Abstract
Animal models of inflammatory pain are characterized by the release of inflammatory mediators such as cytokines and neurotrophic factors, and enhanced analgesic sensitivity to opioids. In this study, we examine the mechanisms underlying this effect, in particular the roles of cholecystokinin (CCK) and nerve growth factor (NGF), in an animal model of central nervous system (CNS) inflammation induced by spinal administration of lipopolysaccharide (LPS). Although spinal administration of LY-225910 (25 ng), a CCK-B antagonist, enhanced morphine analgesia in naïve rats, it was unable to do so in LPS-treated animals. Conversely, spinal CCK-8S administration (1 ng) decreased morphine analgesia in LPS-treated rats, but not in naıve animals. Further, spinal anti-NGF (3 mg) was able to reduce morphine analgesia in LPS-treated rats, but not in naïve animals, an effect that was reversed by spinal administration of LY-225910. While CCK-8S concentration was increased in spinal cord extracts of LPS animals as compared to controls, morphine-induced spinal CCK release in the extracellular space, as measured by in-vivo spinal cord microdialysis was inhibited in LPS animals as compared to controls, and this was reversed by anti-NGF pretreatment. Finally, chronic spinal administration of b-NGF (7 mg/day) for 7 days enhanced spinal morphine analgesia, possibly by mimicking a CNS inflammatory state. We suggest that in intrathecally LPS-treated rats, spinal CCK release is altered resulting in enhanced morphine analgesia, and that this mechanism may be regulated to an important extent by NGF.
Introduction
Cholecystokinin (CCK) is implicated in a variety of physiological processes including cardiovascular and reproductive functions, feeding, sleep, learning and memory, anxiety, and nociception, particularly as an anti-opioid peptide (Crawley & Corwin, 1994; Cesselin, 1995; Fink et al., 1998). Anatomical evidence suggests a functional relationship between opioid peptides and CCK due to an overlap of these peptides in areas such as the periaqueductal gray matter, thalamus, and superficial lamina of spinal cord (Gall et al., 1987). Binding of the active sulfated form of CCK, CCK-8S, to the CCK receptors, mainly to the CCK-B receptor, reduces the binding affinity of morphine ligands (Wang et al., 1989; Wang & Han, 1990). Behavioural studies show that intrathecal (i.t.) CCK attenuates antinociception induced by both endogenous and exogenous opioids (Faris et al., 1983; Wiesenfeld-Hallin et al., 1999). On the other hand, i.t. administration of CCK-B receptor antagonists and receptor antisense oligonucleotides enhance morphine antinociception (Dourish et al., 1990; Vanderah et al., 1994). Under normal conditions, endogenous CCK does not appear to influence basal pain sensitivity, as spinal treatment with CCK-B receptor antagonists produces antinociception only in the presence of opioids (Watkins et al., 1985).
Inflammatory pain states enhance the antinociceptive potency of opioids. Two major mechanisms that account for the increased potency of morphine in inflammatory animal models are increases in μ-opioid receptors in dorsal root ganglia (Ji et al., 1995) and alterations in the expression of the anti-opioid peptide cholecystokinin (Wiesenfeld-Hallin et al., 1999; Wiesenfeld-Hallin et al., 2002). Similarly, the reduced effectiveness of opioids in neuropathic pain has been attributed to a decrease in spinal opioid receptors (Kohno et al., 2005) and to increased spinal CCK release. (Wiesenfeld-Hallin et al., 2002)
Enhanced opioid analagesia during inflammation has been well established for peripherally (Stein et al., 2001), systemically (Kayser & Guilbaud, 1983; Joris et al., 1990) and spinally (Hylden et al., 1991; Stanfa et al., 1992) administered opioids. While i.t. administration of CCK attenuates morphine antinociception in the carrageenan model of peripheral inflammation, spinally administered CCK-B antagonists have little or no effect on opioid analgesia in this model (Stanfa & Dickenson, 1993), or in a model of visceral inflammmation (Friedrich & Gebhart, 2000). In neuropathic pain models, however, co-administration of morphine with a CCK-B antagonist is able to enhance morphine analgesia so that the analgesia is comparable to that seen in naïve animals (Nichols et al., 1995).
Inflammation is known to trigger the release of a variety of mediators including neurotrophins and cytokines (Lewin & Mendell, 1993; Safieh-Garabedian et al., 1995). In particular, nerve growth factor (NGF) contributes to hyperalgesia in peripheral inflammation (Woolf et al., 1994; McMahon, 1996). Upregulation of NGF receptors is also responsible for the maintenance of long-lasting inflammatory pain (Pezet et al., 2001). In a complete Freund’s adjuvant (CFA) animal model of peripheral inflammation, increased NGF and enhanced expression of NGF receptors have been shown to contribute to upregulation of sensory neuron opioid receptors (Mousa et al., 2007).
Although many behavioural studies have examined the effects of peripheral inflammation on opioid analgesia, little is known about mechanisms underlying changes in opioid sensitivity in inflammatory pain of the CNS, a condition commonly associated with neurodegenerative conditions. A model of CNS inflammation has been developed using spinal administration of the endotoxin lipopolysaccharide (LPS). Previous work has shown that i.t. administration of LPS produces an enhanced production of cytokines such as tumour necrosis factor-α (TNF-α) in the cerebrospinal fluid (CSF) (Tanazawa et al., 1994), as well as significant thermal hyperalgesia and movement-evoked hyperalgesia (Meller et al., 1994; Kehl et al., 2004), and enhanced activity of dorsal horn neurons (Reeve et al., 2000). A two-injection LPS protocol consisting of a low dose “priming” i.t. LPS injection followed by a “challenge” higher dose LPS injection 24 hours later leads to a more robust mechanical allodynia, mechanical hyperalgesia, and heat hyperalgesia which are reversible by a p38 MAPK inhibitor, a caspase 1 inhibitor, and an IL-1 receptor antagonists (Cahill et al., 1999, 2003a; Clark et al., 2006). Further, this animal model of CNS inflammation displays enhanced morphine sensitivity (Cahill et al., 2003a), similar to that seen after peripheral inflammation.
Interestingly, chronic NGF administration leads to a normalization of CCK expression in neuropathic rats (Verge et al., 1995), which is known to be upregulated after nerve injury (Hökfelt et al., 1994). Consistent with this, our laboratory has also shown that NGF administration can protect against the decreased opioid analgesia seen with nerve injury, and that intrathecal morphine efficacy can also be augmented by a CCK-B antagonist in naïve animals chronically treated anti-NGF antibodies (Cahill et al., 2003b). We hypothesize that following CNS inflammation induced by intrathecal LPS, increased spinal NGF levels may lead to decreased CCK activity that results in increased opioid analgesia.
To test this hypothesis in the current study, we examine the effects of i.t. administration of CCK-8S and the CCK-B antagonist LY-225910 on morphine analgesia in LPS-treated rats. As upregulation of NGF is commonly found in models of inflammation (Pezet et al., 2001), and our past evidence suggests that spinal NGF can influence opioid analgesia (Cahill et al., 2003b), we also examine the effect of i.t. anti-NGF administration on morphine analgesia in the LPS model. To examine a potential interaction between NGF and CCK, we test whether a CCK antagonist is able to enhance morphine analgesia in animals treated with LPS and anti-NGF. Further, we determine whether chronic spinal administration of β-NGF, which mimics a spinal inflammatory state, enhances morphine analgesia. Finally, we measure differences in spinal CCK levels and morphine-stimukated CCK release in control and LPS animals, as well as in LPS animals after pretreatment with anti-NGF.
