Abstract
Essential oils were obtained by hydrodistillation of the flowers+leaves and stems of Artemisia kotuchovii Kupr. (AKEOf+l and AKEOstm, respectively) and analyzed by gas chromatography (GC) and gas chromatography-mass spectrometry (GC/MS). The primary components of the oils were estragole, (E)- and (Z)-β-ocimenes, methyl eugenol, limonene, spathulenol, β-pinene, myrcene, and (E)-methyl cinnamate. Seventy four constituents were present at concentrations from 0.1 to 1.0%, and 34 compounds were identified in trace (<0.1%) amounts in one or both plant components. Screening of the essential oils for biological activity showed that AKEOstm, but not AKEOf+l, inhibited N-formyl-Met-Leu-Phe (fMLF)-stimulated Ca2+ flux and chemotaxis and phorbol-12-myristate-13-acetate (PMA)-induced reactive oxygen species (ROS) production in human neutrophils. Selected pure constituents, representing >96% of the AKEOstm composition, were also tested in human neutrophils and HL-60 cells transfected with N-formyl peptide receptor 1 (FPR1). We found that one component, 6-methyl-3,5-heptadien-2-one (MHDO), inhibited fMLF- and interleukin 8 (IL-8)-stimulated Ca2+ flux, fMLF-induced chemotaxis, and PMA-induced ROS production in human neutrophils. MHDO also inhibited fMLF-induced Ca2+ flux in FPR1-HL60 cells. These results suggest that MHDO may be effective in modulating some innate immune responses, possibly by an inhibition of neutrophil migration and ROS production.
Keywords: Artemisia kotuchovii, calcium flux, chemotaxis, essential oil, neutrophil, N-formyl peptide receptor 1 (FPR1), reactive oxygen species
Introduction
Artemisia is one of largest genus in the Asteraceae family, with approximately 500 species. Many of these species produce essential oils used in folk and modern medicine and in the cosmetics and pharmaceutical industries.1,2 Extracts or compounds isolated from Artemisia have been shown to exhibit a wide range of biological properties, including anti-inflammatory activities that help to reduce inflammation.3,4 Essential oils from several species of this genus have also been reported to decrease the production of proinflammatory mediators.5,6 Extensive studies of the chemical components of these oils have demonstrated that they contain diverse bioactive secondary metabolites, including volatile monoterpenes and sesquiterpenes, with artemisinin as one of best studied.1,2,7,8 However, the chemical composition and biological properties of many endemic Artemisia spp. have not been evaluated.
Artemisia kotuchovii Kupr. is endemic in the Kazakhstan Altai. This perennial herb was described in 1999 and is closely related to A. dracunculus L. (tarragon).9 To date, the chemical composition and biological properties of A. kotuchovii have not been analyzed. Although ethnobotanical and ethnopharmacological data on medicinal usage of this herb are unknown, the related herb tarragon has been reported to have a wide range of applications in traditional medicine because of its therapeutic properties for a variety of ailments.1 For example, pharmacological evaluation of tarragon extracts demonstrated anti-inflammatory activity.1,10 Although tarragon essential oil was inactive in modulating human neutrophil phagocytosis,11 essential oils from other Artemisia spp. exhibited some immunomodulatory properties, such as inhibition of nuclear factor (NF)-κB transcriptional activity and stimulation of nitric oxide and prostaglandin E2 production by macrophages.5,6
Neutrophils are a key cellular component of the immune response to infection or tissue injury.12 These phagocytes are recruited to sites of injury or infection by a variety of factors, including formyl-Met-Leu-Phe (fMLF), a bacterial or mitochondria-derived peptide, and chemokines such as interleukin 8 (IL-8).13 IL-8 and fMLF via activate G-protein coupled receptors (GPCR) to induce neutrophil chemotaxis and the release of various mediators, such as reactive oxygen species (ROS), proteases, and cytokines.13 Growing evidence supporting the anti-inflammatory and tissue-protective effects of chemokine antagonists led to the design and screening of synthetic compounds and plant-derived constituents for novel small-molecule N-formyl peptide receptor (FPR) and α-chemokine receptor (CXCR) antagonists.14,15 Human neutrophil elastase (HNE) also plays an important role in regulation of inflammation, and some plant-derived compounds have been identified as elastase inhibitors.16
In the present work, we defined the composition of essential oils from A. kotuchovii and evaluated their biological activity.
Materials and Methods
Chemicals
The major and several minor constituents of essential oils were obtained from commercial sources. Sabinene, (−) linalool, hexanal, p-cymene-8-ol, 3,7-dimethyl-1,3,6-octatriene (β-ocimene), and MHDO were purchased from Sigma-Aldrich Chemical Co. (St. Loius, MO). Citronellol, (+)-limonene, trans-anethole, (−)-caryophyllene oxide, 4-allylanisole (estragole), 1,2-dimethoxy-4-(2-propenyl) (methyl eugenol), myrcene, isopropyl myristate, p-cymene, α-terpinene, and (−)-limonene were purchased from Acros Organics (Geel, Belgium). Myristic acid, α-pinene, and terpinolene were from Santa Cruz Biotechnology (Dallas, TX). Methyl cinnamate, 4-methoxystyrene (4-vinylanisole), palmitic (hexadecanoic) acid, and (1S)-(−)-β-pinene were from Alfa Aesar (Ward Hill, MA). For biological evaluation, compounds were dissolved in DMSO (20 mM stock solutions) and stored at −20°C.
Materials
8-Amino-5-chloro-7-phenylpyridol[3,4-d]pyridazine-1,4(2H,3H)-dione (L-012) was obtained from Wako Chemicals (Richmond, VA). Dimethyl sulfoxide (DMSO), fMLF, HEPES, bacterial lipopolysaccharide (LPS) from Escherichia coli K-235, phorbol-12-myristate-13-acetate (PMA), and Histopaque 1077 were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO). Roswell Park Memorial Institute (RPMI) 1640 medium and penicillin-streptomycin solution were purchased from Mediatech (Herdon, VA). Human neutrophil elastase, N-methylsuccinyl-Ala-Ala-Pro-Val-7-amino-4-methylcoumarin, ionomycin, and bovine serum albumin were purchased from EMD Biosciences (San Diego, CA). Fetal bovine serum (FBS) was purchased from Atlas Biologicals (Fort Collins, CO). Human IL-8 was purchased from Peprotech Inc. (Rocky Hill, NJ). Tween-20 was from VWR (Radnor, PA). Tris was from J.T. Baker (Phillipsburg, NJ). Blasticidin S and zeocin were obtained from Invivogen (San Diego, CA). Hanks' balanced-salt solution (HBSS; 0.137 M NaCl, 5.4 mM KCl, 0.25 mM Na2HPO4, 0.44 mM KH2PO4, 4.2 mM NaHCO3, 5.56 mM glucose, and 10 mM HEPES, pH 7.4), and G418 were from Life Technologies (Grand Island, NY). HBSS containing 1.3 mM CaCl2 and 1.0 mM MgSO4 is designated as HBSS+; HBSS without ions Ca2+ and Mg2+ is designated as HBSS−.
Plant material
Aerial parts of A. kotuchovii were collected at the end of the blossoming stage in August 2013 in the southern Altai on the Tarbagatai range at altitude 1,709 m above sea level (latitude N 49°03'52", longitude W 85°59'35"). The plant was identified by Dr. Yuriy Kotukhov, and voucher specimens were deposited at the Institute of Plant Biology and Biotechnology (Almaty, Kazakhstan). Plants components were air-dried for 7–10 days at room temperature away from direct sunlight. Weighed samples were cut under laboratory conditions before hydrodistillation.
Essential Oil Extraction
Two types of air dried material (inflorescence+leaves or stems) were separately subjected to hydrodistillation for 3 h using a Clevenger type apparatus to yield essential oils. Conventional hydrodistillation is considered the primary method for essential oil extraction.17 Although hydrodistillation could lead to artifacts at when performed at higher temperatures over long hydrodistillation times at low pH,18 we only applied conditions accepted by the European Pharmacopoeia (European Directorate for the Quality of Medicines, Council of Europe, Strasbourg, France, 2014) and thus avoided these artifacts. Solutions of the essential oils in DMSO (10 mg/ml stock solutions) for biological evaluation and in n-hexane (10% w/v) for gas-chromatographic analysis.
GC/MS analysis
Gas chromatography–mass spectrometry (GC/MS) analysis of the oils was carried out with an Agilent 5975 GC/MSD system, as reported previously.19,20 An Innowax FSC column (60 m × 0.25 mm, 0.25 µm film thickness) was used with He as carrier gas (0.8 mL/min). GC oven temperature was kept at 60°C for 10 min, increased to 220°C at a rate of 4°C/min, kept constant at 220°C for 10 min, and then increased to 240°C at a rate of 1°C/min. The split ratio was adjusted to 40:1, and the injector temperature was at 250°C. MS were taken at 70 eV. Mass range was from m/z 35 to 450.
