Abstract
Background
Urinary tract infections (UTI) are common in renal transplant recipients. Trimethoprim/Sulfamethoxazole (TMP/SMZ) in moderate to high daily doses prevents Pneumocystis jiroveci (PCP) and reduces the risk of UTI in renal transplant patients. Low dose TMP/SMZ also reduces the risk of PCP, though its ability to reduce the risk of UTI is uncertain.
Design
Retrospective review of 158 patients who received a renal transplant without corticosteroids for maintenance immunosuppression.
Results
Forty percent of patients initially prescribed TMP/SMZ ultimately stopped this medication early due to an adverse reaction. Urinary infection occurred in 16%, without a significant difference in the risk of UTI between those treated with dapsone versus those treated with TMP/SMZ (HR[95%CI]: 1.7 [0.75, 3.9], p=0.2). In the subset of patients who were older than age 47 years, (mean age for this cohort, SD +/−6.2 years) those treated with dapsone originally or who switched from TMP/SMZ to dapsone had a greater risk of UTI compared to patients who remained on TMP/SMZ (HR[95%CI]: 4.3 [1.2, 15.5], p=0.024).
Conclusions
For renal transplant recipients over the age of 47 years, treated without long-term glucocorticoids, our retrospective data suggest low dose TMP/SMZ is associated with a lower risk of UTI compared to dapsone prophylaxis.
Background
Infections of the urinary tract are the most common type of infection affecting renal transplant recipients (1). Urinary tract infections (UTI) occur in at least a quarter of patients after renal transplantation, a rate that far exceeds that of the general population (2,3). The use of immunosuppression medications and ureteral stents along with ureteral reflux and altered bladder function may account for this increased risk (4,5,6,7).
The impact of UTI in this population is substantial, increasing both the risk of graft failure and death (8,9). In a previous study from our institution, the development of a UTI after renal transplantation was associated with a 3.5 fold increased likelihood of death compared with renal transplant recipients who did not develop a UTI (9). Other groups have linked the incidence of UTI with higher rates of acute rejection and allograft loss (8,10,11).
Trimethoprim/sulfamethoxazole (TMP/SMZ) has routinely been used as first-line prophylaxis against Pneumocystis jiroveci pneumonia (PCP, previously known as Pneumocystis carinii) in immunocompromised hosts (12). In the renal transplant population, high-dose TMP/SMZ, (one double-strength tablet twice daily, 320mg/1600mg total dose) has been shown to reduce UTI rates (13). The effect of lower doses of TMP/SMZ on UTI suppression remains less clear. Studies evaluating the benefits of medium-dose TMP/SMZ (one double strength tablet daily, 160mg/800mg) have been mixed (14,15), and no trial has shown low dose TMP/SMZ (80mg/400mg) to lower the risk of UTI.
Historical infection rates in renal transplant recipients mostly reflect data from patients receiving a typical three-drug immunosuppression regimen (for example, the combination of a calcineurin inhibitor, an anti-proliferative agent and a corticosteroid). Since the early era of organ transplantation, the use of corticosteroids has been a mainstay, but this trend is changing as more patients are treated with early withdrawal of steroids or without steroids altogether (16). In 2006, a meta-analysis by Tan et al. found significantly lower rates of UTI in patients treated with a steroid-withdrawal regimen compared to those who remained on steroids, with a relative risk reduction of 20% (17).
In light of the reduced risk of UTI development in patients not prescribed long-term corticosteroids, we sought to describe the risk of UTI in renal transplant patients treated with low-dose TMP/SMZ compared to those treated with dapsone prophylaxis. We also aimed to describe the reasons for the discontinuation of TMP/SMZ therapy or selection of dapsone as initial prophylaxis.