Materials and Methods
Animals
Experiments were performed on male Long Evans hooded rats (200–250 g; Charles River, Quebec, Canada) housed in groups of three per cage. Rats were maintained on a 12/12-h light/dark cycle, and were allowed free access to food and water. Each rat was habituated to the animal facility for at least 3 days prior to testing, was used only once, and sacrificed with an overdose of chloral hydrate upon completion of experiments. All efforts were taken to minimize animal suffering and reduce number of animals used. Experiments were carried out according to a protocol approved by the animal care committee at McGill University, and were in accordance with guidelines from the Canadian Council on Animal Care and the International Association for the Study on Pain.
Plantar Test
For all morphine analgesia trials, baseline and post-morphine withdrawal latencies from noxious heat were assessed using the plantar test as previously described by Hargreaves et al. (1998). Rats were placed in Plexiglas® boxes (22 cm X 34 cm) positioned on a heated (30° C) glass surface. Animals were allowed to habituate for 20 min before testing. Paw withdrawal latency in response to radiant heat was measured using the plantar test apparatus (Stoelting, Wood Dale, IL). The heat source was positioned under the plantar surface of one hindpaw, and both hindpaws were tested in succession with a 10 sec interval. The two readings obtained were averaged for each time point. A digital timer activated simultaneously with the heat source was stopped by a photocell activation and automatically recorded the response latency for paw withdrawal to the nearest 0.01 sec. The intensity of the light beam was chosen to give baseline latencies of 10–12 sec in naive rats. A cut-off time of 40 sec was imposed to prevent tissue damage, and each hindpaw was tested only once at each time point.
Drugs
Drugs used included: LPS (E. Coli. Lipopolysacharride; Sigma-Aldrich, Oakville, ON), anti-NGF (antibodies to NGF; Tocris, Ellisville, MO), IgG (Immunoglobulin G; Tocris), LY225-910 (CCK-B antagonist; Tocris), CCK-8S (Cholecystokinin 8-sulfated; Tocris), β-NGF (beta nerve growth factor, Sigma) and morphine sulfate (Sabex, Boucherville, QC). All drugs were given intrathecally and administrations were performed by injecting a 30 μl volume into the subarachnoid space via a lumbar puncture (between vertebrae L4 and L5) using a Hamilton syringe, while the rats were briefly anesthetized with halothane.
CNS inflammation induced by i.t. LPS
CNS inflammation was induced by injecting a 2 μg intrathecal “priming” dose of LPS 24 hours prior to testing day, and a 20 μg intrathecal “challenge” dose of LPS on the testing day. Control animals received two i.t. saline (30 μl) injections separated by 24 hrs. This method has previously been shown to result in enhanced thermal and mechanical nociceptive responses (Cahill et al., 1998, 2003a).
Intrathecal catheter surgery
Rats were implanted with a chronic indwelling intrathecal (i.t.) catheter, which was inserted between the L5 and L-6 vertebrae during a lumbar puncture according to the methods of Pogatzki et al. (2000). The lumbar puncture was performed using a 23-gauge needle, and PE-32 polyurethane tubing was pushed 3 cm through the needle to the lumbar enlargement, before the needle was removed. The tubing was then anchored to the back muscles using 3-0 silk sutures.
Morphine analgesic trials
Pilot studies as well as past studies by Cahill et al. (1998) showed that animals did not develop thermal hyperalgesia 24 hours after a single LPS injection, vehicle, or anti-NGF. Withdrawal latencies in the plantar test in LPS or control rats were measured prior to LPS/saline “challenge” as baseline reading and prior to morphine administration at 40 min post-LPS/saline “challenge”. Withdrawal latencies were again assessed 30 min post-morphine administration, or 70 min post-LPS/saline challenge. All drugs in experiments 1–4 were administered i.t. via lumbar puncture. In experiment 5, β-NGF was administered by spinal catheter attached to an osmotic pump, while morphine was administered via lumbar puncture.
Experiment 1
A morphine analgesic dose-response curve was generated on the plantar test for LPS-treated and vehicle-treated rats that were administered the CCK-B antagonist LY-225910. Thus, five doses of morphine (0.3 μg, 1.0 μg, 3.0 μg, 10 μg, and 30 μg) were administered i.t. to rats in the following four groups: non-LPS/vehicle, non-LPS/LY-225910, LPS/vehicle, and LPS/LY-225910. A 25 ng dose of LY-225910 was used, and LY-225910 or vehicle (40% dimethyl sulfoxide (DMSO) in 0.9% saline) was co-administered with morphine in a single i.t. injection. In preliminary trials, this dose was found to significantly enhance the antinociception produced by 3 μg i.t. morphine.
Experiment 2
The effect of CCK-8S on morphine analgesia in the plantar test was also evaluated in LPS-treated and non-LPS-treated rats. A 1.0 ng dose of CCK-8S was used, and CCK-8S or vehicle (0.9% saline) was co-administered with morphine. Consistent with the results of Fukazawa et al. (2007) and as observed in our pilot studies, this dose of CCK-8S was a low dose that did not affect the antinociception produced by 3 μg i.t. morphine in naïve rats.
Experiment 3
Anti-NGF (3 μg) was administered i.t. immediately prior to the “priming” LPS injection as well as the “challenge” LPS injection. Control rats received i.t. IgG vehicle prior to the LPS injections. As above, 3 μg morphine i.t. was administered 40 min post “challenge” (LPS and anti-NGF or IgG) and thermal withdrawal latencies were assessed 30 min post-morphine administration.
Experiment 4
The effect of the CCK-B antagonist, LY-225910 (25 ng, i.t.), on morphine (3 μg) analgesia in the plantar test was further examined in rats that received both anti-NGF (3 μg) and LPS treatment. LPS-treated animals that received only anti-NGF (and vehicle) were used as controls.
Experiment 5
The effect of chronic β-NGF administration (Sigma), on morphine (3 μg) analgesia in the plantar test was also examined. β-NGF was dissolved in artificial CSF (as per Gustafsson et al. (1999b) with Krebs–Ringer solution: 138 mM NaCl, 5 mM KCl, 1 mM MgCl2, 1 mM CaCl2, 11 mM NaHCO3, 1 mM NaH2PO4, containing 0.2% BSA, 0.2% glucose and 0.03% bacitracin, pH 7.4), and administered at a dose of 7 μg/day continuously for 7 days via intrathecal catheter attached to an osmotic pump (Alzet model 2001, Durect Corporation, Cupertino, CA). A group of control animals was also included which received artificial CSF alone. Baseline measurements were taken prior to i.t. catheter surgery and morphine analgesic trials were taken 7 days later, 30 min after i.t. morphine administration.
Experiment 6
Spinal cord extract preparation
For preparation of spinal cord extracts, animals were treated with LPS or vehicle as per the protocol to match morphine analgesic trials. On the second day, at 70 minutes post- injection, animals were decapitated and the spinal cord was expulsed by propulsion. The cervical region was removed and the tissue was snap frozen in isopentane and stored at −80°C. Tissue was weighed and homogenized in boiling distilled water. Centrifugation was performed at 4°C and supernatant was collected. Tissue was re-extracted in 1 mol/L acetic acid. The two supernatants were then combined, lyophilized, and stored at −80°C until assayed.
