Abstract
BACKGROUND AND OBJECTIVE:
Delayed cord clamping (DCC) is recommended for premature infants to improve blood volume. Most preterm infants are born by cesarean delivery (CD), and placental transfusion may be less effective than in vaginal delivery (VD). We sought to determine whether infants <32 weeks born by CD who undergo umbilical cord milking (UCM) have higher measures of systemic blood flow than infants who undergo DCC.
METHODS:
This was a 2-center trial. Infants delivered by CD were randomly assigned to undergo UCM or DCC. Infants delivered by VD were also randomly assigned separately. UCM (4 strippings) or DCC (45–60 seconds) were performed. Continuous hemodynamic measurements and echocardiography were done at site 1.
RESULTS:
A total of 197 infants were enrolled (mean gestational age 28 ± 2 weeks). Of the 154 infants delivered by CD, 75 were assigned to UCM and 79 to DCC. Of the infants delivered by CD, neonates randomly assigned to UCM had higher superior vena cava flow and right ventricular output in the first 12 hours of life. Neonates undergoing UCM also had higher hemoglobin, delivery room temperature, blood pressure over the first 15 hours, and urine output in the first 24 hours of life. There were no differences for the 43 infants delivered by VD.
CONCLUSIONS:
This is the first randomized controlled trial demonstrating higher systemic blood flow with UCM in preterm neonates compared with DCC. UCM may be a more efficient technique to improve blood volume in premature infants delivered by CD.
What’s Known on This Subject:
Delayed cord clamping is recommended for all premature births, despite some studies suggesting a decreased placental transfusion at cesarean delivery.
What This Study Adds:
Umbilical cord milking appears to improve systemic blood flow and perfusion in preterm infants delivered by cesarean delivery more efficiently than delayed cord clamping.
In December 2012, the American College of Obstetricians and Gynecologists (ACOG) recommended a 30- to 60-second delay in umbilical cord clamping for all preterm deliveries.1 Although delayed cord clamping (DCC) has decreased the overall incidence of intraventricular hemorrhage (IVH), the enthusiasm for DCC is tempered by the lack of benefit for severe IVH and the small number of infants included in these trials.2 The lack of benefit could reflect the lack of adequate placental transfusion during DCC for infants delivered by cesarean delivery (CD).3–5 Three trials of DCC that stratified by mode of delivery found no difference in hematocrit levels or tagged red blood cells in infants delivered by CD.3–5 The ACOG statement acknowledges that there are limited data indicating whether DCC performed during CD can improve placental transfusion.1
An alternative to DCC is umbilical cord milking (UCM), or stripping, in which the unclamped umbilical cord is grasped and blood is pushed toward the infant several times before it is clamped to autoinfuse blood into the preterm neonate. This procedure can be performed within 20 seconds.6 A recent meta-analysis of 7 randomized controlled trials of UCM in infants delivered at <33 weeks demonstrated that infants who undergo UCM have higher hemoglobin (Hb) and a lower risk for oxygen requirement at 36 weeks and IVH of all grades compared with those who undergo immediate cord clamping (ICC).7
A recently published study compared UCM with DCC (n = 58) in infants delivered at <33 weeks’ gestational age.8 Although no major clinical differences were found between UCM and DCC, this trial did not analyze any outcomes by mode of delivery. To date there have been no trials comparing UCM and DCC at CD. Given that up to 90% of preterm infants are delivered by CD, there is a critical need to determine which therapy should be given to preterm infants.9 We hypothesized that UCM at CD could improve perfusion, or systemic blood flow (SBF), in preterm neonates as measured by superior vena cava (SVC) flow and right ventricular output (RVO) and would be associated with decreased neonatal morbidities compared with DCC, and that UCM may stabilize blood pressure and cerebral oxygenation as measured by near-infrared spectroscopy (NIRS).
Methods
This randomized controlled trial was conducted at 2 tertiary care centers (Sharp Mary Birch Hospital for Women and Newborns [SMBHWN] and Loma Linda University Medical Center) and was approved by each institutional review board. Pregnant women dated by their earliest ultrasound or last menstrual period at <32 weeks’ gestation were identified and recruited from the labor and delivery and antepartum floors. Both interventions were part of standard practice and were considered minimally risky.
In this study, antenatal consent was not practical because it would exclude the potentially sickest newborns.10,11 UCM and DCC are both standard practices at SMBHWN and as such are left to the preference of the obstetrician. There are no current guidelines for their use in the preterm infant, and they are of equivalent low risk. Our study met the criteria for delayed consent based on the inability to conduct the trial without a waiver and minimal risk of either intervention. As with our previous trial using delayed consent, parents were notified of the intervention by the obstetrician or research team and were approached immediately after birth to provide written consent to enroll their newborn for continued data collection before any study-related procedures (eg, ultrasounds, additional monitoring).12 If a parent did not want to enroll their child in the study, we removed the subject from our study and destroyed all data that had been collected.
