Abstract
Background
It has been suggested that video-assisted (VATS) lobectomy is safer than open lobectomy in patients with compromised lung function, but data regarding this are limited. We assessed acute outcomes of VATS compared to open lobectomy in these high-risk patients using a systematic literature review and meta-analysis of data.
Methods
The databases PubMed and Scopus were searched for studies published between 2000 and 2013 that reported mortality and morbidity of VATS in high-risk lung cancer patients defined as having compromised pulmonary or cardiopulmonary function. Study selection, data collection and critical assessment of the included studies were performed according to the recommendations of the Cochrane Collaboration.
Results
Three case-control studies and three case series that included 330 VATS and 257 open patients were identified for inclusion. Operative mortality, overall morbidity and pulmonary morbidity were 2.5%, 39.3%, 26.2% in VATS patients and 7.8%, 57.5%, 45.5% in open lobectomy group, respectively. VATS lobectomy patients experienced significantly lower pulmonary morbidity (RR = 0.45; 95% CI, 0.30 to 0.67; p = 0.0001), somewhat reduced operative mortality (RR = 0.51; 95% CI, 0.24 to 1.06; p = 0.07), but no significant difference in overall morbidity (RR = 0.68; 95% CI, 0.41 to 1.14; p = 0.14).
Conclusion
The existing data suggest that VATS lobectomy is associated with lower risk for pulmonary morbidity compared with open lobectomy in lung cancer patients with compromised lung function.
Introduction
Up to 25% of patients with stage I non-small cell lung cancer (NSCLC) are considered ineligible for open lobectomy due to severe medical comorbidity [1]. Patients with compromised pulmonary function or cardiopulmonary reserve are considered high risk for postoperative complications and therefore more likely subjected to alternative treatment modalities, the outcomes of which are currently not as favorable as surgical resection [2–5]. Video-assisted (VATS) lobectomy has at least equal oncological efficacy and long-term outcomes in comparison to open lobectomy for early stage NSCLC [6–10]. Due to the theoretical advantage of preserved chest wall mechanics and the demonstrated advantage of less postoperative pain, VATS lobectomy is associated with lower postoperative morbidity compared to open lobectomy, and has been increasingly used since its introduction in early 1990s [5,11–15]. These benefits are believed to be greater for high-risk patients, defined as having compromised pulmonary function or cardiopulmonary reserve, and may broaden the applicability of curative lobectomy for this patient group [2,3,16].
To date, there is no published randomized controlled trial concerning VATS lobectomy specifically in NSCLC patients with compromised lung function. In large-scale retrospective studies reporting acute outcomes of VATS lobectomy, data for these high risk patients were not extractable [17–19]. The existing studies regarding this are limited by small sample size and variations in definitions and results, resulting in controversy over the clinical benefits of the minimally invasive approach compared to the open approach for this specific patient group [3,4,16,20–22]. We assessed the operative mortality and postoperative morbidity of VATS lobectomy for NSCLC patients with compromised lung function and compared them with open lobectomy using a systematic literature review and meta-analyses.
Materials and Methods
Methods of the present systematic review and meta-analysis were specified in advance and documented in a protocol.
Protocol for systematic review
A PICO-formatted matrix was developed to guide selection of appropriate search terms [23]. The population of interest (P) was physiologic high-risk NSCLC patients with compromised pulmonary function or cardiopulmonary reserve. Compromised pulmonary function was defined as predicted postoperative forced expiratory volume in the first second (FEV1) or diffusing capacity for carbon monoxide (DLCO) expressed as a percent predicted (ppoFEV1% or ppoDLCO%) ≤ 40. If ppoFEV1% or ppoDLCO% were not available, pulmonary function was considered compromised for preoperative FEV1% or DLCO% < 50 or FEV1 < 0.8 L. Compromised cardiopulmonary reserve was defined as peak oxygen consumption during exercise (VO2) < 40% predicted or < 12 mL·kg-1·min-1. The intervention (I) was VATS lobectomy. All studies reporting the mortality and morbidity following VATS lobectomy in patients with compromised pulmonary function or cardiopulmonary reserve were eligible for inclusion. The comparator (C) was similar patients undergoing open lobectomy. Primary outcomes (O) measured in the present systematic review were 1) operative mortality, defined as death during the hospitalization for lung resection or within 30 days of the operation; and 2) overall morbidity, defined as the occurrence of at least one major postoperative complication. The secondary outcomes were 1) pulmonary morbidity, defined as pneumonia, atelectasis requiring bronchoscopy, adult respiratory distress syndrome, air leak >5 days, initial ventilator support >24 hours, reintubation, and tracheostomy; and 2) cardiac morbidity, defined as acute myocardial infarction based on electrocardiographic or biochemical findings, congestive heart failure, and atrial or ventricular arrhythmia requiring intervention.
