Background: HCMV gH/gL/pUL128–131 pentameric complex is a main target for potent neutralizing antibody responses.
Results: Soluble gH/gL/pUL128–131 inhibits viral entry and presents neutralizing epitopes.
Conclusion: The biological activities of gH/gL/pUL128–131 confirm the critical role of the native complex as an immunogen.
Significance: The functional differences between gH/gL and gH/gL/pUL128–131 must be considered when designing vaccines and assessing immune responses.
Keywords: glycoprotein, protein complex, protein purification, vaccine, virus entry, HCMV, gH/gL/pUL128–131, neutralizing epitope, pentameric complex
Abstract
Congenital infection of human cytomegalovirus (HCMV) is one of the leading causes of nongenetic birth defects, and development of a prophylactic vaccine against HCMV is of high priority for public health. The gH/gL/pUL128–131 pentameric complex mediates HCMV entry into endothelial and epithelial cells, and it is a major target for neutralizing antibody responses. To better understand the mechanism by which antibodies interact with the epitopes of the gH/gL/pUL128–131 pentameric complex resulting in viral neutralization, we expressed and purified soluble gH/gL/pUL128–131 pentameric complex and gH/gL from Chinese hamster ovary cells to >95% purity. The soluble gH/gL, which exists predominantly as (gH/gL)2 homodimer with a molecular mass of 220 kDa in solution, has a stoichiometry of 1:1 and a pI of 6.0–6.5. The pentameric complex has a molecular mass of 160 kDa, a stoichiometry of 1:1:1:1:1, and a pI of 7.4–8.1. The soluble pentameric complex, but not gH/gL, adsorbs 76% of neutralizing activities in HCMV human hyperimmune globulin, consistent with earlier reports that the most potent neutralizing epitopes for blocking epithelial infection are unique to the pentameric complex. Functionally, the soluble pentameric complex, but not gH/gL, blocks viral entry to epithelial cells in culture. Our results highlight the importance of the gH/gL/pUL128–131 pentameric complex in HCMV vaccine design and emphasize the necessity to monitor the integrity of the pentameric complex during the vaccine manufacturing process.
Introduction
Human cytomegalovirus (HCMV),2 a prototype β-herpesvirus, is ubiquitous in human populations (1). HCMV infection in healthy subjects with intact immune systems rarely causes any symptoms but results in lifelong persistent or latent infection. However, in immunosuppressed individuals, such as those under immunosuppression regimens following solid organ or hematopoietic stem cell transplantation, HCMV infection can lead to fatal organ-specific HCMV diseases including hepatitis, pneumonitis, and enterocolitis. Another significant unmet medical need is that HCMV infection in women during pregnancy may result in viral transmission to the fetus, leading to developmental and neurological abnormalities in newborns with lifelong disabilities. Meta-analysis reveals that an estimated 0.64% (95% confidence interval: 0.60–0.69%) of birth cohort each year in the United States is born with congenital HCMV infection, leading to over 5000 children with sequelae of congenital HCMV infection (2). Thus, developing a prophylactic vaccine for prevention of congenital HCMV infection has been designated to the highest category on the list of vaccine priorities by the Institute of Medicine in the United States (3, 4).
HCMV infects a wide range of cell types in vivo, and its entry is mediated by four major glycoprotein complexes including gB, gM/gN, gH/gL/gO, and gH/gL/pUL128–131 (5). Based on HSV-1 virus entry studies (6, 7), HCMV viral fusion machinery is likely composed of gB and gH/gL complex, but the role of gO and pUL128–131 in the entry process is unknown. Recently, a gO-null virus was shown to be infectious in culture, raising the possibility that gH/gL might also exist on mature infectious virions (8, 9). The gH/gL/pUL128–131 pentameric complex is the viral determinant required for HCMV infection in epithelial and endothelial cells and leukocytes (10–12) and is likely to interact with receptors on the cell surface (13). During natural infection, antibodies binding to the pUL128–131 components are generally potent neutralizers (14, 15). These antibodies may neutralize HCMV infection by multiple distinct mechanisms that are yet to be determined.
We recently described a vaccine candidate based on AD169 strain with restored expression of gH/gL/pUL128–131 complex (16). AD169 strain was developed previously as an attenuated virus vaccine but failed to effectively induce neutralizing antibodies in clinical evaluations (17, 18). Comparative immunogenicity evaluations demonstrated that restoring expression of gH/gL/pUL128–131 complex is associated with significantly improved neutralizing titers against viral epithelial entry, and the resulting neutralizing antibodies target the pentameric complex similarly to those found in natural immune human sera (16, 19). However, it remains to be determined what subcomponents of the pentameric complex are important for the neutralizing activities induced by either vaccination or natural HCMV infection. To address these questions directly, we expressed and purified soluble gH/gL and gH/gL/pUL128–131 pentameric complex. Detailed biochemical and immunological analyses confirmed that these complexes are folded in their native conformation. We further explored the functions of these soluble complexes for their abilities to directly block viral entry to epithelial cells, as well as their relative abilities to adsorb neutralizing antibodies against viral epithelial entry. The results indicate that the soluble pentameric complex, but not gH/gL, contains domains important for HCMV viral entry into epithelial cells and native epitopes responsible for the majority of the neutralizing activity in HCMV hyperimmune globulin (CMV-HIG). The implication of these results and the utility of these soluble gH complexes for vaccine development are discussed.
Experimental Procedures
Expression of gH/gL and gH/gL/pUL128–131 Pentameric Complex
Two vectors were constructed, with vector pGS-gH-gLtwn containing the independent expression cassettes for gH and gL of Towne strain and vector pGS-UL128-UL-130–131 containing the independent expression cassettes for UL128, UL130, and UL131 from Merlin strain. The gH open reading frame was modified with a truncation of its cytoplasmic and transmembrane domains for production of secreted gH/gL complex. In addition, a Strep tag and a His tag were fused to the C terminus of gH via a flexible linker. Each vector contained glutamine synthetase (GS) selection markers. The coding sequence of each of the complex genes was optimized for mammalian expression. The host CHOK1-SV cell line (Lonza) was either transfected with the vector pGS-gH-gLtwn for production of soluble gH/gL or both vectors for pentameric complex. The cells were subsequently selected with methionine-sulfoximine in medium lacking glutamine per the manufacturer's instructions. Single clones were obtained by cell sorting using a flow cytometer. The positive clones were identified by capturing ELISA (see below), and high producing clones were subsequently expanded and seeded into cell culture flasks to confirm cell growth and productivity. Research cell banks were generated and subsequently used for fed batch cultures to generate materials for purification.
Purification of gH/gL and gH/gL/pUL128–131 Pentameric Complex
For both gH/gL and gH/gL/pUL128–131 pentameric complex purification, the clarified cell culture supernatant was loaded onto a HisTrapFF crude column (GE Healthcare) in 50 mm HEPES, pH 7.5, 100 mm NaCl, 5 mm imidazole, and the gH complexes were eluted with a gradient of 25–500 mm imidazole. The semipurified complex was then loaded onto a Strep Tactin column (StrepTrap-HP; GE Healthcare) in 50 mm HEPES, 100 mm NaCl, pH 7.3. Proteins were eluted in a 1 column volume gradient of 0–100% elution buffer containing 2.5 mm d-desthiobiotin. The gH/gL-containing fractions were loaded onto HiLoad 16/60 Superdex 200 (GE Healthcare), eluted in 25 mm HEPES, 150 mm NaCl, pH 7.5, and concentrated with an Amicon Ultra centrifugal 30K filter (EMD Millipore) to 1.0 mg/ml. For the purification of the pentameric complex, the Strep Tactin product was diluted with 50 mm HEPES to decrease the concentration of NaCl. The diluted sample was loaded onto a 10-ml HiTrap SP HP column (GE Healthcare). The column was washed and eluted in 15 column volume in a gradient of 0–100% mobile phase B containing 50 mm HEPES, 1 m NaCl, pH 7.3. Protein containing fractions were pooled and diluted with 50 mm HEPES to contain ∼150 mm NaCl then concentrated to 0.63 mg/ml. All samples were immediately aliquoted and stored at −70 °C.