In vivo microdialysis
For in vivo microdialysis, male Long Evans rats (approximately 300 grams) were anesthetized with sodium pentobarbital [65 mg/kg, intraperitoneal (i.p.)] and a microdialysis fiber was implanted according to the method as previously described (Zahn et al., 2002; Skilling et al., 1988). After shaving the fur and sterilizing the skin on the back, the T13 thoracic vertebra was exposed, and a small hole was made on each side of the vertebra at the posterior pedicle. A single dialysis fiber (200-μm outer diameter, 45 000 mol. wt. cut-off, Hospal AN69-HF) was passed transversely through the spinal cord. The dialysis fiber was coated with epoxy except for a 2-mm active zone, which was positioned within the grey matter of the spinal cord (approximately lamina III–VI). Each end of the fiber was connected to PE-20 tubing with Super glue® and epoxy and fixed to the vertebral bone with the dental acrylic cement (Lang Dental Mfg. Co., Wheeling, IL, USA). The inlet and outlet tubing were tunnelled subcutaneously and externalized at the back of the neck. Artificial CSF was perfused through the probe and tubing throughout the procedure (except while gluing the tubing). Only rats with normal behaviour and no paralysis of hind limbs after the surgery were included in the microdialysis studies.
After surgery, the animals were allowed to recover overnight and habituate in a Raturn® Interactive System (Bioanalytic Systems, Inc. West Lafayette, IN, USA) with a tethering system which allows tubing from the microdialysis catheter to be connected to a syringe pump on one end and to a refrigerated fraction collector on the other. The “priming” i.t. injection of either 2 μg LPS or saline, as well as 3 μg anti-NGF or vehicle IgG was then administered via lumbar puncture. The next day, flow was initiated (3.75 μl/min), a 2 hour equilibration period was allowed, and a 30-minute baseline sample was collected. The “challenge” i.t. injection of either 20 μg LPS or saline, as well 3 μg anti-NGF or vehicle IgG was administered via lumbar puncture. Sample collection at 30-minute intervals began at 30 minutes post-injection. Morphine (100 μM) was administered continuously via the dialysis probe starting at 40 minutes post-injection and continuing until 120 minutes post-injection. This dose has been shown previously to evoke significant CCK release in control animals (Gustafsson et al., 1999a; Gustafsson et al., 1999b). Three 30-minute samples were collected. Samples were frozen at −20° C immediately after collection and stored at −80° C until lyophilisis and assay. Animals were sacrificed immediately upon completion of experiments.
CCK-8S radioimmunoassay
Concentrations of immunoreactive CCK in spinal cord extracts and microdialysates were measured by radioimmunoassay (RIA) in the manner described by Gustavsson et al. (1999b). In brief, standards containing synthetic sulphated CCK octapeptide (CCK-8S, Neosystem, France) were diluted in physiological Krebs medium containing 0,2% bovine serum albumine (BSA) and 0.1% triton X-100. The dialysate and standard samples were preincubated for 48 h at 4°C with the C-terminal directed CCK/gastrin antiserum 2609/10 (Rehfeld, 1978) (generously provided by Prof. Jens Rehfeld). After addition of 2500 cpm of chloramine-T labeled and HPLC-purified [125I] gastrin to each vial, all samples were incubated at 4 °C for another 24 h. The detection limit of the assay was 1 pmol/L and the intra- and interassay coefficients of variation were 6 and 8%, respectively.
Statistical Analysis
All data are expressed as means ± S.E.M. Dose response effects of morphine in the plantar test were expressed as percentage maximum possible effect (%MPE) using the following formula:
Analysis of % MPE in experiment 1 was performed using two-way analysis of variance and analysis of simple main effects (SPSS 7.0, Chicago, IL). ED50 calculations were also performed after non-linear regression using Prism 2.01 (GraphPad Software Inc., San Diego, CA), and unpaired t-tests were performed to compare the ED50’s of the treatment groups. Analysis of the effects of morphine on heat withdrawal latencies in experiment 2 through 5 were performed using repeated measures two-way analysis of variance followed by Tukey’s test for post-hoc comparisons (GB-Stat 3.0, Silver Springs, MD). Total change from baseline during morphine stimulation was calculated by subtracting the CCK concentration of the last baseline and that of each collected morphine-stimulated sample (30 mins to 120 mins post-treatment) and summing all the differences. Due to non-homogeneity of variance, we used non-parametric Kruskal-Wallis one-way ANOVA followed by Mann-Whitney U-tests for individual group comparisons. For the spinal cord CCK measurement experiment, sample CCK concentrations (pmol/L) were converted to pmol/g, based on the pre-assay tissue weight. Statistical analysis was performed using a two-tailed t-test.
Results
Experiment 1: to determine the effect of administration of a CCK-B antagonist on morphine analgesia in LPS-treated rats
Fig. 1 illustrates the %MPE for morphine analgesia in the plantar test in response to administration of different doses of morphine in rats with or without LPS treatment (LPS vs non-LPS), and with or without a CCK-B antagonist (LY-225910 vs vehicle). Analysis of variance revealed a significant effect of treatment group (F(3,112) = 8.83, P < 0.001) and of morphine dose (F(4,112) = 22.12, P < 0.001), but there was no significant interaction between treatment group and dose (P > 0.05). Analysis of simple main effects revealed that across morphine doses, the %MPE scores after non-LPS/vehicle treatment were significantly lower than %MPE scores in all other treatment groups (non-LPS/vehicle treatment versus LPS/vehicle (t(1,112) = 29.90, P < 0.001), versus non-LPS/LY-225910 (t(1,112) = 16.57, P < 0.01), and versus LPS/LY-225910 (t(1,112) = 23.22, P < 0.001)).
Fig. 1.
Dose-response curve for the analgesic effect of morphine (0.3 μg, 1 μg, 3 μg, 10 μg, 30 μg) in 4 conditions: 1) non-LPS/vehicle (open circles; n=6); 2) non-LPS/LY225910 (open squares; n=6); 3) LPS/vehicle (dark circles; n=6); and 4) LPS/LY225910 (dark squares; n=6). Morphine analgesia expressed as % maximum possible effect (%MPE) based on thermal withdrawal latencies in the plantar test. LPS treatment enhances morphine analgesia, and LY225910 enhances morphine analgesia in non-LPS animals, but not in LPS animals. (**P < 0.01)
The following ED50s were also calculated for the four groups above: 1) non-LPS/vehicle: 9.21 μg (95% confidence intervals (C.I.) 3.78 to 22.45); 2) non-LPS/LY-225910: 1.98 μg (C.I. 0.79 to 4.96) 3) LPS/vehicle: 0.83 μg (C.I. 0.47 to 1.48) 4) LPS/LY-225910: 1.214 μg (C.I. 0.54 to 2.71). As with the simple main effect comparisons, unpaired t-tests between ED50’s revealed a significantly greater ED50 for the non-LPS/vehicle treatment vs non-LPS/LY-225910 (t(8) = 2.47, P < 0.05), vs LPS/vehicle (t(8) = 4.65, P < 0.01), and vs LPS/LY-225910 (t(8) = 3.47, P < 0.01).
Experiment 2: to determine the effect of CCK-8S administration on morphine analgesia in LPS-treated rats
In the second experiment, we examined the effect of spinal CCK-8S administration on morphine analgesia in the plantar test in LPS-treated and saline-treated animals. As determined by two-way repeated ANOVA, there was no effect of treatment group (F(3, 19) = 1.22, P > 0.05), but a significant effect of time (F(1, 19) = 139.92, P < 0.001), and a significant group X time interaction (F(3, 19) = 10.4, P < 0.001). Post-hoc analysis revealed significantly higher withdrawal latencies for LPS/saline vs non-LPS/saline (P < 0.01), significantly lower withdrawal latencies for LPS/CCK vs LPS/saline (P < 0.01), and significantly lower withdrawal latencies for LPS/CCK vs non-LPS/CCK treatment (P < 0.05). Consistent with results of our pilot studies, no significant difference was found for the non-LPS/saline vs non-LPS/CCK treatment groups (P > 0.05) (Fig. 2).
