Conservation of gene and protein sequence, and, therefore, conservation of the resulting molecular interactions that mediate biological processes, is foundational to our understanding of biology. This conservation allows discovery in one organism, such as worms or mice, to inform our understanding of the biology in another organism, such as humans. However, there is emerging recognition that many biological processes involve important non-conserved elements, and that de novo gene birth provides an important mechanism for functional evolution [1–3]. Understanding how these novel elements incorporate into gene regulatory networks and alter the network architecture is an important area for theoretical research [4–5], but few experimental examples have been described.
Formation of the dauer larva by nematodes is an adaptation that is responsive to a variety of environmental cues and alterations, features that suggest the underlying gene regulatory network might benefit from enhanced robustness and evolvability [6]. The dauer larva is a relatively dormant, alternative developmental stage that nematodes enter under stressful conditions, such as low food or crowding (signaled by pheromone), that confers increased longevity and tolerance to stressors [7]. In many parasitic nematodes, this stage corresponds to the infective larval stage when the larvae transition to new hosts. There are important conserved components of the gene network that regulate dauer formation, one being the nuclear hormone receptor DAF-12, which is important in free-living as well as parasitic nematode species [8–10]. However, it is also clear that this pathway is subject to considerable inter- and intraspecific differences [11–13]. Recent work on natural isolates of the nematode Pristionchus pacificus identified strain-specific phenotypic differences in dauer formation, and argued that genetically distinct populations exhibit greater sensitivity to pheromone from other populations (pheromone cross-preference [14]). The genetic alterations responsible for these phenotypic differences, however, had not yet been identified.
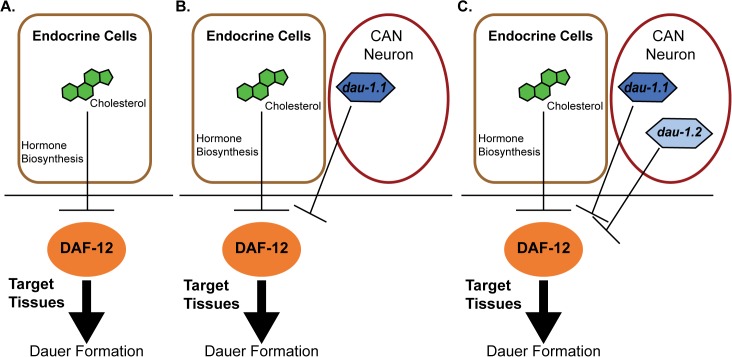
A new paper in this issue of PLOS Genetics [15] provides an answer, and at the same time highlights how “orphan” genes can be incorporated into conserved regulatory networks. The authors started with two strains of P. pacificus that exhibited marked differences in response to pheromone signaling: the RS2333/California strain, with low dauer formation in response to pheromone, and the RS5134/Ohio strain, with a high response. They then generated 911 recombinant inbred lines (RIL) between the strains, and used Quantitative-Trait-Loci (QTL) mapping to identify a novel gene with no apparent orthologs outside of Pristionchus, dauerless (dau-1), that had undergone a duplication event in the RS2333/California strain. This suggested that dau-1 functions as a repressor of dauer formation in a dose-dependent manner. To test this hypothesis, the authors created transgenic lines with multiple copies of dau-1, and created deletion mutants using CRISPR/Cas9 technology. Their data support the hypothesis as P. pacificus animals with more than two gene copies show no dauer formation in response to pheromone, whereas the deletion of one or both gene copies in RS2333 results in significantly higher dauer formation. The authors also show that this function is mediated by the CAN neurons. dau-1 is expressed in these cells, and ablation of these neurons results in increased dauer formation similar to that seen in dau-1 mutants. Finally, epistasis experiments show that dau-1 acts either downstream or in parallel to steroid hormone signaling, but is dependent on the nuclear hormone receptor gene daf-12 (Fig 1).
Fig 1. Proposed model for genetic evolution in P. pacificus dauer formation.
A) Conserved features of the dauer formation pathway. In endocrine cells, cholesterol is a substrate for the biosynthesis of dafachronic acid (DA), a DAF-12 ligand that suppresses the ability of DAF-12 to promote dauer formation. B) Model for the RS5134/Ohio strain with one copy of dauerless (dau-1.1). dau-1.1 is expressed in the CAN neuron cells and acts (genetically) to inhibit DAF-12 function and thereby inhibit dauer formation. C) The RS2333/California strain has two copies of dauerless (dau-1.1 and dau-1.2), and a corresponding double effect of inhibition of dauer formation.
Discovery of the dau-1 genes provides insight for how orphan genes can play an important role in the function and the evolution of biological networks that are conserved across species. The presence—and the duplication—of this gene is interpreted to provide a selective advantage in the context of intraspecific competition because it would allow individuals to continue with a reproductive life cycle even in the presence of crowding. Indeed, Mayer et al. have uncovered additional dau-1 paralogs in RS2333, indicating a potential for ongoing duplication of these novel genes [15]. A second important finding of this paper is that these orphan genes influence dauer formation by modulating a pathway with highly conserved elements (Fig 1). The results provide an important experimental example to complement the theoretical models for how the evolution of novel genes can add functional modifications to conserved regulatory networks. In this case, orphan gene evolution contributes to the evolutionary arms race between competing strains.
