Abstract
CD4+ T cell counts of HIV-infected individuals with pulmonary TB (PTB) are higher than with other opportunistic infections suggesting that progression to PTB is not merely due to T cell depletion but also dysfunction. There are limited data examining T cell functional signatures in human HIV-TB co-infection particularly in PTB which accounts for about 80% of active TB disease overall. We examined a cohort of HIV-infected anti-retroviral naïve individuals in Kampala, Uganda, a TB endemic area using multi-parametric flow cytometry analysis to determine IFN-γ, IL-2, IL-17, and TNF-α production in CD4+ memory T cell subsets. The cytokine frequency and polyfunctionality profile of Mycobacterium tuberculosis (MTB)-specific CD4+ T cells in HIV-infected persons with latent TB infection (LTBI) or PTB is comparable. This similarity suggests that LTBI may represent a smoldering state of persistent MTB replication rather than dormant infection. This may be a contributory mechanism to the significantly increased risk of progression to PTB in this population.
Keywords: Tuberculosis, HIV-1, CD4+ T cell, Polyfunctionality
1. Introduction
Both reactivation of latent tuberculosis infection (LTBI) and progressive primary pulmonary TB (PTB) are substantially more common in HIV-infected individuals as compared to HIV-uninfected persons. In fact, untreated HIV-infected persons who are latently infected with MTB have on the order of a 10% annual risk of developing active PTB compared to a 5–10% lifetime risk in HIV-uninfected individuals [1]. Observations gathered from HIV/TB co-infected persons have offered some insight into the basic immunology of TB latency. The role of CD4+ T cells in controlling TB has been highlighted since these cells are preferentially targeted by HIV-1 infection, resulting in early peripheral depletion of MTB-specific CD4+ T cells after HIV infection [2]. However, the increased risk of active TB begins soon after HIV infection when total CD4+ T cell counts are preserved, suggesting that progression of TB disease in the setting of HIV infection is not merely due to CD4+ T cell depletion, but also due to functional impairment of MTB-specific T cells [3].
Multicolor flow cytometry has offered the opportunity to examine polyfunctional cytokine response patterns on a single cell level, enabling more sophisticated studies to examine the relationship between complex cytokine profiles and disease progression. Polyfunctional T cells in the lungs of mice boosted with recombinant adenovirus-expressing MTB antigen 85A correlated with reduced mycobacterial burden in MTB aerosol challenged mice [4]. A number of recent reports contrast the polyfunctional profile of MTB-specific T cell responses in HIV-uninfected subjects with active PTB and LTBI [5–7]. Most of the reports examining T cell functional signatures in human HIV/TB co-infected patients do not focus on comparing LTBI and PTB [8–12]. The exception is Pollock et al., who found PPD-specific CD4+ T cells with an effector phenotype secreting only TNF-α could distinguish active TB disease and LTBI [8]. The goal of this study was to perform a larger study on untreated HIV-infected subjects in a TB endemic area with the focus on PTB, the most common form of TB disease.
2. Methods
2.1. Recruitment of study subjects
All study recruitment occurred in Kampala, Uganda, at the HIV outpatient clinic of the Infectious Diseases Institute and TB Research Unit at Makerere University, local medical clinics, and the Tuberculosis Clinic at Mulago Hospital [13,14]. Following voluntary counseling and testing for HIV-1 infection, subjects provided informed written consent. The study was approved by the ethical committees of the Joint Clinical Research Center, Uganda Council for Science and Technology, and Case Western Reserve University.
