Abstract
Background:
Since correlations have been found between body composition and physical activity in different parts of the world, inactivity and poor physical condition likely contributes main factors in childhood obesity. This study was implemented to determine the relationship between cardiovascular fitness and body-composition in a group of Iranian middle school students.
Methods:
In this descriptive study, subjects comprised of 12,946 students (10,531 girls and 2,415 boys aged 11–13 years) in the city of Isfahan. Height, weight, body-fat percent, body mass index, and cardiovascular fitness of the aforesaid students were measured by valid tests.
Results:
This study showed that fat percentage and aerobic fitness (VO2 max) of girls were 24.73%, and 29.5 (ml/kg/min) and boys19.32% and 36.4 (ml/kg/min) respectively. Results also revealed that there was a negative significant correlation between fat percent and aerobic fitness of boys (r = −0.81), and girls (r = −0.77) respectively.
Conclusions:
To conclude, this study signifies that fat percentage augmentation leads to a decrease in aerobic fitness of children. Thus, fat percentage can be associated with different chronic diseases such as cardiovascular ones.
Keywords: Body composition, aerobic fitness, Isfahan, obesity, students
INTRODUCTION
Incidence of obesity and overweight in children of some societies had increased dramatically; this is one of the main factors seems related to the development of obesity.[1,2] Different studies in this area indicate that one in four children aged 6–14 years (from 11% to 39%) in developed and developing countries, is overweight or obese.[3] On the other hand, low levels of physical activity are not only associated with the failure of health related physical fitness, but also inactivity correlates with the development of hypercholesterolemia, hypertension, metabolic syndrome, type 2 diabetes and cardiovascular diseases, in both adults and children.[4] In addition, it seems plausible to assume that higher level of physical fitness in children results in a more favorable body composition. Nonetheless, lack of ample data and applicable examinations, in regards to the relationship between aerobic fitness and body fatness, obscure the correlation between cardiovascular fitness and body composition in Iran.[5] Moreover, little is known about the relationship between physical fitness and different body composition indicators such as body mass index (BMI), and the percentage of body-fat in children. To the best of our knowledge, no information is available demonstrating interdependence between obesity and physical activity indicators in Isfahan school children. Therefore, this study was carried out to determine the relationship between cardiovascular fitness and body-composition in school children aged 10–13 years in Isfahan.
METHODS
Subjects
This cross-sectional study comprised 12,946 students of middle schools from the city of Isfahan. Evaluations were performed during February to May 2010 school program, and stratified random sampling was used. At first, 30 schools were identified and randomly selected from the city of Isfahan, and their pupils were become familiar with the aims of the study. Not only, informed consents were obtained from guardians of students, but also medical history questionnaires were filled by parents. Participants and their guardians were informed that they could withdraw from the study at any time. Furthermore, the aims of the study explained to both physical education teachers and school administrators. All of the Participants, at the time of the project implementation, were in good health status comprising different aspects of locomotive, cardiovascular, and endocrine systems. In addition, no participant was on medication at the time of the study.
Measurements
Each child underwent a 1-day testing session. During this session, anthropometric assessment and physical fitness test were carried out. Height and weight were measured by using a stadiometer and calibrated balance beam scale (Novin, Iran) while scale intervals were 0.5 cm and 0.2 kg respectively. Skin-fold tests at three sites were implemented by a validated caliper. The skin-fold site included triceps, subscapular, and medial calf. The landmarks were identified and measured in accordance with Lohman and Timothy procedure.[6] The percentage of body-fat was determined in accordance with gender-specific equations: (If triceps and sub-scapular >35 mm; %fat = 0.783 ∑ SF + I males and %fat = 0.546 ∑ SF + 9.7 females; and If triceps and subscapular <35 mm; %fat = 1.21 [∑SF] – 0.008 [∑SF] 2 + I males and %fat = 1.33 [∑SF] – 0.013 [∑SF] 2 + 2.5 females).[6] BMI was calculated as weight (kg)/height (m)2. Children were considered overweight or obese based on age-specific BMI reference guidelines.[7,8] Both Waist and hip circumferences were measured by a flexible tape at the level of the narrowest point between the lower costal border and the iliac crest, and the widest region of the hip, respectively.[6] Aerobic fitness or VO2 max of subjects was determined by pacer 20-m shuttle-run test.[9] Subjects were required to run back and forth on a 20 m course and be on the 20 m line at the same time a beep is emitted from tape. The frequency of sound signals increased in such a way that running speed starts at 8.5 km/h and increased by 0.5 km/h each min. When the subjects could no longer follow the pace, the stage, the subjects were able to run for, was recorded and used for calculating the VO2 max.[9]
Statistical analysis
Descriptive statistics were run on all the variables. The relationships between body-fat, BMI, and waist-hip ratio (WHR)and aerobic fitness (VO2 max) were examined by Pearson's product-moment correlation coefficient, and P < 0.05 considered as statistically significant. Data were analyzed using the SPSS-PC software (version 20, IBM SPSS inc., USA).
