Abstract
Cnidarians are the oldest extant lineage of venomous animals. Despite their simple anatomy, they are capable of subduing or repelling prey and predator species that are far more complex and recently evolved. Utilizing specialized penetrating nematocysts, cnidarians inject the nematocyst content or “venom” that initiates toxic and immunological reactions in the envenomated organism. These venoms contain enzymes, potent pore forming toxins, and neurotoxins. Enzymes include lipolytic and proteolytic proteins that catabolize prey tissues. Cnidarian pore forming toxins self-assemble to form robust membrane pores that can cause cell death via osmotic lysis. Neurotoxins exhibit rapid ion channel specific activities. In addition, certain cnidarian venoms contain or induce the release of host vasodilatory biogenic amines such as serotonin, histamine, bunodosine and caissarone accelerating the pathogenic effects of other venom enzymes and porins. The cnidarian attacking/defending mechanism is fast and efficient, and massive envenomation of humans may result in death, in some cases within a few minutes to an hour after sting. The complexity of venom components represents a unique therapeutic challenge and probably reflects the ancient evolutionary history of the cnidarian venom system. Thus, they are invaluable as a therapeutic target for sting treatment or as lead compounds for drug design.
Keywords: cnidarians, venom, enzymes, pore forming toxins, neurotoxins, vasodilatory biogenic amines, human envenomation
1. Introduction
The phylum Cnidaria (corals, sea pens, sea anemones, jellyfish and hydroids) includes about 10,000 species living in aquatic habitats worldwide [1]. They range in size from the tiny Hydra spp, at less than 1 cm in length, to the massive lion’s mane jellyfish, Cyanea capillata with the bell diameter exceeding 2 m [2]. Envenomation hazard to humans also varies widely from non-hazardous to the infamous Chironex fleckeri (Australian box jellyfish), one of the most venomous animal dangerous to humans, as a meter of tentacle contact can provoke immediate cardiovascular collapse and death even within minutes after a sting [3]. The majority of cnidarians live in salt water habitats at different water depths. However, approximately 40 species, mostly hydrozoans [4] live in freshwater. Cnidarians are characteristically radially symmetrical [5], although they can also exhibit directional asymmetry or bilateral symmetry. For example, morphological studies on Siphonophores (class Hydrozoa) suggest that directional asymmetry has been gained and/or lost on multiple occasions [6], whilst most anthozoan polyps exhibit bilateral symmetry possessing two orthogonal body axes [5].
Despite the variety in size, toxicity, habitat and morphology several cellular characters are common to the members of Cnidaria, such as two unicellular layers (ectoderm and endoderm) separated by an extra-cellular matrix (mesoglea), neuromuscular systems and multiple sensory systems [7,8]. Molecular evidence and fossil data place the origin of cnidarins prior to the Ediacaran period ~750 million years ago, and major taxa diversification from the remaining metazoans prior to the Cambrian ~550 million years ago [9,10,11].
Since Cnidaria is an ancient clade of animals and the complexity and diversification of their venoms serve a unique therapeutic challenge (e.g., box jellyfishes (Cubozoa) venoms), transcriptomics and proteomics data for the identification and characterizing of their venom components is rapidly accumulating in recent times [12,13,14].
2. Cnidarian Phylogeny
Based upon mitochondrial DNA (mtDNA) data [15] and life cycles [8,16], cnidarians are divided into two extant subphyla: Anthozoa and Medusozoa. Anthozoans possess circular mtDNA, similar to that of other metazoans while medusozoans have atypical linear mtDNA. The members of medusozoan classes Hydrozoa, Schyphozoa, Cubozoa and Staurozoa display a triphasic life cycle in transition of generations: a free-swimming planula larva, a sessile polyp stage and sexual pelagic medusa stage. In anthozoans the medusa stage is lost and sessile adults represent the sexually propagating stage. The life cycle will be discussed in more details further in this review.
3. Cnidarian Life Cycle
There is significant morphological diversity in the cnidarian life cycle, as a single species may display a variety of forms whether it is sessile, polyp, tiny free-swimming planula larva or a pelagic medusoid. The life cycle of both medusozoans and anthozoans comprises sexual reproduction and an asexually budding phase. In medusozoans, the adult medusa is either male or female, and the fertilized egg (zygote) is retained inside the female’s gastric cavity [17,18]. However, in anthozoans, the polyp colonies may be single sex [19] or both male and female [20]. In general, the asexual life cycle of medusozoans includes a fertilized egg, which forms tiny pelagic planula larva that settles down to the sea floor and form a sessile polyp. These polyps further develop a hydroid polyp colony, which liberates medusae by budding from the trunk [18]. Amongst the medusozoans, hydrozoans have the greatest variation in life cycle. For example, species in the Campanulariidae family lack the medusa stage [21] and the members of the order Trachymedusae never form polyps [16]. The asexual life cycle of anthozoans is straightforward including four main stages: the fertilized egg, planula larvae, polyp and sessile sea anemone [16,22].
4. Cnidarian Venom Delivery System
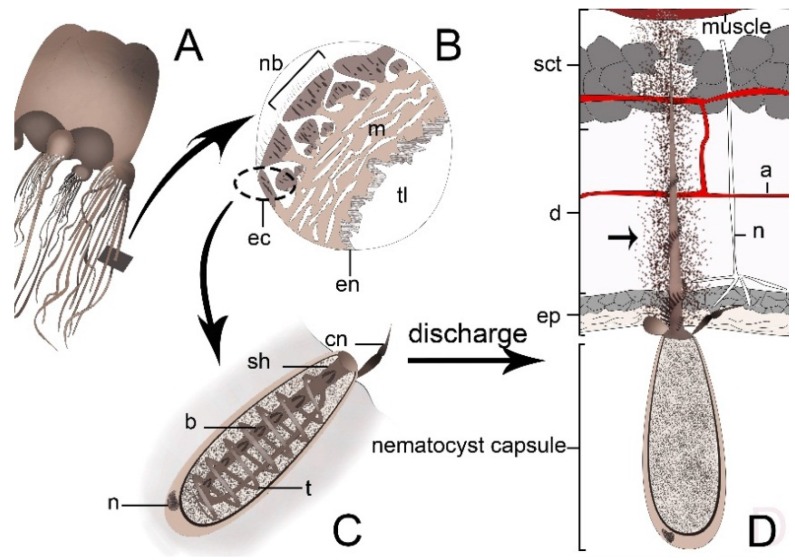
Cnidae are the defining subcellular specialisation of the phylum Cnidaria. They are specialized cellular structures capable of explosive discharge upon activation of cnidocytes (Figure 1). Cnidae contain elaborate structural elements and complex mixtures of bioactive compounds or “venom” for entrapping, subduing and digesting prey as well as deterring and repelling predators and competitors [23]. Cnidae are distributed in various parts of the cnidarian body and are classified into three major types: penetrant nematocysts, the volvent spirocyst and the glutinant ptychocysts.
Figure 1.
Overview of the Cnidarian venom delivery system. (A) Schematic picture of a jellyfish; (B) Transverse section of a tentacle showing two epithelial cell layers, ectoderm (ec) and endoderm (en), divided by the mesoglea (m), various types of cnidocytes placed in nematocyst batteries (nb) and in the cells lining the tentacular lumen (tl); (C) Undischarged nematocyst. The nematocyst capsule contains a cocktail of toxins, shaft (sh), coiled-hollow tubule (t), barbs (b), and nucleus (n). The cnidocil (cn) acts as a mechanoreceptor that upon activation stimulates the discharge mechanism; (D) Discharged nematocyst. Epidermis (ep), dermis (d), subcutaneous tissue (sct), artery (a), nerve (n). Toxin mixture is injected into the prey’s skin and subcutaneous tissues (arrow). The figure was drawn according to the data in references [14,29,34,35].
Cnidae comprise an eversible hollow tubule that is coiled and sometime spine laden. These phylum-specific organelles are synthesized in specialised precursor cells called cnidoblasts. Following their secretion from the Golgi apparatus, the cnidae undergo further structural modifications in the extracellular matrix before migrating to the tentacle surface [24,25,26].
Penetrant nematocysts inject venom into the target organism and are the most studied class of cnidae. Penetrant nematocysts are found in all cnidarians and are morphologically and functionally the most diverse group of cnidae [27]. They are the primary weapon for capturing prey, repelling predators, and intra- and interspecies spatial competition [28]. Although nematocysts are mostly located on the tentacles, they also exist on the outer surface of the bell in certain species of the Alatinidae, and Carybdeidae families [29], oral arms of Catostylus mosaicus (Schyphozoa) [30] and the stomach (gastric cirri) of some cubozoans [29] probably helping to paralyse and digest the prey. Members of family Actiniidae also have nematocysts in the ring around the base of their tentacles, called acrorhagi, which they use for intra- and interspecific competition [31] and at filaments called acontia that are used for defence or paralysis of prey.
Spirocysts are present in most anthozoans and may differ morphologically among different taxonomic groups [32]. The spirocyst capsule wall is thin and the everted tubule is helically folded. The tubule lacks spines, but contains adhering, hydroscopic substances that mechanically immobilize the prey [33]. Ptychocysts have been reported in tube-dwelling members of Actinaria and Ceriantharia. They create the protective tube in which species from these families live [33].
The mechanism of cnidae discharge in response to external stimuli (mechanical and chemical stimuli) is still not completely understood. The general osmotic/tension hypothesis proposes that the osmotic pressure of the intracapsular fluid temporary increases as a result of cnidae exposure to the external solution and subsequent exocytosis of cations from the capsule. The osmotic pressure differences across the capsule wall extant to the point that the preexisting intracapsular pressure exceeds a critical threshold value and triggers the cnidae discharge [36,37,38,39]. The capsule content (venom) is located on the inner surface of the inverted tubule which, during discharge (tubule eversion-extension), everts such that the outside is now exposed and injected into the prey. The inside of the tubule remains contiguous with the inside of the capsule and the cnidae content or venom is then expelled down the axis of the hollow tubule [40].