All study subjects were antiretroviral therapy naïve. Exclusion criteria included prior active TB, isoniazid preventive therapy, steroid therapy, pregnancy, a Karnofsky score ≤60, or current opportunistic infection. The presence of any one of the symptoms including cough, chest pain, recent weight loss, night sweats, fever, loss of appetite, swelling of lymph nodes, or generalized tiredness triggered referral to the Tuberculosis Research Unit where physical examination, sputum examination, and chest radiography was performed. All subjects with PTB had positive sputum culture. Those who ruled out for active PTB by both negative culture and chest X-ray, had tuberculin placed (2TU PPD RT23; Statens Serum Institute, Copenhagen, Denmark), and tuberculin skin test (TST) was measured at 48 h. A TST of ≥5 mm in this HIV-infected cohort is considered positive. Uganda is a TB endemic area, and it is presumed that the vast majority of TST+ adult individuals have LTBI. Both groups, PTB and TST+ included 15 subjects each. The size of induration ranged from 5 mm to 28 mm in the TST+ group. Table 1 describes the clinical information for the subjects.
Table 1.
Baseline characteristics of study subjects. Thirty subjects were included in the study.
| Clinical classification |
Significant P values | ||
|---|---|---|---|
| PTB | TST+ | ||
| N | 15 | 15 | none |
| Mean age | 31 | 33 | none |
| Female n, (%) | 10 (67) | 12 (80) | none |
| Mean CD4 count cells/mm3(range) | 375 (168–724) | 357 (188–646) | none |
| Log10 HIV-1-RNA copies/ml (range) | 5.2 (3.2–5.9) | 4.7 (2.6–5.5) | p = 0.02 |
2.2. Flow cytometry
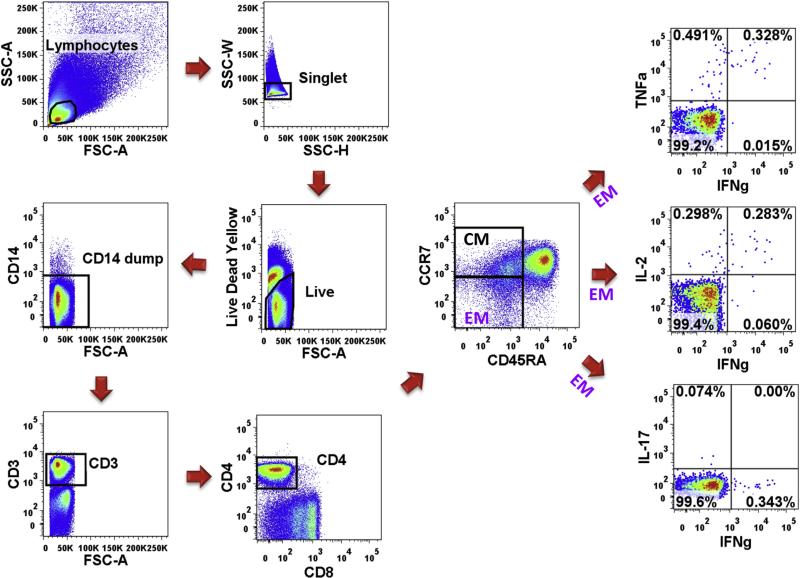
Peripheral blood mononuclear cells (PBMC) were prepared using Ficoll-Hypaque density gradient centrifugation (Eurobio) at enrollment and then cryopreserved in liquid nitrogen until analysis. Thawed PBMC (generally 1 × 106/tube) were stimulated with the superantigen staphylococcal enterotoxin B (SEB, 0.1 μg/ml, SIGMA), CFP10 + ESAT6 soluble antigens (5 μg/ml of each protein, BEI Resources) or no antigen (control) in serum free XVIVO15 (Lonza) medium. After 2 h Brefeldin A (5 μg/ml, SIGMA) was added. Anti-CD28/49d (1 μg/ml each, eBioscience and Biolegend) was also added during stimulation. After overnight stimulation, PBMC were surface stained with anti-CCR7-PE-Cy7 (BD) for 15 min at 37° then live dead yellow, anti-CD14-Qdot 655, CD45RA-PE/Texas Red (all Invitrogen), CD4-APC/Cy7 (Biolegend), and CD8-V500 (BD). They were then washed, fixed, and permeabilized (BD Cytofix/Cytoperm) for intracellular staining with anti-CD3-PerCP, IL-2-APC, IFN-γ-Alexa700, TNF-α-Pacific Blue, and IL-17A-FITC (all Biolegend). Finally cells were washed and fixed in 1% paraformaldehyde. Between 500,000 and 1,000,000 total events were collected from each sample on an LSR-II flow cytometer (BD). An algorithm of analysis is shown in Figure 1. Analysis was performed using Boolean analysis of Flow Jo (Tree Star) and Simplified Presentation of Incredibly Complex Evaluations (SPICE, Mario Roederer, NIAID) to assess polyfunctionality [15].