Fig. 2.
Effect on pre- and post-morphine thermal latencies in the plantar test of the following treatments: 1) non-LPS/vehicle (n=6); 2) LPS/vehicle (n=6); 3) non-LPS/CCK (n=6); and 4) LPS/CCK (n=6). LPS treatment enhances morphine analgesia, and CCK reduces morphine analgesia in the LPS condition. (*P < 0.05, **P < 0.01)
Experiment 3: to determine the effect of neutralizing NGF on morphine analgesia in LPS-treated rats
In the third experiment, morphine analgesia in the plantar test was examined after antibodies to NGF (3 μg) were administered spinally to LPS-treated rats. Two-way repeated ANOVA revealed significant effects of treatment group (F(3, 20) = 3.41, P < 0.05), time (F(1, 20) = 115.8, P < 0.001), and a significant group X time interaction (F(3, 20) = 32.45, P < 0.001). Post-hoc analysis revealed significantly higher withdrawal latencies for LPS/IgG vs non-LPS/IgG (P < 0.01) and significantly lower withdrawal latencies for LPS/anti-NGF vs LPS/IgG (P < 0.01) (Fig. 3).
Fig. 3.

Effect on pre- and post-morphine thermal latencies in the plantar test of the following treatments: 1) non-LPS/IgG (n=6); 2) LPS/IgG (n=6); 3) LPS/anti-NGF (n=6); and 4) non-LPS/anti-NGF (n=6). LPS increases morphine analgesia in IgG-treated rats, and anti-NGF reduces morphine analgesia only in the LPS condtion. (**P < 0.01)
Experiment 4: to determine the effect of a CCK-B antagonist on morphine analgesia in rats treated with anti-NGF and LPS
In order to establish a link behaviourally between NGF and CCK, morphine analgesia was evaluated in LPS animals after spinal administration of both anti-NGF (3 μg) and LY-225910 (25 ng). Two-way repeated ANOVA revealed a non-significant effect of treatment (F(3, 24) = 2.36, P > 0.05), but a significant effect of time (F(1, 24) = 181.46, P < 0.001), and a significant group X time interaction (F(3, 24) = 4.83, P < 0.01). Post-hoc analysis revealed significantly lower withdrawal latencies for LPS/anti-NGF vs LPS/vehicle (P < 0.01) and significantly higher withdrawal latencies for LPS/anti-NGF/LY-225910 vs LPS/anti-NGF (P < 0.01) (Fig. 4).
Fig. 4.

Effect on pre- and post-morphine thermal latencies in the plantar test of the following treatments: 1) non-LPS/LY225910 (n=6) 2) LPS/vehicle (n=6) 3) LPS/anti-NGF (n=6) 4) LPS/anti-NGF/LY225910 (n=6). Anti-NGF again reduces morphine analgesia in LPS-treated rats, and LY-225910 enhances morphine analgesia in LPS animals only in the presence of anti-NGF. (** P < 0.01)
Experiment 5: to determine the effects of chronic spinal NGF treatment on morphine analgesia in naïve rats
To mimic persistent spinal inflammation, β-NGF was administered continuously via osmotic pump for 7 days, and morphine analgesia was subsequently evaluated on the plantar test. Two-way repeated-measures ANOVA revealed a significant effect of treatment (F(1, 24) = 10.04, P < 0.05), time (F(2, 24) = 26.82, P < 0.0001), and a significant group X time interaction (F(3, 24) = 3.6, P < 0.05). Post-hoc comparisons revealed significantly higher withdrawal latencies after morphine in the animals that received chronic β-NGF (P < 0.05) than in those receivingchronic vehicle treatment. No difference was found in pre-morphine withdrawal latencies between the animals that had received chronic β-NGF and those receiving artificial CSF (Fig. 5).
Fig. 5.
Effect on pre- and post-morphine thermal latencies in the plantar test of animals treated for 7 days with chronic β-NGF (7 μg/day; n=6) compared to control animals treated for 7 days with artificial CSF (n=6). Morphine analgesia is enhanced in animals treated with chronic β-NGF. (*P < 0.05)
Experiment 6: to determine spinal CCK levels in LPS-treated rats and morphine-induced CCK release in LPS- and LPS + anti-NGF treated rats
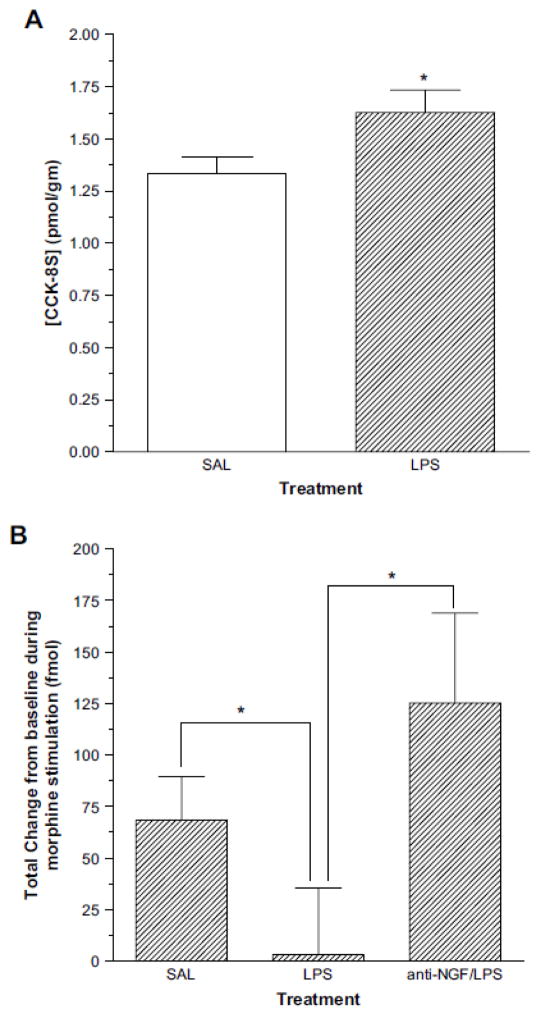
Levels of CCK in spinal cord extracts were measured by radioimmunossay in saline-treated control rats and LPS-treated rats. Two-tailed t-test analysis revealed a significant difference between the two groups (t(11)= 2.24; P<0.05). Concentrations of CCK-8S were significantly higher in LPS-treated rats as compared to saline-treated controls (Fig. 6A).
Fig. 6.
A) Concentration of CCK-8S in spinal cord extracts in saline control and LPS-treated rats (n=6–7). There is a significant increase in CCK-8S levels in LPS-treated animals as compared to control animals. (*P < 0.05) B) Morphine-induced change from baseline of CCK concentration in saline control, LPS-treated, and after anti-NGF pretreatment inI LPS-treated rats (n=6–9). Morphine-induced CCK release is inhibited in LPS-treated animals, an effect that is reversed with anti-NGF treatment. (*P < 0.05)
Morphine-induced CCK release was assessed by in-vivo microdialysis in saline-treated control rats, in LPS-treated rats, and in rats treated with LPS and anti-NGF. One-way ANOVA of estimated CCK-8S concentrations in spinal dialysates revealed no significant differences between the baseline values of the three groups. One-way ANOVA revealed a significant effect of group (χ2(2) = 7.62; P < 0.05) after morphine stimulation. Individual group comparisons (Mann Whitney U) revealed a significant difference between saline controls and LPS-treated animals (P < 0.05), and between LPS-treated animals and rats treated with LPS and anti-NGF (P < 0.05) (Fig. 6B).