Important questions remain. In particular, previous work showed that different P. pacificus isolates exhibit differences in pheromone signaling and dauer survival, in addition to pheromone response, arguing that there are additional genetic modifications in the dauer regulatory network [14]. Whether these functions are influenced by dau-1 or related genes, and how these features of the dauer regulation network may interact, is not known. In addition, whether this represents a unique example or an evolutionary prototype is not clear. Generalizations of the types of changes that are responsible for evolutionary change have focused on the level of individual orthologous (and, therefore, conserved) genes [16–17]. These earlier analyses did, however, highlight how the network position of a gene can influence whether it is likely subject to evolutionary modification, and the types of network nodes that are more likely to be affected by cis-regulatory or protein-coding changes. As more evolutionary examples involving orphan genes are described, it will be interesting to learn if they are preferentially incorporated into particular nodes of regulatory networks, and whether they contribute primarily to intraspecific differences or have a role at longer evolutionary timescales.
Funding Statement
Research in the Chamberlin lab is funded by the US National Science Foundation (DMS-1361251). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Tautz D, Domazet-Lošo T. The evolutionary origin of orphan genes. Nat Rev Genet. 2011;12: 692–702. 10.1038/nrg3053 [DOI] [PubMed] [Google Scholar]
- 2. Carvunis A-R, Rolland T, Wapinski I, Calderwood MA, Yildirim MA, Simonis N, et al. Proto-genes and de novo gene birth. Nature. 2012;487: 370–374. 10.1038/nature11184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Abrusán G. Integration of new genes into cellular networks, and their structural maturation. Genetics. 2013;195: 1407–1417. 10.1534/genetics.113.152256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Aldana M, Balleza E, Kauffman S, Resendiz O. Robustness and evolvability in genetic regulatory networks. J Theor Biol. 2007;245: 433–448. [DOI] [PubMed] [Google Scholar]
- 5. Pechenick DA, Moore JH, Payne JL. The influence of assortativity on the robustness and evolvability of gene regulatory networks upon gene birth. J Theor Biol. 2013;330: 26–36. 10.1016/j.jtbi.2013.03.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Crombach A, Hogeweg P. Evolution of evolvability in gene regulatory networks. PLOS Comput Biol. 2008;4: e1000112 10.1371/journal.pcbi.1000112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Fielenbach N, Antebi A. C. elegans dauer formation and the molecular basis of plasticity. Genes Dev. 2008;22: 2149–2165. 10.1101/gad.1701508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Antebi A, Yeh WH, Tait D, Hedgecock EM, Riddle DL. daf-12 encodes a nuclear receptor that regulates the dauer diapause and developmental age in C. elegans. Genes Dev. 2000;14: 1512–1527. [PMC free article] [PubMed] [Google Scholar]
- 9. Ogawa A, Streit A, Antebi A, Sommer RJ. A conserved endocrine mechanism controls the formation of dauer and infective larvae in nematodes. Curr Biol CB. 2009;19: 67–71. 10.1016/j.cub.2008.11.063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Wang Z, Zhou XE, Motola DL, Gao X, Suino-Powell K, Conneely A, et al. Identification of the nuclear receptor DAF-12 as a therapeutic target in parasitic nematodes. Proc Natl Acad Sci U S A. 2009;106: 9138–9143. 10.1073/pnas.0904064106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Elling AA, Mitreva M, Recknor J, Gai X, Martin J, Maier TR, et al. Divergent evolution of arrested development in the dauer stage of Caenorhabditis elegans and the infective stage of Heterodera glycines. Genome Biol. 2007;8: R211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Stoltzfus JD, Minot S, Berriman M, Nolan TJ, Lok JB. RNAseq analysis of the parasitic nematode Strongyloides stercoralis reveals divergent regulation of canonical dauer pathways. PLOS Negl Trop Dis. 2012;6: e1854 10.1371/journal.pntd.0001854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Crook M. The dauer hypothesis and the evolution of parasitism: 20 years on and still going strong. Int J Parasitol. 2014;44: 1–8. 10.1016/j.ijpara.2013.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Bose N, Meyer JM, Yim JJ, Mayer MG, Markov GV, Ogawa A, et al. Natural variation in dauer pheromone production and sensing supports intraspecific competition in nematodes. Curr Biol CB. 2014;24: 1536–1541. 10.1016/j.cub.2014.05.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Mayer MG, Rodelsperger C, Witte H, Riebesell M, Sommer RJ. The Orphan Gene dauerless Regulates Dauer Development and Intraspecific Competition in Nematodes by Copy Number Variation. PLoS Genet 11(6): e1005146 10.1371/journal.pgen.1005146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Stern DL, Orgogozo V. The loci of evolution: how predictable is genetic evolution? Evol Int J Org Evol. 2008;62: 2155–2177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Stern DL, Orgogozo V. Is genetic evolution predictable? Science. 2009;323: 746–751. 10.1126/science.1158997 [DOI] [PMC free article] [PubMed] [Google Scholar]