Abstract
Determination of the presence of epidermal growth factor receptor (EGFR) gene mutation is useful for predicting the efficacy of gefitinib. However, the survival rate following the initiation of treatment with gefitinib varies among individuals. A retrospective study was conducted to investigate the associations of the pretreatment serum pro-gastrin-releasing peptide (pro-GRP) and plasma neuron-specific enolase (NSE) levels to the patient survival rate following initiation of treatment with gefitinib in non-small cell lung cancer (NSCLC) patients receiving gefitinib treatment. Patients with NSCLC harboring EGFR gene mutations who received gefitinib therapy between 2004 and 2012 were included in the study. Data from a total of 41 patients were analyzed. The serum pro-GRP level was measured in 31 patients and the plasma NSE in 22 patients. The progression-free survival (PFS) (P=0.013) and overall survival (OS) (P=0.014, log-rank test) rates decreased as the plasma NSE level increased. Statistical analysis using a Cox proportional hazards regression model adjusted for age, gender, performance status (PS) and disease stage showed that higher NSE levels were associated with shorter PFS (P=0.021) and OS (P=0.0024). By contrast, no association was detected between the serum level of pro-GRP and survival rate. The results suggest that pretreatment NSE measurement could be clinically useful in patients with NSCLC scheduled to receive gefitinib treatment.
Keywords: epidermal growth factor receptor, gefitinib, prognosis, lung cancer, neuron-specific enolase
Introduction
Epidermal growth factor receptor (EGFR) gene mutation is an important predictor of the response to EGFR-tyrosine kinase inhibitor (EGFR-TKI) treatment. Gefitinib treatment yields a longer progression-free survival (PFS) rate than cytotoxic drug therapy in patients with non-small cell lung cancer (NSCLC) harboring EGFR gene mutations, including exon 19 deletion and exon 21 L858R (1–3). However, an important clinical problem is that in the majority of cases, the tumor cells soon acquire resistance to gefitinib.
Several mechanisms of acquired resistance to gefitinib have been proposed. Hepatocyte growth factor has been shown to induce resistance to gefitinib of lung adenocarcinomas harboring EGFR gene mutations (4). An investigation of 37 cases with acquired resistance to gefitinib revealed the presence of the EGFR T790 mutation, MET amplification, transition from NSCLC to small cell lung cancer (SCLC), or PIK3CA mutation in these cases (5).
Blood levels of pro-gastrin releasing peptide (pro-GRP) and neuron-specific enolase (NSE) have been reported to be frequently elevated in patients with SCLC (6), and their measurement has been utilized for diagnosis or evaluation of the treatment outcomes in these patients. Furthermore, elevation of the pretreatment plasma NSE level has also been associated with poor outcomes in patients with NSCLC (7–11).
We hypothesized that the blood level of pro-GRP and/or NSE may be associated with the clinical course following initiation of gefitinib therapy in patients with NSCLC harboring EGFR gene mutations. To endorse its validity, the present retrospective study was conducted.
Patients and methods
Patient selection
The medical records of patients with NSCLC diagnosed between 2004 and 2012 were reviewed. The inclusion criteria were as follows: i) Patients with cytologically or histologically confirmed NSCLC harboring activating EGFR gene mutations and ii) patients treated with gefitinib. Patients who had received another EGFR-TKI prior to the initiation of treatment with gefitinib were excluded. The study was approved by the Ethics Committee of the University of Toyama (Toyama, Japan).
Analysis for EGFR gene mutations, immunohistochemistry and clinical information
The presence/absence of EGFR gene mutations was analyzed by the PCR-invader assay (BML, Inc., Tokyo, Japan). Plasma NSE and serum pro-GRP were measured by a commercial laboratory (SRL Inc., Tokyo, Japan). Plasma NSE was measured by a radioimmunoassay method before December 2013 and by an electrochemiluminescence method from December 2013. Therefore, the values measured by radioimmunoassay were revised using the following equation: y = 1.060x + 0.665 (y, value measured by the electrochemiluminescence method; x, value measured by the radioimmunoassay method), developed by SRL, Inc. (http://www.srl.info/srlinfo/srlnews/2011/pdf/2011-24.pdf). The serum pro-GRP level was measured by a chemiluminescent enzyme immunoassay method.
From the medical records, the clinical information regarding the patients was reviewed, including the age, gender, performance status (PS) and disease stage. Stage was classified as postoperative recurrence and as stage I–IV according to the tumor-node-metastasis classification. Determination of disease progression was based on computed tomography, according to the Response Evaluation Criteria in Solid Tumors, version 1.1.