Figure 1. Gating strategy for intracellular polyfunctional flow analysis.
Dot plots of a representative subject are shown here.
3. Results
3.1. MTB-antigen specific responses
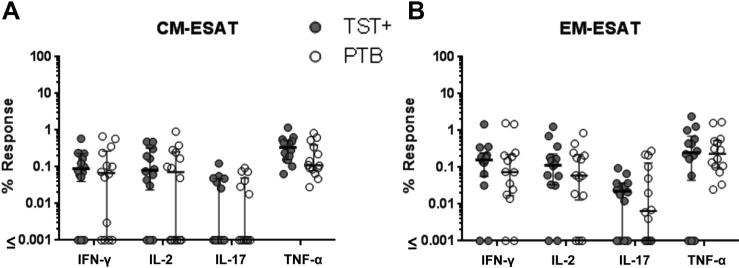
First we examined differences in CD4+ T cell responses after in vitro stimulation with soluble ESAT/CFP between the TST+ and PTB groups (Figure 2). We found no statistical difference between the percent ESAT/CFP-responsive CD4+ T cells in TST+ and PTB subjects when gating on central memory (CM) and effector memory (EM) T cell subsets for any of the 4 cytokines tested (IFN-g, IL-2, IL-17, and TNF-α, Figure 2A and B). Similarly, we found no correlation for any of these cytokines and the CD4 counts in CM and EM subgroups if the TST+ and PTB data are combined by Spearman methods (p > 0.19).
Figure 2. Frequencies of cytokine responses to MTB antigens.
PBMC were stimulated overnight with ESAT/CFP and surface and intracellular staining was performed and cells were gated on (A) CM or (B) EM CD4+ T cells. Filled circles are TST+ and presumed LTBI and open circles are PTB. All points are background subtracted. No statistical differences were observed as determined by SPSS (IBM) with Levene's test for equality of variances applied to each comparison and the appropriate t-test results chosen.
3.2. Polyfunctionality of MTB-specific CD4+ T cells in TST+ and PTB
We next assessed potential differences in the functional profile of MTB-specific CD4+ T cells in HIV-infected persons with LTBI (TST+) vs. those with active disease (PTB) by IFN-γ, IL-2, IL-17, and TNF-α responses to ESAT/CFP and SEB. We employed Boolean analysis of individual functional groups by Flowjo to determine the polyfunctional combinations to each of the stimuli, and expressed the analyzed data using the SPICE program.
