Abstract
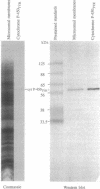
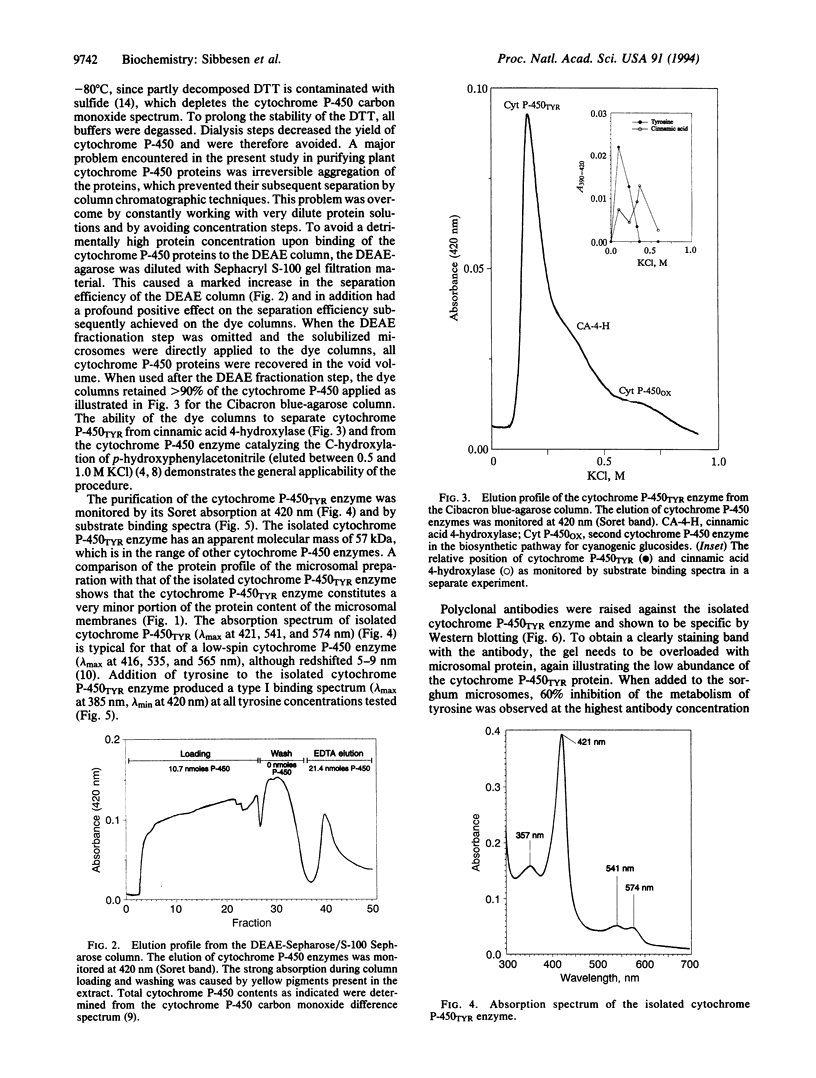
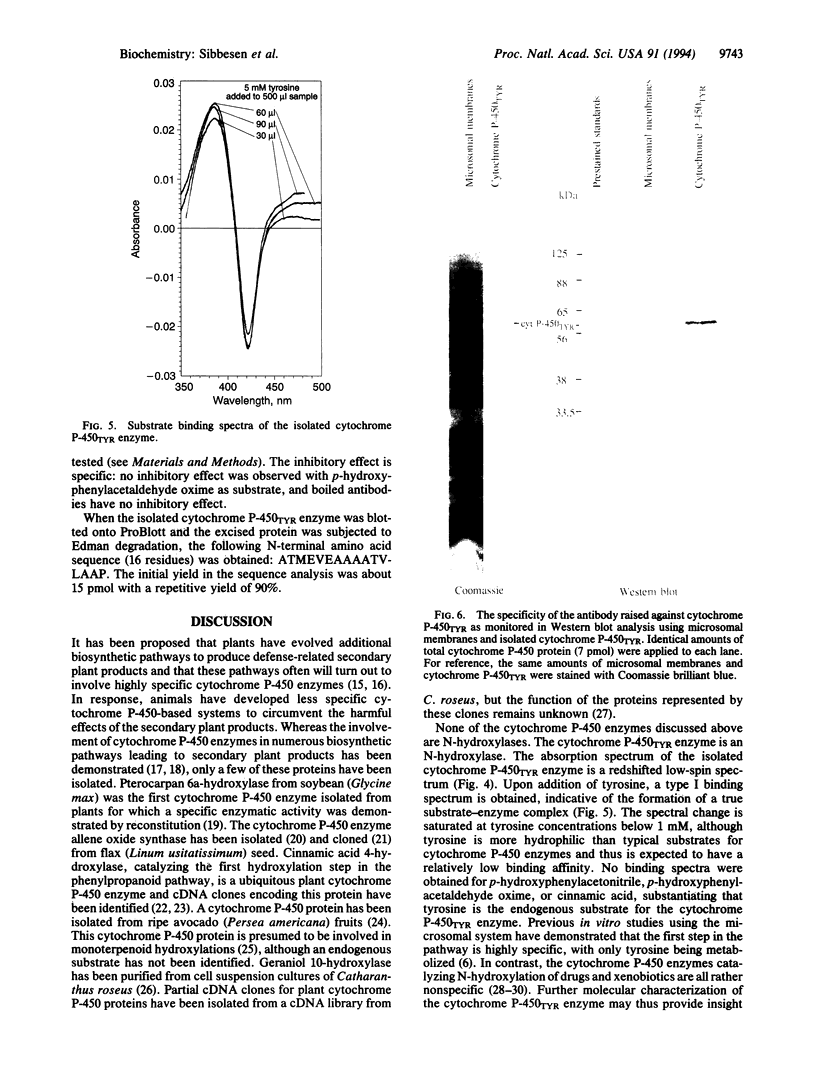
The cytochrome P-450 enzyme (hemethiolate enzyme) that catalyzes the N-hydroxylation of L-tyrosine to N-hydroxytyrosine, the committed step in the biosynthesis of the cyanogenic glucoside dhurrin, has been isolated from microsomes prepared from etiolated seedlings of Sorghum bicolor (L.) Moench. The cytochrome P-450 enzyme was solubilized with the detergents Renex 690, reduced Triton X-100, and 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate and isolated by ion-exchange (DEAE-Sepharose) and dye (Cibacron blue and reactive red 120) column chromatography. To prevent irreversible aggregation of the cytochrome P-450 enzyme, the isolation procedure was designed without any concentration step--i.e., with dilution of the ion-exchange gel with gel filtration material. The isolated enzyme, which we designate the cytochrome P-450TYR enzyme, gives rise to the specific formation of a type I substrate binding spectrum in the presence of L-tyrosine. The microsomal preparation contains 0.2 nmol of total cytochrome P-450/mg of protein. The cytochrome P-450TYR enzyme is estimated to constitute approximately 20% of the total cytochrome P-450 content of the microsomal membranes and about 0.2% of their total protein content. The apparent molecular mass of the cytochrome P-450TYR enzyme is 57 kDa, and the N-terminal amino acid sequence is ATMEVEAAAATVLAAP. A polyclonal antibody raised against the isolated cytochrome P-450TYR enzyme is specific as monitored by Western blot analysis and inhibits the in vitro conversion of L-tyrosine to p-hydroxymandelonitrile catalyzed by the microsomal system. The cytochrome P-450TYR enzyme exhibits high substrate specificity and acts as an N-hydroxylase on a single endogenous substrate. The reported isolation procedure based on dye columns constitutes a gentle isolation method for cytochrome P-450 enzymes and is of general use as indicated by its ability to separate cytochrome P-450TYR from the cytochrome P-450 enzyme catalyzing the C-hydroxylation of p-hydroxyphenylacetonitrile and from cinnamic acid 4-hydroxylase.