Discussion
The purpose of this study was to examine the mechanisms of increased morphine analgesia in an animal model of CNS inflammation. NGF, a neurotrophic factor known to be involved in animal models of peripheral inflammation (Lewin & Mendel, 1993), appears to play a modulatory role on morphine analgesia in the spinal cord LPS-induced CNS inflammation model. Cholecystokinin, a peptide known to possess anti-opioid effects (Cesselin, 1995; McLeane, 2004; Mollereau et al., 2005) also appears to be involved in the sensitivity to morphine of our CNS inflammation model. Increased spinal NGF with CNS inflammation is thought to decrease spinal CCK release, and this is suggested to explain enhanced spinal morphine sensitivity in this animal model. The behavioural results are further supported by in-vivo microdialysis data exhibiting decreased spinal CCK release in LPS treated rats.
Results from the first two experiments suggest that spinal CCK activity may be lower in LPS-treated rats than in naïve animals. Although the CCK antagonist LY225910 was able to increase thermal latencies after morphine in naïve animals, it was unable to do so in LPS-treated rats. The present results with CNS inflammation parallel those with peripheral inflammation, since a similar effect has been shown electrophysiologically in a model of peripheral carrageenan inflammation (Stanfa & Dickenson, 1993), and behaviourally in a model of visceral inflammation (Friedrich & Gebhart, 2000). Thus, i.t. administration of a CCK-B antagonist was unable to enhance morphine analgesia in peripheral inflammatory conditions. Also, consistent with Stanfa & Dickenson (1993) who found that i.t. administration of CCK-8S attenuated the effects of morphine in animals with carrageenan inflammation (but not in normal animals), the present results show that a low dose of CCK-8S (1 ng) decreases morphine analgesia in LPS-treated rats, but not in naïve rats. Consistent with Fukazawa et al. (2007), naïve rats (non-LPS rats) do not show a decrease of morphine sensitivity with this dose of CCK-8S. This suggests that LPS-treated rats show increased responsiveness to the anti-opioid effects i.t. CCK-8S, possibly due to decreased spinal CCK release and a potential resultant supersensitivity of CCK receptors.
The results of our third experiment show that treatment with anti-NGF normalizes morphine analgesia in LPS-treated rats. This suggests that enhanced spinal levels of NGF in the LPS-treated rats contribute to the enhanced morphine analgesia. Peripheral inflammation is known to increase retrograde transport of NGF (Lewin et al., 1993; Djouhri et al., 2001), and therefore may lead to increased spinal levels of NGF. Indeed, previous studies have shown that pretreatment with anti-NGF or anti-TrkA antibodies can reduce thermal hyperalgesia and mechanical allodynia in the carrageenan CFA inflammation models (Woolf et al., 1994; McMahon et al., 1995; Ugolini et al., 2007). We presently report reduced thermal hyperalgesia in the absence of morphine in LPS-treated rats given anti-NGF. We have also observed a transient analgesic effect (lasting 20 minutes only) when anti-NGF is administered to naïve animals (data not shown). The finding that anti-NGF produces the seemingly opposite effects of reducing LPS-induced hyperalgesia and of decreasing morphine analgesia in LPS-treated rats, suggests that anti-NGF is having a specific effect on the mechanism(s) underlying morphine analgesia, rather than a purely hyperalgesic or anti-hyperalgesic effect.
In order to mimic a chronic inflammatory state and investigate the involvement of NGF in morphine analgesia, we infused NGF chronically for 7 days and recorded analgesic effects of morphine at that point. Previous studies have shown that peripheral or systemic administration of NGF elicits mechanical and thermal hyperalgesia (Lewin & Mendell, 1993). One previous study found that intrathecal chronic NGF infusion can result in thermal hyperalgesia (Malcangio et al. 2000). We did not detect such a change in thermal hyperalgesia in our study, possibly because we used a lower dose. However, we did find enhanced morphine analgesia in rats that had been treated with chronic NGF. This may be explained by evidence from recent studies suggesting that NGF can directly influence opioid receptors in-vitro, in transgenic animals, or in inflammatory pain models (McDowell, 2004; Molliver et al., 2005; Mousa et al., 2007). Alternatively, numerous studies show that chronic systemic or intrathecal NGF administration results in multiple changes in neuropeptides involved in nociception (Malcangio et al., 1997; Malcangio et al., 2000; Jongsma Wallin et al., 2001; Ramer et al., 2001; Bowles et al., 2004) and potentially in morphine sensitivity. While we expect that 2 day intrathecal LPS treatment produce increases in spinal NGF, this hypothetical increase may not be the same as that induced by 7 day intrathecal infusion of βNGF.
Past studies have shown that i.t. administration of NGF in neuropathic animals leads to a decrease in neurons that express CCK (Verge et al., 1995), and that anti-NGF administration results in altered neuropeptide expression that resembles axotomy (Christensen & Hulseboch, 1997; Shadiack et al., 2001). Further, while there have been several reports suggesting that CCK-8 may regulate NGF synthesis in inflammation, normal conditions, and possibly neuropathy (Tirassa et al., 1998; Manni et al., 2002; Manni et al., 2003), there has only been one report linking a regulation of CCK by NGF in an inflammatory animal model. In that study, using a model of intestinal inflammation induced by parasite infection, anti-NGF treatment was able to reverse increased intestinal contractile activity in response to CCK (Torrents et al., 2002).
To test the hypothesis that increased spinal NGF in LPS-treated rats may be producing decreased spinal CCK release, and therefore resulting in enhanced morphine analgesia, we administered both anti-NGF and LY225910 together. While LY225910 had been unable to enhance morphine analgesia in LPS-treated rats in the first experiment, it was able to do so in the presence of anti-NGF. We suggest that anti-NGF neutralizes the effects of LPS to the point that the CCK-B antagonist then becomes effective in enhancing morphine analgesia.
Previous studies suggest that reduced levels of CCK in the extracellular space are probably due to alterations in release mechanisms rather than synthesis (Wiesenfeld-Hallin, 2002). Studies have shown that intensity and distribution of CCKLM immunoreactivity, as well as basal release of CCKLM as measured by spinal cord in-vivo microdialysis, does not appear to be altered after peripheral inflammation (de Araujo Lucas et al., 1999; Zhang et al., 2000). However, those studies showed CCK levels to be often below detection threshold. In our study, we chose to extract CCK from whole spinal cord in LPS-treated animals. We found significantly increased CCK levels in LPS-treated animals, suggestive of low basal endogenous release.
While perfusion with high concentrations of potassium in artificial CSF or morphine evokes CCK release in normal rats, the evoked CCK release is reduced or absent in rats with peripheral inflammation. (Gustafsson et al., 1998; de Araujo Lucas et al., 1999; Wiesenfeld-Hallin, 2002). In our studies, we detected significant morphine-stimulated CCK release in control animals but not in LPS animals, suggesting LPS animals have inhibited CCK release mechanisms. Anti-NGF pretreatment immediately before LPS injection significantly restored CCK release in LPS animals to the same levels as control animals. Therefore, consistent with the behavioural results, the microdialysis studies suggest that NGF may modulate CCK release and this may account for the increased morphine sensitivity in the LPS model of central inflammation.