Methods
Using a historical cohort, we reviewed the medical records at our two university-affiliated hospitals. Patients were included in this analysis if they received a kidney or simultaneous kidney and pancreas transplant at our institution between June 2005 and May 2007, were treated with a rapid steroid-withdrawal maintenance immunosuppression regimen, and if they received ongoing medical care in our institutions. Patients were administered thymoglobulin, alemtuzumab or basiliximab along with high-dose methylprednisolone intra-operatively and in the immediate post-operative period for induction immunosuppression. Beginning on post-operative day number three, maintenance immunosuppression consisted of the calcineurin inhibitor tacrolimus along with mycophenolate mofetil (MMF) or mycophenolic acid in all but two patients: one was prescribed cyclosporine and MMF and the other began maintenance immunosuppression with tacrolimus and MMF but was switched to rapamycin early after transplantation. Initial tacrolimus dosing was 0.10-0.15mg/kg twice daily with a goal 12-hour trough serum concentration of 10-12ng/dl for the first two post-transplant months, followed by a goal 12 hour trough level of 8-10ng/dL until the six month post transplant mark. Initial MMF dosing ranged from 500mg to 1000mg twice daily and serum concentration for MMF was not obtained. All patients underwent placement of a ureteral stent at the time of transplantation and stents were removed within the first 30 days after transplantation. In those patients who underwent a pancreas transplant, exocrine pancreatic drainage was enteric. All patients were followed for 180 days after transplantation.
All patients were prescribed once daily dosing of either low-dose TMP/SMZ (80/400mg) or dapsone (100mg). The planned duration of therapy was for the first six months after transplantation as prophylaxis against PCP. Adherence to the prescribed therapy was determined by self-report as documented in the medical record.
Patient demographic characteristics and clinical information detailing reasons for discontinuation of TMP/SMZ therapy were abstracted from the medical record. All patients underwent monthly protocol laboratory evaluations, including urinalysis and urine cultures. Additionally, patients underwent laboratory evaluation if clinically indicated. Urinary tract infection was defined as a urine culture growing more than 50,000 colony forming units (cfu) of bacteria and signs of infection on urinalysis such as five or more leukocytes per high-powered field on microscopic evaluation or by the presence of leukocyte esterase and nitrites on urine dipstick analysis.
The Vanderbilt University Medical Center Institutional Review Board and the Veterans Affairs Tennessee Valley Healthcare System Research and Development Committee approved this study as exempted research.
Statistical Analysis
We performed analyses by using STATA, version 8.0 (Stata, College Park, Texas). Patients’ characteristics were described using medians and interquartile ranges (IQRs) for continuous variables and percentages for categorical variables. For each of the two prophylactic antibiotic therapies, the total number of days used per patient were calculated and described. Comparisons of characteristics between patients with and without UTI were performed by Wilcoxon-rank sum tests or Mann-Whitney U test, as appropriate.
Patients were then categorized by the type of antibiotic prophylaxis used, either TMP/SMX or dapsone, for each day within the 180-day period. The cumulative UTI incidence was then evaluated by using nonparametric Kaplan-Meier estimates by prophylaxis category. Log-rank test was used to test for statistically significant differences between the two groups. Unadjusted and adjusted Cox proportional hazard regression models were applied to describe antibiotic prophylaxis category and its association with UTI. Survival time was calculated from date of transplantation until date of UTI diagnosis, discontinuation of all antibiotic prophylaxis, or at 180-days after transplantation. No patients were lost to follow-up and no patient death or graft loss occurred during this evaluation. Adjusted Cox proportional hazards regression analysis included demographic variables known to predict UTI in patients with a transplant, including sex and diabetes. For the Cox proportional hazards analyses, there was no evidence of collinearity among covariates, and Schoenfeld residual analyses showed the proportionality assumption was not violated (P>0.12 for all). Subgroup analysis by age, dichotomized at the mean age of the overall population, was performed, and P for interaction was determined to evaluate the evidence of effect modification.
Results
From June 2005 through May 2007, 161 patients underwent a renal transplant or simultaneous kidney and pancreas transplant and were treated with a rapid steroid withdrawal immunosuppression protocol. Data from 158 patients were available for analysis. The other three patients were cared for outside of our institutions.
One hundred nine patients (69%) were male and 61 patients (39%) had diabetes mellitus. Diabetes was the leading cause of renal failure (32%) followed by glomerulonephritis, hypertension and cystic renal disease in 19%, 17% and 13% respectively (Table 1). Nearly half the patients underwent a renal transplant from a living donor (48%) and the median exposure to prophylactic antibiotics for PCP was 180 days (IQR 144, 180). Induction immunosuppression included thymoglobulin (84%), basiliximab (11%) or alemtuzumab (5%). Initially, 91% of the patients (n=144) were prescribed TMP/SMZ (80mg/400mg), while the remaining 9% (n=14) were prescribed dapsone 100mg daily. Of those patients initially prescribed TMP/SMZ, 63 discontinued this medication prematurely (40%) due to an adverse event. Forty-seven of those patients (75%) were switched from TMP/SMZ to dapsone for ongoing infection prophylaxis. Of the patients who were initially prescribed dapsone, none switched to TMP/SMZ.