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akazawa T., Miljanich P., Conn E. E. Studies on Cyanogenic Glycoside of Sorghum Vulgare. Plant Physiol. 1960 Jul;35(4):535–538. doi: 10.1104/pp.35.4.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Clement B., Jung F., Pfunder H. N-hydroxylation of benzamidine to benzamidoxime by a reconstituted cytochrome P-450 oxidase system from rabbit liver: involvement of cytochrome P-450 IIC3. Mol Pharmacol. 1993 Mar;43(3):335–342. [PubMed] [Google Scholar]
- Donaldson R. P., Luster D. G. Multiple forms of plant cytochromes p-450. Plant Physiol. 1991 Jul;96(3):669–674. doi: 10.1104/pp.96.3.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleming C. M., Branch R. A., Wilkinson G. R., Guengerich F. P. Human liver microsomal N-hydroxylation of dapsone by cytochrome P-4503A4. Mol Pharmacol. 1992 May;41(5):975–980. [PubMed] [Google Scholar]
- Fling S. P., Gregerson D. S. Peptide and protein molecular weight determination by electrophoresis using a high-molarity tris buffer system without urea. Anal Biochem. 1986 May 15;155(1):83–88. doi: 10.1016/0003-2697(86)90228-9. [DOI] [PubMed] [Google Scholar]
- Halkier B. A., Lykkesfeldt J., Møller B. L. 2-nitro-3-(p-hydroxyphenyl)propionate and aci-1-nitro-2-(p-hydroxyphenyl)ethane, two intermediates in the biosynthesis of the cyanogenic glucoside dhurrin in Sorghum bicolor (L.) Moench. Proc Natl Acad Sci U S A. 1991 Jan 15;88(2):487–491. doi: 10.1073/pnas.88.2.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halkier B. A., Møller B. L. Biosynthesis of the Cyanogenic Glucoside Dhurrin in Seedlings of Sorghum bicolor (L.) Moench and Partial Purification of the Enzyme System Involved. Plant Physiol. 1989 Aug;90(4):1552–1559. doi: 10.1104/pp.90.4.1552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halkier B. A., Møller B. L. Involvement of Cytochrome P-450 in the Biosynthesis of Dhurrin in Sorghum bicolor (L.) Moench. Plant Physiol. 1991 May;96(1):10–17. doi: 10.1104/pp.96.1.