain gene family and has been demonstrated to be the one of the most important transcription factors involved in osteogenic differentiation (20). However, no statistically significant difference in RUNX2 expression was observed at any of the four time points when comparing the transfection and control groups (P>0.05), which indicated that the RUNX2 pathway may not be involved in the mechanism underlying DLX2-induced promotion of MC3T3-E1 osteogenesis. MSX2 belongs to the Hox gene family and is involved in the regulation of osteogenesis. Downregulation of MSX2 has been shown to result in preosteoblast dysmaturity (21). DLX genes have been reported to function in transforming growth factor-β (TGF-β)/BMP-mediated osteogenesis regulation through combining with and antagonizing MSX2 (22,23). In the present study, overexpression of DLX2 was shown to upregulate MSX2 expression. Thus, it was hypothesized that DLX2 and MSX2 regulate the transcription of osteogenesis-associated genes in a synergistic manner by forming a transcription factor complex. However, future investigation is required to further support this hypothesis.
In conclusion, through DLX2 transfection, examination of osteogenesis-associated gene expression, ALP activity and Alizarin red staining, the present study demonstrated that DLX2 overexpression induces the osteogenic differentiation of MC3T3-E1 cells via upregulating bone formation-associated genes, such as ALP and MSX2. Future studies will focus on osseointeractions between DLX2 and MSX2. Further studies will aim to investigate the role of the TGF-β signalling pathway on the induction of osteogenesis by DLX2. Such a study may contribute to potential clinical application of bone mesenchymal stem cells transduce with Dlx2 for bone augmentation in patients suffering from bone deficiency.
Acknowledgements
This study was supported by grants from the Shanghai Leading Academic Discipline Project (no. S30206), the Research Fund of Science and Technology Commission of Shanghai Municipality (no. 10JC1408700), the Research Fund of Shenkang Company (no. SHDC12010205), the Research Fund of Shanghai Municipal Health Bureau (no. 2009077), the Combined Engineering and Medicine Project of Shanghai Jiao Tong University (nos. YG2010MS55 and YG2014QN02) and the National Nature Science Foundation of China (no. 81271122).
Abbreviations
- DLX
distal-less gene
- ALP
alkaline phosphatase
- OCN
osteocalcin
- RUNX2
runt-related transcription factor 2
- MSX2
Msh homeobox 2
- BMP
bone morphogenetic protein
- MSCV
murine stem cell virus
- PCR
polymerase chain reaction
- FCS
fetal calf serum
- PNPP
ρ-nitrophenylphosphate
- MEM
Minimal Essential Medium
- PBS
phosphate-buffered saline
- OD
optical density
References
- 1.Depew MJ, Simpson CA, Morasso M, Rubenstein JL. Reassessing the Dlx code: the genetic regulation of branchial arch skeletal pattern and development. J Anat. 2005;207:501–561. doi: 10.1111/j.1469-7580.2005.00487.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jin Y. 1st. People's Medical Publishing House; Beijing: 2005. Mouse Development Biology and Embryo Research Method. Volume 2; pp. 70–73. [Google Scholar]
- 3.Ferguson CA, Tucker AS, Sharpe PT. Temporospatial cell interactions regulating mandibular and maxillary arch patterning. Development. 2000;127:403–412. doi: 10.1242/dev.127.2.403. [DOI] [PubMed] [Google Scholar]
- 4.Depew MJ, Lufkin T, Rubenstein JL. Specification of jaw subdivisions by Dlx genes. Science. 2002;298:381–385. doi: 10.1126/science.1075703. [DOI] [PubMed] [Google Scholar]
- 5.Qiu M, Bulfone A, Ghattas I, et al. Role of the Dlx homeobox genes in proximodistal patterning of the branchial arches: mutations of Dlx-1, Dlx-2 and Dlx-1 and −2 alter morphogenesis of proximal skeletal and soft tissue structures derived from the first and second arches. Dev Biol. 1997;185:165–184. doi: 10.1006/dbio.1997.8556. [DOI] [PubMed] [Google Scholar]