The hypothesized role of NGF in modulating morphine analgesia via CCK appears consistent with the study by Cahill et al. (2003) in which chronic spinal NGF was found to increase morphine analgesia in an animal model of neuropathic pain. Although not directly investigated in that study, the authors suggested that spinal levels of NGF are low in the neuropathic rats, and that this produces increases in CCK that reduce morphine analgesia. In that study, the neuropathy condition was also mimicked by chronic administration of anti-NGF in naïve rats. A low dose of CCK-B antagonist was able to enhance morphine analgesia in the anti-NGF group, but not in a control IgG group. Cahill et al.’s findings thus suggested that low levels of spinal NGF either via neuropathy or anti-NGF infusion result in enhanced spinal CCK activity, which then allowed a CCK-B antagonist to enhance morphine analgesia. Similar to the current study, it appears that altered spinal NGF levels in neuropathy can influence spinal CCK and hence morphine sensitivity. Increased morphine sensitivity in inflammatory states may be explained by a similar mechanism although in an opposite direction.
In conclusion, behavioural and neurochemical evidence has been presented for a mechanism to explain the phenomenon of increased morphine analgesia in the LPS model of central inflammation. It appears that in LPS-treated rats, increased spinal NGF may inhibit spinal CCK release, which in turn augments morphine analgesia. Elucidating mechanisms that can modulate opioid analgesia in various pain conditions may be helpful in developing new, or improving currently used, opioid therapies.
Acknowledgments
We would like to thank Professor Ernst Brodin for suggestions for the CCK extraction and radioimmunoassay. This work was supported by the Natural Sciences & Engineering Research Council of Canada (NSERC). DX was supported by a NSERC doctoral fellowship.
References
- de Araujo Lucas G, Alster P, Brodin E, Wiesenfeld-Hallin Z. Differential release of cholecystokinin by morphine in rat spinal cord. Neurosci Lett. 1998;245:13–16. doi: 10.1016/s0304-3940(98)00163-3. [DOI] [PubMed] [Google Scholar]
- Bowles WR, Sabino M, Harding-Rose C, Hargreaves KM. Nerve growth factor treatment enhances release of immunoreactive calcitonin gene-related peptide but not substance P from spinal dorsal horn slices in rats. Neurosci Lett. 2004;363:239–242. doi: 10.1016/j.neulet.2004.03.068. [DOI] [PubMed] [Google Scholar]
- Cahill CM, Dray A, Coderre TJ. Priming enhances endotoxin-induced thermal hyperalgesia and mechanical allodynia in rats. Brain Research. 1998;808:13–22. doi: 10.1016/s0006-8993(98)00786-0. [DOI] [PubMed] [Google Scholar]
- Cahill CM, Dray A, Coderre TJ. Enhanced thermal antinociceptive potency and anti-allodynic effects of morphine following spinal administration of endotoxin. Brain Research. 2003a;960:209–218. doi: 10.1016/s0006-8993(02)03885-4. [DOI] [PubMed] [Google Scholar]
- Cahill CM, Dray A, Coderre TJ. Intrathecal nerve growth factor restores opioid effectiveness in an animal model of neuropathic pain. Neuropharmacology. 2003b;45:543–552. doi: 10.1016/s0028-3908(03)00192-8. [DOI] [PubMed] [Google Scholar]
- Cesselin F. Opioid and anti-opioid peptides. Fundamental and Clinical Pharmacology. 1995;9:409–433. doi: 10.1111/j.1472-8206.1995.tb00517.x. [DOI] [PubMed] [Google Scholar]
- Christensen MD, Hulsebosch CE. Spinal cord injury and anti-NGF treatment results in changes in CGRP density and distribution in the dorsal horn in the rat. Experimental Neurology. 1997;147:475–483. doi: 10.1006/exnr.1997.6608. [DOI] [PubMed] [Google Scholar]
- Clark AK, D’Aquisto F, Gentry C, Marchand F, McMahon SB, Malcangio M. Rapid co-release of interleukin 1β and caspase 1 in spinal cord inflammation. Journal of Neurochemistry. 2006;99:868–880. doi: 10.1111/j.1471-4159.2006.04126.x. [DOI] [PubMed] [Google Scholar]
- Crawley JN, Corwin RL. Biological actions of cholecystokinin. Peptides. 1994;15:731–755. doi: 10.1016/0196-9781(94)90104-x. [DOI] [PubMed] [Google Scholar]
- Djouhri L, Dawbarn D, Robertson A, Newton R, Lawson SN. Time course and nerve growth dependence of inflammation-induced alterations in electrophysiological membrane properties in nociceptive primary afferent neurons. Journal of Neuroscience. 2001;21:8722–8733. doi: 10.1523/JNEUROSCI.21-22-08722.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dourish CT, O’Neill MF, Coughlan J, Kitchener SJ, Hawley D, Iversen SD. The selective CCK-B receptor antagonist L-365,260 enhances morphine analgesia and prevents morphine tolerance in the rat. European Journal of Pharmacology. 1990;76:35–44. doi: 10.1016/0014-2999(90)90129-t. [DOI] [PubMed] [Google Scholar]
- Faris PL, Komisaruk BR, Watkins LR, Mayer DJ. Evidence for the neuropeptide cholecystokinin as an antagonist of opiate analgesia. Science. 1983;219:310–312. doi: 10.1126/science.6294831. [DOI] [PubMed] [Google Scholar]
- Fink H, Rex A, Voits M, Voigt JP. Major biological actions of CCK—a critical evaluation of research findings, Exp. Brain Res. 1998;123:77–83. doi: 10.1007/s002210050546. [DOI] [PubMed] [Google Scholar]
- Friedrich AE, Gebhart GF. Effects of spinal cholecystokinin receptor antagonists on morphine antinociception in a model of visceral pain in the rat. Jounal of Pharmacology and Experimental Pharmaceutics. 2000;292:538–544. [PubMed] [Google Scholar]
- Fukazawa Y, Maeda T, Kiguchi N, Tohya K, Kimura M, Kishioka S. Activation of spinal cholecystokinin and neurokinin-1 receptors is associated with the attenuation of intrathecal morphine analgesia following electoacupuncture stimulation in rats. Journal of Pharmacological Sciences. 2007;104:159–166. doi: 10.1254/jphs.fp0070475. [DOI] [PubMed] [Google Scholar]
- Gall C, Lauterborn J, Burks D, Seroogy K. Co-localization of enkephalin and cholecystokinin in discrete areas of rat brain. Brain Research. 1987;403:403–408. doi: 10.1016/0006-8993(87)90085-0. [DOI] [PubMed] [Google Scholar]
- Gustafsson H, Afrah A, Brodin E, Stiller CO. Pharmacological characterization of morphine-induced in vivo release of cholecystokinin in rat dorsal horn: effects of ion channel blockers. Journal of Neurochemistry. 1999a;73:1145–1154. doi: 10.1046/j.1471-4159.1999.0731145.x. [DOI] [PubMed] [Google Scholar]
- Gustafsson H, de Araujo Lucas G, Schott E, Stiller CO, Alster P, Wiesenfeld-Hallin Z, Brodin E. Measurement of cholecystokinin release in vivo in the rat spinal dorsal horn. Brain Research Protocols. 1999b;4:192–200. doi: 10.1016/s1385-299x(99)00016-1. [DOI] [PubMed] [Google Scholar]