Search strategy and study selection
PubMed and Scopus were searched for studies published in English between 2000 and 2013. The potentially eligible studies were selected by combining the search results regarding the population of interest, intervention, comparator and outcome (Table 1). In order to include studies on VATS lobectomy that included NSCLC patients as a subgroup, search terms such as NSCLC, lung cancer were not used. The authors had collected six citations in the past, which address the topic being posed in the present systematic review [3,4,16,20–22]. All of these citations were included in the search results, verifying the precision and the validity of the search strategy.
Table 1. Search terms.
| Domain | Search terms | Boolean operator |
|---|---|---|
| Population of interest | Co-morbidity, co-morbidities, comorbidity, comorbidities, "pulmonary function", "lung function", "pulmonary function test", "pulmonary function tests", "lung function test", "lung function tests", "cardiopulmonary reserve". | OR |
| Intervention | Lobectomy, lobectomies, "lung resection", "lung resections", "pulmonary resection", "pulmonary resections", pneumonectomy a . | OR |
| Comparator | VATS, "minimally invasive thoracic surgery", "minimally invasive thoracic surgeries", "video-assisted thoracic surgery", "video-assisted thoracic surgeries", "video-assisted thoracoscopic surgery", "video-assisted thoracoscopic surgeries", " video-assisted thoracoscopic resection", "video-assisted thoracoscopic resections", thoracoscopic, endoscopic, "thoracic surgery, video-assisted", "minimally invasive surgery", "minimally invasive surgeries", "video-assisted surgery", "video-assisted surgeries", "video-assisted resection", "video-assisted resections", "minimally invasive resection", "minimally invasive resections". | OR |
| Outcome | Outcome, outcomes, complication, complications, treatment outcome | OR |
aMeSH major topic, only for PubMed.
After discarding the duplicates, the records were further selected according to the inclusion and exclusion criteria (Table 2) that were defined a priori. In addition, potentially eligible studies were also identified by reading the review articles and other publications that emerged from the search. The studies arising from the search were reviewed and selected by the two authors independently. Disagreements were resolved by consensus.
Table 2. Criteria for considering studies.
| Inclusion criteria | Exclusion criteria |
|---|---|
| The study reports the mortality and morbidity after VATS lobectomy for NSCLC regardless of stage | Review article |
| The study involves adult physiologic high risk patients | Duplicate report by the same institution |
| The technique of VATS lobectomy is consistent with the CALBG definition (anatomic lobectomy; individual ligation of hilar structures; 1 to 3 ports; no rib spreading; video monitor used for guidance; mediastinal/hilar nodal sampling or dissection) | Fewer than 10 VATS lobectomy cases in the study |
| The contribution of segmental resection or pneumonectomy cases is <10% |
VATS = video-assisted thoracic surgery, NSCLC = non-small cell lung cancer, CALGB = Cancer and Leukemia Group B.
Data collection and critical assessment
In accordance with the recommendation of the Cochrane Collaboration, a data collection form was designed and tested [24]. Using the data collection form, the characteristics, intention, methods and outcomes of interest in each intervention group were assessed. The authors also extracted data of interest indirectly from information presented in the text and tables of full-text articles. Original authors were not contacted with a request to provide data that were not extractable from the publications.
Rather than being summarized in an overall score, the quality of each selected study was evaluated regarding selection bias, performance bias, detection bias, attrition bias and reporting bias according to the recommendation of the Cochrane Collaboration [25]. The risk of bias for each domain was judged as ‘low risk’, ‘high risk', or ‘unclear risk’. In the present systematic review, 'unclear risk' indicates either lack of information or uncertainty over the potential for bias.