The morphological properties of cnidae are taxonomically informative and may aid in distinguishing more distantly related species [33]. For instance, the presence of spirocyst is characteristic of some anthozoans [22]. In addition, the relative abundance of different types of nematocyst, along with variation in the body size in several populations may correlate to environmental factors, such as specific ecosystems, prey size and selectivity in prey capture. For example, Chironex fleckeri has a relatively large body and a higher ratio of mastigophores (the longest of the penetrant cnidae) correlating to the large organisms which they prey upon [41]. In addition to cnidae, venom of Cnidarians was shown to be also synthesized in other cell types such as ectodermal and endodermal gland cells and other, still unknown, cell types [42,43].
5. Venom Composition
Since the early 20th century, numerous analytical experiments and clinical observations have established the toxicological diversity of cnidarian venoms (Table 1). The diversity of venom components range from non-proteinaceous compounds (e.g. purines, biogenic amines) to high molecular weight proteins evolved over the course of hundred million years. Interestingly, some toxin family types identified previously in other venomous animals comprise the venom arsenal of cnidarians. The most striking example is Kunitz peptides are expressed in sea anemones, cone snails, insects, scorpions, spiders, reptiles, ticks and vampire bat [90,91]. Another example of convergent expression of toxins is potassium channel blocker Kv1 which not only evolved convergently in scorpions and sea anemones, but they even use the same residues for blocking the channel pores [92].
Table 1.
Cnidarian venom composition.
| Toxin type | Found in class | Function | MW (kDa) | Reference |
|---|---|---|---|---|
| Enzymes | ||||
| Phospholipase A2 | Anthozoa, Cubozoa, Schyphozoa, Hydrozoa | Cytolytic, hemolytic, prey digestion | 13–45 | [44,45,46] |
| Metalloproteases | Schyphozoa, Cubozoa, Anthozoa | Cytotoxic, cytolytic, local tissue damage | 17–130 | [12,14,47,48] |
| Pore forming toxins (cytolysins) | ||||
| Actinoporins and actinoporin-like proteins | Anthozoa, Hydrozoa | Cytolytic, hemolytic, cardiovascular and respiratory arrest | 20 | [49,50,51,52,53,54] |
| Jellyfish Toxins | Cubozoa | Hemolytic, cardiotoxic, cytolytic, myotoxic, cutaneous inflammation | 42–46 | [13,55,56,57] |
| Hydralysins-related toxins | Hydrozoa, Anthozoa | Cytolytic, prey digestion | 27–31 | [43,58,59,60] |
| Membrane Attack Complex-Perforin | Anthozoa | Cytolytic, hemolytic | 60 | [61,62] |
| Neurotoxins | ||||
| NaTxs (type I-III) | Anthozoa | Neurotoxic, cardiotoxic, insecticide | 3–8 | [63,64,65,66,67] |
| KTxs (type I,III and IV,V KTxs) | Anthozoa | Neurotoxic, hypotensive, cardiotoxic, analgesic, antimicrobial, immunosuppressive, anti obesity | 3–4 | [68,69,70,71,72,73,74,75] |
| Kunitz peptides (type II KTxs) | Anthozoa | Paralytic, serine protease inhibitor | 6 | [76,77,78,79] |
| Small Cysteine-Rich Proteins (SCRiPs) and SCRiPs homologues | Anthozoa | Paralytic | 4.3–5.8 | [80] |
| ASIC Inhibitors | Anthozoa | Analgesic | 3 | [81,82] |
| TRPV1 Inhibitors | Anthozoa | Analgesic | 3 | [83,84] |
| Non-protein bioactive components | ||||
| Serotonin | Hydrozoa, Anthozoa | Vasodilation, sharp pain | - | [85] |
| Histamine | Anthozoa | Vasodilation, sharp pain | - | [86,87] |
| Bunodosine | Anthozoa | Analgesic | - | [88] |
| Caissarone | Anthozoa | Adenosine receptor antagonist | - | [89] |
5.1. Enzymes
5.1.1. Phospholipase A2
Phospholipase A2 (PLA2) activity has been detected in the homogenized tentacles and acontia of cnidarians including subphylums Anthozoa, Schyphozoa, Hydrozoa and Cubozoa [44,46,93]. This enzyme hydrolyzes the sn-2 acyl bond of glycerophospholipids to produce fatty acids including arachidonic acid and lysophospholipid [22,44]. It is commonly found in mammalian tissues where it plays important roles in vital body functions including dietary lipid catabolism, inflammation, signal transduction, and phospholipid remodelling [94]. However, the ubiquitous presence of PLA2 across venomous animal lineages such as reptiles (snakes and anguimorph lizard), centipedes, insects (their bristles, proboscises, and stingers), arachnids (scorpions, spiders, and ticks), cnidarians, and cephalopods [44,90,95,96,97,98] indicates the convergent recruitment of the body proteins into the toxic arsenal of animals. Toxic functions of PLA2 in cnidarian venoms have been proposed to include defence and immobilization and digestion of prey [44,93]. Also, hemolytic activity was demonstrated in a PLA2 fraction recovered from acontial nematocysts of the sea anemone Aiptasia pallida [45]. Although PLA2 enzymes are plentiful amongst cnidarians [44,98], their molecular structure-function relationships remain to be elucidated.
5.1.2. Metalloproteases
Metalloproteases are important venom components of terrestrial animals such as centipedes, snakes, and ticks [90,99]. They induce haemorrhage and necrosis by degrading the extracellular matrix and preventing blood clot formation [90,100]. These functions are commonly associated with several of the recurrent symptoms of sting such as skin damage, oedema, blister formation, myonecrosis and inflammation [100]. Metalloproteases were detected in the venom of jellyfish Stomolophus meleagris [12] and Chironex fleckeri [14]. A recent study focused on the enzymatic and cytotoxic functions of jellyfish metalloproteases and identified diverse proteolytic effects including gelatinolytic, caseinolytic, and fibrinolytic activities [47]. Zinc-dependent metalloproteases of the astacin family were detected in the soluble nematocyst content of the sea anemone Nematostella vectensis and were shown to be expressed in both gland cells and stinging cells. They originate from recruitment of the Bone Morphogenic Protein 1 (BMP1, also known as Tolloid) protein that plays a conserved role in animal development [48].
5.2. Pore Forming Toxins
Pore forming toxins (PFTs) appear to be present in all cnidarian venoms. The mechanism of action of these toxins is penetration through the target cell membrane resulting in diffusion of small molecules and solutes leading to osmotic imbalance and cell lysis. PFTs exhibit dual structure (i) a stable water-soluble structure that is monomeric and binds to the receptors on the target cell; (ii) membrane-bound structure consisting of oligomeric moleculs that form integral membrane pores [101]. Cnidarian PFTs are classified in two groups based on the type of secondary structure they use to penetrate membrane upon pore-forming activity: α-PFTs where they are a rich content of helices and form α-helical barrel structures, and β-PFTs that are rich in β-sheets and form β-barrel pores.
5.2.1. Actinoporins
Actinporins are α-PFTs and are present in Anthozoa and Hydrozoa. They are basic proteins approximately 20 kDa in molecular size lacking in intramolecular disulfide bonds. Actinoporins mediate various types of toxicity and bioactivity, such as cardiovascular and respiratory arrest in rats [50], lysis of chicken, goat, human and sheep erythrocytes [51,53,54] and cytotoxic effects [52], all caused by a pore-forming mechanism. Several studies have reported that actinoporins interact exclusively with sphingomyelin containing membranes [102,103,104], although sticholysin II from Stichodactyla helianthus binds to phosphatidylcholine membrane [105]. The mechanism of membrane penetration requires several steps: (i) initial binding to the target membrane which is accomplished by an exposed aromatic-rich loop; (ii) lateral orientation and consequent oligomerization by a phosphocolin binding site; and (iii) insertion of the N-terminal amphiphilic α-helix segment to the lipid membrane [102,103,106]. High-resolution nuclear magnetic resonance and Fourier transform infrared spectroscopy experiments [107], intrinsic fluorescence measurements [108], X-ray crystallography and electron microscopic analyses of two dimensional crystals [109] have suggested that, during the membrane penetration, the N-terminal amphiphilic α-helix segment of actinoporins detach from the main body and insert into the target membrane to produce pores with a diameter of 1–3 nm (~11–30 Å) [54,110].
5.2.2. Jellyfish Toxins (JFTs)
Cubozoan-related porins are the most potent and rapid-acting toxins secreted by jellyfish species. This toxin family was originally reported in the cubozoan Carybdea alata as CAH1 [111], also reported as CaTX-A/B [112], and subsequently identified in all cubozoans examined including CrTX-A/B from Carybdea rastoni [55], CqTX-A from Chiropsalmus quadrigatus [56], CfTX-1/2 and CfTX-A/B/Bt from Chironex fleckeri [13,113]. However, homologues of cubozoan porins were reported in Scyphozoa (Aurelia aurita), Hydrozoa (Hydra magnipapillata) [13], Anthozoa (Aiptasia pallida) and various hydroids (Hydractinia symbiolongicarpus and Hydra vulgaris), suggesting a common evolutionary origin of these toxins [80]. Characteristically, they are basic proteins with a molecular weight of 40 to 46 kDa and contain both α and β domains. The hypothetical mechanisms underlying pore formation involves oligomerization of several amphiphilic and hydrophobic α-helices in the N-terminal region of the toxin resulting in distortion of the plasma membrane and cell death [13,112,113]. The similarity in the three-dimensional structure of the N-terminal domain of CfTX toxins to that of the α-pore-forming domain (domain I) of the insecticidal δ-endotoxins from Bacillus thuringiensis suggests how toxins of this family insert into membranes. This mechanism of membrane insertion seem to be sufficient to explain the well-defined 12 nm (inner) and 25 nm (outer) diameter pores created in human erythrocytes by CfTX toxins [3].
Interestingly, the members of this toxin family exhibit varying target specify towards various vertebrate tissue. A recent study on CfTX toxins showed that CfTX-1/2 caused cardiovascular collapse within 1 min in anesthetized rats exposed in vivo to the venom, whereas CfTX-A/B more potent in eliciting in vitro hemolytic activity [13]. Although these data suggest that the jellyfish toxin family has undergone functional diversification, more structural/functional data are needed to understand the molecular evolutionary histories that formed this diversification.