RESULTS
The descriptive data (mean ± standard deviation) of the measured variables including height, weight, WHR, BMI, body fat percent and cardiovascular fitness are shown in Table 1, and the results of statistical analysis and comparison of measured variables are shown in Table 2 and Figures 1-4. There were significant differences between male and female students’ fat percentages (21.12% vs. 19.32% P < 0.05, respectively). In comparison with girls, boys showed significantly lower BMI, WHR, and body-fat percent (P < 0.05). Moreover, boys attained better performance in aerobic fitness test and therefore had higher VO2 max (P < 0.05). Relationship between body-fat and VO2 max for each gender is illustrated in Table 2. There were significant and inverse correlations between fat percent and VO2 max of subjects. This inverse correlation was higher in boys (r = −0.81; P < 0.01), whereas girls demonstrated moderate correlation (r = −0.77; P < 0.01).
Table 1.
Descriptive data related to measured variables in students (n=12,946)

Table 2.
Pearson correlation coefficient between aerobic fitness and body composition of subjects

Figure 1.

Body composition distribustion of subjects
Figure 4.
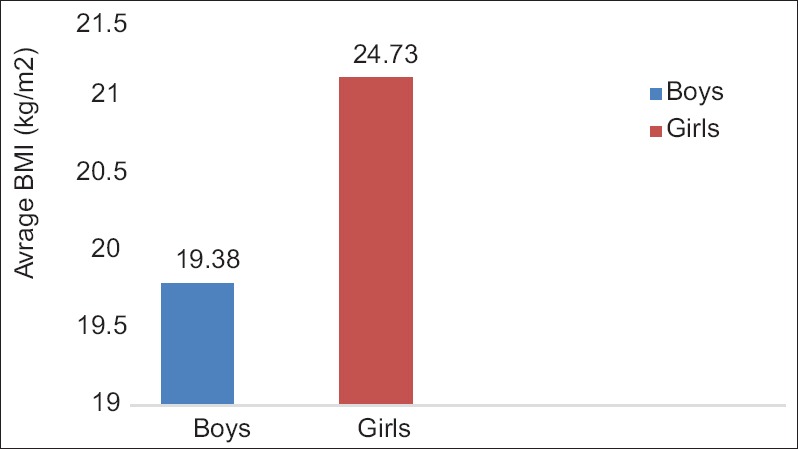
Comparison of body mass index of students
Figure 2.

Comparison of body fat percent of students
Figure 3.

