Abstract
Background and objectives
High body mass index appears protective in hemodialysis patients, but uncertainty prevails regarding which components of body composition, fat or lean body mass, are primarily associated with survival.
Design, setting, participants, & measurements
Data between April 2006 and December 2012 were extracted from the Fresenius Medical Care Europe subset of the international MONitoring Dialysis Outcomes initiative. Fresenius Medical Care Europe archives a unique repository of predialysis body composition measurements determined by multifrequency bioimpedance (BCM Body Composition Monitor). The BCM Body Composition Monitor reports lean tissue indices (LTIs) and fat tissue indices (FTIs), which are the respective tissue masses normalized to height squared, relative to an age- and sex-matched healthy population. The relationship between LTI and FTI and all-cause mortality was studied by Kaplan–Meier analysis, multivariate Cox regression, and smoothing spline ANOVA logistic regression.
Results
In 37,345 hemodialysis patients, median (25th–75th percentile) LTI and FTI were 12.2 (10.3–14.5) and 9.8 (6.6–12.4) kg/m2, respectively. Median (25th–75th percentile) follow-up time was 266 (132–379) days; 3458 (9.2%) patients died during follow-up. Mortality was lowest with both LTI and FTI in the 10th–90th percentile (reference group) and significantly higher at the lower LTI and FTI extreme (hazard ratio [HR], 3.37; 95% confidence interval [95% CI], 2.94 to 3.87; P<0.001). Survival was best with LTI between 15 and 20 kg/m2 and FTI between 4 and 15 kg/m2 (probability of death during follow-up: <5%). When taking the relation between both compartments into account, the interaction was significant (P=0.01). Higher FTI appeared protective in patients with low LTI (HR, 3.37; 95% CI, 2.94 to 3.87; P<0.001 at low LTI–low FTI, decreasing to HR, 1.79; 95% CI, 1.47 to 2.17; P<0.001 at low LTI–high FTI).
Conclusions
This large international study indicates best survival in patients with both LTI and FTI in the 10th–90th percentiles of a healthy population. In analyses of body composition, both lean tissue and fat tissue compartments and also their relationship should be considered.
Keywords: survival, dialysis, bioimpedance, lean tissue, fat tissue
Introduction
Contrary to the general population, higher body mass index (BMI) in hemodialysis (HD) patients is associated with better survival (1). Similar results were found in older adults and in many chronic diseases (2–6). Numerous HD studies have addressed this phenomenon, with largely inconsistent results. Discrepancy exists even regarding the upper BMI limit for survival advantage, with some studies reporting a positive effect even at BMI values >35 kg/m2 and others indicating an upper limit of 25 kg/m2 (7,8). Postulated reasons for improved survival with high BMI include potential benefits of adipose tissue per se, for example as a source of TNF-α–soluble receptors and lipoproteins that counteract the effects of TNF-α itself or the inflammatory effects of circulating endotoxins (8–10) or as a vital energy source in undernourished patients (10). In health, aging is associated with loss of muscle mass in the range of 2–4 kg over 10 years (11). This physiologic process is believed to precede at a faster rate in patients with ESRD where several catabolic factors accelerate muscle wasting (12,13). In addition, anabolic pathways are compromised by low testosterone levels (14) and abnormalities in the insulin growth factor–1 pathways (15,16). It is therefore not surprising that patients with ESRD develop protein-energy wasting (PEW), not because of reduction in fat mass, but because of loss of lean tissue. PEW is present in 18%–75% of patients with CKD (17,18).
BMI measures do not differentiate between fat and lean body mass so it is challenging to understand quantitatively which components of body composition are related to survival in patients with ESRD: fat tissue mass (FTM), lean tissue mass (LTM), or both. Studies with surrogate markers of FTM and LTM suggest a dominant protective role of LTM over FTM (7,17,19,20). However, other studies concluded that survival is dependent more on FTM than LTM (10,21).
Recently, multifrequency bioimpedance spectroscopy (MF-BIS) devices have been introduced to assess body composition in HD patients. MF-BIS directly measures FTM and LTM on a routine basis. In this study, predialysis standard of care body composition assessed by MF-BIS was used to explore the relationship between lean tissue, fat tissue, and survival.
Materials and Methods
The MONitoring Dialysis Outcomes initiative comprises HD databases from Renal Research Institute clinics in the United States; Fresenius Medical Care (FMC) clinics in Europe, Asia Pacific, and Latin America; KfH clinics in Germany; and clinics associated with the Imperial College (United Kingdom), Hadassah Medical Center (Israel), and Maastricht University Medical Center (The Netherlands) (22). Electronic medical records are assembled in a primary database after obtaining informed consent and in accordance with local privacy and data protection regulations. For this study, data from European FMC clinics were extracted for all patients who had at least one routine measurement using the BCM Body Composition Monitor (FMC, Bad Homburg, Germany) between April 1, 2006, and December 31, 2012 (23). In contrast to earlier bioimpedance methodologies, the BCM Body Composition Monitor expresses body composition as a three-compartment model, providing overhydration, lean tissue index (LTI), and fat tissue index (FTI), whereby LTI and FTI are the respective tissue masses normalized to height squared. Also, LTI and FTI percentiles (<10th percentile [low]; 10th–90th percentile [normal]; and >90th percentile [high]) relative to an age- and sex-matched healthy population are supplied. The three-compartment model of the BCM Body Composition Monitor has been validated against standard reference methods for assessment of fluid status and body composition in dialysis patients, albeit partly against gold standard techniques in healthy controls only (24–29).
Baseline was defined as the first BCM Body Composition Monitor measurement available for a patient in the study period (April 1, 2006–December 31, 2012). All other values were averaged for the period ±30 days of the first BCM Body Composition Monitor measurement. Measurements were performed predialysis.