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halkier B. A., Olsen C. E., Møller B. L. The biosynthesis of cyanogenic glucosides in higher plants. The (E)- and (Z)-isomers of p-hydroxyphenylacetaldehyde oxime as intermediates in the biosynthesis of dhurrin in Sorghum bicolor (L.) Moench. J Biol Chem. 1989 Nov 25;264(33):19487–19494. [PubMed] [Google Scholar]
- Hallahan D. L., Nugent J. H., Hallahan B. J., Dawson G. W., Smiley D. W., West J. M., Wallsgrove R. M. Interactions of Avocado (Persea americana) Cytochrome P-450 with Monoterpenoids. Plant Physiol. 1992 Apr;98(4):1290–1297. doi: 10.1104/pp.98.4.1290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Høj P. B., Møller B. L. Acid-labile sulfide and zero-valence sulfur in plant extracts containing chlorophyll and ionic detergents. Anal Biochem. 1987 Aug 1;164(2):307–314. doi: 10.1016/0003-2697(87)90498-2. [DOI] [PubMed] [Google Scholar]
- Jefcoate C. R. Measurement of substrate and inhibitor binding to microsomal cytochrome P-450 by optical-difference spectroscopy. Methods Enzymol. 1978;52:258–279. doi: 10.1016/s0076-6879(78)52029-6. [DOI] [PubMed] [Google Scholar]
- Kochs G., Grisebach H. Phytoalexin synthesis in soybean: purification and reconstitution of cytochrome P450 3,9-dihydroxypterocarpan 6a-hydroxylase and separation from cytochrome P450 cinnamate 4-hydroxylase. Arch Biochem Biophys. 1989 Sep;273(2):543–553. doi: 10.1016/0003-9861(89)90514-6. [DOI] [PubMed] [Google Scholar]
- MacFarlane I. J., Lees E. M., Conn E. E. The in vitro biosynthesis of dhurrin, the cyanogenic glycoside of Sorghum bicolor. J Biol Chem. 1975 Jun 25;250(12):4708–4713. [PubMed] [Google Scholar]
- Meijer A. H., Souer E., Verpoorte R., Hoge J. H. Isolation of cytochrome P-450 cDNA clones from the higher plant Catharanthus roseus by a PCR strategy. Plant Mol Biol. 1993 May;22(2):379–383. doi: 10.1007/BF00014944. [DOI] [PubMed] [Google Scholar]
- Miura S., Degawa M., Hashimoto Y. Renal and hepatic microsomal enzymes responsible for bioactivation of 3-methoxy-4-aminoazobenzene in the rodent. Biochem Pharmacol. 1991 Oct 9;42(9):1655–1659. doi: 10.1016/0006-2952(91)90499-u. [DOI] [PubMed] [Google Scholar]