Exclusion criteria included monochorionic multiples, incarcerated mothers, placenta previa, concern for abruptions, Rh sensitization, hydrops, congenital anomalies, or the obstetrician declining to perform the intervention (ie, unaware of the study protocol). We did included infants with perinatal depression because it would not be feasible to detect perinatal depression at the time of delivery. Entry criteria included a gestational age of 23 0/7 to 31 6/7 weeks.
Infants were randomly assigned by opaque, sealed envelopes immediately before delivery. Computer-generated randomization was stratified by gestational age and mode of delivery (23–27 6/7 or 28–31 6/7 weeks) to ensure equal numbers of neonates born at <28 weeks’ gestation in each arm. The obstetricians were made aware of the randomization by the neonatology team immediately before delivery of the infant. Multiples (twins or triplets) received the same random assignment.
Using the Apgar timer on the resuscitation bed, the advanced life support (ALS) nurses or labor and delivery staff attending the delivery recorded and counted out loud the time elapsed from when the infant was delivered until the time the umbilical cord was clamped by the obstetrician in both arms of the study. UCM was performed by holding the infant at or ∼20 cm below the level of the placenta. The cord was pinched as close to the placenta as possible and milked toward the infant over a 2-second duration. The cord was then released and allowed to refill with blood for a brief 1- to 2-second pause between each milking motion. This was repeated for a total of 4 times. After completion, the cord was clamped, and the neonate was handed to the resuscitation team.8 DCC was performed by holding the infant at or ∼20 cm below the level of the placenta and waiting at least 45 seconds before clamping the cord. In both arms infants were dried and wrapped with sterile towels until the cord was clamped.
Hemodynamic measurements were only performed at site 1 (SMBHWN). These measures included an early echocardiogram (target 6–12 hours of life) and continuous hemodynamic recordings. The echocardiogram was performed with the Vivid E9 cardiovascular ultrasound system with a 12S phased array transducer (GE Medical Systems, Milwaukee, WI). At the time of the first scan all neonates also had a bedside head ultrasound to document evidence of IVH. Formal head ultrasounds were performed on the second or third day of life according to our unit protocol.
Blinded echocardiograms and head ultrasounds were performed mainly (>90%) by the principal investigator (A.C.K.). None of the investigators performing echocardiograms were involved in the randomization or the recording of the intervention. All images were analyzed and measured offline by using EchoPAC software (GE HealthCare, Horten, Norway) and were analyzed without knowledge of the assigned group by the principal investigator. The blinding was achieved by allowing only the ALS nurse attending the delivery and the obstetrician performing the intervention to be aware of the allocation arm. This was possible because all high-risk deliveries occurred in a room adjacent to the resuscitation suite where senior physicians, nurses, and respiratory therapists are in attendance at both sites. No documentation of the intervention was made in the physician or ALS notes. The randomization cards assigned a subject identification number that was kept by the research coordinator.
Measures of superior vena cava (SVC) flow, right ventricular output (RVO), left ventricular output (LVO), diameter and direction of flow through the patent ductus arteriosus, and diameter and direction of flow through the foramen ovale were obtained on each examination.
Flow measures were calculated by using previously described techniques.13
The primary outcome was the SVC flow within the first 12 hours. Infants were resuscitated according to each unit’s protocol. We attached a cerebral oxygen saturation monitor using 4 different wavelengths (FORE-SIGHT; Casmed, Branford, CT) to the infant by placing a sensor on the infant’s forehead within the first 2 hours of life. Cerebral saturation was recorded for the first 24 hours of life.
Electrical cardiometry (EC) was used as a continuous noninvasive measure of cardiac output. EC uses the properties of bioimpedance to noninvasively measure body composition and blood flow via surface adhesive electrodes. Changes in thoracic impedance during delivery of a low-voltage current indicate cardiac output. We validated the use of EC compared with echocardiographic measures of right and left ventricular output and SVC flow.14 All infants had EC sensors placed on their chest by 2 hours and had data recorded for the first 24 hours of life. Cerebral oxygen saturation, pulse oxygen saturation, heart rate, respiratory rate, blood pressure, cardiac output, and stroke volume were recorded every 2 seconds on a purpose-built data acquisition system.
Data Collection (Both Sites)
A single study-related blood draw (hematocrit at 12 hours from a central or venipuncture) and head ultrasound on day 1 were collected as part of the research protocol. Admission complete blood cell count and head ultrasounds on the second or third day of life were part of routine care. All other relevant prenatal and neonatal data were obtained from the medical records of mother and infant.
