Abstract
Acute and chronic injuries are characterized by leukocyte infiltration into tissues. Although matrix metalloproteinase 9 (Mmp9) has been implicated in both conditions, its role in wound repair remains unclear. We previously reported a zebrafish chronic inflammation mutant caused by an insertion in the hepatocyte growth factor activator inhibitor gene 1 (hai1; also known as spint1) that is characterized by epithelial extrusions and neutrophil infiltration into the fin. Here, we performed a microarray analysis and found increased inflammatory gene expression in the mutant larvae, including a marked increase in mmp9 expression. Depletion of mmp9 partially rescued the chronic inflammation and epithelial phenotypes, in addition to restoring collagen fiber organization, as detected by second-harmonic generation imaging. Additionally, we found that acute wounding induces epithelial cell mmp9 expression and is associated with a thickening of collagen fibers. Interestingly, depletion of mmp9 impaired this collagen fiber reorganization. Moreover, mmp9 depletion impaired tissue regeneration after tail transection, implicating Mmp9 in acute wound repair. Thus, Mmp9 regulates both acute and chronic tissue damage and plays an essential role in collagen reorganization during wound repair.
KEY WORDS: Mmp9, Inflammation, Repair, SHG imaging, Zebrafish
Highlighted article: In zebrafish, Mmp9 plays a dual role in wound repair through collagen reorganization: it is overexpressed in chronic tissue damage models and required for acute wound healing in control animals.
INTRODUCTION
Acute tissue damage is characterized by the infiltration of leukocytes that clear pathogens and cellular debris, followed by wound repair (Lieschke et al., 2001; Nathan, 2006; van Furth et al., 1985). Interestingly, many of the molecular signals induced by acute tissue damage, including chemokines and proteases, are also present in chronic tissue damage, suggesting that tight regulation of wound signaling is crucial for wound resolution (Feghali and Wright, 1997). However, the relationship between the molecular signals induced during acute and chronic tissue damage remains unclear.
A key step during normal repair includes the deposition of collagen to form a transient extracellular matrix (ECM) that is subsequently degraded during the remodeling phase (McCarty and Percival, 2013). Specific proteases, such as the matrix metalloproteinases (MMPs), mediate collagen remodeling during tissue repair and are upregulated during both acute and chronic wounding (Castaneda et al., 2005; Fini et al., 1996; Mohan et al., 2002; Vandooren et al., 2013). In particular, Mmp9 is associated with many inflammatory processes, including cardiovascular disease and stroke, as well as epithelial injuries to the eye, skin, gut and lung in various organisms ranging from humans to zebrafish (Bai et al., 2005; Betsuyaku et al., 2000; Castaneda et al., 2005; Fini et al., 1996; Mohan et al., 2002; Wyatt et al., 2009). Mmp9 plays an important role in proper keratinocyte migration in vitro (Hattori et al., 2009) and Mmp9 knockout mice display impaired wound closure (McCawley et al., 1998). In both axolotl and zebrafish, mmp9 expression increases along the wounded edge of the epithelium during early regeneration (Yang et al., 1999; Yoshinari et al., 2009), suggesting a role for Mmp9 during wound healing.
We previously reported zebrafish mutants with chronic epithelial damage and inflammation caused by insertion in the hepatocyte growth factor activator inhibitor gene 1 (hai1; also known as spint1) (Mathias et al., 2007) or the ENTH domain protein clint1 (Dodd et al., 2009). Both mutants are reminiscent of the human condition psoriasis, and exhibit epithelial extrusions, hyperproliferation and chronic neutrophil infiltration into the fin (Mathias et al., 2007). In the current study, we aimed to identify factors that contribute to this chronic tissue damage phenotype. We performed a microarray analysis and found a significant increase in mmp9 expression. Depletion of Mmp9 partially rescued the chronic epithelial damage phenotype.
To investigate the role of Mmp9 during wound repair we used second-harmonic generation (SHG) imaging (Campagnola et al., 2002) to non-invasively assess collagen fiber organization in hai1 morphants. We found that depletion of mmp9 partially rescued the disordered collagen fibers observed in the mutant larvae, suggesting that Mmp9 modulates the organization of collagen matrices. By contrast, depletion of mmp9 impaired regeneration after acute wounding, suggesting that Mmp9 is required for acute wound healing and regeneration in larval zebrafish. Furthermore, with SHG imaging, we determined that mmp9 expression regulates the transition in collagen fiber thickness that occurs during acute wound healing. Thus, mmp9 expression differentially regulates acute and chronic tissue damage and repair, and modulates collagen reorganization during wound repair.
RESULTS
hai1 and clint1 mutants have increased mmp9 expression
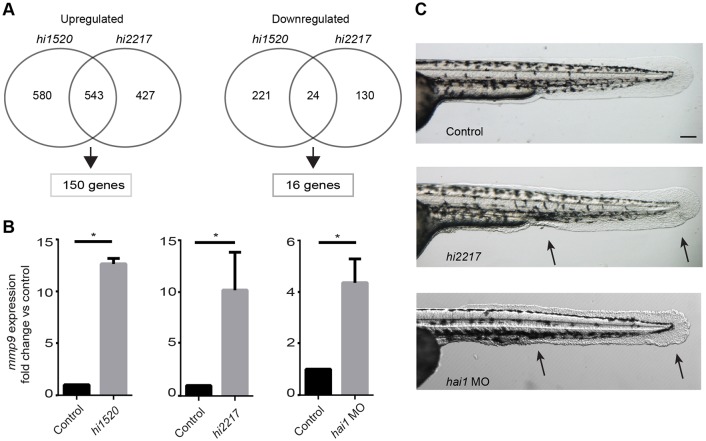
Previous studies from our laboratory identified mutants characterized by chronic epithelial cell damage and persistent neutrophilic infiltration in the epidermis (Dodd et al., 2009; Mathias et al., 2007). To identify mechanisms that contribute to the chronic wound phenotype, we performed microarray analysis of the hai1 (hi2217) and clint1 (hi1520) mutants. Analysis of RNA from 3 days post-fertilization (dpf) hi1520 and hi2217 mutant larvae revealed 166 differentially expressed genes relative to sibling wild-type (WT) larvae (Fig. 1A; supplementary material Fig. S1A,B). We focused on genes involved in pro-inflammatory signaling that modulate ECM remodeling. The most highly overexpressed gene, mmp9, was selected for further analysis. Using quantitative real-time PCR (qRT-PCR) we found a significant overexpression of mmp9 in both the hi2217 (Fig. 1B) and hi1520 mutants (Fig. 1B), which was confirmed by in situ hybridization of hi2217 mutant embryos and their WT siblings (supplementary material Fig. S1C). The heterozygous mutant crosses for the hi2217 fish yield ∼25% of larvae with epithelial defects. To achieve a higher percentage of larvae with the hai1 mutant phenotype, we used a previously established MO to deplete hai1, which recapitulates the mutant phenotype (Carney et al., 2007; Mathias et al., 2007). The hai1 morphants also showed increased mmp9 expression (Fig. 1B), as determined by qRT-PCR.
Fig. 1.
Gene expression profiling reveals elevated inflammatory gene expression, including mmp9, in chronic inflammation mutants. (A) Microarray analysis of inflammation mutants, hi1520 and hi2217, revealed overlapping upregulation of 150 genes and downregulation of 16 genes compared with WT siblings. (B) Upregulation of mmp9 was confirmed by qRT-PCR in the hi2217 and hi1520 mutants and the hai1 morphants. (C) The mutants are characterized by epithelial extrusions and abnormal epithelium development (arrows). Data pooled from experiments performed in triplicate. *P<0.05. Scale bar: 200 µm.