Statistical Analyses
Primary outcome was all-cause death. In survival analysis, baseline values of LTI and FTI were entered as categorical variables. Univariate Kaplan–Meier analyses were conducted to explore the association of combinations of LTI and FTI categories and the time between first body composition measurement and death. Survival differences between groups were assessed using the log-rank test. Multivariate Cox proportional-hazard models adjusted for age, sex, and dialysis vintage assessed the association of LTI, FTI, and different FTI/LTI category combinations with mortality. The robustness of the results was tested with sensitivity analyses controlling for Kt/V and restricted to patients with dialysis vintage <6 months. Cox proportional hazards and linearity assumptions were confirmed using diagnostic plots of scaled Schoenfeld and Martingale residuals. Censoring events were transfer to non-FMC clinics, transplantation, or study end on December 31, 2012. To confirm the results, we fitted a smoothing spline ANOVA logistic regression model for the probability of death 1 year later. This treats both LTI and FTI as continuous variables and allows the data to decide the joint effect of LTI and FTI (30). The same covariates as included in the Cox regression analysis were included in the spline model. In addition, sex-specific spline analyses were also performed to test the observations pertaining to the relationship between BMI and FTI. The R package gss was used to fit the smoothing spline ANOVA model (31). A P value <0.05 was considered significant. Analyses were performed with SAS version 9.3 (Cary, NC) and R statistics software system version 3.0.2.
Results
We analyzed data from 37,345 HD patients (57% men) from 380 clinics in the following 17 European countries: Bosnia and Herzegovina, Czech Republic, France, Hungary, Ireland, Italy, Poland, Portugal, Romania, Russia, Serbia, Slovakia, Slovenia, Spain, Sweden, Turkey, and the United Kingdom. At the time of first body composition measurement, mean patient age was 62.7±15.2 years, and mean BMI was 26.0±5.3 kg/m2. Median (25th–75th percentile) follow-up time was 266 (132–379) days. Median (25th–75th percentile) of LTI and FTI were 12.2 (10.3–14.5) kg/m2 and 9.8 (6.6–12.4) kg/m2, respectively. Between the first body composition measurement and study end 3458 (9.2%) patients died. Table 1 shows key patient characteristics, inflammation markers, and nutritional parameters at baseline and nine distinct body composition categories: (1) low LTI and low FTI; (2) low LTI and normal FTI; (3) low LTI and high FTI; (4) normal LTI and low FTI; (5) normal LTI and normal FTI; (6) normal LTI and high FTI; (7) high LTI and low FTI; (8) high LTI and normal FTI; and (9) high LTI and high FTI. Although 85% of patients had a normal FTI, almost 50% had low LTI (Table 1). Reliable data on residual renal function were not available.
Table 1.
Patient characteristics and indicators of nutritional status and inflammation in an international European cohort of 37,345 hemodialysis patients
| Characteristic | All (N=37,345) | LTI<10th Percentile, FTI<10th Percentile (n=1586) | LTI<10th Percentile, FTI 10th–90th Percentile (n=14,608) | LTI<10th Percentile, FTI>90th Percentile (n=1309) | LTI 10th–90th Percentile, FTI<10th Percentile (n=4855) | LTI 10th–90th Percentile, FTI 10th–90th Percentile (n=12,776) | LTI 10th–90th Percentile, FTI>90th Percentile (n=532) | LTI>90th Percentile, FTI<10th Percentile (n=874) | LTI>90th Percentile, FTI 10th–90th Percentile (n=769) | LTI>90th Percentile, FTI>90th Percentile (n=36) | P Value |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (y) | 62.7±15.2 | 62.2±13.0 | 61.5±15.1 | 58.8±14.6 | 63.3±15.6 | 64.0±15.4 | 59.1±14.0 | 67.3±14.6 | 65.4±13.5 | 61.0±10.0 | <0.001 |
| Vintage (y) | 2.8 (0.7–6.1) | 2.8 (0.5–7.2) | 3.1 (0.9–6.8) | 2.4 (0.9–4.9) | 2.6 (0.4–6.4) | 2.5 (0.7–5.4) | 2.1 (0.7–4.5) | 2.5 (0.5–5.8) | 2.4 (0.5–4.7) | 1.5 (0.7–3.5) | <0.001 |
| BMI (kg/m2) | 26.0±5.3 | 19.0±1.9 | 25.4±3.9 | 36.3±5.0 | 21.2±2.3 | 27.6±4.0 | 40.3±4.4 | 23.9±3.2 | 31.6±4.8 | 48.0±6.4 | <0.001 |
| Men (%) | 57±49 | 72±45 | 64±48 | 61±49 | 60±49 | 51±50 | 40±49 | 43±49 | 26±44 | 19±40 | <0.001 |
| Diabetic (%) | 24±43 | 17±37 | 28±45 | 42±49 | 13±34 | 23±42 | 37±48 | 16±36 | 29±45 | 39±49 | <0.001 |
| Fat mass (kg) | 26.1±11.7 | 14.0±4.1 | 28.8±8.7 | 52.0±10.2 | 12.7±4.6 | 27.0±8.3 | 52.3±8.9 | 10.2±5.0 | 26.5±8.0 | 55.4±10.9 | <0.001 |
| Lean tissue mass (kg) | 34.4±10.5 | 32.0±7.3 | 29.7±8.6 | 27.4±9.5 | 39.6±9.3 | 36.9±9.9 | 36.2±10.3 | 48.8±11.5 | 45.4±10.8 | 45.2±12.1 | <0.001 |
| Albumin (g/dl) | 3.8±0.4 | 3.6±0.5 | 3.8±0.4 | 3.8±0.4 | 3.8±0.5 | 3.9±0.4 | 3.9±0.4 | 3.7±0.5 | 3.8±0.5 | 4.1±0.4 | <0.001 |
| CRP (mg/L) | 6.0 (2.5–15) | 7.8 (2.4–21.6) | 6.7 (2.8–16.9) | 9.5 (4–19.7) | 4.6 (1.6–12.0) | 5.5 (2.3–12.7) | 9.9 (5–17.7) | 5 (2.1–13.2) | 7.0 (3.1–14.8) | 10.3 (3.3–16.6) | <0.001 |
| Creatinine (mg/dl) | 7.7±2.5 | 6.8±2.0 | 7.6±2.4 | 7.9±2.5 | 7.6±2.4 | 8.0±2.6 | 8.0±2.7 | 7.4±2.4 | 7.5±2.6 | 8.5±3.0 | <0.001 |
| Total cholesterol (mg/dl) | 172.0±44.0 | 164.6±41.9 | 169.6±44.2 | 177.0±46.6 | 167.1±40.4 | 175.5±43.9 | 179.3±38.3 | 174.5±46.4 | 187.3±52.2 | 168.7±40.0 | <0.001 |