GC analysis was carried out using an Agilent 6890N GC system. In order to obtain the same elution order as with GC/MS, simultaneous injection was performed using the same column and appropriate operational conditions. Flame ionization detector (FID) temperature was 300°C. The components of essential oils were identified by coinjection with standards (whenever possible), which were purchased from commercial sources and/or isolated from natural sources. In addition, compound identities were confirmed by comparison of their mass spectra with those in Wiley GC/MS Library (Wiley, New York, NY, USA), MassFinder software 4.0 (Dr. Hochmuth Scientific Consulting, Hamburg, Germany), Adams Library, and NIST Library. A C8–C40 n-alkane standard solution (Fluka, Buchs, Switzerland) was used to spike the samples for the determination of relative retention indices (RRI). Relative percentage amounts of the separated compounds were calculated from FID chromatograms.
Cell Culture
Human MonoMac-6 monocytic cells (DSMZ, Germany) were grown in RPMI 1640 supplemented with 10% (v/v) endotoxin-free FBS, 10 µg/ml bovine insulin, 100 µg/ml streptomycin, and 100 U/ml penicillin. Human monocytic THP-1Blue cells obtained from InvivoGen (San Diego, CA) were cultured in RPMI 1640 medium supplemented with 10% (v/v) endotoxin-free FBS, 100 µg/ml streptomycin, 100 U/ml penicillin, 100 µg/ml zeocin, and 10 µg/ml blasticidin S. These cells are stably transfected with a secreted embryonic alkaline phosphatase gene that is under the control of a promoter inducible by NF-κB and AP-1. Human promyelocytic leukemia HL-60 cells stably transfected with human FPR1 (FPR1-HL60 cells) were cultured in RPMI-1640 medium supplemented with 10% heat-inactivated fetal calf serum, 10 mM HEPES, 100 µg/ml streptomycin, 100 U/ml penicillin, and G418 (1 mg/ml).15
Measurement of TNF production
MonoMac-6 cells were plated in culture medium supplemented with 3% (v/v) endotoxin-free FBS in 96-well plates at a density of 2×105 cells in 100 µl per well. The cells were pre-treated with or without essential oil or DMSO (vehicle control) for 30 min, followed by treatment with LPS (200 ng/ml) and incubation for 24 h at 37°C and 5% CO2. An enzyme-linked immunosorbent assay kit for human tumor necrosis factor (TNF) (Biolegend; San Diego, CA) was used to measure cytokine levels in the cell supernatants.
Analysis of NF-κB/AP-1 activation
Activation of NF-κB/AP-1 transcriptional activity was measured using an alkaline phosphatase reporter gene assay in THP1-Blue cells (2×105 cells in 100 µl per well). The cells were pre-treated with or without essential oil or DMSO (vehicle control) for 30 min, followed by treatment with LPS (200 ng/ml) and incubation for 24 h at 37°C and 5% CO2. Alkaline phosphatase activity was measured in cell supernatants using QUANTI-Blue mix (InvivoGen). Activation of NF-κB is reported as absorbance at 655 nm and compared with positive control samples (LPS).
Human Neutrophil Elastase (HNE) Inhibition Assay
Essential oils and individual compounds were dissolved in 100% DMSO at 5 mM stock concentrations. The final concentration of DMSO in the reactions was 1%, and this level of DMSO had no effect on enzyme activity. One of our previously described HNE inhibitors (compound 5b) was used as a positive control.21 The inhibition assay was performed, as described previously.22 Briefly, a buffer solution containing 200 mM Tris-HCl, pH 7.5, 0.01% bovine serum albumin, and 0.05% Tween-20 and 20 mU/mL of human neutrophil elastase was added to black, flat-bottom 96-well microtiter plates containing different concentrations of test compounds. Reactions were initiated by addition of 25 µM elastase substrate N-methylsuccinyl-Ala-Ala-Pro-Val-7-amino-4-methylcoumarin in a final reaction volume of 100 µL/well. Kinetic measurements were obtained every 30 s for 10 min at 25°C using a Fluoroskan Ascent FL fluorescence microplate reader (Thermo Electron, MA) with excitation and emission wavelengths at 355 and 460 nm, respectively. The concentration of inhibitor that caused 50% inhibition of the enzymatic reaction (IC50) was calculated by plotting % inhibition versus logarithm of inhibitor concentration.
Neutrophil Isolation
For isolation of human neutrophils, blood was collected from healthy donors in accordance with a protocol approved by the Institutional Review Board at Montana State University. Neutrophils were purified from the blood using dextran sedimentation, followed by Histopaque 1077 gradient separation and hypotonic lysis of red blood cells, as described previously.15 Isolated neutrophils were washed twice and resuspended in HBSS. Neutrophil preparations were routinely >95% pure, as determined by light microscopy, and >98% viable, as determined by trypan blue exclusion.
Ca2+ Mobilization Assay
Changes in intracellular Ca2+ were measured with a FlexStation II scanning fluorometer using fluorescent dye Fluo-4AM (Invitrogen). Neutrophils or FPR1-HL60 cells, suspended in HBSS, were loaded with Fluo-4AM dye (final concentration, 1.25 µg/ml) and incubated for 30 min in the dark at 37°C. After dye loading, the cells were washed with HBSS, resuspended in HBSS+, separated into aliquots, and deposited into the wells of flat-bottomed, half-area well black microtiter plates (2×105 cells/well). The compound source plate contained dilutions of test essential oil or pure compound in HBSS+, and changes in fluorescence were monitored (λex = 485 nm, λem = 538 nm) every 5 s for 240 s at room temperature after automated addition of compounds. Maximum change in fluorescence, expressed in arbitrary units over baseline, was used to determine agonist response. Responses were normalized to the response induced by 5 nM fMLF, which were assigned a value of 100%. Curve fitting (at least five to six points) and calculation of median effective concentration values (EC50 or IC50) were performed by nonlinear regression analysis of the dose-response curves generated using Prism 5 (GraphPad Software, Inc., San Diego, CA).
Chemotaxis Assay
Human neutrophils were suspended in HBSS+ containing 2% (v/v) heat-inactivated fetal bovine serum (2×106 cells/ml), and chemotaxis was analyzed in 96-well ChemoTx chemotaxis chambers (Neuroprobe, Gaithersburg, MD), as described previously.23 In brief, neutrophils were preincubated with the indicated concentrations of the test sample (oil or pure compound) or DMSO for 30 min at room temperature and added to the upper wells of the ChemoTx chemotaxis chambers. The lower wells were loaded with 30 µl of HBSS+ containing 2% (v/v) fetal bovine serum and the indicated concentrations of test sample, DMSO (negative control), or 1 nM fMLF as a positive control. Neutrophils were added to the upper wells and allowed to migrate through the 5.0-µm pore polycarbonate membrane filter for 60 min at 37°C and 5% CO2. The number of migrated cells was determined by measuring ATP in lysates of transmigrated cells using a luminescence-based assay (CellTiter-Glo; Promega, Madison, WI), and luminescence measurements were converted to absolute cell numbers by comparison of the values with standard curves obtained with known numbers of neutrophils. Curve fitting (at least eight to nine points) and calculation of median effective concentration values (IC50) were performed by nonlinear regression analysis of the dose-response curves generated using Prism 5.
ROS production
ROS production was determined by monitoring L-012-enhanced chemiluminescence, which represents a sensitive and reliable method for detecting superoxide anion (O2−.) production.23 Human neutrophils were resuspended at 2×105 cells/ml in HBSS+ supplemented with 40 µM L-012. Cells (100 µl) were aliquoted into wells of 96-well flat-bottomed microtiter plates containing essential oil or compounds at different concentrations (final DMSO concentration of 1%). Cells were preincubated for 30 min, and 200 nM PMA was added to each well to stimulate ROS production. Luminescence was monitored for 60 min (2-min intervals) at 37°C using a Fluroscan Ascent FL microtiter plate reader (Thermo Electron, Waltham, MA). The curve of light intensity (in relative luminescence units) was plotted against time, and the area under the curve was calculated as total luminescence. The compound concentration that inhibited ROS production by 50% of the PMA-induced response (positive control) was determined by graphing the percentage inhibition of ROS production versus the logarithm of concentration of test sample (IC50). Each curve was determined using five to seven concentrations.
Compound Cytotoxicity
Cytotoxicity was analyzed with a CellTiter-Glo Luminescent Cell Viability Assay Kit (Promega), according to the manufacturer's protocol. Briefly, MonoMac-6 cells were cultured at a density of 1×105 cells/well with different concentrations of essential oil or compound for 4 or 24 h at 37°C and 5% CO2. Following treatment, the cells were allowed to equilibrate to room temperature for 30 min, substrate was added, and the samples were analyzed with a Fluoroscan Ascent FL microplate reader.