Comparison of aerobic fitness of students
DISCUSSION
This study demonstrated that aerobic fitness increases in inverse proportion to body-fat in Isfahan school children aged 11–14 years, also findings demonstrated that the prevalence of overweight differed between boys and girls. Moreover, the latter ones had lower levels of aerobic fitness compared with their male counterparts. Previous studies reported that the prevalence of obesity in Belgian adolescents was 7.5%. However the prevalence of overweight and obesity differed significantly by level of education.[8,9] It has been reported that prevalence of overweight in Mexican boys and girls using Centers for Disease Control and Prevention percentiles ranged from 10.8% to 16.1% and 14.3% to 19.1% respectively, whereas their obesity prevalence altered differently (from 9.2 to 14.7% and 6.8 to 10.6% in boys and girls respectively). Furthermore, obesity prevalence among Spanish children ranged from 5% to 8%.[9] This prevalence in Qatari adolescents were 7.9% 4.7% in males and females, respectively.[4,10]
Childhood obesity was emerging as a worldwide epidemic problem (38% for boys and 63% girls) when children, who were obese and overweight by BMI criteria, were considered together. Another study reported that over 37% of children from Birmingham fell into overweight and obese classifications. Indeed, one in five children (22%) had more than 30% body fat, and obese group comprised almost girls (90%).[11] This report also showed that 71% of children had a percentage of body-fat in excess of 20%, which was recognized as the upper end of the optimal range. In fact, the above-mentioned study indicates that boys tend to have a higher prevalence of obesity than girls (17% vs. 15% respectively).[11] On the other hand, an interesting gender difference was suggested considering sexual maturation, with a positive association with overweight and obesity in girls but the associations were reverse for boys.[12] One of the primary behavioral and environmental factors are food consumption patterns, sedentary behavior, and socioeconomic status,[12] that led to gradual increase in body weight and obesity, that is also the consequence of a prolonged positive energy balance, that is, when energy intake exceeds energy loss.[2] However, there are many factors that can influence energy balance and therefore, be identified as contributors to the current obesity epidemic in children, with biological, behavioral, environmental, and social being most cited.[13,14] Although the relative contribution of energy intake to the obesity epidemic is controversial, available data clearly indicate that physical activity plays an integral role in the prevention of obesity.[14,15]
Obese children are at an increased risk of acute medical illnesses and chronic diseases, particularly osteoarthritis, diabetes mellitus, and cardiovascular diseases, which can lead to poor quality of life, an increased personal and financial burden to individuals, families, and society, as well as a shortened lifespan.[15,16]
Aerobic fitness or VO2 max average for children assessed in the present study (32.9 ml/kg/min) was lower than previous reports. For example, VO2 max for children aged 8–12 years ranged between 34 and 58 ml/kg/min,[8,6,11] while boys had higher values of VO2 max.[6,11] However, differences in body-composition and level of physical activity, can result in inconsistent reports.[17] For example, Léger and Lambert reported aerobic capacity from 38 to 52 ml/kg/min for children aged 6–18 years.[9]
Cardiovascular fitness or VO2 max changed by gender criteria (29.5 ± 5.6 vs. 36.4 ± 7.3 for girls and boys, respectively) in accordance with the results of previous studies. In fact, aerobic capacity is consistently greater in boys throughout childhood and this difference exceeded at puberty.[11] The observed difference has been ascribed to cumulative effects of different factors including body composition and cardiac size and function.[11] Regular physical activity plays an important role in the maintenance of body weight and composition as well as in the regulation of skeletal muscle and adipose tissue metabolism.
Several investigators reported an association between physical activity and body composition in children.[4,11,18] Precise assessment of habitual physical activity is critical for accurately descriptive epidemiology of the physical activity-obesity relationship for designing appropriate interventions aimed at modifying body composition and related risk factors and for promoting change in lifestyles.[17,19] Assessment of aerobic fitness and body fat are probably a more valid approach than measurement of self-reported physical activity and body-weight, particularly in children.[17,19] It has been reported that overweight and obese children showed a lower physical fitness than normal children.[9,15,20] However, studies analyzing association between aerobic fitness and different measures of body-composition in this population are scarce. Indeed, results of the present study revealed a moderate-strong inverse relationship (r = –0.81 for boys vs. r = –77 for girls) between aerobic fitness and fat percentage of subjects.
Since people who have sedentary lifestyles also have low levels of fitness and excessive body fat,[21] optimal altering of adiposity in children should begin with augmentation of physical activity and fitness, which leads to reductions in body-fat. Moreover, children, who improve their aerobic fitness during childhood, have less overall adiposity and abdominal adiposity than their counterparts during adolescence and adulthood.[21] Due to the fact that BMI poorly correlates with aerobic fitness, other indicators of adiposity (body fat percentage or WHR) rather than weight has been shown to be associated with aerobic fitness.[22,23]
Limitations
One of the main limitations of the study was that the number of male subjects participated in this research, were about one-fifth of female subjects, because of educational curriculum end at May. It seems that more accurate and specific fitness tests are required to determine if there is association between fitness level of children and their body composition. Moreover, analysis of the relationship between aerobic fitness and body-composition needs adjustments for pubertal development factors, which requires further investigation.