Immunohistochemical (IHC) staining of the tumor specimens was performed for NSE and cluster of differentiation 56 (CD56). NSE staining was performed using the anti-rabbit NSE polyclonal antibody (rabbit polyclonal, prediluted; Nichirei Biosciences, Inc., Tokyo, Japan) and CD56 staining used the anti-mouse monoclonal antibody (Clone 1B6, prediluted; Nichirei Biosciences, Inc.). Immunoperoxidase reactions were performed using the Ventana BenchMark GX automated immunostainer (Ventana Medical System, Tucson, AZ, USA), in accordance with the manufacturer's instructions. The pathological diagnoses and results of assessment of the tumor immunoreactivity were reviewed by two investigators and reported by consensus.
Statistical analysis
Survival curves were drawn using the Kaplan-Meier method to analyze the PFS and overall survival (OS) rate of the patients. The PFS was calculated as the time from the initiation of treatment with gefitinib to the date of mortality or detection of PD and censored at the date of the last visit of the patients not confirmed to have PD. The OS was calculated from the initiation of treatment with gefitinib to the date of mortality and censored at the date of the last visit of the patients who had not succumbed. The PFS and OS were compared by the log-rank test. Patients were divided based on the median tumor marker levels. Independent associations were analyzed using a Cox proportional hazards regression model adjusted for age, gender, PS, EGFR status and the disease stage. Statistical analysis was performed using the statistical package JMP 10.0.2 (SAS Institute, Cary, NC, USA). P<0.05 was considered to indicate a statistically significant difference.
Results
Patient characteristics
A total of 554 patients were diagnosed as having NSCLC between 2004 and 2012 at the First Department of Internal Medicine, University of Toyama. Of these, 74 patients were treated with gefitinib, of whom 41 with activating EGFR gene mutations were included in this study.
Table I shows the patient characteristics. Seventeen patients (41.5%) were male and 19 (46.3%) had a history of smoking. The histological diagnosis was adenocarcinoma in 38 patients (92.7%). In regard to the EGFR gene mutation, exon 19 deletion was detected in 23 (56.1%) tumors, exon 21 L858R point mutation in 15 (36.6%) cases and exon 18 point mutation in 3 (7.3%) patients. Of the patients, 32 (78.0%) were classified as having stage IIIB or IV disease and 9 (22.0%) were classified as having postoperative recurrence. Gefitinib was administered as the first-line therapy in 34 (82.9%) patients and as second-line therapy in seven (17.1%). The median (interquartile range) serum value of pro-GRP in 31 patients was 31.4 (25.8–44.8) pg/ml and the median (interquartile range) plasma value of NSE in the 22 patients was 12.1 (9.8–16.0) ng/ml. The patients were divided into three groups according to the median levels of the tumor markers, as follows: i) High level group, comprising those with high serum levels of the tumor markers, ii) low level group, comprising those with low serum levels of the tumor markers and iii) an ‘unknown’ group, comprising patients whose tumor marker levels were unknown.
Table I.
Patient characteristics.
| Characteristics | Values |
|---|---|
| Patients, n | 41 |
| Age, median years (interquartile range) | 68 (64–79.5) |
| ≥70, n (%) | 18 (43.9) |
| Gender, n (%) | |
| Male | 17 (41.5) |
| Female | 24 (58.5) |
| Histology, n (%) | |
| Adenocarcinoma | 38 (92.7) |
| Non-adenocarcinoma | 3 (7.3) |
| EGFR status, n (%) | |
| Major mutation | |
| Exon 19 del | 23 (56.1) |
| Exon 21 L858R | 15 (36.6) |
| Minor mutation | |
| Exon 18 point mutation | 3 (7.3) |
| PS, n (%) | |
| 0–1 | 28 (68.3) |
| ≥2 | 13 (31.7) |
| Smoking status, n (%) | |
| Yes | 19 (46.3) |
| No | 22 (53.7) |
| Stage, n (%) | |
| IIIB/IV | 32 (78.0) |
| Postoperative recurrence | 9 (22.0) |
| Prior regimen, n (%) | |
| 0 | 34 (83.0) |
| 1 | 7 (17.0) |
EGFR, epidermal growth factor receptor; PS, performance status.