Mmp9 depletion partially rescues the hai1 mutant phenotype
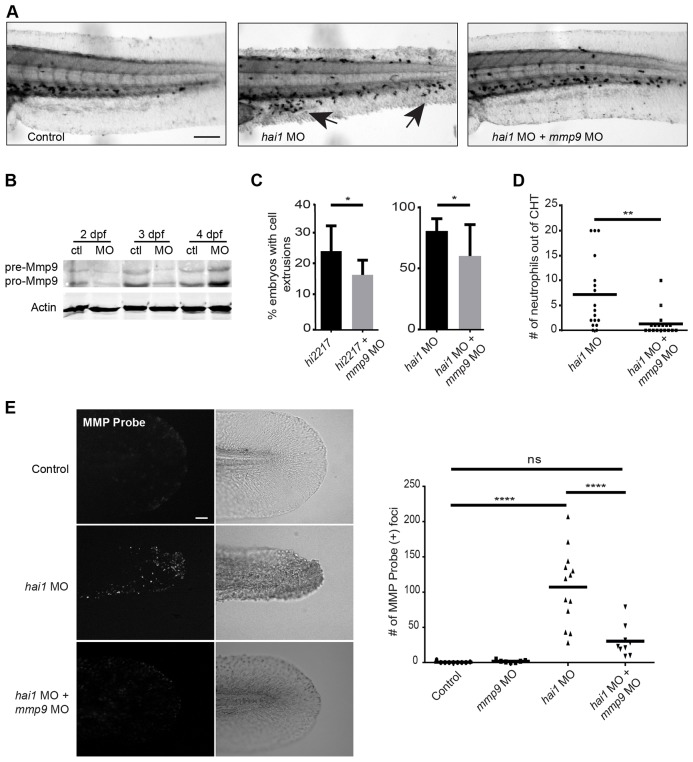
Activation of Mmp9 plays an important role during ECM remodeling (Collier et al., 1988; Fosang et al., 1992; Senior et al., 1991) and restoration of epithelial morphology after tissue damage (Yoshinari et al., 2009). Moreover, inhibition of Mmp9 activity reduces the inflammatory response (Volkman et al., 2010). To determine whether Mmp9 contributes to the abnormal epithelial morphology (Fig. 1C) and neutrophil infiltration (Fig. 2A) observed in the hai1 morphants, we depleted Mmp9 (Fig. 2B) in the hai1 mutants using a previously published mmp9 morpholino (MO1) (Volkman et al., 2010). We found the proportion of mutant hi2217 larvae characterized by epithelial extrusions at the yolk sac extension was reduced when mmp9 was depleted (Fig. 2C). A similar reduction was observed with double injection of mmp9 MO1 and hai1 MO (Fig. 2C), suggesting that overexpression of mmp9 contributes to the hyper-inflammation and epithelial defects in hai1-deficient larvae.
Fig. 2.
Inflammatory phenotypes associated with hai1 mutants are partially rescued by knockdown of mmp9. (A) Sudan Black staining of the hai1 mutants show increased neutrophilic infiltration of the epithelium (arrows), a phenotype that could be rescued by mmp9 knockdown. (B) Western blot analysis of mmp9 MO1. (C) Morpholino knockdown of mmp9 (MO1) decreased the proportion of hi2217 and hai1 morphants displaying epithelial extrusions and (D) decreased the number of neutrophils infiltrating the epithelium in the hai1 morphants. (E) MMPSense showed hyper-activation of MMPs in the hai1 morphants. Hyper-activation of MMPs in the hai1 morphants could be partially rescued by the knockdown of mmp9 expression (MO1). *P<0.05, **P<0.01 and ****P<0.0001. Scale bars: 300 µm in A; 40 µm in E. C and D represent data from a single experiment performed in triplicate.
To investigate whether Mmp9 also contributes to neutrophil infiltration of the epithelium in the hai1 mutants, we performed Sudan Black staining (Le Guyader et al., 2008) and quantified neutrophils outside of the caudal hematopoietic tissue (CHT). The hai1 morphants co-injected with mmp9 MO1 had significantly fewer neutrophils outside of the CHT compared with siblings injected with the hai1 MO alone (Fig. 2A,D), indicating that Mmp9 depletion partially rescues neutrophil infiltration into the epithelium of hai1-deficient larvae. To confirm that the mmp9 MO reduced Mmp9 activity, we utilized a recently reported MMP activity assay in zebrafish (Hall et al., 2014). Depletion of hai1 was associated with increased MMP activation in the fin compared with control larvae (Fig. 2E). We rescued the increased MMP activity in hai1 morphants by performing dual injection of hai1 and mmp9 MOs (Fig. 2E). Together, these data indicate that Mmp9 mediates neutrophilic infiltration and epithelial defects in hai1-deficient larvae.
SHG imaging reveals Mmp9-dependent collagen remodeling in the hai1 morphants
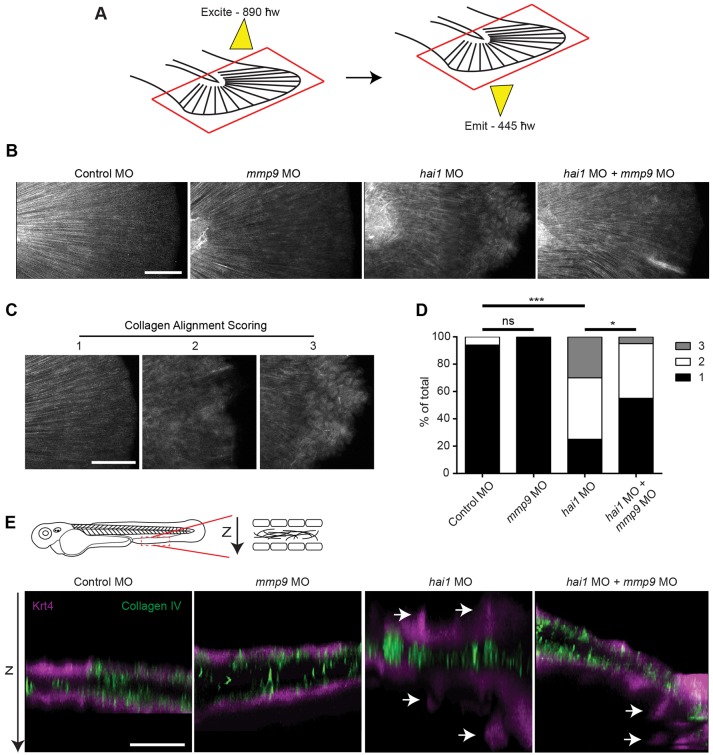
It is known that Mmp9 alters the ECM through the cleavage of matrix components, including collagen (Collier et al., 1988; O'Farrell and Pourmotabbed, 2000; Van den Steen et al., 2002). To determine whether Mmp9 modulates the collagen matrix in hai1 morphants we performed SHG imaging (Campagnola et al., 2002; Mohler et al., 2003). SHG imaging (Fig. 3A) is a label-free microscopy technique that exploits the interaction of light with ordered biological structures to visualize endogenous proteins, such as fibrillar collagen types I and III. Embryos injected with either control MO, hai1 MO, mmp9 MO1 or co-injected with hai1 MO and mmp9 MO1 at the one-cell stage were fixed at 3 dpf for SHG imaging (Fig. 3B). Single-blind analysis of fiber organization on pooled and randomized images was performed (κ=0.78). Each image was scored from 1 to 3, with 1 representing fibers that are aligned and 3 representing fins with unaligned fibers (Fig. 3C). Analysis revealed that hai1 morphants display a significant increase in the proportion of fins with unaligned fibers compared with control (Fig. 3D), a phenotype that could be partially rescued by co-injection of mmp9 MO1 (Fig. 3D). Thus, Mmp9 appears to influence the arrangement of collagen fibers in the fins of hai1 morphants (supplementary material Movies 1-3). Slight changes in collagen fiber alignment were also observed in a small number of mmp9 morphants and are thought to be the result of minor unresolved abrasions due to the lack of Mmp9. As SHG is only sensitive to fibrillar collagen types (Mohler et al., 2003), we also performed immunohistochemistry (IHC) to visualize non-fibrillar type IV collagen. Using IHC we did not observe any obvious differences in collagen organization or structure in the hai1 morphants compared with control (Fig. 3E). Taken together, our data suggest that the alignment of collagen fibers is altered in hai1 morphants, at least in part due to the overexpression of mmp9.
Fig. 3.
SHG imaging shows altered collagen alignment in hai1 morphants that is partially rescued by mmp9 depletion. (A) SHG imaging of type I/III collagen. Caudal fins were excited with an 890 ħw laser and collagen was detected at 445 ħw. (B) Representative z-projected images of stitched multiple ROI SHG z-stacks. The hai1 morphants display irregular collagen alignment. (C) In order to quantify alignment, SHG images illustrate scoring scheme, ranking severity of collagen mis-alignment for analysis from 1 to 3. (D) The hai1 morphants display a significant decrease in collagen alignment that could be partially rescued upon knockdown of mmp9 expression (MO1). (E) IHC of type IV collagen in the transgenic zebrafish Tg(krt4:tdTom). Epithelial extrusions were observed (arrows) in the hai1 morphants. Knockdown of mmp9 resulted in fewer observed extrusions. *P<0.05 and ***P<0.001. Scale bars: 50 µm in B,C; 20 µm in E. D represents the data from experiments performed in quadruplicate and scored by an individual, single-blind analyzer. D represents pooling with experimental numbers for Control MO=18, mmp9 MO=18, hai1 MO=20, hai1 MO+mmp9 MO=20.
