Abstract
Background
This study aimed to investigate new bone formation using recombinant human bone morphogenetic protein 2 (rhBMP-2) and locally applied bisphosphonate in rat calvarial defects.
Methods
Thirty-six rats were studied. Two circular 5 mm diameter bony defect were formed in the calvaria using a trephine bur. The bony defect were grafted with Bio-Oss® only (group 1, n = 9), Bio-Oss® wetted with rhBMP-2 (group 2, n = 9), Bio-Oss® wetted with rhBMP-2 and 1 mM alendronate (group 3, n = 9) and Bio-Oss® wetted with rhBMP-2 and 10 mM alendronate (group 4, n = 9). In each group, three animals were euthanized at 2, 4 and 8 weeks after surgery, respectively. The specimens were then analyzed by histology, histomorphometry and immunohistochemistry analysis.
Results
There were significant decrease of bone formation area (p < 0.05) between group 4 and group 2, 3. Group 3 showed increase of new bone formation compared to group 2. In immunohistochemistry, collagen type I and osteoprotegerin (OPG) didn’t show any difference. However, receptor activator of nuclear factor κB ligand (RANKL) decreased with time dependent except group 4.
Conclusion
Low concentration bisphosphonate and rhBMP-2 have synergic effect on bone regeneration and this is result from the decreased activity of RANKL of osteoblast.
Keywords: Bisphophonate, Bone morphogenetic protein 2, Bone regeneration, Histology, Alendronate
Background
In oral and maxillofacial field, restoring bony defect and enhancing bone regeneration is one of the most interesting subjects. Grafting bone materials such as autogenous, allogenic, and heterogenous bone are most widely used to restore bony defect. There have been studied to improve the efficiency of the bone regeneration. In recent times, there are reports of studies using growth factors (BMP, PDGF, TGF-β, IGF etc.) to enhance the efficiency of such bone grafting [1].
Bone morphogenetic protein (BMP) is one of the TGF-β super family, known to form new bones and cartilage. It is reported that BMP −2, −4, −5, −6, −7 etc. have been revealed to have osseoinductibility [2–4]. Out of these, rhBMP-2 which is obtained from recombinant DNA technology from mammalian cells is known to stimulate differentiation of osteoblast in cell experiments and to induce osteogenesis in animal experiments [5–7].
Bisphosphonate is widely used in the treatment of osteoporosis and osseous metastasis of cancer, and it is known to play a role of reducing bone loss by reducing or stopping the function of osteoclast [8, 9]. As increased use of bisphosphonate, there have been reported bisphosphonate related osteonecrosis of jaw (BRONJ) due to suppressed osteoclastogenesis [10, 11]. On the other hand, there are studies regarding low concentration of locally applied bisphosphonate accelerates the healing of the tooth extraction socket, bone fracture or increase the bone density of the interface after titanium implant placement [12–14]. We hypothesized that rhBMP-2 and low concentration bisphosphonate have synergic effect for bone regeneration. The aim of this study was to investigate the combined effect of rhBMP-2 and locally applied bisphosphonate of bone regeneration in a rat model.
Methods
Preparation of animal
Thirty-six Sprague-Dawley male rats (15 weeks old, Koatech, INC. Korea) weighing between 250 g and 300 g comprised the animal experimental model used. The animals were housed individually in standard rat cages maintained under an ambient temperature of 24 °C to 26 °C and a 12/12 h light/dark cycle. The animals had free access to drinking water and standard laboratory pellets. This study was conducted at the Pusan National University Institutional Animal Care and Use Committee (PNU-2011-000254 ).
Experimental materials and surgical procedures
Each bony defect was stuffed by Bio-Oss®, inorganic bovine bone, as xenograft material. rhBMP-2 was produced in E. coli using genetic engineering (Cowellmedi Co, Busan, Korea). As for rhBMP-2, 100 μg/1 mL concentrations were used. Alendronate (Sigma, St. Louis, MO, USA) was used as bisphosphonate. 1 mM (low concentration) and 10 mM (high concentration) alendronate were conducted this study.