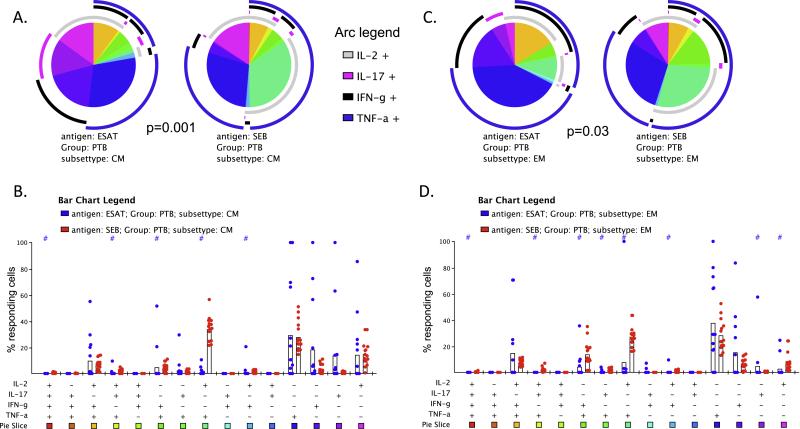
We found no significant difference in the overall polyfunctional cytokine profile of ESAT/CFP-specific CM and EM CD4+ T cells in TST+ and PTB groups (Figure 3A p = 0.19 and 3C p = 0.99). A few small differences were observed with higher single producing IL-17 + cells in EM (Figure 3B) and IFN-γ/TNF-α+ and IL-2/TNF-α+ cells in CM (Figure 3D) in TST+. Most notably the ESAT/CFP responding triple functional IFN-γ/IL-2/TNF-α+ a and single function TNF-α+ cells did not differ significantly between groups in either memory subsets (Figure 3B and D).
Figure 3. Polyfunctionality of ESAT/CFP-stimulated CD4+ T cells in TST + vs. PTB.
The pies and bar graphs depict the proportion of ESAT/CFP-responding CD4+ T cells in TST+ and PTB individuals with each distinct cytokine pattern for CM (A, B) and EM (C, D) subsets. The arc around each pie represents the individual cytokines as noted in the Arc legend. The pie slice color represents the polyfunctional combination as labeled in the bar graph. The bar graph shows the individual subjects’ proportions for each possible polyfunctional combination. Statistical comparisons between the pies were performed using Monte Carlo permutation analysis by SPICE. A # at the top of the bar graph indicates a statistically significant difference (p < 0.05) of that individual functional combination between TST+ and PTB as determined by Wilcoxon sign rank test.
As shown in Figure 4, we did find a significant difference between the overall polyfunctional cytokine profile of the ESAT/CFP-specific CD4+ T cells and cells stimulated with SEB in both T cell subsets (Figure 4A, CM p = 0.001 and Figure 4C, EM p = 0.03) in the PTB group. As shown in Figure 4A and C, there was greater poly-functionality in the SEB activated population compared to those cells that respond to ESAT/CFP. Finally, the pattern of results in TST+ subjects was nearly identical to those of the PTB group (data not shown), further supporting an overall similarity in the overall patterns of responses to MTB (or mitogen) between HIV-infected individuals with LTBI and PTB.
Figure 4. Polyfunctionality of ESAT/CFP vs. SEB-stimulated CD4+ T cells.
The pies and bar graphs depict the proportion of ESAT/CFP- or SEB-responding cells in PTB individuals with each distinct cytokine pattern for CD4+ CM (A, B) and EM (C, D) subsets. A # at the top of the bar graph indicates a statistically significant difference (p < 0.05) of that individual functional combination between CFP/ESAT or SEB-responding cells.
4. Discussion
In this study of HIV/TB dually infected subjects in Uganda, we found no significant differences in any of the cytokine polyfunctional profiles between the TST+ and PTB groups. This is in contrast to several previous studies comparing LTBI and active TB in HIV-uninfected subjects that had somewhat divergent results. Three shared the finding that there were clear differences between LTBI and active TB disease. Caccamo et al. had higher polyfunctional cells in PTB than in LTBI [6]. By contrast, Harari et al. had nearly an opposite result with higher polyfunctionality in LTBI than active TB [5]. The third study had a mixed result with more polyfunctionality in PTB than LTBI but also more single TNF-α cells in PTB [7]. A fourth very recent study by Kim et al. has shown that LTBI and active TB were not distinguished by flow cytometry assessing IFN-γ, IL-2 and TNF-α+ [16]
There are several studies that examine HIV/TB co-infected individuals. Day et al. observed loss in IL-2 secretion capacity by MTB-specific CD4+ T cells in HIV-infected individuals [9]. Kalsdorf et al. studied bronchoalveolar lavage lymphocytes from HIV-infected persons that had presumed LTBI [10]. Lymphocytes from this site were enriched in EM cells and found to have similar proportions of PPD-responding triple functional CD4+ T cells (IFN-γ/TNF-α/IL-2+) in HIV-infected subjects which is confirmed in the current study.