Survival rates
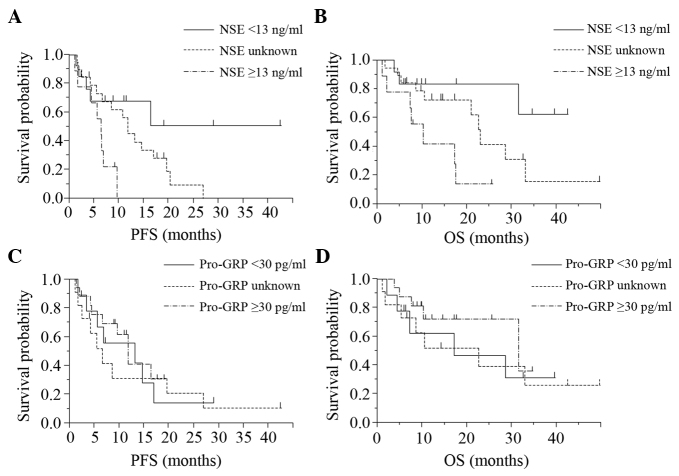
The PFS and OS in the 41 patients were 9.6 and 22.6 months, respectively (Fig. 1). Patients with higher NSE levels (high level group) showed shorter PFS (P=0.013) and OS (P=0.014) values according to the log-rank test, than those of the patients with lower NSE levels (low level group) and the group in which the NSE levels were unknown (‘unknown group), whereas no association was observed between the serum pro-GRP level and the PFS or OS (Fig. 2). Table II shows the results of analysis using the Cox proportional hazards regression model. This analysis identified pretreatment NSE as being significantly associated with the PFS (P=0.021) and OS (P=0.0024), independent of the age, gender, EGFR status, PS or disease stage.
Figure 1.
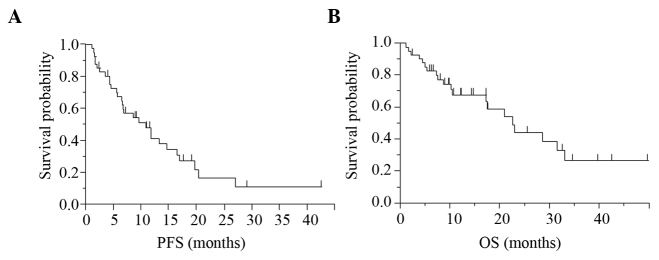
Kaplan-Meier curve for all 41 patients with NSCLC receiving gefitinib therapy. The (A) PFS and (B) OS in the 41 patients were 9.6 and 22.6 months, respectively. NSCLC, non-small cell lung cancer; PFS, progression-free survival; OS, overall survival.
Figure 2.
Kaplan-Meier curve for each patient group divided according to the tumor marker level. Patients with (A) higher NSE levels had a shorter PFS than those with (B) lower PFS levels. No association was observed in the pro-GRP levels in (A) PFS and (B) OS. PFS, progression-free survival; OS, overall survival; NSE, neuron-specific enolase; pro-GRP, pro-gastrin-releasing peptide.
Table II.
Analysis using a Cox proportional hazards regression model for assessing the association between plasma NSE and survival rate.
| PFS | OS | |||||
|---|---|---|---|---|---|---|
| Characteristics | HR | 95% CI | P-value | HR | 95% CI | P-value |
| Age, years | ||||||
| ≥70 | 0.41 | 0.15–1.10 | 0.079 | 1.25 | 0.39–3.87 | 0.700 |
| <70 | Reference | Reference | ||||
| Gender | ||||||
| Male | 1.84 | 0.71–4.83 | 0.209 | 1.37 | 0.44–4.29 | 0.583 |
| Female | Reference | Reference | ||||
| EGFR status | ||||||
| Major | 0.89 | 0.18–5.23 | 0.891 | 0.70 | 0.09–6.71 | 0.740 |
| Minor | Reference | Reference | ||||
| PS | ||||||
| ≥2 | 3.23 | 1.17–9.07 | 0.024 | 8.74 | 2.35–35.7 | 0.0013 |
| 0–1 | Reference | Reference | ||||
| Stage | ||||||
| Recurrence | 0.47 | 0.10–1.81 | 0.285 | 0.30 | 0.02–1.94 | 0.230 |
| IIIB/IV | Reference | Reference | ||||
| NSE | ||||||
| ≥13 | 4.69 | 1.27–19.12 | 0.021 | 10.31 | 2.26–59.2 | 0.0024 |
| Unknown | 2.72 | 0.87–10.14 | 0.088 | 3.08 | 0.80–15.33 | 0.103 |
| <13 | Reference | Reference | ||||
NSE, neuron specific enolase; PFS, progression-free survival; OS, overall survival; HR, hazard ratio; CI, confidence interval; EGFR, epidermal growth factor receptor; Major, major mutation including exon 19 del and exon 21 L858R; Minor, minor mutation; PS, performance status.