Statistical Analysis
An initial pilot study of 30 infants was completed to determine the feasibility and efficacy of the study and revealed a 25% difference in SVC flow at 6 hours between infants treated with UCM and controls. We based our sample calculations on the basis of our previous results and the preliminary evaluation of the first pilot infants. A sample size calculation determined that ≥40 neonates in each group were needed to demonstrate at least a 25% difference in SVC at 12 hours between neonates treated with UCM those treated with DCC with a 2-sided α of .05 and 80% power. However, when a second site that could not perform hemodynamics was added, the sample size was increased (n = 600) to detect change in the incidence of IVH, based on a 50% reduction in IVH from Rabe et al (22% vs 11%, P = .32). This change was added as a secondary outcome to the trial.
We performed statistical analyses by using PASW Statistics 18.0 (Chicago, IL). Normally distributed continuous outcome variables were compared with the unpaired Student’s t test, and nonparametric continuous outcome variables were analyzed with the Mann–Whitney U test. For repeated measurements tests, R (R Foundation for Statistical Computing, Vienna, Austria) and lme4 (linear mixed-effects models using Eigen and S4) were used to perform linear mixed-effects regression (lmer) analysis of the relationship between the respective treatments (ie, UCM and DCC). Two-sided P <.05 was considered significant.
Results
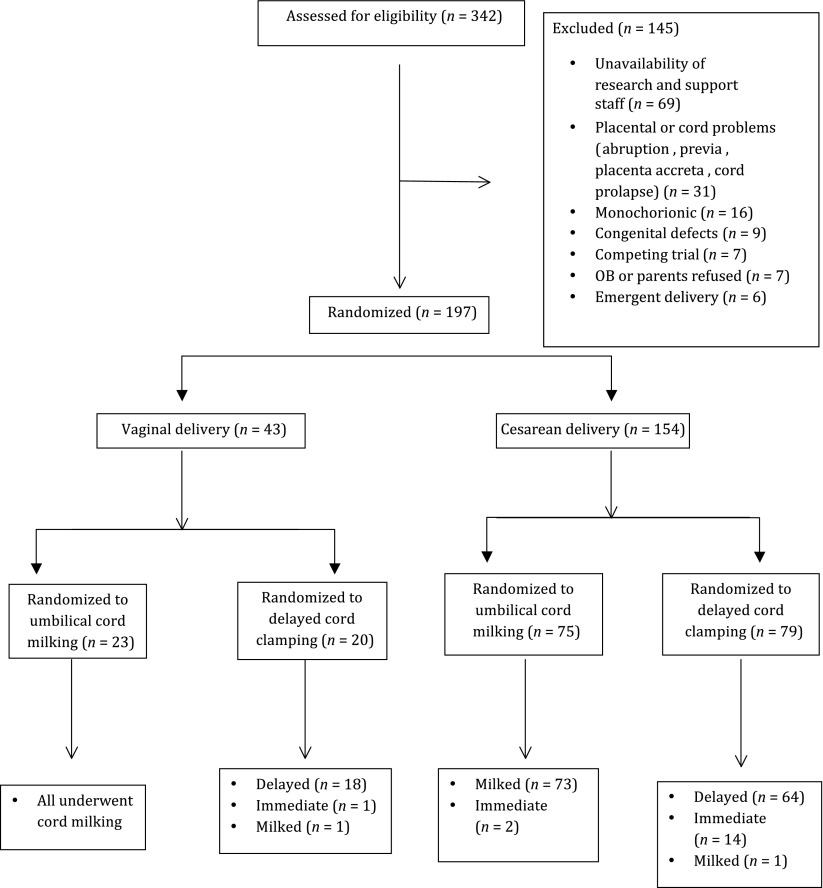
Interim analysis after 197 infants were enrolled (August 2013–August 2014) demonstrated that although the primary outcome (SVC flow) was statistically different (Table 4), the incidence of IVH was much lower than anticipated (expected 22% vs 11%; Rabe et al).8 Based on an actual observed IVH difference of 6% (13% vs 7%), with α = .05 and β = .8, we would have needed at least 780 in each arm. This would have taken 7 years for a 2-center study to complete. Therefore, the trial was stopped on the grounds of futility for the outcome of IVH (154 were delivered by CD; Fig 1). All data were analyzed as intent to treat. From site 1, 150 subjects were enrolled; 140 (n = 70 each group) had complete hemodynamics (functional echocardiography, NIRS, EC, and bedside monitoring performed), and 10 had no or partial hemodynamic data because of unavailability of staff. For site 2 clinical outcomes were collected.
TABLE 4.