5.2.3. Hydralysins-Related Toxins
In addition to porins found in cnidarians, recent studies have described a novel β-PFT family secreted from the digestive endodermal cells of green hydra (Chlorohydra viridissima) [43,58]. These non-nematocyst, body-derived toxins secreted during feeding are suggested to play a role in lysing prey tissues [59]. These toxins are not active on membrane phospholipids or carbohydrates, but rather bind to specific membrane receptors and form pores [58]. A recent study characterized aerolysin, a hydralysin homologue, secreted from pharyngeal ectodermal cell of N. vectensis and suggested a role in prey disintegration [60].
5.2.4. Membrane Attack Complex-Perforin
This group of β-PFT toxins have been detected in the venoms extracted from the sea anemones Phyllodiscus semoni and Actinaria villosa. The toxins may be used in prey capture/predator defence [61,62]. Membrane attack complex (MAC) proteins have been identified in the complement system produced by T-cells and NK cells. The proteins create a transmembrane pore into the target cell and initiate various apoptotic cell death pathways [114].
5.3. Neurotoxins
Cnidarian neurotoxins (voltage-gated ion channel toxins) are a group of low molecular weight peptides and are among the best characterized toxins in terms of the mechanism of action. They are produced by sea anemones and have a fundamental role in the venom to help these sessile animals to immobilize the prey rapidly and to defend against predators. They prolong the action potential of the excitable and non-excitable membranes in sensory neurons and cardiac and skeletal muscle cells [64,115] via modifying the sodium channel gating [64,116,117] or blocking the potassium channel gating during the repolarisation stage [115]. This causes the cell to become hyperactive and to release massive amounts of neurotransmitter at synapses and neuromuscular junctions that can cause initial spastic stage followed by descending flaccid paralysis. Sea anemone voltage-gated ion channel toxins have been studied extensively because they are valuable bioresources to study the structure and function of sodium and potassium channels and also to be used for development of drugs [73,74] and bioinsecticides [66,67].
Several cnidarian neurotoxin polypeptides are exclusively block Vanilloid Receptor 1 (TRPV1) and acid-sensing ion channel 3 (ASIC3), which take part in initiation and transduction of pain and hyper-algesia. These peptides are thus promising tools for the development of novel pain reducers.
5.3.1. Voltage-Gated Sodium (Nav) Channel Toxins
Voltage-gated sodium (Nav) channel toxins (NaTxs) are transmembrane complexes composed of several subunits. The highly conserved α-subunit consists of four homologous domains (D1–D4) each containing six hydrophobic transmembrane regions (S1–S6) [117]. Anthozoan NaTxs and several other groups of toxins from scorpion and spiders bind to site 3 (loop S3–S4 in D4) of the Nav channels [64,118] resulting in considerable neurotransmitter release in synapses. One possible explanation underlying its toxicity is the electrostatic interaction between a cluster of basic amino acids on the toxin with acidic amino acids at site 3 [119] locking S4 segment in its inward position, thus inhibiting the conformational changes of the channel necessary for fast inactivation [117]. These toxins are divided into three groups: (i) type I NaTx; and (ii) type II NaTxs exhibit extensive sequence similarity and share similar function. In addition to type I and II NaTxs there is an orphan NaTx clade, that exhibits only a partial homology to both type I and type II NaTxs and shows similar mechanism of action to those [120,121]. Lastly, there are short (~30 amino acids long) peptides in sea anemone venom that exhibit a similar activity to the rest of NaTxs families despite lacking any shared sequence motifs with the rest of them [118]. These peptides form the type III family and exhibit very high selectivity towards arthropod sodium channels [122].
5.3.2. Voltage-gated Potasium (Kv) Channel Toxins and Kunitz peptides
Sea anemone voltage-gated potasoim (Kv) channel toxins (KTxs) are categorized in five groups based on their sequence similarity and binding affinity towards different Kv channel families [72]. Type I KTxs have a molecular weight of 4 kDa with three disulfide bridges. They inhibit the potassium current through channel Kv1. and Kv3. subfamilies and intermediate conductance calcium-activated potasoim channels [22]. Type II KTxs are Kunitz peptides with a molecular weight of 6 kDa that are cross-linked with three disulfide bridges and are remarkable in their dual function of activity. Usually they act against trypsin and chymotrypsin proteinases in order to inhibit the rapid degradation of the venom protease by endogenous enzymes of the animals themselves or of the prey [31,78,79]. Interestingly, several sea anemone Kunitz peptides possess both Kv blocking activity similar to dendrotoxin and protease inhibiting activity [76,123]. For example, kalicludin 1-3 from A. sulcata binds competitively to Kv1.2 channels to paralyse the prey rapidly. Type III KTxs are 3–4 kDa peptides with three disulfide bridges that have evolved from NaTxs under the regime of positive selection [80]. They block a variety of distinct potassium ion channels such as Kv3.4 channel belonging to the rapidly inactivating Kv channel [68] and hERG (the human Ether-à-go-go-Related Gene) [69,75]. Interestingly, the influence of positive selection does not result in the complete loss of sodium ion channel blocking activity, where several type III KTxs target both hERG and acid-sensing ion channel 3 (ASIC3, H+-gated Nav channels) [75]. Type IV are structurally new peptides from the sea anemone Stichodactyla haddoni displaying crab paralyzis activity and are cross-linked with two disulfide bridges [124]. Type V has been found in the sea anemone Bunodosoma caissarum and is cross-linked by four disulfide bridges. This novel peptide is active on Drosophila Shaker IR channels [72].
5.3.3. Small Cysteine-Rich Peptides (SCRiPs)
This newly discovered group of neurotoxins have been reported in the ectoderm of reef-building corals Acropora millepora and SCRiP homologs have been retrieved in sea anemones, Anemonia viridis and Metridium senile [80]. The injection of recombinantly expressed A. millepora SCRiPs in zebrafish (Danio rerio) larvae resulted in severe paralysis, suggesting the first peptide neurotoxin family described from scleractinian corals [80].
5.3.4. ASIC Inhibitors
ASICs are sodium-selective acid-sensing ion channels (ASIC) expressed in peripheral neuronal system. They have been associated with acidic pain during pathological conditions such as inflammation and ischemia. Recently, a novel peptide π-AnmTX Ugr 9a-1 from the venom of the sea anemone Urticina grebelnyi [81] and PhcrTx1 from Phymanthus crucifer [82] have been reported to target ASIC channels. These peptides are cross-linked by two disulfide bridges and have no sequence homology to other sea anemone neurotoxin peptides.
5.3.5. TRPV1 Inhibitors
TRPV1s are non-selective cation channels expressed in mammalians peripheral and central neuronal systems. They initiate neuronal response during inflammation stimuli, which allow them to be regarded as one of the most important molecular triggers of pain stimuli. The first peptidic TRPV1 inhibitor isolated from sea anemone venom was τ-SHTX-Hcr2b (APHC1) from Heteractis crispa [83]. Subsequently, two homologous peptides (τ-SHTX-Hcr2c (APHC2) and τ-SHTX-Hcr2d (APHC3) that target TRPV1 were isolated from the same species [84]. These novel neurotoxins are promising new models for designing a new generation of analgesic drugs.
5.4. Non-Protein Bioactive Components
In addition to the protein and peptide compounds mentioned above, a number of pharmacologically active low molecular weight compounds have been detected in cnidarian venoms. Large amounts of 5-hydroxytryptamine (5-HT, serotonin) have been reported in the water where hydra was stimulated electrically [85]. This presumably nematocyst-derived substance causes instant pain in predators and thus may have a defensive role [85]. Also the vasodilatation enhanced by serotonin may potentiate the effect of other venom components [85]. Histamine has been detected in the homogenised tentacles of the sea anemones Anemonia viridis (previously known as Anemonia sulcata) and Actinia equina [86,87]. Like serotonin, histamine produces sharp pain and increases vascular permeability. Although these compounds have not been directly detected in isolated nematocysts, it is likely that they are involved in cnidarian envenomation, and treatment of sting victims with antihistamines has been recommended to relieve the symptoms [125]. Bunodosine, an N-acylamino acid, was purified from the venom of the sea anemone Bunodosoma cangicum. This compound exhibits a potent analgesic activity by activation of serotonin receptors [88]. Caissarone is a quaternary purine derivative isolated from the sea anemone Bunodosoma caissarum by extracting the whole animal in acetone. This marine product has a high antagonist activity towards guinea-pig ileum adenosine receptors which stimulate gut function [89].
6. The Role of Cnidarians Venoms in Drug Discovery
Toxic compounds isolated from Cnidaria have been viewed to produce several serious implications to human health due to their neurotoxicity, cytotoxicity and tissue damage. However, novel findings have demonstrated that their toxins might offer a tool to study cell physiology [126] and provide promising sources of pharmacological lead/active agents for therapy of human diseases.
Palytoxin is a highly potent non-protein toxin isolated from order Zoantharia (soft corals) of the genus Palythoa and Zoanthus and the sea anemone Radianthus macrodactylus [127]. The interaction of palytoxin with Na+, K+-ATPase pump in almost every excitable tissue induces passive ion conductance which triggers K efflux, Na influx and massive membrane depolarization and tissue contraction [127,128]. Palytoxin has been reported for anti-cancer activity against head and neck carcinoma cells [129], Ehrlich ascites tumour and P-388 lymphocytic leukaemia cells [130]. The mechanism of action of its cytotoxicity and tumour suppressor activity has been established by actin filament distortion and apoptosis [131]. Conversely, palytoxin has been identified as a tumor promoter by disrupting the regulation of cellular signalling cascades [132].
Over the past decade, several cytolysins and protease inhibitors have been extracted from the sea anemone A. equina. Equinatoxin II (EqT II) is a pore-forming protein that has been shown to have significant toxicity against Ehrlich ascites tumour and L1210 leukaemia cell lines [133] and diploid lung fibroblasts of the Chinese hamster [134]. Equistatin is a potent inhibitor of papain-like cysteine proteinase and aspartic proteinase cathepsin D [135]. Overexpression and hypersecretion of cathepsin-D has been reported in breast carcinoma cells [136], and papain-like cysteine proteases are involved in diseases of the central nervous system [137].