- Møller B. L., Conn E. E. The biosynthesis of cyanogenic glucosides in higher plants. N-Hydroxytyrosine as an intermediate in the biosynthesis of dhurrin by Sorghum bicolor (Linn) Moench. J Biol Chem. 1979 Sep 10;254(17):8575–8583. [PubMed] [Google Scholar]
- Nebert D. W., Gonzalez F. J. P450 genes: structure, evolution, and regulation. Annu Rev Biochem. 1987;56:945–993. doi: 10.1146/annurev.bi.56.070187.004501. [DOI] [PubMed] [Google Scholar]
- O'keefe D. P., Leto K. J. Cytochrome P-450 from the Mesocarp of Avocado (Persea americana). Plant Physiol. 1989 Apr;89(4):1141–1149. doi: 10.1104/pp.89.4.1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OMURA T., SATO R. THE CARBON MONOXIDE-BINDING PIGMENT OF LIVER MICROSOMES. I. EVIDENCE FOR ITS HEMOPROTEIN NATURE. J Biol Chem. 1964 Jul;239:2370–2378. [PubMed] [Google Scholar]
- Potts J. R., Weklych R., Conn E. E., Rowell J. The 4-hydroxylation of cinnamic acid by sorghum microsomes and the requirement for cytochrome P-450. J Biol Chem. 1974 Aug 25;249(16):5019–5026. [PubMed] [Google Scholar]
- Song W. C., Brash A. R. Purification of an allene oxide synthase and identification of the enzyme as a cytochrome P-450. Science. 1991 Aug 16;253(5021):781–784. doi: 10.1126/science.1876834. [DOI] [PubMed] [Google Scholar]
- Song W. C., Funk C. D., Brash A. R. Molecular cloning of an allene oxide synthase: a cytochrome P450 specialized for the metabolism of fatty acid hydroperoxides. Proc Natl Acad Sci U S A. 1993 Sep 15;90(18):8519–8523. doi: 10.1073/pnas.90.18.8519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teutsch H. G., Hasenfratz M. P., Lesot A., Stoltz C., Garnier J. M., Jeltsch J. M., Durst F., Werck-Reichhart D. Isolation and sequence of a cDNA encoding the Jerusalem artichoke cinnamate 4-hydroxylase, a major plant cytochrome P450 involved in the general phenylpropanoid pathway. Proc Natl Acad Sci U S A. 1993 May 1;90(9):4102–4106. doi: 10.1073/pnas.90.9.4102. [DOI] [PMC free article] [PubMed] [Google Scholar]