Another study looked at extrapulmonary TB in HIV-infected individuals and found essentially no triple functional CD4+ T cells in the blood but an increased but very low frequency of triple functional cells in the MTB-infected pericardial space [12].
The report most similar in design to ours is that of Pollock et al., who examined HIV-infected and uninfected individuals with LTBI and active TB [8]. In contrast to our findings, they found that PPD-specific TNF-α single functional EM cells accurately predicted active TB disease vs. LTBI. However, there were key differences in their cohort design that complicate the interpretation of their findings compared to ours. In their final analysis of subjects with TB disease, they grouped together HIV-infected and uninfected subjects that were a mix of PTB and extra-pulmonary disease rather than all PTB in the current study. Also, they had a smaller set of dually infected subjects with active TB (n = 7) all of whom had lower CD4 counts of ≤250 cells/μl (mean 166). Perhaps most importantly, nearly half the HIV-infected subjects within their LTBI and TB disease cohorts had undetectable HIV viral loads, raising the possibility that they were on anti-retroviral therapy, although that was not specifically noted. The combination of these factors may explain the outcome that our study does not support the notion that MTB-antigen specific single TNF-α producing EM cells are strongly predictive of active TB disease in HIV-infected subjects. Our study is focused exclusively on anti-retroviral therapy naïve HIV-infected persons (median CD4 counts >350 in the groups) with LTBI and PTB in a TB endemic area. This clinical scenario is very common in the TB endemic areas throughout the underdeveloped areas worldwide.
One limitation of most flow based studies including this study is that they require stimulation of the cells to identify and then characterize MTB-antigen specific cells. Differences in antigen presenting cell (APC) function and costimulatory molecule and receptor expression on the T cells and APC may be playing a role. We feel that our studies and the others in the field are still useful however as they are reading out an end result of antigen eliciting T cell responses that must also occur in vivo for control of MTB infection.
One potential explanation for the difference between our findings in HIV-infected subjects compared to other studies in HIV-uninfected subjects may be that LTBI represents a smoldering state of MTB infection in HIV-infected persons. The presence of increased MTB antigen (due to uncontrolled MTB infection) may provoke a more active immune response in latently infected subjects that are also HIV-1 co-infected. This might result in minimal or no immunologic difference as determined by PBMC studies between active and “latent” disease among HIV/TB dually infected subjects. In the primate dual infection model, the polyfunctionality of T cells better correlates with antigen load than protection from disease [17] supporting our contention that the higher polyfunctional T cell proportions are more a reflection of a subacute or smoldering process with persistent antigen rather than an effective immune response that has led to bacterial eradication or dormancy.
The data with the mitogen demonstrate that the MTB-specific memory T cell populations (both CM and EM) have a different profile than the overall memory populations that are activated by the superantigen SEB, as shown in Figure 4B and D. The patterns though are similar between PTB and LTBI (data not shown). This suggests that those T cells presumably exposed to MTB in the lung and draining lymph node acquire a distinct memory phenotype compared to the other memory populations present. The meaning and significance of this is yet to be defined.
Overall, our data supports the need for larger studies with a very careful immunologic and clinical examination of HIV-infected persons in endemic areas to understand if TB is commonly in a smoldering rather than truly latent state. Flow cytometry based polyfunctional analysis may be useful in differentiating HIV-infected persons with latent TB who might benefit from full anti-TB chemotherapy due to subclinical infection from those who truly have a latent infection. This is based on the finding that polyfunctional analysis suggests smoldering MTB infection rather than a dormant or well-contained status as in HIV-uninfected subjects. Also, our findings suggest caution and the need for further studies to evaluate and refine the predictive potential of polyfunctional profiling specifically in HIV-infected individuals to discriminate LTBI from active PTB.