| HDL cholesterol (mg/dl) | 42.1±14.6 | 45.9±16.1 | 41.2±14.8 | 39.1±14.4 | 46.1±15.0 | 41.5±13.8 | 36.4±10.1 | 46.8±16.2 | 41.7±14.0 | 38.6±11.3 | <0.001 |
| LDL cholesterol (mg/dl) | 100.3±37.6 | 94.7±34.4 | 99.3±37.6 | 101.4±39.0 | 97.9±35.2 | 102.2±38.2 | 105.2±39.1 | 97.9±38.3 | 109.7±38.4 | 97.0±36.1 | <0.001 |
| Triglycerides (mg/dl) | 161.8±99.4 | 121.9±62.9 | 159.992.3 | 200.6±120.3 | 123.3±67.0 | 174.8±107.9 | 224.2±134.0 | 149.9±103.3 | 191.6±122.3 | 240.1±102.1 | <0.001 |
| Predialysis systolic BP (mmHg) | 136.4±19.5 | 137.2±20.5 | 134.8±20.4 | 134.6±20.8 | 139.3±18.4 | 137.0±18.5 | 137.5±19.2 | 138.5±18.7 | 138.7±18.0 | 128.6±15.7 | <0.001 |
| Serum sodium (mmol/L) | 137.9±3.4 | 137.5±3.5 | 137.7±3.5 | 137.8±3.4 | 138.1±3.5 | 138.2±3.3 | 138.2±3.3 | 138.4±3.3 | 138.3±3.3 | 139.4±3.4 | <0.001 |
| Body surface area (m2) | 1.8±0.2 | 1.6±0.2 | 1.8±0.2 | 2.0±0.2 | 1.6±0.2 | 1.8±0.2 | 2.1±0.2 | 1.7±0.2 | 1.8±0.2 | 2.1±0.3 | <0.001 |
| FTI (kg/m2) | 9.8±4.5 | 5.1±1.5 | 10.6±3.4 | 19.6±4.2 | 4.7±1.7 | 10.2±9.3 | 20.2±3.9 | 3.9±1.9 | 10.5±3.2 | 22.8±4.3 | <0.001 |
| LTI (kg/m2) | 12.5±3.1 | 11.4±1.9 | 10.7±2.3 | 10.0±2.6 | 14.4±2.4 | 13.6±2.5 | 13.6±2.4 | 18.4±3.0 | 17.7±3.2 | 18.3±3.0 | <0.001 |
| Died (%) | 9±29 | 17±40 | 10±30 | 9±28 | 10±30 | 7±25 | 7±26 | 10±30 | 9±28 | 8±28 |
Values are mean baseline values±SDs. Median baseline values (interquartile range) are reported for CRP and dialysis vintage (25th–75th percentiles). Baseline was defined as the first BCM Body Composition Monitor measurement available for a patient in the study period (April 1, 2006–December 31, 2012), whereby all other values were averaged for the period ±30 days of the first BCM Body Composition Monitor measurement. P values were estimated with one-way ANOVA (standard with Gaussian assumption or Kruskal–Wallis test). BMI, body mass index; CRP, C-reactive protein; FTI, fat tissue index; LTI, lean tissue index.
Of the six groups with normal or high FTI, BMI values were higher than the cohort average value (i.e., 26.0±5.3 kg/m2) in five groups (41.3% of the study population). A BMI value slightly lower than the cohort average was observed for the remaining group with normal FTI and low LTI (39.1% of the study population; BMI, 25.41±3.89 kg/m2). Sex-specific spline analyses of the relationship between BMI and FTI revealed that BMI increases with increasing FTI (Supplemental Figures 1 and 2). Of the six groups with normal or high LTI, BMI values were higher than the cohort average in four groups (37.8% of the study population). Lower than cohort average BMI was observed for the remaining two groups with low FTI (15.3% of the study population). Table 2 provides a summary of the BMI categories as defined by the World Health Organization (WHO) (in 2004) and the LTI and FTI groups analyzed in this study. BMI levels were higher than one would expect given the lean and fat tissue amounts: respectively, 20% and 47% of patients had FTI and LTI below the 10th percentile, but they had normal to obese BMI levels.
Table 2.
Matrix presentation of body mass index values in the nine LTI and FTI percentile categories
| LTI (kg/m2) | FTI (kg/m2) | |||
|---|---|---|---|---|
| <10th Percentile | 10th–90th Percentile | >90th Percentile | ||
| <10th percentile | 19.0±1.9 | 25.4±3.9 | 36.3±5.0 | WHO: normal–obese class II |
| 10th–90th percentile | 21.2±2.3 | 27.6±4.0 | 40.3±4.4 | WHO: normal–obese class III |
| >90th percentile | 23.9±3.2 | 31.6±4.8 | 48.0±6.4 | WHO: normal–obese class III |
| WHO: normal | WHO: overweight–obese class I | WHO: obese class II–III | ||
Mean body mass index±SD are presented. Definitions of normal to obese class III are according to the WHO classification for the general population and are on the basis of the group means. LTI, lean tissue index; FTI, fat tissue index; WHO, World Health Organization.
Univariate Kaplan–Meier analyses indicated a survival benefit for patients with normal LTI and concomitant normal FTI. The worst survival was observed for patients with a combination of low LTI and low FTI.
The results of the multivariate Cox proportional-hazard analyses are presented in Table 3. First, we constructed three explanatory models with LTI and FTI as categorical variables and with normal FTI and normal LTI as references. Model 1 included LTI but not FTI; model 2 included FTI but not LTI; and model 3 included combinations of LTI and FTI categories. Model 1 indicated that both low and high LTIs were associated with higher mortality (hazard ratio [HR], 1.68; 95% confidence interval [95% CI], 1.56 to 1.80; P<0.001 and HR, 1.20; 95% CI, 1.01 to 1.41; P=0.03, respectively). Model 2 indicated that also both low and high FTIs were associated with higher mortality (HR, 1.34; 95% CI, 1.24 to 1.45; P<0.001 and HR, 1.19; 95% CI, 1.01 to 1.40; P=0.03, respectively). Model 3 indicated the highest HR for the combination of low LTI and low FTI; the lowest HR was for normal LTI and FTI. Sensitivity analyses controlling for Kt/V and restricting dialysis vintage confirmed the robustness of these results (data not shown).