Recently antibutyrylcholinestrasic activity was detected in the crude venom extracted from the tentacle material of the Mediterranean jellyfish Pelagia noctiluca. Inhibition of butylcholinestrase in the central nervous system may prove useful in the treatment of neurodegenerative diseases such as Alzheimer’s disease and senile dementia [138].
ShK is a potent Kv1.3 channel blocker toxin that isolated from the sea anemone Stoichactis helianthus. Since this channel is crucial in the activation (proliferation and cytokine production) of human effector memory Tcells (TEM), ShK could provide a valuable immunosuppressant for the treatment of autoimmune diseases mediated by T cells [73]. Kv1.3 blockers are also considered as a therapeutic target for the treatment of obesity, thus highlighting the potential use of ShK in treatment of obesity and insulin resistance [74].
7. Envenomation
Fortunately, due to the biophysical properties of the discharge event many cnidarians lack the capacity to penetrate human skin. Further, stings by most species with cnidae capable of perforating human tissue lead only to a negligible to moderate transient irritation/burning sensation. In contrast, contact with several jellyfish, sea anemone and coral species can cause severe pain, tissue damage and even cardiovascular collapse and death. The venom of the hazardous animals is introduced into the target tissue (epidermis, dermis, vasculature, lymphatic system and probably in some cases subcutaneous or muscular structures) (Figure 1) [125] and initiates immediate and delayed immunological and toxicological responses [139,140,141]. Envenomation symptoms are determined by the contents of the venom, the volume of the injection, the health of the patient and the duration of the tentacle-skin contact [125].
One of the most important steps in the treatment of human envenomation is to use specific fluids to prevent further nematocyst discharge, since physical attempts to detach remaining tentacles from the victim’s skin might cause massive discharge [34,142]. Since 1908 numerous traditional remedies such as urea, seawater, vinegar, methylated spirits etc. have been used to inactivate the undischarged nematocysts on the adhered tentacles and/or to alleviate pain. These chemicals have been used to varying degrees of success, ranging from complete success in certain species to complete failure in others. For instance, while certain chemicals such as ethanol, or 5% acetic acid in distilled water, cause massive nematocyst discharge in Hydrozoa and Cubozoa species [14,143,144], some other such as a food grade vinegar was found to inactivate the penetrating nematocysts of C. fleckeri rapidly and completely [34].
Avoidance and treatment of human envenomation by cnidarian species is an increasingly important objective within the broader scientific community. It also represents a collaborative aim of medical, pharmacological, toxicological and biological disciplines towards developing improved understanding of field ecologies as well as effective therapeutics to minimize pathogenic impact on bathers. It also can improve our understanding of the food web and biome dynamics of these ancient and intriguing animals.
8. Conclusions
Members of Cnidaria phylum are ancestral venomous Eumetazoan with a unique phylogeny. In spite of recent efforts to extract and characterize novel toxins from cnidarians, much more remains to be done to investigate their toxins and their potential as new sources of therapeutic substances. Of the approximately 10,000 cnidarian species, only 156 toxins from 10 pharmacological families have been defined. Potentially useful new toxicological information is gradually accumulating. One promising solution towards broadening the knowledge on cnidarian toxins is to apply high throughput sequencing technologies to identify novel structural and pharmacological groups of toxins [145,146]. Nevertheless, the enormous potential of cnidarian venoms for understanding the activity of different receptors and channels in health and disease conditions, as well as their use as sources of novel pharmaceutical lead compounds, appears to offer endless possibilities for future scientific research.
Acknowledgments
Mahdokht Jouiaei was funded by University of Queensland Ph.D. scholarships. Bryan G. Fry was funded by the Australian Research Council. Timo J. Nevalainen was funded by the European Community’s Seventh Framework Programme (FP7/2007-2013) under grant agreement n° 227799-ASSEMBLE. We thank Yehu Moran for his constructive criticism.
Abbreviation
- rDNA
ribosomal DNA
- mtDNA
mitochondrial DNA
- PLA2
Phospholipase A2
- MACP
Membrane Attack Complex-Perforin
- SCRiPs
Small cysteine-rich peptides
Author Contributions
Mahdokht Jouiaei wrote and revised the manuscript and provided figures. Angel A. Yanagihara, Bruno Madio, Yehu Moran, Timo J. Nevalainen, Paul Alewood and Bryan G. Fry revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Zhang Z. Animal biodiversity: An introduction to higher-level classification and taxonomic richness. Zootaxa. 2011;3148:7–12. doi: 10.11646/zootaxa.3703.1.1. [DOI] [PubMed] [Google Scholar]
- 2.Higgins J., Ford M., Costello J. Transitions in morphology, nematocyst distribution, fluid motions, and prey capture during development of the scyphomedusa Cyanea capillata. Biol. Bull. 2008;214:29–41. doi: 10.2307/25066657. [DOI] [PubMed] [Google Scholar]
- 3.Yanagihara A.A., Shohet R.V. Cubozoan venom-induced cardiovascular collapse is caused by hyperkalemia and prevented by zinc gluconate in mice. PLoS ONE. 2012;7:e51368. doi: 10.1371/journal.pone.0051368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jankowski T., Collins A.G., Campbell R. Global diversity of inland water cnidarians. Hydrobiologia. 2008;595:35–40. doi: 10.1007/s10750-007-9001-9. [DOI] [Google Scholar]
- 5.Finnerty J.R., Pang K., Burton P., Paulson D., Martindale M.Q. Origins of bilateral symmetry: Hox and dpp expression in a sea anemone. Science. 2004;304:1335–1337. doi: 10.1126/science.1091946. [DOI] [PubMed] [Google Scholar]
- 6.Dunn C.W., Wagner G.P. The evolution of colony-level development in the Siphonophora (Cnidaria: Hydrozoa) Dev. Genes Evol. 2006;216:743–754. doi: 10.1007/s00427-006-0101-8. [DOI] [PubMed] [Google Scholar]
- 7.Moroz L.L., Kocot K.M., Citarella M.R., Dosung S., Norekian T.P., Povolotskaya I.S., Grigorenko A.P., Dailey C., Berezikov E., Buckley K.M., et al. The ctenophore genome and the evolutionary origins of neural systems. Nature. 2014;510:109–114. doi: 10.1038/nature13400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Technau U., Steele R.E. Evolutionary crossroads in developmental biology: Cnidaria. Development. 2011;138:1447–1458. doi: 10.1242/dev.048959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iten H.V., Leme J.d.M., Marques A.C., Simões M.G. Alternative interpretations of some earliest Ediacaran fossils from China. Acta Palaeontol. Pol. 2013;58:111–113. [Google Scholar]
- 10.Park E., Hwang D.S., Lee J.S., Song J.I., Seo T.K., Won Y.J. Estimation of divergence times in cnidarian evolution based on mitochondrial protein-coding genes and the fossil record. Mol. Phylogenet. Evol. 2012;62:329–345. doi: 10.1016/j.ympev.2011.10.008. [DOI] [PubMed] [Google Scholar]
- 11.Park T.Y., Woo J., Lee D.J., Lee D.C., Lee S.B., Han Z., Chough S.K., Choi D.K. A stem-group cnidarian described from the mid-Cambrian of China and its significance for cnidarian evolution. Nat. Commun. 2011;2:442. doi: 10.1038/ncomms1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li R., Yu H., Xue W., Yue Y., Liu S., Xing R., Li P. Jellyfish Venomics and Venom Gland Transcriptomics Analysis of Stomolophus meleagris to Reveal the Toxins Associated with Sting. J. Proteomics. 2014;106:17–29. doi: 10.1016/j.jprot.2014.04.011. [DOI] [PubMed] [Google Scholar]
- 13.Brinkman D.L., Konstantakopoulos N., McInerney B.V., Mulvenna J., Seymour J.E., Isbister G.K., Hodgson W.C. Chironex fleckeri (Box Jellyfish) venom proteins: expansion of a cnidarian toxin family that elicits variable cytolytic and cardiovascular effects. J. Biol. Chem. 2014;289:4798–4812. doi: 10.1074/jbc.M113.534149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jouiaei M., Casewell N.R., Yanagihara A.A., Nouwens A., Cribb B.W., Whitehead D., Jackson T.N., Ali S.A., Wagstaff S.C., Koludarov I. Firing the sting: chemically induced discharge of cnidae reveals novel proteins and peptides from Box Jellyfish (Chironex fleckeri) venom. Toxins. 2015;7:936–950. doi: 10.3390/toxins7030936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kayal E., Roure B., Philippe H., Collins A.G., Lavrov D.V. Cnidarian phylogenetic relationships as revealed by mitogenomics. BMC. Evol. Biol. 2013;13:5. doi: 10.1186/1471-2148-13-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Collins A.G. Phylogeny of Medusozoa and the evolution of cnidarian life cycles. J.Evol. Biol. 2002;15:418–432. doi: 10.1046/j.1420-9101.2002.00403.x. [DOI] [Google Scholar]