Acknowledgments
None.
Funding: Funding was provided by NIH AI-80313, AI-36219, NHLBI-05636, and VA.
Footnotes
Competing interests: None declared.
Ethical approval: The study was approved by the ethical committees of the Joint Clinical Research Center, Uganda Council for Science and Technology, and Case Western Reserve University.
References
- 1.Selwyn PA, Hartel D, Lewis VA, Schoenbaum EE, Vermund SH, Klein RS, Walker AT, Friedland GH. A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection [see comments] N Engl J Med. 1989;320:545–50. doi: 10.1056/NEJM198903023200901. [DOI] [PubMed] [Google Scholar]
- 2.Geldmacher C, Ngwenyama N, Schuetz A, Petrovas C, Reither K, Heeregrave EJ, Casazza JP, Ambrozak DR, Louder M, Ampofo W, Pollakis G, Hill B, Sanga E, Saathoff E, Maboko L, Roederer M, Paxton WA, Hoelscher M, Koup RA. Preferential infection and depletion of Mycobacterium tuberculosis-specific CD4 T cells after HIV-1 infection. J Exp Med. 2010;207:2869–81. doi: 10.1084/jem.20100090. http://dx.doi.org/10.1084/jem.20100090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sonnenberg P, Glynn JR, Fielding K, Murray J, Godfrey-Faussett P, Shearer S. How soon after infection with HIV does the risk of tuberculosis start to increase? a retrospective cohort study in South African gold miners. J Infect Dis. 2005;191:150–8. doi: 10.1086/426827. [DOI] [PubMed] [Google Scholar]
- 4.Forbes EK, Sander C, Ronan EO, McShane H, Hill AV, Beverley PC, Tchilian EZ. Multifunctional, high-level cytokine-producing Th1 cells in the lung, but not spleen, correlate with protection against Mycobacterium tuberculosis aerosol challenge in mice. J Immunol. 2008;181:4955–64. doi: 10.4049/jimmunol.181.7.4955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Harari A, Rozot V, Enders FB, Perreau M, Stalder JM, Nicod LP, Cavassini M, Calandra T, Blanchet CL, Jaton K, Faouzi M, Day CL, Hanekom WA, Bart PA, Pantaleo G. Dominant TNF-alpha+ Mycobacterium tuberculosis-specific CD4+ T cell responses discriminate between latent infection and active disease. Nat Med. 2011;17:372–6. doi: 10.1038/nm.2299. http://dx.doi.org/10.1038/nm.2299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Caccamo N, Guggino G, Joosten SA, Gelsomino G, Di Carlo P, Titone L, Galati D, Bocchino M, Matarese A, Salerno A, Sanduzzi A, Franken WP, Ottenhoff TH, Dieli F. Multifunctional CD4(+) T cells correlate with active Mycobacterium tuberculosis infection. Eur J Immunol. 2010;40:2211–20. doi: 10.1002/eji.201040455. http://dx.doi.org/10.1002/eji.201040455. [DOI] [PubMed] [Google Scholar]
- 7.Sutherland JS, Adetifa IM, Hill PC, Adegbola RA, Ota MO. Pattern and diversity of cytokine production differentiates between Mycobacterium tuberculosis infection and disease. Eur J Immunol. 2009;39:723–9. doi: 10.1002/eji.200838693. http://dx.doi.org/10.1002/eji.200838693. [DOI] [PubMed] [Google Scholar]
