Abstract
Programmed cell death (PCD) pathways, including apoptosis and regulated necrosis, are required for normal cell turnover and tissue homeostasis. Mis-regulation of PCD is increasingly implicated in aging and aging-related disease. During aging the cell turnover rate declines for several highly-mitotic tissues. Aging-associated disruptions in systemic and inter-cell signaling combined with cell-autonomous damage and mitochondrial malfunction result in increased PCD in some cell types, and decreased PCD in other cell types. Increased PCD during aging is implicated in immune system decline, skeletal muscle wasting (sarcopenia), loss of cells in the heart, and neurodegenerative disease. In contrast, cancer cells and senescent cells are resistant to PCD, enabling them to increase in abundance during aging. PCD pathways limit life span in fungi, but whether PCD pathways normally limit adult metazoan life span is not yet clear. PCD is regulated by a balance of negative and positive factors, including the mitochondria, which are particularly subject to aging-associated malfunction.
Keywords: apoptosis, necrosis, aging, sarcopenia, mitochondria, senescence
1. Types of cell death
Cell death mechanisms have traditionally been divided into two types, programmed cell death (PCD) mechanisms that require energy, and necrotic cell death mechanisms that do not(Elmore, 2007). In addition, necrotic cell death typically causes a strong immune response, whereas PCD does not (Proskuryakov and Gabai, 2010; Taylor et al., 2008). However, more recently it has become clear that the PCD and necrosis mechanisms can sometimes overlap, and that necrosis is a more regulated process than was previously assumed(Galluzzi et al., 2012).
The canonical form of PCD is apoptosis(Elmore, 2007; Kerr et al., 1972), sometimes called type I PCD, in which the cell undergoes a characteristic series of molecular, biochemical and morphological changes. One of the earliest molecular markers of apoptosis is the externalization of cell membrane phosphatidylserine, which can be detected by staining with the reagent Annexin V(Hankins et al., 2015; Leventis and Grinstein, 2010). The exposed phosphatidylserine ultimately serves as a signal to nearby macrophages to engulf the dying cell. The first visible changes of apoptosis are the shrinkage of the cell, and a condensation of the nucleus and chromatin called pyknosis. This is followed by a fragmentation of the nucleus and chromatin called karyorrhexis, cell membrane blebbing, and finally the budding of the cell into a series of membrane-bound structures called apoptotic bodies. The small size of the apoptotic bodies facilitates their engulfment by nearby macrophages and other cells through phagocytosis, where the materials are further broken down in phagolysosomes for recycling. Because no cellular material is released into the interstitial space, and the engulfing cells do not release inflammatory cytokines, there is no immune reaction to apoptosis.
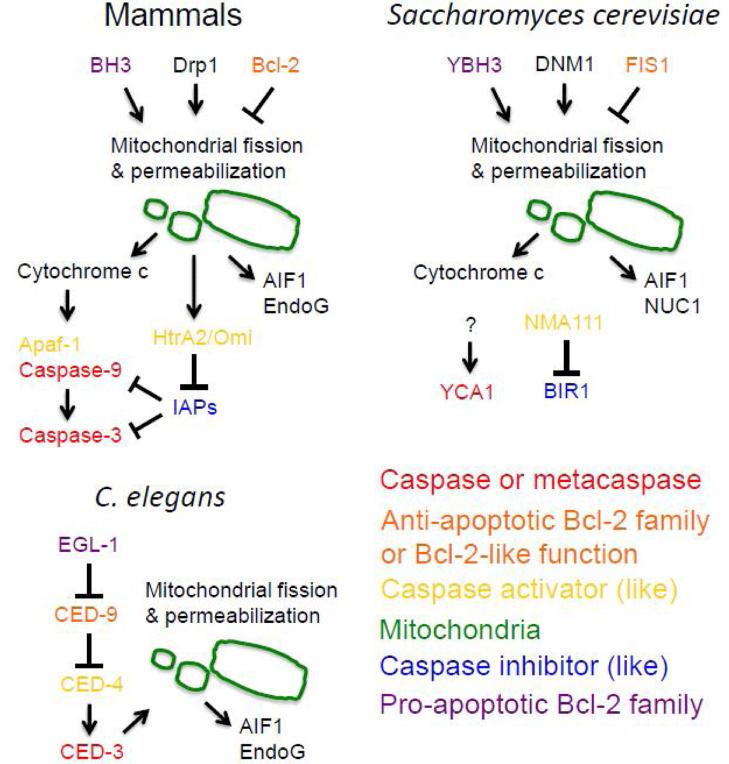
The molecular mechanism of apoptosis includes the sequential activation of a series of cysteine proteases called the caspases (Figure 1)(Shalini et al., 2015). The caspases are present in the cytoplasm of most cells, mostly in an inactive form (procaspase). Under appropriate conditions the caspases can activate themselves through autocatalytic cleavage, and can also cleave and activate other caspases, thereby creating a self-amplifying cascade. Initiator caspases (e.g., caspase-9) cleave and activate downstream effector caspases (e.g., caspase-3), which in turn cleave numerous cellular proteins including those of the cytoskeleton. The mitochondria play a central role in regulating the apoptotic pathway and caspase activation in mammalian cells. Cells contain a mix of pro-apoptotic and anti-apoptotic “Bcl-2 family” proteins and are poised for entry into the apoptotic pathway if and when the balance is sufficiently altered. The pro-apoptotic “Bcl-2 family” proteins include several of the “BH3-only” class (e.g., Bid, Bim, Puma)that promote permeabilization of the mitochondria by activating “Bcl-2 family” members Bad and Bax to form a pore structure in the mitochondrial membrane(Westphal et al., 2014). The anti-apoptotic “Bcl-2 family” proteins(e.g., Bcl-2, Bcl-x and Bcl-xL) can bind to the pro-apoptotic proteins to inhibit pore formation. In normal cells the mitochondria undergo dynamic changes in morphology regulated in part by the fission factor Drp1(Otera et al., 2013). During apoptosis Drp1 directs dramatic fission that is required formitochondrial membrane permeabilization (Frank et al., 2001). In addition, the cell cytoplasm also contains one or more IAPs (inhibitor of apoptosis proteins), which are a conserved family of caspase inhibitors. Recent studies in mice reveal that in vivo inhibition of caspase activity does not prevent cell death, but rather shifts the mechanism towards a more necrotic phenotype (Galluzzi et al., 2015).
Figure 1.
Cross-species comparison of PCD factors implicated in aging. The factors and regulatory relationships that appear similar between mammals, C. elegans and S. cerevisiae are emphasized; additional regulatory factors exist for each species but are not indicated for sake of clarity. For mammals the canonical intrinsic caspase-dependent apoptotic pathway is outlined. AIF and EndoG are caspase-independent and translocate to the nucleus where they mediate DNA fragmentation and chromatin condensation. Please see text for details.
Apoptosis is initiated in response to death signals that can be either intrinsic or extrinsic. The intrinsic apoptosis pathway is promoted by cellular stresses including DNA damage, activated oncogenes, hypoxia, oxidative stress and irradiation. These stimuli shift the balance of cytoplasmic activities to favor the pro-apoptotic factors by altering protein expression and/or stability, resulting in mitochondrial membrane permeabilization and the release of mitochondrial pro-apoptotic factors into the cytoplasm (Figure 1). These mitochondrial factors include cytochrome c, Smac/DIABLO and the HtrA2/Omi serine protease. The cytochrome c binds and activates the cytoplasmic proteins Apaf-1 and procaspase-9. Together these proteins form a structure called the apoptosome, which in turn leads to cleavage of procaspase-9 to form the active caspase-9 protease. Smac/DIABLO and HtrA2/Omi inactivate specific IAPs, thereby further tipping the balance towards apoptosis. Subsequently another group of proteins is released from the mitochondria, including AIF, endonuclease G (EndoG) and CAD, which act in the nucleus to promote DNA fragmentation and nuclear condensation. As discussed below, AIF and EndoG also participate in several forms of caspase-independent PCD. In many cell types the intrinsic pathway can be activated by the withdrawal of specific hormones that normally act to suppress the apoptotic pathway(Raff et al., 1994).
The extrinsic pathway for activation of apoptosis involves signaling through transmembrane receptors of the tumor necrosis factor (TNF) receptor family. These receptors share an intracellular protein domain involved in signaling called the “death domain”, and are sometime called “death receptors”. For example, in cells that express TNFR1 (TNF receptor 1), the hormone TNF-alpha activates the receptor and downstream signaling resulting in activation of the initiator caspase-8 and subsequent apoptosis.
In certain cell types the initiation of PCD is associated with up-regulation of cell cycle markers, leading to the suggestion that PCD in these cells may involve a partial entry into the cell cycle. For example, serum withdrawal from quiescent cultures of mouse 3T3 cells caused apoptosis associated with up-regulation of G1 phase proteins c-myc, c-jun, c-fos and cdc-2, as well as BrdU incorporation and PCNA expression, indicative of an abortive G1 traverse(Pandey and Wang, 1995). Treatment of transformed cultured lymphoid cells with glucocorticoids causes G1 arrest and apoptosis, implicating G1 regulators in both processes(King and Cidlowski, 1995, 1998). These early studies were with cultured cells, however as mentioned below, maladaptive cell-cycle entry is also implicated in neuronal PCD in vivo.
The necrosis pathway for cell death is characterized by cell swelling and disruptions of the cell membrane(Proskuryakov and Gabai, 2010). The release of cytoplasm into the interstitial space typically results in inflammation. Necrosis was traditionally thought to be an energy-independent toxic process where the cell is passively destroyed. For example, in many cell types moderate levels of heat or radiation stress will induce apoptosis, whereas extreme heat or radiation will cause necrosis. However, recent studies suggest that except for under the most extreme stress conditions, necrosis is also a regulated process that is activated by specific pathological and physiological stimuli, and is sometimes referred to as type III PCD, programmed necrosis, regulated necrosis, or “necroptosis”(Degterev et al., 2014). Programmed necrosis is caspase-independent and can be further subdivided depending on the involvement of specific factors. For example, in certain cell types stresses including ischemia, hypoxia, and signaling through TNF-receptor family members can induce a necroptosis pathway involving the RIP1 kinase, the JNK signaling pathway and the mitochondria, ultimately leading to cell membrane disruption (Galluzzi et al., 2009; Pasparakis and Vandenabeele, 2015). Several forms of programmed necrosis involve cleavage of mitochondrial AIF by cysteine proteases such as calpain and/or cathepsin, followed by release of the AIF from permeabilized mitochondria. The AIF then undergoes translocation to the nucleus, and this translocation is positively regulated by cyclophilin A and negatively regulated by Hsp70 (Delavallee et al., 2011; Gurbuxani et al., 2003; Zhu et al., 2007). In the nucleus the AIF interacts with translocated mitochondrial EndoG to mediate nuclear DNA fragmentation. Ongoing studies of cell death reveal that there are multiple types of apoptosis and programmed necrosis, and that these mechanisms can overlap depending on the cell type and the nature of the stress.
Another type of programmed cell death is autophagic cell death, sometimes called type II PCD(Galluzzi et al., 2012; Kroemer et al., 2009). Macroautophagy is a cellular “self-eating” process in which bulk cytoplasm and organelles become sequestered inside double-membrane vesicles called autophagosomes. The autophagosomes in turn fuse with lysosomes to promote degradation of the cellular materials. Autophagic cell death is characterized by the absence of chromatin condensation and by extensive autophagic vacuolization of the cytoplasm. Notably, macroautophagy is typically a protective and anti-apoptotic mechanism, and therefore autophagic cell death may involve hyper-activation of a normally beneficial process.
Mechanisms of PCD can be further divided into sub-types based on a variety of criteria, including caspase-dependence and the degree of overlap with necrosis (Galluzzi et al., 2012). For example, “aponecrosis” shares features of both apoptosis and necrosis, and may represent a midpoint in a continuum between the two forms of cell death (Papucci et al., 2004; Yakovlev and Faden, 2004). “Paraptosis” is caspase-independent and characterized by cytoplasmic vacuolization and the requirement for gene transcription and translation. Paraptosis can be induced by IGF-I signaling and other factors in cultured mammalian cells, and may also occur in vivo (Sperandio et al., 2004). “Anoikis” refers to apoptotic PCD that is induced upon detachment of the cell from the extracellular matrix (Paoli et al., 2013). Anoikis can proceed through either the intrinsic or extrinsic apoptosis pathway depending on the cell type, and importantly, resistance to anoikis is a hallmark of metastatic cancers (Taddei et al., 2012; Zhong and Rescorla, 2012). The fact that most (and perhaps all) normal cells are dependent on circulating factors and/or ECM contacts to prevent PCD may be one mechanism that promotes multi-cellularity. Finally, “Cornification” is a specialized form of PCD that occurs in the keratinocytes in the epidermis and that involves caspase-14(Eckhart et al., 2013; Galluzzi et al., 2012). In cornification the dead cell material is maintained to form the outermost layer of the skin, and is ultimately shed through desquamation.