Results
Composition of the essential oils from A. kotuchovii
Essential oils were obtained by conventional hydrodistillation of the dried stems or flowers+leaves of A. kotuchovii (designated as AKEOstm and AKEOf/l, respectively) and analyzed by GC and GC/MS techniques simultaneously to determine their chemical compositions. Hydrodistillation of the stems and flowers+leaves produced 0.096 and 1.010% (v/w on the basis of the weight of dried material) essential oils, respectively. The main constituents of AKEOf/l and AKEOstm were estragole (75.1 and 76.6%), (E and Z)-β-ocimene (9.2 and 8.2%), methyl eugenol (4.3 and 4.6%), limonene (1.0 and 1.0%), spathulenol (0.8 and 1.0%), β-pinene (0.9 and 1.0%), myrcene (1.5 and 0.7%), and (E)-methyl cinnamate (1.9 and 0.5%, respectively). Seventy four constituents were present at concentrations of 0.1 to <1.0% in one or both plant sources. The remaining 34 volatile compounds were identified in trace amounts, and their relative percentage amounts are indicated in Table 1. Thus, the major components of the AKEOf/l and AKEOstm are phenylpropanoids (81.5 and 79.6%, respectively) and monoterpene hydrocarbons (13.0 and 14.4%, respectively). Sesquiterpenes compose around 2–2.2% in both plant components. It should be noted that AKEOstm had more oxygenated monoterpenes and fatty acids compared to AKEOf/l (0.5 vs. 0.1% and 0.5 vs. 0.2%, respectively).
Table 1.
Composition of the volatile compounds identified in the essential oils from A. kotuchovii
| Cmpd | RRI | Compound name | A (%) |
B (%) |
Cmpd | RRI | Compound name | A (%) |
B (%) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1032 | α-Pinene* | 0.6 | 0.6 | 59 | 1783 | β-Sesquiphellandrene** | 0.2 | 0.2 |
| 2 | 1035 | α-Thujene** | t | t | 60 | 1786 | ar-Curcumene** | 0.1 | 0.1 |
| 3 | 1093 | Hexanal* | t | 0.1 | 61 | 1798 | Methyl salicylate* | - | t |
| 4 | 1118 | β-Pinene* | 0.9 | 1.0 | 62 | 1804 | Myrtenol* | - | t |
| 5 | 1132 | Sabinene* | 0.3 | 0.3 | 63 | 1815 | 2,6-Dimethyl-3(E),5(Z),7-octatriene-2-ol** | t | t |
| 6 | 1174 | Myrcene* | 1.5 | 0.7 | 64 | 1827 | (E,E)-2,4-Decadienal | - | t |
| 7 | 1188 | α-Terpinene* | t | 0.4 | 65 | 1830 | 2,6-Dimethyl-3(E),5(E),7-octatriene-2-ol** | 0.1 | t |
| 8 | 1195 | Dehydro-1,8-cineole** | t | 0.1 | 66 | 1845 | (E)-Anethole* | t | 0.1 |
| 9 | 1203 | Limonene* | 1.0 | 1.0 | 67 | 1857 | Geraniol* | - | t |
| 10 | 1218 | β-Phellandrene** | 0.2 | 0.1 | 68 | 1864 | p-Cymen-8-ol* | 0.1 | t |
| 11 | 1244 | Amyl furan ** | t | t | 69 | 1868 | (E)-Geranyl acetone* | t | t |
| 12 | 1246 | (Z)-β-Ocimene* | 3.9 | 3.8 | 70 | 1885 | 1-Phenyl-3-methylpenta-1,2,4-triene** | 0.1 | - |
| 13 | 1255 | γ-Terpinene* | t | t | 71 | 1893 | (Z)- Methyl cinnamate** | t | t |
| 14 | 1266 | (E)-β-Ocimene* | 5.3 | 4.4 | 72 | 1958 | (E)-β-Ionone* | t | t |
| 15 | 1280 | p-Cymene* | 0.3 | 0.5 | 73 | 1988 | Isopropyl myristate* | - | 0.1 |
| 16 | 1290 | Terpinolene* | 0.3 | 0.1 | 74 | 1990 | Anisaldehyde* | - | t |
| 17 | 1348 | 6-Methyl-5-hepten-2-one* | - | t | 75 | 2001 | Isocaryophyllene oxide** | t | t |
| 18 | 1362 | cis-Rose oxide** | - | t | 76 | 2008 | Caryophyllene oxide* | 0.4 | 0.2 |
| 19 | 1376 | trans-Rose oxide** | - | t | 77 | 2014 | (E)-Cinnamaldehyde* | t | - |
| 20 | 1382 | cis-Alloocimene** | t | t | 78 | 2030 | Methyl eugenol* | 4.3 | 4.6 |
| 21 | 1413 | Roze furan** | 0.1 | 0.1 | 79 | 2037 | Salvial-4(14)-en-1-one** | t | 0.1 |
| 22 | 1416 | 4,8-Dimethyl-1,3,7-nonatriene** | t | - | 80 | 2041 | Pentadecanal** | - | t |
| 23 | 1429 | Perillene** | t | t | 81 | 2065 | Salviadienol** | t | - |
| 24 | 1446 | 2,6-Dimethyl-1,3(E),5(Z),7-octatetraene** | t | - | 82 | 2071 | Humulene epoxide-II** | t | - |
| 25 | 1452 | α,p-Dimethylstyrene* | t | - | 83 | 2096 | (E)-Methyl cinnamate* | 1.9 | 0.5 |
| 26 | 1452 | 1-Octen-3-ol* | - | t | 84 | 2097 | (E)-Ethyl cinnamate** | - | t |
| 27 | 1458 | cis-1,2-Limonene epoxide* | t | - | 85 | 2124 | Zingiberenol** | t | t |
| 28 | 1460 | 2,6-Dimethyl-1,3(E),5(E),7-octatetraene** | - | t | 86 | 2131 | Hexahydrofarnesyl acetone** | t | t |
| 29 | 1468 | trans-1,2-Limonene epoxide** | t | - | 87 | 2139 | Chavibetol** | t | - |
| 30 | 1474 | trans-Sabinene hydrate** | t | - | 88 | 2144 | Spathulenol** | 0.8 | 1.0 |
| 31 | 1476 | (Z)-β-Ocimene epoxide** | t | t | 89 | 2179 | 3,4-Dimethyl-5-pentylidene-2(5H)- furanone** | - | t |
| 32 | 1479 | (E,Z)-2,4-Heptadienal** | t | - | 90 | 2187 | T-Cadinol** | t | t |
| 33 | 1487 | Isoneroloxide** | t | - | 91 | 2204 | Alismol** | 0.1 | 0.2 |
| 34 | 1498 | (E)-β-Ocimene epoxide** | t | t | 92 | 2214 | ar-Turmerol** | t | - |
| 35 | 1520 | 3,5-Octadien-2-one** | t | t | 93 | 2228 | Isospathulenol** | t | - |
| 36 | 1535 | β-Bourbonene** | t | - | 94 | 2232 | α-Bisabolol* | t | - |
| 37 | 1541 | Benzaldehyde* | t | t | 95 | 2245 | Elemicin** | 0.2 | 0.1 |
| 38 | 1542 | α- Isocomene** | - | t | 96 | 2247 | trans-α-Bergamotol** | t | 0.1 |
| 39 | 1553 | Linaloola | - | 0.2 | 97 | 2255 | α-Cadinol** | t | - |
| 40 | 1586 | Pinocarvone** | - | t | 98 | 2257 | β-Eudesmol* | - | t |
| 41 | 1602 | 6-Methyl-3,5-heptadien-2 one (MHDO)* | - | 0.1 | 99 | 2259 | Eudesma-4(15),7-dien-1-ol* | - | 0.1 |
| 42 | 1611 | Terpinen-4-ol* | t | t | 100 | 2268 | Torilenol** | t | t |
| 43 | 1612 | β-Caryophyllene* | 0.1 | t | 101 | 2269 | (6S,7R)-Bisabolone** | - | t |
| 44 | 1639 | trans-p-Mentha-2,8-dien-1-ol** | t | - | 102 | 2281 | Chavicol** | t | - |
| 45 | 1670 | Benzeneacetaldehyde* | t | - | 103 | 2290 | (2E,6Z)-Farnesol* | t | - |
| 46 | 1672 | trans-Pinocarveol* | t | - | 104 | 2375 | Eudesma-4(15),7-dien-1-β-ol** | 0.1 | 0.1 |
| 47 | 1671 | Acetophenone* | t | - | 105 | 2300 | Tricosane* | - | t |
| 48 | 1687 | α-Humulene* | t | t | 106 | 2309 | Farnesylacetone* | t | - |
| 49 | 1687 | Estragole* | 75.1 | 76.6 | 107 | 2360 | 6-Dodecen-4-olide** | t | - |
| 50 | 1694 | p-Vinylanisole* | - | 0.1 | 108 | 2369 | (2E,6E)-Farnesol* | - | t |
| 51 | 1706 | α-Terpineol* | t | 0.1 | 109 | 2392 | Caryophylla-2(12),6-dien-5β-ol ** | t | - |
| 52 | 1726 | α-Zingiberene** | - | t | 110 | 2415 | Demethoxyencecaline** | 0.1 | 0.1 |
| 53 | 1726 | Germacrene D** | 0.2 | 0.1 | 111 | 2500 | Pentacosane* | - | t |
| 54 | 1733 | Neryl acetate* | t | - | 112 | 2528 | (E)-p-Methoxy-cinnamaldehyde** | t | - |
| 55 | 1755 | Bicyclogermacrene** | - | t | 113 | 2622 | Phytol* | t | - |
| 56 | 1765 | Geranyl acetate* | t | t | 114 | 2670 | Myristic acid* | 0.2 | 0.1 |
| 57 | 1772 | Citronellol* | t | 0.1 | 115 | 2700 | Heptacosane* | - | t |
| 58 | 1773 | δ-Cadinene** | t | - | 116 | 2931 | Palmitic acid* | t | 0.4 |
The data are presented as relative % for components of essential oil from flowers+leaves (A) and essential oil from stems (B). RRI: Relative retention indices calculated against n-alkanes; % calculated from flame ionization detector data. Trace amounts (tr) are present at < 0.1 %.