The animals were anesthetized with a mixture of 10 mg/kg of xylazine hydrochloride (Rumpun® Bayer, Korea) and 100 mg/kg of ketamine chloride (Ketalar®, Yuhan Corporation, Korea). The dorsal area of the rat cranium was shaved before surgery, and the surgical field was prepared with an iodine solution. A midline skin incision was performed on the skull, and the periosteum with the temporalis muscle was reflected laterally. Two symmetrical round 5 mm diameter bony defects were then formed in the calvaria using 5 mm diameter trephine (Hee Sung Corp., Seoul, Korea). The bony defect were grafted with Bio-Oss® only (group 1, n = 9), Bio-Oss® wetted with rhBMP-2 (group 2, n = 9), Bio-Oss® wetted with rhBMP-2 and 1 mM alendronate (group 3, n = 9) and Bio-Oss® wetted with rhBMP-2 and 10 mM alendronate (group 4, n = 9). Then, the muscle layer was closed with 4–0 Vicryl® sutures in a continuous fashion, and the skin with 3-0 VicrylⓇ sutures. Gentamycin at 5 mg/kg was injected for prevention of infection after surgery. The animals from each group were sacrificed at 2, 4 and 8 weeks after surgery. The skin was dissected, the calvaria harvested and immediately immersed in a 10 % tempered solution of formaldehyde.
Histology
Each specimen was fixed in 10 % formaldehyde solution, decalcified in formic acid for 48 h, and embedded in paraffin. Serial cross-sections (5 μm) were cut through the larger diameter of the defect and stained with hematoxylin-eosin (H-E). The H-E stains reveal the cellular reactions indicating bone formation. The slides were photographed with the use of a virtual slide system (Scanscope CS system, Aperio Technologies, Vista,CA).
Histomorphometric analysis
The Aperio Technologies Scanscope CS system is useful for calculating new bone formation areas on H-E stained slides. The calculation, involving just the drawing of the newly formed bone outlines, is easily done. Slides in each group were scanned by virtual slide system microscopy (X100), and then 2 slides from each group were selected in the 2, 4, 8 weeks. To calculate the new bone formation area, 4 sites were randomly selected for each slide, the photographs of which were 0.600 mm × 0.500 mm. In this study, we applied 2 statistical methods to the significance testing of each group. The dependent variables of the control and experimental groups were averages and standard deviations. The difference of the dependent variables in each group for the 2, 4, 8 weeks was analyzed by Kruskal-Wallis test and Tukey’s post hoc test. The collected data were analyzed with the use of SPSS 18.0 software (SPSS Inc., Chicago, IL, USA).
Immunohistochemical analysis
The 5 μm thick slice maintained at 60 °C in an oven for an hour. After that, the sample was washed with distilled water after being hydrated using several steps of alcohol after the paraffin was removed. This treatment was repeated four times for five minutes with xylene. And then the sample was washed three times with the buffer solution for three minutes. In order to remove the intrinsic peroxide within the tissue, the sample was reacted with 0.3 % H2O2 solution. The sample was washed four times with buffer solution in order to inhibit the unusual combination within the tissue, after reacting with blocking serum (goat ImmunoCruz staining system, Santa Cruz Biotechnology, Inc., USA) for an hour, and reacting for a night with a dilution of each primary antibody, OPG, RANKL, Collagen type I (Santa Cruz biotechnology, OPG 1:120, RANKL 1:40, collagen type I 1:100). After being washed with the buffer solution, the sample was reacted with a secondary antibody labeled Biotin for an hour, washed four times again with the buffer solution, and reacted with the enzyme conjugate streptavidin (goat immunocruz staining system, Santa Cruz Biotechnology) for an hour at room temperature.
The tissue section was again washed in PBS three times for 10 min and was colored for 3 min in a solution mixed with diaminobenzidine chromogen and hydrogen peroxidase. The section was then washed in Tris buffer, PBS and distilled water for 10 min each. After that, it was counterstained with Harris hematoxylin and then mounted.
After each staining we evaluated the slides by immunohistochemical reactivity for RANKL using a scoring system of −, +, ++, and +++, which corresponded to absent staining, weak staining (<25 % of cells), moderate staining (<50 % of cells), and strong staining (>50 %), respectively. In the evaulation of OPG, collagen Type I expression, the boundary of cell counting was using 50 % and 75 % instead of 25 % and 50 %.