Hemodynamic Outcomes for Infants Delivered by CD
| UCM, N = 75 | DCC, N = 79 | |
|---|---|---|
| Time of echocardiogram, h:min | 7:23 ± 5:53 | 6:28 ± 5:15 |
| SVC flow, mL/kg per min* | 93 ± 24 | 81 ± 29 |
| RVO, mL/kg per min** | 261 ± 80 | 216 ± 73 |
| Diameter of atrial shunt, mm | 1.78 ± 0.81 | 1.58 ± 0.65 |
| Diameter of PDA, mm | 1.44 ± 0.67 | 1.45 ± 0.66 |
| LVO, mL/kg per min | 189 ± 67 | 206 ± 79 |
| Need for pressors | 9 | 18 |
| PDA necessitating treatment | 17 | 25 |
| PDA ligation | 4 | 1 |
Data are presented as mean ± SD unless otherwise stated. PDA, patent ductus arteriosus.
P < .05; **P < .001.
FIGURE 1.
CONSORT diagram: study enrollment, intended randomization, and actual randomization.
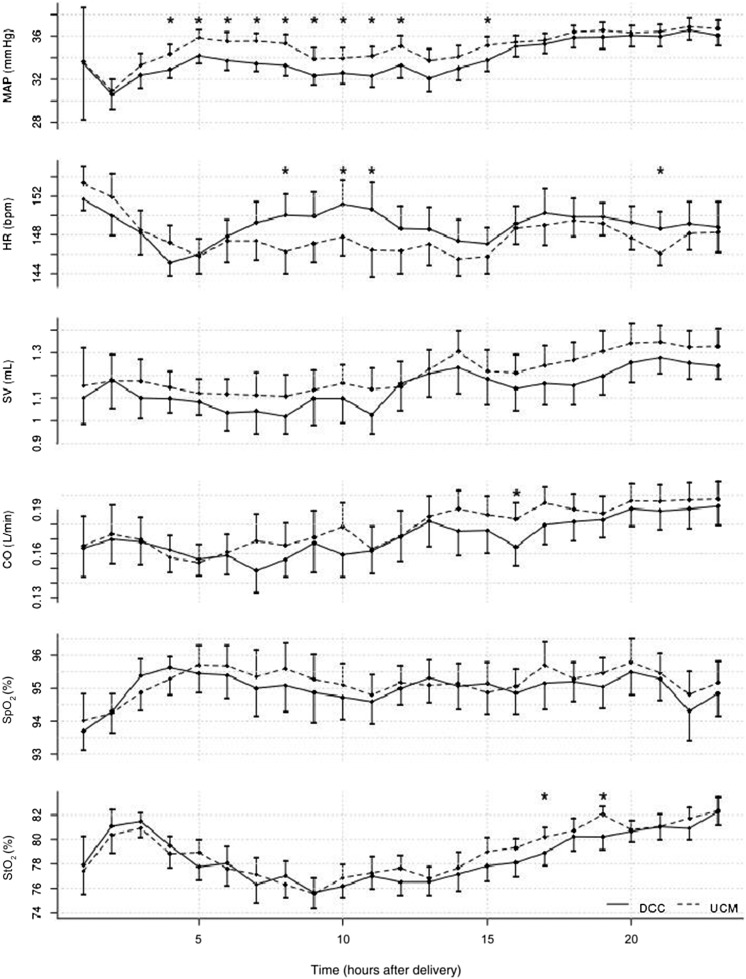
Data for infants delivered by CD are shown in Tables 1, 2, 3, and 4. Neonates who underwent UCM had higher admission Hb, higher urine output (Table 3), and higher measures of SBF (SVC flow and RVO, Table 4). There were no differences in cerebral saturation, pulse oxygen saturation, cardiac output by impedance, or heart rate over 24 hours (Fig 2). Blood pressure was higher in the first 15 hours of life in the UCM group (Fig 2).
TABLE 1.
Perinatal Outcomes for Infants Delivered by CD
| UCM (n = 75) | DCC (n = 79) | |
|---|---|---|
| Maternal age, y | 31 ± 5 | 30 ± 6 |
| Gestational age, wk | 28 ± 2 | 28 ± 2 |
| Birth wt, g | 1255 ± 413 | 1132 ± 392 |
| Placenta wt, g | 257 ± 95 | 245 ± 109 |
| Female | 46 | 48 |
| Diabetes (gestational or type 1 or 2) | 11 | 15 |
| Chorioamnionitis | 20 | 20 |
| Pregnancy-induced hypertension | 22 | 25 |
| Labor or uterotonics before delivery | 27 | 20 |
| Narcotics given before delivery | 16 | 19 |
| Lowest maternal Hb (48 h after delivery), g/dL | 10 ± 2.7 | 9.8 ± 1.6 |
| Duration of rupture of membranes, h | 63 ± 169 | 67 ± 147 |
| General anesthesia | 6 | 2 |
| Full course antenatal steroids | 69 | 75 |
| Antenatal magnesium | 72 | 64 |
Data are presented as mean ± SD. There were no significant group differences at baseline.
TABLE 2.