The details of the apoptotic pathway differ between mammals and the model organisms Drosophila and C. elegans, however the major players are largely conserved (Figure 1). In both Drosophila and C. elegans the Bcl-2 family members regulate apoptosome formation and caspase activation (Denton et al., 2013; Estaquier et al., 2012). In C. elegans the apoptotic pathway was first characterized by its role in the programmed elimination of specific somatic cells during development (Metzstein et al., 1998). The pro-apoptotic Bcl-2-related protein EGL-1 inhibits the activity of the anti-apoptotic Bcl-2-related factor CED-9 (Figure 1). CED-9 inhibits the activity of the Apaf-1-related factor CED-4, which in turn activates the caspase CED-3, which is related to mammalian caspase 3. EGL-1 and CED-3 are implicated in promoting mitochondrial fission and causing release of pro-apoptotic factors AIF and EndoG from the mitochondria (Parrish et al., 2001; Wang et al., 2002). Since then additional types of C. elegans developmental cell deaths have been identified (Kinet and Shaham, 2014); for example, cell deaths in the female germ line and during male tale development are EGL-1 independent(Gumienny et al., 1999; Maurer et al., 2007).
2. PCD in normal tissue development, adult homeostasisand aging
PCD plays important roles during human development, including the destruction of cells to create the spaces between the digits of the hands and feet (Chimal-Monroy et al., 2011; Hernandez-Martinez and Covarrubias, 2011), and the structures of the brain (Burek and Oppenheim, 1996; Oppenheim, 1991). In the adult, PCD is required for the normal turnover of cells in numerous tissues that are maintained by cell division, including the intestinal epithelium, the blood, the epidermis, kidney and lungs(Elmore, 2007; Gunther et al., 2013; Kile, 2014). It has been estimated that about 10 billion cells are created in the adult each day to replace those turned over by PCD(Renehan et al., 2001). Aging appears to be associated with mis-regulation of PCD, in that PCD appears to be more active than is optimal in certain cell types, and less active than is optimal in other cell types (Gupta, 2005; Higami and Shimokawa, 2000; Lu et al., 2012; Salminen et al., 2011; Shen and Tower, 2009; Warner, 1997).
Aging is associated with decreased PCD in several cell types. For example, in humans, aging caused decreased expression of apoptosis genes and increased expression of senescence-related genes in adipose mesenchymal stem cells (Alt et al., 2012). Similarly, in mouse, bone marrow mesenchymal stem cells had decreased expression of both cell cycle and PCD genes during aging(Wilson et al., 2010). A decrease in PCD response with age was observed in the liver of rats challenged with the DNA-damaging agent methyl methanesulfonate (MMS) (Suh et al., 2002), and this reduced PCD was associated with increased phosphorylation of ERKs (extracellular signal-regulated protein kinases) in the old animal tissues (Suh, 2002). Radiation challenge will induce apoptosis in isolated peripheral blood lymphocytes, and this response is significantly reduced in cells isolated from aged mice (Polyak et al., 1997), as well as cells isolated from older human patients (Camplejohn et al., 2003). Consistent with these observations, human serum shows a reduction in markers of apoptosis during normal aging (Kavathia et al., 2009).
In humans vascular injury can lead to neointima (scar) formation. This response is greater in older patients due in part to the excessive proliferation and reduced apoptosis of vascular smooth muscle cells (VSMCs), and the same result is observed in aging mice (Vazquez-Padron et al., 2004). Interestingly, the reduced apoptosis in the VSMCs during aging may involve reduced signaling from adjacent endothelial cells (Qian et al., 2011).
Correctly regulated PCD and phagocytosis of the resulting cell debris is required to prevent an inappropriate immune response (Devitt and Marshall, 2011). Aging-associated disruptions of PCD regulation and phagocytosis may contribute to aging-associated inflammation, including activation of the NFκB signaling pathway (Gupta et al., 2006; Salvioli et al., 2013), (Shaw et al., 2010). Because NFκB signaling can be either pro-apoptotic or anti-apoptotic depending on the cellular context, this could couple inflammation to further alterations in PCD in certain tissues.
Some cell types are characterized by increased PCD during aging. Apoptosis plays a critical role in the development of the immune system, as well as in adult immune homeostasis. Apoptosis functions in the selection of T-cells in the thymus, and the deletion of self-reactive T and B cells. In addition apoptosis is required to regulate immunological memory and to reduce the number of effector T cells after an immune response. Finally, natural killer cells and cytotoxic T lymphocytes kill target cells by inducing apoptosis, for example, virus-infected target cells. During aging there is a significant loss of cells in the thymus (thymus involution) and bone marrow, associated with an age-related increase in the number of apoptotic lymphocytes (Sainz et al., 2003). Increased apoptosis is implicated in the age-related reduction of T-cell subtypes in the human immune system (T-cell lymphopenia), including both CD4+ and CD8+ T-cells (Gupta, 2005). In contrast, in vitro studies report that certain T-cell subtypes (CD8+ CD28−) resulting from repeated immune stimulation exhibit senescence-like changes that include reduced response to stress and resistance to apoptosis(Chou and Effros, 2013). Aging in the mammalian heart is characterized by a loss of myocytes and hypertrophy of the remaining myocytes (Olivetti et al., 1991). Both apoptotic and necrotic mechanisms are implicated in myocyte cell loss during normal heart aging as well as in heart failure (Kajstura et al., 1996; Kung et al., 2011).
Apoptosis is critical in wound healing, and wound healing is generally impaired during aging, often associated with local inflammation (Hiebert and Granville, 2012; Rai et al., 2005). For example, mis-regulated apoptosis has been implicated in the age-associated increase in fibrotic lung disease in two ways: increased sensitivity to apoptosis in alveolar epithelial cells may lead to greater injury, whereas reduced sensitivity to apoptosis in fibroblasts and myofibroblasts may promote fibrosis (Kapetanaki et al., 2013).
In summary, aging-associated disruptions in systemic and inter-cell signaling combined with cell-autonomous effects result in increased susceptibility to PCD in some cell types, and decreased PCD in other cell types. One possible interpretation of these results is that the general decrease in PCD markers during aging may result in part from a slowing of the normal homeostatic cell turnover rate for several adult tissues. For example, dramatically decreased cell turnover rate with age is observed for the skin (Rinnerthaler et al., 2014), and reduced cell turnover rates with age are also reported for the heart (Bergmann et al., 2009) breast epithelium (Misell et al., 2005) and pituitary (Nolan et al., 1999; Richardson et al., 2014). At the same time, alterations in systemic signaling, including inflammation and ECM disruptions, may combine with cell-autonomous changes such as increased oxidative stress and genomic damage to make certain cell types more sensitive to PCD, particularly when challenged by acute or chronic stress.
3. Suppression of PCD in cancerous and senescent cells
PCD is an important anti-cancer mechanism and the p53 pathway is a critical mediator of this response (Vazquez et al., 2008). p53 is activated in response to cellular stress including DNA damage and increased ROS levels. Functioning as a transcription factor p53 positively regulates expression of pro-apoptotic factors, including the pro-apoptotic Bcl-2 family member Puma, thereby shifting the balance to favor apoptosis. In addition, p53 protein can physically translocate to the mitochondria where it promotes membrane permeabilization and the release of the mitochondrial pro-apoptotic factors(Green and Kroemer, 2009). The importance of p53-induced apoptosis as an anti-cancer mechanism is supported by the fact that p53 is mutated in the majority of human cancers. Most of the mutations are located in the p53 protein DNA binding domain, suggesting that p53 transcriptional activity is particularly important for tumor suppression. Moreover, in cancers where p53 is intact, there is typically a mutation in some other component of the p53 pathway. For example, many cancers have mutations that cause increased expression of the p53 negative regulator MDM2 (Vazquez et al., 2008). These results suggest that inactivation or suppression of the p53 pathway is one requirement for the cancer phenotype. Other cancers are found to have dramatically up-regulated expression of one or more heat shock proteins (hsps) that can bind to and inactivate pro-apoptotic factors including Apaf-1, cytrochrome c and AIF (Jego et al., 2013; Rerole et al., 2011). A reduced sensitivity to apoptosis is observed in cells isolated from patients with the premature aging-like syndrome called Werner's disease (Spillare et al., 1999), and this may be related to the increased cancer incidence observed in those patients. Most cancer chemotherapies, including ionizing radiation, are thought to act by hyper-stimulating the otherwise suppressed PCD pathways, including activating any remaining p53 (Stegh, 2012; Zhao et al., 2012). Consistent with this idea, the efficiency of induction of apoptosis by chemotherapy has been shown to correlate with clinical outcomes for several cancers (Bosserman et al., 2012). However, it should be noted that other studies suggest that, at least for certain cell types, the tumor-suppressive function of p53 is independent of transcriptional induction of apoptosis, and instead may involve p53 regulation of DNA repair and/or metabolism(Valente et al., 2013). Finally a promising therapy for cancer is treatment with TRAIL (tumor necrosis factor-related apoptosis-inducing ligand). TRAIL is a naturally-occurring cytokine that binds to the DR4 and DR5 death receptors to activate the extrinsic apoptosis pathway, and it does this preferentially in cancer cells (Refaat et al., 2014).
Cellular senescence is also an important anti-cancer mechanism(Childs et al., 2014). Cellular senescence is an irreversible cell cycle arrest that can result from repeated cell divisions and telomere erosion, as well as from activated oncogenes, disrupted chromatin structure and oxidative stress. Activation of p53 can cause cells to undergo either apoptosis or cellular senescence depending on the type of cell and the nature of the stress (Zuckerman et al., 2009). Senescent cells are resistant to apoptosis (Wang, 1995) and this may be one reason that senescent cells are observed to accumulate during aging in several tissues(Kirkland and Tchkonia, 2014). Senescent cells exhibit a “senescence-associated secretory phenotype” (SASP) involving the release of pro-inflammatory cytokines and extracellular matrix degrading enzymes (Tchkonia et al., 2013). Because of this the accumulation of senescent cells during aging is implicated as a causal factor in aging-associated inflammation and disruptions of ECM. The resistance to apoptosis has been examined in senescent human fibroblasts in vitro. These cells fail to reduce Bcl-2 levels upon apoptotic stimuli including serum starvation (Wang, 1995, 1997)and oxidative stress (Ryu et al., 2007; Yeo et al., 2000), and have reduced caspase-3 expression(Marcotte et al., 2004) and increased expression of the anti-apoptotic factor Bcl-xL (Rochette and Brash, 2008). The resistance of senescent cells to apoptosis might also involve the down-regulation of cell cycle factors that participate in both cell division and in cell death (King and Cidlowski, 1995). Recently both genetic and chemical interventions have been described that can preferentially kill senescent cells in vivo in mice, and these interventions had health benefits for mouse models with elevated levels of senescent cells (Zhu et al., 2015). Despite the potential negative effects of increasing the abundance of senescent cells, cellular senescence has emerged as an important therapeutic target in cancer(Lee and Lee, 2014). Cancer chemotherapy and ionizing radiation can cause either apoptosis or cellular senescence depending on the cell type, and both outcomes have been shown to positively correlate with clinical outcomes.
4. PCD in neurodegenerative disease
The number of neurons does not decrease on a global scale during normal aging in humans or other mammals, indicating a general absence of PCD during normal brain aging (Andersen et al., 2003; Giannaris and Rosene, 2012; Long et al., 1999; Merrill et al., 2001; Rapp and Gallagher, 1996). However, PCD is increasingly implicated in several aging-associated neurodegenerative diseases(Ghavami et al., 2014). Nervous tissue has several properties that may contribute to a susceptibility to maladaptive PCD. For example, in the normal adult brain, activated caspase-3 functions in the remodeling of neuronal processes important for synaptic plasticity and certain forms of memory (Snigdha et al., 2012). The presence of activated caspase-3 may create a risk for inappropriate PCD activation in neurons challenged by stress. Moreover, neural tissue is rich in iron, and iron levels have been observed to further increase with age and in neurodegenerative disease(Oshiro et al., 2011). Iron can promote Fenton chemistry and oxidative stress, thereby favoring PCD. Neurons appear to suppress PCD in part by using glucose metabolism to generate reducing equivalents that prevent oxidation and activation of cytochrome c (Vaughn and Deshmukh, 2008). This may make neurons particularly susceptible to PCD upon metabolic disruption.