The concentrated samples were tested by cation exchange chromatography (CEX) using a 1-ml HiTrap SP HP column to confirm purity. Equal amounts of gH/gL and pentameric complex were injected on an Agilent 1200 series HPLC equipped with a diode array detector set to 280 nm. The mobile phases were identical to the CEX purification. The LC gradient was performed as follows (min/% of mobile phase B): 0.0/0, 10/0, 40/100, and 45/100. The HPLC was then re-equilibrated to initial conditions for 10 min. The flow rate was kept at 1 ml/min.
Reducing and Nonreducing SDS-PAGE and Deglycosylation
SDS-PAGE was performed using 4–20% precast Novex Tris-glycine gels. Samples were mixed with Laemmli sample buffer. Reduced samples contained 100 mm DTT and nonreduced samples contained 25 mm iodoacetamide. The samples were heated at 70 °C for 10 min and electrophoresed. Gels were fixed with 12% trichloroacetic acid, rinsed with water, stained with Pierce GelCode Stain, and subsequently destained. Gels were scanned using Molecular Dynamic Personal Densitometer SI and imaged using Image Quant software (GE Healthcare). Samples were deglycosylated by adding 1% SDS (w/v) and 13.5 mm 2-mercaptoethanol. The samples were heated at 37 °C for 10 min before adding 0.5% of NP-40 (w/v) and 5 units of proteomic grade PNGase F (Sigma-Aldrich). The samples were incubated at 25 °C overnight. Sialic acid residues were removed with 2 units of α (2–3,6,8,9)-neuraminidase according to the manufacturer's instructions (Sigma-Aldrich).
In-gel Digestion and Protein Identification by LC-MS/MS
In-gel digestion of each protein band was performed as described previously (20), and the extracted peptides were analyzed by nano-LC-MS/MS with a Waters nanoAcquity coupled to a Thermo LTQ-XL. A Waters BEH C18 column (75 μm × 100 mm) with 1.7-μm particles was used for separation with a New Objective PicoTip emitter (Woburn, MA). A Waters nanoAcquity trap column (180 μm × 20 mm; Symmetry C18, Waters) was used for desalting. A 5-min desalting step at 15 μl/min with 0.1% formic acid (Pierce), 0.02% TFA (Pierce), and 1% acetonitrile in water was used after samples were loaded onto the HPLC. Next, peptides were eluted from the column with a linear gradient over 85 min at 300 nl/min to 0.1% formic acid, 0.02% TFA, and 65% acetonitrile in water. The spray voltage was 2.5 kV. The capillary temperature was 220 °C. The capillary voltage was 47 V, and the tube lens was 155 V. A total of 5 μl of digest was analyzed for each band. Data-dependent analysis was performed using a single full MS scan from 300 to 2000 m/z followed by MS/MS analysis of the five most intense ions detected in the full scan. Dynamic exclusion was applied after two MS/MS scans of the same ion.
LC-MS/MS data were searched in Proteome Discover (version 1.4, Thermo) against a custom CMV database using Sequest HT. Searches were performed using a full trypsin restriction with up to two missed cleavage sites. The precursor mass tolerance was set to 1.5 Da, and the fragment mass tolerance was 0.8 Da. Spectra were matched based on b and y ions. Carbamidomethyl modification on cysteine was the only modification used.
Sandwich ELISA
Rabbit monoclonal antibodies targeting the pentameric complex (15) were immobilized at 2 μg/ml in PBS on a 96-well FluoroNunc MaxiSorp plate at 4 °C overnight. The plate was then washed with PBS, 0.05% Tween 20 (PBST-20) and blocked with 3% nonfat milk in PBST-20 for 1 h. Cell culture supernatant or purified solution was added to the wells in titration and incubated for 2 h. The captured complex was detected with anti-His Tag HRP and visualized with a fluorogenic HRP substrate, 10-acetyl-3,7-dihydroxyphenoxazine (Virolabs, Chantilly, VA). Fluorescent signals with excitation at 531 nm and emission at 595 nm were measured with a plate reader (Victor III, PerkinElmer Life Sciences).
Capillary Isoelectric Focusing Immunoassay (NanoPro)
Protein samples of gH/gL and gH/gL/pUL128–131 pentameric complex were analyzed on the ProteinSimple Peggy system (NanoPro) according to manufacturer's instructions with minor modification as previously described (21). Briefly, samples were diluted to the same concentration in Bicine CHAPS buffer (ProteinSimple). The samples were then mixed with ampholytes premix, pH 3–10, and fluorescence labeled pI standards. Primary antibodies, rabbit mAb 57.4 and 124.4, were diluted 1:100 in antibody diluent (ProteinSimple). The secondary anti-rabbit HRP-conjugated antibody was from ProteinSimple and diluted 1:100 in Antibody Diluent. Luminol and peroxide XDR was mixed at a 1:1 ratio. The automated separation and detection were performed using the default charge separation settings. The capillaries were imaged and analyzed with Compass software (ProteinSimple).
Size Exclusion Chromatography with Multiangle Light Scattering Detection
Size exclusion chromatography was performed on an Agilent 1200 series HPLC equipped with a variable wavelength detector set to 280 nm, a Wyatt Optilab rEX refractive index detector, and a Wyatt DAWN HELEOS-II multiangle light scattering (MALS) detector. Concentrations of gH/gL and pentameric complex samples were determined using the extinction coefficients 0.907 and 1.093 mg−1·ml·cm−1, respectively. Light scattering data were processed using the protein conjugate template in Wyatt Astra software. The dn/dc values were 0.185 and 0.133 ml/g for protein and glycan, respectively. Chromatographic separations were completed using a Phenomenex BioSep SEC s4000 (7.8 × 300 mm) and Tosoh TSKgel G3000SWxl (7.8 × 300 mm) connected in series. The column temperature was 30 °C. The flow rate was maintained at 0.5 ml/min under isocratic conditions during a complete run time of 65 min. The mobile phase was 25 mm sodium phosphate buffer, 150 mm NaCl, 0.05% NaN3 (w/v), pH 7.3. The gH/gL and gH/gL/pUL128–131 pentameric complex samples were injected at equal concentrations and were analyzed for absolute mass.
Reversed Phase (RP)-HPLC
The RP-HPLC separation was performed on an Agilent 1200 Series with diode array detector set to 280 nm. Prior to injection, the samples were treated with 1% SDS for nonreducing conditions or 1% SDS, 100 mm DTT for reducing conditions. The analytical column was an Applied Biosystems Poros R2, 2.1 mm × 30 mm with a 10-μm particle size. The mobile phases were 0.05% TFA in water (mobile phase A) and 0.1% TFA in acetonitrile (mobile phase B). The separation gradient was performed as follows (min/% of mobile phase B): 0/20, 10/40, 11/40, 12/50, 13/50, 13.1/55, and 17/55. The HPLC column was then re-equilibrated to initial conditions for 4.5 min. The flow rate was kept at 2 ml/min, and the column temperature was maintained at 75 °C.