CONCLUSIONS
Our results suggest that cardiovascular fitness, as an indicator of physical activity, is linked to the increased level of adiposity in children. Inactivity is interconnected with obesity; however, it is likely to be as one of the easiest factors to be modified. Although guidelines for physical activity in youths recommend involvement in moderate to vigorous physical activities for at least 60 min a day for health promotion and from a weight-control perspective.[1,24] According to the results of the present study and the fact that academic curriculum of most countries of the world and Iranian middle school students included only 90-min physical activity per week, initiatives should be put forward to promote physical activity in children in both school and out-of-school environment. Certainly, diet modifications also have to be considered in implementation of school-based physical activity interventions, and it is vital that researchers and practitioners target alternative venues that still service a large amount of children, but are also accessible.
ACKNOWLEDGMENTS
The authors would also like to thank the study participants for their cooperation and dedication. We wish to thank the Isfahan Provincial Health Office's administrators and personnel that helped and supported us in this study. This study was financially supported by the Provincial Health Office, Isfahan University of Medical Sciences, Isfahan, Iran.
Footnotes
Source of Support: This study was financially supported by the Provincial Health Office, Isfahan University of Medical Sciences, Isfahan - Iran
Conflict of Interest: None declared.
REFERENCES
- 1.Wang Y, Monteiro C, Popkin BM. Trends of obesity and underweight in older children and adolescents in the United States, Brazil, China, and Russia. Am J Clin Nutr. 2002;75:971–7. doi: 10.1093/ajcn/75.6.971. [DOI] [PubMed] [Google Scholar]
- 2.Wang G, Dietz WH. Economic burden of obesity in youths aged 6 to 17 years: 1979-1999. Pediatrics. 2002;109:E81–1. doi: 10.1542/peds.109.5.e81. [DOI] [PubMed] [Google Scholar]
- 3.Ostojic SM, Stojanovic MD, Stojanovic V, Maric J, Njaradi N. Correlation between fitness and fatness in 6-14-year old Serbian school children. J Health Popul Nutr. 2011;29:53–60. doi: 10.3329/jhpn.v29i1.7566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.King AC, Parkinson KN, Adamson AJ, Murray L, Besson H, Reilly JJ, et al. Correlates of objectively measured physical activity and sedentary behaviour in English children. Eur J Public Health. 2011;21:424–31. doi: 10.1093/eurpub/ckq104. [DOI] [PubMed] [Google Scholar]
- 5.Pate RR, Saunders R, Dishman RK, Addy C, Dowda M, Ward DS. Long-term effects of a physical activity intervention in high school girls. Am J Prev Med. 2007;33:276–80. doi: 10.1016/j.amepre.2007.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eston R, Thomas R. 3rd ed. Vol. 1. Abingdon, Oxon, UK: Routledge; 2009. Kinanthropometery and exercise phyisiology laboratory manual: TL, tests, procedures and data. Anthropometry. [Google Scholar]
- 7.Ara I, Moreno LA, Leiva MT, Gutin B, Casajús JA. Adiposity, physical activity, and physical fitness among children from Aragón, Spain. Obesity (Silver Spring) 2007;15:1918–24. doi: 10.1038/oby.2007.228. [DOI] [PubMed] [Google Scholar]
- 8.Ogden CL, Flegal KM, Carroll MD, Johnson CL. Prevalence and trends in overweight among US children and adolescents, 1999-2000. JAMA. 2002;288:1728–32. doi: 10.1001/jama.288.14.1728. [DOI] [PubMed] [Google Scholar]
- 9.Léger LA, Lambert J. A maximal multistage 20-m shuttle run test to predict VO 2 max. Eur J Appl Physiol Occup Physiol. 1982;49:1–12. doi: 10.1007/BF00428958. [DOI] [PubMed] [Google Scholar]