Each of the aging-associated neurodegenerative diseases Alzheimer's disease (AD), Parkinson's disease (PD) and Huntington's disease (PD) involves the accumulation of abnormal proteins and aggregates. Protein aggregates are normally cleared by the macroautophagy pathway (autophagy), and this pathway also functions in the normal turnover of mitochondria (mitophagy). Abnormal mitochondrial function and oxidative stress are implicated in the etiology of each disease. The data suggest that a disruption of normal autophagy/mitophagy may contribute to mitochondrial malfunction and thereby promote PCD (Ghavami et al., 2014; Hroudova et al., 2014; Nixon and Yang, 2012).
It is not yet clear what are the precise mechanism(s) for cell death in the aging-associated neurodegenerative diseases. Increased or altered p53 activity is found to correlate with AD (Lanni et al., 2012), and animal model studies implicate a PARP-dependent programmed-necrosis pathway in PD (Lee et al., 2014). Cell cycle re-entry may be particularly important (Folch et al., 2012). Brain tissue from neurodegenerative disease patients shows extensive up-regulation of cell cycle proteins including CDKs. Studies of cultured neuronal cells indicate that oxidative stress and DNA damage can induce an abortive cell cycle entry and consequent apoptosis, suggesting that this might also occur in vivo. Taken together, these studies suggest that only a sub-set of neurons are at risk for loss by PCD-like events during aging.
5. PCD-like events in tissues of old animals
During aging several tissues exhibit cell loss that is attributed to PCD or to PCD-like processes. Mammals exhibit an aging-associated skeletal muscle atrophy called sarcopenia that involves both reductions in muscle fiber size and loss of fibers(Marzetti et al., 2008). The precise mechanisms for sarcopenia are not yet known, however studies in rodents implicate mitochondrial malfunction and abnormal PCD signaling (Marzetti et al., 2013). The expression of PCD markers correlates with aging and sarcopenia in specific rodent muscle tissues. For example, aging rat gastrocnemius muscle showed increased levels of Bax, decreased Bcl-2, activation of caspase-3 and DNA fragmentation (Song et al., 2006). In contrast, aging rat soleus muscle showed translocation of Endonuclease G from the mitochondria to the nucleus suggesting a caspase-independent process (Leeuwenburgh et al., 2005). Mitochondrial mutations are implicated in sarcopenia because mtDNA deletions co-localized with mitochondrial malfunction in skeletal muscle fibers of aged rats (Wanagat et al., 2001), and PCD-like events were elevated in the skeletal muscle of aging mice engineered to have greater mitochondrial mutation load (Kujoth et al., 2005). Age-related alterations in systemic signaling are also implicated in muscle aging, based on the results of several striking interventions. For example, fusion of young and old mouse circulatory systems promoted the proliferative and regenerative capacity of skeletal muscle in the old mouse (Conboy et al., 2005). Old mice exhibit a decrease in circulating levels of the growth hormone GDF-11, and restoring GDF-11 levels in the old mouse was sufficient for much of the benefit for muscle function (Sinha et al., 2014). Loss of myocytes is observed in the aging heart, and studies in rodent models implicate a PCD mechanism involving increased Bax/Bcl-2 ratio, cytochrome c release from mitochondria and Caspase-3 activation (Kwak, 2013). In mice, over-expression of the mitochondrial antioxidant enzyme MnSOD reduced PCD in the aging heart, thereby implicating mitochondrial function and oxidative stress (Kwak et al., 2014). In addition, mutation of mouse cathepsin K reduced AIG translocation and PCD during aging, resulting in improved heart function and implicating both caspase-dependent and caspase-independent mechanisms(Hua et al., 2015). Finally, increasing GDF-11 levels in the aging mouse had a benefit for heart function, indicating a role for altered systemic signaling in heart aging.
Several additional tissues show increased sensitivity to induction of PCD during human aging. For example, the aging kidney has increased sensitivity to stress and PCD, and rodent models show decreased levels of Bcl-2, and increases in Bax, cytochrome c and caspase-3 (Wang et al., 2014). Age-related hearing loss involves loss of sensory hair cells via PCD, and mitochondrial genome mutations are implicated in this process(Fujimoto and Yamasoba, 2014). Aging-associated hair greying involves loss of melanocyte stem cells by PCD, and decreased Bcl-2 and increased ROS are implicated in this process (Seiberg, 2013). Finally, increased sensitivity to stress and PCD is observed in aging gastric mucosa, and rodent studies implicate caspase-3 activation (Tarnawski et al., 2014).
In the model organism Drosophila, several studies indicate altered PCD regulation during aging. Increased PCD-like events were observed during aging in muscle tissue and in fat tissue, as indicated by DNA fragmentation and caspase activity (Zheng et al., 2005). In contrast there was no evidence for a global increase in PCD in Drosophila nervous tissue using these assays. However, a transgenic reporter for caspase-3 activity was activated during aging in a subset of nervous tissue cell types, including the ellipsoid body that functions in visual memory, and the antennal lobe and a sub-set of olfactory receptor neurons involved in olfaction(Chihara et al., 2014). The olfactory receptor neurons also had increased PARP levels and DNA fragmentation, and were sometimes lost through PCD. These studies suggest that only a specific sub-set of neurons are at risk for PCD during aging in Drosophila.
In the model organism C. elegans the muscles undergo an aging-related ultrastructural deterioration (also referred to as sarcopenia), whereas nervous system ultrastructure remains largely intact(Herndon et al., 2002). The possible role of PCD in the C. elegans sarcopenia is not known, however a loss of normal signaling from motorneurons has been implicated (Liu et al., 2013). During aging the C. elegans gonad undergoes hypertrophy marked by increased cell DNA content and increased cell number. This phenotype is more severe in cep-1 (p53-related gene) mutants, suggesting an aging-associated loss of normal germ-line PCD activity (Andux and Ellis, 2008; McGee et al., 2012). Taken together the studies with the model systems support the conclusion that aging can cause increased susceptibility to apoptosis in some cell types, and decreased apoptosis in other cell types.
6. PCD in life span regulation
Because PCD is required for normal tissue homeostasis in the adult, it is expected to be important for optimal life span in species such as mammals where multiple tissues are maintained by constant cell turnover. In contrast in C. elegans all of the adult somatic cells are post-mitotic. C. elegans contains a single caspase gene ced-3 that is required for developmentally regulated PCD (Reddien and Horvitz, 2004). Mutation of ced-3 was reported not to affect adult life span, suggesting that C. elegans life span is not regulated by a canonical caspase-dependent PCD(Garigan et al., 2002), however a possible role for caspase-independent PCD or necrosis pathways in regulating C. elegans life span cannot yet be ruled out. Intriguingly, the canonical caspase-dependent C. elegans PCD pathway was recently found to be required for increased life span caused by ETC mutants and retrograde ROS signaling (Yee et al., 2014), in the absence of overt PCD. The data suggest a potentially ancient function for the pathway in mediating retrograde mitochondrial ROS signaling and altered nuclear gene expression independent of its function in regulating PCD.
In Drosophila, both the gut (Jiang et al., 2011; Micchelli and Perrimon, 2006; Ohlstein and Spradling, 2006) and Malphigian tubules (equivalent to kidney nephrons) (Singh et al., 2007)are maintained by stem cell division and cell turnover, however it has not yet been determined if the rate of cell turnover is important for normal longevity. Over-expression of caspase inhibitors p35 and DIAP1 in adult flies did not affect life span, suggesting that adult fly life span is not limited by a canonical caspase-dependent PCD(Shen et al., 2009), however a possible role for caspase-independent PCD or necrosis pathways in regulating adult fly life span cannot yet be ruled out.
In mammals PCD is required for normal development and adult tissue homeostasis, which complicates investigation of possible roles for PCD in mammalian life span regulation. Drugs that inhibit PCD and necrosis show promise as potential interventions in injury and disease (Degterev et al., 2014), however as yet there are no reports of benefits for normal longevity.
7. PCD in fungal aging
Aging has been studied in Saccharomyces cerevisiae using two different approaches, the replicative life span assay and the chronological life span assay (Longo et al., 2012), and with both methods the cells appear to die through a type of PCD. The replicative aging assay counts the number of times the mother cell can produce a bud (daughter cell). Approaching 25 divisions the mother cell becomes larger, buds less frequently and ultimately dies by lysis(Egilmez and Jazwinski, 1989). The pathways reported to regulate life span in this assay include the regulation of rDNA stability and damaged protein retention in the mother cell by Sir2 (Delaney et al., 2011). In addition, the Ras/PKA and TOR/Sch9 signaling pathways were found to promote translation and growth and suppress longevity in response to nutrients (Powers et al., 2006). Mitochondrial stress and retrograde signaling can also affect the Ras/PKA pathway and increase life span, depending on the yeast genotype (Jazwinski, 2014; Kirchman et al., 1999). Aged mother cells showed characteristics of PCD including increased markers of ROS, nuclear DNA fragmentation as indicated by TUNEL labeling, and phosphatidylserine externalization as indicated by Annexin V staining (Laun et al., 2001).
The other approach for studying aging in Saccharomyces cerevisiae is the chronological aging assay. Here the yeast culture is grown until glucose is depleted, which causes the cells to withdraw from the cell cycle (Fabrizio and Longo, 2003). The cells remain metabolically active and the number of viable cells in the sample can be measured at various time points by plating the cells on a more rich media and counting the number of colonies. As a function of time the number of viable cells in the sample decreases as cells are lost to lysis. The pathways thought to regulate life span in the chronological aging assay include Tor/Sch9 and Ras/PKA (Wei et al., 2008). Cell death in the Saccharomyces cerevisiae chronological aging assay has also been described as a PCD based on both morphological and genetic criteria (Fabrizio et al., 2004; Herker et al., 2004). The morphological criteria included chromatin condensation and fragmentation, increased markers of ROS, DNA fragmentation indicated by TUNEL, and phosphatidylserine externalization indicated by Annexin V staining (Carmona-Gutierrez et al., 2010a).
Several genes have been identified that function in yeast PCD similarly to PCD regulators in mammals (Figure 1)(Carmona-Gutierrez et al., 2010a). A protease related to caspases called a metacaspase plays a role in yeast PCD(Hill and Nystrom, 2015). Deletion of the Saccharomyces cerevisiae metacaspase YCA1 increased resistance to PCD caused by stressors such as hydrogen peroxide and also increased chronological life span (Herker et al., 2004; Madeo et al., 2002). An AIF-like gene called AIF1 was identified and deletion of AIF1 was reported to delay chronological aging (Wissing et al., 2004). A yeast homolog of EndoG was identified (NUC1) that has opposing roles in cell death in the chronological assay: on glucose media deletion of NUC1 increased cell death, whereas on glycerol media deletion of NUC1 increased life span and increased resistance to hydrogen peroxide induced PCD (Buttner et al., 2007). Notably the PCD mediated by NUC1 did not require the metacaspase YCA1. An IAP-like protein called BIR1 was identified, and over-expression of BIR1 could both reduce PCD and extend chronological life span (Walter et al., 2006). BIR1 is in turn regulated by a yeast homolog of the mammalian serine protease HtrA2/Omi, called NMA111(Figure 1)(Fahrenkrog, 2011; Fahrenkrog et al., 2004); however, unlike mammalian HtrA2/Omi, which is released from the mitochondria, the yeast NMA111 functions in the nucleus. So far no interaction between BIR1 and YCA1 has been detected (Walter et al., 2006). The specific molecular activator(s) of YCA1 during PCD remain to be identified (Carmona-Gutierrez et al., 2010a; Carmona-Gutierrez et al., 2010b; Hill and Nystrom, 2015), however Cytochrome c has been implicated(Silva et al., 2005).
Mitochondrial fission appears to play a role in yeast PDC similar to the situation in mammals. The gene DNM1 promoted mitochondrial fission and PCD in response to hydrogen peroxide, whereas the conserved mitochondrial morphology regulator FIS1 inhibited fission and YCA1-dependent cell death (Fannjiang et al., 2004). Notably deletion of DNM1 also increased replicative life span (Scheckhuber et al., 2007). Interestingly, human Bcl-2 could substitute for FIS1, consistent with a Bcl-2-like function for FIS1. Finally, a BH3-only protein called YBH3 was identified (Buttner et al., 2011). Upon toxic stress YBH3 associated with the mitochondria and disrupted mitochondrial membrane potential. Over-expression of YBH3 sensitized cells to PCD caused by hydrogen peroxide, however this death was not dependent upon YCA1, AIF1, NMA111 or NUC1. Deletion of YBH3 was reported to increase both replicative and chronological life span (Buttner et al., 2011). The studies with yeast therefore indicate multiple mechanisms for PCD involving factors released from the mitochondria, similar to the situation in mammals.