Negative Staining EM
Samples were applied to a layer of continuous carbon supported by nitrocellulose on a 400-mesh copper grid and immediately stained with uranyl formate. Electron microscopy was performed using an FEI Tecnai T12 electron microscope, operating at 120 keV equipped with an FEI Eagle 4k × 4k CCD camera. Image sets were acquired over a magnification range of 52,000–110,000× with nominal defocus from −1 μm and −3 μm and doses between 24–45 e/Å2. Individual particles in the 67,000× magnification images were selected using automated picking protocols (22). A reference-free alignment strategy based on the XMIPP processing package was used to classify the selected particles into self-similar groups (23).
Inhibition of Viral Entry into Epithelial Cells
ARPE-19 cells were seeded in DMEM/F12 at a density of 1.5 × 104 cells/well and incubated at 37 °C overnight. The soluble gH/gL, soluble gH/gL/pUL128–131 pentameric complex, CMV-HIG (Cytogam, CSL Behring), and negative control IgG were 2-fold serially diluted with DMEM/F12 complete medium in 96-well plates. Each dilution was preincubated with ∼100 pfu pentameric complex restored AD169 strain at room temperature for 1 h. The mixtures were then transferred to the ARPE-19 cell plate and cultured at 37 °C, 5% CO2 for 20 h. The highest concentrations tested for each reagent were: CMV-HIG 195 μg/ml, pentameric complex 158 μg/ml, gH/gL 93 μg/ml, and negative control IgG 166 μg/ml.
At the end of incubation, cells were fixed with methanol, blocked with 3% nonfat milk in PBST-20, and stained with rabbit anti-IE2 sera and Vactastain ABC kit. The HCMV infection foci were counted with ImagePro (Media Cybernetics). Inhibition percentage for each well was calculated with the following formula: inhibition percentage = (1 − foci counts in test well/foci counts in virus only control well) × 100%. Inhibition curves were constructed for each tested reagent. Four-parameter logistic curve fitting was used to extract IC50 values for each reagent tested.
Depletion of Pentameric Complex or gH/gL Specific Antibodies from Anti-HCMV Hyperimmune Globulin
Purified soluble pentameric complex and gH/gL were coated on a 96-well MaxiSorp plate at 5 μg/ml in PBS for 1 h. The plate was blocked with 1% BSA in HEPES, 0.1% P188. CMV-HIG was diluted to 200 μg/ml in PBS, 0.5% BSA, and serially incubated (four times) with the coated plate for 1 h at room temperature for each incubation to deplete antibodies that bind to the complex. Mock depletion was performed by incubating CMV-HIG on a plate coated with PBS only and then blocked. The extent of depletion was monitored by direct ELISA using a MaxiSorp plate coated with the pentameric complex and blocked with HEPES, 1% BSA. The depleted CMV-HIG samples were serially diluted from the nominal 100 μg/ml to less than 0.1 μg/ml in 2-fold dilutions and incubated with the pentameric complex coated plate for 1 h at room temperature. The plate was washed and incubated with alkaline phosphatase-conjugated goat anti-human IgG (Jackson Immunoresearch). After washing, 4-methylumbelliferyl phosphate (Virolabs) liquid substrate was added, and fluorescence was detected after 1 h with excitation at 360 nm and emission at 465 nm.
ARPE-19 cells were seeded in DMEM/F12, 1% FBS, 1% penicillin/streptomycin in a 384-well Corning clear-bottomed black plate at 8000 cells/well and incubated for 24 h. CMV-HIG and depleted CMV-HIG were serially diluted from the nominal 50 μg/ml to less than 0.1 μg/ml in 2-fold dilutions and incubated with pentameric complex-restored AD169 virus at multiplicity of infection of 0.7 at room temperature for 1 h. The mixture was transferred to the ARPE-19 cell plate, and the cell plate was incubated for 21 h. The culture supernatant was then removed, and the cells were fixed with 3.7% formaldehyde in PBS for 30 min and permeabilized with 0.5% Triton X-100 in PBS for 20 min. The plate was stained with clone L-14 (mouse α-IE1 mAb; ATCC) and AlexaFluor 488-conjugated chicken anti-mouse IgG (Life Technologies) to fluorescently label infected cells. Cell DNAs were stained with Hoechst 33342 (Life Technologies). The plate was imaged by a BioTek Cytation3, and all nuclei were counted using Gen5.
Results
Expression and Purification of Soluble gH/gL and gH/gL/pUL128–131 Pentameric Complex
CHO cells transfected with pGS-gH-gLtwn vector produced soluble gH/gL in a fed-batch culture. Because gH was fused with a His tag and a Strep tag, immobilized metal affinity chromatography (IMAC) and Strep Tactin procedures were followed to purify gH/gL from the clarified supernatant with a purification yield of ∼5 mg/liter culture. The gH/gL purity was assessed as 98% by reducing SDS-PAGE (Fig. 1F). The gH and gL proteins were identified by in-gel digestion followed by LC-MS/MS.
FIGURE 1.
Purification of gH/gL and gH/gL/pUL128–131 pentameric complex (PC). A, UV trace of Strep Tactin chromatogram. B, UV trace of CEX chromatogram to purify pentameric complex. C, reinjection of gH/gL and pentameric complex on the HPLC CEX column. D, reducing SDS-PAGE of pentameric complex purification fractions. E, nonreducing SDS-PAGE of pentameric complex purification fractions. F, reducing SDS-PAGE of deglycosylated gH/gL and pentameric complex by PNGase and α(2–3,6,8,9)-neuraminidase (NM). Protein identification was confirmed by in-gel digestion followed by nano LC-MS/MS. For SDS-PAGE, samples were loaded at 10 μg/lane. IMAC, product from immobilized metal affinity chromatography; StrepFT, flowthrough from Strep Tactin column. Strep, product from Strep Tactin purification.
CHO cells transfected with both pGS-gH-gLtwn and pGS-UL128-UL-130–131 vectors produced both soluble gH/gL and gH/gL/pUL128–131 pentameric complex. Both complexes were enriched after IMAC and Strep Tactin chromatography because gH was His- and Strep-tagged (Fig. 1A). Since the sequence analysis predicted a pI of 7.5 for the pentameric complex and 6.2 for gH/gL, the two complexes could be separated based on their charge differences. Initially, we explored CEX using SP-Sepharose at pH 7.0. Although gH/gL immediately eluted from the column and was present in the flow through fraction (CEXFT) as expected, the pentameric complex bound strongly to the column and required 0.7 m NaCl to be eluted. To minimize potential damage to the pentameric complex in the high salt buffer, we increased the mobile phase pH to 7.3 and found that the pentameric complex eluted with 0.3 m NaCl (Fig. 1B). Eluted pentameric complex was diluted immediately with 50 mm HEPES, pH 7.3, to decrease the salt concentration as a precaution to stabilize noncovalently bound proteins in the pentameric complex. The purification yield for pentameric complex was ∼0.6 mg/liter culture, and the purity was 95%. The purified gH/gL and pentameric complex were reinjected onto an analytical scale CEX column, and their distinct retention properties were confirmed (Fig. 1C).