Mmp9 influences early leukocyte recruitment during caudal fin wound healing
Previous studies have shown that caudal fin amputation leads to increased expression of mmp9 at the wound edge (Yoshinari et al., 2009). To determine whether the wounded epithelium was a source of the increased mmp9 expression, we utilized a transgenic zebrafish, Tg(kr4:l10a-gfp), which labels the ribosomal subunit l10a in keratinocytes, and performed translating ribosomal affinity purification (TRAP) (Lam et al., 2013). At 6 h post-wounding (hpw), a significant increase in mmp9 expression in epithelial cells was observed (Fig. 4A). To determine whether Mmp9 alters leukocyte recruitment to acute injury, as we found with the hai1 mutant, we performed caudal fin transection. Mmp9-deficient larvae have an early impairment of macrophage recruitment at 1, 6 and 12 hpw, although resolution was not affected (supplementary material Fig. S2A). Similarly, the mmp9 morphants displayed a significant reduction in the number of neutrophils recruited to the site of wounding at 1 hpw, as assessed by Sudan Black staining (supplementary material Fig. S2B). Total neutrophil counts were performed on Mmp9-deficient larvae at 2 dpf and a significant reduction in the total number of neutrophils was observed (supplementary material Fig. S2C). However, no increase in neutrophil retention at the wound was observed at 24 or 48 hpw, indicating that Mmp9 depletion did not affect resolution of neutrophil inflammation at wounds.
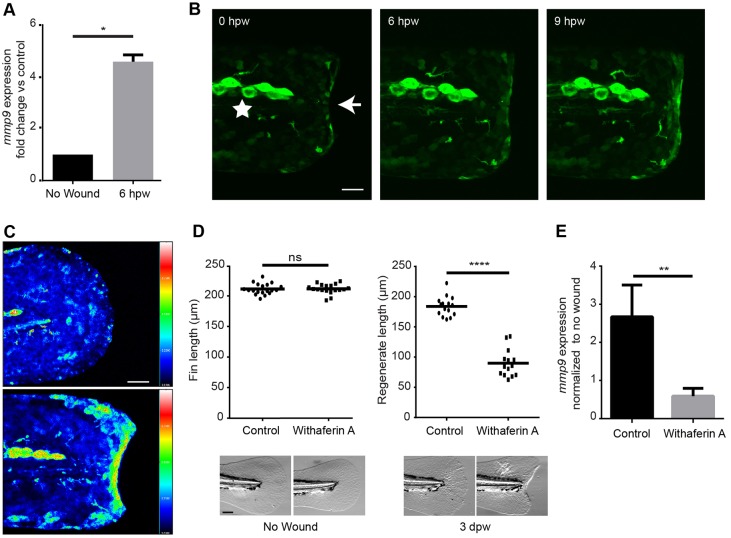
Fig. 4.
Acute wounding induces NFκB-dependent expression of mmp9. (A) An increased expression of mmp9 is observed by 6 h post wounding (hpw) in the 2-dpf embryo. (B) A visible increase in NFκB activation is seen by 6 hpw using the transgenic Tg(NFκB:gfp). The star denotes neuromast cells expressing high levels of gfp independent of wounding. The arrow indicates the edge of the wounded caudal fin and the site of increased NFκB activation. (C) Ratiometric image analysis performed in a cross of Tg(NFκB:gfp) and Tg(krt4:tdTom). (D) Early inhibition of the NFκB pathway with 30 µM withaferin A resulted in a significant reduction in regeneration at 3 dpw but had no effect on developmental fin length. (E) Early inhibition of the NFκB pathway resulted in an abrogation of the increased mmp9 expression in the Tg(krt4:l10a-gfp) TRAP line. *P<0.05, **P<0.01 and ****P<0.0001. Scale bars: 40 µm in B,C; 100 μm in D. A represents data combined from experiments performed in triplicate; D represents data combined from experiments performed in quadruplicate and normalized to unwounded treatments.
Macrophage recruitment and function are required for complete regeneration in multiple models (Li et al., 2012; Lieschke et al., 2001; Martin et al., 2003; Petrie et al., 2014; van Furth et al., 1985). To determine whether the early reduction in macrophage presence alters regeneration in our system we performed morpholino knockdown of the transcription factor Irf8, which has been used to deplete macrophages with a concomitant increase in neutrophils (Li et al., 2011). Following caudal fin amputation, the irf8 morphants displayed no defect in regenerate length at 3 days post-wounding (dpw) (supplementary material Fig. S2D). To further validate this result we targeted the Pu.1 transcription factor by morpholino, which blocks the development of both neutrophils and macrophages. The Pu.1-deficient embryos displayed no defect in caudal fin regeneration at 3 dpw (supplementary material Fig. S2D), consistent with a previous study using Pu.1 knockdown in larval zebrafish (Mathew et al., 2007). These findings indicate that the early leukocyte recruitment defect in Mmp9-deficient larvae is unlikely to affect regeneration at 3 dpw.
NFκB signaling mediates mmp9 expression following caudal fin amputation
We took a candidate approach to identify the upstream signaling pathways that mediate wound-induced expression of mmp9 (Fig. 1B and Fig. 4A). NFκB is a known regulator of mmp9 expression in other systems (Eberhardt et al., 2002; Ganguly et al., 2013). Using a previously published NFκB reporter line, Tg(NFκB:gfp), we observed an increase in NFκB activity after tail transection by 6 hpw (Fig. 4B) (Kanther et al., 2011). Ratiometric imaging was performed by crossing the NFκB reporter with the keratinocyte-labeled transgenic Tg(krt4:tdTom), and confirmed an increase in reporter activity after tail transection (Fig. 4C). This is in agreement with a recent report that identified increased NFκB activity after wounding in zebrafish larvae downstream of wound-induced H2O2 (Candel et al., 2014; de Oliveira et al., 2014). Because early wound signals like H2O2 have been shown to modulate regeneration (Yoo et al., 2012), we sought to determine whether NFκB activation was required for mmp9 expression. Using a pharmacological inhibitor of the NFκB pathway, withaferin A, we found that early NFκB inhibition impaired caudal fin wound healing at 3 dpw (Fig. 4D). To determine whether the activation of NFκB was driving the expression of mmp9 in the epithelium, we performed TRAP to isolate mRNA from control and NFκB-inhibited larvae. Pharmacological inhibition of NFκB was sufficient to block the increase in mmp9 expression as quantified by qRT-PCR (Fig. 4E). Taken together, our findings suggest that NFκB signaling mediates the expression of mmp9 following caudal fin amputation.
Wound-induced changes to collagen fibers are mediated by Mmp9
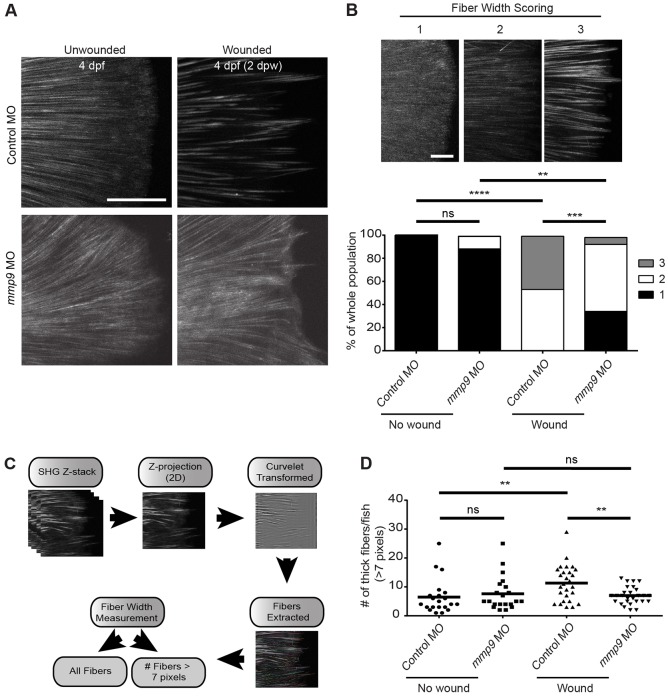
To determine whether Mmp9 also mediates changes to collagen matrices during acute injury we performed IHC and SHG imaging to visualize collagen organization following acute wounding. No clear changes in type IV collagen were observed by IHC after wounding (supplementary material Fig. S3A). By contrast, SHG imaging after tail transection revealed a significant increase in collagen fiber width during wound healing by 2 dpw (Fig. 5A). The increase in fiber width was not observed in unwounded age-matched controls (Fig. 5A). We depleted mmp9 using morpholino to determine whether Mmp9 mediated this change. The Mmp9-deficient larvae had impaired fiber thickening at 2 dpw compared with control, suggesting that Mmp9 is involved in wound-induced fiber thickening (Fig. 5A). To quantify the change in fiber thickness we performed two forms of analysis. We used both single-blind fiber-width scoring (κ=0.58) (Fig. 5B) and a computational measurement of fiber thickness (Fig. 5C). Both methods of analysis revealed a significant increase in fiber width at 2 dpw in control animals that was impaired in Mmp9-deficient wounded larvae (Fig. 5B,D). Fiber alignment was also analyzed, but we found no statistical difference between wounded control and Mmp9-deficient groups (supplementary material Fig. S3B). These data indicate that caudal fin amputation leads to a thickening of collagen fibers through an Mmp9-dependent mechanism.