Table 1.
Baseline Characteristics
| Characteristic | |
|---|---|
| Age (median [IQR}) | 47 [37, 57] |
| Male | 69% (n=109) |
| Diabetes mellitus | 39% (n=61) |
| Etiology of ESRD | |
| Diabetes mellitus | 32% (50) |
| Cystic renal disease | 13% (21) |
| Glomerulonephritis | 19% (31) |
| Hypertension | 17% (26) |
| Unknown | 8% (13) |
| Reflux/ Congenital | 5% (8) |
| Interstitial nephritis | 3% (5) |
| CNI* toxicity | 1% (2) |
| Alport's syndrome | 0.5% (1) |
| Acute kidney injury | 0.5% (1) |
| Type of Transplant | |
| Living Donor | 48.1% (76) |
| Deceased Donor | 42.4% (67) |
| With/after Pancreas | 9.5% (15) |
| Total Days of Antibiotics | Med [IQR]: 180 [144, 180] Range:(3, 180) |
| Type of Antibiotics (n=158) | |
| TMP/SMZ** only | 61.4% (97) |
| Dapsone only | 8.7% (14) |
| Both | 29.8% (47) |
Calcineurin Inhibitor
Trimethoprim/Sulfamethoxazole
Reasons for stopping TMP/SMZ prematurely included drug hypersensitivity reaction (allergy, rash or non-infectious fever) (n=16, 25%), elevated liver function tests (n=13, 21%) and acute interstitial nephritis (AIN) (n=7, 11%). Of the seven patients with AIN, four patients had an interstitial infiltrate with eosinophils and granulomata on renal biopsy, while the other three were diagnosed and treated empirically due to the presence of elevated serum or urine eosinophil counts. All seven patients had resolution of symptoms upon cessation of TMP/SMZ (Table 2). Sixteen patients (25%) discontinued TMP/SMZ for unknown reasons. There was no difference in the likelihood of discontinuing TMP/SMZ in our study based on age, gender, diagnosis of diabetes or type of transplant received, living or deceased donor. Ten patients (16%) discontinued dapsone at least 30 days prematurely, most commonly due to persistent anemia. There was no difference in the rate of TMP/SMZ discontinuation between age groups. Thirty seven percent (n=28) in the group younger than the mean age and 27% (n=19) in the group older than the mean age discontinued this medication prematurely (p=0.19).
Table 2.
Reasons for prematurely discontinuing medication
| Adverse Event | TMP/SMZ | Dapsone |
|---|---|---|
| Total | n=63 | n=14 |
| Drug allergy or non-infectious fever | 25% (16) | |
| Unknown/Not documented | 25% (16) | 50% (7) |
| Elevated liver function tests* | 21% (13) | |
| Hematologic process** | 16% (10) | 29% (4) |
| Acute interstitial nephritis | 11% (7) | 7% (1) |
| Hyperkalemia | 4% (3) | |
| Acute Kidney Injury | 4% (1) | |
| Poor Appetite | 7% (1) | |
| Oral Ulcers | 7% (1) |
Patients were considered to have stopped antibiotics prematurely if they discontinued TMP/SMZ or dapsone more than 30 days prior to reaching the 180 day post-transplant mark.
Aspartate aminotransferas (AST) or alanine aminotransferase (ALT) greater than three times the upper limit of normal
Hematologic processes included prolonged leucopenia or anemia
Urinary tract infection occurred in 25 patients overall (16%). The median (IQR) time to UTI diagnosis was 22 (7, 94) days after transplantation. The leading causative microorganism responsible for UTI in this population was Enterococcus faecalis (n=7, 28%), followed by Escherichia coli (n=6, 24%) and Klebsiella pneumoniae (n=4, 16%). Bacteria with in vitro resistance to TMP/SMZ were responsible for 80% of the urinary infections (n=20) (Table 3).
Table 3.