SDS-PAGE showed that although the CEXFT sample migrated as gH (one band) and gL (two bands) under the reducing condition (Fig. 1D), two protein bands (∼240 and ∼120 kDa) were observed under nonreducing condition (Fig. 1E). Both bands contained only gH and gL. This suggested: 1) the gH/gL complex did not bind to the CEX column at pH 7.3 (Fig. 1C), consistent with the prediction based on its theoretical pI of 6.2; 2) gH and gL were covalently bound in the gH/gL complex; and 3) a portion of gH/gL formed (gH/gL)2 homodimer through disulfide linkage. It was noted that the ratio of (gH/gL)2 homodimer to gH/gL monodimer on SDS-PAGE did not change with different sample preparation temperature or different alkylation reagents (data not shown). This suggested that the two populations of ∼240 and ∼120 kDa detected by SDS-PAGE were not caused by a disulfide scrambling artifact from sample preparation for SDS-PAGE.
The CEX eluted sample (pentameric complex) was confirmed to contain gH, gL, pUL128, pUL130, and pUL131 by LC-MS/MS analysis. In reducing SDS-PAGE, pUL130 co-migrated with gL and was detected as an unresolved band. The poor resolution was due to heterogeneous glycosylation on both pUL130 and gL. Upon deglycosylation by PNGase F, pUL130 migrated as a sharp band at ∼24 kDa, whereas the gL bands migrated as two with a minor shift in molecular mass (Fig. 1F). This shift difference is expected because gL has only one N-linked glycosylation site, whereas pUL130 has three. The higher gL band observed after deglycosylation by PNGase likely has sialic acid-containing O-glycans as it shifted toward the small molecular mass band after neuraminidase cleavage (Fig. 1F). pUL131 co-migrated with pUL128 with an apparent molecular mass of 17 kDa. Upon deglycosylation with PNGase F, pUL131 migrated to the expected molecular mass of 13 kDa, whereas the observed molecular mass of pUL128 did not change. Again this is expected because pUL131 contains one N-linked glycosylation site, whereas pUL128 has none. In nonreducing SDS-PAGE, gH, gL, and pUL128 co-migrated as a ∼140 kDa complex, suggesting that gH-gL-pUL128 were covalently linked through disulfide bonds, whereas the other two proteins pUL130 and pUL131 are noncovalently associated.
Binding Activities of gH/gL/pUL128–131 and gH/gL Complexes Monitored during Purification
To confirm the enrichment of the pentameric complex during the purification process, we tested the binding of each sample to gH/gL specific mAb 124.4 and pentameric complex specific mAb 57.4 by ELISA (Fig. 2). In Fig. 2A, all samples except StrepFT bound to mAb 124.4, suggesting that the Strep Tactin column removed impurities. In Fig. 2B, the CEX sample showed a strong binding to mAb 57.4, whereas CEXFT bound poorly, suggesting that pentameric complex was enriched in CEX. For each sample, the fold difference of binding activity compared with the IMAC sample was calculated and summarized in Fig. 2C. The Strep sample had slightly increased binding activities toward both mAb 124.4 and 57.4, consistent with its increased purity. The CEXFT sample exhibited more than 4-fold decrease in binding to mAb 57.4, whereas the CEX sample had a 6-fold increase in binding. This result clearly indicated that gH/gL was enriched in the CEXFT sample, and the pentameric complex was enriched in the CEX sample. The low binding activity of the CEXFT sample toward mAb 57.4 was likely caused by the presence of residual amounts of pentameric complex in this sample fraction.
FIGURE 2.

Binding activity of purification samples to gH/gL specific mAb 124.4 and pentameric complex specific mAb 57.4. A, dose-response curve of purification samples binding to mAb 124.4. B, dose-response curve of purification samples binding to mAb 57.4. In the Sandwich ELISA assay, either mAb 124.4 or 57.4 was immobilized on the plates at a fix concentration of 2 μg/ml, and a serial dilution of samples were tested for binding. C, relative binding activities compared with sample IMAC. Pentameric complex is enriched in the CEX by 6-fold. Low binding activity of CEXFT sample toward mAb 57.4 suggests a low level of pentameric complex is remaining in the CEXFT.
Soluble gH/gL and gH/gL/pUL128–131 Pentameric Complex Retain Native Neutralizing Epitopes Comparable with the Membrane-bound Forms
We previously isolated 11 elite neutralizing mAbs from a rabbit vaccinated with pentameric complex restored AD169 (revertant virus), and seven of them recognized the pentameric complex (15). The seven mAbs were screened against the soluble gH/gL and gH/gL/pUL128–131 and distinguished into two categories. Category 1 mAbs 57.4 and 276.1 recognized the soluble pentameric complex, but not gH/gL (Fig. 3, A and C), consistent with their binding to the AD169 revertant virus only. Category 2 mAbs 124.4, 316.2, 270.7, 70.7, and 324.4, recognized both soluble gH/gL and the pentameric complex, consistent with their binding to both AD169 parental and revertant virus (Fig. 3, B and C). The binding pattern of each mAb toward the soluble complexes resembles its binding to the parental and revertant virus, suggesting that the soluble gH/gL and pentameric complexes retain native neutralizing epitopes comparable with the membrane-bound forms.
FIGURE 3.

Binding activities of seven elite neutralizing antibodies to the soluble gH/gL and pentameric complex. The elite neutralizing mAbs were isolated from a rabbit vaccinated with the pentameric complex restored AD169 (15). Among the seven elite neutralizers, two bind to the pentameric complex only (A and C) consistent with their binding to the AD169 revertant virus (category 1), and five bind to both gH/gL and the pentameric complex (B and C) consistent with their binding to both AD169 parental and revertant virus (category 2).
Biophysical Characterization of gH/gL and gH/gL/pUL128–131 Pentameric Complex
The charge heterogeneities of gH/gL and the pentameric complex were evaluated by NanoPro technology. Both complexes were separated based on their pI in a capillary containing a pH gradient. After separation, proteins were immobilized to the capillary wall and detected with primary and HRP-conjugated secondary antibodies. Both gH/gL and pentameric complex appeared as broad peaks, suggesting that both were heterogeneous in charge, a common phenomenon for CHO expressed heavily glycosylated macromolecules (24, 25). As expected, gH/gL migrated between pH 6.0–6.5 consistent with its theoretical pI of 6.2. It was recognized only by gH/gL specific mAb 124.4 and not pentameric complex specific mAb 57.4 (Fig. 4A). The pentameric complex migrated between pH 7.4 to 8.1, consistent with its theoretical pI of 7.5. The pentameric complex was recognized by both mAb 124.4 and mAb 57.4. Because the CEX sample did not have peaks migrating between pH 6.0 and 6.5, we concluded that CEX sample contained pure pentameric complex.
FIGURE 4.

Biophysical characterization of the soluble gH/gL and gH/gL/pUL128–131 PC. A, charge heterogeneity of gH/gL and PC by imaged capillary isoelectric focusing (NanoPro Technology). Electropherograms show that gH/gL is detected by mAb 124.4 only and PC is detected by both mAb 124.4 and mAb 57.4. B, size exclusion chromatogram of gH/gL (solid line) and PC (dashed line) measured at UV 280 nm. The calculated molecular mass is shown as dots above each peak. The gH/gL complex has a total molecular mass of 250 kDa, and the PC has a total molecular mass of 180 kDa. The dotted line shows a PC purchased from a reagent vendor. It reveals co-purified (gH/gL)2 homodimer present in the PC preparation. C, two-dimensional class average calculated using particles selected from the EM images of gH/gL overlaid with the coordinates of the HSV-2 gH/gL structure (Protein Data Bank code 3M1C) is on the left, and the two-dimensional class average of PC is on the right.