Fig. 5.
SHG imaging reveals a defect in collagen thickening in the caudal fin of mmp9 morphants at 2 days post amputation. (A) Amputation of the larval caudal fin results in a thickening of type I/III collagen fibers by 2 dpw (top). Fiber thickening does not appear to occur in the mmp9 morphants (MO1) by 2 dpw (bottom). (B) Scoring system for fiber width, from 1 to 3 (top). A significant defect in collagen fiber thickness is seen in the mmp9 morphants at 2 dpw (bottom). (C) Schematic illustrating the quantification of SHG images. All fibers were measured and the number of fibers above a given threshold width (i.e. 7 pixels) can be determined, see Materials and Methods. (D) Quantification of the number of fibers >7 pixels in diameter per caudal fin validates a defect in collagen fiber thickening in the mmp9 morphants at 2 dpw. **P<0.01, ***P<0.001 and ****P<0.0001. Scoring in B from a single-blind analyzer from pooled experiments performed in triplicate. Data in D are from experiments performed in quadruplicate and scored by a single-blind analyzer. Scale bars: 50 µm in A; 20 µm in B. Experimental numbers for B and D: no wound Control MO=21, no wound mmp9 MO=25, wounded Control MO=31, wounded mmp9 MO=32.
Mmp9 is necessary for optimal caudal fin wound healing
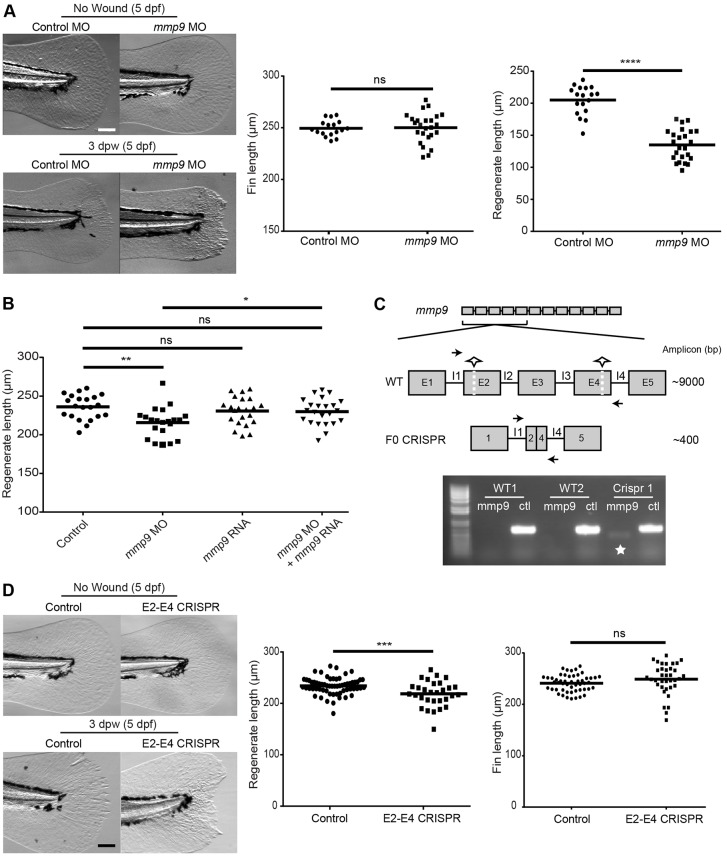
The zebrafish caudal fin is known to undergo complete epimorphic regeneration following amputation (Poss et al., 2003) and we sought to determine whether the defect in collagen remodeling in the Mmp9-deficient embryos was associated with a deficit in caudal fin wound healing. We depleted mmp9 expression (MO 1 and 2) and found that Mmp9-deficient larvae had impaired regenerate length (Fig. 6A; supplementary material Fig. S4A), indicating that Mmp9 is necessary for proper wound healing. The Mmp9 dependency was wound specific, as unwounded morphants did not display a defect in fin length compared with age-matched controls (Fig. 6A; supplementary material Fig. S4A). To confirm that the regeneration defect was not due to off-target effects of MO, we rescued mmp9 expression using co-injection of mmp9 MO and zebrafish mmp9 RNA. Co-injection of mmp9 RNA rescued the regeneration defect of the morphants, supporting a requirement for Mmp9 during acute wound healing (Fig. 6B).
Fig. 6.
Mmp9 regulates caudal fin wound healing following amputation. (A) Morpholino knockdown of mmp9 (MO1) causes a regenerative defect at 3 dpw without influencing developmental fin length. (B) Co-injection of mmp9 MO2 and zebrafish mmp9 RNA (125 ng/µl) rescues the healing defect at 3 dpw (5 dpf). (C) Schematic of two-site CRISPR-Cas9 targeting of mmp9 (top). Half-stars indicate CRISPR sites and arrows represent primer binding for screening. PCR amplification results in the presence of a ∼400 bp amplicon indicated by the star, with the ctl lane representing a housekeeping gene to ensure the presence of gDNA (bottom). (D) Mosaic F0 larvae did not have altered fin lengths at 5 dpf but displayed a significant defect in caudal fin wound healing. *P<0.05, **P<0.01, ***P<0.001 and ****P<0.0001. Graphs represent a single experiment performed in triplicate. Scale bars: 100 µm in A,D.
To further confirm a role for Mmp9 in acute wound healing, we utilized a two-cut CRISPR-Cas9 mutagenesis technique to create mosaic F0 mmp9 mutants. In short, two separate exon targets were identified (exon 2 and exon 4). Co-injection at the one-cell stage resulted in excision of a ∼9000 bp segment of the mmp9 genomic DNA (Fig. 6C) in a mosaic fashion in the developing larvae. At 2 dpf or at the completion of an experiment, genomic DNA was isolated from individual larvae and PCR screened for the presence of a ∼400 bp segment to identify F0 mutant embryos (Fig. 6C). The amplicon was then purified and sequenced for further confirmation (supplementary material Fig. S4B). We found that the mosaic F0 mutants displayed a regeneration defect at 3 dpw (Fig. 6D) but had no effect on unwounded fin length (Fig. 6D). Taken together, these findings suggest that Mmp9 is necessary for acute wound healing and regeneration in the zebrafish larvae.
DISCUSSION
Here, we have demonstrated that Mmp9 plays important roles in acute and chronic tissue damage. In both cases, Mmp9 is involved in the infiltration of leukocytes; however, in the case of hai1-deficient larvae and chronic inflammation, Mmp9 contributed to abnormal collagen matrix composition and epidermal morphology. By contrast, in acute tissue damage associated with tail transection, Mmp9 was necessary for wound resolution and regeneration. Taken together, these findings suggest that a balance in Mmp9 activity is essential for normal tissue homeostasis and repair.
Resolution of inflammation is an essential step during wound repair (Starnes and Huttenlocher, 2012). In some human diseases, like pyoderma gangrenosum, inflammation fails to resolve, contributing to chronic wounds and tissue damage (Powell et al., 1985). These types of inflammatory disorders suggest that a tightly regulated inflammatory response is crucial to ensure proper wound healing (Eming et al., 2007) and that inflammation can contribute to compromised wound repair. However, substantial evidence indicates that leukocyte response to acute damage can be essential for the normal healing process (LeBert and Huttenlocher, 2014). Leukocyte recruitment is necessary to limit infection at the site of tissue damage (Nathan, 2006), and macrophage recruitment is required for optimal wound healing through the clearance of cellular debris (Leibovich and Ross, 1975; Li et al., 2012; Martin et al., 2003; Shiratori et al., 1996; Tidball and Wehling-Henricks, 2007; van Furth et al., 1985). Support for an important role for macrophages in wound repair has also been shown during regeneration in both larval zebrafish (Li et al., 2012) and the adult fin (Petrie et al., 2014).
Our findings suggest that increased mmp9 expression regulates leukocyte infiltration to the inflammatory site, as depletion of mmp9 impaired leukocyte recruitment in the context of both early acute inflammation and chronic tissue damage. This is in accordance with recent reports that suggest that Mmp9 promotes leukocyte recruitment to wounds (Hall et al., 2014; Parks et al., 2004). Interestingly, depletion of leukocytes (pu.1 morpholino) or macrophages specifically (irf8 morpholino) did not impair regeneration after tail transection. This agrees with recent data suggesting that the absence of macrophages impairs regeneration in adult zebrafish but does not result in a defect in wound repair in the larval zebrafish until 120 hpw (Petrie et al., 2014). Thus, our findings suggest that the defect in regeneration observed in Mmp9-deficient larvae was unlikely to be due to a defect in macrophage recruitment.