Delivery Room Outcomes for Infants Delivered by CD
| UCM, N = 75 | DCC, N = 79 | |
|---|---|---|
| Time clamp cord, s** | 20 ± 10 | 42 ± 12 |
| Delivery room temperature, °C | 36.8 ± 0.4 | 36.6 ± 0.4 |
| Admission temperature, °C | 36.8 ± 0.6 | 36.7 ± 0.4 |
| Apgar score, 1 min, 5 min (median) | 5, 7 | 5, 7 |
| Needed PPV | 57 | 56 |
| Intubation in delivery room | 28 | 33 |
Data are presented as mean ± SD unless otherwise stated. PPV, positive-pressure ventilation.
P < .001.
TABLE 3.
Neonatal Outcomes for Infants Delivered by CD
| UCM, N = 75 | DCC, N = 79 | |
|---|---|---|
| Birth Hb, g/dL* | 16.3 ± 2.4 | 15.6 ± 2.2 |
| Polycythemia (hematocrit >65%) | 2 | 4 |
| Urine output first 24 h, mL/kg per h* | 4.42 ± 1.3 | 3.99 ± 1.2 |
| Need for transfusion | 31 | 41 |
| Peak bilirubin, mg/dL | 8.1 ± 2.9 | 7.3 ± 2.2 |
| Necrotizing enterocolitis | 1 | 0 |
| Retinopathy necessitating surgery | 1 | 2 |
| Spontaneous intestinal perforation | 3 | 2 |
| Oxygen at 36 wk corrected | 16 | 12 |
| Any IVH | 5 | 10 |
| Severe IVH (≥grade 3)a | 3 | 3 |
| Sepsis (culture positive) | 5 | 3 |
| Death | 2 | 6 |
Data are presented as mean ± SD unless otherwise stated. * P < .05.
One infant had early-onset sepsis (blood culture positive at birth); otherwise, all sepsis was late onset.
FIGURE 2.
Continuous hemodynamic measurements over 24 hours of life. Dotted line, UCM; solid line, DCC; CO, cardiac output by electrical cardiometry; HR, heart rate; MAP, mean arterial pressure; Spo2, pulse oxygen saturation; SV, stroke volume by electrical cardiometry; Sto2, cerebral saturations by near-infrared spectroscopy. *Paired t test = P < .05 for hourly averages. MAP was significantly higher in UCM infants treated with UCM for the first 15 hours (HR 3–15 beats per minute; χ2 = 5.05, P = .02).
There were no differences in vaginally delivered infants (Supplemental Table 5).
Discussion
Our study is the largest study comparing UCM with DCC in infants delivered by CD and the first to demonstrate improvements in placental transfusion, as seen by higher Hb at birth, improved hemodynamics (higher measures of blood flow and improved blood pressure), and improved urine output with UCM compared with DCC in premature infants delivered by CD. In addition, although there are 15 randomized controlled trials of a ≥60-second delay in cord clamping in term infants, this is the first study of preterm infants evaluating a delay >30 seconds.15
Rabe et al randomly assigned 58 neonates born at <33 weeks’ gestation to UCM (4 times) or to a 30-second delay in cord clamping. Although they did not find any differences in outcomes or Hb levels, the infants treated with DCC had a lower CD rate (58% vs 78%).8 Because a greater number of infants undergoing DCC were delivered by VD, the lower proportion of CD in this group may have reduced the difference seen between the 2 approaches. Aladangady et al3 reported lower circulating red cell volume with DCC in neonates born by CD compared with VD. One could speculate whether more blood remains in the placenta when a neonate is delivered by CD because the anesthetic and surgical interventions interfere with the active contraction of the uterine muscles to expel the placenta.
SVC flow and RVO were higher in infants treated with UCM. SVC flow, a measure of SBF, is an important marker of neonatal transition. SVC flow represents cardiac input and therefore is not affected by the presence of fetal shunts. It is therefore a useful measure of SBF in the newborn heart. We also found that RVO was higher after UCM but not LVO. Previous investigators have demonstrated that decreased RVO was closely associated with worsening respiratory disease (defined by fraction of inspired oxygen requirement), severe IVH, and death.11 The higher overall SBFs are probably related to an increased blood volume from placental transfusion, resulting in improved hemodynamics.
We did not demonstrate a difference in LVO by echocardiography or cardiac output measured by electrical impedance. We demonstrated in a previous trial that cardiac output correlated better with LVO than with RVO or SVC flow.14 The main concern with LVO as a measure of early SBF during the cardiovascular transition from fetal to newborn life is the extent to which it is confounded by a left to right ductal shunt, which may be particularly large in preterm infants. In the presence of a ductal shunt, LVO measures both SBF and the flow across the patent ductus arteriosus and therefore significantly overestimates SBF. Therefore, it is not surprising that there was no improvement in cardiac output with UCM.