Sexual reproduction is also associated with PCD in Saccharomyces cerevisiae. Mating type pheromone can induce PCD in Saccharomyces cerevisiae haploid cells of the corresponding mating type that fail to mate (Severin and Hyman, 2002); low levels of hormone promote mating whereas high levels promote PCD. The mating type pheromone binds to its receptor (either Ste2p or Ste3p) and activates a MAPK signaling pathway involving the Ste20p kinase. There follows an increase in cytoplasmic Ca++ levels, increased mitochondrial activity and ROS production, mitochondrial fission dependent upon mitochondrial protein Ysp1p, and finally cytochrome c release and cell death (Pozniakovsky et al., 2005). Cytochrome c release is also implicated in PCD caused by acetic acid (Ludovico et al., 2002). PCD can also occur during meiosis of diploid yeast cells, where it is regulated by histone H2B phosphorylation (Ahn et al., 2006; Ahn et al., 2005). It is interesting to note that hormones involved in sexual differentiation in mammals, such as testosterone and estrogen, are also regulators of MAPK signaling and PCD (Vasconsuelo et al., 2011), and sexual differentiation is associated with aging in several species (Tower, 2006, 2014), suggesting possible conservation of mechanisms.
Saccharomyces cerevisiae was also found to undergo a form of programmed necrosis (Carmona-Gutierrez et al., 2011). The yeast gene Pep4 is related to mammalian cathepsin D, and deletion of Pep4 shortened chronological life span. Shortened life span was associated with increased ROS production and increased apoptotic-like PCD, as indicated by phosphatidylserine externalization, as well as increased necrotic-like PCD, as indicated by loss of membrane integrity. In turn, over-expression of Pep4 increased chronological life span associated with decreased necrotic-like PCD.
The fungus Podospora anserina has been a pioneering model for the study of aging (Osiewacz and Borghouts, 2000). For several genotypes the culture will grow for 3-5 weeks and then enter senescence marked by mitochondrial DNA instabilities, increased ROS, mitochondrial fission, accumulation of pigment and cell death (Gredilla et al., 2006; Osiewacz et al., 2010). Genetic studies indicate that PCD factors conserved with Saccharomyces cerevisiae and with mammals play a role in Podospora anserina aging. Deletion of the conserved mitochondrial fission regulator PaDnm1 dramatically extended Podospora anserina life span (Scheckhuber et al., 2007). Deletion of the metacaspase gene PaMca1 caused increased life span, indicative a role for metacaspase-dependent PCD. In addition, deletion of mitochondrial AIF-like factors PaAIF2 and PaAMID2 caused increased resistance to oxidative stress and increased life span independent of metacaspase activity, indicative of a metacaspase-independent PCD (Brust et al., 2010b). In mammals, the peptidyl-cis,trans-prolyl-isomerase called Cyclophilin D interacts with the F1F0-ATP synthase and regulates mitochondrial membrane permeabilization and a programmed-necrosis form of PCD (Elrod and Molkentin, 2013; Giorgio et al., 2013). In Podospora anserina, the expression of Cyclophilin D is increased during aging, and treatment with the Cyclophilin D inhibitor cyclosporin A could increase life span in both wild-type and Cyclophilin D-over-expressing strains(Brust et al., 2010a), suggesting a conserved mechanism.
Taken together the studies in fungi suggest conservation of several aspects of PCD with mammals, including a critical regulatory role for the mitochondria. Interestingly, fungal PCD is regulated by sex hormones and during gametogenesis, similar to the situation in flies, nematodes and mammals. In the fungi PCD appears to limit life span in both replicative and chronological assays. The pro-apoptotic role of ROS, mitochondrial DNA rearrangements and release of pro-apoptotic factors from permeabilized mitochondria suggests that a failure in mitochondrial maintenance may contribute to the increased sensitivity to PCD during fungal aging(Bernhardt et al., 2014), similar to the situation in mammals.
8. Why is PCD mis-regulated during aging?
There are several aspects to PCD that may make this process particularly susceptible to disruptions during aging. First, as discussed above, mammalian PCD is a highly-regulated process that is key to the homeostasis of numerous tissues, including the immune system, and is therefore regulated by numerous inputs. These inputs include multiple hormonal, ECM and cell-cell signals, thereby providing many potential targets for aging-associated disruptions. Second, the normal regulation of PCD involves a delicate balance of multiple pro-PCD and anti-PCD factors, and cells appear poised for PCD upon appropriate signaling. This may make PCD pathways particularly vulnerable to maladaptive signals during aging. Third, the important role of the mitochondria and ROS in PCD is likely a factor, because mitochondria appear to be particularly susceptible to damage and malfunction during aging across species (Bernhardt et al., 2014; Hur et al., 2014; Wallace, 2010).
Several aspects of the observed mis-regulation of PCD during aging support the idea that mitochondrial malfunctionis a key underlying factor. First, mitochondria are increasingly implicated in the regulation of PCD across species (Figure 1), and mitochondrial malfunction is consistent with either increased or decreased PCD depending on the cellular context. In humans the observed systemic decrease in PCD markers during aging(Kavathia et al., 2009)is likely to be linked to the decrease in cell turnover rate observed for highly-mitotic tissues such as skin and blood. This global decrease in coordinated cell birth and PCD might in turn result from mitochondrial malfunction and the aging-associated decrease in resting metabolic rate (Fukagawa et al., 1990). The metabolism of cells is linked to the metabolism of the organism(Savage et al., 2007), and during aging metabolic capacity may become insufficient to support robust turnover of the highly mitotic tissues. At the same time, aging-associated failures in mitochondrial maintenance, such as reduced mitochondrial turnover rate (mitophagy), may predispose mitochondria to the loss of membrane potential and membrane permeabilization that promote PCD, leading to increased PCD in cell types such as the muscle. Decreased mitophagy will result in longer-lived and more damage-prone mitochondria, and decreased mitophagy is associated with aging across multiple species (Gelino and Hansen, 2012; Liu et al., 2014; Richard et al., 2013). The mitochondrial genome has a high mutation rate, and mitochondrial mutations are increasingly implicated in mitochondrial malfunction during aging(Schon et al., 2012; Sevini et al., 2014). Because mitophagy is important in clearing mutated mitochondrial genomes from the cell, the decrease in mitophagy with age may be involved in the accumulation of mutated mitochondrial genomes that favor mitochondrial malfunction, including maladaptive PCD(Tower, 2014). Finally, the uni-parental inheritance of mitochondrial genomes may create genetic conflicts (sexual antagonistic pleiotropy) that cause mitochondrial functions, including PCD, to be particularly susceptible to aging-associated disruptions, including sex-biased mis-regulation(Maklakov and Lummaa, 2013; Tower, 2006, 2014).
Acknowledgements
This work was supported by a grant from the Department of Health and Human Services (AG011833) and by pilot project funds from the Southern California Environmental Health Sciences Center (5P30ES007048).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ahn SH, Diaz RL, Grunstein M, Allis CD. Histone H2B deacetylation at lysine 11 is required for yeast apoptosis induced by phosphorylation of H2B at serine 10. Mol Cell. 2006;24:211–220. doi: 10.1016/j.molcel.2006.09.008. [DOI] [PubMed] [Google Scholar]
- Ahn SH, Henderson KA, Keeney S, Allis CD. H2B (Ser10) phosphorylation is induced during apoptosis and meiosis in S. cerevisiae. Cell Cycle. 2005;4:780–783. doi: 10.4161/cc.4.6.1745. [DOI] [PubMed] [Google Scholar]
- Alt EU, Senst C, Murthy SN, Slakey DP, Dupin CL, Chaffin AE, Kadowitz PJ, Izadpanah R. Aging alters tissue resident mesenchymal stem cell properties. Stem cell research. 2012;8:215–225. doi: 10.1016/j.scr.2011.11.002. [DOI] [PubMed] [Google Scholar]
- Andersen BB, Gundersen HJ, Pakkenberg B. Aging of the human cerebellum: a stereological study. The Journal of comparative neurology. 2003;466:356–365. doi: 10.1002/cne.10884. [DOI] [PubMed] [Google Scholar]
- Andux S, Ellis RE. Apoptosis maintains oocyte quality in aging Caenorhabditis elegans females. PLoS Genet. 2008;4:e1000295. doi: 10.1371/journal.pgen.1000295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergmann O, Bhardwaj RD, Bernard S, Zdunek S, Barnabe-Heider F, Walsh S, Zupicich J, Alkass K, Buchholz BA, Druid H, Jovinge S, Frisen J. Evidence for cardiomyocyte renewal in humans. Science. 2009;324:98–102. doi: 10.1126/science.1164680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernhardt D, Hamann A, Osiewacz HD. The role of mitochondria in fungal aging. Current opinion in microbiology. 2014;22C:1–7. doi: 10.1016/j.mib.2014.09.007. [DOI] [PubMed] [Google Scholar]
- Bosserman L, Prendergast F, Herbst R, Fleisher M, Salom E, Strickland S, Raptis A, Hallquist A, Perree M, Rajurkar S, Karimi M, Rogers K, Davidson D, Willis C, Penalver M, Homesley H, Burrell M, Garrett A, Rutledge J, Chernick M, Presant CA. The microculture-kinetic (MiCK) assay: the role of a drug-induced apoptosis assay in drug development and clinical care. Cancer research. 2012;72:3901–3905. doi: 10.1158/0008-5472.CAN-12-0681. [DOI] [PubMed] [Google Scholar]
- Brust D, Daum B, Breunig C, Hamann A, Kuhlbrandt W, Osiewacz HD. Cyclophilin D links programmed cell death and organismal aging in Podospora anserina. Aging Cell. 2010a;9:761–775. doi: 10.1111/j.1474-9726.2010.00609.x. [DOI] [PubMed] [Google Scholar]
- Brust D, Hamann A, Osiewacz HD. Deletion of PaAif2 and PaAmid2, two genes encoding mitochondrial AIF-like oxidoreductases of Podospora anserina, leads to increased stress tolerance and lifespan extension. Current genetics. 2010b;56:225–235. doi: 10.1007/s00294-010-0295-1. [DOI] [PubMed] [Google Scholar]
- Burek MJ, Oppenheim RW. Programmed cell death in the developing nervous system. Brain Pathol. 1996;6:427–446. doi: 10.1111/j.1750-3639.1996.tb00874.x. [DOI] [PubMed] [Google Scholar]
- Buttner S, Eisenberg T, Carmona-Gutierrez D, Ruli D, Knauer H, Ruckenstuhl C, Sigrist C, Wissing S, Kollroser M, Frohlich KU, Sigrist S, Madeo F. Endonuclease G regulates budding yeast life and death. Mol Cell. 2007;25:233–246. doi: 10.1016/j.molcel.2006.12.021. [DOI] [PubMed] [Google Scholar]
