Abstract
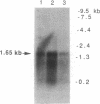
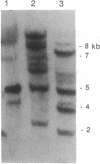
Comparison of bovine alpha 1-antichymotrypsin (ACT) protease inhibitor with that in human was achieved by cloning a nearly full-length bovine ACT cDNA of 1.5 kb, obtained by screening a bovine liver cDNA library with the human liver ACT cDNA. The deduced primary sequence indicated that the 1456-bp bovine ACT cDNA encodes a protein of 416 amino acids that contains the predicted full-length ACT with a 26-residue NH2-terminal signal sequence. Overall, the primary sequence of bovine ACT possesses a high degree of homology (55%) with human ACT; both bovine and human ACTs share common sequences in the reactive-site domains. Importantly, the reactive site of bovine ACT possesses serine as the predicted P1 position (residue at the NH2-terminal side of the cleaved peptide bond) of the reactive site, whereas human ACT contains leucine in the P1 position. Interestingly, further evidence for heterogeneity in P1 residues was provided by a second partial 0.9-kb bovine liver ACT cDNA clone (pHHK11) that contains isoleucine as P1 residue and shares only partial homology (68%) with the deduced primary sequence of the full-length bovine liver ACT cDNA clone (pHHK12). These findings suggest that isoforms of ACT in bovine liver vary in reactive-site P1 residues; the P1 position of the reactive site is often involved in protease inhibitor specificity. Consistent with the hypothesis of ACT isoforms was the demonstration of multiple copies of the bovine ACT gene by genomic blots.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abraham C. R., Selkoe D. J., Potter H. Immunochemical identification of the serine protease inhibitor alpha 1-antichymotrypsin in the brain amyloid deposits of Alzheimer's disease. Cell. 1988 Feb 26;52(4):487–501. doi: 10.1016/0092-8674(88)90462-x. [DOI] [PubMed] [Google Scholar]
- Aronsen K. F., Ekelund G., Kindmark C. O., Laurell C. B. Sequential changes of plasma proteins after surgical trauma. Scand J Clin Lab Invest Suppl. 1972;124:127–136. doi: 10.3109/00365517209102760. [DOI] [PubMed] [Google Scholar]
- Carrell R. W., Pemberton P. A., Boswell D. R. The serpins: evolution and adaptation in a family of protease inhibitors. Cold Spring Harb Symp Quant Biol. 1987;52:527–535. doi: 10.1101/sqb.1987.052.01.060. [DOI] [PubMed] [Google Scholar]
- Chandra T., Stackhouse R., Kidd V. J., Robson K. J., Woo S. L. Sequence homology between human alpha 1-antichymotrypsin, alpha 1-antitrypsin, and antithrombin III. Biochemistry. 1983 Oct 25;22(22):5055–5061. doi: 10.1021/bi00291a001. [DOI] [PubMed] [Google Scholar]
- Coombes E. J., Shakespeare P. G., Batstone G. F. Urine proteins after burn injury. Clin Chim Acta. 1979 Jul 16;95(2):201–209. doi: 10.1016/0009-8981(79)90360-7. [DOI] [PubMed] [Google Scholar]
- Gierasch L. M. Signal sequences. Biochemistry. 1989 Feb 7;28(3):923–930. doi: 10.1021/bi00429a001. [DOI] [PubMed] [Google Scholar]
- Hill R. E., Hastie N. D. Accelerated evolution in the reactive centre regions of serine protease inhibitors. Nature. 1987 Mar 5;326(6108):96–99. doi: 10.1038/326096a0. [DOI] [PubMed] [Google Scholar]
- Hill R. E., Shaw P. H., Boyd P. A., Baumann H., Hastie N. D. Plasma protease inhibitors in mouse and man: divergence within the reactive centre regions. Nature. 1984 Sep 13;311(5982):175–177. doi: 10.1038/311175a0. [DOI] [PubMed] [Google Scholar]
- Hook V. Y., Purviance R. T., Azaryan A. V., Hubbard G., Krieger T. J. Purification and characterization of alpha 1-antichymotrypsin-like protease inhibitor that regulates prohormone thiol protease involved in enkephalin precursor processing. J Biol Chem. 1993 Sep 25;268(27):20570–20577. [PubMed] [Google Scholar]
- Kelly U. L., Cooper E. H., Alexander C., Stone J. The assessment of antichymotrypsin in cancer monitoring. Biomedicine. 1978 Jul-Aug;28(4):209–215. [PubMed] [Google Scholar]
- Kornfeld R., Kornfeld S. Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem. 1985;54:631–664. doi: 10.1146/annurev.bi.54.070185.003215. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Okayama H., Kawaichi M., Brownstein M., Lee F., Yokota T., Arai K. High-efficiency cloning of full-length cDNA; construction and screening of cDNA expression libraries for mammalian cells. Methods Enzymol. 1987;154:3–28. doi: 10.1016/0076-6879(87)54067-8. [DOI] [PubMed] [Google Scholar]
- Ray C. A., Black R. A., Kronheim S. R., Greenstreet T. A., Sleath P. R., Salvesen G. S., Pickup D. J. Viral inhibition of inflammation: cowpox virus encodes an inhibitor of the interleukin-1 beta converting enzyme. Cell. 1992 May 15;69(4):597–604. doi: 10.1016/0092-8674(92)90223-y. [DOI] [PubMed] [Google Scholar]
- Rubin H., Wang Z. M., Nickbarg E. B., McLarney S., Naidoo N., Schoenberger O. L., Johnson J. L., Cooperman B. S. Cloning, expression, purification, and biological activity of recombinant native and variant human alpha 1-antichymotrypsins. J Biol Chem. 1990 Jan 15;265(2):1199–1207. [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha S., Dovey H. F., Seubert P., Ward P. J., Blacher R. W., Blaber M., Bradshaw R. A., Arici M., Mobley W. C., Lieberburg I. The protease inhibitory properties of the Alzheimer's beta-amyloid precursor protein. J Biol Chem. 1990 Jun 5;265(16):8983–8985. [PubMed] [Google Scholar]
- Travis J., Salvesen G. S. Human plasma proteinase inhibitors. Annu Rev Biochem. 1983;52:655–709. doi: 10.1146/annurev.bi.52.070183.003255. [DOI] [PubMed] [Google Scholar]
- Tsuda M., Masuyama M., Katsunuma T. Inhibition of human DNA polymerase alpha by alpha 1-antichymotrypsin. Cancer Res. 1986 Dec;46(12 Pt 1):6139–6142. [PubMed] [Google Scholar]
- Van Nostrand W. E., Wagner S. L., Farrow J. S., Cunningham D. D. Immunopurification and protease inhibitory properties of protease nexin-2/amyloid beta-protein precursor. J Biol Chem. 1990 Jun 15;265(17):9591–9594. [PubMed] [Google Scholar]
- Wei A., Rubin H., Cooperman B. S., Schechter N., Christianson D. W. Crystallization, activity assay and preliminary X-ray diffraction analysis of the uncleaved form of the serpin antichymotrypsin. J Mol Biol. 1992 Jul 5;226(1):273–276. doi: 10.1016/0022-2836(92)90140-f. [DOI] [PubMed] [Google Scholar]