Identification based on comparison with co-injected with standards.
Tentatively identified using Wiley, MassFinder mass spectra libraries and published RRI. Compounds that were selected for further biological screening are indicated in bold.
Evaluation of essential oil effect on phagocyte function
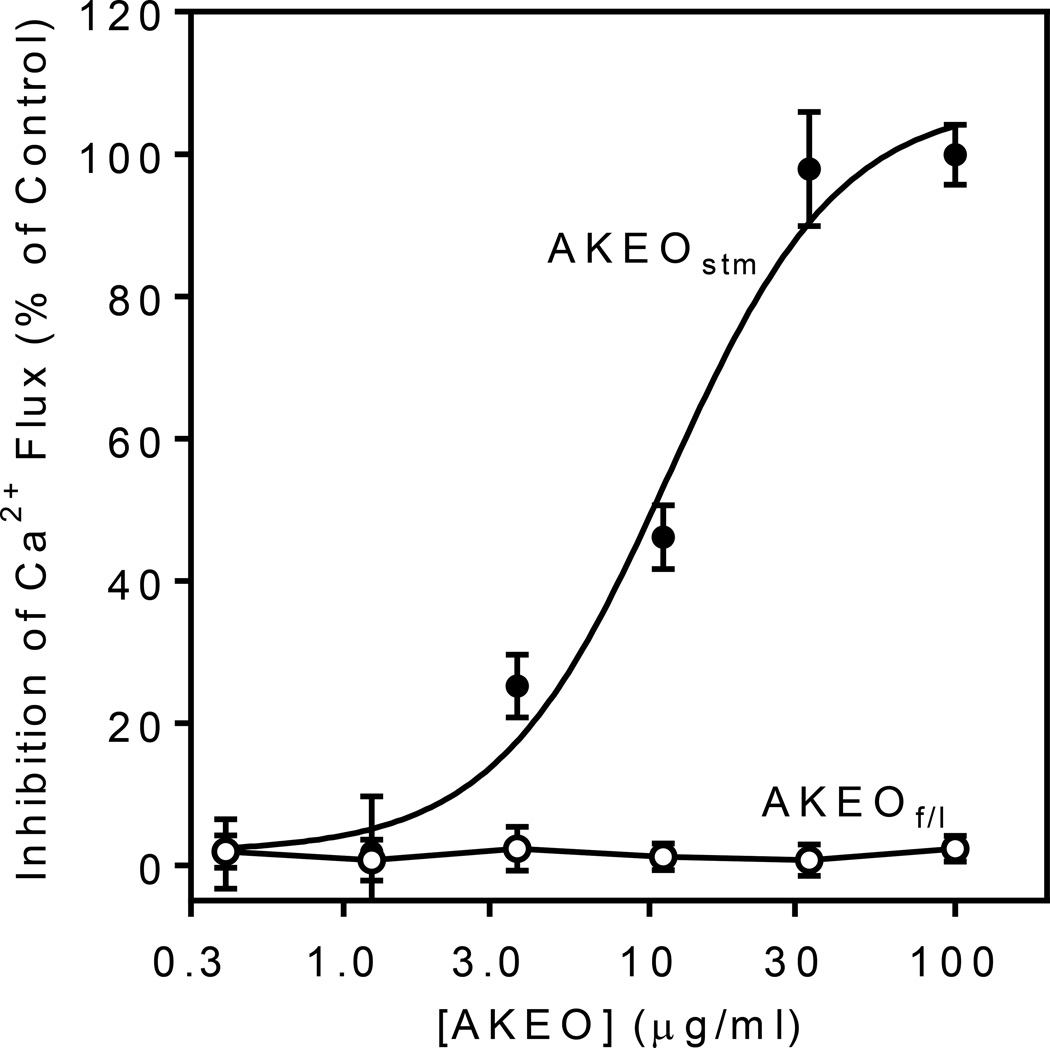
The essential oils from A. kotuchovii were screened for modulatory activity in various cell and enzymatic systems related to mechanisms of innate immunity (Table 2). Essential oils and their components have been reported previously to modulate intracellular Ca2+ levels 24 and inhibit of leukocyte migration.25,26 We found that AKEOstm, but not AKEOf/l, inhibited fMLF-stimulated Ca2+ flux and fMLF-induced chemotaxis in human neutrophils, with IC50 values of 12.5 and 10.1 µg/ml, respectively (Figure 1 and Table 2). Recently, several essential oils were reported to modulate ROS production in neutrophils.27,28 Thus, we evaluated the effect of AKEOstm and AKEOf/l on PMA-induced ROS production by human neutrophils and found that AKEOstm inhibited ROS production with an IC50 of 49.2 µg/ml, whereas AKEOf/l had no activity (Table 2). Although some essential oils and their components were previously identified as HNE inhibitors,29 evaluation of AKEOstm and AKEOf/l showed that they did not inhibit HNE, even at concentrations up to 50 µg/ml.
Table 2.
Biological screening of the essential oils from A. kotuchovii
| Biological Activity | AKEOf/l | AKEOstm |
|---|---|---|
| IC50 (µg/ml) | ||
| LPS-induced NF-κB/AP-1 activity in THP-1Blue cells | N.A. | N.A. |
| LPS-induced TNF production in MonoMac-6 cells | N.A. | N.A. |
| Neutrophil elastase enzymatic activity | N.A. | N.A. |
| fMLF-induced Ca2+ flux in human neutrophilsa | N.A. | 12.5 ± 2.7 |
| fMLF-induced chemotaxis in human neutrophilsb | N.A. | 10.1 ± 3.1 |
| PMA-induced ROS production in human neutrophilsc | N.A. | 49.2 ± 5.4 |
Activity was evaluated as inhibition of Ca2+ flux induced by 5 nM fMLF in neutrophils.
Inhibition of chemotactic activity in neutrophils was evaluated in the presence of 0.5 nM fMLF.
Inhibition of ROS production in neutrophils was evaluated in the presence of 200 nM PMA and 40 µM L-012 . N.A.: no activity was observed, even at the highest concentration tested (50 µM).
Figure 1.
Effect of essential oils from A. kotuchovii on Ca2+ mobilization in human neutrophils. Neutrophils were preincubated with the indicated concentrations of essential oils isolated from stems (AKEOstm) (closed circles) and flowers+leaves (AKEOf/l) (open circles) of A. kotuchovii for 30 min at 25 °C and then stimulated with 5 nM of fMLF. The response induced by the peptide agonist alone was assigned a value of 100%. Values are the mean ± S.D. of triplicate samples from one experiment that is representative of three independent experiments.
Because various essential oils have been reported to inhibit NF-κB/AP-1 transcriptional activity and production of pro-inflammatory cytokines,30,31 we also evaluated the effects of AKEOstm and AKEOf/l on these responses using cultures of monocytic cells. However, we found that AKEOstm and AKEOf/l did not alter NF-κB/AP-1 activity or TNF production in monocytic cells (Table 2).
Although our functional cell-based assays suggested that the essential oils were relatively nontoxic, we evaluated their potential cytotoxic effects to determine if the results might be influenced by background cytotoxicity. Using a cytotoxicity assay, we determined that neither of the essential oils significantly affected viability of MonoMac-6 cells over a concentration range up to 50 µg/ml, further demonstrating that AKEOstm and AKEOf/l were not cytotoxic (data not shown).
Effect of AKEOstm components on neutrophil function
Because AKEOstm inhibited neutrophil functional responses, we focused on analysis of the effects of AKEOstm constituents to possibly identify the active compound(s). Twenty five commercially available components of AKEOstm, including eight major (≥1%) and seventeen minor compounds, were tested. Note that among the minor constituents tested, hexanal, α-terpinene, p-cymene, MHDO, (E)-anethole, isopropyl myristate, and palmitic acid were all present and higher levels in AKEOstm compared to AKEOf/l (Table 1).