- Buttner S, Ruli D, Vogtle FN, Galluzzi L, Moitzi B, Eisenberg T, Kepp O, Habernig L, Carmona-Gutierrez D, Rockenfeller P, Laun P, Breitenbach M, Khoury C, Frohlich KU, Rechberger G, Meisinger C, Kroemer G, Madeo F. A yeast BH3-only protein mediates the mitochondrial pathway of apoptosis. EMBO J. 2011;30:2779–2792. doi: 10.1038/emboj.2011.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camplejohn RS, Gilchrist R, Easton D, McKenzie-Edwards E, Barnes DM, Eccles DM, Ardern-Jones A, Hodgson SV, Duddy PM, Eeles RA. Apoptosis, ageing and cancer susceptibility. British journal of cancer. 2003;88:487–490. doi: 10.1038/sj.bjc.6600767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmona-Gutierrez D, Bauer MA, Ring J, Knauer H, Eisenberg T, Buttner S, Ruckenstuhl C, Reisenbichler A, Magnes C, Rechberger GN, Birner-Gruenberger R, Jungwirth H, Frohlich KU, Sinner F, Kroemer G, Madeo F. The propeptide of yeast cathepsin D inhibits programmed necrosis. Cell death & disease. 2011;2:e161. doi: 10.1038/cddis.2011.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmona-Gutierrez D, Eisenberg T, Buttner S, Meisinger C, Kroemer G, Madeo F. Apoptosis in yeast: triggers, pathways, subroutines. Cell Death Differ. 2010a;17:763–773. doi: 10.1038/cdd.2009.219. [DOI] [PubMed] [Google Scholar]
- Carmona-Gutierrez D, Frohlich KU, Kroemer G, Madeo F. Metacaspases are caspases. Doubt no more. Cell Death Differ. 2010b;17:377–378. doi: 10.1038/cdd.2009.198. [DOI] [PubMed] [Google Scholar]
- Chihara T, Kitabayashi A, Morimoto M, Takeuchi K, Masuyama K, Tonoki A, Davis RL, Wang JW, Miura M. Caspase inhibition in select olfactory neurons restores innate attraction behavior in aged Drosophila. PLoS Genet. 2014;10:e1004437. doi: 10.1371/journal.pgen.1004437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Childs BG, Baker DJ, Kirkland JL, Campisi J, van Deursen JM. Senescence and apoptosis: dueling or complementary cell fates? EMBO reports. 2014 Oct 13; doi: 10.15252/embr.201439245. doi: 10.15252/embr.201439245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chimal-Monroy J, Abarca-Buis RF, Cuervo R, Diaz-Hernandez M, Bustamante M, Rios-Flores JA, Romero-Suarez S, Farrera-Hernandez A. Molecular control of cell differentiation and programmed cell death during digit development. IUBMB Life. 2011;63:922–929. doi: 10.1002/iub.563. [DOI] [PubMed] [Google Scholar]
- Chou JP, Effros RB. T cell replicative senescence in human aging. Curr Pharm Des. 2013;19:1680–1698. doi: 10.2174/138161213805219711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature. 2005;433:760–764. doi: 10.1038/nature03260. [DOI] [PubMed] [Google Scholar]
- Degterev A, Zhou W, Maki JL, Yuan J. Assays for necroptosis and activity of RIP kinases. Methods in enzymology. 2014;545:1–33. doi: 10.1016/B978-0-12-801430-1.00001-9. [DOI] [PubMed] [Google Scholar]
- Delaney JR, Sutphin GL, Dulken B, Sim S, Kim JR, Robison B, Schleit J, Murakami CJ, Carr D, An EH, Choi E, Chou A, Fletcher M, Jelic M, Liu B, Lockshon D, Moller RM, Pak DN, Peng Q, Peng ZJ, Pham KM, Sage M, Solanky A, Steffen KK, Tsuchiya M, Tsuchiyama S, Johnson S, Raabe C, Suh Y, Zhou Z, Liu X, Kennedy BK, Kaeberlein M. Sir2 deletion prevents lifespan extension in 32 long-lived mutants. Aging Cell. 2011;10:1089–1091. doi: 10.1111/j.1474-9726.2011.00742.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delavallee L, Cabon L, Galan-Malo P, Lorenzo HK, Susin SA. AIF-mediated caspase-independent necroptosis: a new chance for targeted therapeutics. IUBMB Life. 2011;63:221–232. doi: 10.1002/iub.432. [DOI] [PubMed] [Google Scholar]
- Denton D, Aung-Htut MT, Kumar S. Developmentally programmed cell death in Drosophila. Biochimica et biophysica acta. 2013;1833:3499–3506. doi: 10.1016/j.bbamcr.2013.06.014. [DOI] [PubMed] [Google Scholar]
- Devitt A, Marshall LJ. The innate immune system and the clearance of apoptotic cells. Journal of leukocyte biology. 2011;90:447–457. doi: 10.1189/jlb.0211095. [DOI] [PubMed] [Google Scholar]
- Eckhart L, Lippens S, Tschachler E, Declercq W. Cell death by cornification. Biochimica et biophysica acta. 2013;1833:3471–3480. doi: 10.1016/j.bbamcr.2013.06.010. [DOI] [PubMed] [Google Scholar]
- Egilmez NK, Jazwinski SM. Evidence for the involvement of a cytoplasmic factor in the aging of the yeast Saccharomyces cerevisiae. J Bacteriol. 1989;171:37–42. doi: 10.1128/jb.171.1.37-42.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35:495–516. doi: 10.1080/01926230701320337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elrod JW, Molkentin JD. Physiologic functions of cyclophilin D and the mitochondrial permeability transition pore. Circulation journal : official journal of the Japanese Circulation Society. 2013;77:1111–1122. doi: 10.1253/circj.cj-13-0321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estaquier J, Vallette F, Vayssiere JL, Mignotte B. The mitochondrial pathways of apoptosis. Adv Exp Med Biol. 2012;942:157–183. doi: 10.1007/978-94-007-2869-1_7. [DOI] [PubMed] [Google Scholar]
- Fabrizio P, Battistella L, Vardavas R, Gattazzo C, Liou LL, Diaspro A, Dossen JW, Gralla EB, Longo VD. Superoxide is a mediator of an altruistic aging program in Saccharomyces cerevisiae. J Cell Biol. 2004;166:1055–1067. doi: 10.1083/jcb.200404002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabrizio P, Longo VD. The chronological life span of Saccharomyces cerevisiae. Aging Cell. 2003;2:73–81. doi: 10.1046/j.1474-9728.2003.00033.x. [DOI] [PubMed] [Google Scholar]
- Fahrenkrog B. Nma111p, the pro-apoptotic HtrA-like nuclear serine protease in Saccharomyces cerevisiae: a short survey. Biochemical Society transactions. 2011;39:1499–1501. doi: 10.1042/BST0391499. [DOI] [PubMed] [Google Scholar]
- Fahrenkrog B, Sauder U, Aebi U. The S. cerevisiae HtrA-like protein Nma111p is a nuclear serine protease that mediates yeast apoptosis. J Cell Sci. 2004;117:115–126. doi: 10.1242/jcs.00848. [DOI] [PubMed] [Google Scholar]
- Fannjiang Y, Cheng WC, Lee SJ, Qi B, Pevsner J, McCaffery JM, Hill RB, Basanez G, Hardwick JM. Mitochondrial fission proteins regulate programmed cell death in yeast. Genes Dev. 2004;18:2785–2797. doi: 10.1101/gad.1247904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Folch J, Junyent F, Verdaguer E, Auladell C, Pizarro JG, Beas-Zarate C, Pallas M, Camins A. Role of cell cycle re-entry in neurons: a common apoptotic mechanism of neuronal cell death. Neurotoxicity research. 2012;22:195–207. doi: 10.1007/s12640-011-9277-4. [DOI] [PubMed] [Google Scholar]
- Frank S, Gaume B, Bergmann-Leitner ES, Leitner WW, Robert EG, Catez F, Smith CL, Youle RJ. The role of dynamin-related protein 1, a mediator of mitochondrial fission, in apoptosis. Dev Cell. 2001;1:515–525. doi: 10.1016/s1534-5807(01)00055-7. [DOI] [PubMed] [Google Scholar]
- Fujimoto C, Yamasoba T. Oxidative stresses and mitochondrial dysfunction in age-related hearing loss. Oxidative medicine and cellular longevity. 2014;2014:582849. doi: 10.1155/2014/582849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukagawa NK, Bandini LG, Young JB. Effect of age on body composition and resting metabolic rate. The American journal of physiology. 1990;259:E233–238. doi: 10.1152/ajpendo.1990.259.2.E233. [DOI] [PubMed] [Google Scholar]
- Galluzzi L, Bravo-San Pedro JM, Vitale I, Aaronson SA, Abrams JM, Adam D, Alnemri ES, Altucci L, Andrews D, Annicchiarico-Petruzzelli M, Baehrecke EH, Bazan NG, Bertrand MJ, Bianchi K, Blagosklonny MV, Blomgren K, Borner C, Bredesen DE, Brenner C, Campanella M, Candi E, Cecconi F, Chan FK, Chandel NS, Cheng EH, Chipuk JE, Cidlowski JA, Ciechanover A, Dawson TM, Dawson VL, De Laurenzi V, De Maria R, Debatin KM, Di Daniele N, Dixit VM, Dynlacht BD, El-Deiry WS, Fimia GM, Flavell RA, Fulda S, Garrido C, Gougeon ML, Green DR, Gronemeyer H, Hajnoczky G, Hardwick JM, Hengartner MO, Ichijo H, Joseph B, Jost PJ, Kaufmann T, Kepp O, Klionsky DJ, Knight RA, Kumar S, Lemasters JJ, Levine B, Linkermann A, Lipton SA, Lockshin RA, Lopez-Otin C, Lugli E, Madeo F, Malorni W, Marine JC, Martin SJ, Martinou JC, Medema JP, Meier P, Melino S, Mizushima N, Moll U, Munoz-Pinedo C, Nunez G, Oberst A, Panaretakis T, Penninger JM, Peter ME, Piacentini M, Pinton P, Prehn JH, Puthalakath H, Rabinovich GA, Ravichandran KS, Rizzuto R, Rodrigues CM, Rubinsztein DC, Rudel T, Shi Y, Simon HU, Stockwell BR, Szabadkai G, Tait SW, Tang HL, Tavernarakis N, Tsujimoto Y, Vanden Berghe T, Vandenabeele P, Villunger A, Wagner EF, Walczak H, White E, Wood WG, Yuan J, Zakeri Z, Zhivotovsky B, Melino G, Kroemer G. Essential versus accessory aspects of cell death: recommendations of the NCCD 2015. Cell Death Differ. 2015;22:58–73. doi: 10.1038/cdd.2014.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galluzzi L, Kepp O, Kroemer G. RIP kinases initiate programmed necrosis. Journal of molecular cell biology. 2009;1:8–10. doi: 10.1093/jmcb/mjp007. [DOI] [PubMed] [Google Scholar]
- Galluzzi L, Vitale I, Abrams JM, Alnemri ES, Baehrecke EH, Blagosklonny MV, Dawson TM, Dawson VL, El-Deiry WS, Fulda S, Gottlieb E, Green DR, Hengartner MO, Kepp O, Knight RA, Kumar S, Lipton SA, Lu X, Madeo F, Malorni W, Mehlen P, Nunez G, Peter ME, Piacentini M, Rubinsztein DC, Shi Y, Simon HU, Vandenabeele P, White E, Yuan J, Zhivotovsky B, Melino G, Kroemer G. Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ. 2012;19:107–120. doi: 10.1038/cdd.2011.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garigan D, Hsu AL, Fraser AG, Kamath RS, Ahringer J, Kenyon C. Genetic analysis of tissue aging in Caenorhabditis elegans: a role for heat-shock factor and bacterial proliferation. Genetics. 2002;161:1101–1112. doi: 10.1093/genetics/161.3.1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelino S, Hansen M. Autophagy - An Emerging Anti-Aging Mechanism. Journal of clinical & experimental pathology. 2012 Jul 12;(Suppl 4) doi: 10.4172/2161-0681.s4-006. pii: 006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghavami S, Shojaei S, Yeganeh B, Ande SR, Jangamreddy JR, Mehrpour M, Christoffersson J, Chaabane W, Moghadam AR, Kashani HH, Hashemi M, Owji AA, Los MJ. Autophagy and apoptosis dysfunction in neurodegenerative disorders. Prog Neurobiol. 2014;112:24–49. doi: 10.1016/j.pneurobio.2013.10.004. [DOI] [PubMed] [Google Scholar]
- Giannaris EL, Rosene DL. A stereological study of the numbers of neurons and glia in the primary visual cortex across the lifespan of male and female rhesus monkeys. The Journal of comparative neurology. 2012;520:3492–3508. doi: 10.1002/cne.23101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giorgio V, von Stockum S, Antoniel M, Fabbro A, Fogolari F, Forte M, Glick GD, Petronilli V, Zoratti M, Szabo I, Lippe G, Bernardi P. Dimers of mitochondrial ATP synthase form the permeability transition pore. Proc Natl Acad Sci U S A. 2013;110:5887–5892. doi: 10.1073/pnas.1217823110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gredilla R, Grief J, Osiewacz HD. Mitochondrial free radical generation and lifespan control in the fungal aging model Podospora anserina. Exp Gerontol. 2006;41:439–447. doi: 10.1016/j.exger.2006.01.010. [DOI] [PubMed] [Google Scholar]
- Green DR, Kroemer G. Cytoplasmic functions of the tumour suppressor p53. Nature. 2009;458:1127–1130. doi: 10.1038/nature07986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gumienny TL, Lambie E, Hartwieg E, Horvitz HR, Hengartner MO. Genetic control of programmed cell death in the Caenorhabditis elegans hermaphrodite germline. Development. 1999;126:1011–1022. doi: 10.1242/dev.126.5.1011. [DOI] [PubMed] [Google Scholar]
- Gunther C, Neumann H, Neurath MF, Becker C. Apoptosis, necrosis and necroptosis: cell death regulation in the intestinal epithelium. Gut. 2013;62:1062–1071. doi: 10.1136/gutjnl-2011-301364. [DOI] [PubMed] [Google Scholar]
- Gupta S. Molecular mechanisms of apoptosis in the cells of the immune system in human aging. Immunological reviews. 2005;205:114–129. doi: 10.1111/j.0105-2896.2005.00261.x. [DOI] [PubMed] [Google Scholar]