We found no difference in cerebral oxygenation as measured by NIRS. Because infants had no difference in oxygenation or cardiac output, it is not surprising that cerebral oxygenation was not different. In addition, there may have been a component of cerebral autoregulation that could have affected our results.
Despite no difference in cerebral saturations or impedance-derived cardiac output or stroke volume, infants undergoing UCM had higher urine output and blood pressure, suggesting that these infants had some improvement in organ perfusion within the first 24 hours of life. Improved perfusion during this critical time period may prevent IVH from occurring by stabilizing the fluctuations in SBF that have been proposed as a mechanism for late IVH.16 Although we did not see a statistically significant difference (P = .29) in IVH between groups, we did not have the power to assess this outcome based on the observed occurrence in our study. However, there were fewer absolute numbers of neonates with total and severe IVH in the UCM group, which is consistent with Rabe et al8 comparing UCM with DCC.
An additional advantage for UCM is the rapid time frame for UCM to occur. This not only allows a minimal delay of resuscitation but also reduces the time for the newborn to be cooled in the operative field. Although the difference between the 2 groups in our trial was only 20 seconds, infants undergoing UCM had higher temperatures in the delivery room (at 5 minutes) than infants undergoing DCC. Studies have shown that low birth weight infants undergoing DCC are warmer than those undergoing ICC, possibly because of the warm placental blood entering the newborn.17 UCM allows rapid placement of the newborn under the radiant warmer. The admission temperatures were not different between groups. We suggest that centers with concerns about hypothermia that are considering implementing DCC or UCM monitor temperatures in the delivery room. We have since implemented the use of a warming bed (Lifestart Trolley; Inditherm, Rotherham, UK) that can be used on the sterile field in addition to warm sterile towels to ensure that there is minimal temperature drop in infants in the operating room.
An important limitation of our trial was the lack of an ICC group. Because both DCC and UCM provide a placental transfusion regardless of the mode of delivery, it was expected that we would not see substantial differences in clinical outcomes. Because the recommendations by ACOG demonstrated that DCC improved clinical outcomes,1 we did not have equipoise to randomly assign infants to a third group that did not receive a placental transfusion.
However, 18 infants delivered by CD who were randomly assigned to DCC were thought to be too unstable in the opinion of the obstetrician and underwent ICC. Therefore, the mean time in our trial, by intent to treat, for DCC was 42 seconds. There were 2 neonates in the UCM group who underwent early clamping. When these neonates, who were randomly assigned to either group (UCM or DCC) but underwent ICC, were removed from the analysis there was still a difference in Hb, SVC flow, RVO, and urine output (data not shown).
Another limitation of our study is the small number of infants born at <29 weeks’ gestational age (n = 94). Recently, a pilot study of 75 extremely premature neonates (born at <29 weeks’ gestation age) randomly assigned to UCM or ICC demonstrated a 50% reduction in total IVH.18 In a recent retrospective study of UCM in 318 infants born at <30 weeks, UCM was associated with reductions in IVH, necrotizing enterocolitis, and death before hospital discharge.19 As suggested by these authors, it is possible that UCM may have greater benefits in smaller, more immature neonates.
Despite the concerns that UCM may provide a rapid bolus of blood, our data are consistent with other studies that found that UCM is beneficial with minimal risk. Placental blood during UCM is directed toward the lungs during a time when there is a rapid fall in pulmonary resistance unlike any other period when volume is given. Concerns about rapid changes in venous pressure during cord milking were addressed in an early trial that demonstrated no greater increase in venous pressures with UCM compared with uterine contractions or a newborn cry during intact placental circulation.20
Although there are limited data on neurodevelopmental outcomes in premature infants,21 UCM has been studied in 7 randomized controlled trials and 9 controlled trials over the past 60 years in term and preterm infants (n = 1904), documenting its safety and efficacy.6,20,22–31 A recent meta-analysis evaluating the safety and efficacy of UCM at birth concluded that there was a lower risk for oxygen requirement at 36 weeks and IVH of all grades.7 Therefore, UCM should no longer be considered experimental; rather, it is a proven intervention that ensures that premature newborns receive an adequate placental transfusion at birth.
Conclusions
UCM provides a greater placental transfusion, as demonstrated by higher initial Hb, higher blood pressure, and improved SBF and urine output for infants delivered by CD. UCM may be preferable in preterm infants delivered by CD, particularly in newborns when immediate resuscitation is needed. Although more larger trials are needed to confirm our observations, UCM should be considered as a beneficial option for preterm infants delivered by CD.
Supplementary Material
Acknowledgments
The authors acknowledge Wade Rich, Kathy Arnell, Jane Steen, and Deb Poeltler for their assistance in designing the protocol, collecting and analyzing the data, and editing the manuscript. We also acknowledge the San Diego neonatology physicians and advanced life support nurses for their assistance in randomizing, ensuring compliance with the protocol, and collecting data in the delivery room.