Statistical analysis
A meta-analysis was performed using Review Manager Version 5.0 (Cochrane Collaboration, Software Update, Oxford, United Kingdom). The risk ratio (RR) between VATS lobectomy and open lobectomy groups with 95% confidence intervals was used as a summary statistic for effect measures. The chi-squared test was used to assess heterogeneity between studies. I2 statistic was used to estimate the percentage of total variation across studies due to heterogeneity. I2 of more than 50% was considered substantial heterogeneity. A fixed-effects model was adopted if the level of heterogeneity was acceptable (p > 0.10, or p ≤ 0.10 but I2 ≤ 50%); otherwise, a random-effects model was adopted. Sensitivity analyses were performed to evaluate the robustness of the results of meta-analyses bases on study quality. When data were available, an intention-to-treat analysis was preferred to reduce the selection bias. Continuous data are presented as median and range. A p-value <0.05 was considered statistically significant.
Results
Description of studies
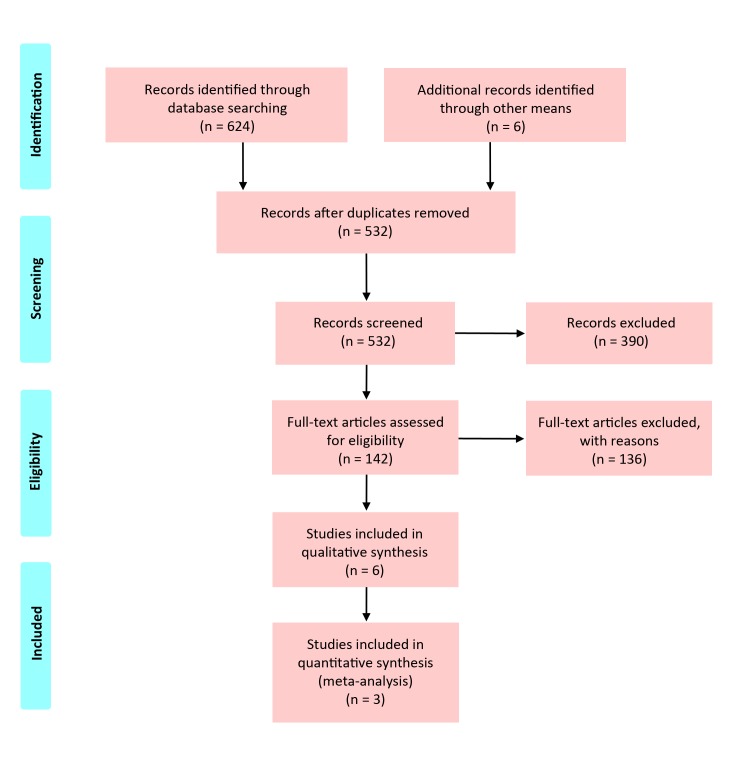
A total of 368 and 256 publications were identified in PubMed and Scopus databases, respectively. After discarding duplicates, 526 publications were screened based on title and abstract. Of those, 142 publications were further assessed for eligibility based on full-text articles. Six publications ultimately were included for data collection and critical assessment [3,4,16,20,22,26]. A list identifying reasons for exclusion of the 136 publications is available as supporting information (S1 Table [https://drive.google.com/folderview?id=0B8uBM3vcuLv8d2p1cEFBc3NGZU0&usp=sharing]). No additional eligible publication was identified after assessing review articles. The diagram of literature selection is depicted in Fig 1. The publications were excluded mainly because they did not involve the population of interest (n = 316, 50.6%) or the clinical data of the population of interest were not extractable (n = 88, 14.1%).
Fig 1. Diagram of literature selection.
The included publications consisted of three case-control studies and three case series. The characteristics of the studies are listed in Table 3. A total of 620 NSCLC patients with compromised pulmonary function were involved, of whom 330 underwent VATS lobectomy and 257 underwent open lobectomy. Most studies were published in the last four years [4,16,20,22,26]. In patients with initially attempted VATS lobectomy, the conversion rate was 4.4%, ranging from 0% to 33.3%. The proportion of patients with pathologic stage I NSCLC was 61.4%, ranging from 45.9% to 82.1%. In the study of Berry et al., data regarding pulmonary morbidity were extracted from the subgroup patients with FEV1% or DLCO% ≤ 45 as the population of interest [20]. The intervention group in the study of Lau and co-workers comprised 27 patients undergoing open segmentectomies by thoracotomy in addition to 18 patients undergoing VATS lobectomies [4].