- 17.Freeman G., Miller R.L. Hydrozoan eggs can only be fertilized at the site of polar body formation. Dev. Biol. 1982;94:142–152. doi: 10.1016/0012-1606(82)90077-X. [DOI] [PubMed] [Google Scholar]
- 18.Galliot B., Schmid V. Cnidarians as a model system for understanding evolution and regeneration. Int. J. Dev. Biol. 2002;46:39–48. [PubMed] [Google Scholar]
- 19.Hand C., Uhlinger K.R. The culture, sexual and asexual reproduction, and growth of the sea anemone Nematostella vectensis. Biol. Bull. 1992;182:169–176. doi: 10.2307/1542110. [DOI] [PubMed] [Google Scholar]
- 20.Loya Y. Settlement, mortality and recruitment of a red sea scleractini an coral population. Coelenterate Ecol. Behav. 1976;3:89. [Google Scholar]
- 21.Govindarajan A.F., Boero F., Halanych K.M. Phylogenetic analysis with multiple markers indicates repeated loss of the adult medusa stage in Campanulariidae (Hydrozoa, Cnidaria) Mol. Phylogenet. Evol. 2006;38:820–834. doi: 10.1016/j.ympev.2005.11.012. [DOI] [PubMed] [Google Scholar]
- 22.Frazao B., Vasconcelos V., Antunes A. Sea anemone (Cnidaria, Anthozoa, Actiniaria) toxins: An overview. Mar. Drugs. 2012;10:1812–1851. doi: 10.3390/md10081812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Orts D.J., Peigneur S., Madio B., Cassoli J.S., Montandon G.G., Pimenta A.M., Bicudo J.E., Freitas J.C., Zaharenko A.J., Tytgat J. Biochemical and Electrophysiological Characterization of Two Sea Anemone Type 1 Potassium Toxins from a Geographically Distant Population of Bunodosoma caissarum. Mar. Drugs. 2013;11:655–679. doi: 10.3390/md11030655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Beckmann A., Ozbek S. The nematocyst: A molecular map of the cnidarian stinging organelle. Int. J. Dev. Biol. 2012;56:577–582. doi: 10.1387/ijdb.113472ab. [DOI] [PubMed] [Google Scholar]
- 25.Ozbek S. The cnidarian nematocyst: A miniature extracellular matrix within a secretory vesicle. Protoplasma. 2011;248:635–640. doi: 10.1007/s00709-010-0219-4. [DOI] [PubMed] [Google Scholar]
- 26.Fautin D.G. Structural diversity, systematics, and evolution of cnidae. Toxicon. 2009;54:1054–1064. doi: 10.1016/j.toxicon.2009.02.024. [DOI] [PubMed] [Google Scholar]
- 27.David C.N., Ozbek S., Adamczyk P., Meier S., Pauly B., Chapman J., Hwang J.S., Gojobori T., Holstein T.W. Evolution of complex structures: minicollagens shape the cnidarian nematocyst. Trends Genet. 2008;24:431–438. doi: 10.1016/j.tig.2008.07.001. [DOI] [PubMed] [Google Scholar]
- 28.Tardent P. The cnidarian cnidocyte, a hightech cellular weaponory. BioEssays. 1995;17:351–362. doi: 10.1002/bies.950170411. [DOI] [Google Scholar]
- 29.Gershwin L. Nematocysts of the Cubozoa. Zootaxa. 2006;1232:1–57. [Google Scholar]
- 30.Peach M.B., Pitt K.A. Morphology of the nematocysts of the medusae of two scyphozoans, Catostylus mosaicus and Phyllorhiza punctata (Rhizostomeae): implications for capture of prey. Invertebrate Biol. 2005;124:98–108. doi: 10.1111/j.1744-7410.2005.00012.x. [DOI] [Google Scholar]
- 31.Minagawa S., Sugiyama M., Ishida M., Nagashima Y., Shiomi K. Kunitz-type protease inhibitors from acrorhagi of three species of sea anemones. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 2008;150:240–245. doi: 10.1016/j.cbpb.2008.03.010. [DOI] [PubMed] [Google Scholar]
- 32.Rifkin J. A study of the spirocytes from the Ceriantharia and Actiniaria (Cnidaria: Anthozoa) Cell Tissue Res. 1991;266:365–373. doi: 10.1007/BF00318192. [DOI] [Google Scholar]
- 33.Östman C. A guideline to nematocyst nomenclature and classification, and some notes on the systematic value of nematocysts. Sci. Mar. 2000;64:31–46. [Google Scholar]
- 34.Hartwick R., Callanan V., Williamson J. Disarming the box-jellyfish: nematocyst inhibition in Chironex fleckeri. Med. J. Aust. 1980;1:15–20. [PubMed] [Google Scholar]
- 35.Rifkin J.F., Burnett J.W., Fenner P.J. Venomous and poisonous marine animals: A medical and biological handbook. NewSouth Publishing; Sydney, Australia: 1996. [Google Scholar]
- 36.Endean R., Rifkin J., Daddow L. Envenomation by the box-jellyfish Chironex fleckeri: How nematocysts discharge. Hydrobiologia. 1991;216:641–648. doi: 10.1007/BF00026525. [DOI] [Google Scholar]
- 37.Hidaka M. Advances in comparative and environmental physiology. Springer-Verlag; Berlin, Germany: 1993. Mechanism of nematocyst discharge and its cellular control; pp. 45–76. [Google Scholar]
- 38.Holstein T., Tardent P. An ultrahigh-speed analysis of exocytosis: Nematocyst discharge. Science. 1984;223:830–833. doi: 10.1126/science.6695186. [DOI] [PubMed] [Google Scholar]
- 39.Özbek S., Balasubramanian P.G., Holstein T.W. Cnidocyst structure and the biomechanics of discharge. Toxicon. 2009;54:1038–1045. doi: 10.1016/j.toxicon.2009.03.006. [DOI] [PubMed] [Google Scholar]
- 40.Lotan A., Fishman L., Loya Y., Zlotkin E. Delivery of a nematocyst toxin. Nature. 1995;375:456. doi: 10.1038/375456a0. [DOI] [PubMed] [Google Scholar]
- 41.Carrette T., Alderslade P., Seymour J. Nematocyst ratio and prey in two Australian cubomedusans, Chironex fleckeri and Chiropsalmus sp. Toxicon. 2002;40:1547–1551. doi: 10.1016/S0041-0101(02)00168-X. [DOI] [PubMed] [Google Scholar]
- 42.Moran Y., Genikhovich G., Gordon D., Wienkoop S., Zenkert C., Ozbek S., Technau U., Gurevitz M. Neurotoxin localization to ectodermal gland cells uncovers an alternative mechanism of venom delivery in sea anemones. Proc. Biol. Sci. 2012;279:1351–1358. doi: 10.1098/rspb.2011.1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang M., Fishman Y., Sher D., Zlotkin E. Hydralysin, a novel animal group-selective paralytic and cytolytic protein from a noncnidocystic origin in hydra. Biochemistry. 2003;42:8939–8944. doi: 10.1021/bi0343929. [DOI] [PubMed] [Google Scholar]
- 44.Nevalainen T.J., Peuravuori H.J., Quinn R.J., Llewellyn L.E., Benzie J.A., Fenner P.J., Winkel K.D. Phospholipase A2 in cnidaria. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2004;139:731–735. doi: 10.1016/j.cbpc.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 45.Hessinger D.A., Lenhoff H.M. Mechanism of hemolysis induced by nematocyst venom: Roles of phospholipase A and direct lytic factor. Arch. Biochem. Biophys. 1976;173:603–613. doi: 10.1016/0003-9861(76)90297-6. [DOI] [PubMed] [Google Scholar]
- 46.Mariottini G.L., Pane L. Cytotoxic and cytolytic cnidarian venoms. A review on health implications and possible therapeutic applications. Toxins. 2014;6:108–151. doi: 10.3390/toxins6010108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee H., Jung E.-S., Kang C., Yoon W.D., Kim J.-S., Kim E. Scyphozoan jellyfish venom metalloproteinases and their role in the cytotoxicity. Toxicon. 2011;58:277–284. doi: 10.1016/j.toxicon.2011.06.007. [DOI] [PubMed] [Google Scholar]
- 48.Moran Y., Praher D., Schlesinger A., Ayalon A., Tal Y., Technau U. Analysis of soluble protein contents from the nematocysts of a model sea anemone sheds light on venom evolution. Mar. Biotechnol. 2013;15:329–339. doi: 10.1007/s10126-012-9491-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Uechi G.-I., Toma H., Arakawa T., Sato Y. Biochemical and physiological analyses of a hemolytic toxin isolated from a sea anemone Actineria villosa. Toxicon. 2005;45:761–766. doi: 10.1016/j.toxicon.2005.01.015. [DOI] [PubMed] [Google Scholar]
- 50.Hu B., Guo W., Wang L.H., Wang J.G., Liu X.Y., Jiao B.H. Purification and characterization of gigantoxin-4, a new actinoporin from the sea anemone Stichodactyla gigantea. Int. J. Biol. Sci. 2011;7:729–739. doi: 10.7150/ijbs.7.729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ravindran V.S., Kannan L., Venkateshvaran K. Biological activity of sea anemone proteins: II. Cytolysis and cell line toxicity. Indian J. Exp. Biol. 2010;48:1233–1236. [PubMed] [Google Scholar]
- 52.Fedorov S., Dyshlovoy S., Monastyrnaya M., Shubina L., Leychenko E., Kozlovskaya E., Jin J.-O., Kwak J.-Y., Bode A.M., Dong Z. The anticancer effects of actinoporin RTX-A from the sea anemone Heteractis crispa (Radianthus macrodactylus) Toxicon. 2010;55:811–817. doi: 10.1016/j.toxicon.2009.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kohno Y., Satoh H., Iguchi A., Nagai H. Characterization of a new hemolytic protein toxin from the sea anemone Anthopleura asiatica. Fish. Sci. 2009;75:1049–1054. doi: 10.1007/s12562-009-0112-2. [DOI] [Google Scholar]