- Gustafsson H, Lucas GA, Schött E, Stiller CO, Alster P, Wiesenfeld-Hallin Z, Brodin E. Peripheral axotomy influences the in vivo release of cholecystokinin in the spinal cord dorsal horn - possible involvement of cholecystokinin-B receptors. Brain Research. 1998;790:141–150. doi: 10.1016/s0006-8993(98)00060-2. [DOI] [PubMed] [Google Scholar]
- Hargreaves K, Dubner R, Brown F, Flores C, Joris J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain. 1988;32:77–88. doi: 10.1016/0304-3959(88)90026-7. [DOI] [PubMed] [Google Scholar]
- Hökfelt T, Zhang X, Wiesenfeld-Hallin Z. Messenger plasticity in primary sensory neurons following axotomy and its functional implication. Trends in Neuroscience. 1994;17:22–30. doi: 10.1016/0166-2236(94)90031-0. [DOI] [PubMed] [Google Scholar]
- Hylden JL, Thomas DA, Iadarola MJ, Nahin RL, Dubner R. Spinal opioid analgesic effects are enhanced in a model of unilateral inflammation/hyperalgesia: possible involvement of noradrenergic mechanisms. European Journal of Pharmacology. 1991;194:135–143. doi: 10.1016/0014-2999(91)90097-a. [DOI] [PubMed] [Google Scholar]
- Ji RR, Zhang Q, Law PY, Low HH, Elde R, Hökfelt T. Expression of mu-, delta-, and kappa-opioid receptor-like immunoreactivities in rat dorsal root ganglia after carrageenan-induced inflammation. Journal of Neuroscience. 1995;15:8156–8166. doi: 10.1523/JNEUROSCI.15-12-08156.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jongsma Wallin H, Danielsen N, Johnston JM, Gratto KA, Karchewski LA, Verge VMK. Exgoenous NT-3 and NGF differentially modulate PACAP expression in adult sensory neurons, suggesting distinct roles in injury and inflammation. European Journal of Neuroscience. 2001;14:267–274. doi: 10.1046/j.0953-816x.2001.01641.x. [DOI] [PubMed] [Google Scholar]
- Joris J, Costello A, Dubner R, Hargreaves KM. Opiates suppress carrageenan- induced edema and hyperthermia at doses that inhibit hyperalgesia. Pain. 1990;43:95–103. doi: 10.1016/0304-3959(90)90054-H. [DOI] [PubMed] [Google Scholar]
- Kayser V, Guilbaud G. The analgesic effects of morphine, but not those of enkephalinase-inhibitor thiorphan, are enhanced in arthritic rats. Brain Research. 1983;267:131–138. doi: 10.1016/0006-8993(83)91046-6. [DOI] [PubMed] [Google Scholar]
- Kehl LJ, Kovacs KJ, Larson AA. Tolerance develops to the effect of lipopolysaccharides on movement-evoked hyperalgesia when administered chronically by a systemic route but not an intrathecal route. Pain. 2004;111:104–115. doi: 10.1016/j.pain.2004.06.014. [DOI] [PubMed] [Google Scholar]
- Kohno T, Ji R-R, Ito N, Allchorne AJ, Befort K, Karchewski LA, Woolf CJ. Peripheral axonal injury results in reduced μ opioid receptor pre- and post-synaptic action in the spinal cord. Pain. 2005;117:77–87. doi: 10.1016/j.pain.2005.05.035. [DOI] [PubMed] [Google Scholar]
- Lewin GR, Mendell LM. Nerve growth factor and nociception. Trends in Neuroscience. 1993;16:353–358. doi: 10.1016/0166-2236(93)90092-z. [DOI] [PubMed] [Google Scholar]
- Malcangio M, Garrett NE, Tomlinson DR. Nerve growth factor treatment increases stimulus-evoked release of sensory neuropeptides in the rat spinal cord. European Journal of Neuroscience. 1997;9:1101–1104. doi: 10.1111/j.1460-9568.1997.tb01462.x. [DOI] [PubMed] [Google Scholar]
- Malcangio M, Ramer MS, Boucher TJ, McMahon SB. Intrathecally injected neurotrophins and the release of substance P from the rat isolated spinal cord. European Journal of Neuroscience. 2000;12:139–144. doi: 10.1046/j.1460-9568.2000.00890.x. [DOI] [PubMed] [Google Scholar]
- Manni L, Lundeberg T, Tirassa P, Aloe L. Role of cholecystokinin-8 in nerve growth factor and nerve growth factor mRNA expression in carrageenan-induced joint inflammation in adult rats. Rheumatology. 2002;41:787–792. doi: 10.1093/rheumatology/41.7.787. [DOI] [PubMed] [Google Scholar]
- Manni L, Lundeberg T. Effects of cholecystokinin-8 in peripheral neuropathies: a nerve growth factor mediated action? Archives of Italian Biology. 2003;141:117–126. [PubMed] [Google Scholar]
- McCleane G. Cholecystokinin antagonists: a new way to improve the analgesia from old analgesics. Current Pharmaceutical Design. 2004;10:303–314. doi: 10.2174/1381612043386419. [DOI] [PubMed] [Google Scholar]
- McDowell TS. Exogenous nerve growth factor attenuates opioid-induced inhibition of voltage-activated Ba2+ currents in rat sensory neurons. Neuroscience. 2004;125:1029–1037. doi: 10.1016/j.neuroscience.2004.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon SB, Bennett DL, Priestley JV, Shelton DL. The biological effects of endogenous nerve growth factor on adult sensory neurons revealed by a trkA-IgF fusion molecule. Nature Medicine. 1995;1:774–780. doi: 10.1038/nm0895-774. [DOI] [PubMed] [Google Scholar]
- McMahon SB. NGF as a mediator of inflammatory pain. Philosophical Transactions of the Royal Society of London B: Biological Sciences. 1996;351:431–440. doi: 10.1098/rstb.1996.0039. [DOI] [PubMed] [Google Scholar]
- Meller S, Dykstra C, Grzybycki D, Murphy S, Gebhart GF. The possible role of glia in nociceptive processing and hyperalgesia in the spinal cord of the rat. Neuropharmacology. 1994;33:1471–1478. doi: 10.1016/0028-3908(94)90051-5. [DOI] [PubMed] [Google Scholar]
- Mollereau C, Roumy M, Zajac JM. Opioid-modulating peptides : mechanisms of action. Current Topics in Medicinal Chemistry. 2005;5:341–355. doi: 10.2174/1568026053544515. [DOI] [PubMed] [Google Scholar]
- Molliver DC, Lindsay J, Albers KM, Davis BM. Overexpression of NGF or GDNF alters transcriptional plasticity evoked by inflammation. Pain. 2005;113:277–284. doi: 10.1016/j.pain.2004.10.025. [DOI] [PubMed] [Google Scholar]
- Mousa SA, Cheppudira BP, Shaqura M, Fischer O, Hofmann J, Hellweg R, Schäfer M. Nerve growth factor governs the enhanced ability of opioids to suppress inflammatory pain. Brain. 2007;130:502–513. doi: 10.1093/brain/awl330. [DOI] [PubMed] [Google Scholar]
- Nichols ML, Bian D, Ossipov MH, Lai J, Porreca F. Regulation of morphine antiallodynic efficacy by cholecystokinin in a model of neuropathic pain in rats. Journal of Pharmcology and Experimental Therapeutics. 1995;5:1339–1345. [PubMed] [Google Scholar]
