Abstract
Disruptions in circadian rhythms and dopaminergic activity are involved in the pathophysiology of bipolar disorder, though their interaction remains unclear. Moreover, a lack of animal models that display spontaneous cycling between mood states has hindered our mechanistic understanding of mood switching. Here we find that mice with a mutation in the circadian Clock gene (ClockΔ19) exhibit rapid mood-cycling, with a profound manic-like phenotype emerging during the day following a period of euthymia at night. Mood cycling coincides with abnormal daytime spikes in ventral tegmental area (VTA) dopaminergic activity, tyrosine hydroxylase (TH) levels, and dopamine synthesis. To determine the significance of daytime increases in VTA dopamine activity to manic behaviors, we developed a novel optogenetic stimulation paradigm that produces a sustained increase in dopamine neuronal activity and find that this induces a manic-like behavioral state. Time-dependent dampening of TH activity during the day reverses manic-related behaviours in ClockΔ19 mice. Finally, we show that CLOCK acts as a negative regulator of TH transcription, revealing a novel molecular mechanism underlying cyclic changes in mood-related behaviour. Taken together, these studies have identified a mechanistic connection between circadian gene disruption and the precipitation of manic episodes in bipolar disorder.
Keywords: circadian rhythms, ventral tegmental area, dopamine, optogenetics, anxiety, depression, bipolar disorder
Introduction
Bipolar disorder is a complex psychiatric illness that presents with abnormal mood states ranging from anxiety and depression to altered activity levels and disrupted sleep-wake cycles. Mood-cycling, or switching, between manic, depressive, and euthymic mood states is a hallmark feature of bipolar disorder, yet insight into its mechanistic driving force has remained highly elusive. Compelling evidence suggests that altered circadian clocks are involved in the psychopathology of bipolar disorder1, as disrupted circadian rhythms often predict the onset of mood episodes and can, themselves, precipitate depression or mania in susceptible individuals2, 3. Previously we demonstrated that mice with a point mutation in the Clock gene (ClockΔ19)4, 5, a core regulatory component of the circadian transcriptional machinery, display a behavioural profile during the daytime (i.e. their inactive period) that is strikingly similar to the manic phase of human bipolar disorder, and includes hyperactivity, increased reward-related behaviors, and decreased anxiety- and depressive-like behaviours3, 6. This behavioral profile is concurrent with increased firing of ventral tegmental area (VTA) dopamine cells6, 7. Indeed, converging lines of evidence point to altered mesolimbic dopaminergic activity as a prominent mediator of human mania. For instance, psychostimulant exposure produces a behavioral state similar to bipolar mania and the dopamine precursor, L-dopa can precipitate mania in patients8. In turn, administration of alpha-methyl-para tyrosine (AMPT), an inhibitor of the rate-limiting enzyme in dopamine synthesis tyrosine hydroxylase (TH), can decrease manic symptoms 8. Interestingly, direct knock-down of Clock in the VTA of wild-type mice recapitulates the reduced anxiety-related phenotypes typical of manic-like behavior inClockΔ19 mice, but also leads to increased depression-related behaviour9, hinting at a potential role for Clock in regulating VTA dopaminergic activity and distinct mood states. However, it is currently unclear how manic-episodes are precipitated and what molecular mechanisms underlie this process. Given this, we first sought to establish a direct and functionally significant link between the diurnal activity of VTA dopaminergic activity and mood-related behaviour. Optogenetics was used to enable direct control of VTA dopamine neuronal firing to determine to what extent VTA dopamine activity drives select features of manic-like behaviour. Whereas most studies using optogenetics have examined the effects of acute neural circuit modulation, here we introduce a novel optogenetic paradigm to induce chronic changes in VTA neural activity. In addition, we sought to determine the molecular mechanisms by which the circadian Clock gene regulates dopaminergic activity.
Materials and Methods
Animals
ClockΔ19 mutant mice were created by N-ethyl-N-nitrosurea mutagenesis that produces a dominant-negative CLOCK protein defective in transcriptional activity5. For all experiments using ClockΔ19 mutants, adult male mutant (Clock/Clock) and wild-type (+/+) littermate controls, on a mixed BALBc, C57BL/6 background, were group housed in sets of 2–4 per cage. Optogenetic studies were carried out on adult male Tyrosine hydroxylase (TH)::IRES-Cre transgenic mice (EM:00254) 10. TH::Cre mice were individually housed following surgery to reduce damage to the permanent fiber optic implants. All mice were maintained on a 12/12-h light dark cycle (lights on 7:00 a.m. = Zeitgeber (ZT) 0, lights off at 7:00 p.m. = ZT 12) with food and water provided ad libitum. Cohorts of animals that underwent behavioural testing at night were maintained on a reverse light dark cycle (lights on at 7:00 p.m.). Adult male C57BL/6 mice (Jackson Laboratories) were used for chromatin immunoprecipitation assays. All animal use was conducted in accordance with the National Institute of Health guidelines and approved by the Institutional Animal Care and Use Committees of the University of Pittsburgh, UT Southwestern Medical Center, Duke University, and Stanford University.
Electrophysiology
Surgeries
Male ClockΔ19 and WT littermate controls (20–30 weeks old) were used for electrophysiological experiments. Mice were anesthetized with ketamine (100mg/kg) and xylazine (10mg/kg), placed in a stereotaxic device, and metal ground screws were secured to the cranium. A total of 14 tungsten microwires were arranged in bundle arrays of 4–10 wires (each wire separated by at least 250μm), and implanted to target the dorsal hippocampus and VTA. Implanted coordinates were as follows (all coordinates are measured from bregma): dorsal hippocampus, AP −2.3mm, ML +1.63mm, DV−1.3mm; VTA, AP −3.2mm, ML+0.3mm, DV −4.25mm. Implanted electrodes were anchored to ground screws using dental acrylic. After a two-week recovery period, mice were then separated into individual cages and their cage placed into the recording chamber. Following a two-day habituation period, neurophysiological data was recorded throughout the entire 24 hour light-dark cycle of each mouse.
Neuronal and LFP data acquisition
Neuronal activity was sorted online and recorded using the Multi-Neuron Acquisition Processor system (Plexon Inc, TX). Local field potentials (LFPs) were pre-amplified (500X), filtered (0.7–170 Hz), and digitized at 500 Hz using a Digital Acquisition card (National Instruments, Austin, TX) and a Multi-Neuron Acquisition Processor (Plexon, Dallas, TX). Bursting and firing rate analyses were performed using Neuroexplorer (Nex Technologies, Littleton, MA). Electrophysiological recordings were referenced to two ground screws located above the cerebellum and anterior cortex. VTA channels were thresholded to select for dopaminergic neurons based on standard physiological criteria including waveforms > 2ms in length and bursting6, 11, 12. Cells were sorted using an offline-sorting algorithm (Plexon, Dallas, TX) to confirm the quality of recorded cells. For firing analysis, data was collected during 60 sec epochs of REM sleep occurring during each of the 6 hr intervals: ZT 0-6, ZT 6-12, ZT 12-18, and ZT 18-24. As several mutant mice failed to display a 60 sec epoch of REM sleep during each of the four time windows, additional inclusion criteria were applied such that neurons had to display a minimum firing rate of 1.5Hz and at least 1 bursting episode during the 60 sec period. Burst events were initiated by a pair of spikes having an interspike interval of 80 ms and terminated by interspike intervals of 160 ms. Following initial analysis, we also performed within neuronal analysis where only neurons recorded during the first 6 h of the light and dark cycle were included.
Behavioural state identification
Behavioural states were identified using a two-dimensional state map as previously described13. The state map generated cluster separation based on the high amplitude theta (4–9Hz) and gamma (33–55Hz) oscillations characteristic of REM sleep, the absence of gamma oscillations and high amplitude delta (1–4Hz) characteristic of slow-wave sleep, and the high amplitude gamma and theta oscillations characteristic of waking. Behavioural state maps were generated from each of the 2–4 hippocampal LFPs acquired; the map that produced the best cluster separation was utilized for behavioural state identification.
Optrode Construction and Implantation
Two- to three-month old TH::Cre mice were anesthetized with isoflourane and placed in a stereotaxic device. Metal ground screws were secured to the skull above the cerebellum and anterior cranium. A total of 8 tungsten microwires were arranged in a bundle (each wire separated by at least 200μm), and implanted in the VTA as follows (AP −3.4mm, ML +0.3mm, and DV −4.25mm, relative to bregma). Full details of the procedures for electrode construction and surgical implantation have been previously described14. A mono fiberoptic cannula coupled to a 2.5mm metal ferrule (NA 0.22; 100μm [inner diameter], 125μm buffer [outer diameter], MFC_100/125-0.22, Doric Lenses, Quebec) was built directly into the microarray bundle prior to surgical implantation. The tip of the fiber was secured 500μm above the tip of the tungsten microwires such that the optical fiber lay just above the VTA following implantation. Implanted electrodes were anchored to ground screws using dental acrylic. Optogenetic stimulation experiments were initiated following a 25-day recovery period.
Optogenetics
Virus construction and packaging
Recombinant adeno-associated virus type 5(AAV5)-DIO-Ef1a-hChR2 (C128S/D156A)-eYFP vector and its control vector, AAV5-DIO-Ef1a-eYFP, were constructed by cloning DIO-Ef1a-hChR2 (C128S/D156A)-eYFP and DIO-Ef1a-eYFP, respectively, into an AAV backbone using Mlul and EcoR1 restriction sites. The resulting plasmid DNA was serotyped with AAV5coat proteins and packaged by the University of North Carolina viral vector core.
Light transmission calculations for optical fiber placement
Fiber optic placement was calculated to ensure light propagation throughout the entire extent of the VTA while maintaining adequate light power to achieve optimal opsin activation based on known SSFO photocurrent responses15. Fiber placement was determined by taking into account both the medial-lateral (ML) spread of light and the dorsal-ventral (DV) transmission of light. ML spread was calculated using trigonometry based on light emission from a single point source, where the half angle of divergence (θdiv) is given by:
where NA is the numerical aperture of the fiber (0.22) and ntissue is the refractive index of light as it travels through gray matter (1.36)16. To calculate distance, the fractional change in light power (mW) that occurs as light travels through a medium was taken into consideration. This is based on both the fractional loss of light that occurs with conical spread and light scattering17, 18. The corresponding power densities (mW/mm2) were then calculated for varying distances of fiber tip from viral placement through the entire DV extent of the VTA using the online calculator for light transmission (http://www.stanford.edu/group/dlab/cgi-bin/graph/chart.php). ML spread (radius of illumination) was then calculated using the most ventral distance within the VTA that achieved power densities required to activate SSFO.