Table 3. Characteristics of studies.
| Study | Year of publica-tion | Design | Country | Total number of patients | Number of VATS patients | Definition of compromised lung function |
|---|---|---|---|---|---|---|
| Berry et al.20 | 2010 | CCS | USA | 340 | 173 | FEV1% or DLCO% ≤ 60 |
| Lau et al.4 | 2010 | CCS | UK | 84 | 18 | ppoFEV1 <40 |
| Kachare et al.16 | 2011 | CCS | USA | 60 | 47 | ppoFEV1% or ppoDLCO%<40 |
| Garzon et al.3 | 2006 | CS | China | 25 | 13 | FEV1 <0.8L or FEV1% <50 |
| Paul et al.26 | 2013 | CS | USA | 50 | 18 | ppoDLCO% ≤40 |
| Wang et al.22 | 2013 | CS | China | 61 | 61 | FEV1% <50 |
CCS = case-control study, CS = case series, FEV1 = forced expiratory volume in the first second, DLCO = diffusing capacity of the lung for carbon monoxide, FEV1% = FEV1 as a percent predicted DLCO% = DLCO as a percent predicted, ppoFEV1% = predicted postoperative FEV1 expressed as a percent predicted, ppoDLCO% = predicted postoperative DLCO expressed as a percent predicted.
Risk of bias assessment
All included case-control studies suffer from the inherent bias of retrospective trials. Table 4 shows judgments about each type of bias for the included studies. As no cohort study or randomized, controlled trial was included, the assessment of attrition bias was omitted. Regarding selection bias, the data in all studies were collected based on a retrospective review of a clinical database and/or medical charts in a single center. The allocation to VATS or open approaches depended mainly on clinical decision making, the preference of surgeons, and patients’ preferences.
Table 4. Risk of bias summary.
| Study | Selection bias | Performance bias | Detection bias | Reporting bias |
|---|---|---|---|---|
| Berry et al.20 | High | High | High | Low |
| Lau et al.4 | High | High | High | High |
| Kachare et al.16 | High | High | High | High |
| Garzon et al.3 | High | High | High | High |
| Paul et al.26 | High | High | High | High |
| Wang et al.22 | High | High | High | High |
All case-control studies demonstrated significant imbalance in baseline characteristics [4,16,20]. In the study of Berry et al., the patients undergoing VATS lobectomy were significantly older and had more congestive heart failure, but had less advanced stage NSCLC, less previous thoracic surgery, and less preoperative chemotherapy or radiation compared to those undergoing open lobectomy [20]. In the study of Kachare et al., the patients in VATS group were also older and there was no report on co-morbidities [16]. In the study of Lau et al., the patients in the VATS group had more early-stage NSCLC, and there was no report on cardiac co-morbidities and previous malignancies [4]. Only the study of Kachare et al. presented the results of intention-to-treat analysis [16].
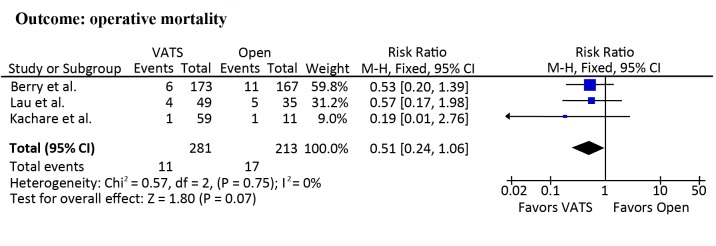
Operative mortality
Operative mortality was 2.5% (0% to 8.2%) and 7.8% (0% to 14.3%) in patients undergoing VATS lobectomy and open lobectomy, respectively. Meta-analyses demonstrated a trend towards reduced risk of operative mortality in patients undergoing VATS lobectomy (RR = 0.51; 95% CI 0.24 to 1.06; p = 0.07; Fig 2). The publication of Paul et al. was not included in this meta-analysis because there was no operative mortality in either the intervention or control groups [26]. In addition, the VATS group (n = 49) in the study of Lau et al. included 27 patients undergoing open segmentectomy [4]. The sensitivity analysis, from which the study of Lau et al. was removed, did not show a difference between the two approaches (RR = 0.48; 95% CI, 0.19 to 1.20; p = 0.12).