Results
Histologic finding
H-E stain
After grafting, at 2 week collagen fiber arrangement was finding in all group. Osteoblast proliferation was observed that occurred in early stage of new bone formation . At 4 week, relatively small bone formation was observed at group 2, 3, 4. At 8 week, all group showed the aspect of complete mineralization (Fig. 1).
Fig. 1.
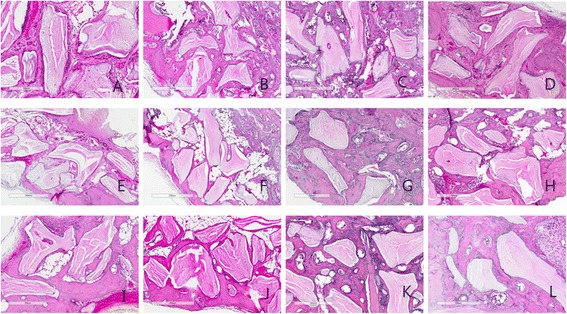
Histological view at 2, 4 and 8 weeks. a 2 week group 1; b 2week group 2; c 2week group 3; d 2 week group 4; e 4 week group 1; f 4 week group 2; g 4 week group 3; h 4 week group 4; i 8 week group 1; j 8 week group 2; k 8 week group 3; l 8 week group 4. (H-E stain, Magnification X 100)
Degrees of inflammatory reaction
Relative inflammatory reaction intensity was evaluated for each groups. We examined the specimens at 2 week in each group because most of healing process and inflammatory phase are almost finished within 2 week. In group 1, 2, 3, any remarkable inflammatory reaction was not observed, but mild inflammation was observed in group 4 (Fig. 2, Table 1).
Fig. 2.
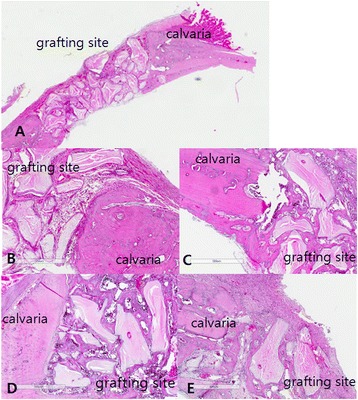
Inflammatory reaction of grafted site at 2 week. a Gross specimen; b Group 1; c Group 2; d group 3; e Group 4. In group 4, inflammatory cells and capillaries were observed. (H-E stain, Magnification X 100)
Table 1.
Inflammatory reaction in each group
| Time | Group 1 | Group 2 | Group 3 | Group 4 |
|---|---|---|---|---|
| 2 week | ± | ± | ± | + |
(±; minimal, +; mild)
Histomorphometric analysis
After measuring the area where bones were formed by randomly designating 4 locations from one specimen, an average was calculated and shown by ratio. The width of the designated rectangle was set to 0.3 mm [2] (Fig. 3).
Fig. 3.

Measurement of bone formation area by Aperio imaging scope. New bone formation can be calculated just drawing the outlines of newly formed bone. H-E stain, Magnification X 100
In 2 weeks, mean extent of bone formation was 10.46 % in group 1, 17.63 % in group 2, 19.73 % in group 3 and 12.41 % in group 4. In 4 weeks, that was 18.79 % in group 1, 23.21 % in group 2, 28.49 % in group 3 and 21.27 % in group 4. In 8 weeks, the extent was 31.93 % in group 1, 34.55 % in group 2, 38.98 % in group 3 and 30.68 % in group 4.
The bone formation area was calculated from bone formation ratio and expressed as mean ± standard deviation. In 2, 4 week, there exist significant difference among each groups. In 8 week there were not any difference among groups. In 2 week, group 2 and 3 showed increased bone formation significantly compared to group 1. And group 3 increased larger than group 4 significantly. In 4 week, group 3 showed increased bone formation area compred to group 1 and 4 (Tables 2 and 3).
Table 2.
Measurement of bone formation area
| Time | Group 1 | Group 2 | Group 3 | Group 4 | P value |
|---|---|---|---|---|---|
| 2 week | 0.031 ± 0.006 | 0.052 ± 0.004 | 0.059 ± 0.008 | 0.037 ± 0.006 | 0.002* |
| 4 week | 0.056 ± 0.004 | 0.069 ± 0.01 | 0.085 ± 0.006 | 0.063 ± 0.008 | 0.004* |
| 8 week | 0.096 ± 0.010 | 0.104 ± 0.013 | 0.117 ± 0.01 | 0.092 ± 0.009 | 0.072 |
(Unit ; mm2, *p < 0.05)
Table 3.