Stereotaxic viral injection and optical fiber implantation
TH::Cre mice were anaesthetized with 1.5–3.0% isoflurane and kept on a self-regulating heating pad throughout the duration of the stereotactic surgery. Purified AAV5virus (viral titer = 4×1012 viral molecules/ml) was unilaterally injected into the VTA at AP −3.3, ML +0.44, DV −4.2 and −4.7 (relative to bregma) using a 5mm long 33-gauge metal beveled needle, facing medial. A 10μl Hamilton microsyringe (nanofil; WPI, Sarasota, FL) and its controller (Micro4; WPI, Sarasota, FL) delivered 1μl of virus at each DV coordinate at a rate of 0.1 μl/min. The needle was left in place for 10 min between injections and slowly withdrawn. Immediately following, a Doric chronically implantable optical fiber (NA=0.22, 200μm inner core, Doric Lenses Inc., Quebec, Canada) was cut to a length of ~5mm and unilaterally implanted over the VTA at −4.1mm DV. Light transmission through each fiber was measured before implantation and confirmed post-mortem. Only fibers with ≥80% transmission efficiency were used. Adhesive cement (C&B metabond, Parkell, Edgewood, NY) and cranioplastic cement (Dental cement, Stoelting, Wood Dale, IL) were used to secure the fiber optic implant to the skull and the incision was closed and sealed with tissue adhesive (Vetbond; Fisher, Pittsburgh, PA). All surgeries were performed using strict aseptic techniques. Mice were administered 0.5ml of 0.9% saline and Buprenex (0.1mg/kg) sub-cutaneously immediately following surgery. After recovery, mice were left undisturbed for 4 weeks to allow for sufficient viral expression prior to behavioural testing.
In vivo light delivery
For chronic stimulation, TH::Cre mice were individually placed into KinderScientific locomotor boxes (see Behavioural Assays for more details) and simultaneously received a single unilateral 5 sec pulse of blue light every 15 min for 1 hour between ZT 6-10 (daytime) or ZT 18-22 (night time) for 7 consecutive days (Figure 3e). A multi-mode fiber optic black-jacketed patchcord (NA 0.22, 200μm inner core, Doric Lenses Inc. Quebec, Canada) connected a 100mW 473nm diode-pumped solid-state laser or a 100mW 447nm diode laser (OEM Lasers Systems, East Lansing, MI) to permanently implanted fiber optics. Commutators (Doric Lenses Inc. Quebec, Canada) were used to reduce torque on patchcords that occurs with animal movement. A standard power meter (Thorlabs, Newton, NJ) was used to measure the emitted light power prior to fiber tethering to achieve a minimum of 3mW at fiber tip (corresponding power densities of 39-3mw/mm2 at VTA depths of 0.1–0.6m). Laser output was controlled using a Rigol pulse generator (DG1022, Rigol USA, Oakwood Village, OH).
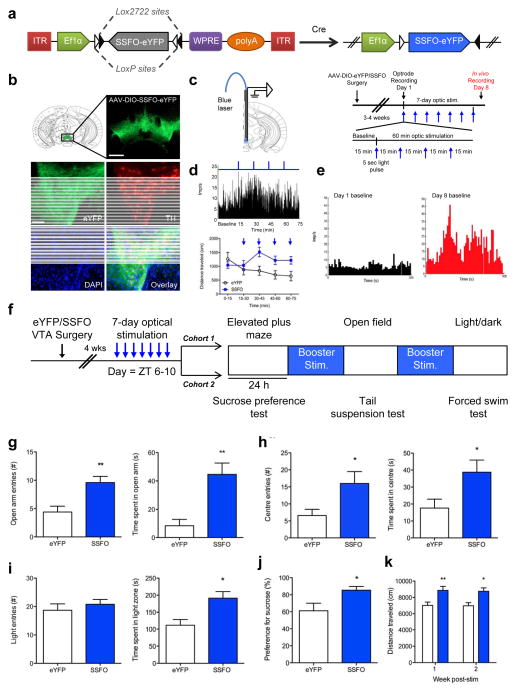
Figure 3. Effect of chronic VTA optical stimulation on manic-related behaviours.
a) Schematic of the Cre-dependent stable step function opsin (SSFO) viral construct tagged with enhanced yellow florescent protein (eYFP) used for long-term optical control of neural activity. Two incompatible LoxP sites surround a double-inverted open reading frame that ensures specificity of viral transduction and SSFO expression in cre-containing cells. (b) Expression of SSFO [AAV5-ChR2(C128S/D156A)-eYFP] in the ventral tegmental area (VTA) of a tyrosine hydroxylase (TH)::Cre mouse showing specificity of SSFO expression (green) in TH+ cells (red). TH::Cre mice lacking cre-recombinase (−/−) were used as a negative control and showed no viral expression in TH+ cells (data not shown). Scale bar: 4X=500μm; 20X=100μm. (c) Left panel: Schematic of unilateral optrode implantation for simultaneous optic stimulation and electrophysiological recording of VTA dopamine neurons. Right panel: Chronic stimulation paradigm: three-four weeks following surgery, mice received 7 days of optic stimulation. On each day, mice were given a 5 sec pulse of 473nm or 447nm light every 15 min for a total of 60 min. Electrophysiological recordings were conducted in a separate cohort of mice that underwent the chronic stimulation paradigm whereby VTA DA recordings were performed in awake behaving mice on Day 1 and again on Day 8 for comparison. (d) Top panel: Single neuron recording on Day 1 of the stimulation paradigm in an SSFO-mouse confirmed increased VTA dopamine neural firing during the 5 sec light pulse and a sustained increase in firing over the 60 min stimulation paradigm (blue lines atop graph represent single 5 sec pulses of blue light delivered 15 min apart). Bottom panel:1 hroptic stimulation of the VTA in SSFO-mice increased locomotor activity throughout the course of stimulation (main effect of treatment: F1,60=5.84, p=0.019), with a significant peak in activity occurring 15–30min (p<0.05) after the start of stimulation (first stimulation occurred at the 15 min mark)(e) Representative 5 min baseline (i.e. independent of concurrent optic stimulation) in vivo recording from an awake freely-moving TH::Cre mouse expressing SSFO confirmed that 7 days of optic stimulation (as depicted above in Figure 3c, right panel) increased the mean baseline firing rate of VTA dopamine neurons on Day 8 (red trace) compared to Day 1 (black trace). Firing rates were averaged across 5 sec time bins.(f) Chronic optical stimulation paradigm for behavioural testing: four weeks following viral-mediated delivery of SSFO or eYFP to the VTA, TH::Cre mice were optically stimulated with 473nm or 447nm light for 1hr/day for 7 days within the ZT 6-10 time window, followed by behavioural testing to assess anxiety-related (cohort 1) and depressive-related (cohort 2) behaviours. Behavioural tests were separated by 48 hours with a “booster” optical stimulation given between test days to sustain alterations in VTA dopamine neural activity. Note that mice did not receive optical stimulation on the day of behavioural testing.(g–i) TH::Cre mice that received chronic optical stimulation exhibited significantly decreased anxiety-related behaviour as evident by increased entries into, and time spent in,(g) the open arms of the elevated plus maze (entries: t8=3.49, p=0.008; time: t8=4.3, p=0.003), (h) centre of the open field (entries: t7=2.55, p=0.038; time: t7=2.42, p=0.046) and (i) light chamber of the light/dark box (entries: p>0.05; time: t6=3.12, p=0.021).(j) Chronic optical stimulation increased sucrose preference in mice expressing SSFO (t10=2.86, p=0.017).(k) Two-way repeated measures ANOVA revealed a main effect of group (F1,14=13.46, p=0.0025) whereby 7 days of chronic optic stimulation lead to a persistent and sustained increase in baseline locomotor activity at both 1 (p<0.01) and 2 weeks (p<0.05) after cessation of optogenetic stimulation. For behavioural experiments, cohort 1: n=5–6/treatment group; cohort 2, n=5–9/group.
Immunohistochemistry
Mice were perfused with 4% paraformaldehyde in 1XPBS (pH 7.4) and the brains transferred to a 30% sucrose solution. 40 μm sections were taken and processed for TH and DAPI. Briefly, floating sections were rinsed in 1XPBS to remove the cryoprotectant/fixative. Sections were then blocked in PBS++ (3% Normal Donkey Serum, 0.3% Triton-X in PBS) for 30 min and incubated with a chicken anti-tyrosine hydroxylase primary antibody (1:500, Aves Labs) overnight at 4°C on a rotary shaker. The following day, sections were rinsed in 1XPBS and incubated in a donkey anti-chicken secondary conjugated to Cy5 (1:500, Jackson ImmunoResearch Laboratories, West Grove, PA) for 3 hours at room temperature on a rotary shaker. Sections were then incubated with DAPI (1:50,000) for 20 min, washed, and mounted on microscope slides with PVA-DABCO. For cFOS/GFP double labeling shown in Supplementary Figure 3, 30 μm were blocked for 1 hour and incubated with mouse anti-tyrosine hydroxylase primary antibody (1:5000, Sigma, St. Louis, MO) and adjacent sections incubated with rabbit anti-GFP (1:20,000, Abcam, Cambridge, MA) and goat anti-cFOS (1:250, Santa Cruz Biotechnology, Dallas, TX) overnight at room temperature. The following day, sections were rinsed in 1XPBS and incubated in the species-appropriate secondary conjugated to various fluorophores (1:400, Invitrogen, Grand Island, NY) for 2 hours at room temperature on a rotary shaker. Sections were then washed in 1XPBS, mounted and cover slipped with DAPI mounting medium (Vectashield. Burlingame, CA).
Confocal microscopy and analysis
Confocal fluorescence images were acquired on a Leica TCS SP5 scanning laser microscope using a 40X/1.25NA oil immersion objective to visualize the fiber placements and viral-mediated expression. Serial stack images covering a depth of 10μm through multiple sections were acquired using equivalent settings. The Volocity image analysis software (Improvision/PerkinElmer, Waltham, MA) was used for identifying co-localization of DAPI, eYFP and TH staining. Imaging and analysis were performed blind to experimental conditions.
Drug Administration
Alpha-methyl-DL-p-tyrosine methyl ester hydrochloride (AMPT; Sigma-Aldrich, M3281) was prepared fresh in saline (0.9% NaCl, 10 mg/ml) and sterile filtered (0.2 μm) immediately prior to use. Adult male ClockΔ19 mice and WT littermates (24–36 weeks) were weighed and administered an intraperitoneal injection of AMPT (100 mg/kg) 60–90 min prior to behavioural testing. Control groups received an equal injection volume of sterile 0.9% NaCl. AMPT behavioural tests were separated by a minimum of 5 days. Daytime cohorts were administered AMPT within the time window of ZT 5-9; night time cohorts received AMPT between ZT 17-21.
