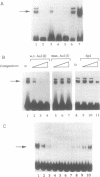
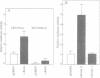
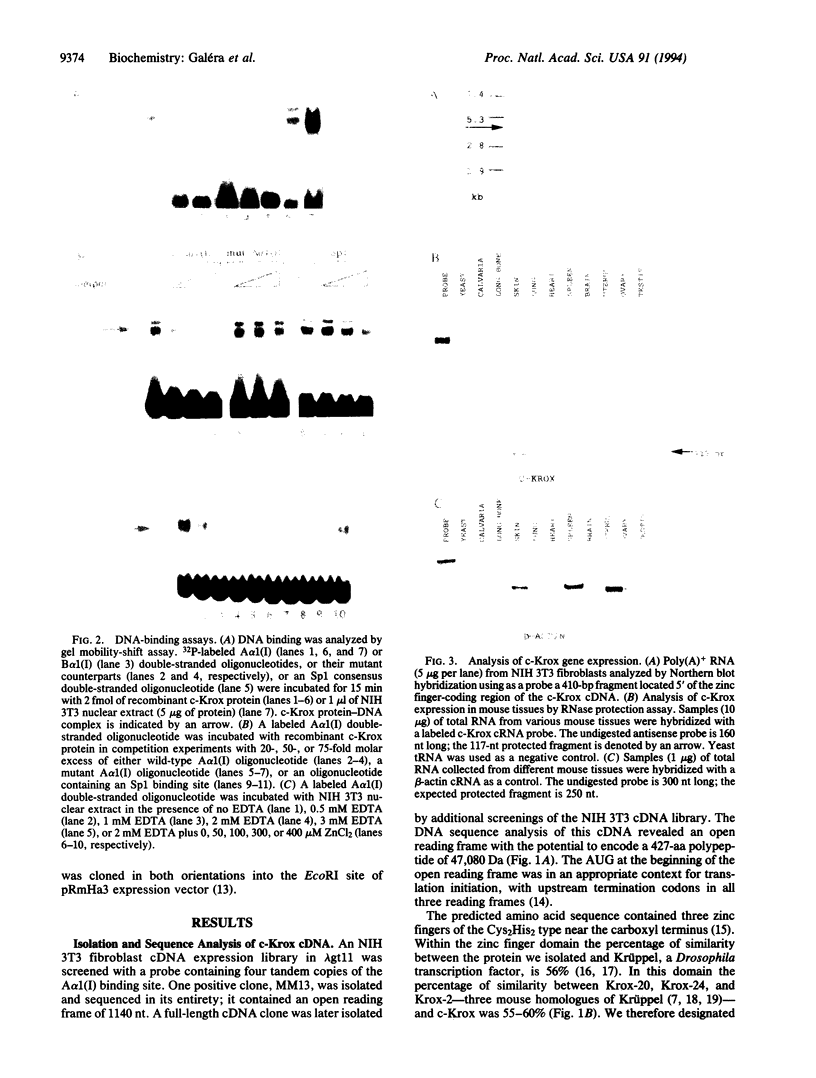
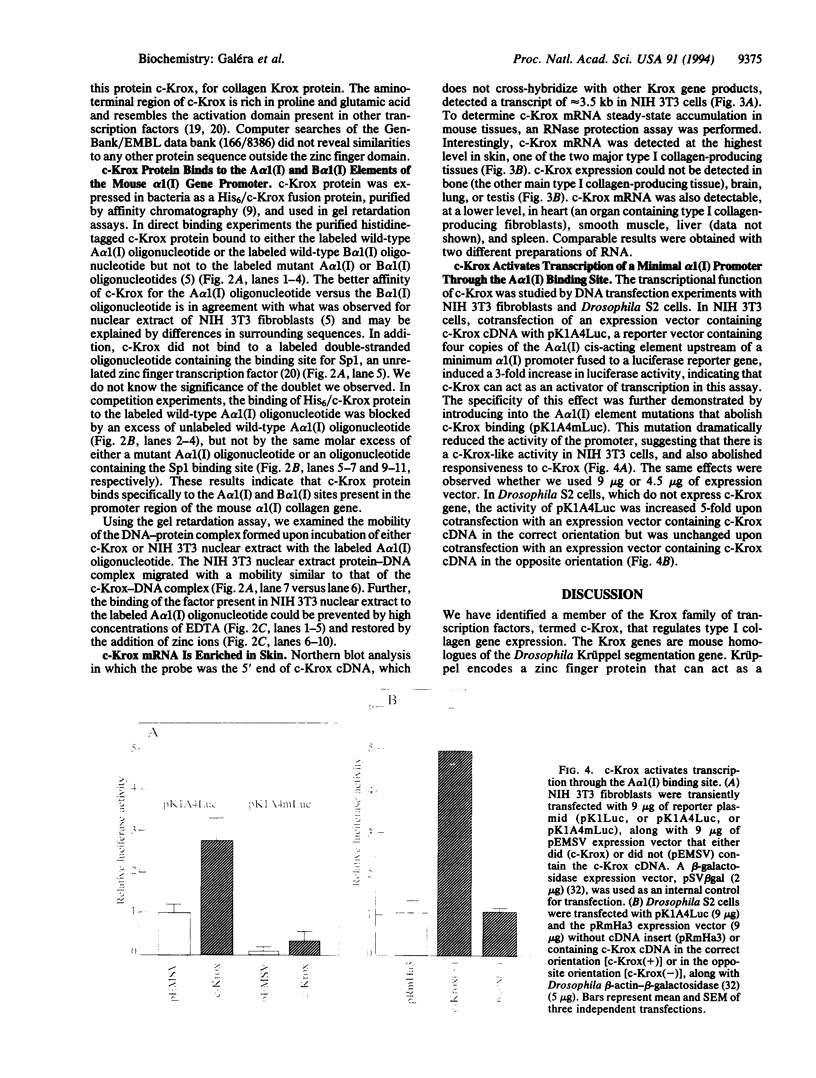
Abstract
We have cloned a mouse cDNA that is a member of the Krox gene family and encodes a protein we have named c-Krox. The c-Krox protein contains three zinc fingers of the Cys2His2 type. c-Krox binds specifically to a guanine-rich cis-acting element present twice in the promoter element of the mouse alpha 1(I) collagen gene. Study of c-Krox gene expression shows that c-Krox is markedly enriched in skin, one of the two major sites of type I collagen synthesis, but is absent in bone, the other main type I collagen-producing tissue, indicating that type I collagen gene expression is differentially regulated in skin and bone. DNA transfection experiments in mouse NIH 3T3 fibroblasts, cells that express the c-Krox gene, or in Drosophila S2 cells, which do not express c-Krox, reveal that c-Krox can activate transcription of a reporter gene linked to several copies of its binding site in the alpha 1(I) collagen promoter. Thus, c-Krox is only the second member of the Krox family for which a target gene has been identified. The selective spatial pattern of expression of its mRNA and its transcription activation ability suggest that c-Krox may be an important regulator of type I collagen skin specific expression in physiologic conditions and in fibrotic diseases such as scleroderma.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berg J. M. Zinc finger domains: hypotheses and current knowledge. Annu Rev Biophys Biophys Chem. 1990;19:405–421. doi: 10.1146/annurev.bb.19.060190.002201. [DOI] [PubMed] [Google Scholar]
- Bunch T. A., Grinblat Y., Goldstein L. S. Characterization and use of the Drosophila metallothionein promoter in cultured Drosophila melanogaster cells. Nucleic Acids Res. 1988 Feb 11;16(3):1043–1061. doi: 10.1093/nar/16.3.1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bush G. L., Tassin A. M., Fridén H., Meyer D. I. Secretion in yeast. Purification and in vitro translocation of chemical amounts of prepro-alpha-factor. J Biol Chem. 1991 Jul 25;266(21):13811–13814. [PubMed] [Google Scholar]