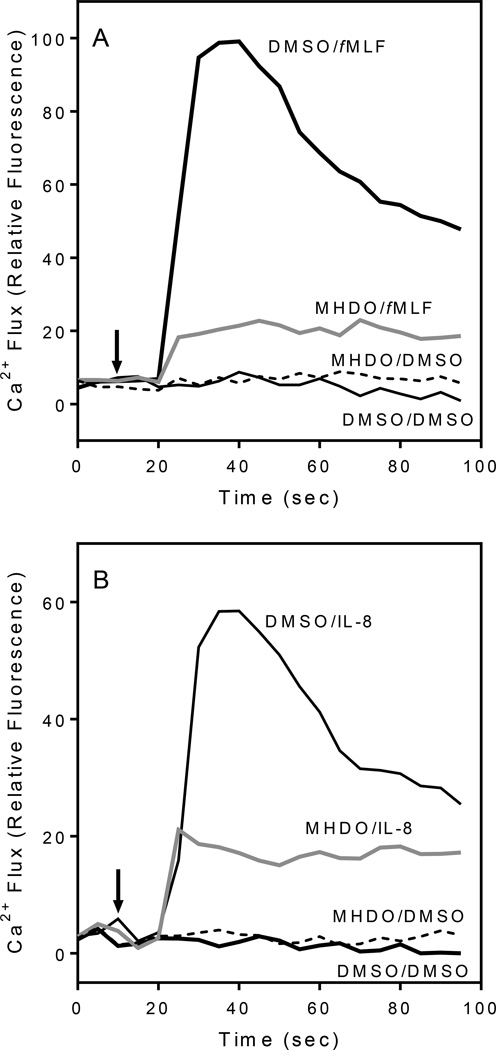
All selected compounds were evaluated for agonist and antagonist effects on Ca2+ flux in human neutrophils. We found that four compounds from different chemical classes, including β-pinene, sabinene, palmitic acid, and myristic fatty acid, had direct but weak agonist activity, as determined by monitoring Ca2+ flux (Table 3). Of all of the component compounds tested, only MHDO inhibited fMLF-stimulated Ca2+ mobilization in neutrophils (IC50 = 8.2 µM), which is far below our common hit threshold of 50 µM,15 and this inhibition was concentration-dependent (Table 2 and Figure 2A). MHDO did not directly activate Ca2+ flux, suggesting it was not down-regulating the subsequent response to fMLF but was directly inhibiting fMLF-stimulated Ca2+ mobilization via a different mechanism. Furthermore, MHDO inhibited Ca2+ flux in fMLF-stimulated FPR1-HL60 cells (Table 2) and IL-8-induced Ca2+ mobilization in human neutrophils (IC50 = 3.2 µM) (Figure 2B). On the other hand, MHDO did not inhibit non-specific Ca2+ flux induced by 10 µM ionomycin (data not shown). Thus, these data indicate that MHDO can modulate intracellular signaling pathways that are common to both FPR1 and CXCR1/2 chemokine receptors, but it is not an ion channel inhibitor or a calcium chelator.
Table 3.
Effect of selected volatile compounds on functional responses in human neutrophil and FPR1-HL60 cells
| Compound Name | Ca2+ flux | Inhibition of Chemotaxisb (IC50) |
Inhibition of ROS Productionc (IC50) |
||
|---|---|---|---|---|---|
| Neutrophils | FPR1-HL60 cells |
||||
| Activation (EC50) |
Inhibitiona (IC50) |
Inhibitiona (IC50) |
|||
| µM (µg/ml) | |||||
| (1R)-(+)-α-Pinene | N.A. | N.A. | N.A. | N.A. | N.A. |
| Hexanal | N.A. | N.A. | N.A. | N.A. | N.A. |
| β-Pinene | 23.8 ± 3.1 (3.2 ± 0.4) |
N.A. | N.A. | 22.7 ± 2.6 (3.1 ± 0.4) |
N.A. |
| Sabinene (+/−) | 49.4 ± 6.3 (6.7 ± 0.86) |
N.A. | N.A. | 37.4 ± 4.3 (5.1 ± 0.6) |
N.A. |
| Myrcene | N.A. | N.A. | N.A. | N.A. | N.A. |
| α-Terpinene | N.A. | N.A. | N.A. | N.A. | N.A. |
| (R)-(+)-Limonene | N.A. | N.A. | N.A. | N.A. | N.A. |
| (S)-(−)-Limonene | N.A. | N.A. | N.A. | N.A. | N.A. |
| (E/Z)-β-Ocimene | N.A. | N.A. | N.A. | N.A. | N.A. |
| p-Cymene | N.A. | N.A. | N.A. | N.A. | N.A. |
| Terpinolene | N.A. | N.A. | N.A. | N.A | N.A. |
| (−)-Linalool | N.A. | N.A. | N.A. | N.A. | N.A. |
| MHDO | N.A. | 8.2 ± 2.5 (1.02 ± 0.31) |
18.0 ± 5.4 (2.2 ± 0.7) |
3.6 ± 0.5 (0.45 ± 0.16) |
2.8 ± 0.4 (0.35 ± 0.05) |
| Estragol | N.A. | N.A. | N.A. | N.A. | N.A. |
| 4-Vinylanisole | N.A. | N.A. | N.A. | N.A. | N.A. |
| (+/−)-β-Citronellol | N.A. | N.A. | N.A. | 48.6 ± 3.7 (7.6 ± 0.6) |
N.A. |
| (E)-Anethole | N.A. | N.A. | N.A. | N.A. | N.A. |
| p-Cymen-8-ol | N.A. | N.A. | N.A. | N.A. | N.A. |
| Isopropyl myristate | N.A. | N.A. | N.A. | N.A. | N.A. |
| (−)-Caryophyllene oxide | N.A. | N.A. | N.A. | N.A. | N.A. |
| Methyl eugenol | N.A. | N.A. | N.A. | N.A. | N.A. |
| (E)-Methyl cinnamate | N.A. | N.A. | N.A. | N.A. | N.A. |
| Elemicine | N.A. | N.A. | N.A. | 46.9 ± 4.2 (9.8 ± 0.9) |
N.A. |
| Myristic acid | 46.7 ± 5.3 (10.7 ± 1.2) |
N.A. | 12.9 ± 3.4 (3.0 ± 0.8) |
N.A. | N.A. |
| Palmitic acid | 40.7 ± 4.6 (10.4 ± 1.2) |
N.A. | N.A. | N.A. | N.A. |
Inhibition of Ca2+ flux induced by 5 nM fMLF.
Inhibition of chemotactic activity in neutrophils was evaluated in the presence of 0.5 nM fMLF.
Inhibition of ROS production in neutrophils was evaluated in the presence of 200 nM PMA and 40 µM L-012. N.A.: no activity was observed, even at the highest concentration tested (50 µM).
Figure 2.
Effect of MHDO on fMLF-induced Ca2+ mobilization in human neutrophils. Panel A. Human neutrophils were preincubated for 30 min (at 25°C) with 15 µM MHDO or DMSO (vehicle control), followed by treatment with 5 nM fMLF or DMSO (vehicle control). Panel B. Effect of MHDO on IL-8-induced Ca2+ mobilization in human neutrophils. Human neutrophils were preincubated for 30 min (at 25°C) with 15 µM MHDO or DMSO (vehicle control), followed by treatment with 25 nM IL-8 or DMSO (vehicle control). Arrows indicate time of treatment additions. The data shown in both panels are representative of three independent experiments.
Consistent with its effect on Ca2+ mobilization, MHDO also inhibited neutrophil chemotaxis with an IC50 in the low micromolar range (Table 3). Among the other compounds tested, β-pinene, sabinene, β-citronellol, and elemicin were weak inhibitors of neutrophil chemotaxis (Table 3).
All 25 compounds were also evaluated for their effect on neutrophil ROS production. As with the other responses, only MHDO inhibited PMA-induced ROS production in human neutrophils (IC50 = 2.8 µM) (Table 3).