Behavioural Assays
Behavioural testing occurred between ZT 6-10 (daytime cohorts) or ZT 18-22 (night time cohorts). Mice were habituated to the testing room for 30 minutes prior to testing, unless otherwise specified.
Locomotor Activity
Mice were individually placed into clear plexiglas chambers (Kinderscientific Smart Cage Rack System; field dimensions: 9.5″×18.0″) containing apparatus-embedded photobeams which measured horizontal locomotor activity for 60 min. Distance travelled was additionally measured in the open field and elevated plus maze (below) using Phenotracker video tracking software (www.tse-systems.com).
Elevated plus maze
The plus maze consisted of two plastic open arms perpendicular to two closed arms (arms: 30 X 5 cm) and was elevated from the ground at a height of 60cm. Mice were habituated to the testing room for 30 min and then placed into the center of the plus-maze facing a closed arm and were allowed to explore the maze for 5 min. Behaviour was video recorded and manually scored by a trained, blinded observer.
Open field
The open field consisted of a large 60 cm3 plastic box. Mice were habituated to the testing room for 30 min and then individually placed into a corner in the periphery of the open field box and allowed to explore the arena for 15 min. The centre was designated as a 20 cm2area in the centre of the open field. Behaviour was video recorded and a trained blinded observer scored both time spent in, and entries into, the centre of the arena.
Light/Dark Test
Anxiety-like behaviour was assessed with a light/dark test using the KinderScientific Smart Cage Rack System interfaced to a PC running MotorMonitor software. Each chamber was equipped with a black Plexiglas box on one side of the chamber; a small doorway allowed free exploration of both dark and light chambers. Mice were initially habituated to the dark chamber for 2 min and then allowed free exploration of both light and dark chambers for the remainder of the 10 min test.
Forced Swim Test
Mice underwent a single day 6 min forced swim test. Mice were habituated to the testing room for 30 min and then placed into a beaker filled with water (23–26°c; 20 cm depth). The time spent struggling, between 2–6 min, was measured by a blinded observer. Immobility was defined as all cessation of movement except those that were necessary to keep the animal afloat.
Tail Suspension Test
Depressive-like behaviour was measured in optically stimulated mice using a 6 min tail suspension test. Mice were suspended by gently taping the entire extent of their tail, from the base to the tip, to a horizontally positioned 10 ml pipette situated over a large beaker. An observer blinded to treatment group recorded the total time spent struggling between 2–6 min.
Sucrose Preference Test
Mice were habituated for three days to a bottle containing a 1% sucrose solution that was placed in their homecage adjacent to a standard water bottle. The amount of water and sucrose solution consumed was measured daily and the % preference for sucrose relative to total fluid intake was calculated. The position of the bottles was balanced across groups to avoid potential side (left vs. right) preferences. Daytime sucrose preference was measured on day four between ZT 5-11; night time preference was measured between ZT 16-23.
Gene Expression
Animals were sacrificed at six time points across the light-dark cycle (ZT 0, 4, 8, 12, 16, 20) and brains frozen on dry ice. VTA tissue was punched on a frozen stage from 300μM slices taken on a cryostat (Leica). RNA isolation and cDNA synthesis was carried out as previously described6. RNA was isolated using TRIzol reagent (Invitrogen) with remaining DNA digested using the DNA free system (Ambion, Austin, TX). cDNA was synthesized with an oligo dT primer and superscript III reverse transcriptase enzyme (Invitrogen). Real-time PCR was performed in duplicate on the ABI 7500 Real-Time PCR system (Applied Biosystems) using the Fast start SYBR green PCR master mix (Roche Applied Science, Indianapolis) with primers specific for TH, Gapdh, Clock, Per2 and Cryprochrome 2 (Cry2)(see Supplementary Table). Relative levels of mRNA were calculated by the 2 ΔΔCt method and normalized to corresponding Gapdh mRNA levels19. Ct values represent the mean of 2–5 biological replicates of the same reaction.
SDS-PAGE and Western Blots
Mice were placed in a plexiglas restrainer and euthanized by microwave irradiation aimed at the head (5 kW, 1.2 s, Murimachi Kikai Co., LTD. Tokyo, Japan). VTA tissue was dissected from 1–2 mm slices with a 16-gauge tissue punch and immediately frozen for later use. For protein extraction, tissue samples were homogenized by sonication on wet ice in a buffer containing 320mM sucrose, 5mM HEPES, phosphatase inhibitor cocktail I and II (Sigma, St. Louis, MO), protease inhibitor (Sigma, St. Louis, MO), 5% SDS, and 50mM NaF. Protein homogenate was spun at 12,000 RPM for 10 minutes at 4°C and the supernatant carefully removed. DC assays (Biorad, Hercules, CA) were performed to quantitate protein levels. Aliquots of sample were combined in Laemmli SDS sample buffer (Bio-World, Dublin, OH), and heated at 65°C for 20 minutes. Samples were loaded (10μg total protein per lane) and run on a pre-cast 4–15% Tris-glycine (TG) extended gel (Biorad, Hercules, CA) at 100V for ~ 90 minutes in 1XTGS buffer (Biorad, Hercules, CA). Proteins were transferred overnight at 4°C onto Immobilon PVDF membranes (Millipore, Bedford, MA) at 35V in 1XTG buffer. Membranes were re-wet briefly in a series of methanol, MilliQ water and 1XPBS and blocked in Odyssey Blocking Buffer (LI-COR Biosciences, Lincoln, NE) for 1 hour at room temperature (RT). Membranes were incubated overnight at RT with the following primary antibodies diluted in blocking buffer + 0.2% Tween20: mouse anti-TH (1:2000, Sigma, St. Louis, MO), rabbit anti-phospho-ser31 TH (1:400, Millipore, Bedford, MA), rabbit anti-phospho-ser40 TH (1:1000, Millipore, Bedford, MA) and mouse anti-GAPDH (1:20,000, Fitzgerald, Acton, MA). Blots were stripped between application of the two rabbit phospho-antibodies and re-probed (NewBlot Stripping buffer, LI-COR Biosciences, Lincoln, NE). Blots were washed in 1XPBS + 0.1% Tween20 and incubated for 1 hour at RT with infrared (IR) Dye 680 conjugated goat anti-rabbit and IRDye 800 conjugated goat anti-mouse secondary antibodies (1:5000, LI-COR Biosciences, Lincoln, NE) diluted in 0.2% Tween20 + 0.02% SDS. Blots were washed in 1XPBS + 0.1% Tween20 with a final wash in 1XPBS. Blots were scanned using the Odyssey Infrared Imaging System (LI-COR Biosciences, Lincoln, NE) interfaced to a PC running Odyssey 2.1 software for quantification. Values are expressed as a ratio to the corresponding GAPDH integrated intensity to control for potential discrepancies in amount of protein loaded. Values that were +/−1.5 standard deviations from the group mean were excluded from analyses.
Tissue L-DOPA High-Performance Liquid Chromatography Electrochemical Detection (HPLC-EC) Analysis
Mice were injected with the amino acid decarboxylase inhibitor NSD-1015 (Sigma, 100 mg/kg, i.p.) and euthanized by cervical dislocation 60 min later. The nucleus accumbens and dorsal striatum were punched from separate tissue slabs using 2 mm diameter circular tissue punches (nucleus accumbens: +1.7-1.0; dorsal striatum: +1.0-0.0 mm, relative to bregma). Tissue punches were weighed and then frozen and stored at −80°C until processing. Tissue was homogenized by sonication in 99 volumes (w/v) of ice-cold 100 mM HClO4. Homogenate was centrifuged for 20 min at 15,000 g, 4°C. Supernatants were recovered and passed through 0.2 μm filters and L-DOPA was quantified in the filtrates by HPLC-EC. The HPLC system consisted of a BASi (West Lafayette, IN) LC-4C detector coupled to a BASi LCEC radial flow cell. Flow was provided by a Shimadzu (Columbia, MD) LC-20AD solvent delivery module and preceded by an online degasser series 1100 from Agilent (Santa Clara, CA). The chromatograms were analyzed using PowerChrom software (eDAQ, Colorado Springs, CO). Monoamines in 10μl tissue filtrate were separated on a 1 x 100 mm UniJet microbore 5 μm C-8 column (BASi). The mobile phase consisted of 24 mM Na2HPO4, 3 mM sodium octyl sulfate, 27.4 mM citric acid, 107 μM EDTA and 18.75 % (v/v) MeOH, pH adjusted to 2.4 with NaOH. The flow was set at 50 μl/min and the potential at + 750 mV relative to an Ag/Cl reference electrode. Elution time for L-DOPA was 7 min.
Chromatin Immunoprecipitation (ChIP)
ChIP assays were performed according to the methods described previously20. Briefly, 2 mm VTA containing midbrain dissections from C57BL/6 mice or ClockΔ19 mutant mice and their wild type littermates were cross-linked with 1% formaldehyde for 15 minutes. Cross-linking was quenched with 2 M glycine for 5 minutes. The chromatin was sheared to 200–1000 bp fragments by 8 rounds of sonication and cleared with Protein A beads (Thermo Scientific, Rockford, IL). Between 60 and 100 μg of chromatin was used for each pull-down with 5–12 μg of each of the following rabbit primary antibodies: CLOCK-H276x (Santa Cruz Biotechnology, Santa Cruz, CA); acetylated H3, (no. 06-599, Millipore, Bedford, MA); IgG (no. 12-370, Millipore, Bedford, MA) and anti-phospho-Creb s133 (no. 06-519, Millipore, Bedford, MA). Antibody–chromatin complexes were immunoprecipitated with Protein A beads according to the manufacturer’s instructions. Following reverse cross-linking of input and precipitated samples, levels of protein binding at each promoter of interest were determined by measuring the amount of associated DNA by quantitative PCR (Applied Biosystems Prism 7700, Foster City, CA) with primer sets for the TH proximal and distal site promoters (sequences are listed in Supplementary Table 1). Input DNA and immunoprecipitated DNA were amplified in duplicate in the presence of SYBR Green on the ABI 7500 Real-Time PCR system (Applied Biosystems, Carlsbad, CA). Relative quantification of template DNA was performed using the fold enrichment method.