IHC staining
IHC staining was performed in 27 cases. All 27 showed positive staining for NSE and negative staining for CD56. Thus, there were no significant associations between the plasma NSE levels and the findings of IHC.
Discussion
In the present study, the univariate and multivariate analyses revealed the existence of an association between the pretreatment plasma NSE level and patient survival rate. However, no association was observed between the serum pro-GRP level and survival rate. To investigate the neuroendocrine properties of the tumor cells, IHC analysis was conducted.
NSE is an isozyme of the intracytoplasmic enzyme enolase, which was first identified in an extract of mouse brain tissue (12) and later shown to be elevated in neuroendocrine tumors, including SCLC. By contrast, in patients with NSCLC, the plasma NSE level has been shown to be associated with the patient survival rate. This association was observed in patients with NSCLC, regardless of whether they were treated by surgical resection, radiation therapy or chemotherapy (7–11). Furthermore, recently it has been reported that a higher pretreatment NSE level was associated with a shorter survival duration following the initiation of gefitinib in non-selective patients with NSCLC (13). In the present study the association between the plasma NSE level and the prognosis was examined in a specific patient population and confirmed the existence of the same association in patients with NSCLC harboring EGFR mutations and receiving gefitinib treatment. This association is noteworthy, in view of transition to SCLC being reported as one of the mechanisms of acquisition of resistance to gefitinib (5).
According to previous studies, plasma NSE elevation in NSCLC patients may reflect the heterogeneity of NSCLC and a neuroendocrine nature of the tumor. However, consistent with previous studies (14,15), no association was found between the plasma NSE level and the presence of neuroendocrine markers, including NSE and CD56, as determined by IHC. Although the reason for this inconsistency is unclear, it could be attributable to the difficulty in quantitative measurements by IHC. Indeed, although neuroendocrine markers have been detected in a considerable proportion of NSCLCs by IHC (16–25), positive (18–22) and negative findings (23–25) have been reported in terms of the association between the IHC characteristics and the prognosis. Another explanation is that NSE expression is not identical to NSE secretion into the blood circulation, as NSE expression has also been detected by immunohistochemistry in various normal human tissues other than nervous and neuroendocrine tissues, including type II alveolar epithelial cells (26). It is possible that in all the patients in the present study, the tumor expressed NSE, as the tumor was an adenocarcinoma in the majority of patients. However, the mechanism of NSE secretion remains unclear.
Pro-GRP is superior in sensitivity for the diagnosis of SCLC, but its association with the prognosis is weak (27,28). The present finding of a lack of any association between the serum pro-GRP level and survival rate is in line with previous studies.
There were several limitations of the present study. Pretreatment plasma NSE was not measured in certain patients and the study sample was very small in size. Although we conducted multivariate analyses to adjust for confounding factors, it may be difficult to exclude the influences of factors other than the plasma NSE level on the patient survival rate. These findings should be interpreted with caution and further study in a larger study population is necessary. Second, we cannot speculate on the mechanism of elevation of the plasma NSE, as the IHC analysis revealed no significant findings. Third, although the pretreatment plasma NSE level is likely to be a prognostic factor in patients with NSCLC based on previous studies, we cannot conclude from our observations whether it may be a predictive factor in patients with NSCLC receiving gefitinib treatment.
In conclusion, the results of the present study indicate the existence of an association between the pretreatment NSE level and survival rate in NSCLC patients receiving treatment with gefitinib. In the practical treatment of NSCLC, gefitinib therapy is one of the important treatment options for patients with NSCLC harboring EGFR gene mutations, regardless of the plasma NSE level. However, these findings suggest that measurement of the pretreatment plasma NSE level can contribute to follow-up planning in patients receiving gefitinib treatment.