Table 3.
Results of Cox proportional-hazards models
| Model | Models Adjusted for Age, Vintage, and Sex | Fully Adjusted Modelsa | ||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% LCI | 95% UCI | P Value | HR | 95% LCI | 95% UCI | P Value | |
| Model 1: LTI without FTI | ||||||||
| Low LTI | 1.68 | 1.56 | 1.80 | <0.001 | 1.53 | 1.40 | 1.66 | <0.001 |
| Normal LTI (reference) | ||||||||
| High LTI | 1.20 | 1.01 | 1.41 | 0.03 | 1.02 | 0.84 | 1.24 | 0.85 |
| Model 2: FTI without LTI | ||||||||
| Low FTI | 1.34 | 1.24 | 1.45 | <0.001 | 1.19 | 1.08 | 1.31 | <0.001 |
| Normal FTI (reference) | ||||||||
| High FTI | 1.19 | 1.01 | 1.40 | 0.03 | 1.23 | 1.02 | 1.47 | 0.03 |
| Model 3: LTI and FTI combined | ||||||||
| Low LTI, low FTI | 3.37 | 2.94 | 3.87 | <0.001 | 2.51 | 2.12 | 2.96 | <0.001 |
| Low LTI, normal FTI | 1.81 | 1.67 | 1.97 | <0.001 | 1.63 | 1.48 | 1.81 | <0.001 |
| Low LTI, high FTI | 1.79 | 1.47 | 2.17 | <0.001 | 1.74 | 1.40 | 2.17 | <0.001 |
| Normal LTI, low FTI | 1.57 | 1.40 | 1.75 | <0.001 | 1.42 | 1.25 | 1.62 | <0.001 |
| Normal LTI, normal FTI (reference) | ||||||||
| Normal LTI, high FTI | 1.36 | 0.99 | 1.89 | 0.06 | 1.41 | 0.99 | 2.01 | 0.06 |
| High LTI, low FTI | 1.42 | 1.14 | 1.76 | 0.002 | 0.99 | 0.75 | 1.32 | 0.95 |
| High LTI, normal FTI | 1.28 | 1.00 | 1.64 | 0.05 | 1.31 | 1.00 | 1.73 | 0.05 |
| High LTI, high FTI | 1.73 | 0.56 | 5.38 | 0.34 | 1.91 | 0.48 | 7.65 | 0.36 |
Outcome was all-cause mortality; predictors were categories of low (<10th percentile) and high (>90th percentile) LTI and FTI, respectively. Models were adjusted for age, vintage, and sex or as otherwise indicated. LTI, lean tissue index; FTI, fat tissue index; reference, normal LTI and FTI (10th–90th percentile of age- and sex-matched healthy population); HR, hazard ratio; 95% LCI, lower 95% confidence interval; 95% UCI; upper 95% confidence interval.