- 54.Glasser E., Rachamim T., Aharonovich D., Sher D. Hydra actinoporin-like toxin-1, an unusual hemolysin from the nematocyst venom of Hydra magnipapillata which belongs to an extended gene family. Toxicon. 2014;91:103–113. doi: 10.1016/j.toxicon.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 55.Nagai H., Takuwa K., Nakao M., Ito E., Miyake M., Noda M., Nakajima T. Novel proteinaceous toxins from the box jellyfish (sea wasp) Carybdea rastoni. Biochem. Biophys. Res. Commun. 2000;275:582–588. doi: 10.1006/bbrc.2000.3353. [DOI] [PubMed] [Google Scholar]
- 56.Nagai H., Takuwa-Kuroda K., Nakao M., Oshiro N., Iwanaga S., Nakajima T. A novel protein toxin from the deadly box jellyfish (Sea Wasp, Habu-kurage) Chiropsalmus quadrigatus. Biosci. Biotechnol. Biochem. 2002;66:97–102. doi: 10.1271/bbb.66.97. [DOI] [PubMed] [Google Scholar]
- 57.Ramasamy S., Isbister G.K., Seymour J.E., Hodgson W.C. The in vitro effects of two chirodropid (Chironex fleckeri and Chiropsalmus sp.) venoms: efficacy of box jellyfish antivenom. Toxicon. 2003;41:703–711. doi: 10.1016/s0041-0101(03)00046-1. [DOI] [PubMed] [Google Scholar]
- 58.Sher D., Fishman Y., Zhang M., Lebendiker M., Gaathon A., Mancheno J.M., Zlotkin E. Hydralysins, a new category of β-pore-forming toxins in cnidaria. J. Biol. Chem. 2005;280:22847–22855. doi: 10.1074/jbc.M503242200. [DOI] [PubMed] [Google Scholar]
- 59.Sher D., Fishman Y., Melamed-Book N., Zhang M., Zlotkin E. Osmotically driven prey disintegration in the gastrovascular cavity of the green hydra by a pore-forming protein. FASEB J. 2008;22:207–214. doi: 10.1096/fj.07-9133com. [DOI] [PubMed] [Google Scholar]
- 60.Moran Y., Fredman D., Szczesny P., Grynberg M., Technau U. Recurrent horizontal transfer of bacterial toxin genes to eukaryotes. Mol. Biol. Evol. 2012;29:2223–2230. doi: 10.1093/molbev/mss089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Oshiro N., Kobayashi C., Iwanaga S., Nozaki M., Namikoshi M., Spring J., Nagai H. A new membrane-attack complex/perforin (MACPF) domain lethal toxin from the nematocyst venom of the Okinawan sea anemone Actineria villosa. Toxicon. 2004;43:225–228. doi: 10.1016/j.toxicon.2003.11.017. [DOI] [PubMed] [Google Scholar]
- 62.Nagai H., Oshiro N., Takuwa-Kuroda K., Iwanaga S., Nozaki M., Nakajima T. Novel proteinaceous toxins from the nematocyst venom of the Okinawan sea anemone Phyllodiscus semoni Kwietniewski. Biochem. Biophys. Res. Commun. 2002;294:760–763. doi: 10.1016/S0006-291X(02)00547-8. [DOI] [PubMed] [Google Scholar]
- 63.Norton T.R. Cardiotonic polypeptides from Anthopleura xanthogrammica (Brandt) and A. elegantissima (Brandt) Fed. Proc. 1981;40:21–25. [PubMed] [Google Scholar]
- 64.Smith J.J., Blumenthal K.M. Site-3 sea anemone toxins: molecular probes of gating mechanisms in voltage-dependent sodium channels. Toxicon. 2007;49:159–170. doi: 10.1016/j.toxicon.2006.09.020. [DOI] [PubMed] [Google Scholar]
- 65.Bruhn T., Schaller C., Schulze C., Sanchez-Rodriguez J., Dannmeier C., Ravens U., Heubach J.F., Eckhardt K., Schmidtmayer J., Schmidt H., et al. Isolation and characterisation of five neurotoxic and cardiotoxic polypeptides from the sea anemone Anthopleura elegantissima. Toxicon. 2001;39:693–702. doi: 10.1016/S0041-0101(00)00199-9. [DOI] [PubMed] [Google Scholar]
- 66.Yan F., Cheng X., Ding X., Yao T., Chen H., Li W., Hu S., Yu Z., Sun Y., Zhang Y., et al. Improved insecticidal toxicity by fusing Cry1Ac of Bacillus thuringiensis with Av3 of Anemonia viridis. Curr. Microbiol. 2014;68:604–609. doi: 10.1007/s00284-013-0516-1. [DOI] [PubMed] [Google Scholar]
- 67.Bosmans F., Tytgat J. Sea anemone venom as a source of insecticidal peptides acting on voltage-gated Na+ channels. Toxicon. 2007;49:550–560. doi: 10.1016/j.toxicon.2006.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Diochot S., Schweitz H., Beress L., Lazdunski M. Sea anemone peptides with a specific blocking activity against the fast inactivating potassium channel Kv3.4. J. Biol. Chem. 1998;273:6744–6749. doi: 10.1074/jbc.273.12.6744. [DOI] [PubMed] [Google Scholar]
- 69.Diochot S., Loret E., Bruhn T., Beress L., Lazdunski M. APETx1, a new toxin from the sea anemone Anthopleura elegantissima, blocks voltage-gated human ether-a-go-go-related gene potassium channels. Mol. Pharmacol. 2003;64:59–69. doi: 10.1124/mol.64.1.59. [DOI] [PubMed] [Google Scholar]
- 70.Diochot S., Baron A., Rash L.D., Deval E., Escoubas P., Scarzello S., Salinas M., Lazdunski M. A new sea anemone peptide, APETx2, inhibits ASIC3, a major acid-sensitive channel in sensory neurons. EMBO J. 2004;23:1516–1525. doi: 10.1038/sj.emboj.7600177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ovchinnikova T.V., Balandin S.V., Aleshina G.M., Tagaev A.A., Leonova Y.F., Krasnodembsky E.D., Men’shenin A.V., Kokryakov V.N. Aurelin, a novel antimicrobial peptide from jellyfish Aurelia aurita with structural features of defensins and channel-blocking toxins. Biochem. Biophys. Res. Commun. 2006;348:514–523. doi: 10.1016/j.bbrc.2006.07.078. [DOI] [PubMed] [Google Scholar]
- 72.Orts D.J., Moran Y., Cologna C.T., Peigneur S., Madio B., Praher D., Quinton L., de Pauw E., Bicudo J.E., Tytgat J., et al. BcsTx3 is a founder of a novel sea anemone toxin family of potassium channel blocker. FEBS J. 2013;280:4839–4852. doi: 10.1111/febs.12456. [DOI] [PubMed] [Google Scholar]
- 73.Chi V., Pennington M.W., Norton R.S., Tarcha E.J., Londono L.M., Sims-Fahey B., Upadhyay S.K., Lakey J.T., Iadonato S., Wulff H. Development of a sea anemone toxin as an immunomodulator for therapy of autoimmune diseases. Toxicon. 2012;59:529–546. doi: 10.1016/j.toxicon.2011.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Upadhyay S.K., Eckel-Mahan K.L., Mirbolooki M.R., Tjong I., Griffey S.M., Schmunk G., Koehne A., Halbout B., Iadonato S., Pedersen B., et al. Selective Kv1.3 channel blocker as therapeutic for obesity and insulin resistance. Proc. Natl. Acad. Sci. USA. 2013;110:E2239–E2248. doi: 10.1073/pnas.1221206110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jensen J.E., Cristofori-Armstrong B., Anangi R., Rosengren K.J., Lau C.H., Mobli M., Brust A., Alewood P.F., King G.F., Rash L.D. Understanding the molecular basis of toxin promiscuity: The analgesic sea anemone peptide APETx2 interacts with acid-sensing ion channel 3 and hERG channels via overlapping pharmacophores. J. Med. Chem. 2014;57:9195–9203. doi: 10.1021/jm501400p. [DOI] [PubMed] [Google Scholar]
- 76.Peigneur S., Billen B., Derua R., Waelkens E., Debaveye S., Beress L., Tytgat J. A bifunctional sea anemone peptide with Kunitz type protease and potassium channel inhibiting properties. Biochem. Pharmacol. 2011;82:81–90. doi: 10.1016/j.bcp.2011.03.023. [DOI] [PubMed] [Google Scholar]
- 77.Gladkikh I., Monastyrnaya M., Leychenko E., Zelepuga E., Chausova V., Isaeva M., Anastyuk S., Andreev Y., Peigneur S., Tytgat J., Kozlovkaya E. Atypical reactive center Kunitz-type inhibitor from the sea anemone Heteractis crispa. Mar. Drugs. 2012;10:1545–1565. doi: 10.3390/md10071545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Minagawa S., Ishida M., Shimakura K., Nagashima Y., Shiomi K. Isolation and amino acid sequences of two Kunitz-type protease inhibitors from the sea anemone Anthopleura aff. xanthogrammica. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1997;118:381–386. doi: 10.1016/S0305-0491(97)00174-0. [DOI] [PubMed] [Google Scholar]
- 79.Delfin J., Martinez I., Antuch W., Morera V., Gonzalez Y., Rodriguez R., Marquez M., Saroyan A., Larionova N., Diaz J. Purification, characterization and immobilization of proteinase inhibitors from Stichodactyla helianthus. Toxicon. 1996;34:1367–1376. doi: 10.1016/S0041-0101(96)00114-6. [DOI] [PubMed] [Google Scholar]
- 80.Jouiaei M., Sunagar K., Federman Gross A., Scheib H., Alewood P.F., Moran Y., Fry B.G. Evolution of an ancient venom: Recognition of a novel family of cnidarian toxins and the common evolutionary origin of sodium and potassium neurotoxins in sea anemone. Mol. Biol. Evol. 2015;32:1598–1610. doi: 10.1093/molbev/msv050. [DOI] [PubMed] [Google Scholar]