Glossary
- ACOG
American College of Obstetricians and Gynecologists
- ALS
advanced life support
- CD
cesarean delivery
- DCC
delayed cord clamping
- EC
electrical cardiometry
- Hb
hemoglobin
- ICC
immediate cord clamping
- IVH
intraventricular hemorrhage
- LVO
left ventricular output
- NIRS
near-infrared spectroscopy
- RVO
right ventricular output
- SBF
systemic blood flow
- SMBHWN
Sharp Mary Birch Hospital for Women and Newborns
- SVC
superior vena cava
- UCM
umbilical cord milking
- VD
vaginal delivery
Footnotes
Dr Katheria conceptualized and designed the study, drafted the initial manuscript, designed the data collection instruments, and coordinated and supervised data collection at two of the sites; Drs Truong, Cousins, Oshiro, and Finer carried out the initial analyses and reviewed and revised the manuscript; and all authors approved the final manuscript as submitted.
This trial has been registered at clinicaltrials.gov (identifier NCT01866982).
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
FUNDING: All phases of this study were supported by a National Institutes of Health (NIH) grant 5R03HD072934-02. Funded by the National Institutes of Health (NIH).
POTENTIAL CONFLICT OF INTEREST: The authors have indicated they have no potential conflicts of interest to disclose.
COMPANION PAPER: A companion to this article can be found on page 177, and online at www.pediatrics.org/cgi/doi/10.1542/peds.2015-1545.
References
- 1.Committee on Obstetric Practice, American College of Obstetricians and Gynecologists . Committee opinion no. 543: Timing of umbilical cord clamping after birth. Obstet Gynecol. 2012;120(6):1522–1526 [DOI] [PubMed] [Google Scholar]
- 2.Rabe H, Diaz-Rossello JL, Duley L, Dowswell T. Effect of timing of umbilical cord clamping and other strategies to influence placental transfusion at preterm birth on maternal and infant outcomes. Cochrane Database Syst Rev. 2012;8:CD003248. [DOI] [PubMed] [Google Scholar]
- 3.Aladangady N, McHugh S, Aitchison TC, Wardrop CA, Holland BM. Infants’ blood volume in a controlled trial of placental transfusion at preterm delivery. Pediatrics. 2006;117(1):93–98 [DOI] [PubMed] [Google Scholar]
- 4.Strauss RG, Mock DM, Johnson K, et al. Circulating RBC volume, measured with biotinylated RBCs, is superior to the Hct to document the hematologic effects of delayed versus immediate umbilical cord clamping in preterm neonates. Transfusion. 2003;43(8):1168–1172 [DOI] [PubMed] [Google Scholar]
- 5.McDonnell M, Henderson-Smart DJ. Delayed umbilical cord clamping in preterm infants: a feasibility study. J Paediatr Child Health. 1997;33(4):308–310 [DOI] [PubMed] [Google Scholar]
- 6.Katheria AC, Leone TA, Woelkers D, Garey DM, Rich W, Finer NN. The effects of umbilical cord milking on hemodynamics and neonatal outcomes in premature neonates. J Pediatr. 2014;164(5):1045–1050, e1041 [DOI] [PubMed] [Google Scholar]
- 7.Al-Wassia H, Shah PS. Efficacy and safety of umbilical cord milking at birth: a systematic review and meta-analysis. JAMA Pediatr. 2015;169(1):18–25 [DOI] [PubMed] [Google Scholar]
- 8.Rabe H, Jewison A, Alvarez RF, et al. Brighton Perinatal Study Group . Milking compared with delayed cord clamping to increase placental transfusion in preterm neonates: a randomized controlled trial. Obstet Gynecol. 2011;117(2 pt 1):205–211 [DOI] [PubMed] [Google Scholar]
- 9.Bettegowda VR, Dias T, Davidoff MJ, Damus K, Callaghan WM, Petrini JR. The relationship between cesarean delivery and gestational age among US singleton births. Clin Perinatol. 2008;35(2):309–323, v–vi [DOI] [PubMed] [Google Scholar]
- 10.Rich W, Finer NN, Gantz MG, et al. SUPPORT and Generic Database Subcommittees of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network . Enrollment of extremely low birth weight infants in a clinical research study may not be representative. Pediatrics. 2012;129(3):480–484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rich WD, Auten KJ, Gantz MG, et al. National Institute of Child Health and Human Development Neonatal Research Network . Antenatal consent in the SUPPORT trial: challenges, costs, and representative enrollment. Pediatrics. 2010;126(1). Available at: www.pediatrics.org/cgi/content/full/126/1/e215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Katheria AC, Sauberan JB, Akotia D, Rich W, Durham J, Finer NN. A pilot randomized controlled trial of early versus routine caffeine in extremely premature infants [published online ahead of print January 21, 2015]. Am J Perinatol. 2015. Available at: http://www.ncbi.nlm.nih.gov/pubmed/25607226 [DOI] [PubMed] [Google Scholar]