Mmp9 is known to have an essential role in remodeling the ECM, in particular through the proteolysis of specific targets like collagen IV (Collier et al., 1988). Using SHG imaging, we found that Mmp9 modulated the collagen organization in the context of both acute and chronic tissue damage. In the chronic injury model, with the hai1 morphants, collagen fibers were characterized by poor organization; depletion of mmp9 in these hai1 morphants partially restored collagen fiber alignment, suggesting that Mmp9 contributes to the abnormal collagen architecture in the mutants. However, it is not clear whether this was due to a direct effect of Mmp9 on matrix organization or indirectly through its effects on epithelial cells, fibroblasts or leukocytes. As SHG only detects fibrillar collagen types such as I and III and not non-fibrillar collagen IV, it seems likely that the effects are indirect. Regardless of the mechanism, the observation that depletion of mmp9 partially rescues the altered collagen organization in the mutant suggests that Mmp9 plays a role in regulating ECM architecture in the context of chronic tissue damage.
To determine whether Mmp9 also affects collagen reorganization induced by acute wounding, we performed SHG imaging after tail transection. Surprisingly, SHG imaging after acute injury did not reveal changes to collagen fiber alignment as was observed in the chronic injury model. Instead, acute wounding induced a thickening of collagen fibers in the larval fin. Interestingly, the fiber thickening induced by acute injury was dependent upon mmp9 expression, as morphants did not show wound-induced collagen fiber thickening. Taken together, these findings suggest that Mmp9 mediates the changes in collagen fiber organization that occur in both acute and chronic tissue damage. However, in the case of chronic injury, Mmp9 has detrimental effects, whereas, with acute injury, Mmp9 mediates the changes in collagen fiber organization that are part of the normal repair process.
In the current study we show that Mmp9 regulates collagen structure after wounding. However, as of yet, we do not know the significance of the fiber thickening that occurs after acute injury. In future studies it will be informative to perform live imaging of fiber architecture over time to determine whether Mmp9 regulates the transition in fiber thickness as wound healing progresses. It is known that, following injury in mammalian models, surrounding fibroblasts rapidly produce collagen (Eickelberg et al., 1999; Yates et al., 2012), providing a transient matrix for stability. The type of collagen produced appears to be age dependent and coincides with whether a wound scars or regenerates. Adult wound-induced ECM is predominately the thicker type I collagen, whereas fetal wound-induced collagen is the thinner type III collagen (Yates et al., 2012). SHG imaging does not discriminate between type I and III collagen, and it is therefore possible that Mmp9 is involved in a switch in collagen type during repair. Taken together, our findings demonstrate that Mmp9 promotes acute wound resolution and collagen fiber remodeling, and supports the idea that proper regulation of Mmp9 is required to prevent the progression towards chronic tissue damage and inflammation.
MATERIALS AND METHODS
Zebrafish maintenance and handling
All protocols using zebrafish in this study were approved by the University of Wisconsin-Madison Research Animals Resource Center. Adult zebrafish and embryos were maintained as described previously (Yoo et al., 2010). For wounding assays, 2-dpf larvae were anesthetized in E3 containing 0.2 mg/ml tricaine (ethyl 3-aminobenzoate; Sigma-Aldrich). To prevent pigment formation, some larvae were maintained in E3 containing 0.2 mM N-phenylthiourea (Sigma-Aldrich).
Microarray design and analysis
Adults heterozygous for the hi2217 retroviral insertion in the hai locus (Mathias et al., 2007) were crossed and RNA samples were extracted from WT siblings or hi2217 homozygous mutants. Adults heterozygous for the hi1520 retroviral insertion within the clint1 locus (Dodd et al., 2009) were crossed and RNA samples from WT sibling and hi1520 mutants were collected. Pools of 30-50 embryos of WT siblings and hi2217 or hi1520 mutants were collected at 3 dpf for RNA isolation. Embryos were homogenized in RNA Stat60 (Teltest). RNA was purified using standard chloroform phase separation, isopropanol precipitation and ethanol wash steps. RNA from the hi2217 or hi1520 mutant embryos was labelled with Cy5 and hybridized against Cy3-labeled RNA from the corresponding WT sibling embryos. Experiments were performed in triplicate. RNA isolation, synthesis of amino allyl-labeled aRNA, dye coupling and hybridization conditions were as previously described (Stockhammer et al., 2009). Microarray analysis was performed using custom-designed 44-k Agilent chips (GEO platform accession: GPL7735), described elsewhere (Stockhammer et al., 2009). Microarray data were processed and analyzed as described (Zakrzewska et al., 2010). Significance cut-offs for differentially expressed probe sequences were set at 1.5-fold change at P<10−5. The data were submitted to the GEO database (www.ncbi.nlm.nih.gov/geo) and are available under accession number GSE28110.
Regeneration assays
For regeneration assays, tail transection was performed on 2- to 2.5-dpf larvae using a surgical blade (Feather, no. 10). Regenerate length was quantified by measuring the distance between the caudal tip of the notochord and the caudal tip of the tail fin at 3 dpw. Withaferin A (Tocris) treatment for 1 h before and after wounding at 30 µM was performed to pharmacologically inhibit NFκB activity.
Immunofluorescence and Sudan Black staining
Larvae (2.5-3.5 dpf) were fixed with 1.5% paraformaldehyde in 0.1 M Pipes (Sigma-Aldrich), 1.0 mM MgSO4 (Sigma-Aldrich) and 2 mM EGTA (Sigma-Aldrich) overnight at 4°C and immunolabeled as previously described (Yoo and Huttenlocher, 2011). The following primary antibody was used: rabbit anti-collagen type IV (PAb, Ab6586; Abcam) at 1:300. Dylight 488- or 549-conjugated mouse anti-rabbit IgG antibodies (Jackson ImmunoResearch Laboratories, 211-482-171, 211-502-171) were used as secondary antibodies at 1:250. Immunofluorescence images were acquired with a confocal microscope (FluoView FV1000; Olympus) using a NA 0.75/20× objective. Each fluorescence channel was acquired by sequential line scanning. Z-series were acquired using 180- to 280-µm pinhole and 0.5- to 5-µm step sizes. Z-series images were stacked or three-dimensionally (3D) reconstructed by the FluoView FV1000 software. For Sudan Black staining, embryos (1, 6, 24, 48 h after wounding) were fixed in 4% paraformaldehyde in PBS overnight at 4°C, rinsed in PBS and incubated in 0.03% Sudan Black, followed by extensive washing in 70% ethanol. After rehydration in PBST, pigments were removed by incubation in a 1% H2O2 (Fisher Scientific) and 1% KOH (Sigma-Aldrich) solution. Embryos were observed using a stereoscopic zoom microscope (SMZ1500; Nikon). To image MMP activity, we used MMPSense 645 FAST (PerkinElmer, NEV10100, absorption/emission maxima of 649/666), following a previously established protocol (Hall et al., 2014). Immunofluorescence images were acquired with a confocal microscope (FluoView FV1000; Olympus) using a NA 0.75/20× objective.
Ratiometric image analysis
Images were acquired with a confocal microscope (FluoView FV1000; Olympus) using a NA 0.75/20× objective and performing sequential line scanning. Ratiometric analysis was performed by using FluoView FV1000 software. Two-dimensional (2D) ratiometric images were created after z-series stacking. After complete loss of background by subtraction in the numerator channel, ratio images were created by dividing the numerator channel with the denominator channel and processed by a median filter to remove background noise.
Translating ribosome affinity purification (TRAP)
TRAP was performed as described previously (Lam et al., 2013). Pools of 30-50 3-dpf Tg(krt4:l10a-gfp) larvae were collected for each condition. RNA was purified using the miRvana RNA purification kit (Ambion) and eluted in 30-50 µl of nuclease-free water (Ambion).
Whole-embryo RNA extraction for qRT-PCR
RNA was extracted from whole embryos using the miRvana RNA purification kit (Ambion). One-step qRT-PCR was performed in triplicate using Super Script III Platinum SYBR Green (Invitrogen) from purified TRAP or whole-embryo RNA. Fold change was determined using efficiency-corrected comparative quantitation. Data were normalized to control samples.
Primers: Ef1α: F – 5′-TGCCTTCGTCCCAATTTCAG-3′, R – 5′-TACCCTCCTTGCGCTCAATC-3′; Mmp9: F – 5′-TGATGTGCTTGGACCACGTAA-3′, R – 5′-ACAGGAGCACCTTGCCTTTTC-3′; Il-1β: F – 5′-CACATTTGAAGGCCGTCACACT-3′, R – 5′-CTCGGCGGGGCAACAGG-3′; Tnf-β: F – 5′-CCTCAGACCACGGAAAAGT-3′, R – 5′-GCCCTGTTGGAATGCCTGAT-3′; Mmp13: F – 5′-AAGGAATAAGGCAAATGGATAAAG-3′, R – 5′-TTTGATCCACTGAAGAGGTA-3′; Myd88: F – 5′-GTGTAAGAGGATGGTGGTG-3′, R – 5′-GTAGACGACAGGGATTAGC-3′.