The absolute molecular masses of gH/gL and pentameric complex were analyzed under native conditions using size exclusion HPLC coupled with MALS detection (Fig. 4B). The pentameric complex eluted ∼32.5 min with a measured total molecular mass of 180 kDa. Protein contributed 160 kDa, consistent with the expected molecular mass. The gH/gL eluted ∼29.5 min with a measured molecular mass of 250 kDa. Protein contributed 220 kDa, and glycans contributed 30 kDa. Because the theoretical molecular mass of a monomeric gH/gL is 110 kDa, the measured molecular mass, as well as the retention time, indicated that gH/gL existed predominantly as (gH/gL)2 homodimer under native conditions. The homodimer formed through both covalent and noncovalent interactions as shown by nonreducing SDS-PAGE (Fig. 1E).
Negative staining EM confirmed that gH/gL existed primarily as (gH/gL)2 homodimer, and monomeric gH/gL was observed only in small quantity. The (gH/gL)2 homodimer was ∼22 nm long with a symmetrical arrangement of four lobes. Overlaying the crystal structure of HSV-2 gH/gL complex (Protein Data Bank code 3M1C) (26) onto the HCMV gH/gL image suggests that the two gH/gL are joined at the N termini of gH (Fig. 4C, left panel). EM also revealed that gH/gL/pUL128–131 pentameric complex was ∼15 nm long and 4–5 nm in width and had a characteristically curved morphology different from the homodimer (Fig. 4C, right panel). In all of the obtained two-dimensional class averages, the pentameric complex had a lobe with tenuous connection to the rest of the structure. Additional imaging studies are being performed to better understand the binding of different neutralizing antibodies to the pentameric complex.
Stoichiometry of gH/gL/pUL128–131 Pentameric Complex
Although the gH/gL/pUL128–131 pentameric complex is known to contain five proteins, there is no report so far of the stoichiometry of the five components. Traditional methods to quantitate protein stoichiometry involve protein cross-linking and molecular mass analysis. However, the small size of pUL128 and pUL131 has made this analysis impossible (27). LC-MS quantitation requires purified proteins or peptides as standards. N-terminal sequencing is challenging for a five-protein complex, and it becomes impossible if the N-terminal amino acid is blocked with modification such as pyroglutamate. RP-HPLC can be quantitative if baseline separation and good mass recovery can be achieved (20). We separated the five proteins using RP-HPLC (Fig. 5A) and achieved a mass recovery of 87%. Protein peaks were collected, analyzed on reducing SDS-PAGE, and identified by in-gel digestion followed by nano LC-MS/MS (Fig. 5B). Based on the UV absorbance at 280 nm and the theoretical extinction coefficient of each protein, we confirmed the molar ratio of gH:gL to be 1:1 in the gH/gL complex and gH:gL:pUL128:pUL130:pUL131 to be 1:1:1:1:1 in the pentameric complex (Table 1).
FIGURE 5.
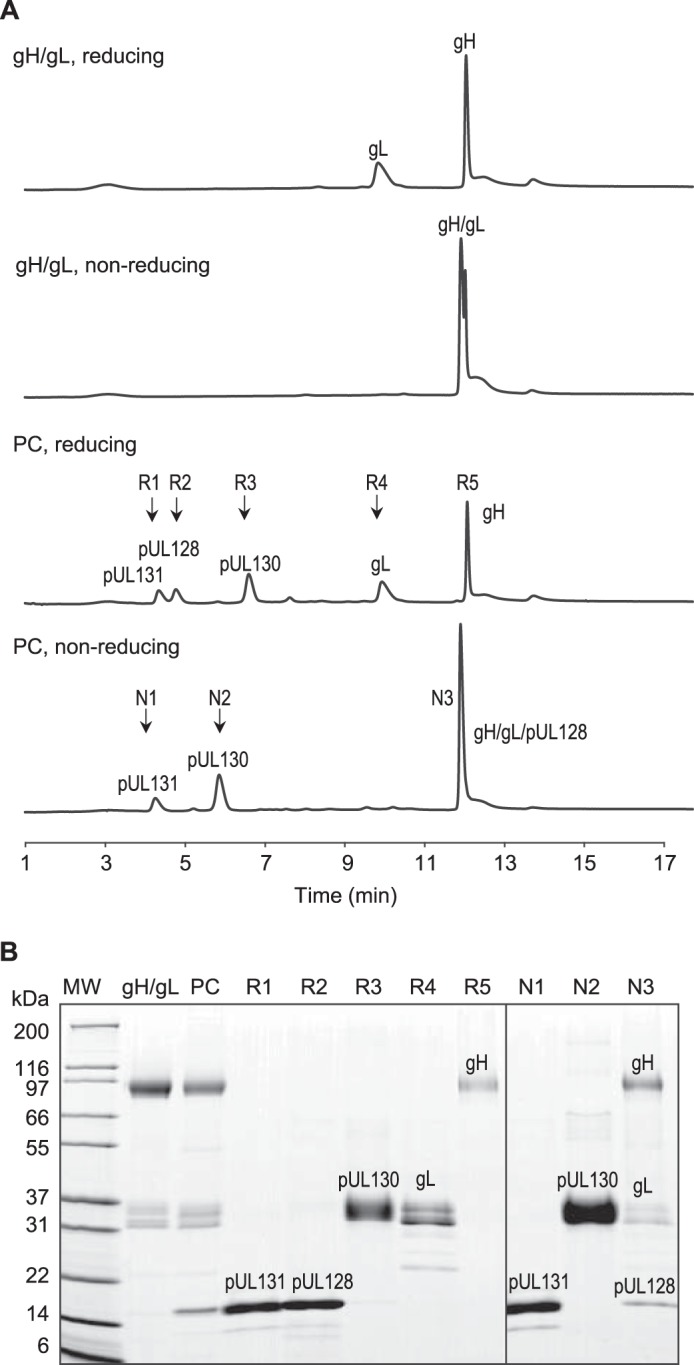
A, RP-HPLC separation of gH/gL and gH/gL/pUL128–131 PC under reducing and nonreducing conditions. Samples were prepared with 1% SDS for nonreducing condition and 1% SDS and 100 mm DTT for reducing condition. Samples of 30 μg of gH/gL and PC were separated on Applied Biosystems Poros R2 column at 75 °C. The chromatogram shows baseline separation among pUL131, pUL128, pUL130, gL, and gH peaks. B, peak identification by SDS-PAGE and nano LC-MS/MS. Samples R1–R5 were collected from PC under reducing condition. Samples N1–N3 were collected from PC under nonreducing condition.
TABLE 1.