- Pezet S, Onteniente B, Jullien J, Junier MP, Grannec G, Rudkin BB, Calvino B. Differential regulation of NGF receptors in primary sensory neurons by adjuvant-induced arthritis in the rat. Pain. 2001;90:113–125. doi: 10.1016/s0304-3959(00)00393-6. [DOI] [PubMed] [Google Scholar]
- Pogatzki EM, Zahn PK, Brennan TJ. Lumbar catheterization of the subarachnoid space with a 32-gauge polyurethane catheter in the rat. European Journal of Pain. 2000;4:111–113. doi: 10.1053/eujp.1999.0157. [DOI] [PubMed] [Google Scholar]
- Ramer MS, Bradbury EJ, McMahon SB. Nerve growth factor induces P2X(3) expression in sensory neurons. Journal of Neurochemistry. 2001;77:864–75. doi: 10.1046/j.1471-4159.2001.00288.x. [DOI] [PubMed] [Google Scholar]
- Reeve A, Patel S, Fox A, Walker K, Urban L. Intrathecally administered endotoxin or cytokines produce allodynia, hyperalgesia and changes in spinal cord neuronal responses to nociceptive stimuli in the rat. European Journal of Pain. 2000;4:247–257. doi: 10.1053/eujp.2000.0177. [DOI] [PubMed] [Google Scholar]
- Rehfeld JF. Immunochemical studies on cholecystokinin: I. Development of sequence- specific radioimmunoassays for porcine triacontatriapeptide cholecystokinin. Journal of Biological Chemistry. 1978;253:4016–4021. [PubMed] [Google Scholar]
- Safieh-Garabedian B, Poole S, Allchorne A, Winter J, Woolf CJ. Contribution of interleukin-1 beta to the inflammation-induced increase in nerve growth factor levels and inflammatory hyperalgesia. British Journal of Pharmacology. 1995;115:1265–1275. doi: 10.1111/j.1476-5381.1995.tb15035.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shadiack AM, Sun Y, Zigmond RE. Nerve growth factor antiserum induces axotomy-like changes in neuropeptide expression in intact sympathetic and sensory neurons. Journal of Neuroscience. 2001;21:363–371. doi: 10.1523/JNEUROSCI.21-02-00363.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skilling SR, Smullin DH, Beitz AJ, Larson AA. Extracellular amino acid concentrations in the dorsal spinal cord of freely moving rats following veratridine and nociceptive stimulation. Journal of Neurochemistry. 1988;51:127–132. doi: 10.1111/j.1471-4159.1988.tb04845.x. [DOI] [PubMed] [Google Scholar]
- Stanfa LC, Sullivan AF, Dickenson AH. Alterations in neuronal excitability and potency of spinal mu, delta and kappa opioids after carrageenan-induced inflammation. Pain. 1992;50:245–254. doi: 10.1016/0304-3959(92)90040-I. [DOI] [PubMed] [Google Scholar]
- Stanfa LC, Dickenson AH. Cholecystokinin as a factor in the enhanced potency of spinal morphine following carageenan inflammation. British Journal of Pharmacology. 1993;108:967–973. doi: 10.1111/j.1476-5381.1993.tb13493.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein C, Machelska H, Binder W, Schafer M. Peripheral opioid analgesia. Current Opinion in Pharmaocology. 2001;1:62–65. doi: 10.1016/s1471-4892(01)00005-4. [DOI] [PubMed] [Google Scholar]
- Tanazawa T, Suzuki Y, Anzai M, Tsugane S, Takauasu M, Shibuya M. Vasodilation by intrathecal lipopolysaccharide of the cerebral arteries after subarachnoid haemorrhage in dogs. Acta Neurochirurgica. 1996;138:330–337. doi: 10.1007/BF01411745. [DOI] [PubMed] [Google Scholar]
- Tirassa P, Stenfors C, Lundeberg T, Aloe L. Cholecystokinin-8 regulation of NGF concentrations in adult mouse brain through a mechanism involving CCK(A) and CCK(B) receptors. British Journal of Pharmacology. 1998;123:1230–1236. doi: 10.1038/sj.bjp.0701718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torrents D, Torres R, De Mora F, Vergara P. Antinerve growth factor treatment prevents intestinal dysmotility in trichinella spiralis-infected rats. Journal of Pharmacology and Experimental Therapeutics. 2002;302:659–665. doi: 10.1124/jpet.102.035287. [DOI] [PubMed] [Google Scholar]
- Ugolini G, Marinelli S, Covaceuszach S, Cattaneo A, Pavone F. The function neutralizing anti-TrkA antibody MNAC13 reduces inflammatory and neuropathic pain. PNAS. 2007;104:2985–2990. doi: 10.1073/pnas.0611253104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanderah TW, Lai J, Yamamura HI, Porreca F. Antisense oligodeoxynucleotide to the CCKB receptor produces naltindole and [Leu5]enkephalin antiserum-sensitive enhancement of morphine antinociception. Neuroreport. 1994;5:1–5. doi: 10.1097/00001756-199412000-00049. [DOI] [PubMed] [Google Scholar]
- Verge VM, Richardson PM, Wiesenfeld-Hallin Z, Hökfelt T. Differential influence of nerve growth factor on neuropeptide expression in vivo: a novel role in peptide suppression in adult sensory neurons. Journal of Neuroscience. 1995;15:2081–2096. doi: 10.1523/JNEUROSCI.15-03-02081.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang XJ, Han JS. Modification by cholecystokinin octapeptide of the binding of mu-, delta-, and kappa opioid receptors. Journal of Neurochemistry. 1990;55:1379–1382. doi: 10.1111/j.1471-4159.1990.tb03149.x. [DOI] [PubMed] [Google Scholar]
- Wang XJ, Fan S, Ren MF, Han JS. Cholecystokinin-8 suppressed 3H-etorphine binding to rat brain opiate receptors. Life Sciences. 1989;45:117–123. doi: 10.1016/0024-3205(89)90285-3. [DOI] [PubMed] [Google Scholar]
- Watkins LR, Kinscheck IB, Mayer DJ. Potentiation of morphine analgesia by the cholecystokinin antagonist proglumide. Brain Research. 1985;327:169–180. doi: 10.1016/0006-8993(85)91511-2. [DOI] [PubMed] [Google Scholar]
- Wiesenfeld-Hallin Z, Xu XJ. Opioid-antiopioid interactions. In: Stein C, editor. Opioids in pain control – Basic and Clinical Aspects. Cambridge University Press; Cambridge: 1999. pp. 131–142. [Google Scholar]
- Wiesenfeld-Hallin Z, Xu XJ, Hökfelt T. The role of spinal cholecystokinin in chronic pain states. Pharmacology & Toxicology. 2002;91:398–403. doi: 10.1034/j.1600-0773.2002.910619.x. [DOI] [PubMed] [Google Scholar]
- Woolf CJ, Safieh-Garabedian B, Ma QP, Crilly P, Winter J. Nerve growth factor contributes to the generation of inflammatory sensory hypersensitivity. Neuroscience. 1994;62:327–331. doi: 10.1016/0306-4522(94)90366-2. [DOI] [PubMed] [Google Scholar]
- Zahn PK, Sluka KA, Brennan TJ. Excitatory amino acid release in the spinal cord caused by plantar incision in the rat. Pain. 2002;100:65–76. doi: 10.1016/s0304-3959(02)00241-5. [DOI] [PubMed] [Google Scholar]
- Zhang X, Lucas GA, Elde R, Wiesenfeld-Hallin Z, Hökfelt T. Effect of morphine on cholecystokinin and μ-opioid receptor-like immunoreactivities in rat spinal dorsal horn neurons after peripheral axotomy and inflammation. Neuroscience. 2000;95:197–207. doi: 10.1016/s0306-4522(99)00419-4. [DOI] [PubMed] [Google Scholar]