Morpholino injections
Morpholino oligonucleotides (Gene Tools) were re-suspended in water to a stock concentration of 1 mM. Final morpholino concentrations were injected into one- to two-cell stage embryos in 3-nl amounts and embryos were maintained at 28.5°C. Morpholino sequences used have been previously described as follows: mmp9 MO1 (Volkman et al., 2010), mmp9 MO2 (Hall et al., 2014), pu.1 MO (Rhodes et al., 2005), hai1 splice-blocking MO (Mathias et al., 2007) and irf8 MO (Li et al., 2011); control MO: 5′-CCTCTTACCTCAGTTACAATTTATA-3′.
In situ hybridization
A plasmid (pcs2.1, Invitrogen) containing mmp9 was grown and harvested (Maxi-prep, Clontech). The plasmid was linearized using restriction enzymes to serve as template for transcription of digoxigenin-labeled anti-sense probes using T3 RNA polymerase. Whole-mount in situ hybridization was performed as described on 2-dpf embryos (Jowett and Yan, 1996). Following staining, embryos were placed in 80% glycerol for microscopy. Pictures were recorded with a Nikon Digital Sight camera on the Nikon SMZ1500 microscope.
SHG imaging and processing
Tails were removed (∼1- to 2-mm fragments) from the main body of fixed zebrafish embryos (so tail fins lay approximately flat) in a drop of PBS in a glass-bottomed imaging dish (Fluodishes, World Precision Instruments). Tails were imaged on a custom-built multiphoton microscope at the Laboratory for Optical and Computational Instrumentation (Conklin et al., 2009). Briefly, the microscope consists of a tunable titanium sapphire laser (Mira, Coherent) with an inverted microscope (TE2000, Nikon) driven by WiscScan software (http://loci.wisc.edu/software/wiscscan). For SHG imaging of the tails, a 40× water immersion lens (1.25 NA, Nikon) was used, the laser was tuned to 890 nm and backwards SHG was collected through a 445/20 emission filter and detected on a H7422 GaAsP photomultiplier tube (PMT) (Hamamatsu, Japan). Brightfield images were simultaneously collected in transmission mode with a photodiode-based detector (Bio-Rad). Imaging parameters were kept constant across all imaging days. Images were collected as z-stacks, with optical sections 1 µm apart, at 512×512 resolution. Images were collected from the tip of the notochord to the tip of the tail or to the wound site. A minimum of six tails per treatment per replicate were collected. Because the tail is three-dimensional and angled, for visualization and analyses sum projections of the image z-stacks were generated in FIJI (Schindelin et al., 2012). Sum projections were analyzed for fiber width using CT-FIRE software (Bredfeldt et al., 2014; 2. CT-FIRE | LOCI, available at http://loci.wisc.edu/software/ctfire) (see also Fig. 5C). Briefly, individual fibers were extracted from each projected image. The width of each fiber was calculated as the average width of the points automatically selected to represent the fiber as assessed by the algorithm in CT-FIRE. Using these width measurements, the number of fibers ≥7 pixels was determined for each projection.
Statistical analyses
Assuming a Gaussian distribution of the overall population of values, P-values were driven by two-tailed paired t-test (two comparisons) or one-way analysis of variance (ANOVA) (multiple comparisons) comparing means of each sample. Data are representative of at least three separate experiments. The single-blind SHG was analyzed using chi-squared test on data scored by a single scorer, pooled from all experiments. Reliability of single-blind analysis was assessed by determining the κ index.
Western blot
Approximately 60 embryos were collected at 2, 3 and 4 dpf, de-yolked and homogenized using a 1 ml Dounce homogenizer. Samples were sonicated five times for 3 s at 20% amplitude and centrifuged at 18,000 g for 10 min at 4°C. Samples were run on a 10% acrylamide gel and blocked with 5% milk/TS-T. The primary antibodies anti-Mmp9 at 1:250 (Abcam, ab38898) and anti-Actin at 1:2000 (Sigma-Aldrich, a5441) were used; for secondary antibodies, anti-rabbit 800 (Rockland, 611-131-122) and anti-mouse 600 (Invitrogen, A21057) were used at 1:1000. The blots were imaged with an infrared imaging system (Odyssey, LI-COR Biosciences).
CRISPR-Cas9
CRISPR guide RNAs were designed using the Joung Lab zifit site: http://zifit.partners.org/ZiFiT/Disclaimer.aspx.
Exon 2: Oligo1-TAGGTGGGCTGGTCAAGCAAGC, Oligo2-AAACGCTTGCTTGACCAGCCCA; Exon 4: Oligo1-TAGGTGATCCCTACCCCTTTGA, Oligo2-AAACTCAAAGGGGTAGGGATCA.
The pT7 gRNA vector (Addgene 46759) was digested with BsmBI, BglII and SalI (New England Biolabs) and diluted to 5 ng/µl in ddH2O. Annealed oligos were ligated into vector using quick ligase (New England Biolabs). Single colonies were selected following transformation and digest-confirmed with BglII. Candidate plasmids were sequenced using M13 primer. Sequence-confirmed plasmids were linearized with BamHI (New England Biolabs) and in vitro-transcribed using MAXIScript T7 kit (Ambion, Life Technologies). The resulting guide RNAs were injected into the yolk at the one-cell stage at a volume of 2 nl. The final concentrations of the injection mixes were as follows: gRNAs at ∼40 ng/μl and Cas9 protein (New England Biolabs) at ∼55 ng/μl. To confirm, 2- to 5-dpf larval zebrafish were individually lysed in 50 µl of 50 mM NaOH at 95°C for 10 min. Samples were cooled to 4°C and mixed with 1 M Tris-HCl (pH 8.0). Supernatant was collected after 5 min, 2000 g centrifugation. PCR was performed on gDNA to confirm with the following primers for mmp9: intron 1 forward: 5′-GTTTGGGTTTCTCCTCTCGGTTCTCTACT-3′; intron 4 reverse: 5′-AACTGCAGAGGGAGCTATTGCTCCTC-3′.
Supplementary Material
Acknowledgements
We thank the laboratory members for their critical reading of the manuscript.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Funding
This work was supported by the National Institutes of Health [GM074827 and GM102924 to A.H.] and by the European Commission [LSHG-2006-037220 to A.Z. and A.H.M.]. Further support came from the Laboratory for Optical and Computational Instrumentation (LOCI) and support from the UW-UWM Intercampus research program (to K.W.E.). Deposited in PMC for release after 12 months.
Author contributions
D.C.L. performed most of the experiments, collected and analyzed data, prepared figures and wrote the manuscript. J.M.S. performed the SHG imaging and revised the manuscript. J.R. performed the in situ hybridization and isolated RNA for the microarray analysis. E.B. assisted the performing of experiments and collection of data. Y.L. developed the program for analysis of SHG imaging. A.Z. performed the microarray analysis. K.W.E. advised on the imaging, imaging study design and computational analysis. A.H. contributed to data analysis and co-wrote the manuscript.