Microbiology of urinary infections and antibiotic resistance
| Organism (species) | Younger* | Older** | Total | Resistant*** |
|---|---|---|---|---|
| Escherichia | 3 | 3 | 6 | 83% |
| Klebsiella | 2 | 1 | 3 | 33% |
| Enterobacter | 0 | 1 | 1 | 100% |
| Pseudomonas | 3 | 1 | 4 | 100% |
| Streptococcus | 1 | 0 | 1 | 100% |
| Enterococcus | 5 | 2 | 7 | 100% |
| Staphylococcus | 1 | 0 | 1 | 100% |
| Mixed flora | 0 | 2 | 2 | N/A |
Younger than the mean age of 47 years
Older than the mean age of 47 years
Percentage with in vitro resistance to TMP/SMZ
Overall, risk factors for developing a UTI included female gender (p=0.001) and abnormal urinary anatomy (p=0.001), defined as an atypical genito-urinary (GU) system for a renal transplant recipient (Table 4). Examples of atypical GU system included a history of ureteral reflux, neobladder formation, urostomy or similar anatomic abnormalities.
Table 4.
Characteristics associated with UTI
| Variable | UTI n=25 | No UTI n=133 | p-Value* |
|---|---|---|---|
| Age | 42.8 (15.7) | 47.5 (11.7) | 0.09 |
| Male | 40.0% | 74.4% | 0.001 |
| Diabetes | 40.0% | 38.4% | 0.88 |
| Type of Transplant | |||
| Living Donor | 56.0% | 46.6% | |
| Deceased Donor | 32.0% | 44.4% | 0.51 |
| With/after pancreas | 12.0% | 9.0% | |
| Dapsone use | 40.0% | 38.4% | 0.88 |
| Abnormal Anatomy | 50.0% | 13.0% | 0.001 |
Wilcoxon-rank sum or Mann-Whitney U test
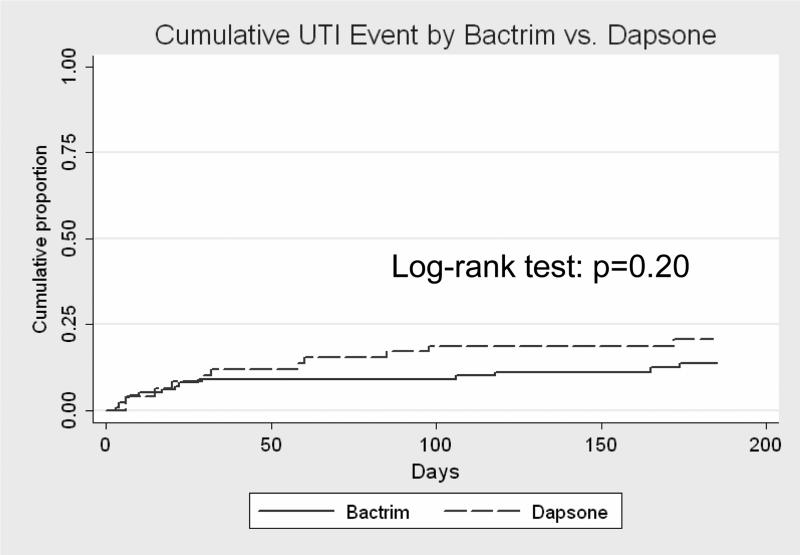
Among all patients who received TMP/SMZ, 10% (n=15) developed a UTI, while 16% (n=10) of patients who received dapsone developed a UTI. Kaplan-Meier estimates of UTI by type of antibiotic prophylaxis are shown in Figure 1. This difference was not statistically significant (Log-rank test p=0.20).
Figure 1.