Adjusted for age, vintage, sex, geographic region, albumin, hemoglobin, diabetes, and BP.
All three explanatory models were only adjusted for age, sex, and vintage to avoid collinearity, but the addition of other potential variables were included in a predictive model (fully adjusted model, Table 3): only the statistical significance at high LTI–low FTI was reduced.
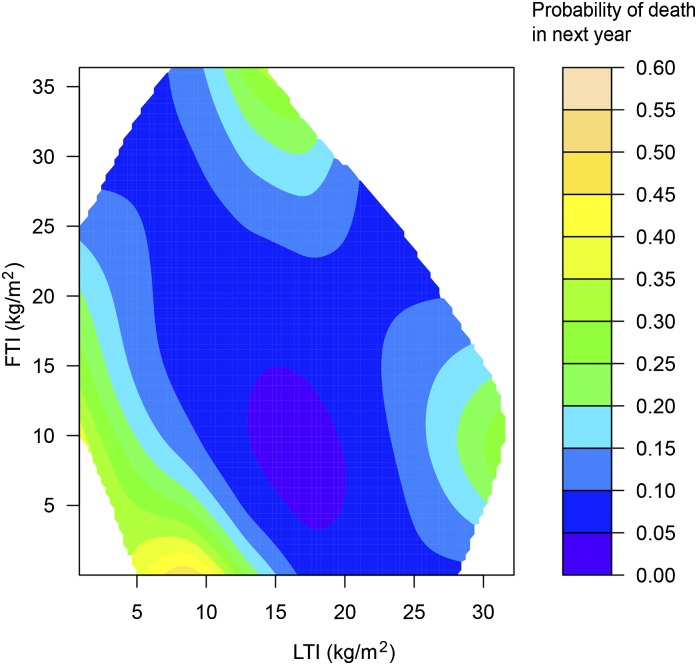
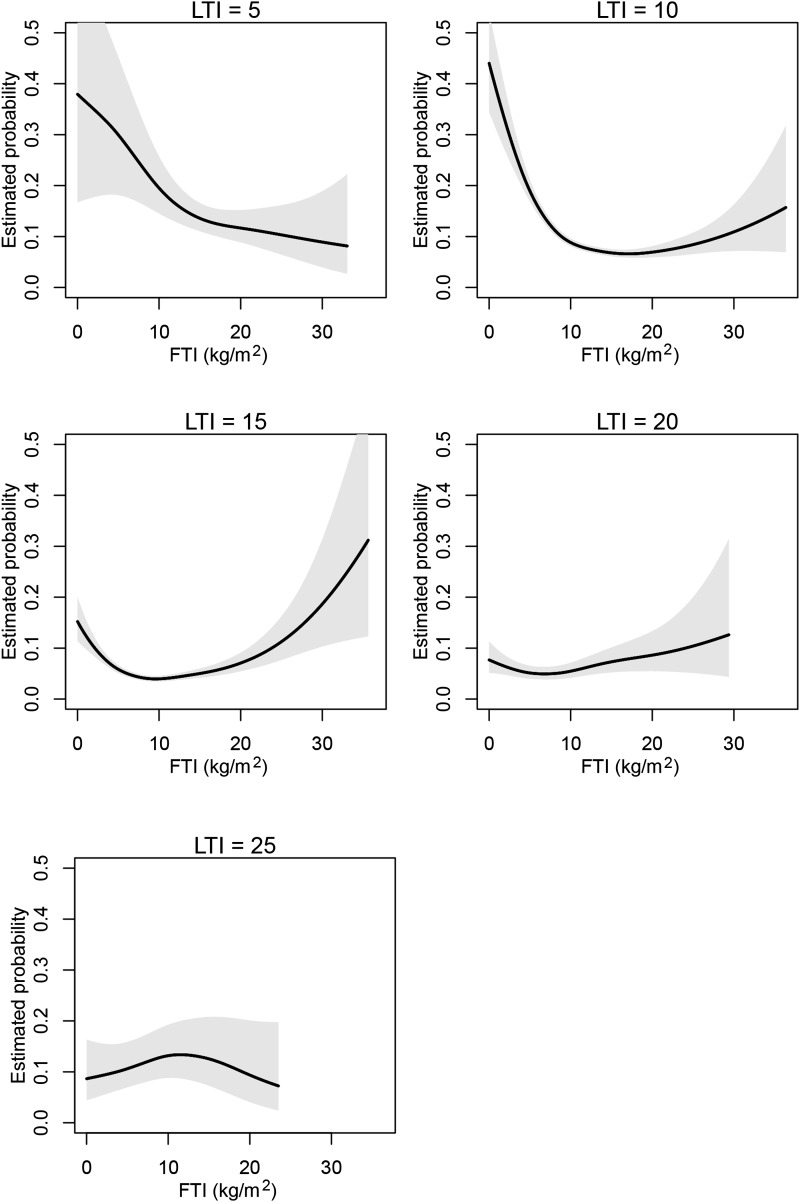
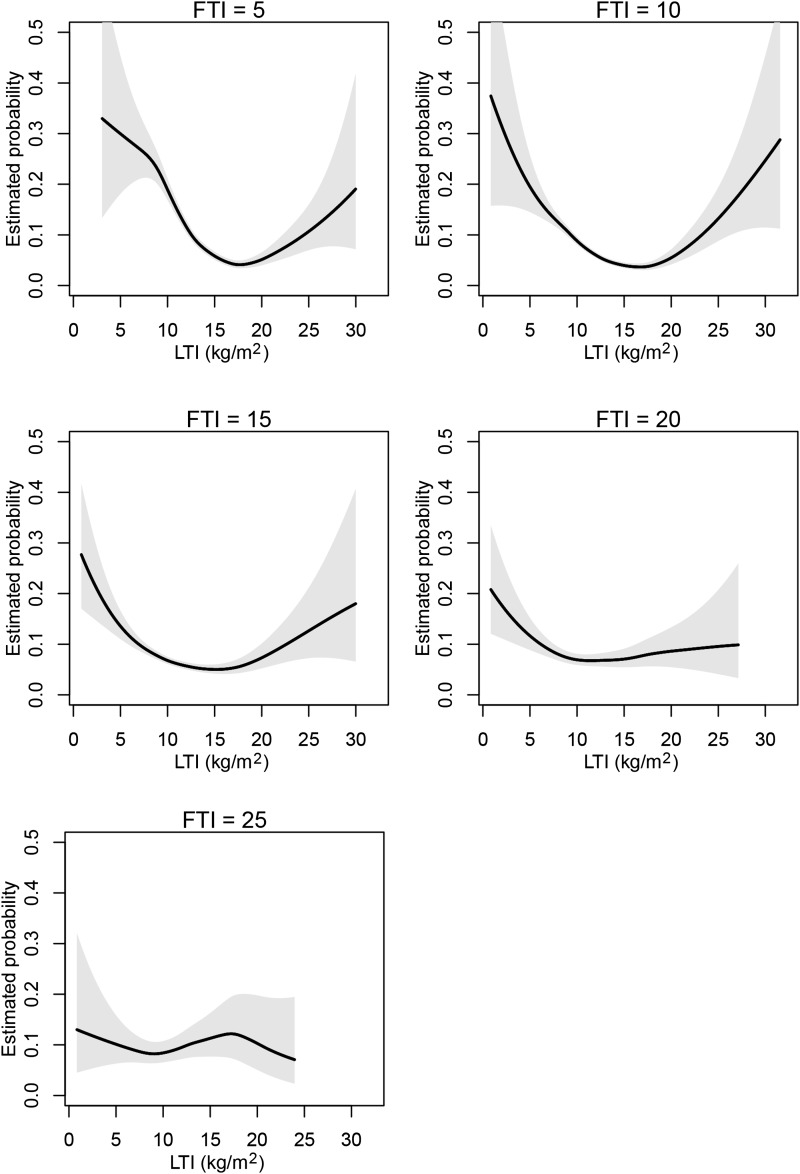
Next we conducted a 1-year survival analysis for the joint effect of LTI and FTI as continuous variables. The contour plot of the estimated death probability as a function of LTI and FTI with other covariates fixed on the basis of the smoothing spline ANOVA logistic regression model is shown in Figure 1. The result of FTI and LTI slices through the contour plot at discrete LTI and FTI values (5, 10, 15, 20, and 25 kg/m2) are displayed in Figures 2 and 3, respectively. The results corroborate the Cox regression results, revealing optimal survival at LTI and FTI in the middle ranges. However, although the range 15–20 kg/m2 appeared optimal for LTI, the optimal range for FTI appeared to be lower (4–15 kg/m2).
Figure 1.
Risk of death across levels of FTI and LTI. Contour plot of the estimated probability of death in the next year as a joint function of LTI and FTI for female patients with age and body mass index fixed at their median values. Estimates of the joint effects are shown in a region with sufficient data decided by posterior SDs. FTI, fat tissue index; LTI, lean tissue index.
Figure 2.
FTI slices at different LTI ranges. Sections though the contour plot (Figure 1) at five LTI levels. FTI, fat tissue index; LTI, lean tissue index.
Figure 3.
LTI slices at different FTI ranges. Sections though the contour plot (Figure 1) at five FTI levels. FTI, fat tissue index; LTI, lean tissue index.