Fig 2. Meta-analyses of operative mortality.
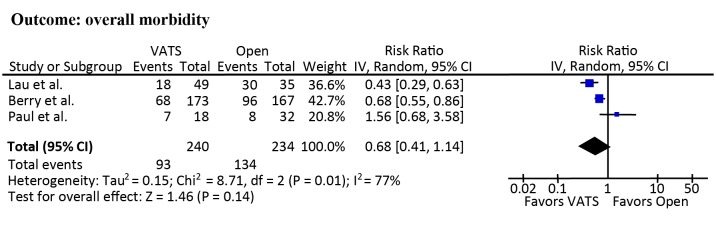
Overall morbidity
Other than the study of Kachare et al., data on overall morbidity could be extracted directly and indirectly from the included studies [16]. The overall morbidity was 39.3% (36.7% to 46.2%) and 57.5% (25.0% to 85.7%) in patients undergoing VATS lobectomy and open lobectomy, respectively. The overall morbidity in the study of Kachare et al. could not be accurately calculated because only the incidence of individual postoperative complications was reported [16]. Meta-analyses demonstrated the risk of overall morbidity between VATS and open lobectomy was comparable (RR = 0.68; 95% CI, 0.41 to 1.14; p = 0.14; Fig 3).
Fig 3. Meta-analyses of overall morbidity.
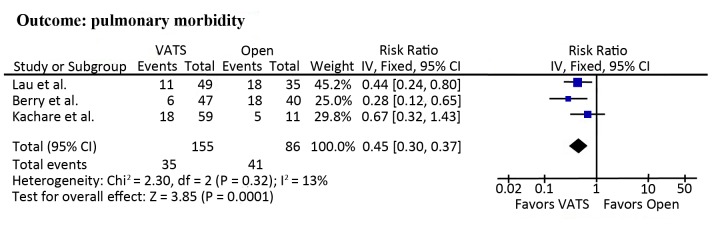
Pulmonary morbidity
All included studies reported pulmonary morbidity, which was 26.2% (12.8% to 36.1%) and 45.5% (45.0% to 51.4%) in patients undergoing VATS lobectomy and open lobectomy, respectively. Meta-analyses demonstrated a significantly lower risk of pulmonary morbidity in patients undergoing VATS lobectomy compared to those undergoing open lobectomy (RR = 0.45; 95% CI, 0.30 to 0.67; p = 0.0001; Fig 4). Sensitivity analysis was performed due to absence of a clear definition of pulmonary morbidity in the study of Lau et al. [4]. However, the results remained highly significant (RR = 0.45; 95% CI, 0.26 to 0.79; p = 0.005).
Fig 4. Meta-analyses of pulmonary morbidity.
Cardiac morbidity
Only one case-control study and two case series reported cardiac morbidity [16,21,22]. While no myocardial infarction occurred in the two case series, the incidence of myocardial infarction was 1.7% and 9.1% in patients undergoing VATS lobectomy and open lobectomy, respectively, in the case-control study of Kachare et al. [16]. The incidence of atrial fibrillation was 9.0% (4.9% to 12.0%) and 8.6% (0% to 17.1%) in patients undergoing VATS lobectomy and open lobectomy, respectively. A meta-analysis was not performed because comparisons were only available in two studies.
Discussion
Despite the advance in the non-surgical therapy modalities, lobectomy remains the therapy of choice for early-stage lung cancer due to the favorable oncological outcomes and survival rates [12,27]. In the last decade VATS lobectomy has been increasingly adopted for treatment of early-stage NSCLC and has achieved at least equal oncological efficacy and long-term outcomes in comparison to open lobectomy [6–10]. An ample body of research demonstrates that VATS lobectomy is associated with better preservation of lung function in the initial postoperative period and fewer pulmonary complications [5,11,14–16,22,28]. On the other hand, a substantial proportion of early-stage lung cancer patients is of questionable eligibility for open lobectomy according to conventional risk assessment due to compromised pulmonary function or cardiopulmonary reserve [1–5]. In this context, the clinical benefits of VATS lobectomy may permit a safe anatomic resection in high-risk NSCLC patients with compromised pulmonary function or cardiopulmonary reserve, who traditionally would have been offered non-surgical therapy [29,30]. However, there is insufficient information about the potential advantages of VATS lobectomy for this specific patient group.