- 81.Osmakov D.I., Kozlov S.A., Andreev Y.A., Koshelev S.G., Sanamyan N.P., Sanamyan K.E., Dyachenko I.A., Bondarenko D.A., Murashev A.N., Mineev K.S., et al. Sea anemone peptide with uncommon β-hairpin structure inhibits acid-sensing ion channel 3 (ASIC3) and reveals analgesic activity. J. Biol. Chem. 2013;288:23116–23127. doi: 10.1074/jbc.M113.485516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rodríguez A.A., Salceda E., Garateix A.G., Zaharenko A.J., Peigneur S., López O., Pons T., Richardson M., Díaz M., Hernández Y. A novel sea anemone peptide that inhibits acid-sensing ion channels. Peptides. 2014;53:3–12. doi: 10.1016/j.peptides.2013.06.003. [DOI] [PubMed] [Google Scholar]
- 83.Andreev Y.A., Kozlov S.A., Koshelev S.G., Ivanova E.A., Monastyrnaya M.M., Kozlovskaya E.P., Grishin E.V. Analgesic compound from sea anemone Heteractis crispa is the first polypeptide inhibitor of vanilloid receptor 1 (TRPV1) J. Biol. Chem. 2008;283:23914–23921. doi: 10.1074/jbc.M800776200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kozlov S.A., Andreev Ya.A., Murashev A.N., Skobtsov D.I., D’Iachenko I.A., Grishin E.V. New polypeptide components from the Heteractis crispa sea anemone with analgesic activity. Russ. J. Bioorg. Chem. 2009;35:711–719. doi: 10.1134/s1068162009060065. [DOI] [PubMed] [Google Scholar]
- 85.Sher D., Zlotkin E. A hydra with many heads: Protein and polypeptide toxins from hydra and their biological roles. Toxicon. 2009;54:1148–1161. doi: 10.1016/j.toxicon.2009.02.036. [DOI] [PubMed] [Google Scholar]
- 86.Novak V., Sket D., Cankar G., Lebez D. Partial purification of a toxin from tentacles of the sea anemone Anemonia sulcata. Toxicon. 1973;11:411–417. doi: 10.1016/0041-0101(73)90116-5. [DOI] [PubMed] [Google Scholar]
- 87.Mathias A., Ross D., Schachter M. The distribution of 5-hydroxytryptamine, tetramethylammonium, homarine, and other substances in sea anemones. J. Physiol. 1960;151:296–311. doi: 10.1113/jphysiol.1960.sp006439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zaharenko A.J., Picolo G., Ferreira W.A., Jr., Murakami T., Kazuma K., Hashimoto M., Cury Y., de Freitas J.C., Satake M., Konno K. Bunodosine 391: An analgesic acylamino acid from the venom of the sea anemone Bunodosoma cangicum. J. Nat. Prod. 2011;74:378–382. doi: 10.1021/np100738m. [DOI] [PubMed] [Google Scholar]
- 89.Cooper R.A., de Freitas J., Porreca F., Eisenhour C.M., Lukas R., Huxtable R.J. The sea anemone purine, caissarone: Adenosine receptor antagonism. Toxicon. 1995;33:1025–1031. doi: 10.1016/0041-0101(95)00047-P. [DOI] [PubMed] [Google Scholar]
- 90.Fry B.G., Roelants K., Champagne D.E., Scheib H., Tyndall J.D., King G.F., Nevalainen T.J., Norman J.A., Lewis R.J., Norton R.S., et al. The toxicogenomic multiverse: Convergent recruitment of proteins into animal venoms. Annu. Rev. Genomics Hum. Genet. 2009;10:483–511. doi: 10.1146/annurev.genom.9.081307.164356. [DOI] [PubMed] [Google Scholar]
- 91.Low D.H., Sunagar K., Undheim E.A., Ali S.A., Alagon A.C., Ruder T., Jackson T.N., Pineda Gonzalez S., King G.F., Jones A., et al. Dracula’s children: Molecular evolution of vampire bat venom. J. Proteomics. 2013;89:95–111. doi: 10.1016/j.jprot.2013.05.034. [DOI] [PubMed] [Google Scholar]
- 92.Gilquin B., Racape J., Wrisch A., Visan V., Lecoq A., Grissmer S., Menez A., Gasparini S. Structure of the BgK-Kv1.1 complex based on distance restraints identified by double mutant cycles. Molecular basis for convergent evolution of Kv1 channel blockers. J. Biol. Chem. 2002;277:37406–37413. doi: 10.1074/jbc.M206205200. [DOI] [PubMed] [Google Scholar]
- 93.Talvinen K.A., Nevalainen T.J. Cloning of a novel phospholipase A2 from the cnidarian Adamsia carciniopados. Comp. Biochem. Physiol..B Biochem. Mol. Biol. 2002;132:571–578. doi: 10.1016/S1096-4959(02)00073-8. [DOI] [PubMed] [Google Scholar]
- 94.Six D.A., Dennis E.A. The expanding superfamily of phospholipase A2 sub enzymes: Classification and characterization. Biochim. Biophys. Acta. 2000;1488:1–19. doi: 10.1016/S1388-1981(00)00105-0. [DOI] [PubMed] [Google Scholar]
- 95.Undheim E.A., King G.F. On the venom system of centipedes (Chilopoda), a neglected group of venomous animals. Toxicon. 2011;57:512–524. doi: 10.1016/j.toxicon.2011.01.004. [DOI] [PubMed] [Google Scholar]
- 96.Fry B.G., Vidal N., Norman J.A., Vonk F.J., Scheib H., Ramjan S.F., Kuruppu S., Fung K., Hedges S.B., Richardson M.K., et al. Early evolution of the venom system in lizards and snakes. Nature. 2006;439:584–588. doi: 10.1038/nature04328. [DOI] [PubMed] [Google Scholar]
- 97.Fry B.G., Wuster W. Assembling an arsenal: Origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences. Mol. Biol. Evol. 2004;21:870–883. doi: 10.1093/molbev/msh091. [DOI] [PubMed] [Google Scholar]
- 98.Nevalainen T.J. Phospholipases A 2 in the genome of the sea anemone Nematostella vectensis. Comp. Biochem. Physiol. D Genomics Proteomics. 2008;3:226–233. doi: 10.1016/j.cbd.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 99.Undheim E.A., Jones A., Clauser K.R., Holland J.W., Pineda S.S., King G.F., Fry B.G. Clawing through evolution: Toxin diversification and convergence in the ancient lineage Chilopoda (Centipedes) J. Mol. Biol. 2014;31:2124–2148. doi: 10.1093/molbev/msu162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Fox J.W., Serrano S.M. Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon. 2005;45:969–985. doi: 10.1016/j.toxicon.2005.02.012. [DOI] [PubMed] [Google Scholar]
- 101.Parker M.W., Feil S.C. Pore-forming protein toxins: From structure to function. Prog. Biophys. Mol. Biol. 2005;88:91–142. doi: 10.1016/j.pbiomolbio.2004.01.009. [DOI] [PubMed] [Google Scholar]
- 102.Garcia-Ortega L., Alegre-Cebollada J., Garcia-Linares S., Bruix M., Martinez-Del-Pozo A., Gavilanes J.G. The behavior of sea anemone actinoporins at the water-membrane interface. Biochim. Biophys. Acta. 2011;1808:2275–2288. doi: 10.1016/j.bbamem.2011.05.012. [DOI] [PubMed] [Google Scholar]
- 103.Kristan K.C., Viero G., Dalla Serra M., Macek P., Anderluh G. Molecular mechanism of pore formation by actinoporins. Toxicon. 2009;54:1125–1134. doi: 10.1016/j.toxicon.2009.02.026. [DOI] [PubMed] [Google Scholar]
- 104.Bakrac B., Gutierrez-Aguirre I., Podlesek Z., Sonnen A.F., Gilbert R.J., Macek P., Lakey J.H., Anderluh G. Molecular determinants of sphingomyelin specificity of a eukaryotic pore-forming toxin. J. Biol. Chem. 2008;283:18665–18677. doi: 10.1074/jbc.M708747200. [DOI] [PubMed] [Google Scholar]
- 105.Martínez D., Otero A., Alvarez C., Pazos F., Tejuca M., Eliana Lanio M., Gutiérrez-Aguirre I., Barlic A., Iloro I., Luis Arrondo J. Effect of sphingomyelin and cholesterol on the interaction of St II with lipidic interfaces. Toxicon. 2007;49:68–81. doi: 10.1016/j.toxicon.2006.09.019. [DOI] [PubMed] [Google Scholar]
- 106.Mechaly A.E., Bellomio A., Gil-Cartón D., Morante K., Valle M., González-Mañas J.M., Guérin D. Structural insights into the oligomerization and architecture of eukaryotic membrane pore-forming toxins. Structure. 2011;19:181–191. doi: 10.1016/j.str.2010.11.013. [DOI] [PubMed] [Google Scholar]
- 107.Drechsler A., Potrich C., Sabo J.K., Frisanco M., Guella G., Dalla Serra M., Anderluh G., Separovic F., Norton R.S. Structure and activity of the N-terminal region of the eukaryotic cytolysin equinatoxin II. Biochemistry. 2006;45:1818–1828. doi: 10.1021/bi052166o. [DOI] [PubMed] [Google Scholar]
- 108.Gutierrez-Aguirre I., Barlic A., Podlesek Z., Macek P., Anderluh G., Gonzalez-Manas J.M. Membrane insertion of the N-terminal α-helix of equinatoxin II, a sea anemone cytolytic toxin. Biochem. J. 2004;384(Pt 2):421–428. doi: 10.1042/BJ20040601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Mancheno J.M., Martin-Benito J., Martinez-Ripoll M., Gavilanes J.G., Hermoso J.A. Crystal and electron microscopy structures of sticholysin II actinoporin reveal insights into the mechanism of membrane pore formation. Structure. 2003;11:1319–1328. doi: 10.1016/j.str.2003.09.019. [DOI] [PubMed] [Google Scholar]
- 110.Tejuca M., Dalla Serra M., Potrich C., Alvarez C., Menestrina G. Sizing the radius of the pore formed in erythrocytes and lipid vesicles by the toxin sticholysin I from the sea anemone Stichodactyla helianthus. J. Membr. Biol. 2001;183:125–135. doi: 10.1007/s00232-001-0060-y. [DOI] [PubMed] [Google Scholar]
- 111.Chung J.J., Ratnapala L.A., Cooke I.M., Yanagihara A.A. Partial purification and characterization of a hemolysin (CAH1) from Hawaiian box jellyfish Carybdea alata venom. Toxicon. 2001;39:981–990. doi: 10.1016/S0041-0101(00)00237-3. [DOI] [PubMed] [Google Scholar]
- 112.Nagai H., Takuwa K., Nakao M., Sakamoto B., Crow G.L., Nakajima T. Isolation and characterization of a novel protein toxin from the Hawaiian box jellyfish (sea wasp) Carybdea alata. Biochem. Biophys. Res. Commun. 2000;275:589–594. doi: 10.1006/bbrc.2000.3352. [DOI] [PubMed] [Google Scholar]