Luciferase Assays
Plasmid construction
Wild-type and E-box mutant TH luciferase reporter gene constructs:The TH promoter reporter genes were prepared by inserting promoter fragments (containing the putative transcriptional start site) into the pGL3-luc vector. An initial TH promoter fragment (~1 kb) was generated by PCR from mouse genomic DNA with primers: 5′-TCCTGAACCATTGCCTGAAGGAAG-3′ upstream and 5′-GGTCCCGAGTTCTGTCTCCA-3′ downstream. This fragment was then inserted into pGEM-T Easy vector (Promega cat. #A1360) and subsequently digested with EcoR1 to release the 1 kb promoter or SacI and SalI to liberate the 250 bp fragment. These were cloned into pGL3-luc to generate −1 kb and −250 bp reporter constructs. Mutagenic primers were designed to produce point mutations at the putative E-boxes of the TH-luc constructs. The proximal E-box sequence of the −250 TH luc construct was changed from CAGGTG to CCCGGG (this resulted in production of a diagnostic XmaI site), using complementary primers (coding sequence: 5′-CCCTTACATGGGGGCcccgggAGAATGGGGCTGCC-3′). The distal E-box sequence of the −1 kb TH luc construct was changed from CAGCTG to CCCGGG (this resulted in a production of a diagnostic XmaI site), using complementary primers (coding sequence: 5′-CCCTTACATGGGGGCcccgggAGAATGGGGCTGCC-3′). The manufacturer’s recommended protocol was followed for QuikChange II Site-Directed Mutagenesis (Stratagene cat. # 200555) using a 6.5 min extension time. WT and mutant E-box TH-luc constructs were confirmed by DNA sequencing. CLOCK and CLOCKΔ19 expression plasmids: Mouse Clock ORF was amplified by PCR from a cDNA kindly provided by J. Takahashi (UT Southwestern, Dallas, TX), cloned in the pSG5 vector (Stratagene) with a FLAG epitope sequences at the 5′ end. Clock-Δ19 cDNA was generated by subcloning of full-length Clock cDNA into pGEX-4-T3 (Pharmacia Biotech) for oligonucleotide-directed deletion of exon 19 with the U.S.E. Mutagenesis kit (Pharmacia Biotech). VP-16 CREB plasmids were kindly provided by E. Nestler (Mount Sinai School of Medicine, NY).
Cell culture and Luciferase assay
Rat pheochromocytoma (PC12) cells were cultured in Dulbeco’s modified Eagle’s Medium F-12 supplemented with 10% horse serum, 5% fetal bovine serum, and 1% penicillin/streptomycin in a 37°C, 5% CO2 incubator. The assay was performed using PC12 cells transfected by electroporation (BTX 360) with 5 μg of either construct. For CLOCK/CLOCKΔ19 experiments, rat pheochromocytoma (PC12) cells were maintained in Dulbecco’s modified Eagle’s medium F-12, supplemented with 10% horse serum, 5% fetal bovine serum, and 1% penicillin/streptomycin at 37 °C and 5% CO2. Cells were plated in 12-well, collagen-coated plates at 80% confluence for 24 hours before transfection. Cells were transfected by 3.5ul Lipofectamine LTX (Invitrogen) according to manufacturer’s instructions with 5μg TH-luc reporter plasmid, 1μ or 10μg CLOCK, 10μg CLOCKΔ19 or 5 μg CREB expression plasmid, or a combination, depending on the experimental group. Empty vector plasmid (pCDNA) was used to normalize total amounts of DNA. After transfection, the cells were grown on 12-well collagen-coated dishes for 24 hours before luciferase assays. Three days post-transfection, cells were collected in lysis buffer and centrifuged to clear cellular debris. Thirty microliters of lysate was combined with 140 μl of luciferase assay buffer. Luminescence was measured using an FLx-800 microplate fluorescence reader after an automated injection of 40 μL of 1 mM luciferin/well. Luciferase activity was normalized to total protein levels.
Statistical Analysis
Binary data were analyzed using an unpaired student’s t-test while comparisons of three or more group means were conducted using an analysis of variance (ANOVA) followed by a Bonferroni post-hoc test. Analyses over time were conducted using a two-way repeated measures-ANOVA followed by a Bonferroni post-hoc test to control for multiple comparisons. Interactions that reached significance or trended towards significance were followed up with post-hoc student’s t-tests and are indicated in the corresponding figure legends. All datasets were tested for Gaussian distribution prior to analysis using the F test. Adequate sample size for each test was calculated by power analyses based on previous behavioral and molecular work. Where applicable, animals were randomly assigned to treatment groups through manual randomization, although care was taken to ensure that littermates were equally distributed amongst treatment groups. For behavioral assays, measurements that were +/−1.5 standard deviations from the group mean were excluded. Analyses were conducted using the GraphPad Prism 5 statistical software for Windows. The CircWave v1.4 software (courtesy of Dr. Roelof Hut, http://www.euclock.org/) was used to analyze diurnal patterns of gene expression and protein levels. Data are presented as mean ± standard error of the mean (SEM) with a two-tailed p-value ≤ 0.05 considered statistically significant. *p≤0.05, **p≤0.01, ***p≤0.001, ****p≤0.0001. Significant treatment and time effects are denoted as #p≤0.05, ##p≤0.01, ####p≤0.0001.
Results
ClockΔ19 mice exhibit rapid mood-cycling across the light-dark cycle
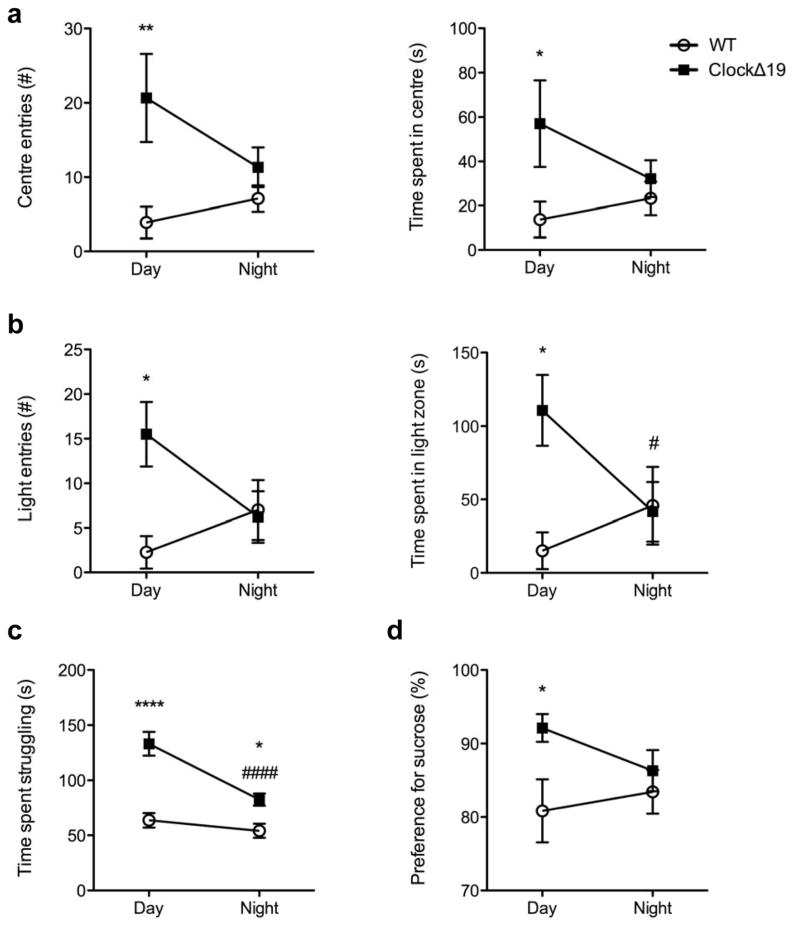
Previously we found that ClockΔ19 mice exhibited a robust manic-like phenotype when tested during the daytime3, however, we had yet to explore how behaviour may change over the light-dark cycle. Consistent with previous findings3, ClockΔ19 mice exhibited reduced anxiety-related behaviour during the daytime as indicated by increased exploration of the centre of the open field (center entries, p<0.01; center time, p<0.05; Figure 1a) and light chamber of the light/dark box (light entries, p<0.05; light time, p<0.05; Figure 1b). ClockΔ19 mice also exhibited an antidepressive-like phenotype in the forced swim test (FST; p<0.0001; Figure 1c) and sucrose-preference test (p<0.05;Figure 1d), two well-validated behavioural assays that measure motivational and anhedonic facets of depressive-like behavior, respectively. Quite surprisingly, however, the behavioural profile of ClockΔ19 mutant mice was time-dependent as manic-related behaviours were normalized to near wild-type (i.e. euthymic-like) levels during the night time, resulting in a cyclic pattern in mood-related states over the light-dark cycle (night, mutant vs. WT: open field, light/dark, and sucrose preference test, p’s>0.05; mutant, day vs. night: light/dark test, p<0.05; FST, p<0.0001; Figure 1a–d). To confirm that behavioral changes were not due to locomotor changes across the light-dark cycle (day vs. night: WT, p=0.04; mutants, p<0.05; Supplementary Figure 1a), the data were re-analyzed to control for locomotor differences in the open field and light/dark test and yielded a consistent pattern of results (day, mutant vs. WT: open field, p<0.01; light/dark, p<0.05; night, mutant vs. WT: p’s>0.05; Supplementary Figure 1b,c).
Figure 1. Behavioural mood-cycling across the light-dark cycle.
Anxiety-like (a, b) and depressive-like (c, d) behaviours were measured during the day (ZT 6-10) and night (ZT 18-22) in wild-type (WT) and ClockΔ19 mutant mice. (a) ClockΔ19 mutant mice exhibited a significant increase in entries into the centre of the open field during the day (main effect of genotype, F1,28=11.72, p=0.0019; post-hoc, p<0.01) that decreased to WT levels at night (genotype by time interaction, F1,28=4.19, p=0.05). ClockΔ19 mice also spent more time in the centre of the open field during the day (main effect of genotype, F1,28=6.17, p=0.02; post-hoc, p<0.05) with differences no longer apparent at night.(b) This daytime specific anxiolytic profile was also found in the light-dark test, where ClockΔ19 mice entered the light chamber significantly more than WT controls during the day (main effect of genotype, F1,28=4.23, p=0.049; post-hoc, p<0.05) but significantly decreased exploration to WT levels at night (genotype by time interaction, F1,28=5.35, p=0.03; post-hoc student’s t-test, p=0.06, ClockΔ19 day vs. night). ClockΔ19 mice also spent more time in the light chamber of the light/dark box during the day (main effect of genotype, F1,28=4.29, p=0.048; post-hoc, p<0.05), with a significant decrease to WT levels at night (genotype by time interaction, F1,28=5.03, p=0.03; post-hoc student’s t-test, p=0.049, ClockΔ19 day vs. night). (c) ClockΔ19 mice exhibited decreased depressive-like behaviors during the day as evidenced by increased struggling time in the forced swim test (main effect of genotype, F1,28=44.7, p<0.0001; post-hoc, p<0.0001) and (d) an increased preference for a 1% sucrose solution (main effect of genotype F1,27=5.46, p=0.027; post-hoc, p<0.05). Differences in depressive-like behaviours were no longer evident at night as ClockΔ19mutant mice significantly decreased struggling time to near WT levels in the forced swim test (c:genotype by time interaction, F1,28=7.84, p=0.0092; post-hoc, p<0.0001) and no longer showed a preference for sucrose relative to WT mice (d). Day and night cohorts, n=8–9/group.