Survival analysis of time to first UTI
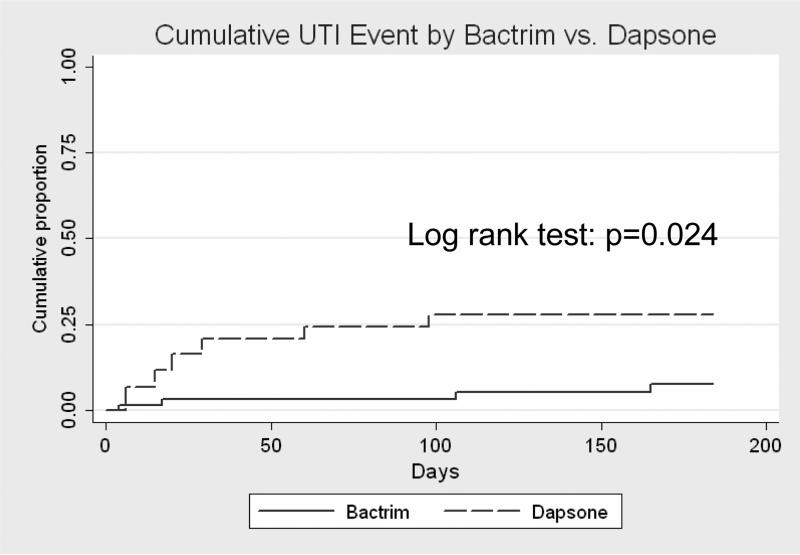
In patients over the age of 47 years (mean, standard deviation 6.2 years), those treated with dapsone originally or who switched from TMP/SMZ to dapsone had a greater risk of UTI compared to patients who remained on TMP/SMZ prophylaxis after transplantation (HR [95% CI]: 4.3 [1.2,15.5], p=0.024) (Figure 2). This association persisted after adjustment for sex and diabetes status (HR{95% CI]: 4.3 [1.2,16], p=0.30). Five patients developed a UTI after switching from TMP/SMZ to dapsone prophylaxis. In that small group, urinary infection occurred an average of 47 days after the antibiotic change, with the earliest infection occurring 11 days after discontinuing TMP/SMZ.
Figure 2.
Survival analysis of time to first UTI, older age
Sixty percent of all UTIs occurred in the first 30 days after transplantation, with the ureteral stent in place. During this early time period, in patients over age 47 years, dapsone use was associated with a higher occurrence of UTI (HR [95% CI]: 7.44 [1.3,41.6], p=0.02). There was no difference in the subgroup of patients younger than the mean age, nor was there a statistically significant difference in the group as a whole during this time period based on antibiotic selection (HR [95% CI]: 1.18 [0.4,3.8], p=0.77). Further adjustment for sex and diabetes status was not possible in this cohort of individuals with early UTI.
Discussion
In this retrospective cohort of renal transplant recipients who were not prescribed corticosteroids for maintenance immunosuppression, 16% of patients developed a UTI within the first 180 days after transplant surgery. Risk factors for development of UTI in this cohort included female gender and abnormal genitor-urinary anatomy (as described previously). For renal transplant recipients over the age of 47 years, treated without long-term glucocorticoids, our retrospective data suggest low dose TMP/SMZ is associated with a lower risk of UTI compared to dapsone prophylaxis.
The overall incidence of UTI in our two institutions was less than that previously reported in the literature (1, 2, 3). This reduced risk may reflect several differences in our study compared with previous trials. We evaluated data pertaining only to the first occurance of UTI, censoring data after the primary urinary infection or at the six-month post-transplant mark, when antibiotics for PCP prophylaxis were discontinued. In total, gram-negative enterobacteriaceae, such as Escherichia coli and Klebsiella pneumoniae accounted for the largest proportion of UTI-forming bacteria (n=14, 56%). Of these, only three (21%) were susceptible to TMP/SMZ. According to antiobiograms from our institution, resistance of these organisms to TMP/SMZ is growing. In 2005, 23% of clinic patients with Escherichia coli UTIs were resistant to TMP/SMZ. By 2007, resistance had increased to 33%. Resistance of Klebsiella pneumoniae increased from 13% to 18% during this time period. We hypothesize that UTIs in our patients on TMP/SMZ prophylaxis may have been suppressed when patients contracted a susceptible strain of either Escherichia coli or Klebsiella pneumoniae. We have no evidence that our routine use of TMP/SMZ altered resistance patterns in the community.
Enterococcus faecalis accounted for over a quarter (n=7, 28%) of the urinary infections and all were sensitive to treatment with the antibiotic vancomycin. We hypothesize that even low dose TMP/SMZ may effectively diminish the risk of developing a TMP/SMZ-sensitive urinary tract infection. Additionally, patients not exposed to long-term corticosteroids may indeed have reduced urinary infection risk compared to those who receive maintenance steroids (17).