In the present systematic review we assessed the operative mortality and postoperative morbidity of VATS lobectomy for high-risk NSCLC patients based on the available published data. A total of three case-control studies and three case series published between 2000 and 2013 were included after searching the most comprehensive medical databases. A multitude of randomized clinical trials and large-scale retrospective studies on VATS lobectomy in NSCLC patients exist for this period [7,18,19,31–33]. However, high risk patients were either not involved, or the data for this special patient group were not extractable. In a case-control study based on the STS General Thoracic Database, Ceppa et al. analyzed the clinical data of 12,970 patients who underwent an anatomic pulmonary resection (either lobectomy or segmentectomy) by either thoracotomy (n = 8439) or VATS (n = 4531) between 2000 and 2010 [17]. They found that thoracotomy was associated with markedly increased pulmonary complications in patients with impaired pulmonary function when compared with VATS patients. Besides the significant baseline imbalances as one of the major limitations, the exact number of patients with impaired pulmonary function, which was defined as predicted FEV1% < 60, was not extractable in this study.
As one of our major findings, the risk for pulmonary morbidity after VATS lobectomy in high-risk patients was less than half of that in their counterparts undergoing open lobectomy. In the last decade, a number of randomized clinical trials and large propensity-matched studies have shown that VATS lobectomy for early-stage NSCLC resulted in lower pulmonary morbidity compared to the open approach [7,18,19,31–33]. The reasons behind these advantages may be related to less chest wall trauma, reduced acute postoperative pain and earlier removal of chest tube, resulting in better preservation and faster recovery of pulmonary function after VATS lobectomy [5,11,14,15,28]. Indeed, better preserved pulmonary function in the immediate postoperative phase has been found to be strongly associated with lower pulmonary morbidity after lobectomy [34]. Moreover, VATS lobectomy enables improved deep breathing, early expectoration and ambulation in the postoperative period, which contribute to a decreased risk for pulmonary morbidity [16,22].
Due to advances in perioperative care in the last decade, operative mortality in high-risk patients has decreased after VATS and open lobectomy. Our meta-analyses did not demonstrate advantages of VATS lobectomy in high-risk patients in this regard. This finding might be explained by the limited patient numbers involved in the present systematic review and meta-analysis, and is similar to findings previously reported in a meta-analysis of outcomes for lobectomy patients in all risk levels [35].
Currently, non-surgical treatments including stereotactic body radiotherapy and radiofrequency ablation are increasingly employed in high-risk NSCLC patients due to low procedural mortality and morbidity [12,36]. However, accumulating evidence indicates that these treatment modalities are associated with increased risk for involved lobe and regional recurrence compared with lobectomy [1]. In contrast, concerns regarding the oncologic efficacy and long-term outcome of VATS lobectomy for early-stage NSCLC have not been supported by evidence in the recent literature [10,37,38]. In a prospective randomized clinical trial a total of 100 patients with clinical stage IA NSCLC underwent either VATS lobectomy or open lobectomy [37]. The 3- and 5-year survival rates as well as incidence of recurrences of VATS group were found equivalent to that of open group. Recently Su and colleagues performed a secondary analysis of clinical data in 1,018 patients enrolled in a large-scale multicenter, randomized trial (ACOSOG Z0030), which was conducted to determine the long-term clinical outcomes of patients undergoing surgical treatment for early stage NSCLC [10]. They found no difference in overall survival, disease-free survival, survival based on pattern of recurrence or time to locoregional recurrence between the VATS and open lobectomy groups during a median follow-up period of 6.7 years.
The authors acknowledge the limitations of the present systematic review. Besides the inherent bias of the retrospective studies and relatively small number of involved patients, the included case-control studies demonstrated significant imbalances in baseline characteristics. Also of concern is the fact that the definition of compromised pulmonary function varied in the included studies, resulting in increased heterogeneity of the population of interest. In addition, intention-to-treat analysis of the acute outcome of VATS lobectomy was reported only in one out of six included studies [16]. In the remaining studies, patients intended for VATS lobectomy but converted to open thoracotomy were included in the open lobectomy group, resulting in reduced validity of the pooled outcomes and increased bias in favor of VATS lobectomy [7,39]. Despite the imperfect data of the included studies, the results of the present systematic review and meta-analysis are intriguing and suggestive of a potential benefit of VATS lobectomy in high-risk patients.