Supplementary material
Supplementary material available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.121160/-/DC1
References
- Bai S., Thummel R., Godwin A. R., Nagase H., Itoh Y., Li L., Evans R., McDermott J., Seiki M. and Sarras M. P. Jr (2005). Matrix metalloproteinase expression and function during fin regeneration in zebrafish: analysis of MT1-MMP, MMP2 and TIMP2. Matrix Biol. 24, 247-260. 10.1016/j.matbio.2005.03.007 [DOI] [PubMed] [Google Scholar]
- Betsuyaku T., Fukuda Y., Parks W. C., Shipley J. M. and Senior R. M. (2000). Gelatinase B is required for alveolar bronchiolization after intratracheal bleomycin. Am. J. Pathol. 157, 525-535. 10.1016/S0002-9440(10)64563-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bredfeldt J. S., Liu Y., Pehlke C. A., Conklin M. W., Szulczewski J. M., Inman D. R., Keely P. J., Nowak R. D., Mackie T. R. and Eliceiri K. W. (2014). Computational segmentation of collagen fibers from second-harmonic generation images of breast cancer. J. Biomed. Opt. 19, 016007 10.1117/1.JBO.19.1.016007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campagnola P. J., Millard A. C., Terasaki M., Hoppe P. E., Malone C. J. and Mohler W. A. (2002). Three-dimensional high-resolution second-harmonic generation imaging of endogenous structural proteins in biological tissues. Biophys. J. 82, 493-508. 10.1016/S0006-3495(02)75414-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Candel S., de Oliveira S., López-Muñoz A., García-Moreno D., Espín-Palazón R., Tyrkalska S. D., Cayuela M. L., Renshaw S. A., Corbalán-Vélez R., Vidal-Abarca I. et al. (2014). Tnfa signaling through tnfr2 protects skin against oxidative stress–induced inflammation. PLoS Biol. 12, e1001855 10.1371/journal.pbio.1001855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carney T. J., von der Hardt S., Sonntag C., Amsterdam A., Topczewski J., Hopkins N. and Hammerschmidt M. (2007). Inactivation of serine protease Matriptase1a by its inhibitor Hai1 is required for epithelial integrity of the zebrafish epidermis. Development 134, 3461-3471. 10.1242/dev.004556 [DOI] [PubMed] [Google Scholar]
- Castaneda F. E., Walia B., Vijay-Kumar M., Patel N. R., Roser S., Kolachala V. L., Rojas M., Wang L., Oprea G., Garg P. et al. (2005). Targeted deletion of metalloproteinase 9 attenuates experimental colitis in mice: central role of epithelial-derived MMP. Gastroenterology 129, 1991-2008. 10.1053/j.gastro.2005.09.017 [DOI] [PubMed] [Google Scholar]
- Collier I. E., Wilhelm S. M., Eisen A. Z., Marmer B. L., Grant G. A., Seltzer J. L., Kronberger A., He C. S., Bauer E. A. and Goldberg G. I. (1988). H-ras oncogene-transformed human bronchial epithelial cells (TBE-1) secrete a single metalloprotease capable of degrading basement membrane collagen. J. Biol. Chem. 263, 6579-6587. [PubMed] [Google Scholar]
- Conklin M. W., Provenzano P. P., Eliceiri K. W., Sullivan R. and Keely P. J. (2009). Fluorescence lifetime imaging of endogenous fluorophores in histopathology sections reveals differences between normal and tumor epithelium in carcinoma in situ of the breast. Cell Biochem. Biophys. 53, 145-157. 10.1007/s12013-009-9046-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Oliveira S., Lopez-Munoz A., Candel S., Pelegrin P., Calado A. and Mulero V. (2014). ATP modulates acute inflammation in vivo through dual oxidase 1-derived H2O2 production and NF-kappaB activation. J. Immunol. 192, 5710-5719. 10.4049/jimmunol.1302902 [DOI] [PubMed] [Google Scholar]
- Dodd M. E., Hatzold J., Mathias J. R., Walters K. B., Bennin D. A., Rhodes J., Kanki J. P., Look A. T., Hammerschmidt M. and Huttenlocher A. (2009). The ENTH domain protein Clint1 is required for epidermal homeostasis in zebrafish. Development 136, 2591-2600. 10.1242/dev.038448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eberhardt W., Schulze M., Engels C., Klasmeier E. and Pfeilschifter J. (2002). Glucocorticoid-mediated suppression of cytokine-induced matrix metalloproteinase-9 expression in rat mesangial cells: involvement of nuclear factor-kappaB and Ets transcription factors. Mol. Endocrinol. 16, 1752-1766. 10.1210/me.2001-0278 [DOI] [PubMed] [Google Scholar]
- Eickelberg O., Kohler E., Reichenberger F., Bertschin S., Woodtli T., Erne P., Perruchoud A. P. and Roth M. (1999). Extracellular matrix deposition by primary human lung fibroblasts in response to TGF-beta1 and TGF-beta3. Am. J. Physiol. 276, L814-L824. [DOI] [PubMed] [Google Scholar]
- Eming S. A., Krieg T. and Davidson J. M. (2007). Inflammation in wound repair: molecular and cellular mechanisms. J. Invest. Dermatol. 127, 514-525. 10.1038/sj.jid.5700701 [DOI] [PubMed] [Google Scholar]
- Feghali C. A. and Wright T. M. (1997). Cytokines in acute and chronic inflammation. Front. Biosci. 2, d12-d26. [DOI] [PubMed] [Google Scholar]
- Fini M. E., Parks W. C., Rinehart W. B., Girard M. T., Matsubara M., Cook J. R., West-Mays J. A., Sadow P. M., Burgeson R. E., Jeffrey J. J. et al. (1996). Role of matrix metalloproteinases in failure to re-epithelialize after corneal injury. Am. J. Pathol. 149, 1287-1302. [PMC free article] [PubMed] [Google Scholar]
- Fosang A. J., Neame P. J., Last K., Hardingham T. E., Murphy G. and Hamilton J. A. (1992). The interglobular domain of cartilage aggrecan is cleaved by PUMP, gelatinases, and cathepsin B. J. Biol. Chem. 267, 19470-19474. [PubMed] [Google Scholar]
- Ganguly K., Rejmak E., Mikosz M., Nikolaev E., Knapska E. and Kaczmarek L. (2013). Matrix metalloproteinase (MMP) 9 transcription in mouse brain induced by fear learning. J. Biol. Chem. 288, 20978-20991. 10.1074/jbc.M113.457903 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Hall C. J., Boyle R. H., Sun X., Wicker S. M., Misa J. P., Krissansen G. W., Print C. G., Crosier K. E. and Crosier P. S. (2014). Epidermal cells help coordinate leukocyte migration during inflammation through fatty acid-fuelled matrix metalloproteinase production. Nat. Commun. 5, 3880 10.1038/ncomms4880 [DOI] [PubMed] [Google Scholar]
- Hattori N., Mochizuki S., Kishi K., Nakajima T., Takaishi H., D'Armiento J. and Okada Y. (2009). MMP-13 plays a role in keratinocyte migration, angiogenesis, and contraction in mouse skin wound healing. Am. J. Pathol. 175, 533-546. 10.2353/ajpath.2009.081080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jowett T. and Yan Y.-L. (1996). Double fluorescent in situ hybridization to zebrafish embryos. Trends Genet. 12, 387-389. 10.1016/S0168-9525(96)90091-8 [DOI] [PubMed] [Google Scholar]
- Kanther M., Sun X., Mühlbauer M., Mackey L. C., Flynn E. J. III, Bagnat M., Jobin C. and Rawls J. F. (2011). Microbial colonization induces dynamic temporal and spatial patterns of NF-kappaB activation in the zebrafish digestive tract. Gastroenterology 141, 197-207. 10.1053/j.gastro.2011.03.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam P.-Y., Harvie E. A. and Huttenlocher A. (2013). Heat shock modulates neutrophil motility in zebrafish. PLoS ONE 8, e84436 10.1371/journal.pone.0084436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Guyader D., Redd M. J., Colucci-Guyon E., Murayama E., Kissa K., Briolat V., Mordelet E., Zapata A., Shinomiya H. and Herbomel P. (2008). Origins and unconventional behavior of neutrophils in developing zebrafish. Blood 111, 132-141. 10.1182/blood-2007-06-095398 [DOI] [PubMed] [Google Scholar]
- LeBert D. C. and Huttenlocher A. (2014). Inflammation and wound repair. Semin. Immunol. 26, 315-320. 10.1016/j.smim.2014.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leibovich S. J. and Ross R. (1975). The role of the macrophage in wound repair. A study with hydrocortisone and antimacrophage serum. Am. J. Pathol. 78, 71-100. [PMC free article] [PubMed] [Google Scholar]
- Li L., Jin H., Xu J., Shi Y. and Wen Z. (2011). Irf8 regulates macrophage versus neutrophil fate during zebrafish primitive myelopoiesis. Blood 117, 1359-1369. 10.1182/blood-2010-06-290700 [DOI] [PubMed] [Google Scholar]
- Li L., Yan B., Shi Y.-Q., Zhang W.-Q. and Wen Z.-L. (2012). Live imaging reveals differing roles of macrophages and neutrophils during zebrafish tail fin regeneration. J. Biol. Chem. 287, 25353-25360. 10.1074/jbc.M112.349126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieschke G. J., Oates A. C., Crowhurst M. O., Ward A. C. and Layton J. E. (2001). Morphologic and functional characterization of granulocytes and macrophages in embryonic and adult zebrafish. Blood 98, 3087-3096. 10.1182/blood.V98.10.3087 [DOI] [PubMed] [Google Scholar]