- Chavrier P., Lemaire P., Revelant O., Bravo R., Charnay P. Characterization of a mouse multigene family that encodes zinc finger structures. Mol Cell Biol. 1988 Mar;8(3):1319–1326. doi: 10.1128/mcb.8.3.1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chavrier P., Zerial M., Lemaire P., Almendral J., Bravo R., Charnay P. A gene encoding a protein with zinc fingers is activated during G0/G1 transition in cultured cells. EMBO J. 1988 Jan;7(1):29–35. doi: 10.1002/j.1460-2075.1988.tb02780.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C., Okayama H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol. 1987 Aug;7(8):2745–2752. doi: 10.1128/mcb.7.8.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Jimenez S. A., Feldman G., Bashey R. I., Bienkowski R., Rosenbloom J. Co-ordinate increase in the expression of type I and type III collagen genes in progressive systemic sclerosis fibroblasts. Biochem J. 1986 Aug 1;237(3):837–843. doi: 10.1042/bj2370837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karsenty G., de Crombrugghe B. Two different negative and one positive regulatory factors interact with a short promoter segment of the alpha 1 (I) collagen gene. J Biol Chem. 1990 Jun 15;265(17):9934–9942. [PubMed] [Google Scholar]
- Kozak M. The scanning model for translation: an update. J Cell Biol. 1989 Feb;108(2):229–241. doi: 10.1083/jcb.108.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemaire P., Revelant O., Bravo R., Charnay P. Two mouse genes encoding potential transcription factors with identical DNA-binding domains are activated by growth factors in cultured cells. Proc Natl Acad Sci U S A. 1988 Jul;85(13):4691–4695. doi: 10.1073/pnas.85.13.4691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Licht J. D., Grossel M. J., Figge J., Hansen U. M. Drosophila Krüppel protein is a transcriptional repressor. Nature. 1990 Jul 5;346(6279):76–79. doi: 10.1038/346076a0. [DOI] [PubMed] [Google Scholar]
- Maity S. N., Golumbek P. T., Karsenty G., de Crombrugghe B. Selective activation of transcription by a novel CCAAT binding factor. Science. 1988 Jul 29;241(4865):582–585. doi: 10.1126/science.3399893. [DOI] [PubMed] [Google Scholar]