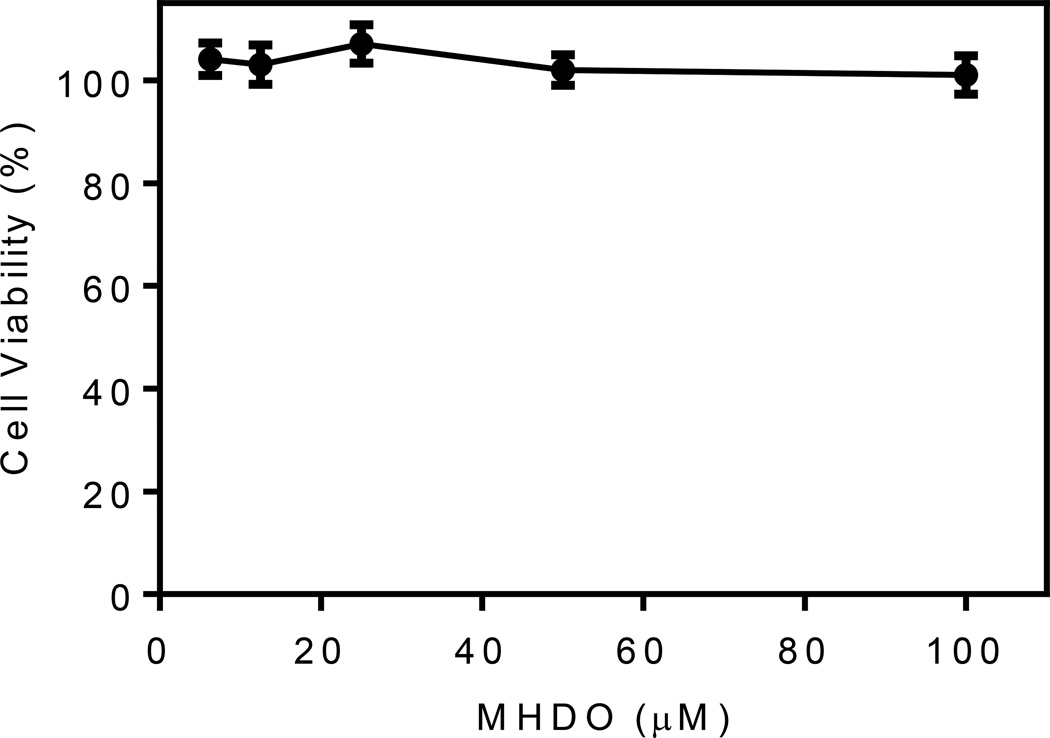
To ensure that the results on inhibition of Ca2+ flux and neutrophil chemotaxis were not influenced by possible compound toxicity, cytotoxicity of MHDO was evaluated at various concentrations (up to 100 µM) in MonoMac-6 cells during a 4-h incubation, which is relevant for relatively short times used to measure Ca2+ mobilization (up to 35 min) and chemotaxis (up to 90 min). As shown in Figure 3, MHDO did not affect cell viability, even at the highest tested concentration, thereby verifying that this compound, like the parent essential oil, was also not cytotoxic, at least during the 4 hr incubation period.
Figure 3.
Effect of MHDO on cell viability. MonoMac-6 cells were incubated for 4 hr with the indicated concentrations of MHDO, and cell viability was determined using a luminescent cell viability assay kit, as described. Values are the mean ± S.D. of triplicate samples from one experiment that is representative of two independent experiments. No statistically significant differences between untreated cells (DMSO alone) and cells treated with the compound were found.
Discussion
Essential oils are natural mixtures of terpenes, mainly monoterpenes and sesquiterpenes, which have a wide-range of pharmacological activities.11,32–34 Furthermore, essential oils from various plants, including Artemisia spp., have been reported to exhibit various biological effects in vitro and in vivo.2,35 In the present study, we defined the chemical profile of essential oils isolated from A. kotuchovii and evaluated their effects on phagocyte function.
Similar to the composition of essential oils from A. dracunculus,1 we found that estragole, (E) and (Z) β-ocimenes, and methyl eugenol were the primary constituents of A. kotuchovii essential oils. Other primary constituents present at lower concentrations (1–1.9% total weight) were limonene, spathulenol, β-pinene, myrcene, and methyl cinnamate. These compounds, with the exception of methyl cinnamate, are also present in tarragon essential oils.1,36
Although various essential oils have previously been found to inhibit NF-κB/AP-1 transcription and production of pro-inflammatory cytokines 5,30 and HNE activity,29,37 we did not find these activities in A. kotuchovii essential oils. Indeed, none of the major components of A. kotuchovii essential oils (>1% by total weight) have been reported previously to inhibit HNE, NF-κB/AP-1 activity, or TNF production. Among the minor compounds (~ 1%), only limonene has been reported to inhibit NF-κB activation in vivo during acute lung injury.38
Evaluation of biological activities of A. kotuchovii essential oils revealed that only AKEOstm inhibited Ca2+ flux and chemotaxis in human neutrophils. To date, there are no publications on modulation of Ca2+ mobilization in neutrophils by essential oils. Since intercellular Ca2+ flux is involved in chemotaxis, the inhibitory effect of AKEOstm on neutrophil chemotaxis is likely due to a primary effect on Ca2+ flux. Previously, other essential oils or their constituents have been reported to inhibit cell migration. For example, rosemary and geranium essential oils inhibited leukocyte recruitment and chemotaxis.39,40 In addition, Thymus vulgaris L. and Citrus latifolia (Yu.Tanaka) Tanaka essential oils also inhibited leukocyte migration.25,41 Although we found that AKEOstm also inhibited PMA-induced ROS production in human neutrophils, the potency of this effect was relatively low compared to essential oils from Sideritis italica (Miller) Greuter et Burdet.27
To further define the active component(s) in AKEOstm, we evaluated 25 of its constituents and found that β-citronellol, elemicin, β-pinene, and sabinene had low activity, whereas MHDO was a relatively potent inhibitor of neutrophil chemotaxis (Table 3), suggesting that it could be one of the primary essential oil components responsible for the inhibitory effects of AKEOstm on human neutrophils in vitro (Table 2). Indeed, only MHDO inhibited the other neutrophil responses tested (i.e., PMA-induced ROS production and fMLF- and IL-8-induced Ca2+ flux). Furthermore, MHDO was not present in AKEOf/l (Table 1). Thus, these data provide a molecular basis to explain at least part of inhibitory activity of AKEOstm on neutrophil chemotaxis. However, based on molecular weight of MHDO (124.18 Da), its relative percentage (~ 0.1%) in AKEOstm, and the IC50 values for biological activities of AKEOstm (~ 10–50 µg/ml) (see Tables 1 and 2), effective concentrations of this component would be in the range of 0.08–0.4 µM, which is several fold lower than the IC50 values for the biological activity profiles of MHDO (2.8 −8.2 µM, see Table 3). Thus, we could not exclude possible synergetic effects of other minor compounds, which are higher in AKEOstm (for example dehydro-1,8-cineole, trans-α-bergamotol, etc.) versus AKEOf/l. Since β-pinene and sabinene directly activated Ca2+ mobilization in neutrophils, albeit with low efficacy, it is possible that they could contribute to receptor desensitization and/or intracellular Ca2+ store depletion. Recently, Siani et al.42 reported that effective inhibition of eosinophil migration by essential oils from Syzygium cumini Skells and Psidium guajava L. correlated with the presence of β-pinene. Because α-pinene was completely inactive in all of our test-systems (Table 3), we suggest that human neutrophils can discriminate between α- and β- isomers of this bicyclic monoterpene. In support of this isomer preference, α-pinene inhibited inflammatory pathways in human chondrocytes, whereas β-pinene was inactive.43 Thus, various cell and/or molecular targets may determinate potency of the biological activities of these isomers.
Although previous reports indicate estragole and limonene can inhibit neutrophil and eosinophil migration,25,26,44 we did not observe inhibition of neutrophil function by R- and S- enantiomers of limonene. Likewise, we did not observe any effects of anethole on neutrophil function, although it was reported to inhibit paw edema in mice in acute and persistent inflammation models.45 These discrepancies may be explained by differences in methodologies and species specificity. For example, Kummer et al.26 used murine neutrophils, which respond differently than human neutrophils to fMLF and other agonists.
Neutrophils express a large number of receptors for the recognition of pathogen invasion and communication, including chemosensory, chemoattractant, and chemokine GPCR, Fc-receptors, various cytokine receptors, adhesion receptors, as well as innate immune receptors, such as Toll-like receptors.46,47 Here, we found that MHDO inhibited both fMLF- and IL-8-induced Ca2+ fluxes in neutrophils and the fMLF-induced response in FPR1-HL60 cells. Thus, MHDO may interfere with intracellular signaling pathways common for both FPR1 and CXCR1/2. Nevertheless, further studies will be necessary in the future to determine the precise molecular targets for MHDO in human neutrophils. It should be noted that MHDO is found in paprika and tomato oleoresins 48 and can be formed by thermal degradation of lycopene (ψ,ψ-carotene),49 a red pigment of some fruits and vegetables. Although there are many publications describing the anti-inflammatory and antioxidant effects of lycopene,50 our studies suggest that inhibition of leukocyte activation and/or migration could also contribute to the biological effects of paprika and tomato oleoresins, including lycopene metabolites and products of its thermal degradation.
In summary, we have defined the essential oil composition of A. kotuchovii and show that these essential oils and a primary component compound are able to inhibit human neutrophil responses, including Ca2+ mobilization, chemotaxis, and ROS production. Further studies are now needed to determine the molecular targets for MHDO, as well as the biological activity of other minor constituents of the oil and evaluation of therapeutic efficacy of AKEOstm and MHDO in animal models of acute or chronic inflammatory diseases.