Discussion
In this study of prevalent HD patients, body composition was determined as part of standard of care using a validated multifrequency bioimpedance device. LTI and FTI within the 10th–90th percentile of an age- and sex-matched healthy population were associated with best survival, whereas low FTI and low LTI, and especially the combination of both, were associated with higher mortality. The average patient BMI was 26.0±15.2 kg/m2, which is overweight for the general population (normal BMI defined by WHO: 18.5–24.9 kg/m2). In fact, the BMI distribution was found to approximately parallel the FTI distribution, but starting already at WHO normal status and progressing to overweight and then even obese: irrespective of LTI, patients with low FTI had on average a WHO normal BMI (normal, 18.5–24.9 kg/m2); patients with normal FTI had a high WHO BMI (overweight, 25–29.9 kg/m2); and patients with high FTI were obese according to the WHO classification (30 to ≥40 kg/m2) (Table 2). This observation, which was also confirmed by sex-specific analyses, indicates a shift toward higher BMIs in this HD population and can explain some previous interpretations of a protective effect of high BMI per se. Furthermore, patients in our study with even a low LTI did not appear to be underweight, but rather to have normal weight or even be obese according to the WHO BMI classification. Therefore, BMIs of HD patients are incompletely informative in predicting body composition. This can only be partially explained by the effect of fluid loading on BMI in dialysis patients (e.g., approximately +0.6 kg/m2 for an interdialytic weight gain from 75 to 77 kg in a patient of height 1.75 m), but appears to be mainly related to an altered relation between fat and lean tissue in this population.
These findings add valuable insight to understanding the observation that HD patients with high BMI experience a better survival compared with fellow patients with lower BMI. In essence, our results indicate that body composition, and not just BMI, is related to outcome and that patients with the same BMI but different ratios of lean to fat mass may have different outcomes. The robustness of the result was tested with various models incorporating LTI and FTI as categorical and continuous variables and by a sensitivity analysis when Kt/V is controlled for or when restricted to patients with dialysis vintage <6 months.
Almost half of the patients in our study had LTIs below the 10th percentile of an age- and sex-matched healthy population, whereas only 4.5% had LTIs above the 90th percentile (Table 1). Low lean body mass is a component of the PEW syndrome, and it is thought to be associated with a parallel decrease in BMI. However, our results show that most patients with low LTI had normal FTI and BMI levels ranging between normal and obese. Honda et al. demonstrated that low lean BMI (defined in their study as lean body mass determined by dual energy x-ray absorptiometry and divided by height squared) is not exclusively a characteristic of patients with low BMI (21). In fact, the prevalence of patients with PEW according to the Subjective Global Assessment was 60% in patients with a BMI<20 kg/m2, 39% in patients in the normal 20–25 kg/m2 group, and 16% in patients in the overweight group (BMI 25–29.9 kg/m2). Patients with PEW in the latter group were characterized by a higher fat BMI over lean BMI, a condition labeled obese sarcopenia. On the basis of the current findings, we suggest that the presence of a normal BMI is insufficient to refute diagnose of malnutrition and that LTI, FTI, and their relation must be taken into account.
Knowledge of predictors of low and high LTI and FTI may help to develop interventional corrective strategies. Inflammatory markers are known to be higher in patients with PEW; therefore, it is not surprising that a low LTI is associated with mortality (21). This relationship suggests that malnourished (even obese) HD patients have a low LTI because of the catabolic effects of inflammatory mediators. The C-reactive protein levels in Table 1 support this. It has also recently been demonstrated that an excess of fat mass in obese patients can amplify the oxidative stress and inflammation caused by renal insufficiency (32). This is in line with the results of our model 2, but not model 3 (whereby the latter may be because of the small numbers of patients in the upper categories). As shown in this study, the relation between LTI and FTI has to be taken into account. In patients with normal LTI, higher FTI was associated with a borderline significant outcome (P=0.06) compared with normal FTI, and in patients with low LTI it was associated with improved outcome. Fat mass can also serve as an energy reservoir in case of energy depletion, which might provide an explanation for the latter phenomenon because patients with low LTI are likely to be protein-energy wasted. Therefore, there may be a discrepancy between the long-term health risks with increased fat mass and short-term positive effects of higher fat mass in patients with low LTI and protein energy wasting (10,33). Interventions aimed to reduce inflammation may translate into a higher LTI. One such intervention would be to reduce catheter use because even subclinical infections can trigger inflammation (34). Another intervention would be timely and effective treatment of periodontitis, a frequently neglected source of inflammation (35). Strategies to improve nutritional competence may include oral supplemental nutrition programs. Strategies to increase creatinine by increasing muscle mass could possibly benefit both LTI and FTI (36).
An unexpected finding of this study is the adverse association between high LTI and survival in the simple model, which was only adjusted for age, sex, and vintage. Theoretically, underdialysis might explain this (10,33,37,38). However, adjustment for Kt/V did not materially change the results. Adjustment for age, vintage, sex, geographic region, albumin, hemoglobin, diabetes, and BP in the background predictive models resulted in a loss of statistical significance for high LTIs. This could either indicate a problem of multicollinearity in the extended model or a low degree of robustness of the association at high LTI.
This study had certain limitations. The number of patients with high LTI and FTI was small (n=36), which could compromise achievement of statistical significance here. Prevalent HD patients were studied, and the dialysis vintage at the time of first body composition measurement varied between patients. In an attempt to address this we adjusted all Cox models for dialysis vintage. MF-BIS by the BCM Body Composition Monitor was the sole source of body composition assessment. However, this specific methodology has been validated in several studies (24–29) (although fat mass was validated against gold standard techniques in healthy controls only), and the strong relationship to outcome supports the functional validity of the device and its measurements. Moreover, the BCM Body Composition Monitor is not able to distinguish between appendicular and visceral muscle mass. LTM and adipose tissue mass were normalized to height squared for reporting of LTI and FTI, but other normalizations could better reflect differences in body compositions between races and geographic areas. However, inclusion of geographic area into the model did not change the results substantially. Also, indexing to height squared is common in sarcopenia studies (39), and use of this facilitates comparison of study results. BCM Body Composition Monitor measurements were performed for clinical reasons, and supervising clinicians could use these for interventions (not blinded). However, we do not believe that this should be considered a confounder because all clinicians followed a common policy (i.e., to target relative overhydration to be ≤15%); therefore, an individual effect may be overruled. Strengths of this research lie in its multinational and diverse study population. The fact that this is the largest study on this subject conducted so far improves generalizability of the findings. Moreover, the BCM Body Composition Monitor is used on a routine basis, which reduces the chances of bias by indication.