- 10.Molnár D, Livingstone B. Physical activity in relation to overweight and obesity in children and adolescents. Eur J Pediatr. 2000;159(Suppl 1):S45–55. doi: 10.1007/pl00014365. [DOI] [PubMed] [Google Scholar]
- 11.Meigs JB, Muller DC, Nathan DM, Blake DR, Andres R. Baltimore Longitudinal Study of Aging. The natural history of progression from normal glucose tolerance to type 2 diabetes in the Baltimore Longitudinal Study of Aging. Diabetes. 2003;52:1475–84. doi: 10.2337/diabetes.52.6.1475. [DOI] [PubMed] [Google Scholar]
- 12.Al-Nakeeb Y, Duncan MJ, Lyons M, Woodfield L. Body fatness and physical activity levels of young children. Ann Hum Biol. 2007;34:1–12. doi: 10.1080/03014460601079886. [DOI] [PubMed] [Google Scholar]
- 13.Kimm SY, Glynn NW, Obarzanek E, Kriska AM, Daniels SR, Barton BA, et al. Relation between the changes in physical activity and body-mass index during adolescence: A multicentre longitudinal study. Lancet. 2005;366:301–7. doi: 10.1016/S0140-6736(05)66837-7. [DOI] [PubMed] [Google Scholar]
- 14.Centers for Disease Control and Prevention (CDC). Overweight among students in grades K-12 – Arkansas, 2003-04 and 2004-05 school years. MMWR Morb Mortal Wkly Rep. 2006;55:5–8. [PubMed] [Google Scholar]
- 15.Krebs NF, Jacobson MS. American Academy of Pediatrics Committee on Nutrition. Prevention of pediatric overweight and obesity. Pediatrics. 2003;112:424–30. doi: 10.1542/peds.112.2.424. [DOI] [PubMed] [Google Scholar]
- 16.Krassas GE, Tzotzas T, Tsametis C, Konstantinidis T. Prevalence and trends in overweight and obesity among children and adolescents in Thessaloniki, Greece. J Pediatr Endocrinol Metab. 2001;14 Suppl 5:1319–26. [PubMed] [Google Scholar]
- 17.Kain J, Uauy R, Vio F, Albala C. Trends in overweight and obesity prevalence in Chilean children: Comparison of three definitions. Eur J Clin Nutr. 2002;56:200–4. doi: 10.1038/sj.ejcn.1601301. [DOI] [PubMed] [Google Scholar]
- 18.Goran MI, Reynolds KD, Lindquist CH. Role of physical activity in the prevention of obesity in children. Int J Obes Relat Metab Disord. 1999;23 Suppl 3:S18–33. doi: 10.1038/sj.ijo.0800880. [DOI] [PubMed] [Google Scholar]
- 19.Lee SJ, Arslanian SA. Cardiorespiratory fitness and abdominal adiposity in youth. Eur J Clin Nutr. 2007;61:561–5. doi: 10.1038/sj.ejcn.1602541. [DOI] [PubMed] [Google Scholar]
- 20.Brunet M, Chaput JP, Tremblay A. The association between low physical fitness and high body mass index or waist circumference is increasing with age in children: The ‘Québec en Forme’ Project. Int J Obes (Lond) 2007;31:637–43. doi: 10.1038/sj.ijo.0803448. [DOI] [PubMed] [Google Scholar]
- 21.Goran MI, Bergman RN, Avila Q, Watkins M, Ball GD, Shaibi GQ, et al. Impaired glucose tolerance and reduced beta-cell function in overweight Latino children with a positive family history for type 2 diabetes. J Clin Endocrinol Metab. 2004;89:207–12. doi: 10.1210/jc.2003-031402. [DOI] [PubMed] [Google Scholar]
- 22.Shaya FT, Flores D, Gbarayor CM, Wang J. School-based obesity interventions: A literature review. J Sch Health. 2008;78:189–96. doi: 10.1111/j.1746-1561.2008.00285.x. [DOI] [PubMed] [Google Scholar]
- 23.del Río-Navarro BE, Velázquez-Monroy O, Sánchez-Castillo CP, Lara-Esqueda A, Berber A, Fanghänel G, et al. The high prevalence of overweight and obesity in Mexican children. Obes Res. 2004;12:21. doi: 10.1038/oby.2004.28. [DOI] [PubMed] [Google Scholar]
- 24.Grydeland M, Bergh IH, Bjelland M, Lien N, Andersen LF, Ommundsen Y, et al. Intervention effects on physical activity: The HEIA study – A cluster randomized controlled trial. Int J Behav Nutr Phys Act. 2013;10:17. doi: 10.1186/1479-5868-10-17. [DOI] [PMC free article] [PubMed] [Google Scholar]