Stoichiometry of gH/gL and gH/gL/pUL128–131 pentameric complex
| Peak | R1 | R2 | R3 | R4 | R5 |
|---|---|---|---|---|---|
| Retention time (min) | 4.3 | 4.8 | 6.6 | 9.8–9.9 | 12.0–12.5 |
| Protein ID | pUL131 | pUL128 | pUL130 | gL | gH |
| Theoretical extinction coefficient (m−1 cm−1) | 15,930 | 18,910 | 40,910 | 28,880 | 69,220 |
| Peak area in gH/gL | 183.0 | 403.5 | |||
| Molar ratio compared with R5 | 1.3 | 1.0 | |||
| Peak area in pentameric complex | 56.67 | 64.56 | 131.3 | 130.7 | 290.5 |
| Molar ratio compared with R5 | 0.8 | 0.8 | 0.8 | 1.1 | 1.0 |
Furthermore, the RP-HPLC also confirmed the covalent linkage in the complexes. Under nonreducing but denaturing conditions, gH/gL eluted as one peak containing both gH and gL proteins. Under the same condition, the pentameric complex eluted as three peaks N1–N3. Peak identification showed that N3 fraction contained gH/gL/pUL128, confirming the observed covalent linkages.
Inhibition of Viral Entry into Epithelial Cells
To study the effect of the soluble complexes on viral entry, the pentameric complex restored AD169 virus was incubated with gH/gL, the pentameric complex, CMV-HIG, and a negative control IgG, respectively. CMV-HIG was used as a positive control for viral entry inhibition. Viral entry was quantitated at 20 h postinfection by the expression of IE2 protein. As expected, CMV-HIG effectively inhibited viral entry with an IC50 of 1.5 μg/ml. Pentameric complex inhibited 91% at the highest concentration tested (158 μg/ml) with an estimated IC50 of 53 μg/ml. No inhibition was observed for gH/gL or the negative control IgG (Fig. 6). Inhibition in fibroblast cells was not evaluated as the pentameric complex is not required for entry into fibroblast.
FIGURE 6.

Evaluation of viral entry inhibition by soluble gH/gL and gH/gL/pUL128–131 PC. Pentameric complex restored AD169 viruses were preincubated with soluble gH/gL, soluble gH/gL/pUL128–131 PC, CMV-HIG, and a negative control IgG, and the mixtures were used to infect ARPE-19 cells. Viral entry was quantitated at 20 h postinfection by IE2 protein expression.
Depletion of Neutralizing Activities in CMV-HIG by gH/gL and gH/gL/pUL128–131 Pentameric Complex
To deplete gH/gL or pentameric complex specific antibodies, CMV-HIG was serially incubated with purified gH/gL or pentameric complex. The mock depletion was performed using PBS as the mock antigen. The extent of depletion was measured by direct ELISA using plates coated with the pentameric complex. Fig. 7A shows the dose-response curve of depleted CMV-HIG binding to the pentameric complex. The mock control had similar binding activity as the untreated CMV-HIG, suggesting that there was minimum activity loss because of sample handling. Depletion with pentameric complex reduced up to 85% of the pentameric complex binding activity in CMV-HIG comparing to the mock control. As expected, gH/gL partially depleted (63%) the binding activity to the pentameric complex. These results suggest that there are pentameric complex specific antibodies in CMV-HIG that cannot be removed by gH/gL.
FIGURE 7.

Evaluation of CMV-HIG depleted by soluble gH/gL and gH/gL/pUL128–131 PC. A, the efficiency of depletion as evaluated by the binding activities of depleted CMV-HIG toward PC. The purified PC was immobilized on the plate for direct ELISA. B, neutralization (Neut) of HCMV in infecting epithelial ARPE-19 cells by depleted CMV-HIG. Infectivity was quantitated at 21 h postinfection by IE1 protein expression. C, reduction of binding and neutralization activities of gH/gL and PC depleted CMV-HIG compared with mock control. Conc., concentration.
The potency of the depleted CMV-HIG was evaluated by measuring its ability to neutralize HCMV infection of epithelial ARPE-19 cells. Only CMV-HIG depleted by the pentameric complex showed significant decrease in neutralizing activity (Fig. 7B). The relative binding and neutralizing activities compared with the control were summarized in Fig. 7C. Although gH/gL-depleted CMV-HIG lost 63% of the binding activity, it had almost the same neutralizing activity as the mock control. This indicated that gH/gL specific antibodies do not contribute to the main neutralizing activity in CMV-HIG. In contrast, pentameric complex-depleted CMV-HIG showed a 76% reduction in its ability to neutralize infection of epithelial cells, similar to the 85% loss in binding activity. The remaining neutralizing activity could be a result of incomplete depletion. The result suggests that pentameric complex specific antibodies, not including those targeting the gH/gL portion of the complex, correlate strongly with neutralizing potency and these pentameric complex specific antibodies comprise at least 76% of the neutralizing activity in CMV-HIG. Our observation matches well with the finding from Fouts et al. (28), which demonstrated that the neutralization titer of CMV-HIG decreases 85% when it is depleted by cell surface-expressed pentameric complex antigens. Our depletion study further confirms that the purified soluble pentameric complex contains the key native epitopes that are important to the neutralizing activity in naturally infected human sera.
Discussion
The HCMV gH/gL/pUL128–131 pentameric complex is currently under intensive evaluation for its vaccine potential. Multiple modalities of HCMV vaccine containing the pentameric complex have been shown to be immunogenic in preclinical models (16, 29–32). A replication defective virus expressing the complex is currently being tested in a phase I clinical trial (ClinicalTrials.gov Identifier NCT01986010). Better understanding the size, charge, composition, stoichiometry, and neutralizing epitope of the pentameric complex provides insight into how to define and maintain what is expected to be a critical antigenic attribute of the HCMV vaccine.
Using the recombinant expression system, we are able to produce two stable complexes: gH/gL and the gH/gL/pUL128–131 pentameric complex. Although gH/gL may only be present in certain HCMV strains (8, 9), the complex is stable in vitro and capable of eliciting neutralizing antibodies in mice (31, 33, 34). The soluble gH/gL complex tends to homodimerize to form (gH/gL)2, and it has been hypothesized that the formation of (gH/gL)2 on the virus surface may increase the binding avidity for cellular receptors and facilitate viral entry (35). In our study, gH/gL dimerizes in the absence of pUL128–131, consistent with the observations of others (31, 34). We speculate that gO and pUL128–131 shield the N terminus of gH/gL from dimerization in HCMV and (gH/gL)2 exists only in the absence of a sufficient amount of gO or pUL128–131.
Our work highlights the importance of fully characterizing the recombinant gH/gL/pUL128–131 pentameric complex. In our experience, cells overexpressing gH, gL, pUL128, pUL130, and pUL131 not only produced the pentameric complex but also gH/gL. It is therefore imperative to carefully evaluate the purity of any recombinant pentameric complex. As shown in Fig. 4B, the pentameric complex purchased from a reagent vendor contains co-purified (gH/gL)2 homodimers. We have not been able to identify gH/gL specific antibodies that do not recognize the pentameric complex as shown in Fig. 3C. Analysis such as size exclusion HPLC-MALS, CEX, NanoPro, and RP-HPLC can be used quantitatively to assess the purity of gH/gL/pUL128–131 pentameric complex.
We show for the first time the stoichiometry of 1:1:1:1:1 for the pentameric complex by PR-HPLC. In addition, both RP-HPLC and SDS-PAGE results suggest that pUL128 is disulfide-linked to gH/gL, whereas pUL130 and pUL131 interact with gH/gL/pUL128 noncovalently, a model that is consistent with the results by Wang and Shenk (11), but different from reports by Ryckman et al. (27). While we were writing this manuscript, Ciferri et al. (34) published the importance of Cys-144 in gL, which is disulfide linked with Cys-162 in pUL128. Hence, our work agrees well with Ciferri's data. Our EM images of gH/gL and pentameric complex also match with Ciferri's results. Finally, we have confirmed the molecular mass of gH/gL and pentameric complex by size exclusion HPLC-MALS, the charge properties by CEX, and the immunospecificity toward mAb 124.4 and 57.4 by NanoPro.