Although this was a small cohort and included only incident renal transplant patients maintained on a steroid sparing immunosuppressive regimen, in the subset of individuals older than the mean age, the use of low-dose TMP/SMZ was associated with a lower risk of UTI formation compared to those prescribed dapsone therapy. This association was not observed in younger patients. One explanation may be differences in the type of causative organism between older and younger patients in this cohort. Younger patients in our cohort were more likely to develop a UTI from organisms not generally susceptible to TMP/SMZ, such as pseudomonas or enterococcus. Seventy-one percent of patients with an Enterococcus species cultured from the urine and 75% of those with a Pseudomonas species were younger than 47 years. This finding parallels the results of a previous case series, which evaluated the microbiologic etiology of both community- and nosocomial-acquired urinary tract infections in multiple age groups at a university-based hospital. That study reported that the distribution of gram negative and gram positive UTIs was similar between patients younger than age 65 years and those over age 65. However, similar to our findings, younger patients had a higher rate of Enterococcus species-induced urinary infections compared to those over age 65 (7.4% vs. 4%)(18).
Although there is no comparison to patients without antibiotic prophylaxis, our findings suggest that TMP/SMZ 80mg/400mg daily may indeed be an effective prophylaxis against UTI in immunocompromised hosts. Alternatively, the use of TMP/SMZ may increase the likelihood of UTI from traditionally TMP/SMZ-resistant organisims, such as Enterococcus or Pseudomonas species.
In this cohort, over a third of the patients initially prescribed TMP/SMZ for prophylaxis had to discontinue this medication prematurely and the likelihood of stopping this medication was similar in both age groups. Though discontinuing this medication early may reflect some practice bias on the part of our physicians, other studies evaluating long-term use of TMP/SMZ have also reported high rates of drug intolerance (19,20). A quarter of our patients discontinued TMP/SMZ as a result of a hypersensitivity reaction and 11% of patients developed acute interstitial nephritis (AIN). Of the seven patients suspected of having AIN, four had evidence of granulomata and interstitial eosinophil infiltration on renal allograft biopsy. Fortunately, no patient developed irreversible kidney damage or other long-term morbidity from treatment with this medication.
Acute interstitial nephritis is a rare cause of renal allograft dysfunction and when described, has often been attributed drugs such as TMP/SMZ (21,22). Although results are mixed, some studies have suggested that early steroid therapy may be beneficial in cases of drug-induced AIN (21,23,24). While we do not have historical data in which to compare, we postulate that if steroids are protective against drug-induced AIN, the occurrence of this hypersensitivity reaction may increase as more transplant centers move away from routine use of glucocorticoids.
Limitations of this study include the retrospective design and small size of our cohort which may limit our ability to describe a significant difference in UTI incidence in all patients and precludes further subclassification based on age. Additionally, the definition of UTI in prior publications is variable. We chose a definition that was consistent with the practice patterns of our physicians regarding treatment of pyuria and bacturia. It is possible that either a more liberal or more strict definition of UTI may have altered the outcomes of this study. Also, although we request that laboratory tests performed outside of our institution be sent to our physicians routinely for review, it is possible that we did not capture every UTI, if diagnosed and treated at an outside facility. Finally, this work represents only one center's experience and therefore the generalizability of the findings may be limited.
Additionally, we did not observe enough UTI outcomes to adjust for more than two variables without violating the modeling statistics. We decided a priori to adjust for both sex and diabetes status, as these have a previously demonstrated relationship with UTI(2,25). When substituting the variable of anatomical abnormality for diabetes status, the results overall were similar.
In summary, initial treatment with daily, low-dose TMP/SMZ may lower the risk of urinary infections in certain renal transplant patients undergoing early steroid withdrawal for maintenance immunosuppression. Larger prospective studies or randomized controlled trials are needed to clarify the risk of incident UTI in kidney transplant recipients treated with steroid-sparing immunosuppression regimens.
Acknowledgements
The authors acknowledge Anthony Langone, MD, and J. Harold Helderman, MD, for their assistance in this research. Dr. Kerri Cavanaugh was supported by NIH grant support (NIDDK K23K080952).
Contributor Information
Jeffrey Allen Giullian, South Denver Nephrology Associates. Denver, CO.
Kerri Cavanaugh, Division of Nephrology, Vanderbilt University Medical Center. Nashville, TN.
Heidi Schaefer, Division of Nephrology, Vanderbilt University Medical Center. Nashville, TN.