Daytime-specific increase in VTA dopamine activity in ClockΔ19 mice
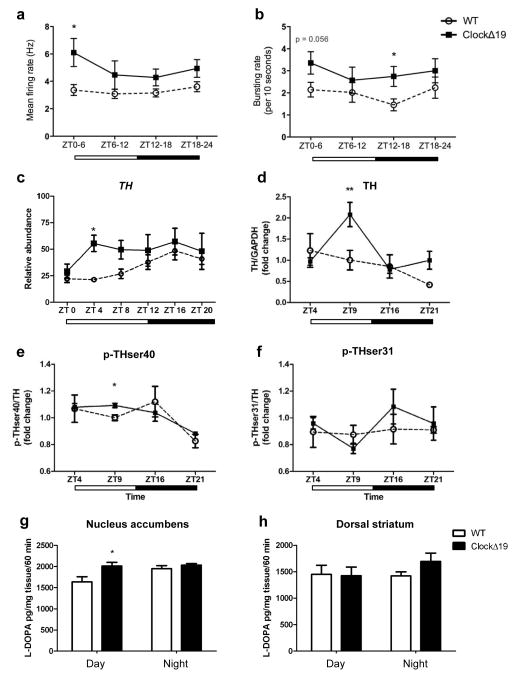
To determine how VTA dopamine cell firing changes over the course of the light-dark cycle in tandem with behavioural cycling, in vivo electrophysiological recordings were performed across the 24-hour cycle. VTA dopamine neurons (WT: n=14/14/16/12 and ClockΔ19 mice: n=14/14/17/12 for the number of dopaminergic neurons analyzed across each of the four time bins) were recorded during REM sleep to ensure locomotor activity would not influence dopaminergic firing (Supplemental Figure 2). In addition, neuronal firing was measured during REM sleep rather than anesthetization, as REM sleep is a normal physiological state21, 22. Consistent with previous reports23, ClockΔ19 mice spent significantly less time in REM sleep during the day compared to WT littermates (ZT 0-6, p<0.05; ZT 6-12, p<0.05) and displayed a highly disrupted diurnal rhythm in overall REM sleep (Supplemental Figure 2). Firing rates were elevated in ClockΔ19 mutants throughout the light-dark cycle, reaching significance at ZT 0-6 (p<0.05; Figure 2a). Bursting rates were also significantly elevated in ClockΔ19 mice with the largest effect at ZT 12-18 (p<0.05; Figure 2b). Since our data suggested that genotypic differences in bursting rate were largely driven by a reduced bursting rate in WT dopaminergic neurons, additional firing pattern analyses were performed. When bursting rates were compared in WT mice between ZT 0-6 versus ZT 12-18, a significant reduction in bursting rate was found during the night (ZT 0-6: 2.3±0.4bursts/10 seconds; ZT 12-18: 1.4±0.3bursts/10 seconds) suggesting a diurnal rhythm in activity. Conversely, no differences in bursting rates were observed in ClockΔ19 mice, indicating a loss of diurnal rhythmicity. When differences in firing rate between day and night were compared, ClockΔ19 mice displayed higher dopamine neuron firing during ZT 0-6 (6.5±1.1Hz) than ZT 12-18 (4.8±0.4Hz), with no differences detected in WT mice (ZT 0-6: 3.4±0.5Hz; ZT 12-18: 3.4±0.4Hz). This data demonstrates a significant daytime elevation in tonic dopaminergic activity and a loss in the diurnal rhythm of dopaminergic cell bursting in ClockΔ19 mice.
Figure 2. Time specific alterations in VTA dopaminergic activity in ClockΔ19 mice.
(a) Two-way ANOVA of firing rate found a significant genotype effect (F1,112=12.67, p=0.0006). Post-hoc tests revealed significant differences in the firing rate of dopaminergic neurons during the first 6 h of the light cycle and the last 6 h of the dark cycle (p<0.05 using student’s t-test; n=14/14/16/12 and 14/14/17/12 for the number of dopaminergic neurons analyzed in WT (n=8) and ClockΔ19 mice (n=9), respectively). (b) Two-way ANOVA of bursting rate found a significant genotype effect (F1,112=8.6, p=0.004) with post-hoc analyses revealing significant differences in the bursting rate of dopaminergic neurons during the first 6 h of the dark cycle (p<0.05, by student’s t-test; n=14/14/16/12 and 14/14/17/12 for the number of dopaminergic neurons analyzed in WT and ClockΔ19 mice, respectively). (c) Relative abundance of TH mRNA normalized to the expression of Gapdh. Two-way ANOVA revealed a significant genotype effect (F(1,45)=9.42, p=0.004) with a specific increase in TH expression at ZT 4 in ClockΔ19 mice (p< 0.05, n = 3–5 animals/genotype/time point). Diurnal variation was significant in wild-type (WT) mice (CircWave: F(2,27)=10.63, p=0.0004) but not in ClockΔ19 mutants (p>0.05).(d) A significant main effect of time was found for total TH (F(3,28)=5.34, p=0.005), with an increase in ClockΔ19 mouse TH levels at ZT 9 (p < 0.01). Diurnal variation was statistically significant in mutants (CircWave: F(2,17)=5.15, p=0.02). (e) There was a significant effect of time on phosphorylated TH (ser 40) protein (F(3,32)= 6.50, p=0.002), with ClockΔ19 mice exhibiting a specific increase in THser40 levels at ZT 9 (p<0.05, student’s t-test). Diurnal variation in THser40 was statistically significant in mutants (Circwave: F(2,17)=7.18, p=0.005). (f) No differences in phosphorylated TH (ser31) protein levels were found at any time point measured. Inset depicts average protein levels over 24 h. (e–f) Dopamine synthesis assay.(g) Dopamine synthesis was significantly increased in ClockΔ19 mutant mice as measured by L-Dopa in the nucleus accumbens after NSD-1015 administration during the light phase, at ZT 4 (t9 = 2.546, p=0.03). (h) Dopamine synthesis was unaltered in the dorsal striatum of ClockΔ19 mutants (p>0.05, n=5–8 per group; dark phase = ZT 16). White and dark bars below graph represent daytime and night time measurements, respectively.
As dopamine neural activity often parallels TH activity and dopamine synthesis24–26, we measured TH transcript levels, markers of TH activity, and dopamine synthesis in the VTA of WT and ClockΔ19 across the light-dark cycle. Q-PCR analysis revealed that WT mice exhibited a significant diurnal variation in TH mRNA expression with low levels during the day and higher expression at night (p=0.0004; Figure 2c). ClockΔ19 mutant mice exhibited an overall increase in TH levels, with the greatest increase in expression observed during the day (p< 0.05; Figure 2c).
To determine whether increased TH gene expression results in altered TH protein levels, total TH protein and two of its phosphorylated forms, p-TH at Ser 40 and p-TH at Ser 3125, were measured in the VTA at four time points (day: ZT 4, ZT 9; night: ZT 16, ZT 21). The pattern of diurnal TH protein expression in WT mice was consistent with previous findings, demonstrating peak levels of TH during the day that decreased at night27. ClockΔ19 mice exhibited a daytime-specific increase in total TH (p<0.01; Figure 2d) and p-THser40/TH at ZT 9 (p<0.05; Figure 2e), representing a time slightly lagging the increase in TH mRNA expression. No differences in p-THser31/TH protein were found (p>0.05; Figure 2f).
Dopamine synthesis assays were performed to determine if increased TH activity in ClockΔ19 mice leads to increased dopamine synthesis in the nucleus accumbens (NAc). HPLC analysis revealed a 20% increase in dopamine synthesis in the NAc of ClockΔ19 mice during the day with no difference detected at night (p=0.03; Figure 2g). No significant alterations were found in the dorsal striatum (p>0.05; Figure 2h), consistent with previous work showing no differences in TH protein levels in the substantia nigra of ClockΔ19 mice6.
Chronic optic stimulation of VTA dopaminergic activity induces manic-like behaviour
We next sought to determine whether daytime-specific increases in VTA dopamine firing are sufficient to reproduce manic-related behaviours. To accomplish this, we used optogenetic techniques to chronically manipulate VTA dopamine activity in TH::Cre mice. A chronic paradigm was employed to recapitulate the chronicity of increased daytime dopaminergic activity in ClockΔ19 mice. Due to the long-term nature of the stimulation paradigm employed, a double-mutated form of channel rhodopsin, the stable step-function opsin (SSFO), ChR2(C128S/D156A), was used15. The slower-off channel kinetics and enhanced sensitivity of SSFO offers the advantage that less frequent and intense stimulation is required to maintain a constant state of depolarization15. To restrict opsin expression to midbrain dopamine neurons, a double-floxed inverted open-reading-frame (DIO) Cre-dependent virus, tagged with enhanced yellow florescence protein (eYFP), was injected into the VTA of TH::Cre mice (Figure 3a,b). Specificity and efficiency of opsin expression, and accuracy of viral and optical fiber placement was confirmed in vivo (Supplementary Figures 3–6). cFOS immunohistochemistry was used as an indicator of neural activity and verified increased activity in SSFO-expressing VTA neurons following a single 5 sec pulse of blue light (Supplementary Figure 7). Optrode recordings, which combine simultaneous optogenetic stimulation with electrophysiological recordings in awake behaving mice, additionally confirmed that a single 5 sec pulse of blue light delivered every 15 minutes for one hour (Figure 3c) increased dopamine neural firing concurrent with increased locomotor activity over the course of this stimulation period, with a peak in both neural and locomotor activity occurring 15–30minutes following stimulation (p<0.05; Figure 3d). This acute stimulation-induced hyperactivity was most pronounced on day 7, following 6 days of stimulation (Supplemental Figure 8). Indeed, 6 successive days of stimulation (1 hr/day) were required before a change in baseline locomotor activity was observed (p<0.05; Supplementary Figure 8). Similarly, in vivo recordings confirmed that chronic optic stimulation led to a sustained increase in baseline VTA dopamine firing on Day 8, independent of any further stimulation (Figure 3e).
Once this timeline was established, a unique stimulation paradigm was used whereby mice received 7 successive days of 1hr/day optogenetic stimulation prior to behavioural testing. To control for the diurnal specificity of neural firing observed in ClockΔ19 mice, TH::Cre mice were optically stimulated during the day, when VTA dopamine cell firing is high in ClockΔ19 mice. Following completion of 7 days of optic stimulation, mice were tested in a behavioural battery to assess features of manic-like behaviour, including locomotor, anxiety-, and depressive-like behaviours (Figure 3f).