- Martin P., D'Souza D., Martin J., Grose R., Cooper L., Maki R. and McKercher S. R. (2003). Wound healing in the PU.1 null mouse – tissue repair is not dependent on inflammatory cells. Curr. Biol. 13, 1122-1128. 10.1016/S0960-9822(03)00396-8 [DOI] [PubMed] [Google Scholar]
- Mathew L. K., Sengupta S., Kawakami A., Andreasen E. A., Lohr C. V., Loynes C. A., Renshaw S. A., Peterson R. T. and Tanguay R. L. (2007). Unraveling tissue regeneration pathways using chemical genetics. J. Biol. Chem. 282, 35202-35210. 10.1074/jbc.M706640200 [DOI] [PubMed] [Google Scholar]
- Mathias J. R., Dodd M. E., Walters K. B., Rhodes J., Kanki J. P., Look A. T. and Huttenlocher A. (2007). Live imaging of chronic inflammation caused by mutation of zebrafish Hai1. J. Cell Sci. 120, 3372-3383. 10.1242/jcs.009159 [DOI] [PubMed] [Google Scholar]
- McCarty S. M. and Percival S. L. (2013). Proteases and delayed wound healing. Adv. Wound Care 2, 438-447. 10.1089/wound.2012.0370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCawley L. J., O'Brien P. and Hudson L. G. (1998). Epidermal growth factor (EGF)- and scatter factor/hepatocyte growth factor (SF/HGF)-mediated keratinocyte migration is coincident with induction of matrix metalloproteinase (MMP)-9. J. Cell. Physiol. 176, 255-265. 10.1002/(SICI)1097-4652(199808)176:2<255::AID-JCP4>3.0.CO;2-N [DOI] [PubMed] [Google Scholar]
- Mohan R., Chintala S. K., Jung J. C., Villar W. V. L., McCabe F., Russo L. A., Lee Y., McCarthy B. E., Wollenberg K. R., Jester J. V. et al. (2002). Matrix metalloproteinase gelatinase B (MMP-9) coordinates and effects epithelial regeneration. J. Biol. Chem. 277, 2065-2072. 10.1074/jbc.M107611200 [DOI] [PubMed] [Google Scholar]
- Mohler W., Millard A. C. and Campagnola P. J. (2003). Second harmonic generation imaging of endogenous structural proteins. Methods 29, 97-109. 10.1016/S1046-2023(02)00292-X [DOI] [PubMed] [Google Scholar]
- Nathan C. (2006). Neutrophils and immunity: challenges and opportunities. Nat. Rev. Immunol. 6, 173-182. 10.1038/nri1785 [DOI] [PubMed] [Google Scholar]
- O'Farrell T. J. and Pourmotabbed T. (2000). Identification of structural elements important for matrix metalloproteinase type V collagenolytic activity as revealed by chimeric enzymes. Role of fibronectin-like domain and active site of gelatinase B. J. Biol. Chem. 275, 27964-27972. [DOI] [PubMed] [Google Scholar]
- Parks W. C., Wilson C. L. and López-Boado Y. S. (2004). Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat. Rev. Immunol. 4, 617-629. 10.1038/nri1418 [DOI] [PubMed] [Google Scholar]
- Petrie T. A., Strand N. S., Tsung-Yang C., Rabinowitz J. S. and Moon R. T. (2014). Macrophages modulate adult zebrafish tail fin regeneration. Development 141, 2581-2591. 10.1242/dev.098459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poss K. D., Keating M. T. and Nechiporuk A. (2003). Tales of regeneration in zebrafish. Dev. Dyn. 226, 202-210. 10.1002/dvdy.10220 [DOI] [PubMed] [Google Scholar]
- Powell F. C., Schroeter A. L., Su W. P. and Perry H. O. (1985). Pyoderma gangrenosum: a review of 86 patients. Q. J. Med. 55, 173-186. [PubMed] [Google Scholar]
- Rhodes J., Hagen A., Hsu K., Deng M., Liu T. X., Look A. T. and Kanki J. P. (2005). Interplay of pu.1 and gata1 determines myelo-erythroid progenitor cell fate in zebrafish. Dev. Cell 8, 97-108. 10.1016/j.devcel.2004.11.014 [DOI] [PubMed] [Google Scholar]
- Schindelin J., Arganda-Carreras I., Frise E., Kaynig V., Longair M., Pietzsch T., Preibisch S., Rueden C., Saalfeld S., Schmid B. et al. (2012). Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676-682. 10.1038/nmeth.2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Senior R. M., Griffin G. L., Fliszar C. J., Shapiro S. D., Goldberg G. I. and Welgus H. G. (1991). Human 92- and 72-kilodalton type IV collagenases are elastases. J. Biol. Chem. 266, 7870-7875. [PubMed] [Google Scholar]
- Shiratori Y., Hongo S., Hikiba Y., Ohmura K., Nagura T., Okano K., Kamii K., Tanaka T., Komatsu Y., Ochiai T. et al. (1996). Role of macrophages in regeneration of liver. Dig. Dis. Sci. 41, 1939-1946. 10.1007/BF02093593 [DOI] [PubMed] [Google Scholar]
- Starnes T. W. and Huttenlocher A. (2012). Neutrophil reverse migration becomes transparent with zebrafish. Adv. Hematol. 2012, 398640 10.1155/2012/398640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stockhammer O. W., Zakrzewska A., Hegedus Z., Spaink H. P. and Meijer A. H. (2009). Transcriptome profiling and functional analyses of the zebrafish embryonic innate immune response to Salmonella infection. J. Immunol. 182, 5641-5653. 10.4049/jimmunol.0900082 [DOI] [PubMed] [Google Scholar]
- Tidball J. G. and Wehling-Henricks M. (2007). Macrophages promote muscle membrane repair and muscle fibre growth and regeneration during modified muscle loading in mice in vivo. J. Physiol. 578, 327-336. 10.1113/jphysiol.2006.118265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Steen P. E., Proost P., Grillet B., Brand D. D., Kang A. H., Van Damme J. and Opdenakker G. (2002). Cleavage of denatured natural collagen type II by neutrophil gelatinase B reveals enzyme specificity, post-translational modifications in the substrate, and the formation of remnant epitopes in rheumatoid arthritis. FASEB J. 16, 379-389. 10.1096/fj.01-0688com [DOI] [PubMed] [Google Scholar]
- van Furth R., Nibbering P. H., van Dissel J. T. and Diesselhoff-den Dulk M. M. C. (1985). The characterization, origin, and kinetics of skin macrophages during inflammation. J. Invest. Dermatol. 85, 398-402. 10.1111/1523-1747.ep12277056 [DOI] [PubMed] [Google Scholar]
- Vandooren J., Van den Steen P. E. and Opdenakker G. (2013). Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9): the next decade. Crit. Rev. Biochem. Mol. Biol. 48, 222-272. 10.3109/10409238.2013.770819 [DOI] [PubMed] [Google Scholar]
- Volkman H. E., Pozos T. C., Zheng J., Davis J. M., Rawls J. F. and Ramakrishnan L. (2010). Tuberculous granuloma induction via interaction of a bacterial secreted protein with host epithelium. Science 327, 466-469. 10.1126/science.1179663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wyatt R. A., Keow J. Y., Harris N. D., Haché C. A., Li D. H. and Crawford B. D. (2009). The zebrafish embryo: a powerful model system for investigating matrix remodeling. Zebrafish 6, 347-354. 10.1089/zeb.2009.0609 [DOI] [PubMed] [Google Scholar]
- Yang E. V., Gardiner D. M., Carlson M. R. J., Nugas C. A. and Bryant S. V. (1999). Expression of Mmp-9 and related matrix metalloproteinase genes during axolotl limb regeneration. Dev. Dyn. 216, 2-9. 10.1002/(SICI)1097-0177(199909)216:1<2::AID-DVDY2>3.0.CO;2-P [DOI] [PubMed] [Google Scholar]
- Yates C. C., Hebda P. and Wells A. (2012). Skin wound healing and scarring: fetal wounds and regenerative restitution. Birth Defects Res. C Embryo Today 96, 325-333. 10.1002/bdrc.21024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo S. K. and Huttenlocher A. (2011). Spatiotemporal photolabeling of neutrophil trafficking during inflammation in live zebrafish. J. Leukoc. Biol. 89, 661-667. 10.1189/jlb.1010567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo S. K., Deng Q., Cavnar P. J., Wu Y. I., Hahn K. M. and Huttenlocher A. (2010). Differential regulation of protrusion and polarity by PI(3)K during neutrophil motility in live zebrafish. Dev. Cell 18, 226-236. 10.1016/j.devcel.2009.11.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo S. K., Freisinger C. M., LeBert D. C. and Huttenlocher A. (2012). Early redox, Src family kinase, and calcium signaling integrate wound responses and tissue regeneration in zebrafish. J. Cell Biol. 199, 225-234. 10.1083/jcb.201203154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshinari N., Ishida T., Kudo A. and Kawakami A. (2009). Gene expression and functional analysis of zebrafish larval fin fold regeneration. Dev. Biol. 325, 71-81. 10.1016/j.ydbio.2008.09.028 [DOI] [PubMed] [Google Scholar]
- Zakrzewska A., Cui C., Stockhammer O. W., Benard E. L., Spaink H. P., Meijer A. H. (2010). Macrophage-specific gene functions in Spi1-directed innate immunity. Blood 116, e1-e11. 10.1182/blood-2010-01-262873 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.