The soluble gH/gL and gH/gL/pUL128–131 pentameric complexes are essential reagents for vaccine development by enabling quick epitope screening of pentameric complex binding antibodies. Fig. 3 illustrates how the two soluble complexes, folded in the native conformation, can be used in an ELISA assay to screen gH/gL and pentameric complex specific antibodies. Furthermore, the two soluble glycoprotein complexes can be used in conformational epitope mapping experiments such as hydrogen-deuterium exchange coupled with MS.
Functions of the gH/gL/pUL128–131 pentameric complex have not yet been defined, and it has been speculated that the pentameric complex interacts with a cellular receptor to mediate viral entry into endothelial and epithelial cells. The viral entry inhibition studies with the soluble pentameric complex support the hypothesis because we show that the soluble recombinant pentameric complex, not gH/gL, blocks viral infection of epithelial cells. Based on the stoichiometry and molecular mass determined for the pentameric complex, the IC50 is estimated as ∼0.3 μm under the current experimental conditions.
Antibodies specific to the pentameric complex may have therapeutic applications. It is important to emphasize that not all pentameric complex binding antibodies are potent neutralizers. Some pentameric complex binding antibodies in human hyperimmune globin may bind to the gH/gL portion of the pentameric complex but do not contribute to virus neutralization (Fig. 7C, gH/gL depleted CMV-HIG). Therefore, it is important to use both pentameric complex and gH/gL to screen antibodies. Antibodies that bind only to the pentameric complex are expected to be more potent neutralizers than those that bind both the pentameric complex and gH/gL.
Lastly, the gH/gL/pUL128–131 pentameric complex can be used to develop functionally relevant serologic assays to support vaccine clinical development. Currently, the commonly available serologic test uses the whole virus preparation as the antigens in ELISA, and this approach may not be sufficient to judge the quality of subjects' anti-HCMV antibody responses. Because epitopes on the pentameric complex are critical for neutralizing infection of key cell types, the recombinant pentameric complex could be used as an alternative antigen in the serologic test to quantitate the anti-pentameric complex titer. Additionally, it would be extremely valuable to understand whether the serological titer against the pentameric complex might serve as a surrogate correlate of protection.
In conclusion, we have described a panel of biochemical, immunological and in vitro functional characterizations of a recombinant soluble gH/gL/pUL128–131 pentameric complex. We show that 1) the pentameric complex has a stoichiometry of 1:1:1:1:1; 2) it contains neutralizing epitopes recognized by HCMV-HIG; and 3) it can block viral entry to epithelial cells. Our results confirm the importance of the gH/gL/pUL128–131 pentameric complex as a critical component for vaccine design and indicate that the recombinant soluble form described here retains the critical antigenic attributes previously described for the membrane-bound gH/gL/pUL128–131 complex. This soluble pentameric complex enables future structural and functional studies to better understand the viral entry process.
Acknowledgments
We gratefully acknowledge the assistance of Deba Saha, Gregory Keil, Zubia Naji, Pravien Abeywickrema, and Noel Byrne of Merck Research Laboratories with stable CHO-GS cell line generation and discussion on suggesting purification columns, NanoImaging Services Inc. with imaging gH/gL and pentameric complex using EM, and performing crystal structure overlay based on simulated map. We also thank Richard W. Peluso for critical review of this manuscript.
The authors declare that they have no conflicts of interest with the contents of this article.
- HCMV
- human cytomegalovirus
- PC
- pentameric complex
- HIG
- hyperimmune globulin
- CEX
- cation exchange chromatography
- CEXFT
- flowthrough from CEX column
- MALS
- multiangle light scattering
- RP
- reversed phase
- GS
- glutamine synthetase
- Bicine
- N,N-bis(2-hydroxyethyl)glycine
- IMAC
- immobilized metal affinity chromatography
- TFA
- trifluoroacetic acid
- PNGase F
- peptide N-glycosidase F.
References
- 1. Edward S., Mocarski J., Shenk T., Griffiths P. D., Pass R. F. (2013) Cytomegaloviruses. In Fields Virology (Knipe D. M., Howley P. M., eds) 6th Ed., Lippincott Williams & Wilkins, Philadelphia [Google Scholar]
- 2. Kenneson A., Cannon M. J. (2007) Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev. Med. Virol. 17, 253–276 [DOI] [PubMed] [Google Scholar]
- 3.Institute of Medicine CtSPfV (ed) (2001) Vaccines for the 21st century: a tool for decisionmaking, National Academies Press, Washington, D. C. [PubMed] [Google Scholar]
- 4. Arvin A. M., Fast P., Myers M., Plotkin S., Rabinovich R., (2004) Vaccine development to prevent cytomegalovirus disease: report from the National Vaccine Advisory Committee. Clin. Infect. Dis. 39, 233–239 [DOI] [PubMed] [Google Scholar]
- 5. Sinzger C., Digel M., Jahn G. (2008) Cytomegalovirus cell tropism. Curr. Top. Microbiol. Immunol. 325, 63–83 [DOI] [PubMed] [Google Scholar]
- 6. Krummenacher C., Carfí A., Eisenberg R. J., Cohen G. H. (2013) Entry of herpesviruses into cells: the enigma variations. Adv. Exp. Med. Biol. 790, 178–195 [DOI] [PubMed] [Google Scholar]
- 7. Stampfer S. D., Heldwein E. E. (2013) Stuck in the middle: structural insights into the role of the gH/gL heterodimer in herpesvirus entry. Curr. Opin. Virol. 3, 13–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zhou M., Yu Q., Wechsler A., Ryckman B. J. (2013) Comparative analysis of gO isoforms reveals that strains of human cytomegalovirus differ in the ratio of gH/gL/gO and gH/gL/UL128–131 in the virion envelope. J. Virol. 87, 9680–9690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ryckman B. J., Chase M. C., Johnson D. C. (2010) Human cytomegalovirus TR strain glycoprotein O acts as a chaperone promoting gH/gL incorporation into virions but is not present in virions. J. Virol. 84, 2597–2609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Wang D., Shenk T. (2005) Human cytomegalovirus UL131 open reading frame is required for epithelial cell tropism. J. Virol. 79, 10330–10338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Wang D., Shenk T. (2005) Human cytomegalovirus virion protein complex required for epithelial and endothelial cell tropism. Proc. Natl. Acad. Sci. U.S.A. 102, 18153–18158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Hahn G., Revello M. G., Patrone M., Percivalle E., Campanini G., Sarasini A., Wagner M., Gallina A., Milanesi G., Koszinowski U., Baldanti F., Gerna G. (2004) Human cytomegalovirus UL131–128 genes are indispensable for virus growth in endothelial cells and virus transfer to leukocytes. J. Virol. 