SSFO-mice explored the open arms of the EPM (entries, p=0.008; time, p=0.003; Figure 3g), centre of the open field arena (entries, p=0.038; time, p=0.046; Figure 3h) and light chamber of a light/dark box (time, p=0.021; Figure 3i) more than eYFP controls, indicating anxiolysis, or increased exploratory and risk-taking behaviour. As SSFO mice exhibited hyperactivity during behavioural testing (EPM, p<0.05; light/dark, p<0.01; Supplementary Figure 9), percent distance traveled was additionally used as a read-out of anxiety-like behaviour in order to avoid potential confounds of hyperactivity on locomotor-based exploratory measures; the results were consistent with those reported above. Both the anhedonic and motivational features of depressive-like behavior were assayed using the sucrose preference test, to measure reward behaviour, and the tail suspension and forced swim tests to measure motivation to escape an inescapable paradigm. SSFO-mice exhibited an increased preference for sucrose following 7 days of chronic stimulation (p=0.017; Figure 3j). Conversely, chronic enhancement of VTA dopamine activity had no impact on depressive-like measures in the tail suspension test or forced swim test (p’s>0.05; Supplementary Figure 9a,b). Notably, behavioral changes in response to chronic stimulation persisted for up to 2 weeks following cessation of optic stimulation (week 1, p<0.01; week 2, p<0.05; Figure 3k).
CLOCK binds the TH promoter in the VTA
To determine whether increased VTA dopamine levels and activity in ClockΔ19 mice result from CLOCK acting as a direct transcriptional regular of TH, we first characterized the transcript profile of Clock over the light-dark cycle. Interestingly, Clock gene expression was anti-phasic to TH expression in WT mice, with a peak occurring during the light phase, when TH expression is lowest, and a trough during the dark phase when TH expression was highest (Supplementary Figure 10a). To ensure that CLOCK maintains its function as a transcriptional activator in brain regions outside of the supra chiasmatic nucleus (SCN), two well-established downstream targets of SCN CLOCK transcriptional activation were measured in the VTA. As expected28, VTA Per2 and Cry2 expression were decreased in ClockΔ19 mice, confirming that CLOCK can act as a transcriptional activator at gene targets external to the SCN (Per2, p<0.001; Cry2, p<0.001; Supplementary Figure 10b,c).
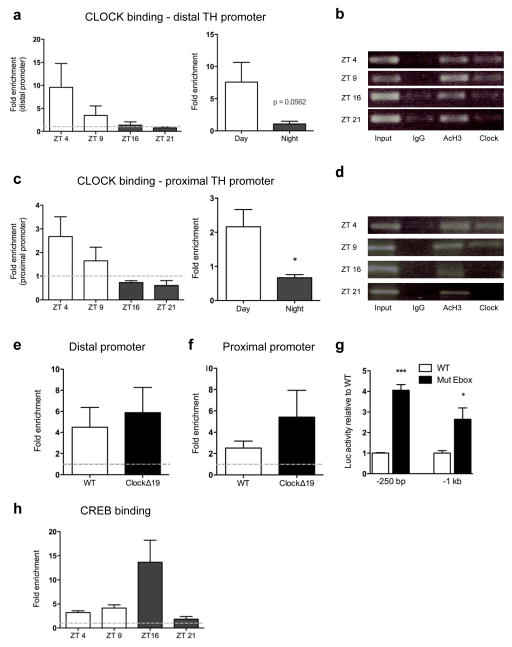
We next determined the ability of CLOCK to directly bind E-box elements in the TH promoter (CANNTG) by performing chromatin immunoprecipitation (ChIP) assays on VTA-containing midbrain tissue. Two primer sets were created to amplify distal and proximal regions of the promoter containing putative E-Boxes (Supplementary Table 1; Supplementary Figure 11). ChIP analyses were performed on C57BL/6 mice at 4 time points: daytime = ZT 4 and ZT 9 and night time = ZT 16 and ZT 21. CLOCK was present at both TH promoter sites during the day (distal: Figure 4a; proximal: Figure 4c), with no enrichment observed above background at night. CLOCK binding was then assessed in ClockΔ19 mutants during the day at ZT 4, when binding is expected based on the results in Figures 4a,c. Consistent with the WT data, the CLOCKΔ19 mutated protein was enriched at both TH promoter sites in ClockΔ19 mice (distal: Figure 4e; proximal: 4f). This data indicates that the mutated CLOCK protein is capable of binding DNA, however the missing exon 19 appears critical for conferring proper transcriptional regulation. To test the functional significance of CLOCK binding at the TH promoter, we interfered with CLOCK’s ability to properly bind TH. Luciferase assays were performed with TH-luc reporter plasmids containing either an intact or mutated E-box. Disruption of CLOCK binding through mutation of either E-box site significantly increased TH-luc reporter activity (proximal, p<0.0001 and distal, p<0.01; Figure 4g), demonstrating that the repression of TH transcription is E-box dependent.
Figure 4. CLOCK binding at the TH promoter.
(a) Fold enrichment, as calculated from Ct values, at distal promoter region across ZT time following chromatin immunoprecipitation with a CLOCK-specific antibody in C57BL/6 mice (n =3–4 per time point). (a, right) Comparison of CLOCK binding at the distal TH promoter during the light (averaged values of ZT 4 and ZT 9 from graph, left) versus dark phase (averaged values of ZT 16 and ZT 21 from graph, left) shows a strong trend toward a diurnal variation in promoter occupancy (t13=2.113, p=0.056). (b) Representative agarose gels of q-PCR products from ChIP assay for graphs in (a). (c) Binding at the proximal TH promoter. (c, right) Significant diurnal variation in CLOCK occupancy of the TH proximal promoter (t13=2.713, p=0.02).(d) Representative agarose gels of q-PCR products from ChIP assay for graphs in (b). (e,f) Fold enrichment at distal (e) and proximal promoter region (f) following ChIP with a CLOCK-specific antibody comparing ClockΔ19 mutants to wild-type (WT) controls; (n =4–6 per time point).(g) Relative luciferase activity in PC12 cells transfected with WT TH-luc constructs (250 bp or 1000 bp) or TH-luc constructs containing mutant E-boxes. Mutating the E-boxes significantly increased luciferase activity at the proximal (t16=11.30, p<0.0001) and distal site (t9=3.158, p<0.01).(h) Differential levels of p-CREB (s133) binding at the proximal TH promoter across ZT time with increased binding observed during the beginning of the dark phase (ZT 16) in C57BL/6 mice.
We hypothesized that phosphorylated cAMP Response Element Binding Protein (p-CREB) may compete with CLOCK for binding at the proximal TH promoter given the proximity of the CRE binding site upstream of the proximal CLOCK E-box binding site (Supplementary Figure 11) and the well-established role of CREB in the regulation of TH transcription29. Indeed, an increase in p-CREB binding was found at the TH proximal promoter site during the night relative to the day that represents an opposite pattern of binding from that of CLOCK at the TH promoter (Figure 4h). This suggests an antagonistic relationship between CLOCK and p-CREB in regulating TH transcription.
Daytime TH inhibition reverses manic-like behaviors
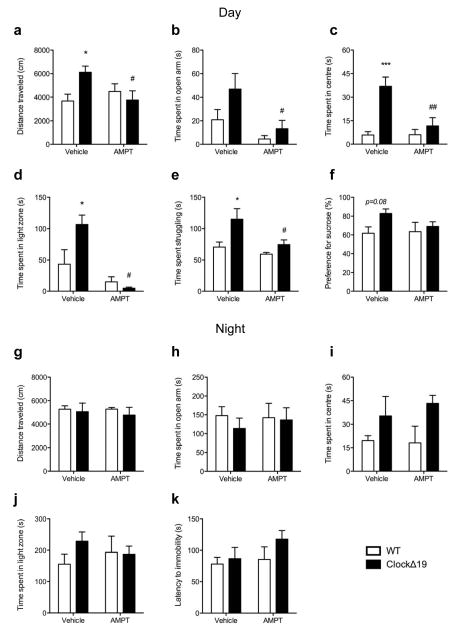
Finally, we sought to determine if daytime increases in TH activity are necessary to produce a switch to a manic-like state inClockΔ19 mice through direct pharmacological inhibition of TH activity. Mice were administered Alpha-methyl-DL-p-tyrosine methyl ester hydrochloride (AMPT) either during the day, at a time corresponding to peak levels of dopaminergic activity in ClockΔ19 mice, or during the dark phase when no difference in dopaminergic activity is observed. As expected, vehicle-treated ClockΔ19 mice exhibited a hyperactive phenotype (p<0.05; Figure 5a) and an overall anxiolytic behavioural profile compared to WT mice during the daytime as characterized by increased exploration of the open arms of the EPM (main effect of genotype, p=0.03; Figure 5b), centre of the open field (p<0.001; Figure 5c) and light chamber of the light/dark test (p<0.05;Figure 5d). ClockΔ19 mice also exhibited an anti-depressive phenotype during the daytime as demonstrated by increased time spent struggling in the FST (p<0.05; Figure 5e) and an increased preference for sucrose (main effect of genotype, p=0.08;Figure 5f). Importantly, all behavioural components of the ClockΔ19 mouse manic-like phenotype were reversed to WT levels with daytime AMPT treatment (p’s<0.05, vehicle vs. AMPT; Figure 5a–e). AMPT did not alter general locomotor activity (p>0.05; Supplementary Figure 12a,b) except in the light/dark test where it decreased overall distance traveled in ClockΔ19 mice (p<0.01; Supplementary Figure 12c). In this case, data were also plotted as %distance traveled and yielded similar results to those reported above (p<0.01; Supplementary Figure 12d). Conversely, AMPT had no behavioural effects when administered during the dark phase (p’s>0.05, vehicle vs. AMPT;Figure 6g–k and Supplementary Figure 12e–g), when ClockΔ19 mice were found to be in a euthymic-like state.
Figure 5. Daytime-specific tyrosine hydroxylase inhibition reverses manic-like behaviours.