References
- 1.Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–957. doi: 10.1056/NEJMoa0810699. [DOI] [PubMed] [Google Scholar]
- 2.Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I, et al. North-East Japan Study Group: Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362:2380–2388. doi: 10.1056/NEJMoa0909530. [DOI] [PubMed] [Google Scholar]
- 3.Mitsudomi T, Morita S, Yatabe Y, Negoro S, Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, et al. West Japan Oncology Group: Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): An open label, randomised phase 3 trial. Lancet Oncol. 2010;11:121–128. doi: 10.1016/S1470-2045(09)70364-X. [DOI] [PubMed] [Google Scholar]
- 4.Yano S, Wang W, Li Q, Matsumoto K, Sakurama H, Nakamura T, Ogino H, Kakiuchi S, Hanibuchi M, Nishioka Y, et al. Hepatocyte growth factor induces gefitinib resistance of lung adenocarcinoma with epidermal growth factor receptor-activating mutations. Cancer Res. 2008;68:9479–9487. doi: 10.1158/0008-5472.CAN-08-1643. [DOI] [PubMed] [Google Scholar]
- 5.Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, Bergethon K, Shaw AT, Gettinger S, Cosper AK, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011;3:75ra26. doi: 10.1126/scitranslmed.3002003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Molina R, Auge JM, Filella X, Viñolas N, Alicarte J, Domingo JM, Ballesta AM. Pro-gastrin-releasing peptide (proGRP) in patients with benign and malignant diseases: Comparison with CEA, SCC, CYFRA 21-1 and NSE in patients with lung cancer. Anticancer Res. 2005;25:1773–1778. [PubMed] [Google Scholar]
- 7.van Zandwijk N, Jassem E, Bonfrer JM, Mooi WJ, van Tinteren H. Serum neuron-specific enolase and lactate dehydrogenase as predictors of response to chemotherapy and survival in non-small cell lung cancer. Semin Oncol. 1992;19:37–43. (Suppl 2) [PubMed] [Google Scholar]
- 8.Diez M, Torres A, Ortega L, Maestro M, Hernando F, Gomez A, Picardo A, Granell J, Balibrea JL. Value of serum neuron-specific enolase in nonsmall cell lung cancer. Oncology. 1993;50:127–131. doi: 10.1159/000227163. [DOI] [PubMed] [Google Scholar]
- 9.Pujol JL, Boher JM, Grenier J, Quantin X. Cyfra 21-1, neuron specific enolase and prognosis of non-small cell lung cancer: Prospective study in 621 patients. Lung Cancer. 2001;31:221–231. doi: 10.1016/S0169-5002(00)00186-0. [DOI] [PubMed] [Google Scholar]
- 10.Ferrigno D, Buccheri G, Giordano C. Neuron-specific enolase is an effective tumour marker in non-small cell lung cancer (NSCLC) Lung Cancer. 2003;41:311–320. doi: 10.1016/S0169-5002(03)00232-0. [DOI] [PubMed] [Google Scholar]
- 11.Viñolas N, Molina R, Galán MC, Casas F, Callejas MA, Filella X, Grau JJ, Ballesta AM, Estape J. Tumor markers in response monitoring and prognosis of non-small cell lung cancer: Preliminary report. Anticancer Res. 1998;18:631–634. [PubMed] [Google Scholar]
- 12.Rider CC, Taylor CB. Evidence for a new form of enolase in rat brain. Biochem Biophys Res Commun. 1975;66:814–820. doi: 10.1016/0006-291X(75)90582-3. [DOI] [PubMed] [Google Scholar]
- 13.Fiala O, Pesek M, Finek J, Benesova L, Minarik M, Bortlicek Z, Topolcan O. The role of neuron-specific enolase (NSE) and thymidine kinase (TK) levels in prediction of efficacy of EGFR-TKIs in patients with advanced-stage NSCLC. Anticancer Res. 2014;34:5193–5198. [PubMed] [Google Scholar]
- 14.Slodkowska J, Zych J, Szturmowicz M, Demkow U, Rowinska-Zakrzewska E, Roszkowski-Sliz K. Neuroendocrine phenotype of non-small cell lung carcinoma: Immunohistological evaluation and biochemical study. Int J Biol Markers. 2005;20:217–226. doi: 10.1177/172460080502000404. [DOI] [PubMed] [Google Scholar]
- 15.Sørhaug S, Steinshamn S, Haaverstad R, Nordrum IS, Martinsen TC, Waldum HL. Expression of neuroendocrine markers in non-small cell lung cancer. APMIS. 2007;115:152–163. doi: 10.1111/j.1600-0463.2007.apm_542.x. [DOI] [PubMed] [Google Scholar]