78, 10023–10033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ryckman B. J., Chase M. C., Johnson D. C. (2008) HCMV gH/gL/UL128–131 interferes with virus entry into epithelial cells: evidence for cell type-specific receptors. Proc. Natl. Acad. Sci. U.S.A. 105, 14118–14123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Macagno A., Bernasconi N. L., Vanzetta F., Dander E., Sarasini A., Revello M. G., Gerna G., Sallusto F., Lanzavecchia A. (2010) Isolation of human monoclonal antibodies that potently neutralize human cytomegalovirus infection by targeting different epitopes on the gH/gL/UL128–131A complex. J. Virol. 84, 1005–1013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Freed D. C., Tang Q., Tang A., Li F., He X., Huang Z., Meng W., Xia L., Finnefrock A. C., Durr E., Espeseth A. S., Casimiro D. R., Zhang N., Shiver J. W., Wang D., An Z., Fu T.-M. (2013) Pentameric complex of viral glycoprotein H is the primary target for potent neutralization by a human cytomegalovirus vaccine. Proc. Natl. Acad. Sci. U.S.A. 110, E4997–E5005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Fu T.-M., Wang D., Freed D. C., Tang A., Li F., He X., Cole S., Dubey S., Finnefrock A. C., ter Meulen J., Shiver J. W., Casimiro D. R. (2012) Restoration of viral epithelial tropism improves immunogenicity in rabbits and rhesus macaques for a whole virion vaccine of human cytomegalovirus. Vaccine 30, 7469–7474 [DOI] [PubMed] [Google Scholar]
- 17. Neff B. J., Weibel R. E., Buynak E. B., McLean A. A., Hilleman M. R. (1979) Clinical and laboratory studies of live cytomegalovirus vaccine Ad-169. Proc. Soc. Exp. Biol. Med. 160, 32–37 [DOI] [PubMed] [Google Scholar]
- 18. Elek S. D., Stern H. (1974) Development of a vaccine against mental retardation caused by cytomegalovirus infection in utero. Lancet 1, 1–5 [DOI] [PubMed] [Google Scholar]
- 19. Wang D., Li F., Freed D. C., Finnefrock A. C., Tang A., Grimes S. N., Casimiro D. R., Fu T.-M. (2011) Quantitative analysis of neutralizing antibody response to human cytomegalovirus in natural infection. Vaccine 29, 9075–9080 [DOI] [PubMed] [Google Scholar]
- 20. Shytuhina A., Pristatsky P., He J., Casimiro D. R., Schwartz R. M., Hoang V. M., Ha S. (2014) Development and application of a reversed-phase high-performance liquid chromatographic method for quantitation and characterization of a Chikungunya virus-like particle vaccine. J. Chromatogr. A 1364, 192–197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Michels D. A., Tu A. W., McElroy W., Voehringer D., Salas-Solano O. (2012) Charge heterogeneity of monoclonal antibodies by multiplexed imaged capillary isoelectric focusing immunoassay with chemiluminescence detection. Anal. Chem. 84, 5380–5386 [DOI] [PubMed] [Google Scholar]
- 22. Lander G. C., Stagg S. M., Voss N. R., Cheng A., Fellmann D., Pulokas J., Yoshioka C., Irving C., Mulder A., Lau P.-W., Lyumkis D., Potter C. S., Carragher B. (2009) Appion: an integrated, database-driven pipeline to facilitate EM image processing. J. Struct. Biol. 166, 95–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Sorzano C. O., Marabini R., Velázquez-Muriel J., Bilbao-Castro J. R., Scheres S. H., Carazo J. M., Pascual-Montano A. (2004) XMIPP: a new generation of an open-source image processing package for electron microscopy. J. Struct. Biol. 148, 194–204 [DOI] [PubMed] [Google Scholar]
- 24. Dou P., Liu Z., He J., Xu J.-J., Chen H.-Y. (2008) Rapid and high-resolution glycoform profiling of recombinant human erythropoietin by capillary isoelectric focusing with whole column imaging detection. J. Chromatogr. A 1190, 372–376 [DOI] [PubMed] [Google Scholar]
- 25. Anderson C. L., Wang Y., Rustandi R. R. (2012) Applications of imaged capillary isoelectric focussing technique in development of biopharmaceutical glycoprotein-based products. Electrophoresis 33, 1538–1544 [DOI] [PubMed] [Google Scholar]
- 26. Chowdary T. K., Cairns T. M., Atanasiu D., Cohen G. H., Eisenberg R. J., Heldwein E. E. (2010) Crystal structure of the conserved herpesvirus fusion regulator complex gH-gL. Nat. Struct. Mol. Biol. 17, 882–888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Ryckman B. J., Rainish B. L., Chase M. C., Borton J. A., Nelson J. A., Jarvis M. A., Johnson D. C. (2008) Characterization of the human cytomegalovirus gH/gL/UL128–131 complex that mediates entry into epithelial and endothelial cells. J. Virol. 82, 60–70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Fouts A. E., Chan P., Stephan J.-P., Vandlen R., Feierbach B. (2012) Antibodies against the gH/gL/UL128/UL130/UL131 complex comprise the majority of the anti-cytomegalovirus (anti-CMV) neutralizing antibody response in CMV hyperimmune globulin. J. Virol. 86, 7444–7447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wussow F., Yue Y., Martinez J., Deere J. D., Longmate J., Herrmann A., Barry P. A., Diamond D. J. (2013) A vaccine based on the rhesus cytomegalovirus UL128 complex induces broadly neutralizing antibodies in rhesus macaques. J. Virol. 87, 1322–1332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Wussow F., Chiuppesi F., Martinez J., Campo J., Johnson E., Flechsig C., Newell M., Tran E., Ortiz J., La Rosa C., Herrmann A., Longmate J., Chakraborty R., Barry P. A., Diamond D. J. (2014) Human cytomegalovirus vaccine based on the envelope gH/gL pentamer complex. PLoS Pathog. 10, e1004524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Wen Y., Monroe J., Linton C., Archer J., Beard C. W., Barnett S. W., Palladino G., Mason P. W., Carfi A., Lilja A. E. (2014) Human cytomegalovirus gH/gL/UL128/UL130/UL131A complex elicits potently neutralizing antibodies in mice. Vaccine 32, 3796–3804 [DOI] [PubMed] [Google Scholar]
- 32. Saccoccio F. M., Sauer A. L., Cui X., Armstrong A. E., Habib E.-S. E., Johnson D. C., Ryckman B. J., Klingelhutz A. J., Adler S. P., McVoy M. A. (2011) Peptides from cytomegalovirus UL130 and UL131 proteins induce high titer antibodies that block viral entry into mucosal epithelial cells. Vaccine 29, 2705–2711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Loomis R. J., Lilja A. E., Monroe J., Balabanis K. A., Brito L. A., Palladino G., Franti M., Mandl C. W., Barnett S. W., Mason P. W. (2013) Vectored co-delivery of human cytomegalovirus gH and gL proteins elicits potent complement-independent neutralizing antibodies. Vaccine 31, 919–926 [DOI] [PubMed] [Google Scholar]
- 34. Ciferri C., Chandramouli S., Donnarumma D., Nikitin P. A., Cianfrocco M. A., Gerrein R., Feire A. L., Barnett S. W., Lilja A. E., Rappuoli R., Norais N., Settembre E. C., Carfi A. (2015) Structural and biochemical studies of HCMV gH/gL/gO and pentamer reveal mutually exclusive cell entry complexes. Proc. Natl. Acad. Sci. U.S.A. 112, 1767–1772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Fouts A. E., Comps-Agrar L., Stengel K. F., Ellerman D., Schoeffler A. J., Warming S., Eaton D. L., Feierbach B. (2014) Mechanism for neutralizing activity by the anti-CMV gH/gL monoclonal antibody MSL-109. Proc. Natl. Acad. Sci. U.S.A. 111, 8209–8214 [DOI] [PMC free article] [PubMed] [Google Scholar]