Acknowledgements
We would like to thank Professor Andrei Khlebnikov, Tomsk Polytechnic University, Tomsk, Russia for advice on compound structure interpretation. This research was supported in part by National Institutes of Health IDeA Program COBRE grant GM110732; grants 0504/GF3 and 2117/GF4 from The Ministry of Education and Science, Kazakhstan; a USDA National Institute of Food and Agriculture Hatch project; and the Montana State University Agricultural Experiment Station.
Abbreviations
- AKEO
Essential oils of A. kotuchovii
- DMSO
dimethyl sulfoxide
- FBS
fetal bovine serum
- FID
flame ionization detector
- FPR1
N-formyl peptide receptor 1
- GC
gas chromatography
- GPCR
G-protein coupled receptors
- HBSS
Hanks' balanced-salt solution
- HNE
human neutrophil elastase
- IL
interleukin
- LPS
lipopolysaccharide
- MHDO
6-methyl-3,5-heptadien-2-one
- MS
mass spectrometry
- PMA
phorbol-12-myristate-13-acetate
- ROS
reactive oxygen species
- TNF
tumor necrosis factor
References
- 1.Obolskiy D, Pischel I, Feistel B, Glotov N, Heinrich M. Artemisia dracunculus L. (tarragon): a critical review of its traditional use, chemical composition, pharmacology, and safety. J. Agric. Food Chem. 2011;59:11367–11384. doi: 10.1021/jf202277w. [DOI] [PubMed] [Google Scholar]
- 2.Abad MJ, Bedoya LM, Apaza L, Bermejo P. The artemisia L. Genus: a review of bioactive essential oils. Molecules. 2012;17:2542–2566. doi: 10.3390/molecules17032542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mubashir K, Ganai BA, Ghazanfar K, Akbar S, Malik AH, Masood A. Evaluation of Artemisia amygdalina D. for anti-inflammatory and immunomodulatory potential. ISRN Inflammation. 2013;2013:483646. doi: 10.1155/2013/483646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moufid A, Eddouks M. Artemisia herba alba: a popular plant with potential medicinal properties. Pak. J. Biol. Sci. 2012;15:1152–1159. doi: 10.3923/pjbs.2012.1152.1159. [DOI] [PubMed] [Google Scholar]
- 5.Yoon WJ, Moon JY, Song G, Lee YK, Han MS, Lee JS, Ihm BS, Lee WJ, Lee NH, Hyun CG. Artemisia fukudo essential oil attenuates LPS-induced inflammation by suppressing NF-kappaB and MAPK activation in RAW 264.7 macrophages. Food Chem. Toxicol. 2010;48:1222–1229. doi: 10.1016/j.fct.2010.02.014. [DOI] [PubMed] [Google Scholar]
- 6.Cha JD, Moon SE, Kim HY, Lee JC, Lee KY. The Essential Oil Isolated from Artemisia capillaris Prevents LPS-Induced Production of NO and PGE(2) by Inhibiting MAPK-Mediated Pathways in RAW 264.7 Macrophages. Immunol. Invest. 2009;38:783–497. doi: 10.1080/08820130902936307. [DOI] [PubMed] [Google Scholar]
- 7.Li T, Chen H, Wei N, Mei X, Zhang S, Liu DL, Gao Y, Bai SF, Liu XG, Zhou YX. Anti-inflammatory and immunomodulatory mechanisms of artemisinin on contact hypersensitivity. Int. Immunopharmacol. 2012;12:144–150. doi: 10.1016/j.intimp.2011.11.004. [DOI] [PubMed] [Google Scholar]
- 8.Ho WE, Peh HY, Chan TK, Wong WS. Artemisinins: pharmacological actions beyond anti-malarial. Pharmacol. Ther. 2014;142:126–139. doi: 10.1016/j.pharmthera.2013.12.001. [DOI] [PubMed] [Google Scholar]
- 9.Kupriyanov AN. New species of genus Artemisia (Asteraceae) from Altay and Kazakhstan. Artemisia kotuchovii Kupr. Sp. nov. Botanical J. 1999;84:114–116. [Google Scholar]
- 10.Vandanmagsar B, Haynie KR, Wicks SE, Bermudez EM, Mendoza TM, Ribnicky D, Cefalu WT, Mynatt RL. Artemisia dracunculus L. extract ameliorates insulin sensitivity by attenuating inflammatory signalling in human skeletal muscle culture. Diabetes Obes. Metab. 2014;16:728–738. doi: 10.1111/dom.12274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Perez-Roses R, Risco E, Vila R, Penalver P, Canigueral S. Effect of some essential oils on phagocytosis and complement system activity. J. Agric. Food Chem. 2015;63:1496–1504. doi: 10.1021/jf504761m. [DOI] [PubMed] [Google Scholar]
- 12.Beyrau M, Bodkin JV, Nourshargh S. Neutrophil heterogeneity in health and disease: a revitalized avenue in inflammation and immunity. Open Biol. 2012;2:120134. doi: 10.1098/rsob.120134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bokoch GM. Chemoattractant signaling and leukocyte activation. Blood. 1995;86:1649–1660. [PubMed] [Google Scholar]
- 14.Maeda DY, Quinn MT, Schepetkin IA, Kirpotina LN, Zebala JA. Nicotinamide glycolates antagonize CXCR2 activity through an intracellular mechanism. J. Pharmacol. Exp. Ther. 2010;332:145–152. doi: 10.1124/jpet.109.159020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schepetkin IA, Kirpotina LN, Khlebnikov AI, Cheng N, Ye RD, Quinn MT. Antagonism of human formyl peptide receptor 1 (FPR1) by chromones and related isoflavones. Biochem. Pharmacol. 2014;92:627–641. doi: 10.1016/j.bcp.2014.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ryoo IJ, Yun BS, Lee IK, Kim YH, Lee IS, Ahn JS, Bae K, Yoo ID. Hydoroxyhibiscone A, a novel human neutrophil elastase inhibitor from Hibiscus syriacus . J. Microbiol. Biotechnol. 2010;20:1189–1191. doi: 10.4014/jmb.1004.04028. [DOI] [PubMed] [Google Scholar]
- 17.Kubeczka K-H. History and Sources of Essential Oil Research. In: Baser HCKBG, editor. Hanbook of Essential Oils: Science, Technology and Applications. Boca Raton, FL: CRC Press Taylor & Francis Group; 2010. pp. 3–30. [Google Scholar]
- 18.Mastelic J, Politeo O, Jerkovic I. Contribution to the analysis of the essential oil of Helichrysum italicum (Roth) G. Don. Determination of ester bonded acids and phenols. Molecules. 2008;13:795–803. doi: 10.3390/molecules13040795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ali A, Tabanca N, Özek G, Özek T, Aytac Z, Bernier UR, Agramonte NM, Baser KHC, Khan IA. Essential Oils of Echinophora lamondiana (Apiales: Umbelliferae): A Relationship Between Chemical Profile and Biting Deterrence and Larvicidal Activity Against Mosquitoes (Diptera: Culicidae) J. Med. Entomol. 2015;52:93–100. doi: 10.1093/jme/tju014. [DOI] [PubMed] [Google Scholar]
- 20.Özek G, Demirci F, Özek T, Tabanca N, Wedge DE, Khan SI, Baser KH, Duran A, Hamzaoglu E. Gas chromatographic-mass spectrometric analysis of volatiles obtained by four different techniques from Salvia rosifolia Sm., and evaluation for biological activity. J. Chromatogr. A. 2010;1217:741–748. doi: 10.1016/j.chroma.2009.11.086. [DOI] [PubMed] [Google Scholar]
- 21.Crocetti L, Schepetkin IA, Cilibrizzi A, Graziano A, Vergelli C, Giomi D, Khlebnikov AI, Quinn MT, Giovannoni MP. Optimization of N-benzoylindazole derivatives as inhibitors of human neutrophil elastase. J. Med. Chem. 2013;56:6259–6272. doi: 10.1021/jm400742j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schepetkin IA, Khlebnikov AI, Quinn MT. N-benzoylpyrazoles are novel small-molecule inhibitors of human neutrophil elastase. J. Med. Chem. 2007;50:4928–4938. doi: 10.1021/jm070600+. [DOI] [PubMed] [Google Scholar]
- 23.Schepetkin IA, Kirpotina LN, Khlebnikov AI, Quinn MT. High-throughput screening for small-molecule activators of neutrophils: Identification of novel N-formyl peptide receptor agonists. Mol. Pharmacol. 2007;71:1061–1074. doi: 10.1124/mol.106.033100. [DOI] [PubMed] [Google Scholar]
- 24.Chan AS, Pang H, Yip EC, Tam YK, Wong YH. Carvacrol and eugenol differentially stimulate intracellular Ca2+ mobilization and mitogen-activated protein kinases in Jurkat T-cells and monocytic THP-1 cells. Planta Med. 2005;71:634–639. doi: 10.1055/s-2005-871269. [DOI] [PubMed] [Google Scholar]