- 113.Brinkman D., Burnell J. Identification, cloning and sequencing of two major venom proteins from the box jellyfish, Chironex fleckeri. Toxicon. 2007;50:850–860. doi: 10.1016/j.toxicon.2007.06.016. [DOI] [PubMed] [Google Scholar]
- 114.Voskoboinik I., Dunstone M.A., Baran K., Whisstock J.C., Trapani J.A. Perforin: structure, function, and role in human immunopathology. Immunol. Rev. 2010;235:35–54. doi: 10.1111/j.0105-2896.2010.00896.x. [DOI] [PubMed] [Google Scholar]
- 115.Castaneda O., Harvey A.L. Discovery and characterization of cnidarian peptide toxins that affect neuronal potassium ion channels. Toxicon. 2009;54:1119–1124. doi: 10.1016/j.toxicon.2009.02.032. [DOI] [PubMed] [Google Scholar]
- 116.Catterall W.A. Neurotoxins that act on voltage-sensitive sodium channels in excitable membranes. Annu. Rev. Pharmacol. Toxicol. 1980;20:15–43. doi: 10.1146/annurev.pa.20.040180.000311. [DOI] [PubMed] [Google Scholar]
- 117.Catterall W.A., Cestèle S., Yarov-Yarovoy V., Yu F.H., Konoki K., Scheuer T. Voltage-gated ion channels and gating modifier toxins. Toxicon. 2007;49:124–141. doi: 10.1016/j.toxicon.2006.09.022. [DOI] [PubMed] [Google Scholar]
- 118.Moran Y., Gordon D., Gurevitz M. Sea anemone toxins affecting voltage-gated sodium channels-molecular and evolutionary features. Toxicon. 2009;54:1089–1101. doi: 10.1016/j.toxicon.2009.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Loret E.P., del Valle R.M., Mansuelle P., Sampieri F., Rochat H. Positively charged amino acid residues located similarly in sea anemone and scorpion toxins. J. Biol. Chem. 1994;269:16785–16788. [PubMed] [Google Scholar]
- 120.Cariello L., de Santis A., Fiore F., Piccoli R., Spagnuolo A., Zanetti L., Parente A. Na+ channel toxinsCalitoxin, a neurotoxic peptide from the sea anemone Calliactis parasitica: amino-acid sequence and electrophysiological properties. Biochemistry. 1989;28:2484–2489. doi: 10.1021/bi00432a020. [DOI] [PubMed] [Google Scholar]
- 121.Nir N., Eli S., Daniel S., Yehu M., Liora T., Ana L.T.-M., Michal H., Binyamin H., Eliahu Z. AdE-1, a new inotropic Na+ channel toxin from Aiptasia diaphana, is similar to, yet distinct from, known anemone Na+ channel toxins. Biochem. J. 2013;451:81–90. doi: 10.1042/BJ20121623. [DOI] [PubMed] [Google Scholar]
- 122.Moran Y., Kahn R., Cohen L., Gur M., Karbat I., Gordon D., Gurevitz M. Molecular analysis of the sea anemone toxin Av3 reveals selectivity to insects and demonstrates the heterogeneity of receptor site-3 on voltage-gated Na+ channels. Biochem. J. 2007;406:41–48. doi: 10.1042/BJ20070233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Bruhn T. Kalicludines and Kaliseptine. J. Biol. Chem. 1995;270:25121–25126. doi: 10.1074/jbc.270.42.25121. [DOI] [PubMed] [Google Scholar]
- 124.Honma T., Kawahata S., Ishida M., Nagai H., Nagashima Y., Shiomi K. Novel peptide toxins from the sea anemone Stichodactyla haddoni. Peptides. 2008;29:536–544. doi: 10.1016/j.peptides.2007.12.010. [DOI] [PubMed] [Google Scholar]
- 125.Burnett J.W. Treatment of Atlantic cnidarian envenomations. Toxicon. 2009;54:1201–1205. doi: 10.1016/j.toxicon.2009.02.020. [DOI] [PubMed] [Google Scholar]
- 126.Morabito R., La Spada G., Crupi R., Esposito E., Marino A. Crude Venom from Nematocysts of the Jellyfish Pelagia noctiluca as a Tool to Study Cell Physiology. Cent. Nerv. Syst. Agents. Med. Chem. 2015;15:68–73. doi: 10.2174/1871524914666141028150212. [DOI] [PubMed] [Google Scholar]
- 127.Wu C.H. Palytoxin: Membrane mechanisms of action. Toxicon. 2009;54:1183–1189. doi: 10.1016/j.toxicon.2009.02.030. [DOI] [PubMed] [Google Scholar]
- 128.Habermann E. Palytoxin acts through Na+, K+-ATPase. Toxicon. 1989;27:1171–1187. doi: 10.1016/0041-0101(89)90026-3. [DOI] [PubMed] [Google Scholar]
- 129.Gorogh T., Beress L., Quabius E.S., Ambrosch P., Hoffmann M. Head and neck cancer cells and xenografts are very sensitive to palytoxin: Decrease of C-jun N-terminale kinase-3 expression enhances palytoxin toxicity. Mol. Cancer. 2013;12:12. doi: 10.1186/1476-4598-12-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Quinn R.J., Kashiwagi M., Moore R.E., Norton T.R. Anticancer activity of zoanthids and the associated toxin, palytoxin, against Ehrlich ascites tumor and P-388 lymphocytic leukemia in mice. J. Pharm. Sci. 1974;63:257–260. doi: 10.1002/jps.2600630217. [DOI] [PubMed] [Google Scholar]
- 131.Louzao M.C., Ares I.R., Cagide E., Espiña B., Vilariño N., Alfonso A., Vieytes M.R., Botana L.M. Palytoxins and cytoskeleton: An overview. Toxicon. 2011;57:460–469. doi: 10.1016/j.toxicon.2010.09.017. [DOI] [PubMed] [Google Scholar]
- 132.Wattenberg E.V. Modulation of protein kinase signaling cascades by palytoxin. Toxicon. 2011;57:440–448. doi: 10.1016/j.toxicon.2010.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Giraldi T., Ferlan I., Romeo D. Antitumour Activity of Equinatoxin. Chem.-Biol. Interact. 1976;13:199–203. doi: 10.1016/0009-2797(76)90073-9. [DOI] [PubMed] [Google Scholar]
- 134.Batista U., Macek P., Sedmak B. The cytotoxic and cytolytic activity of equinatoxin II from the sea anemone Actinia equina. Cell Biol. Int. Rep. 1990;14:1013–1024. doi: 10.1016/0309-1651(90)90113-D. [DOI] [PubMed] [Google Scholar]
- 135.Lenarcic B., Turk V. Thyroglobulin type-1 domains in equistatin inhibit both papain-like cysteine proteinases and cathepsin D. J. Biol. Chem. 1999;274:563–566. doi: 10.1074/jbc.274.2.563. [DOI] [PubMed] [Google Scholar]
- 136.Liaudet-Coopman E., Beaujouin M., Derocq D., Garcia M., Glondu-Lassis M., Laurent-Matha V., Prébois C., Rochefort H., Vignon F. Cathepsin D: Newly discovered functions of a long-standing aspartic protease in cancer and apoptosis. Cancer Lett. 2006;237:167–179. doi: 10.1016/j.canlet.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 137.Brömme D., Petanceska S. Role of Proteases in the Pathophysiology of Neurodegenerative Diseases. Springer; New York, NY, USA: 2002. Papain-like cysteine proteases and their implications in neurodegenerative diseases; pp. 47–61. [Google Scholar]
- 138.Ayed Y., Dellai A., Ben Mansour H., Bacha H., Abid S. Analgesic and antibutyrylcholinestrasic activities of the venom prepared from the Mediterranean jellyfish Pelagia noctiluca. Ann. Clin. Microbiol. Antimicrob. 2012;11:15. doi: 10.1186/1476-0711-11-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Hughes R.J., Angus J.A., Winkel K.D., Wright C.E. A pharmacological investigation of the venom extract of the Australian box jellyfish, Chironex fleckeri, in cardiac and vascular tissues. Toxicol. Lett. 2012;209:11–20. doi: 10.1016/j.toxlet.2011.11.025. [DOI] [PubMed] [Google Scholar]
- 140.Tibballs J., Yanagihara A.A., Turner H.C., Winkel K. Immunological and toxinological responses to jellyfish stings. Inflamm. Allergy Drug Targets. 2011;10:438–446. doi: 10.2174/187152811797200650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Bruschetta G., Impellizzeri D., Morabito R., Marino A., Ahmad A., Spano N., Spada G.L., Cuzzocrea S., Esposito E. Pelagia noctiluca (Scyphozoa) crude venom injection elicits oxidative stress and inflammatory response in rats. Mar. Drugs. 2014;12:2182–2204. doi: 10.3390/md12042182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Morabito R., Marino A., Dossena S., La Spada G. Nematocyst discharge in Pelagia noctiluca (Cnidaria, Scyphozoa) oral arms can be affected by lidocaine, ethanol, ammonia and acetic acid. Toxicon. 2014;83:52–58. doi: 10.1016/j.toxicon.2014.03.002. [DOI] [PubMed] [Google Scholar]
- 143.Birsa L.M., Verity P.G., Lee R.F. Evaluation of the effects of various chemicals on discharge of and pain caused by jellyfish nematocysts. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010;151:426–430. doi: 10.1016/j.cbpc.2010.01.007. [DOI] [PubMed] [Google Scholar]
- 144.Rifkin J.F., Fenner P.J., Williamson J.A. First aid treatment of the sting from the hydroid Lytocarpus philippinus: The structure of, and in vitro discharge experiments with its nematocysts. J. Wilderness Med. 1993;4:252–260. doi: 10.1580/0953-9859-4.3.252. [DOI] [Google Scholar]
- 145.Soon W.W., Hariharan M., Snyder M.P. High-throughput sequencing for biology and medicine. Mol. Syst. Biol. 2013;9:640. doi: 10.1038/msb.2012.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Escoubas P., King G.F. Venomics as a drug discovery platform. Expert Rev. Proteomics. 2009;6:221–224. doi: 10.1586/epr.09.45. [DOI] [PubMed] [Google Scholar]