- 8.Pollock KM, Whitworth HS, Montamat-Sicotte DJ, Grass L, Cooke GS, Kapembwa MS, Kon OM, Sampson RD, Taylor GP, Lalvani A. T-cell immunophenotyping distinguishes active from latent tuberculosis. J Infect Dis. 2013;208:952–68. doi: 10.1093/infdis/jit265. http://dx.doi.org/10.1093/infdis/jit265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Day CL, Mkhwanazi N, Reddy S, Mncube Z, van der Stok M, Klenerman P, Walker BD. Detection of polyfunctional Mycobacterium tuberculosis-specific T cells and association with viral load in HIV-1-infected persons. J Infect Dis. 2008;197:990–9. doi: 10.1086/529048. http://dx.doi.org/10.1086/529048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kalsdorf B, Scriba TJ, Wood K, Day CL, Dheda K, Dawson R, Hanekom WA, Lange C, Wilkinson RJ. HIV-1 infection impairs the bronchoalveolar T-cell response to mycobacteria. Am J Respir Crit Care Med. 2009;180:1262–70. doi: 10.1164/rccm.200907-1011OC. doi: 10.1164/rccm.200907-1011OC [pii] 200907-1011OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sutherland JS, Young JM, Peterson KL, Sanneh B, Whittle HC, Rowland-Jones SL, Adegbola RA, Jaye A, Ota MO. Polyfunctional CD4(+) and CD8 T cell responses tuberculosis antigens in HIV-1-infected patients before and after anti-retroviral treatment. J Immunol. 2010;184:6537–44. doi: 10.4049/jimmunol.1000399. http://dx.doi.org/10.4049/jimmunol.1000399. [DOI] [PubMed] [Google Scholar]
- 12.Matthews K, Ntsekhe M, Syed F, Scriba T, Russell J, Tibazarwa K, Deffur A, Hanekom W, Mayosi BM, Wilkinson RJ, Wilkinson KA. HIV-1 infection alters CD4+ memory T-cell phenotype at the site of disease in extrapulmonary tuberculosis. Eur J Immunol. 2012;42:147–57. doi: 10.1002/eji.201141927. http://dx.doi.org/10.1002/eji.201141927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Leidl L, Mayanja-Kizza H, Sotgiu G, Baseke J, Ernst M, Hirsch C, Goletti D, Toossi Z, Lange C. Relationship of immunodiagnostic assays for tuberculosis and numbers of circulating CD4+ T-cells in HIV infection. Eur Respir J. 2010;35:619–26. doi: 10.1183/09031936.00045509. http://dx.doi.org/10.1183/09031936.00045509. [DOI] [PubMed] [Google Scholar]
- 14.Nanteza MW, Mayanja-Kizza H, Charlebois E, Srikantiah P, Lin R, Mupere E, Mugyenyi P, Boom WH, Mugerwa RD, Havlir DV, Whalen CC. A randomized trial of punctuated antiretroviral therapy in Ugandan HIV-seropositive adults with pulmonary tuberculosis and CD4(+) T-cell counts of >/ 350 cells/muL. J Infect Dis. 2011;204:884–92. doi: 10.1093/infdis/jir503. doi: 10.1093/infdis/jir503 [pii] jir503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Roederer M, Nozzi JL, Nason MC. SPICE: exploration and analysis of post-cytometric complex multivariate datasets. Cytom Part A J Int Soc Anal Cytol. 2011;79:167–74. doi: 10.1002/cyto.a.21015. http://dx.doi.org/10.1002/cyto.a.21015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim K, Perera R, Tan DB, Fernandez S, Seddiki N, Waring J, French MA. Circulating mycobacterial-reactive CD4+ T cells with an immunosuppressive phenotype are higher in active tuberculosis than latent tuberculosis infection. Tuberc (Edinb) 2014;94:494–501. doi: 10.1016/j.tube.2014.07.002. http://dx.doi.org/10.1016/j.tube.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 17.Mattila JT, Diedrich CR, Lin PL, Phuah J, Flynn JL. Simian immunodeficiency virus-induced changes in T cell cytokine responses in cynomolgus macaques with latent Mycobacterium tuberculosis infection are associated with timing of reactivation. J Immunol. 2011;186:3527–37. doi: 10.4049/jimmunol.1003773. http://dx.doi.org/10.4049/jimmunol.1003773. [DOI] [PMC free article] [PubMed] [Google Scholar]