- 25.Hirota R, Roger NN, Nakamura H, Song HS, Sawamura M, Suganuma N. Anti-inflammatory effects of limonene from yuzu (Citrus junos Tanaka) essential oil on eosinophils. J. Food Sci. 2010;75:H87–H92. doi: 10.1111/j.1750-3841.2010.01541.x. [DOI] [PubMed] [Google Scholar]
- 26.Kummer R, Fachini-Queiroz FC, Estevao-Silva CF, Grespan R, Silva EL, Bersani-Amado CA, Cuman RK. Evaluation of anti-inflammatory activity of Citrus latifolia Tanaka essential oil and limonene in experimental mouse models. Evid. Based Complement. Alternat. Med. 2013;2013:859083. doi: 10.1155/2013/859083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Basile A, Senatore F, Gargano R, Sorbo S, Del Pezzo M, Lavitola A, Ritieni A, Bruno M, Spatuzzi D, Rigano D, Vuotto ML. Antibacterial and antioxidant activities in Sideritis italica (Miller) Greuter et Burdet essential oils. J. Ethnopharmacol. 2006;107:240–248. doi: 10.1016/j.jep.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 28.Cosentino M, Luini A, Bombelli R, Corasaniti MT, Bagetta G, Marino F. The essential oil of bergamot stimulates reactive oxygen species production in human polymorphonuclear leukocytes. Phytother. Res. 2014;28:1232–1239. doi: 10.1002/ptr.5121. [DOI] [PubMed] [Google Scholar]
- 29.Mori M, Ikeda N, Kato Y, Minamino M, Watabe K. Inhibition of elastase activity by essential oils in vitro. J. Cosmet. Dermatol. 2002;1:183–187. doi: 10.1111/j.1473-2165.2002.00059.x. [DOI] [PubMed] [Google Scholar]
- 30.Tabanca N, Ma G, Pasco DS, Bedir E, Kirimer N, Baser KH, Khan IA, Khan SI. Effect of essential oils and isolated compounds from Pimpinella species on NF-kappaB: a target for antiinflammatory therapy. Phytother. Res. 2007;21:741–745. doi: 10.1002/ptr.2154. [DOI] [PubMed] [Google Scholar]
- 31.Hua KF, Yang TJ, Chiu HW, Ho CL. Essential oil from leaves of Liquidambar formosana ameliorates inflammatory response in lipopolysaccharide-activated mouse macrophages. Nat. Prod. Commun. 2014;9:869–872. [PubMed] [Google Scholar]
- 32.de Cassia da Silveira ESR, Andrade LN, Dos Reis Barreto de Oliveira R, de Sousa DP. A review on anti-inflammatory activity of phenylpropanoids found in essential oils. Molecules. 2014;19:1459–1480. doi: 10.3390/molecules19021459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seow YX, Yeo CR, Chung HL, Yuk HG. Plant essential oils as active antimicrobial agents. Crit. Rev. Food Sci. Nutr. 2014;54:625–644. doi: 10.1080/10408398.2011.599504. [DOI] [PubMed] [Google Scholar]
- 34.Bhalla Y, Gupta VK, Jaitak V. Anticancer activity of essential oils: a review. J. Sci. Food Agric. 2013;93:3643–3653. doi: 10.1002/jsfa.6267. [DOI] [PubMed] [Google Scholar]
- 35.Takaki I, Bersani-Amado LE, Vendruscolo A, Sartoretto SM, Diniz SP, Bersani-Amado CA, Cuman RK. Anti-inflammatory and antinociceptive effects of Rosmarinus officinalis L. essential oil in experimental animal models. J. Med. Food. 2008;11:741–746. doi: 10.1089/jmf.2007.0524. [DOI] [PubMed] [Google Scholar]
- 36.Sayyah M, Nadjafnia L, Kamalinejad M. Anticonvulsant activity and chemical composition of Artemisia dracunculus L. essential oil. J. Ethnopharmacol. 2004;94:283–287. doi: 10.1016/j.jep.2004.05.021. [DOI] [PubMed] [Google Scholar]
- 37.Khan MS, Ahmad I. In vitro antifungal, anti-elastase and anti-keratinase activity of essential oils of Cinnamomum-, Syzygium- and Cymbopogon-species against Aspergillus fumigatus and Trichophyton rubrum . Phytomedicine. 2011;19:48–55. doi: 10.1016/j.phymed.2011.07.005. [DOI] [PubMed] [Google Scholar]
- 38.Chi G, Wei M, Xie X, Soromou LW, Liu F, Zhao S. Suppression of MAPK and NF-kappaB pathways by limonene contributes to attenuation of lipopolysaccharide-induced inflammatory responses in acute lung injury. Inflammation. 2013;36:501–511. doi: 10.1007/s10753-012-9571-1. [DOI] [PubMed] [Google Scholar]
- 39.Abe S, Maruyama N, Hayama K, Inouye S, Oshima H, Yamaguchi H. Suppression of neutrophil recruitment in mice by geranium essential oil. Mediators Inflamm. 2004;13:21–24. doi: 10.1080/09629350410001664798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nogueira de Melo GA, Grespan R, Fonseca JP, Farinha TO, Silva EL, Romero AL, Bersani-Amado CA, Cuman RK. Rosmarinus officinalis L. essential oil inhibits in vivo and in vitro leukocyte migration. J. Med. Food. 2011;14:944–946. doi: 10.1089/jmf.2010.0159. [DOI] [PubMed] [Google Scholar]
- 41.Fachini-Queiroz FC, Kummer R, Estevao-Silva CF, Carvalho MD, Cunha JM, Grespan R, Bersani-Amado CA, Cuman RK. Effects of thymol and carvacrol, constituents of Thymus vulgaris L. essential oil, on the inflammatory response. Evid. Based Complement. Alternat. Med. 2012;2012:657026. doi: 10.1155/2012/657026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Siani AC, Souza MC, Henriques MG, Ramos MF. Anti-inflammatory activity of essential oils from Syzygium cumini and Psidium guajava . Pharm. Biol. 2013;51:881–887. doi: 10.3109/13880209.2013.768675. [DOI] [PubMed] [Google Scholar]
- 43.Rufino AT, Ribeiro M, Judas F, Salgueiro L, Lopes MC, Cavaleiro C, Mendes AF. Anti-inflammatory and chondroprotective activity of (+)-alpha-pinene: structural and enantiomeric selectivity. J. Nat. Prod. 2014;77:264–269. doi: 10.1021/np400828x. [DOI] [PubMed] [Google Scholar]
- 44.Silva-Comar FMD, Wiirzler LAM, Silva SE, Kummer R, Pedroso RB, Spironello RA, Silva EL, Bersani-Amado CA, Cuman RKN. Effect of estragole on leukocyte behavior and phagocytic activity of macrophages. Evid. Based Complement. Alternat. Med. 2014 doi: 10.1155/2014/784689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ritter AM, Domiciano TP, Verri WA, Jr, Zarpelon AC, da Silva LG, Barbosa CP, Natali MR, Cuman RK, Bersani-Amado CA. Antihypernociceptive activity of anethole in experimental inflammatory pain. Inflammopharmacol. 2013;21:187–197. doi: 10.1007/s10787-012-0152-6. [DOI] [PubMed] [Google Scholar]
- 46.Futosi K, Fodor S, Mocsai A. Neutrophil cell surface receptors and their intracellular signal transduction pathways. Int. Immunopharmacol. 2013;17:638–650. doi: 10.1016/j.intimp.2013.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Malki A, Fiedler J, Fricke K, Ballweg I, Pfaffl MW, Krautwurst D. Class I odorant receptors, TAS1R and TAS2R taste receptors, are markers for subpopulations of circulating leukocytes. J. Leukoc. Biol. 2015;97:533–545. doi: 10.1189/jlb.2A0714-331RR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rios JJ, Fernandez-Garcia E, Minguez-Mosquera MI, Perez-Galvez A. Description of volatile compounds generated by the degradation of carotenoids in paprika, tomato and marigold oleoresins. Food Chem. 2008;106:1145–1153. [Google Scholar]
- 49.Kanasawud P, Crouzet JC. Mechanism of formation of volatile compounds by thermal-degradation of carotenoids in aqueous-medium .2. Lycopene degradation. J. Agric. Food Chem. 1990;38:1238–1242. [Google Scholar]
- 50.He Q, Zhou W, Xiong C, Tan G, Chen M. Lycopene attenuates inflammation and apoptosis in post-myocardial infarction remodeling by inhibiting the nuclear factor-kappaB signaling pathway. Mol. Med. Rep. 2015;11:374–378. doi: 10.3892/mmr.2014.2676. [DOI] [PubMed] [Google Scholar]