- 6.Qiu M, Bulfone A, Martinez S, et al. Null mutation of Dlx-2 results in abnormal morphogenesis of proximal first and second branchial arch derivatives and abnormal differentiation in the forebrain. Genes Dev. 1995;9:2523–2538. doi: 10.1101/gad.9.20.2523. [DOI] [PubMed] [Google Scholar]
- 7.Merlo GR, Zerega B, Paleari L, et al. Multiple functions of Dlx genes. Int J Dev Biol. 2000;44:619–626. [PubMed] [Google Scholar]
- 8.Panganiban G, Rubenstein JL. Developmental functions of the Distal-less/Dlx homeobox genes. Development. 2002;129:4371–4386. doi: 10.1242/dev.129.19.4371. [DOI] [PubMed] [Google Scholar]
- 9.Li H, Marijanovic I, Kronenberg MS, et al. Expression and function of Dlx genes in the osteoblast lineage. Dev Biol. 2008;316:458–470. doi: 10.1016/j.ydbio.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harris SE, Guo D, Harris MA, et al. Transcriptional regulation of BMP-2 activated genes in osteoblasts using gene expression microarray analysis: role of Dlx2 and Dlx5 transcription factors. Front Biosci. 2003;8:s1249–s1265. doi: 10.2741/1170. [DOI] [PubMed] [Google Scholar]
- 11.Lee MH, Kwon TG, Park HS, et al. BMP-2-induced Osterix expression is mediated by Dlx5 but is independent of Runx2. Biochem Biophys Res Commun. 2003;309:689–694. doi: 10.1016/j.bbrc.2003.08.058. [DOI] [PubMed] [Google Scholar]
- 12.Muraglia A, Perera M, Verardo S, et al. DLX5 overexpression impairs osteogenic differentiation of human bone marrow stromal cells. Eur J Cell Biol. 2008;87:751–761. doi: 10.1016/j.ejcb.2008.04.004. [DOI] [PubMed] [Google Scholar]
- 13.Karsenty G, Wagner EF. Reaching a genetic and molecular understanding of skeletal development. Dev Cell. 2002;2:389–406. doi: 10.1016/S1534-5807(02)00157-0. [DOI] [PubMed] [Google Scholar]
- 14.Beck GR, Jr, Zerler B, Moran E. Phosphate is a specific signal for induction of osteopontin gene expression. Proc Natl Acad Sci USA. 2000;97:8352–8357. doi: 10.1073/pnas.140021997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hu Z, Peel SA, Ho SK, et al. Role of bovine bone morphogenetic proteins in bone matrix protein and osteoblast-related gene expression during rat bone marrow stromal cell differentiation. J Craniofac Surg. 2005;16:1006–1014. doi: 10.1097/01.scs.0000170449.72040.ee. [DOI] [PubMed] [Google Scholar]
- 16.Vaes BL, Dechering KJ, Feijen A, et al. Comprehensive microarray analysis of bone morphogenetic protein 2-induced osteoblast differentiation resulting in the identification of novel markers for bone development. J Bone Miner Res. 2002;17:2106–2118. doi: 10.1359/jbmr.2002.17.12.2106. [DOI] [PubMed] [Google Scholar]
- 17.Glowacki J, Rey C, Glimcher MJ, et al. A role for osteocalcin in osteoclast differentiation. J Cell Biochem. 1991;45:292–302. doi: 10.1002/jcb.240450312. [DOI] [PubMed] [Google Scholar]
- 18.Hu JZ, Jiang XQ, Zhang ZY, et al. Effects of Nell-1 gene on bone formation-related gene expression during osteoblastic differentiation of rat bone marrow stromal cells by real-time PCR. [(In Chinese)]. Zhongguo Kou Qiang He Mian Wai Ke Za Zhi. 2008;6:48–53. [Google Scholar]
- 19.Hu JZ, Jiang XQ, Zhang ZY, Zhang XL. Effect of Nell-1 gene on osteogenic differentiation of rat bMSCs: An in vitro study. [(In Chinese)]. Zhongguo Kou Qiang He Mian Wai Ke Za Zhi. 2009;7:38–43. [Google Scholar]
- 20.Li P, Yu SH, Chen D, et al. Overexpression of Runx2 induces osteogenic differentiation in C2C12 cells. [(In Chinese)]. Zhongguo Sheng Wu Hua Xue Yu Fen Zi Sheng Wu Xue Bao. 2010;26:236–242. [Google Scholar]
- 21.Cheng SL, Shao JS, Charlton-Kachigian N, et al. MSX2 promotes osteogenesis and suppresses adipogenic differentiation of multipotent mesenchymal progenitors. J Biol Chem. 2003;278:45969–45977. doi: 10.1074/jbc.M306972200. [DOI] [PubMed] [Google Scholar]
- 22.Bendall AJ, Abate-Shen C. Roles for Msx and Dlx homeoproteins in vertebrate development. Gene. 2000;247:17–31. doi: 10.1016/S0378-1119(00)00081-0. [DOI] [PubMed] [Google Scholar]
- 23.Ryoo HM, Lee MH, Kim YJ. Critical molecular switches involved in BMP-2-induced osteogenic differentiation of mesenchymal cells. Gene. 2006;366:51–57. doi: 10.1016/j.gene.2005.10.011. [DOI] [PubMed] [Google Scholar]