Post hoc test results (Tukey HSD)
| P value | |||
|---|---|---|---|
| Groups | 2 week | 4 week | |
| Group 1 | Group 2 | 0.014* | 0.182 |
| Group 3 | 0.003* | 0.003* | |
| Group 4 | 0.681 | 0.661 | |
| Group 2 | Group 3 | 0.636 | 0.067 |
| Group 4 | 0.065 | 0.765 | |
| Group 3 | Group 4 | 0.012* | 0.020* |
(*P < 0.05)
Immunohistochemistric finding
Collagen type I
With regard to collagen, high expression was examined in group 1,2,3. There was no difference among three groups, but group 4 shows less expression of collagen type I relatively than other group (Fig. 4, Table 4).
Fig. 4.
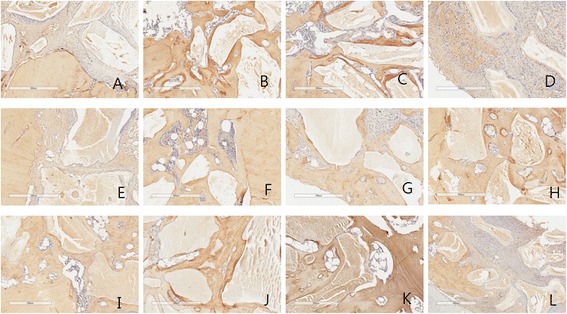
Collagen type I antibody reaction of grafted sites. a 2 week group 1; b 2week group 2; c 2week group 3; d 2 week group 4; e 4 week group 1; f 4 week group 2; g 4 week group 3; h 4 week group 4; i 8 week group 1; j 8 week group 2; k 8 week group 3; l 8 week group 4. Magnification X 100
Table 4.
Immunohistochemistry analysis of collagen type I, OPG and RANKL
| Antibody | Time/week | Groups | |||
|---|---|---|---|---|---|
| Group 1 | Group 2 | Group 3 | Group 4 | ||
| Collagen type I | 2 | ++ | +++ | +++ | + |
| 4 | ++ | ++ | ++ | ++ | |
| 8 | +++ | +++ | +++ | + | |
| 2 | ++ | ++ | ++ | + | |
| OPG | 4 | +/++ | ++ | +/++ | + |
| 8 | ++ | ++ | +++ | + | |
| 2 | ++ | ++ | + | ++ | |
| RANKL | 4 | ++ | +++ | −/+ | ++ |
| 8 | + | + | + | ++ | |
(−; no immunoreactivity, +; weak immunoactivity, ++; moderate immunoactivity, +++; strong immunoactivity, Abbreviations: OPG osteoprotegerin, RANKL receptor activator nuclear factor κB ligand)
OPG
In the analysis of OPG expression, it shows moderate expression at 2, 4 weeks, and decrease at 8 weeks. Like collagen type I, no differeces were examed among group 1,2,3. In group 4, OPG did not manifest well (Fig. 5, Table 4).
Fig. 5.
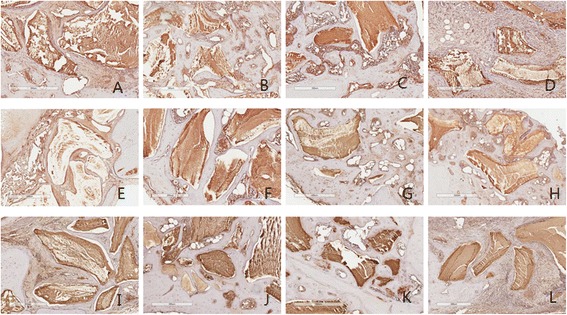
OPG antibody reaction of grafted sites. a 2 week group 1; b 2week group 2; c 2week group 3; d 2 week group 4; e 4 week group 1; f 4 week group 2; g 4 week group 3; h 4 week group 4; i 8 week group 1; j 8 week group 2; k 8 week group 3; l 8 week group 4. Magnification X 100
RANKL
In the case of RANKL, expression of RANKL was decreased with time dependant except group 4. Manifestation seems to be slightly reduced in the 2 and 4 week stages in group 3. It continues to be manifested moderate in group 4 (Fig. 6, Table 4).