References
- 1.Tolkhoff-Rubin NE, Rubin RH. Urinary tract infection in the immunocompromised host: lessons from kidney transplantation and the AIDS epidemic. Infect Dis Clin North Am. 1997;11:707–17. doi: 10.1016/s0891-5520(05)70381-0. [DOI] [PubMed] [Google Scholar]
- 2.Alangaden GH, Thyagarajan R, Gruber SA, Morawski K, Garnick J, El-Amm JM, West MS, Sillix DH, Chandrasekar PH, Haririan A. Infections complications after kidney transplantation: current epidemiology and associated risk factors. Clin Transplant. 2006;20:401–409. doi: 10.1111/j.1399-0012.2006.00519.x. [DOI] [PubMed] [Google Scholar]
- 3.Valera B, Gentil MA, Cabello V, Fijo J, Cordero E, Cisneros JM. Epidemiology of Urinary Infections in Renal Transplant Recipients. Transplantation Proceedings. 2006;38:2414–2415. doi: 10.1016/j.transproceed.2006.08.018. [DOI] [PubMed] [Google Scholar]
- 4.Wilson CH, Bhatti AA, Rix DA, Manas DM, for the Cochrane Database Systematic Review Routine intraoperative ureteric stenting for kidney transplant recipients. 2005;4 doi: 10.1002/14651858.CD004925.pub2. [DOI] [PubMed] [Google Scholar]
- 5.Glazier DB, Jacobs MG, Lyman NW, Whang MI, Manor E, Mulgaonkar SP. Urinary tract infection associated with ureteral stents in renal transplantation. Can J Urol. 1998;5(1):462–466. [PubMed] [Google Scholar]
- 6.Engelstein D, Dorfman B, Yussim A, Shmueli D, Bar Nathan N, Shaharabani E, Shapira Z. A Critical Appraisal of Vesiculoureteral Reflux in Long-Term Renal Transplantation Recipients: Prospective Study. Transplantation Proceedings. 1997;29:136–137. doi: 10.1016/s0041-1345(96)00036-x. [DOI] [PubMed] [Google Scholar]
- 7.Basiri A, Simforoosh N, Khoddam R, Hosseini Moghaddam SM, Shayani Nasab H. A comparison of augmentation cystoplasty before and after renal transplantation with the control group. Urol J. 2004;1:45–48. [PubMed] [Google Scholar]
- 8.Memikoglu KO, Keven K, Sengul S, Soypacaci Z, Erturk S, Erbay B. Urinary Tract Infections Following Renal Transplantation: A Single-Center Experience. Transplantation Proceedings. 2007;39:3131–3134. doi: 10.1016/j.transproceed.2007.10.005. [DOI] [PubMed] [Google Scholar]
- 9.Chuang P, Parikh CR, Langone A. Urinary tract infections after renal transplantation: a retrospective review at two US transplant centers. Clinical Transplantation. 2005;19:230–235. doi: 10.1111/j.1399-0012.2005.00327.x. [DOI] [PubMed] [Google Scholar]
- 10.Ramsey DE, Finch WT, Birtch AG. Urinary tract infections in kidney transplant recipients. Arch Surg. 1979;114:1022–1025. doi: 10.1001/archsurg.1979.01370330044007. [DOI] [PubMed] [Google Scholar]
- 11.Dharnidharka VR, Agodoa LY, Abbott KC. Effects of urinary tract infection on outcomes after renal transplantation in children. CJASN. 2007;2(1):100–106. doi: 10.2215/CJN.01820506. [DOI] [PubMed] [Google Scholar]
- 12.Green H, Paul M, Vidal L, Leibovici L. Prophylaxis of Pneumocystis Pneumonia in Immunocompromised Non-HIV-Infected Pateints: Systematic Review and Meta-analysis of Randomized Controlled Trials. Mayo Clin Proc. 2007;82(9):1052–1059. doi: 10.4065/82.9.1052. [DOI] [PubMed] [Google Scholar]
- 13.Khosroshahi HT, Mogaddam AN, Shoja MM. Efficacy of High-Dose Trimethoprim-Sulfamethoxazol Prophylaxis on Early Urinary Tract Infection After Renal Transplantation. Transplantation Proceedings. 2006;38:2062–1064. doi: 10.1016/j.transproceed.2006.06.111. [DOI] [PubMed] [Google Scholar]