In conclusion, this large multinational study demonstrated that LTM and FTM, as determined by whole-body MF-BIS, are important predictors of survival in chronic HD patients. Both low LTI and FTI, but especially the combination of both, are associated with poorer survival. Although BMI values for HD patients are generally higher than those for the general population, many patients with apparently overweight BMI levels have LTI levels below the 10th percentile of healthy controls. These results suggest that routine assessment of body composition by bioimpedance adds to the clinical care of HD patients. Trials of interventions to maintain lean body mass and fat mass in middle ranges are warranted to further explore the relationship between body composition and survival.
Disclosures
D.M., L.A.U., I.B., B.C., M.E., E.G., A.G., C.M., L.S., and A.S. are employees of Fresenius Medical Care and may hold stock in the company. P.K. holds stock in Fresenius Medical Care.
Supplementary Material
Acknowledgments
J.K. and F.M.vdS. received an unrestricted research grant from FMC EMEA and the Renal Research Institute.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.08550814/-/DCSupplemental.
References
- 1.Chazot C, Gassia JP, Di Benedetto A, Cesare S, Ponce P, Marcelli D: Is there any survival advantage of obesity in Southern European haemodialysis patients? Nephrol Dial Transplant 24: 2871–2876, 2009 [DOI] [PubMed] [Google Scholar]
- 2.Chao FC, Efron B, Wolf P: The possible prognostic usefulness of assessing serum proteins and cholesterol in malignancy. Cancer 35: 1223–1229, 1975 [DOI] [PubMed] [Google Scholar]
- 3.Yeh S, Wu SY, Levine DM, Parker TS, Olson JS, Stevens MR, Schuster MW: Quality of life and stimulation of weight gain after treatment with megestrol acetate: Correlation between cytokine levels and nutritional status, appetite in geriatric patients with wasting syndrome. J Nutr Health Aging 4: 246–251, 2000 [PubMed] [Google Scholar]
- 4.Malvy E, Thiébaut R, Marimoutou C, Dabis F, Groupe d’Epidemiologie Clinique du Sida en Aquitaine : Weight loss and body mass index as predictors of HIV disease progression to AIDS in adults. Aquitaine cohort, France, 1985-1997. J Am Coll Nutr 20: 609–615, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kenchaiah S, Pocock SJ, Wang D, Finn PV, Zornoff LA, Skali H, Pfeffer MA, Yusuf S, Swedberg K, Michelson EL, Granger CB, McMurray JJ, Solomon SD, CHARM Investigators : Body mass index and prognosis in patients with chronic heart failure: Insights from the Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity (CHARM) program. Circulation 116: 627–636, 2007 [DOI] [PubMed] [Google Scholar]
- 6.Bouchard C: BMI, fat mass, abdominal adiposity and visceral fat: Where is the ‘beef’? Int J Obes (Lond) 31: 1552–1553, 2007 [DOI] [PubMed] [Google Scholar]
- 7.Beddhu S, Pappas LM, Ramkumar N, Samore M: Effects of body size and body composition on survival in hemodialysis patients. J Am Soc Nephrol 14: 2366–2372, 2003 [DOI] [PubMed] [Google Scholar]
- 8.Kalantar-Zadeh K, Abbott KC, Salahudeen AK, Kilpatrick RD, Horwich TB: Survival advantages of obesity in dialysis patients. Am J Clin Nutr 81: 543–554, 2005 [DOI] [PubMed] [Google Scholar]
- 9.Hakim RM, Lowrie E: Obesity and mortality in ESRD: Is it good to be fat? Kidney Int 55: 1580–1581, 1999 [DOI] [PubMed] [Google Scholar]
- 10.Ikizler TA: Resolved: Being fat is good for dialysis patients: The Godzilla effect: Pro. J Am Soc Nephrol 19: 1059–1062, 2008 [DOI] [PubMed] [Google Scholar]
- 11.Roubenoff R, Hughes VA: Sarcopenia: Current concepts. J Gerontol A Biol Sci Med Sci 55: M716–M724, 2000 [DOI] [PubMed] [Google Scholar]
- 12.Carrero JJ, Chmielewski M, Axelsson J, Snaedal S, Heimbürger O, Bárány P, Suliman ME, Lindholm B, Stenvinkel P, Qureshi AR: Muscle atrophy, inflammation and clinical outcome in incident and prevalent dialysis patients. Clin Nutr 27: 557–564, 2008 [DOI] [PubMed] [Google Scholar]
- 13.Kooman JP, Broers NJ, Usvyat L, Thijssen S, van der Sande FM, Cornelis T, Levin NW, Leunissen KM, Kotanko P: Out of control: Accelerated aging in uremia. Nephrol Dial Transplant 28: 48–54, 2013 [DOI] [PubMed] [Google Scholar]
- 14.Carrero JJ, Qureshi AR, Nakashima A, Arver S, Parini P, Lindholm B, Bárány P, Heimbürger O, Stenvinkel P: Prevalence and clinical implications of testosterone deficiency in men with end-stage renal disease. Nephrol Dial Transplant 26: 184–190, 2011 [DOI] [PubMed] [Google Scholar]
- 15.Bailey JL, Zheng B, Hu Z, Price SR, Mitch WE: Chronic kidney disease causes defects in signaling through the insulin receptor substrate/phosphatidylinositol 3-kinase/Akt pathway: Implications for muscle atrophy. J Am Soc Nephrol 17: 1388–1394, 2006 [DOI] [PubMed] [Google Scholar]
- 16.Johansen KL, Dalrymple LS, Delgado C, Kaysen GA, Kornak J, Grimes B, Chertow GM: Association between body composition and frailty among prevalent hemodialysis patients: A US Renal Data System special study. J Am Soc Nephrol 25: 381–389, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kalantar-Zadeh K, Ikizler TA, Block G, Avram MM, Kopple JD: Malnutrition-inflammation complex syndrome in dialysis patients: Causes and consequences. Am J Kidney Dis 42: 864–881, 2003 [DOI] [PubMed] [Google Scholar]