Fig. 6.
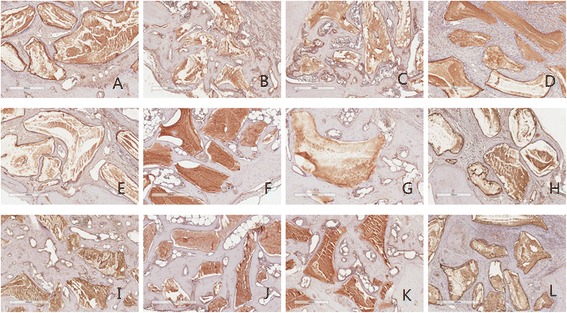
RANKL antibody reaction of grafted sites. a 2 week group 1; b 2week group 2; c 2week group 3; d 2 week group 4; e 4 week group 1; f 4 week group 2; g 4 week group 3; h 4 week group 4; i 8 week group 1; j 8 week group 2; k 8 week group 3; l 8 week group 4. Magnification X 100
Discussion
In this study we aimed to investigate if rhBMP-2 and locally applied bisphosphonate have synergic effect on bone regeneration. Since Urist reported the osteoconductive potential of BMP 2, numerous studies were conducted on rhBMP-2 [2]. Active studies are being carried out after it was approved for use in dental field by the FDA (INFUSE® Bone Graft, Medtronic Spinal and Biologics, Memphis, Tenn) in 2007. BMP-2 have an effect on precursor cells like mesenchymal stem cells which differentiating into osteoblast. It is widely known that BMP, when local applied, accelerates bone regeneration [15].
Phamacologic mechanism of alendronate is inhibition of farnesyl diphosphate synthase in the mevalonate pathway essential for the prenylation of proteins in osteoclasts. This causes mechanical inhibition of osteoclast adhesion on the bone margin and osteoclast apoptosis [16]. The effect of BPs on osteoclasts could also be produced by osteoblasts. Osteoblast/stromal cells regulate osteoclastogenesis by M-CSF, RANKL and OPG [17, 18]. Through cell-to-cell contact of osteoblast/stromal cells with osteoclasts, RANKL and M-CSF induce osteoclast progenitor cells to differentiate into osteoclasts. There are reports that low concentration bisphosphonate stimulate the proliferation and differentiation of osteoblast [19–22]. Also systemic or local short period application of bisphosphonate improves bone regeneration due to pharmacological effects in vivo [23–26].
In the histomorphometry analysis, in the 2 week, significant difference was observed in group 2 and 3 compared to the group 1 and 4. In spite of group 2 was much bone formation, there was not significant difference between group 2 and 3. In the results of the 4 week, group 3 was only significant difference compare to other group and highest bone formation. It seems ordinary BMP was involved in the early stage of osteogenesis and BMP was main role in bone formation on 2 week. In 4 and 8 weeks there was more bone formation in group 3. It seems alendronate was main role in suppression of osteoclastic activity at initiate of bone remodeling with BMP.
RANKL is known as osteoclast differentiation factor and it is known to accelerate and stimulate their differentiation [27]. The manifestation of RANKL appears on the surface of marrow stromal cells, immature osteoblasts or mesenchymal cells [28]. OPG is an important regulator of osteoclastogenesis via it’s binding to RANKL of the effects of RANKL. OPG is role of inhibiting bone resorption. When its concentration is reduced, the resorption processes may prevail and bone loss occurs [29, 30]. In the immunohistochemistry findings, in the case of collagen, most came out uniformly without a significant difference in most of the specimen, but because osteogenesis occurred relatively not well in group 4, it seems collagen manifestation also did not appear well. Likewise, even in the case of OPG, it seems to be not manifested well because of a relatively weak of osteogenesis in the high concentration group. In the case of RANKL, it seems there is a slight reduction in manifestation from the group 3 in the early phase, and this seems to show a suppression of the osteoclastic activity of alendronate. In group 4, RANKL continues to be moderate manifested, this is seems to be relatively less bone formatinon and suppression of osteoclastic activity a weak of osteogenesis, though its mechanism is vague.