- 14.Tolkoff-Rubin NE, Cosimi AB, Russell PS, Rubin RH. A controlled study of trimethoprimsulfamethoxazole prophylaxis of urinary tract infection in renal transplant recipients. Rev Infect Dis. 1982;4(2):614–618. doi: 10.1093/clinids/4.2.614. [DOI] [PubMed] [Google Scholar]
- 15.Fox BC, Sollinger HW, Belzer FO, Maki DG. A prospective, randomized, double-blind study of trimethoprim-sulfamethoxazole for prophylaxis of infection in renal transplantation: clinical efficacy, absorption of trimethoprim-sulfamethoxazole, effects on the microflora, and the const-benefit of prophylaxis. Am J Med. 1990;89(3):255–274. doi: 10.1016/0002-9343(90)90337-d. [DOI] [PubMed] [Google Scholar]
- 16.Augustine JJ, Hricik DE. Steroid Sparing in Kidney Transplantation: Changing Paradigms, Improving Outcomes, and Remaining Questions. CJASN. 2006;1:1080–1089. doi: 10.2215/CJN.01800506. [DOI] [PubMed] [Google Scholar]
- 17.Tan JY, Zhao N, Wu TX, Yang KH, Zhang JD, Tian JH, Liu YL, Wang KJ, Chen F, Li SF, Li YP. Steroid Withdrawal Increases Risk of Acute Rejection by Reduces Infeciotn: A Meta-Analysis of 1681 Cases in Renal Transplantation. Transplantation Proceedings. 2006;38:2054–2056. doi: 10.1016/j.transproceed.2006.06.039. [DOI] [PubMed] [Google Scholar]
- 18.Ackermann RJ, Monroe PW. Bacteremic urinary tract infection in older people. J Am Geriatr Soc. 1996;44:927–33. doi: 10.1111/j.1532-5415.1996.tb01862.x. [DOI] [PubMed] [Google Scholar]
- 19.Fisman JA. Prevention of Infection Caused by Pneumocystis carinii in Transplant Recipients. Clinical Infectious Diseases. 2001;33:1397–1405. doi: 10.1086/323129. [DOI] [PubMed] [Google Scholar]
- 20.Para MF, Finkelstein D, Becker S, Dohn M, Walawander A, Black JR. Reduced toxicity with gradual initiation of trimethoprim-sulfamethoxazole as primary prophylaxis for Pneumocystis carinii pneumonia: AIDS Clinical Trials Group. 268. Journal of Aquired Immune Deficiency Syndrome. 2000;24(4):337–343. doi: 10.1097/00126334-200008010-00007. [DOI] [PubMed] [Google Scholar]
- 21.Josephson MA, Chiu MY, Woodle ES, Thistlethwaite JR, Haas M. Drug-induced acute interstitial nephritis in renal allorgraft: histopathologic features and clinical course in six patients. Am J Kidney Dis. 1999;34(3):540–8. doi: 10.1016/s0272-6386(99)70083-1. [DOI] [PubMed] [Google Scholar]
- 22.Sen S, Bayrak R, Ok E, Basdemir G. Drug-Induced Acute Interstitial Nephritis and Vascultitis or Vascular Rejection in Renal Allografts. America Journal of Kidney Diseases. 2001;37(1):E4. [PubMed] [Google Scholar]
- 23.Laberke HG. Treatment of acute interstitial nephritis. Klischenn Wochenschrift. 1980;58(10):531–532. doi: 10.1007/BF01477071. [DOI] [PubMed] [Google Scholar]
- 24.Gonzalez E, Gutierrez E, Galeano C, Chevia C, de Sequera P, Bernis C, Parra EG, Delgado R, Sanz M, Ortiz M, Goicoechea M, Quereda C, Olea T, Bouarich H, Hernandez Y, Segovia B, Praga M. Early steroid treatment improves the recovery of renal function in patients with drug-induced acute interstitial nephritis. KI. 2008;73(8):940–946. doi: 10.1038/sj.ki.5002776. [DOI] [PubMed] [Google Scholar]
- 25.Abbott K, Swanson S, Richter E, Bohen E, Agodoa L, Peters T, Barbour G, Lipnic R, Cruess D. Late Urinary Tract Infection After Renal Transplantation in the United States. AJKD. 2004;44(2):353–362. doi: 10.1053/j.ajkd.2004.04.040. [DOI] [PubMed] [Google Scholar]