ClockΔ19 mice and wild-type (WT) littermates received an intraperitoneal injection of either 0.9% saline (vehicle) or AMPT (100mg/kg) during the day (ZT 6-10) or night (ZT 18-22) and were tested for anxiety- and depressive-related behaviours 60–90 min later. Daytime cohorts (a–f): (a) Two-way ANOVA revealed a significant interaction of genotype and treatment (F1,20=6.66, p=0.01) on distance traveled in a novel environment where vehicle-treated ClockΔ19 mice exhibited hyperactivity (p<0.05) which decreased to WT levels following AMPT treatment (p<0.05). Vehicle-treated ClockΔ19 mice also showed the expected decrease in anxiety-like behaviour compared to vehicle-treated WT littermates as indicated by increased exploration of the anxiogenic (b) open arms of the elevated plus maze (main effect of genotype: F1,20=5.09, p=0.03), (c) centre of the open field (main effect of genotype: F1,15=16.22, p=0.001; post-hoc p<0.001), and (d) light chamber of the light/dark box (interaction between genotype and treatment: F1,15=6.12, p=0.03; post-hoc p<0.05 for vehicle treated groups) and displayed (e, f) decreased depressive-like behaviour in the forced swim test (e) as indicated by increased time spent struggling (F1,16=8.73, p=0.009; post-hoc p<0.05) and an increased preference for sucrose (f) in the sucrose preference test (genotype effect:F1,25=3.24, p=0.08). Importantly, AMPT treatment significantly reversed ClockΔ19 mice manic-behavioural features to WT levels (a–e). Main effect of treatment in (a) reported above; (b) F1,20=10.48, p=0.004, post-hoc p<0.05; (c) F1,15=7.6, p=0.01, post-hoc p<0.001; (d) F1,15=19.18, p=0.0005, post-hoc p<0.001; (e) F1,16=6.55, p=0.02, post-hoc p<0.05. (g–k) Night time cohorts. Vehicle-treated ClockΔ19 mice did not exhibit manic-related behavioural features when tested at night, nor did AMPT treatment effect behaviour in ClockΔ19 mice or WT littermates. Daytime cohorts, n=5–6/group; Daytime sucrose preference test and night time light/dark test, n=7/8; Night time cohorts, n=5–8/group.
Discussion
The current study demonstrates that disrupted function of the circadian protein, CLOCK, by the Δ19 mutation leads to rapid mood-cycling across the light-dark cycle. To the best of our knowledge, this is the first mouse model that spontaneously switches between two different mood-related states. This behavioural cycling between daytime manic-like behaviour and night time euthymic-like behaviour coincided with time-dependent changes in VTA dopamine neuronal firing and TH activity across the light-dark cycle. Specifically, changes in TH activity were determined by measuring the levels of transcript, markers of protein activity, and dopamine synthesis. Levels of phosphorylated ser40, and not ser31, were increased in ClockΔ19 mice. Phosphorylation at the serine 40 residue, and to a lesser extent, at the serine 31 residue of the TH protein increases TH activity and catecholamine synthesis in vivo24. Although phosphorylation at ser40 and ser31 occur in response to a multitude of acute stimuli30, sustained increases in phosphorylation at ser40 are indicative of chronic changes in TH activity31 and are known to parallel changes in dopamine synthesis. Furthermore, changes in TH expression and phosphorylated ser40 are known to correlate with dopamine neuron activity and depolarization24, 26. The increases in TH activity and dopamine synthesis found here coincided with the daytime increase in VTA dopamine neuronal firing observed in ClockΔ19 mice, suggesting that increased TH activity and dopamine synthesis may act as a precursor or consequence of increased neural firing.
Using a novel optogenetic stimulation paradigm, we demonstrate that chronic stimulation of daytime VTA dopaminergic neurons is sufficient to increase daytime exploratory and reward-related behaviours in WT mice. This behavioural profile is similar to that observed in ClockΔ19 mice, suggesting that chronically increased VTA dopamine neural firing may underlie specific components of the ClockΔ19 mouse manic-like behavioural phenotype. Indeed, tonic but not phasic21,22 activation of VTA-DA neurons in mice has been shown to produce an anxiolytic response32, which is consistent with current findings and suggests that changes to tonic DA firing underlie anxiety-like behavioral responses. Previous work from our group has found that a selective reduction in VTA neuronal firing in ClockΔ19 mice normalizes their anxiety-like, but not depressive-like behaviours7. Interestingly, chronic optic stimulation of VTA dopamine neurons had no impact on depressive-like behavior as measured in the tail suspension and forced swim tests. However, other studies have found that acute VTA optogenetic modulation decreases depressive-like behaviour induced by stressors33, 34. This highlights the complexity of the behaviors and neural circuits under investigation but may also reflect the properties of the different opsins used to modulate neural activity and their physiological effects on neural firing. For instance, the step-function opsins used in the current study do not drive synchronous trains of neural firing in response to light, as channel rhodopsins do, but rather enable step-like control of membrane potential by bringing neurons closer to action potential threshold35, 36. This creates an UP state and can be considered a more physiologically relevant approach to neural circuit manipulation36. Indeed, the chronic stimulation paradigm introduced here lead to a persistent and sustained change in behavior following cessation of all optogenetic stimulation suggesting that the stimulation paradigm altered the physiological properties of VTA dopamine neurons and/or downstream neural circuits without the need for additional circuit modulation. The plastic changes induced by this paradigm and the longevity of the effect will be an interesting avenue of investigation for future studies.
This is the first study to report that the circadian protein, CLOCK, can directly regulate TH. This finding is surprising given that most studies have reported that CLOCK acts as a transcriptional activator at CLOCK-controlled gene promoters, rather than as a suppressor37. Conversely, CREB (a known activator of TH expression) binding to the TH promoter occurred in an opposite diurnal pattern to CLOCK, suggesting an antagonistic relationship between CLOCK and CREB in TH regulation. Importantly, we show that the CLOCKΔ19 mutated protein is still capable of binding DNA. This integral finding has been confirmed in a recent study which demonstrated that the CLOCKΔ19 mutated protein binds DNA, albeit with lower affinity to E-box regulatory sites, resulting in reduced transcriptional efficiency38. In light of the current findings, this would suggest that the CLOCKΔ19 protein is a less effective transcriptional repressor of TH and provides an additional mechanistic explanation for the increased TH activity observed in ClockΔ19 mice. The importance of CLOCK in the regulation of TH is evolutionarily conserved as fruit flies with a mutation in the Clock gene also have an increase in TH expression39. Interestingly a recent study found that another circadian protein, REV-ERBα, also directly regulates the expression of TH in dopamine neurons and that inhibition of REV-ERBα can lead to a very similar manic-like profile and increased TH expression40. These results provide further confirmation of the important role of circadian genes in the regulation of dopamine synthesis and the precipitation of mania.
The importance of this increased TH activity to manic-like symptoms was demonstrated by the complete reversal of manic-related behaviours in ClockΔ19 mice following TH inhibition during the day. Both anxiety and depressive-like features were reversed in ClockΔ19 mice by daytime AMPT treatment whereas VTA optic stimulation in TH::Cre mice reproduced only the anxiolytic and rewarding features of the ClockΔ19 mouse manic-like phenotype. In this respect, AMPT-induced TH inhibition likely targets multiple brain regions, whereas optic stimulation specifically targeted the VTA. Such circuit-specific functional differences will be interesting to explore in future studies.
Conclusions
The current study uncovers a novel mechanism for regulation of TH by CLOCK and underscores the importance of normal patterns of dopaminergic activity in the proper regulation of mood-related behaviours. This has far-reaching implications for bipolar disorder and other psychiatric disorders, where the inability to regulate mood is a central defining feature. Given the paucity of effective treatment options for bipolar disorder41–43, the current findings represent a novel therapeutic avenue and extend to a multitude of diseases that involve dysfunction in limbic dopaminergic circuitry. Furthermore, the use of optogenetic technology with step function opsins opens the door for future studies to examine the effects of chronic stimulation of multiple neurotransmitter systems across various brain regions to determine the impact on complex behavioural phenotypes.
Supplementary Material
Acknowledgments
This work was supported by funding from the McKnight Foundation, the National Alliance for Research on Schizophrenia and Depression, the National Institute of Mental Health (MH082876), the National Institute on Drug Abuse (DA023988), and the National Institute of Neurological Disorders and Stroke (NS058339). We would like to thank Dr. Maisie Lo and members of the Deisseroth Lab for their generosity and assistance in performing the optogenetic experiments. We thank Joe Takahashi for the ClockΔ19 mice. The excellent technical assistance of Heather Buresch, Emily Webster, Edgardo Falcon, Elizabeth Gordon, and Ariel Ketcherside is greatly appreciated.
Footnotes
Author Contributions: M.M.S. designed experiments, performed and analyzed the optogenetic, western blotting, and behavioral studies, assisted with the electrophysiology experiments, and wrote the paper. S.S. performed the PCR and ChIP assays, collected tissue for western blotting, analyzed data, and contributed to writing of the paper. K.D. performed the electrophysiology experiments with assistance by S.K. and contributed to writing of the appropriate sections. P.K.P. performed immunohistochemistry and provided feedback on the manuscript. K.T. and M.R.W provided technical assistance and conceptual advice with optogenetic experiments along with K.D., and provided substantial feedback on the manuscript. R.A. provided technical assistance with the PCR and chromatin immunoprecipitation assays. J.F.E. and E.M.R. performed the luciferase assays. J.P.R.J. and M.C. provided dopamine synthesis data. C.A.M. was responsible for designing and supervising the experiments, providing conceptual guidance, and for editing of the manuscript.
Financial Disclosures/Conflict of Interests
All authors have no conflict of interest to disclose.
Supplementary information is available at Molecular Psychiatry’s website.