- Gupta S, Agrawal A, Agrawal S, Su H, Gollapudi S. A paradox of immunodeficiency and inflammation in human aging: lessons learned from apoptosis. Immunity & ageing : I & A. 2006;3:5. doi: 10.1186/1742-4933-3-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurbuxani S, Schmitt E, Cande C, Parcellier A, Hammann A, Daugas E, Kouranti I, Spahr C, Pance A, Kroemer G, Garrido C. Heat shock protein 70 binding inhibits the nuclear import of apoptosis-inducing factor. Oncogene. 2003;22:6669–6678. doi: 10.1038/sj.onc.1206794. [DOI] [PubMed] [Google Scholar]
- Hankins HM, Baldridge RD, Xu P, Graham TR. Role of flippases, scramblases and transfer proteins in phosphatidylserine subcellular distribution. Traffic. 2015;16:35–47. doi: 10.1111/tra.12233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herker E, Jungwirth H, Lehmann KA, Maldener C, Frohlich KU, Wissing S, Buttner S, Fehr M, Sigrist S, Madeo F. Chronological aging leads to apoptosis in yeast. J Cell Biol. 2004;164:501–507. doi: 10.1083/jcb.200310014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernandez-Martinez R, Covarrubias L. Interdigital cell death function and regulation: new insights on an old programmed cell death model. Dev Growth Differ. 2011;53:245–258. doi: 10.1111/j.1440-169X.2010.01246.x. [DOI] [PubMed] [Google Scholar]
- Herndon LA, Schmeissner PJ, Dudaronek JM, Brown PA, Listner KM, Sakano Y, Paupard MC, Hall DH, Driscoll M. Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature. 2002;419:808–814. doi: 10.1038/nature01135. [DOI] [PubMed] [Google Scholar]
- Hiebert PR, Granville DJ. Granzyme B in injury, inflammation, and repair. Trends Mol Med. 2012;18:732–741. doi: 10.1016/j.molmed.2012.09.009. [DOI] [PubMed] [Google Scholar]
- Higami Y, Shimokawa I. Apoptosis in the aging process. Cell and tissue research. 2000;301:125–132. doi: 10.1007/s004419900156. [DOI] [PubMed] [Google Scholar]
- Hill SM, Nystrom T. The dual role of a yeast metacaspase: What doesn't kill you makes you stronger. Bioessays. 2015 Feb 13; doi: 10.1002/bies.201400208. doi: 10.1002/bies.201400208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hroudova J, Singh N, Fisar Z. Mitochondrial dysfunctions in neurodegenerative diseases: relevance to Alzheimer's disease. BioMed research international. 2014 2014 May 12;:175062. doi: 10.1155/2014/175062. doi: 10.1155/2014/175062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hua Y, Robinson TJ, Cao Y, Shi GP, Ren J, Nair S. Cathepsin K knockout alleviates aging-induced cardiac dysfunction. Aging Cell. 2015 Feb 18; doi: 10.1111/acel.12276. doi: 10.1111/acel.12276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hur JH, Stork DA, Walker DW. Complex-I-ty in aging. Journal of bioenergetics and biomembranes. 2014;46:329–335. doi: 10.1007/s10863-014-9553-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jazwinski SM. The retrograde response: a conserved compensatory reaction to damage from within and from without. Progress in molecular biology and translational science. 2014;127:133–154. doi: 10.1016/B978-0-12-394625-6.00005-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jego G, Hazoume A, Seigneuric R, Garrido C. Targeting heat shock proteins in cancer. Cancer letters. 2013;332:275–285. doi: 10.1016/j.canlet.2010.10.014. [DOI] [PubMed] [Google Scholar]
- Jiang H, Grenley MO, Bravo MJ, Blumhagen RZ, Edgar BA. EGFR/Ras/MAPK signaling mediates adult midgut epithelial homeostasis and regeneration in Drosophila. Cell Stem Cell. 2011;8:84–95. doi: 10.1016/j.stem.2010.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kajstura J, Cheng W, Sarangarajan R, Li P, Li B, Nitahara JA, Chapnick S, Reiss K, Olivetti G, Anversa P. Necrotic and apoptotic myocyte cell death in the aging heart of Fischer 344 rats. The American journal of physiology. 1996;271:H1215–1228. doi: 10.1152/ajpheart.1996.271.3.H1215. [DOI] [PubMed] [Google Scholar]
- Kapetanaki MG, Mora AL, Rojas M. Influence of age on wound healing and fibrosis. J Pathol. 2013;229:310–322. doi: 10.1002/path.4122. [DOI] [PubMed] [Google Scholar]
- Kavathia N, Jain A, Walston J, Beamer BA, Fedarko NS. Serum markers of apoptosis decrease with age and cancer stage. Aging (Albany NY) 2009;1:652–663. doi: 10.18632/aging.100069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerr JF, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. British journal of cancer. 1972;26:239–257. doi: 10.1038/bjc.1972.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kile BT. The role of apoptosis in megakaryocytes and platelets. British journal of haematology. 2014;165:217–226. doi: 10.1111/bjh.12757. [DOI] [PubMed] [Google Scholar]
- Kinet MJ, Shaham S. Noncanonical cell death in the nematode Caenorhabditis elegans. Methods in enzymology. 2014;545:157–180. doi: 10.1016/B978-0-12-801430-1.00007-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King KL, Cidlowski JA. Cell cycle and apoptosis: common pathways to life and death. J Cell Biochem. 1995;58:175–180. doi: 10.1002/jcb.240580206. [DOI] [PubMed] [Google Scholar]
- King KL, Cidlowski JA. Cell cycle regulation and apoptosis. Annu Rev Physiol. 1998;60:601–617. doi: 10.1146/annurev.physiol.60.1.601. [DOI] [PubMed] [Google Scholar]
- Kirchman PA, Kim S, Lai CY, Jazwinski SM. Interorganelle signaling is a determinant of longevity in Saccharomyces cerevisiae. Genetics. 1999;152:179–190. doi: 10.1093/genetics/152.1.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirkland JL, Tchkonia T. Clinical strategies and animal models for developing senolytic agents. Exp Gerontol. 2014 Oct 28; doi: 10.1016/j.exger.2014.10.012. pii: S0531-5565(14)00291-5. doi: 10.1016/j.exger.2014.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri ES, Baehrecke EH, Blagosklonny MV, El-Deiry WS, Golstein P, Green DR, Hengartner M, Knight RA, Kumar S, Lipton SA, Malorni W, Nunez G, Peter ME, Tschopp J, Yuan J, Piacentini M, Zhivotovsky B, Melino G. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ. 2009;16:3–11. doi: 10.1038/cdd.2008.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kujoth GC, Hiona A, Pugh TD, Someya S, Panzer K, Wohlgemuth SE, Hofer T, Seo AY, Sullivan R, Jobling WA, Morrow JD, Van Remmen H, Sedivy JM, Yamasoba T, Tanokura M, Weindruch R, Leeuwenburgh C, Prolla TA. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science. 2005;309:481–484. doi: 10.1126/science.1112125. [DOI] [PubMed] [Google Scholar]
- Kung G, Konstantinidis K, Kitsis RN. Programmed necrosis, not apoptosis, in the heart. Circ Res. 2011;108:1017–1036. doi: 10.1161/CIRCRESAHA.110.225730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwak HB. Effects of aging and exercise training on apoptosis in the heart. Journal of exercise rehabilitation. 2013;9:212–219. doi: 10.12965/jer.130002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwak HB, Lee Y, Kim JH, Van Remmen H, Richardson AG, Lawler JM. MnSOD Overexpression Reduces Fibrosis and Pro-Apoptotic Signaling in the Aging Mouse Heart. J Gerontol A Biol Sci Med Sci. 2014 Jul 11; doi: 10.1093/gerona/glu090. pii: glu090. doi 10.1093/gerona/glu090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanni C, Racchi M, Memo M, Govoni S, Uberti D. p53 at the crossroads between cancer and neurodegeneration. Free Radic Biol Med. 2012;52:1727–1733. doi: 10.1016/j.freeradbiomed.2012.02.034. [DOI] [PubMed] [Google Scholar]
- Laun P, Pichova A, Madeo F, Fuchs J, Ellinger A, Kohlwein S, Dawes I, Frohlich KU, Breitenbach M. Aged mother cells of Saccharomyces cerevisiae show markers of oxidative stress and apoptosis. Mol Microbiol. 2001;39:1166–1173. [PubMed] [Google Scholar]
- Lee M, Lee JS. Exploiting tumor cell senescence in anticancer therapy. BMB reports. 2014;47:51–59. doi: 10.5483/BMBRep.2014.47.2.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y, Kang HC, Lee BD, Lee YI, Kim YP, Shin JH. Poly (ADP-ribose) in the pathogenesis of Parkinson's disease. BMB reports. 2014;47:424–432. doi: 10.5483/BMBRep.2014.47.8.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leeuwenburgh C, Gurley CM, Strotman BA, Dupont-Versteegden EE. Age-related differences in apoptosis with disuse atrophy in soleus muscle. American journal of physiology. Regulatory, integrative and comparative physiology. 2005;288:R1288–1296. doi: 10.1152/ajpregu.00576.2004. [DOI] [PubMed] [Google Scholar]
- Leventis PA, Grinstein S. The distribution and function of phosphatidylserine in cellular membranes. Annual review of biophysics. 2010;39:407–427. doi: 10.1146/annurev.biophys.093008.131234. [DOI] [PubMed] [Google Scholar]
- Liu J, Zhang B, Lei H, Feng Z, Liu J, Hsu AL, Xu XZ. Functional aging in the nervous system contributes to age-dependent motor activity decline in C. elegans. Cell Metab. 2013;18:392–402. doi: 10.1016/j.cmet.2013.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Sakakibara K, Chen Q, Okamoto K. Receptor-mediated mitophagy in yeast and mammalian systems. Cell research. 2014;24:787–795. doi: 10.1038/cr.2014.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long JM, Mouton PR, Jucker M, Ingram DK. What counts in brain aging? Design-based stereological analysis of cell number. J Gerontol A Biol Sci Med Sci. 1999;54:B407–417. doi: 10.1093/gerona/54.10.b407. [DOI] [PubMed] [Google Scholar]
- Longo VD, Shadel GS, Kaeberlein M, Kennedy B. Replicative and chronological aging in Saccharomyces cerevisiae. Cell Metab. 2012;16:18–31. doi: 10.1016/j.cmet.2012.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu B, Chen H, Lu H. The relationship between apoptosis and aging. Advances in Bioscience and Biotechnology. 2012;3:705–711. [Google Scholar]
- Ludovico P, Rodrigues F, Almeida A, Silva MT, Barrientos A, Corte-Real M. Cytochrome c release and mitochondria involvement in programmed cell death induced by acetic acid in Saccharomyces cerevisiae. Mol Biol Cell. 2002;13:2598–2606. doi: 10.1091/mbc.E01-12-0161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madeo F, Herker E, Maldener C, Wissing S, Lachelt S, Herlan M, Fehr M, Lauber K, Sigrist SJ, Wesselborg S, Frohlich KU. A caspase-related protease regulates apoptosis in yeast. Mol Cell. 2002;9:911–917. doi: 10.1016/s1097-2765(02)00501-4. [DOI] [PubMed] [Google Scholar]
- Maklakov AA, Lummaa V. Evolution of sex differences in lifespan and aging: causes and constraints. Bioessays. 2013;35:717–724. doi: 10.1002/bies.201300021. [DOI] [PubMed] [Google Scholar]
- Marcotte R, Lacelle C, Wang E. Senescent fibroblasts resist apoptosis by downregulating caspase-3. Mech Ageing Dev. 2004;125:777–783. doi: 10.1016/j.mad.2004.07.007. [DOI] [PubMed] [Google Scholar]
- Marzetti E, Calvani R, Cesari M, Buford TW, Lorenzi M, Behnke BJ, Leeuwenburgh C. Mitochondrial dysfunction and sarcopenia of aging: from signaling pathways to clinical trials. Int J Biochem Cell Biol. 2013;45:2288–2301. doi: 10.1016/j.biocel.2013.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marzetti E, Lawler JM, Hiona A, Manini T, Seo AY, Leeuwenburgh C. Modulation of age-induced apoptotic signaling and cellular remodeling by exercise and calorie restriction in skeletal muscle. Free Radic Biol Med. 2008;44:160–168. doi: 10.1016/j.freeradbiomed.2007.05.028. [DOI] [PubMed] [Google Scholar]