Unlike previous experiments, this experiment was designed in combination with elements that can be easily applied to preclinical practice. First, as scaffolds or carrier, it has used deproteinized bovine bone, and was not applied by adsorbing alendronate for a long period of time, and direct used 1 mM, 10 mM together with rhBMP-2. And this experiment has attempted to perform application by using BMP and bisphosphonate together. The concentration of BMP was determined from the manufacturer, and the concentration of bisphosphonate is known to use a concentration of 1 mg/ml in many theses, but it was difficult to find grounds for these. Therefore, we used the concentration of 1 mM/ml (0.325 mg/ml) that was suggested purpose of prevent root resorption in the delayed replantation of an extracted tooth as a standard [31–33]. In group 3, there were statistically significant differences in 2 and 4 week compared to group 1 and 4. It seems there is a great bone forming effect since it showed a significant difference in the 4th week compared to group 2. While it is difficult to provide an accurate interpretation of its mechanism since these are not results from substituting one condition but from using compositive methods, the group using a combination of a rhBMP-2 and 1 mM of alendronate seems to have faster and better bone formation than the control group.
Conclusion
This study was conducted to investigate whether alendronate and rhBMP-2 have combined effect on bone regeneration. Our results indicate that low concentration bisphosphonate and rhBMP-2 have synergic effect on bone regeneration and this is result from the decreased activity of RANKL of osteoblast. Though further studies are needed to find out an appropriate concentration, it seems capable of being clinically applied in bony defect.
Abbreviations
- BMP
Bone morphogenetic protein
- rhBMP-2
Recombinant human bone morphogenetic protein 2
- BP
Bisphophonate
- BRONJ
Bisphosphonate related osteonecrosis of jaw
- H-E
Hematoxylin-eosin
- OPG
Osteoprotegerin
- RANKL
Receptor activator of nuclear factor κB ligand
- M-CSF
Macrophage colony-stimulating factor
Footnotes
Ho-Chul Kim and Jae-Min Song contributed equally to this work.
Competing interests
The authors declare that they have no competing interests.
Funding
This work was supported by 2 year research grant of Pusan National University.
Authors’ contributions
HC involved in writing manuscript. JM, CJ and SY contributed in performing animal surgery, analysis of results. IR and BS involved in design animal model, fabricating the slides. SH contributed in study design, writing manuscript. All authors read and approved the final manuscript.
Authors’ information
HC Kim and JM Song are equally contributed as first author.
Contributor Information
Ho-Chul Kim, Email: hl5bwn@hanmail.net.
Jae-Min Song, Email: omssjm@gmail.com.
Chang-Joo Kim, Email: gabriel001@hanmail.net.
Sang-Yong Yoon, Email: 95517866@hanmail.net.
In-Ryoung Kim, Email: biowool77@gmail.com.
Bong-Soo Park, Email: parkbs@pusan.ac.kr.
Sang-Hun Shin, Email: ssh8080@pusan.ac.kr.
References
- 1.Lieberman JR, Daluiski A, Einhorn TA. The role of growth factors in the repair of bone biology and clinical applications. J Bone and Joint Surgery. 2002;84:1032–1044. doi: 10.2106/00004623-200206000-00022. [DOI] [PubMed] [Google Scholar]
- 2.Urist MR, Strates BS. Bone morphogenetic protein. J Dent Res. 1971;50:1392–1406. doi: 10.1177/00220345710500060601. [DOI] [PubMed] [Google Scholar]