- 18.Qureshi AR, Alvestrand A, Danielsson A, Divino-Filho JC, Gutierrez A, Lindholm B, Bergström J: Factors predicting malnutrition in hemodialysis patients: A cross-sectional study. Kidney Int 53: 773–782, 1998 [DOI] [PubMed] [Google Scholar]
- 19.Moreau-Gaudry X, Guebre-Egziabher F, Jean G, Genet L, Lataillade D, Legrand E, Kuentz F, Trolliet P, Fouque D: Serum creatinine improves body mass index survival prediction in hemodialysis patients: A 1-year prospective cohort analysis from the ARNOS study. J Ren Nutr 21: 369–375, 2011 [DOI] [PubMed] [Google Scholar]
- 20.Desmeules S, Lévesque R, Jaussent I, Leray-Moragues H, Chalabi L, Canaud B: Creatinine index and lean body mass are excellent predictors of long-term survival in haemodiafiltration patients. Nephrol Dial Transplant 19: 1182–1189, 2004 [DOI] [PubMed] [Google Scholar]
- 21.Honda H, Qureshi AR, Axelsson J, Heimburger O, Suliman ME, Barany P, Stenvinkel P, Lindholm B: Obese sarcopenia in patients with end-stage renal disease is associated with inflammation and increased mortality. Am J Clin Nutr 86: 633–638, 2007 [DOI] [PubMed] [Google Scholar]
- 22.Usvyat LA, Haviv YS, Etter M, Kooman J, Marcelli D, Marelli C, Power A, Toffelmire T, Wang Y, Kotanko P: The MONitoring Dialysis Outcomes (MONDO) initiative. Blood Purif 35: 37–48, 2013 [DOI] [PubMed] [Google Scholar]
- 23.Marcelli D, Kirchgessner J, Amato C, Steil H, Mitteregger A, Moscardò V, Carioni C, Orlandini G, Gatti E: EuCliD (European Clinical Database): A database comparing different realities. J Nephrol 14[Suppl 4]: S94–S100, 2001 [PubMed] [Google Scholar]
- 24.Chamney PW, Wabel P, Moissl UM, Müller MJ, Bosy-Westphal A, Korth O, Fuller NJ: A whole-body model to distinguish excess fluid from the hydration of major body tissues. Am J Clin Nutr 85: 80–89, 2007 [DOI] [PubMed] [Google Scholar]
- 25.Moissl UM, Wabel P, Chamney PW, Bosaeus I, Levin NW, Bosy-Westphal A, Korth O, Müller MJ, Ellegård L, Malmros V, Kaitwatcharachai C, Kuhlmann MK, Zhu F, Fuller NJ: Body fluid volume determination via body composition spectroscopy in health and disease. Physiol Meas 27: 921–933, 2006 [DOI] [PubMed] [Google Scholar]
- 26.Passauer J, Petrov H, Schleser A, Leicht J, Pucalka K: Evaluation of clinical dry weight assessment in haemodialysis patients using bioimpedance spectroscopy: A cross-sectional study. Nephrol Dial Transplant 25: 545–551, 2010 [DOI] [PubMed] [Google Scholar]
- 27.Moissl U, Bosaeus I, Lemmey A, Hovgesen S, Wabel P, Chamney PW, Bosy-Westphal A, Korth O, Mueller M, Renders L, Ellegard L, Malmros V, Fueller N: Validation of a 3C model for determination of body fat mass. J Am Soc Nephrol 18: 257, 2007 [Google Scholar]
- 28.Moissl U, Wabel P, Chamney PW, Renders L, Bosy-Westphal A, Korth O, Mueller M: Validation of a bioimpedance spectroscopy method for the assessment of fat free mass. NDT Plus 1[Suppl 2]: 215, 2008. 25983885 [Google Scholar]
- 29.Broers NJ, Martens RJ, Cornelis T, Diederen NM, Wabel P, van der Sande FM, Leunissen KM, Kooman JP: Body composition in dialysis patients: A functional assessment of bioimpedance using different prediction models. J Ren Nutr 25: 121–128, 2015 [DOI] [PubMed] [Google Scholar]
- 30.Wang Y: Smoothing Splines: Methods and Applications, Boca Raton, FL, CRC Press, 2011 [Google Scholar]
- 31.Gu C: Smoothing Spline ANOVA Models, New York, Springer, 2013 [Google Scholar]
- 32.Ramos LF, Shintani A, Ikizler TA, Himmelfarb J: Oxidative stress and inflammation are associated with adiposity in moderate to severe CKD. J Am Soc Nephrol 19: 593–599, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Stenvinkel P, Lindholm B: Resolved: Being fat is good for dialysis patients: The Godzilla effect: Con. J Am Soc Nephrol 19: 1062–1064, 2008 [PubMed] [Google Scholar]
- 34.Dittmer ID, Sharp D, McNulty CA, Williams AJ, Banks RA: A prospective study of central venous hemodialysis catheter colonization and peripheral bacteremia. Clin Nephrol 51: 34–39, 1999 [PubMed] [Google Scholar]
- 35.Jofré R, Rodriguez-Benitez P, López-Gómez JM, Pérez-Garcia R: Inflammatory syndrome in patients on hemodialysis. J Am Soc Nephrol 17[Suppl 3]: S274–S280, 2006 [DOI] [PubMed] [Google Scholar]
- 36.Mihaescu A, Avram C, Bob F, Gaita D, Schiller O, Schiller A: Benefits of exercise training during hemodialysis sessions: A prospective cohort study. Nephron Clin Pract 124: 72–78, 2013 [DOI] [PubMed] [Google Scholar]
- 37.Kotanko P, Thijssen S, Kitzler T, Wystrychowski G, Sarkar SR, Zhu F, Gotch F, Levin NW: Size matters: Body composition and outcomes in maintenance hemodialysis patients. Blood Purif 25: 27–30, 2007 [DOI] [PubMed] [Google Scholar]
- 38.Sridharan S, Vilar E, Berdeprado J, Farrington K: Energy metabolism, body composition, and urea generation rate in hemodialysis patients. Hemodial Int 17: 502–509, 2013 [DOI] [PubMed] [Google Scholar]
- 39.Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M, European Working Group on Sarcopenia in Older People : Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 39: 412–423, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.