- Mermod N., O'Neill E. A., Kelly T. J., Tjian R. The proline-rich transcriptional activator of CTF/NF-I is distinct from the replication and DNA binding domain. Cell. 1989 Aug 25;58(4):741–753. doi: 10.1016/0092-8674(89)90108-6. [DOI] [PubMed] [Google Scholar]
- Nardelli J., Gibson T. J., Vesque C., Charnay P. Base sequence discrimination by zinc-finger DNA-binding domains. Nature. 1991 Jan 10;349(6305):175–178. doi: 10.1038/349175a0. [DOI] [PubMed] [Google Scholar]
- Nguyen H. Q., Hoffman-Liebermann B., Liebermann D. A. The zinc finger transcription factor Egr-1 is essential for and restricts differentiation along the macrophage lineage. Cell. 1993 Jan 29;72(2):197–209. doi: 10.1016/0092-8674(93)90660-i. [DOI] [PubMed] [Google Scholar]
- Preiss A., Rosenberg U. B., Kienlin A., Seifert E., Jäckle H. Molecular genetics of Krüppel, a gene required for segmentation of the Drosophila embryo. Nature. 1985 Jan 3;313(5997):27–32. doi: 10.1038/313027a0. [DOI] [PubMed] [Google Scholar]
- Sauer F., Jäckle H. Concentration-dependent transcriptional activation or repression by Krüppel from a single binding site. Nature. 1991 Oct 10;353(6344):563–566. doi: 10.1038/353563a0. [DOI] [PubMed] [Google Scholar]
- Sauer F., Jäckle H. Dimerization and the control of transcription by Krüppel. Nature. 1993 Jul 29;364(6436):454–457. doi: 10.1038/364454a0. [DOI] [PubMed] [Google Scholar]
- Seto E., Lewis B., Shenk T. Interaction between transcription factors Sp1 and YY1. Nature. 1993 Sep 30;365(6445):462–464. doi: 10.1038/365462a0. [DOI] [PubMed] [Google Scholar]
- Seto E., Shi Y., Shenk T. YY1 is an initiator sequence-binding protein that directs and activates transcription in vitro. Nature. 1991 Nov 21;354(6350):241–245. doi: 10.1038/354241a0. [DOI] [PubMed] [Google Scholar]
- Sham M. H., Vesque C., Nonchev S., Marshall H., Frain M., Gupta R. D., Whiting J., Wilkinson D., Charnay P., Krumlauf R. The zinc finger gene Krox20 regulates HoxB2 (Hox2.8) during hindbrain segmentation. Cell. 1993 Jan 29;72(2):183–196. doi: 10.1016/0092-8674(93)90659-e. [DOI] [PubMed] [Google Scholar]