- Maurer CW, Chiorazzi M, Shaham S. Timing of the onset of a developmental cell death is controlled by transcriptional induction of the C. elegans ced-3 caspase-encoding gene. Development. 2007;134:1357–1368. doi: 10.1242/dev.02818. [DOI] [PubMed] [Google Scholar]
- McGee MD, Day N, Graham J, Melov S. cep-1/p53-dependent dysplastic pathology of the aging C. elegans gonad. Aging (Albany NY) 2012;4:256–269. doi: 10.18632/aging.100448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrill DA, Chiba AA, Tuszynski MH. Conservation of neuronal number and size in the entorhinal cortex of behaviorally characterized aged rats. The Journal of comparative neurology. 2001;438:445–456. doi: 10.1002/cne.1327. [DOI] [PubMed] [Google Scholar]
- Metzstein MM, Stanfield GM, Horvitz HR. Genetics of programmed cell death in C. elegans: past, present and future. Trends Genet. 1998;14:410–416. doi: 10.1016/s0168-9525(98)01573-x. [DOI] [PubMed] [Google Scholar]
- Micchelli CA, Perrimon N. Evidence that stem cells reside in the adult Drosophila midgut epithelium. Nature. 2006;439:475–479. doi: 10.1038/nature04371. [DOI] [PubMed] [Google Scholar]
- Misell LM, Hwang ES, Au A, Esserman L, Hellerstein MK. Development of a novel method for measuring in vivo breast epithelial cell proliferation in humans. Breast cancer research and treatment. 2005;89:257–264. doi: 10.1007/s10549-004-2228-5. [DOI] [PubMed] [Google Scholar]
- Nixon RA, Yang DS. Autophagy and neuronal cell death in neurological disorders. Cold Spring Harb Perspect Biol 4. 2012 Oct 1;4(10) doi: 10.1101/cshperspect.a008839. pii: a008839. doi: 10.1101/cshperspect.a008839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolan LA, Lunness HR, Lightman SL, Levy A. The effects of age and spontaneous adenoma formation on trophic activity in the rat pituitary gland: a comparison with trophic activity in the human pituitary and in human pituitary adenomas. Journal of neuroendocrinology. 1999;11:393–401. doi: 10.1046/j.1365-2826.1999.00333.x. [DOI] [PubMed] [Google Scholar]
- Ohlstein B, Spradling A. The adult Drosophila posterior midgut is maintained by pluripotent stem cells. Nature. 2006;439:470–474. doi: 10.1038/nature04333. [DOI] [PubMed] [Google Scholar]
- Olivetti G, Melissari M, Capasso JM, Anversa P. Cardiomyopathy of the aging human heart. Myocyte loss and reactive cellular hypertrophy. Circ Res. 1991;68:1560–1568. doi: 10.1161/01.res.68.6.1560. [DOI] [PubMed] [Google Scholar]
- Oppenheim RW. Cell death during development of the nervous system. Annual review of neuroscience. 1991;14:453–501. doi: 10.1146/annurev.ne.14.030191.002321. [DOI] [PubMed] [Google Scholar]
- Oshiro S, Morioka MS, Kikuchi M. Dysregulation of iron metabolism in Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis. Advances in pharmacological sciences. 2011;2011:378278. doi: 10.1155/2011/378278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osiewacz HD, Borghouts C. Cellular copper homeostasis, mitochondrial DNA instabilities, and lifespan control in the filamentous fungus Podospora anserina. Exp Gerontol. 2000;35:677–686. doi: 10.1016/s0531-5565(00)00142-x. [DOI] [PubMed] [Google Scholar]
- Osiewacz HD, Brust D, Hamann A, Kunstmann B, Luce K, Muller-Ohldach M, Scheckhuber CQ, Servos J, Strobel I. Mitochondrial pathways governing stress resistance, life, and death in the fungal aging model Podospora anserina. Ann N Y Acad Sci. 2010;1197:54–66. doi: 10.1111/j.1749-6632.2010.05190.x. [DOI] [PubMed] [Google Scholar]
- Otera H, Ishihara N, Mihara K. New insights into the function and regulation of mitochondrial fission. Biochimica et biophysica acta. 2013;1833:1256–1268. doi: 10.1016/j.bbamcr.2013.02.002. [DOI] [PubMed] [Google Scholar]
- Pandey S, Wang E. Cells en route to apoptosis are characterized by the upregulation of c-fos, c-myc, c-jun, cdc2, and RB phosphorylation, resembling events of early cell-cycle traverse. J Cell Biochem. 1995;58:135–150. doi: 10.1002/jcb.240580203. [DOI] [PubMed] [Google Scholar]
- Paoli P, Giannoni E, Chiarugi P. Anoikis molecular pathways and its role in cancer progression. Biochimica et biophysica acta. 2013;1833:3481–3498. doi: 10.1016/j.bbamcr.2013.06.026. [DOI] [PubMed] [Google Scholar]
- Papucci L, Formigli L, Schiavone N, Tani A, Donnini M, Lapucci A, Perna F, Tempestini A, Witort E, Morganti M, Nosi D, Orlandini GE, Zecchi Orlandini S, Capaccioli S. Apoptosis shifts to necrosis via intermediate types of cell death by a mechanism depending on c-myc and bcl-2 expression. Cell and tissue research. 2004;316:197–209. doi: 10.1007/s00441-004-0872-z. [DOI] [PubMed] [Google Scholar]
- Parrish J, Li L, Klotz K, Ledwich D, Wang X, Xue D. Mitochondrial endonuclease G is important for apoptosis in C. elegans. Nature. 2001;412:90–94. doi: 10.1038/35083608. [DOI] [PubMed] [Google Scholar]
- Pasparakis M, Vandenabeele P. Necroptosis and its role in inflammation. Nature. 2015;517:311–320. doi: 10.1038/nature14191. [DOI] [PubMed] [Google Scholar]
- Polyak K, Wu TT, Hamilton SR, Kinzler KW, Vogelstein B. Less death in the dying. Cell Death Differ. 1997;4:242–246. doi: 10.1038/sj.cdd.4400226. [DOI] [PubMed] [Google Scholar]
- Powers RW, 3rd, Kaeberlein M, Caldwell SD, Kennedy BK, Fields S. Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes Dev. 2006;20:174–184. doi: 10.1101/gad.1381406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pozniakovsky AI, Knorre DA, Markova OV, Hyman AA, Skulachev VP, Severin FF. Role of mitochondria in the pheromone- and amiodarone-induced programmed death of yeast. J Cell Biol. 2005;168:257–269. doi: 10.1083/jcb.200408145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proskuryakov SY, Gabai VL. Mechanisms of tumor cell necrosis. Curr Pharm Des. 2010;16:56–68. doi: 10.2174/138161210789941793. [DOI] [PubMed] [Google Scholar]
- Qian D, Wu X, Jiang H, Gao P, Kuang C, Wang K, Huang L. Aging reduces susceptibility of vascular smooth muscle cells to H(2)O(2)-induced apoptosis through the down-regulation of Jagged1 expression in endothelial cells. International journal of molecular medicine. 2011;28:207–213. doi: 10.3892/ijmm.2011.671. [DOI] [PubMed] [Google Scholar]
- Raff MC, Barres BA, Burne JF, Coles HS, Ishizaki Y, Jacobson MD. Programmed cell death and the control of cell survival. Philos Trans R Soc Lond B Biol Sci. 1994;345:265–268. doi: 10.1098/rstb.1994.0104. [DOI] [PubMed] [Google Scholar]
- Rai NK, Tripathi K, Sharma D, Shukla VK. Apoptosis: a basic physiologic process in wound healing. The international journal of lower extremity wounds. 2005;4:138–144. doi: 10.1177/1534734605280018. [DOI] [PubMed] [Google Scholar]
- Rapp PR, Gallagher M. Preserved neuron number in the hippocampus of aged rats with spatial learning deficits. Proc Natl Acad Sci U S A. 1996;93:9926–9930. doi: 10.1073/pnas.93.18.9926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddien PW, Horvitz HR. The engulfment process of programmed cell death in caenorhabditis elegans. Annual review of cell and developmental biology. 2004;20:193–221. doi: 10.1146/annurev.cellbio.20.022003.114619. [DOI] [PubMed] [Google Scholar]
- Refaat A, Abd-Rabou A, Reda A. TRAIL combinations: The new ‘trail’ for cancer therapy (Review). Oncology letters. 2014;7:1327–1332. doi: 10.3892/ol.2014.1922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renehan AG, Booth C, Potten CS. What is apoptosis, and why is it important? BMJ. 2001;322:1536–1538. doi: 10.1136/bmj.322.7301.1536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rerole AL, Jego G, Garrido C. Hsp70: anti-apoptotic and tumorigenic protein. Methods Mol Biol. 2011;787:205–230. doi: 10.1007/978-1-61779-295-3_16. [DOI] [PubMed] [Google Scholar]
- Richard VR, Leonov A, Beach A, Burstein MT, Koupaki O, Gomez-Perez A, Levy S, Pluska L, Mattie S, Rafesh R, Iouk T, Sheibani S, Greenwood M, Vali H, Titorenko VI. Macromitophagy is a longevity assurance process that in chronologically aging yeast limited in calorie supply sustains functional mitochondria and maintains cellular lipid homeostasis. Aging (Albany NY) 2013;5:234–269. doi: 10.18632/aging.100547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson RB, Allan DS, Le Y. Greater organ involution in highly proliferative tissues associated with the early onset and acceleration of ageing in humans. Exp Gerontol. 2014;55:80–91. doi: 10.1016/j.exger.2014.03.015. [DOI] [PubMed] [Google Scholar]
- Rinnerthaler M, Streubel MK, Bischof J, Richter K. Skin aging, gene expression and calcium. Exp Gerontol. 2014 Sep 26; doi: 10.1016/j.exger.2014.09.015. pii: S0531-5565(14)00272-1. doi: 10.1016/j.exger.2014.09.015. [DOI] [PubMed] [Google Scholar]
- Rochette PJ, Brash DE. Progressive apoptosis resistance prior to senescence and control by the anti-apoptotic protein BCL-xL. Mech Ageing Dev. 2008;129:207–214. doi: 10.1016/j.mad.2007.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryu SJ, Oh YS, Park SC. Failure of stress-induced downregulation of Bcl-2 contributes to apoptosis resistance in senescent human diploid fibroblasts. Cell Death Differ. 2007;14:1020–1028. doi: 10.1038/sj.cdd.4402091. [DOI] [PubMed] [Google Scholar]
- Sainz RM, Mayo JC, Reiter RJ, Tan DX, Rodriguez C. Apoptosis in primary lymphoid organs with aging. Microscopy research and technique. 2003;62:524–539. doi: 10.1002/jemt.10414. [DOI] [PubMed] [Google Scholar]
- Salminen A, Ojala J, Kaarniranta K. Apoptosis and aging: increased resistance to apoptosis enhances the aging process. Cell Mol Life Sci. 2011;68:1021–1031. doi: 10.1007/s00018-010-0597-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salvioli S, Monti D, Lanzarini C, Conte M, Pirazzini C, Bacalini MG, Garagnani P, Giuliani C, Fontanesi E, Ostan R, Bucci L, Sevini F, Yani SL, Barbieri A, Lomartire L, Borelli V, Vianello D, Bellavista E, Martucci M, Cevenini E, Pini E, Scurti M, Biondi F, Santoro A, Capri M, Franceschi C. Immune system, cell senescence, aging and longevity--inflamm-aging reappraised. Curr Pharm Des. 2013;19:1675–1679. [PubMed] [Google Scholar]
- Savage VM, Allen AP, Brown JH, Gillooly JF, Herman AB, Woodruff WH, West GB. Scaling of number, size, and metabolic rate of cells with body size in mammals. Proc Natl Acad Sci U S A. 2007;104:4718–4723. doi: 10.1073/pnas.0611235104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheckhuber CQ, Erjavec N, Tinazli A, Hamann A, Nystrom T, Osiewacz HD. Reducing mitochondrial fission results in increased life span and fitness of two fungal ageing models. Nat Cell Biol. 2007;9:99–105. doi: 10.1038/ncb1524. [DOI] [PubMed] [Google Scholar]
- Schon EA, DiMauro S, Hirano M. Human mitochondrial DNA: roles of inherited and somatic mutations. Nat Rev Genet. 2012;13:878–890. doi: 10.1038/nrg3275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seiberg M. Age-induced hair greying - the multiple effects of oxidative stress. International journal of cosmetic science. 2013;35:532–538. doi: 10.1111/ics.12090. [DOI] [PubMed] [Google Scholar]
- Severin FF, Hyman AA. Pheromone induces programmed cell death in S. cerevisiae. Curr Biol. 2002;12:R233–235. doi: 10.1016/s0960-9822(02)00776-5. [DOI] [PubMed] [Google Scholar]
- Sevini F, Giuliani C, Vianello D, Giampieri E, Santoro A, Biondi F, Garagnani P, Passarino G, Luiselli D, Capri M, Franceschi C, Salvioli S. mtDNA mutations in human aging and longevity: Controversies and new perspectives opened by high-throughput technologies. Exp Gerontol. 2014 doi: 10.1016/j.exger.2014.03.022. [DOI] [PubMed] [Google Scholar]