- 16.Broers JL, Rot MK, Oostendorp T, Huysmans A, Wagenaar SS, Wiersma-van Tilburg AJ, Vooijs GP, Ramaekers FC. Immunocytochemical detection of human lung cancer heterogeneity using antibodies to epithelial, neuronal, and neuroendocrine antigens. Cancer Res. 1987;47:3225–3234. [PubMed] [Google Scholar]
- 17.Pujol JL, Simony J, Laurent JC, Richer G, Mary H, Bousquet J, Godard P, Michel FB. Phenotypic heterogeneity studied by immunohistochemistry and aneuploidy in non-small cell lung cancers. Cancer Res. 1989;49:2797–2802. [PubMed] [Google Scholar]
- 18.Kibbelaar RE, Moolenaar KE, Michalides RJ, Van Bodegom PC, Vanderschueren RG, Wagenaar SS, Dingemans KP, Bitter-Suermann D, Dalesio O, Van Zandwijk N, et al. Neural cell adhesion molecule expression, neuroendocrine differentiation and prognosis in lung carcinoma. Eur J Cancer. 1991;27:431–435. doi: 10.1016/0277-5379(91)90379-R. [DOI] [PubMed] [Google Scholar]
- 19.Graziano SL, Mazid R, Newman N, Tatum A, Oler A, Mortimer JA, Gullo JJ, DiFino SM, Scalzo AJ. The use of neuroendocrine immunoperoxidase markers to predict chemotherapy response in patients with non-small-cell lung cancer. J Clin Oncol. 1989;7:1398–1406. doi: 10.1200/JCO.1989.7.10.1398. [DOI] [PubMed] [Google Scholar]
- 20.Pujol JL, Simony J, Demoly P, Charpentier R, Laurent JC, Daurès JP, Lehmann M, Guyot V, Godard P, Michel FB. Neural cell adhesion molecule and prognosis of surgically resected lung cancer. Am Rev Respir Dis. 1993;148:1071–1075. doi: 10.1164/ajrccm/148.4_Pt_1.1071. [DOI] [PubMed] [Google Scholar]
- 21.Hiroshima K, Iyoda A, Shibuya K, Toyozaki T, Haga Y, Fujisawa T, Ohwada H. Prognostic significance of neuroendocrine differentiation in adenocarcinoma of the lung. Ann Thorac Surg. 2002;73:1732–1735. doi: 10.1016/S0003-4975(02)03504-X. [DOI] [PubMed] [Google Scholar]
- 22.Pelosi G, Pasini F, Sonzogni A, Maffini F, Maisonneuve P, Iannucci A, Terzi A, De Manzoni G, Bresaola E, Viale G. Prognostic implications of neuroendocrine differentiation and hormone production in patients with Stage I nonsmall cell lung carcinoma. Cancer. 2003;97:2487–2497. doi: 10.1002/cncr.11376. [DOI] [PubMed] [Google Scholar]
- 23.Howe MC, Chapman A, Kerr K, Dougal M, Anderson H, Hasleton PS. Neuroendocrine differentiation in non-small cell lung cancer and its relation to prognosis and therapy. Histopathology. 2005;46:195–201. doi: 10.1111/j.1365-2559.2005.02047.x. [DOI] [PubMed] [Google Scholar]
- 24.Linnoila RI, Piantadosi S, Ruckdeschel JC. Impact of neuroendocrine differentiation in non-small cell lung cancer. The LCSG experience. Chest. 1994;106:367S–371S. doi: 10.1378/chest.106.6_Supplement.367S. (Suppl 6) [DOI] [PubMed] [Google Scholar]
- 25.Hage R, Elbers HR, Brutel de la Rivière A, van den Bosch JM. Neural cell adhesion molecule expression: Prognosis in 889 patients with resected non-small cell lung cancer. Chest. 1998;114:1316–1320. doi: 10.1378/chest.114.5.1316. [DOI] [PubMed] [Google Scholar]
- 26.Haimoto H, Takahashi Y, Koshikawa T, Nagura H, Kato K. Immunohistochemical localization of gamma-enolase in normal human tissues other than nervous and neuroendocrine tissues. Lab Invest. 1985;52:257–263. [PubMed] [Google Scholar]
- 27.Shibayama T, Ueoka H, Nishii K, Kiura K, Tabata M, Miyatake K, Kitajima T, Harada M. Complementary roles of pro-gastrin-releasing peptide (ProGRP) and neuron specific enolase (NSE) in diagnosis and prognosis of small-cell lung cancer (SCLC) Lung Cancer. 2001;32:61–69. doi: 10.1016/S0169-5002(00)00205-1. [DOI] [PubMed] [Google Scholar]
- 28.Nisman B, Biran H, Ramu N, Heching N, Barak V, Peretz T. The diagnostic and prognostic value of ProGRP in lung cancer. Anticancer Res. 2009;29:4827–4832. [PubMed] [Google Scholar]