- Shi Y., Seto E., Chang L. S., Shenk T. Transcriptional repression by YY1, a human GLI-Krüppel-related protein, and relief of repression by adenovirus E1A protein. Cell. 1991 Oct 18;67(2):377–388. doi: 10.1016/0092-8674(91)90189-6. [DOI] [PubMed] [Google Scholar]
- Singh H., LeBowitz J. H., Baldwin A. S., Jr, Sharp P. A. Molecular cloning of an enhancer binding protein: isolation by screening of an expression library with a recognition site DNA. Cell. 1988 Feb 12;52(3):415–423. doi: 10.1016/s0092-8674(88)80034-5. [DOI] [PubMed] [Google Scholar]
- Stanojević D., Hoey T., Levine M. Sequence-specific DNA-binding activities of the gap proteins encoded by hunchback and Krüppel in Drosophila. Nature. 1989 Sep 28;341(6240):331–335. doi: 10.1038/341331a0. [DOI] [PubMed] [Google Scholar]
- Swiatek P. J., Gridley T. Perinatal lethality and defects in hindbrain development in mice homozygous for a targeted mutation of the zinc finger gene Krox20. Genes Dev. 1993 Nov;7(11):2071–2084. doi: 10.1101/gad.7.11.2071. [DOI] [PubMed] [Google Scholar]
- Van Dyke M. W., Sirito M., Sawadogo M. Single-step purification of bacterially expressed polypeptides containing an oligo-histidine domain. Gene. 1992 Feb 1;111(1):99–104. doi: 10.1016/0378-1119(92)90608-r. [DOI] [PubMed] [Google Scholar]
- Vuorio E., de Crombrugghe B. The family of collagen genes. Annu Rev Biochem. 1990;59:837–872. doi: 10.1146/annurev.bi.59.070190.004201. [DOI] [PubMed] [Google Scholar]
- Vuorio T., Mäkelä J. K., Vuorio E. Activation of type I collagen genes in cultured scleroderma fibroblasts. J Cell Biochem. 1985;28(2):105–113. doi: 10.1002/jcb.240280204. [DOI] [PubMed] [Google Scholar]
- Weiner F. R., Czaja M. J., Jefferson D. M., Giambrone M. A., Tur-Kaspa R., Reid L. M., Zern M. A. The effects of dexamethasone on in vitro collagen gene expression. J Biol Chem. 1987 May 25;262(15):6955–6958. [PubMed] [Google Scholar]
- Xiang M., Lu S. Y., Musso M., Karsenty G., Klein W. H. A G-string positive cis-regulatory element in the LpS1 promoter binds two distinct nuclear factors distributed non-uniformly in Lytechinus pictus embryos. Development. 1991 Dec;113(4):1345–1355. doi: 10.1242/dev.113.4.1345. [DOI] [PubMed] [Google Scholar]