- Shalini S, Dorstyn L, Dawar S, Kumar S. Old, new and emerging functions of caspases. Cell Death Differ. 2015;22:526–539. doi: 10.1038/cdd.2014.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw AC, Joshi S, Greenwood H, Panda A, Lord JM. Aging of the innate immune system. Curr Opin Immunol. 2010;22:507–513. doi: 10.1016/j.coi.2010.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen J, Curtis C, Tavare S, Tower J. A screen of apoptosis and senescence regulatory genes for life span effects when over-expressed in Drosophila. Aging (Albany NY) 2009;1:191–211. doi: 10.18632/aging.100018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen J, Tower J. Programmed cell death and apoptosis in aging and life span regulation. Discov Med. 2009;8:223–226. [PubMed] [Google Scholar]
- Silva RD, Sotoca R, Johansson B, Ludovico P, Sansonetty F, Silva MT, Peinado JM, Corte-Real M. Hyperosmotic stress induces metacaspase- and mitochondria-dependent apoptosis in Saccharomyces cerevisiae. Mol Microbiol. 2005;58:824–834. doi: 10.1111/j.1365-2958.2005.04868.x. [DOI] [PubMed] [Google Scholar]
- Singh SR, Liu W, Hou SX. The adult Drosophila malpighian tubules are maintained by multipotent stem cells. Cell Stem Cell. 2007;1:191–203. doi: 10.1016/j.stem.2007.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha M, Jang YC, Oh J, Khong D, Wu EY, Manohar R, Miller C, Regalado SG, Loffredo FS, Pancoast JR, Hirshman MF, Lebowitz J, Shadrach JL, Cerletti M, Kim MJ, Serwold T, Goodyear LJ, Rosner B, Lee RT, Wagers AJ. Restoring systemic GDF11 levels reverses age-related dysfunction in mouse skeletal muscle. Science. 2014;344:649–652. doi: 10.1126/science.1251152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snigdha S, Smith ED, Prieto GA, Cotman CW. Caspase-3 activation as a bifurcation point between plasticity and cell death. Neuroscience bulletin. 2012;28:14–24. doi: 10.1007/s12264-012-1057-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song W, Kwak HB, Lawler JM. Exercise training attenuates age-induced changes in apoptotic signaling in rat skeletal muscle. Antioxid Redox Signal. 2006;8:517–528. doi: 10.1089/ars.2006.8.517. [DOI] [PubMed] [Google Scholar]
- Sperandio S, Poksay K, de Belle I, Lafuente MJ, Liu B, Nasir J, Bredesen DE. Paraptosis: mediation by MAP kinases and inhibition by AIP-1/Alix. Cell Death Differ. 2004;11:1066–1075. doi: 10.1038/sj.cdd.4401465. [DOI] [PubMed] [Google Scholar]
- Spillare EA, Robles AI, Wang XW, Shen JC, Yu CE, Schellenberg GD, Harris CC. p53-mediated apoptosis is attenuated in Werner syndrome cells. Genes Dev. 1999;13:1355–1360. doi: 10.1101/gad.13.11.1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stegh AH. Targeting the p53 signaling pathway in cancer therapy - the promises, challenges and perils. Expert opinion on therapeutic targets. 2012;16:67–83. doi: 10.1517/14728222.2011.643299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suh Y. Cell signaling in aging and apoptosis. Mech Ageing Dev. 2002;123:881–890. doi: 10.1016/s0047-6374(02)00025-8. [DOI] [PubMed] [Google Scholar]
- Suh Y, Lee KA, Kim WH, Han BG, Vijg J, Park SC. Aging alters the apoptotic response to genotoxic stress. Nat Med. 2002;8:3–4. doi: 10.1038/nm0102-3. [DOI] [PubMed] [Google Scholar]
- Taddei ML, Giannoni E, Fiaschi T, Chiarugi P. Anoikis: an emerging hallmark in health and diseases. J Pathol. 2012;226:380–393. doi: 10.1002/path.3000. [DOI] [PubMed] [Google Scholar]
- Tarnawski AS, Ahluwalia A, Jones MK. Increased susceptibility of aging gastric mucosa to injury: the mechanisms and clinical implications. World journal of gastroenterology : WJG. 2014;20:4467–4482. doi: 10.3748/wjg.v20.i16.4467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor RC, Cullen SP, Martin SJ. Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol. 2008;9:231–241. doi: 10.1038/nrm2312. [DOI] [PubMed] [Google Scholar]
- Tchkonia T, Zhu Y, van Deursen J, Campisi J, Kirkland JL. Cellular senescence and the senescent secretory phenotype: therapeutic opportunities. J Clin Invest. 2013;123:966–972. doi: 10.1172/JCI64098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tower J. Sex-specific regulation of aging and apoptosis. Mech Ageing Dev. 2006;127:705–718. doi: 10.1016/j.mad.2006.05.001. [DOI] [PubMed] [Google Scholar]
- Tower J. Mitochondrial maintenance failure in aging and role of sexual dimorphism. Archives of biochemistry and biophysics. 2014 Oct 25; doi: 10.1016/j.abb.2014.10.008. pii: S0003-9861 (14)00375-0. doi: 10.1016/j.abb.2014.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valente LJ, Gray DH, Michalak EM, Pinon-Hofbauer J, Egle A, Scott CL, Janic A, Strasser A. p53 efficiently suppresses tumor development in the complete absence of its cell-cycle inhibitory and proapoptotic effectors p21, Puma, and Noxa. Cell reports. 2013;3:1339–1345. doi: 10.1016/j.celrep.2013.04.012. [DOI] [PubMed] [Google Scholar]
- Vasconsuelo A, Pronsato L, Ronda AC, Boland R, Milanesi L. Role of 17beta-estradiol and testosterone in apoptosis. Steroids. 2011;76:1223–1231. doi: 10.1016/j.steroids.2011.08.001. [DOI] [PubMed] [Google Scholar]
- Vaughn AE, Deshmukh M. Glucose metabolism inhibits apoptosis in neurons and cancer cells by redox inactivation of cytochrome c. Nat Cell Biol. 2008;10:1477–1483. doi: 10.1038/ncb1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vazquez A, Bond EE, Levine AJ, Bond GL. The genetics of the p53 pathway, apoptosis and cancer therapy. Nature reviews. Drug discovery. 2008;7:979–987. doi: 10.1038/nrd2656. [DOI] [PubMed] [Google Scholar]
- Vazquez-Padron RI, Lasko D, Li S, Louis L, Pestana IA, Pang M, Liotta C, Fornoni A, Aitouche A, Pham SM. Aging exacerbates neointimal formation, and increases proliferation and reduces susceptibility to apoptosis of vascular smooth muscle cells in mice. Journal of vascular surgery. 2004;40:1199–1207. doi: 10.1016/j.jvs.2004.08.034. [DOI] [PubMed] [Google Scholar]
- Wallace DC. Mitochondrial DNA mutations in disease and aging. Environmental and molecular mutagenesis. 2010;51:440–450. doi: 10.1002/em.20586. [DOI] [PubMed] [Google Scholar]
- Walter D, Wissing S, Madeo F, Fahrenkrog B. The inhibitor-of-apoptosis protein Bir1p protects against apoptosis in S. cerevisiae and is a substrate for the yeast homologue of Omi/HtrA2. J Cell Sci. 2006;119:1843–1851. doi: 10.1242/jcs.02902. [DOI] [PubMed] [Google Scholar]
- Wanagat J, Cao Z, Pathare P, Aiken JM. Mitochondrial DNA deletion mutations colocalize with segmental electron transport system abnormalities, muscle fiber atrophy, fiber splitting, and oxidative damage in sarcopenia. FASEB J. 2001;15:322–332. doi: 10.1096/fj.00-0320com. [DOI] [PubMed] [Google Scholar]
- Wang E. Senescent human fibroblasts resist programmed cell death, and failure to suppress bcl2 is involved. Cancer research. 1995;55:2284–2292. [PubMed] [Google Scholar]
- Wang E. Regulation of apoptosis resistance and ontogeny of age-dependent diseases. Exp Gerontol. 1997;32:471–484. doi: 10.1016/s0531-5565(96)00156-8. [DOI] [PubMed] [Google Scholar]
- Wang X, Bonventre JV, Parrish AR. The Aging Kidney: Increased Susceptibility to Nephrotoxicity. International journal of molecular sciences. 2014;15:15358–15376. doi: 10.3390/ijms150915358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Yang C, Chai J, Shi Y, Xue D. Mechanisms of AIF-mediated apoptotic DNA degradation in Caenorhabditis elegans. Science. 2002;298:1587–1592. doi: 10.1126/science.1076194. [DOI] [PubMed] [Google Scholar]
- Warner HR. Aging and regulation of apoptosis. Current topics in cellular regulation. 1997;35:107–121. doi: 10.1016/s0070-2137(97)80004-0. [DOI] [PubMed] [Google Scholar]
- Wei M, Fabrizio P, Hu J, Ge H, Cheng C, Li L, Longo VD. Life span extension by calorie restriction depends on Rim15 and transcription factors downstream of Ras/PKA, Tor, and Sch9. PLoS Genet. 2008;4:e13. doi: 10.1371/journal.pgen.0040013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westphal D, Kluck RM, Dewson G. Building blocks of the apoptotic pore: how Bax and Bak are activated and oligomerize during apoptosis. Cell Death Differ. 2014;21:196–205. doi: 10.1038/cdd.2013.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson A, Shehadeh LA, Yu H, Webster KA. Age-related molecular genetic changes of murine bone marrow mesenchymal stem cells. BMC Genomics. 2010;11:229. doi: 10.1186/1471-2164-11-229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wissing S, Ludovico P, Herker E, Buttner S, Engelhardt SM, Decker T, Link A, Proksch A, Rodrigues F, Corte-Real M, Frohlich KU, Manns J, Cande C, Sigrist SJ, Kroemer G, Madeo F. An AIF orthologue regulates apoptosis in yeast. J Cell Biol. 2004;166:969–974. doi: 10.1083/jcb.200404138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yakovlev AG, Faden AI. Mechanisms of neural cell death: implications for development of neuroprotective treatment strategies. NeuroRx : the journal of the American Society for Experimental NeuroTherapeutics. 2004;1:5–16. doi: 10.1602/neurorx.1.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yee C, Yang W, Hekimi S. The intrinsic apoptosis pathway mediates the pro-longevity response to mitochondrial ROS in C. elegans. Cell. 2014;157:897–909. doi: 10.1016/j.cell.2014.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeo EJ, Hwang YC, Kang CM, Choy HE, Park SC. Reduction of UV-induced cell death in the human senescent fibroblasts. Mol Cells. 2000;10:415–422. [PubMed] [Google Scholar]
- Zhao J, Lu Y, Shen HM. Targeting p53 as a therapeutic strategy in sensitizing TRAIL-induced apoptosis in cancer cells. Cancer letters. 2012;314:8–23. doi: 10.1016/j.canlet.2011.09.040. [DOI] [PubMed] [Google Scholar]
- Zheng J, Edelman SW, Tharmarajah G, Walker DW, Pletcher SD, Seroude L. Differential patterns of apoptosis in response to aging in Drosophila. Proc Natl Acad Sci U S A. 2005;102:12083–12088. doi: 10.1073/pnas.0503374102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong X, Rescorla FJ. Cell surface adhesion molecules and adhesion-initiated signaling: understanding of anoikis resistance mechanisms and therapeutic opportunities. Cellular signalling. 2012;24:393–401. doi: 10.1016/j.cellsig.2011.10.005. [DOI] [PubMed] [Google Scholar]
- Zhu C, Wang X, Deinum J, Huang Z, Gao J, Modjtahedi N, Neagu MR, Nilsson M, Eriksson PS, Hagberg H, Luban J, Kroemer G, Blomgren K. Cyclophilin A participates in the nuclear translocation of apoptosis-inducing factor in neurons after cerebral hypoxia-ischemia. J Exp Med. 2007;204:1741–1748. doi: 10.1084/jem.20070193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Tchkonia T, Pirtskhalava T, Gower A, Ding H, Giorgadze N, Palmer AK, Ikeno Y, Borden G, Lenburg M, O'Hara SP, LaRusso NF, Miller JD, Roos CM, Verzosa GC, LeBrasseur NK, Wren JD, Farr JN, Khosla S, Stout MB, McGowan SJ, Fuhrmann-Stroissnigg H, Gurkar AU, Zhao J, Colangelo D, Dorronsoro A, Ling YY, Barghouthy AS, Navarro DC, Sano T, Robbins PD, Niedernhofer LJ, Kirkland JL. The Achilles' Heel of Senescent Cells: From Transcriptome to Senolytic Drugs. Aging Cell. 2015 Mar 9; doi: 10.1111/acel.12344. doi: 10.1111/acel.12344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuckerman V, Wolyniec K, Sionov RV, Haupt S, Haupt Y. Tumour suppression by p53: the importance of apoptosis and cellular senescence. J Pathol. 2009;219:3–15. doi: 10.1002/path.2584. [DOI] [PubMed] [Google Scholar]