- 13.Katheria A, Leone T. Altered transitional circulation in infants of diabetic mothers with strict antenatal obstetric management: a functional echocardiography study. J Perinatol. 2012;32(7):508–513 [DOI] [PubMed] [Google Scholar]
- 14.Song R, Rich W, Kim JH, Finer NN, Katheria AC. The use of electrical cardiometry for continuous cardiac output monitoring in preterm neonates: a validation study. Am J Perinatol. 2014;31(12):1105–1110 [DOI] [PubMed] [Google Scholar]
- 15.McDonald SJ, Middleton P, Dowswell T, Morris PS. Effect of timing of umbilical cord clamping of term infants on maternal and neonatal outcomes. Cochrane Database Syst Rev. 2013;7:CD004074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kluckow M, Evans N. Low superior vena cava flow and intraventricular haemorrhage in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2000;82(3):F188–F194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kaempf JW, Tomlinson MW, Kaempf AJ, et al. Delayed umbilical cord clamping in premature neonates. Obstet Gynecol. 2012;120(2 pt 1):325–330 [DOI] [PubMed] [Google Scholar]
- 18.March MI, Hacker MR, Parson AW, Modest AM, de Veciana M. The effects of umbilical cord milking in extremely preterm infants: a randomized controlled trial. J Perinatol. 2013;33(10):763–767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Patel S, Clark EA, Rodriguez CE, Metz TD, Abbaszadeh M, Yoder BA. Effect of umbilical cord milking on morbidity and survival in extremely low gestational age neonates. Am J Obstet Gynecol. 2014;211(5):519, e511–517 [DOI] [PubMed] [Google Scholar]
- 20.McCausland AM, Holmes F, Schumann WR. Management of cord and placental blood and its effect upon the newborn. Calif Med. 1949;71(3):190–196 [PMC free article] [PubMed] [Google Scholar]
- 21.Ghavam S, Batra D, Mercer J, et al. Effects of placental transfusion in extremely low birthweight infants: meta-analysis of long- and short-term outcomes. Transfusion. 2014;54(4):1192–1198 [DOI] [PubMed] [Google Scholar]
- 22.Colozzi AE. Clamping of the umbilical cord; its effect on the placental transfusion. N Engl J Med. 1954;250(15):629–632 [DOI] [PubMed] [Google Scholar]
- 23.Erickson-Owens DA, Mercer JS, Oh W. Umbilical cord milking in term infants delivered by cesarean section: a randomized controlled trial. J Perinatol. 2012;32(8):580–584 [DOI] [PubMed] [Google Scholar]
- 24.Oh W. Cord milking at delivery improves the iron status of term infants at 6 weeks. Evid Based Med. 2013;18(6):e58. [DOI] [PubMed] [Google Scholar]
- 25.Takami T, Suganami Y, Sunohara D, et al. Umbilical cord milking stabilizes cerebral oxygenation and perfusion in infants born before 29 weeks of gestation. J Pediatr. 2012;161(4):742–747 [DOI] [PubMed] [Google Scholar]
- 26.Upadhyay A, Gothwal S, Parihar R, et al. Effect of umbilical cord milking in term and near term infants: randomized control trial. Am J Obstet Gynecol. 2013;208(2):120, e121–126 [DOI] [PubMed] [Google Scholar]
- 27.Whipple GA, Sisson TR, Lund CJ. Delayed ligation of the umbilical cord; its influence on the blood volume of the newborn. Obstet Gynecol. 1957;10(6):603–610 [DOI] [PubMed] [Google Scholar]
- 28.Lanzkowsky P. Effects of early and late clamping of umbilical cord on infant’s haemoglobin level. BMJ. 1960;2(5215):1777–1782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Siddall RS, Richardson RP. Milking or stripping the umbilical cord; effect on vaginally delivered babies. Obstet Gynecol. 1953;1(2):230–233 [PubMed] [Google Scholar]
- 30.Siddall RS, Crissey RR, Knapp WL. Effect on cesarean section babies of stripping or milking of the umbilical cords. Am J Obstet Gynecol. 1952;63(5):1059–1064 [DOI] [PubMed] [Google Scholar]
- 31.Alan S, Arsan S, Okulu E, et al. Effects of umbilical cord milking on the need for packed red blood cell transfusions and early neonatal hemodynamic adaptation in preterm infants born ≤1500 g: a prospective, randomized, controlled trial. J Pediatr Hematol Oncol. 2014;36(8):e493–e498 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.