- 3.Gitelman SE, Kobrin MS, Ye JQ, Lopez AR, Lee A, Derynck R. Recombinant Vgr-1/BMP-6-expressing tumors induce fibrosis and endochondral bone formation in vivo. J Cell Biol. 1994;126:1595–1609. doi: 10.1083/jcb.126.6.1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wozney JM, Rosen V. Bone morphogenetic protein and bone morphogenetic protein gene family in bone formation and repair. Clin Orthop. 1998;346:26–37. [PubMed] [Google Scholar]
- 5.Takaoka K, Yoshikawa H, Hashimoto J, Ono K, Matsui M, Nakazato H. Transfilter bone induction by Chinese hamster ovary (CHO) cells transfected by DNA encoding bone morphogenetic protein-4. Clin Orthop. 1994;300:269–273. [PubMed] [Google Scholar]
- 6.Cho YJ, Park JW, Bae YC. The effects of synthetic peptide derived from hBMP-2 on bone formation in rabbit calvarial defect. Tissue Eng Regen Me. 2008;5:488–497. [Google Scholar]
- 7.Cook SD, Wolfe MW, Salkeld SL, Rueger DC. Effect of recombinant human osteogenic protein-1 on healing of segmental defects in non-human primates. J Bone and Joint Surgery. 1995;77:734–750. doi: 10.2106/00004623-199505000-00010. [DOI] [PubMed] [Google Scholar]
- 8.Green JR. Bisphosphonates: preclinical review. Oncologist. 2004;9:3–13. doi: 10.1634/theoncologist.9-90004-3. [DOI] [PubMed] [Google Scholar]
- 9.Jakobsen T, Baas J, Kold S, Bechtold JE, Elmengaard B, Soballe K. Local bisphosphonate treatment increases fixation of hydroxyapatite-coated implants inserted with bone compaction. J Orthop Res. 2009;27:189–194. doi: 10.1002/jor.20745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Marx RE. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg. 2003;61:1115–1117. doi: 10.1016/S0278-2391(03)00720-1. [DOI] [PubMed] [Google Scholar]
- 11.Allen MR, Burr DB. The pathogenesis of bisphosphonate-related osteonecrosis of the jaw: so many hypotheses, so few data. J Oral Maxillofac Surg. 2009;67:61–70. doi: 10.1016/j.joms.2009.01.007. [DOI] [PubMed] [Google Scholar]
- 12.Jakobsen T, Kold S, Bechtold JE, Elmengaard B, Soballe K. Effect of topical alendronate treatment on fixation of implants inserted with bone compaction. Clin Orthop Relat Res. 2006;444:229–234. doi: 10.1097/01.blo.0000191273.34786.40. [DOI] [PubMed] [Google Scholar]
- 13.Jakobsen T, Kold S, Bechtold J, Elmengaard B, Søballe K. Local alendronate increases fixation of implants inserted with bone compaction: 12‐week canine study. J Orthop Res. 2007;25:432–441. doi: 10.1002/jor.20276. [DOI] [PubMed] [Google Scholar]
- 14.McLeod K, Anderson GI, Dutta NK, Smart RSC, Voelcker NH, Sekel R, et al. Adsorption of bisphosphonate onto hydroxyapatite using a novel co‐precipitation technique for bone growth enhancement. J Biomed Mater Res A. 2006;79:271–281. doi: 10.1002/jbm.a.30792. [DOI] [PubMed] [Google Scholar]
- 15.McClellan JW, Mulconrey DS, Forbes RJ, Fullmer N. Vertebral bone resorption after transforaminal lumbar interbody fusion with bone morphogenetic protein (rhBMP-2) J Spinal Disord Tech. 2006;19:483–486. doi: 10.1097/01.bsd.0000211231.83716.4b. [DOI] [PubMed] [Google Scholar]
- 16.Kim HK, Kim JH, Abbas AA, Yoon TR. Alendronate enhances osteogenic differentiation of bone marrow stromal cells: a preliminary study. Clin Orthop Relat Res. 2009;467:3121–3128. doi: 10.1007/s11999-008-0409-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lacey D, Timms E, Tan H, Kelley M, Dunstan C, Burgess T, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93:165–176. doi: 10.1016/S0092-8674(00)81569-X. [DOI] [PubMed] [Google Scholar]
- 18.Tay JY, Bay BH, Yeo JF, Harris M, Meghji S, Dheen ST. Identification of RANKL in osteolytic lesions of the facial skeleton. J Dent Res. 2004;83:349–353. doi: 10.1177/154405910408300415. [DOI] [PubMed] [Google Scholar]