References
- 1.McClung CA. Circadian genes, rhythms and the biology of mood disorders. Pharmacol Ther. 2007;114(2):222–232. doi: 10.1016/j.pharmthera.2007.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Plante DT, Winkelman JW. Sleep disturbance in bipolar disorder: therapeutic implications. Am J Psychiatry. 2008;165(7):830–843. doi: 10.1176/appi.ajp.2008.08010077. [DOI] [PubMed] [Google Scholar]
- 3.Roybal K, Theobold D, Graham A, DiNieri JA, Russo SJ, Krishnan V, et al. Mania-like behavior induced by disruption of CLOCK. Proceedings of the National Academy of Sciences. 2007;104(15):6406–6411. doi: 10.1073/pnas.0609625104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gekakis N, Staknis D, Nguyen HB, Davis FC, Wilsbacher LD, King DP, et al. Role of the CLOCK Protein in the Mammalian Circadian Mechanism. Science. 1998;280(5369):1564–1569. doi: 10.1126/science.280.5369.1564. [DOI] [PubMed] [Google Scholar]
- 5.King DP, Vitaterna MH, Chang AM, Dove WF, Pinto LH, Turek FW, et al. The Mouse Clock Mutation Behaves as an Antimorph and Maps Within the W(19H) Deletion, Distal of Kit. Genetics. 1997;146(3):1049–1060. doi: 10.1093/genetics/146.3.1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McClung CA, Sidiropoulou K, Vitaterna M, Takahashi JS, White FJ, Cooper DC, et al. Regulation of dopaminergic transmission and cocaine reward by the Clock gene. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(26):9377–9381. doi: 10.1073/pnas.0503584102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coque L, Mukherjee S, Cao J-L, Spencer S, Marvin M, Falcon E, et al. Specific Role of VTA Dopamine Neuronal Firing Rates and Morphology in the Reversal of Anxiety-Related, but not Depression-Related Behavior in the Clock[Delta]19 Mouse Model of Mania. Neuropsychopharmacology. 2011 doi: 10.1038/npp.2011.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cousins DA, Butts K, Young AH. The role of dopamine in bipolar disorder. Bipolar Disord. 2009;11(8):787–806. doi: 10.1111/j.1399-5618.2009.00760.x. [DOI] [PubMed] [Google Scholar]
- 9.Mukherjee S, Coque L, Cao J-L, Kumar J, Chakravarty S, Asaithamby A, et al. Knockdown of Clock in the Ventral Tegmental Area Through RNA Interference Results in a Mixed State of Mania and Depression-Like Behavior. Biological Psychiatry. 2010;68 (6):503–511. doi: 10.1016/j.biopsych.2010.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tsai H-C, Zhang F, Adamantidis A, Stuber GD, Bonci A, de Lecea L, et al. Phasic Firing in Dopaminergic Neurons Is Sufficient for Behavioral Conditioning. Science. 2009;324(5930):1080–1084. doi: 10.1126/science.1168878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Grace AA, Bunney BS. Intracellular and extracellular electrophysiology of nigral dopaminergic neurons--3. Evidence for electrotonic coupling. Neuroscience. 1983;10(2):333–348. doi: 10.1016/0306-4522(83)90137-9. [DOI] [PubMed] [Google Scholar]
- 12.White FJ. Synaptic regulation of mesocorticolimbic dopamine neurons. Annu Rev Neurosci. 1996;19:405–436. doi: 10.1146/annurev.ne.19.030196.002201. [DOI] [PubMed] [Google Scholar]
- 13.Dzirasa K, Ribeiro S, Costa R, Santos LM, Lin SC, Grosmark A, et al. Dopaminergic Control of Sleep-Wake States. J Neurosci. 2006;26(41):10577–10589. doi: 10.1523/JNEUROSCI.1767-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dzirasa K, Fuentes R, Kumar S, Potes JM, Nicolelis MA. Chronic in vivo multi-circuit neurophysiological recordings in mice. Journal of neuroscience methods. 2011;195(1):36–46. doi: 10.1016/j.jneumeth.2010.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yizhar O, Fenno LE, Prigge M, Schneider F, Davidson TJ, O’Shea DJ, et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature. 2011;477(7363):171–178. doi: 10.1038/nature10360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mobley JaV-DT. Biomedical Photonics Handbook. CRC Press; Boca Raton, FL: 2003. Optical Properties of Tissue; pp. 2–76. [Google Scholar]
- 17.Aravanis AM, Wang LP, Zhang F, Meltzer LA, Mogri MZ, Schneider MB, et al. An optical neural interface: in vivo control of rodent motor cortex with integrated fiberoptic and optogenetic technology. J Neural Eng. 2007;4(3):S143–156. doi: 10.1088/1741-2560/4/3/S02. [DOI] [PubMed] [Google Scholar]
- 18.Tye KM, Prakash R, Kim SY, Fenno LE, Grosenick L, Zarabi H, et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature. 2011;471(7338):358–362. doi: 10.1038/nature09820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Maywood ES, Fraenkel E, McAllister CJ, Wood N, Reddy AB, Hastings MH, et al. Disruption of Peripheral Circadian Timekeeping in a Mouse Model of Huntington’s Disease and Its Restoration by Temporally Scheduled Feeding. The Journal of Neuroscience. 2010;30(30):10199–10204. doi: 10.1523/JNEUROSCI.1694-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tsankova NM, Kumar A, Nestler EJ. Histone Modifications at Gene Promoter Regions in Rat Hippocampus after Acute and Chronic Electroconvulsive Seizures. The Journal of Neuroscience. 2004;24(24):5603–5610. doi: 10.1523/JNEUROSCI.0589-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lena I, Parrot S, Deschaux O, Muffat-Joly S, Sauvinet V, Renaud B, et al. Variations in extracellular levels of dopamine, noradrenaline, glutamate, and aspartate across the sleep-wake cycle in the medial prefrontal cortex and nucleus accumbens of freely moving rats. J Neurosci Res. 2005;81(6):891–899. doi: 10.1002/jnr.20602. [DOI] [PubMed] [Google Scholar]
- 22.Maloney KJ, Mainville L, Jones BE. c-Fos expression in dopaminergic and GABAergic neurons of the ventral mesencephalic tegmentum after paradoxical sleep deprivation and recovery. Eur J Neurosci. 2002;15(4):774–778. doi: 10.1046/j.1460-9568.2002.01907.x. [DOI] [PubMed] [Google Scholar]
- 23.Naylor E, Bergmann BM, Krauski K, Zee PC, Takahashi JS, Vitaterna MH, et al. The Circadian Clock Mutation Alters Sleep Homeostasis in the Mouse. The Journal of Neuroscience. 2000;20(21):8138–8143. doi: 10.1523/JNEUROSCI.20-21-08138.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Haycock JW, Haycock DA. Tyrosine hydroxylase in rat brain dopaminergic nerve terminals. Multiple-site phosphorylation in vivo and in synaptosomes. J Biol Chem. 1991;266(9):5650–5657. [PubMed] [Google Scholar]
- 25.Kumer SC, Vrana KE. Intricate Regulation of Tyrosine Hydroxylase Activity and Gene Expression. Journal of Neurochemistry. 1996;67(2):443–462. doi: 10.1046/j.1471-4159.1996.67020443.x. [DOI] [PubMed] [Google Scholar]
- 26.Aumann TD, Egan K, Lim J, Boon WC, Bye CR, Chua HK, et al. Neuronal activity regulates expression of tyrosine hydroxylase in adult mouse substantia nigra pars compacta neurons. Journal of Neurochemistry. 2011;116(4):646–658. doi: 10.1111/j.1471-4159.2010.07151.x. [DOI] [PubMed] [Google Scholar]
- 27.Webb IC, Baltazar RM, Wang X, Pitchers KK, Coolen LM, Lehman MN. Diurnal Variations in Natural and Drug Reward, Mesolimbic Tyrosine Hydroxylase, and Clock Gene Expression in the Male Rat. Journal of Biological Rhythms. 2009;24(6):465–476. doi: 10.1177/0748730409346657. [DOI] [PubMed] [Google Scholar]
- 28.Lowrey PL, Takahashi JS. Genetics of circadian rhythms in Mammalian model organisms. Adv Genet. 2011;74:175–230. doi: 10.1016/B978-0-12-387690-4.00006-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lewis-Tuffin LJ, Quinn PG, Chikaraishi DM. Tyrosine hydroxylase transcription depends primarily on cAMP response element activity, regardless of the type of inducing stimulus. Mol Cell Neurosci. 2004;25(3):536–547. doi: 10.1016/j.mcn.2003.10.010. [DOI] [PubMed] [Google Scholar]
- 30.Dunkley PR, Bobrovskaya L, Graham ME, von Nagy-Felsobuki EI, Dickson PW. Tyrosine hydroxylase phosphorylation: regulation and consequences. J Neurochem. 2004;91(5):1025–1043. doi: 10.1111/j.1471-4159.2004.02797.x. [DOI] [PubMed] [Google Scholar]
- 31.Bobrovskaya L, Gilligan C, Bolster EK, Flaherty JJ, Dickson PW, Dunkley PR. Sustained phosphorylation of tyrosine hydroxylase at serine 40: a novel mechanism for maintenance of catecholamine synthesis. J Neurochem. 2007;100(2):479–489. doi: 10.1111/j.1471-4159.2006.04213.x. [DOI] [PubMed] [Google Scholar]
- 32.Kim TI, McCall JG, Jung YH, Huang X, Siuda ER, Li Y, et al. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science. 2013;340(6129):211–216. doi: 10.1126/science.1232437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tye KM, Mirzabekov JJ, Warden MR, Ferenczi EA, Tsai HC, Finkelstein J, et al. Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature. 2013;493(7433):537–541. doi: 10.1038/nature11740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chaudhury D, Walsh JJ, Friedman AK, Juarez B, Ku SM, Koo JW, et al. Rapid regulation of depression-related behaviours by control of midbrain dopamine neurons. Nature. 2013;493(7433):532–536. doi: 10.1038/nature11713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yizhar O, Fenno LE, Davidson TJ, Mogri M, Deisseroth K. Optogenetics in neural systems. Neuron. 2011;71(1):9–34. doi: 10.1016/j.neuron.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 36.Berndt A, Yizhar O, Gunaydin LA, Hegemann P, Deisseroth K. Bi-stable neural state switches. Nat Neurosci. 2009;12(2):229–234. doi: 10.1038/nn.2247. [DOI] [PubMed] [Google Scholar]
- 37.Gekakis N, Staknis D, Nguyen HB, Davis FC, Wilsbacher LD, King DP, et al. Role of the CLOCK protein in the mammalian circadian mechanism. Science. 1998;280(5369):1564–1569. doi: 10.1126/science.280.5369.1564. [DOI] [PubMed] [Google Scholar]
- 38.Shimomura K, Kumar V, Koike N, Kim TK, Chong J, Buhr ED, et al. Usf1, a suppressor of the circadian Clock mutant, reveals the nature of the DNA-binding of the CLOCK:BMAL1 complex in mice. Elife. 2013;2:e00426. doi: 10.7554/eLife.00426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kumar S, Chen D, Sehgal A. Dopamine acts through Cryptochrome to promote acute arousal in Drosophila. Genes Dev. 2012;26(11):1224–1234. doi: 10.1101/gad.186338.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chung S, Lee EJ, Yun S, Choe HK, Park SB, Son HJ, et al. Impact of Circadian Nuclear Receptor REV-ERBalpha on Midbrain Dopamine Production and Mood Regulation. Cell. 2014;157(4):858–868. doi: 10.1016/j.cell.2014.03.039. [DOI] [PubMed] [Google Scholar]
- 41.Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156–167. doi: 10.4088/JCP.09r05385gre. [DOI] [PubMed] [Google Scholar]
- 42.Sidor MM, MacQueen GM. An update on antidepressant use in bipolar depression. Curr Psychiatry Rep. 2012;14(6):696–704. doi: 10.1007/s11920-012-0323-6. [DOI] [PubMed] [Google Scholar]
- 43.Geddes JR, Miklowitz DJ. Treatment of bipolar disorder. Lancet. 2013;381(9878):1672–1682. doi: 10.1016/S0140-6736(13)60857-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.