- 19.Sahni M, Guenther HL, Fleisch H, Collin P, Martin TJ. Bisphosphonates act on rat bone resorption through the mediation of osteoblasts. J Clin Invest. 1993;91:2004–2011. doi: 10.1172/JCI116422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nishikawa M, Akatsu T, Katayama Y, Yasutomo Y, Kado S, Kugal N, et al. Bisphosphonates act on osteoblastic cells and inhibit osteoclast formation in mouse marrow cultures. Bone. 1996;18:9–14. doi: 10.1016/8756-3282(95)00426-2. [DOI] [PubMed] [Google Scholar]
- 21.Vitte C, Fleisch H, Guenther HL. Bisphosphonates induce osteoblasts to secrete an inhibitor of osteoclast-mediated resorption. Endocrinology. 1996;137:2324–2333. doi: 10.1210/endo.137.6.8641182. [DOI] [PubMed] [Google Scholar]
- 22.Naidu A, Dechow PC, Spears R, Wright JM, Kessler HP, Opperman LA. The effects of bisphosphonates on osteoblasts in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106:5–13. doi: 10.1016/j.tripleo.2008.03.036. [DOI] [PubMed] [Google Scholar]
- 23.Srisubut S, Teerakapong A, Vattraphodes T, Taweechaisupapong S. Effect of local delivery of alendronate on bone formation in bioactive glass grafting in rats. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104:e11–e16. doi: 10.1016/j.tripleo.2007.04.022. [DOI] [PubMed] [Google Scholar]
- 24.Greiner SH, Wildemann B, Back DA, Alidoust M, Schwabe P, Haas NP, et al. Local application of zoledronic acid incorporated in a poly (D, L-lactide)-coated implant accelerates fracture healing in rats. Acta Orthop. 2008;9:717–725. doi: 10.1080/17453670810016768. [DOI] [PubMed] [Google Scholar]
- 25.Roshan-Ghias A, Arnoldi J, Procter P, Pioletti DP. In vivo assessment of local effects after application of bone screws delivering bisphosphonates into a compromised cancellous bone site. Clin Biomech (Bristol, Avon) 2011;26:1039–1043. doi: 10.1016/j.clinbiomech.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 26.Toker H, Ozdemir H, Ozer H, Eren K. Alendronate enhances osseous healing in a rat calvarial defect model. Arch Oral Biol. 2012;57:1545–1550. doi: 10.1016/j.archoralbio.2012.06.013. [DOI] [PubMed] [Google Scholar]
- 27.Ficarra G, Beninati F. Bisphosphonate - related osteonecrosis of the jaws: the point of view of the oral pathologist. Clin Cases Miner Bone Metab. 2007;4:53–57. [PMC free article] [PubMed] [Google Scholar]
- 28.Hofbauer LC, Heufelder AE. Role of receptor activator of nuclear factor-κB ligand and osteoprotegerin in bone cell biology. J Mol Med. 2001;79:243–253. doi: 10.1007/s001090100226. [DOI] [PubMed] [Google Scholar]
- 29.Hofbauer LC, Heufelder AE. Osteoprotegerin and its cognate ligand: a new paradigm of osteoclastogenesis. Eur J Endocrinol. 1998;139:152–154. doi: 10.1530/eje.0.1390152. [DOI] [PubMed] [Google Scholar]
- 30.Udagawa N, Takahashi N, Yasuda H, Mizuno A, Itoh K, Ueno Y, et al. Osteoprotegerin produced by osteoblasts is an important regulator in osteoclast development and function 1. Endocrinology. 2000;141:3478–3484. doi: 10.1210/endo.141.9.7634. [DOI] [PubMed] [Google Scholar]
- 31.Levin L, Bryson E, Caplan D, Trope M. Effect of topical alendronate on root resorption of dried replanted dog teeth. Dent Traumatol. 2001;17:120–126. doi: 10.1034/j.1600-9657.2001.017003120.x. [DOI] [PubMed] [Google Scholar]
- 32.Shibata T, Komatsu K, Shimada A, Shimoda S, Oida S, Kawasaki K, et al. Effects of alendronate on restoration of biomechanical properties of periodontium in replanted rat molars. J Periodont Res. 2004;39:405–414. doi: 10.1111/j.1600-0765.2004.00755.x. [DOI] [PubMed] [Google Scholar]
- 33.Komatsu K, Shimada A, Shibata T, Shimoda S, Oida S, Kawasaki K, et al. Long‐term effects of local pretreatment with alendronate on healing of replanted rat teeth. J Periodont Res. 2008;43:194–200. doi: 10.1111/j.1600-0765.2007.01012.x. [DOI] [PubMed] [Google Scholar]