Conclusions
The existing clinical studies suggest that VATS lobectomy is associated with a lower incidence of pulmonary morbidity compared to open lobectomy in high risk patients. Our investigation encourages careful consideration of VATS lobectomy for early-stage NSCLC in high-risk patients. Prospective comparative trials are warranted for further evaluation of the potential clinical benefits of VATS lobectomy in this patient population.
Supporting Information
(DOC)
(DOCX)
(DOCX)
(DOC)
Acknowledgments
We acknowledge the kind contributions of Debra Werner in developing the search terms and strategy.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
No funding was required for this study.
References
- 1. Donington J, Ferguson M, Mazzone P, Handy J Jr., Schuchert M, Fernando H, et al. American College of Chest Physicians and Society of Thoracic Surgeons consensus statement for evaluation and management for high-risk patients with stage I non-small cell lung cancer. Chest. 2012:142:1620–35. 10.1378/chest.12-0790 [DOI] [PubMed] [Google Scholar]
- 2. Demmy TL, Curtis JJ. Minimally invasive lobectomy directed toward frail and high-risk patients: a case-control study. Ann Thorac Surg. 1999;68:194–200. [DOI] [PubMed] [Google Scholar]
- 3. Garzon JC, Ng CS, Sihoe AD, Manlulu AV, Wong RH, Lee WT, et al. Video-assisted thoracic surgery pulmonary resection for lung cancer in patients with poor lung function. Ann Thorac Surg. 2006;81:1996–2003. [DOI] [PubMed] [Google Scholar]
- 4. Lau KK, Martin-Ucar AE, Nakas A, Waller DA. Lung cancer surgery in the breathless patient—the benefits of avoiding the gold standard. Eur J Cardiothorac Surg. 2010;38:6–13. 10.1016/j.ejcts.2010.01.043 [DOI] [PubMed] [Google Scholar]
- 5. Seok Y, Jheon S, Cho S. Serial changes in pulmonary function after video-assisted thoracic surgery lobectomy in lung cancer patients. Thorac Cardiovasc Surg. 2014;62:133–9. 10.1055/s-0033-1343980 [DOI] [PubMed] [Google Scholar]
- 6. D'Amico TA, Niland J, Mamet R, Zornosa C, Dexter EU, Onaitis MW. Efficacy of mediastinal lymph node dissection during lobectomy for lung cancer by thoracoscopy and thoracotomy. Ann Thorac Surg. 2011;92:226–32. 10.1016/j.athoracsur.2011.03.134 [DOI] [PubMed] [Google Scholar]
- 7. Flores RM, Park BJ, Dycoco J, Aronova A, Hirth Y, Rizk NP, et al. Lobectomy by video-assisted thoracic surgery (VATS) versus thoracotomy for lung cancer. J Thorac Cardiovasc Surg. 2009;138:11–8. 10.1016/j.jtcvs.2009.03.030 [DOI] [PubMed] [Google Scholar]
- 8. McKenna RJ Jr., Wolf RK, Brenner M, Fischel RJ, Wurnig P. Is lobectomy by video-assisted thoracic surgery an adequate cancer operation? Ann Thorac Surg. 1998;66:1903–8. [DOI] [PubMed] [Google Scholar]
- 9. Roviaro G, Varoli F, Vergani C, Nucca O, Maciocco M, Grignani F. Long-term survival after videothoracoscopic lobectomy for stage I lung cancer. Chest. 2004;126:725–32. [DOI] [PubMed] [Google Scholar]
- 10. Su S, Scott WJ, Allen MS, Darling GE, Decker PA, McKenna RJ, et al. Patterns of survival and recurrence after surgical treatment of early stage non-small cell lung carcinoma in the ACOSOG Z0030 (ALLIANCE) trial. J Thorac Cardiovasc Surg. 2014;147:747–53. 10.1016/j.jtcvs.2013.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Endoh H, Tanaka S, Yajima T, Ito T, Tajima K, Shitara Y, et al. Pulmonary function after pulmonary resection by posterior thoracotomy, anterior thoracotomy or video-assisted surgery. Eur J Cardiothorac Surg. 2010;37:1209–14. 10.1016/j.ejcts.2009.11.016 [DOI] [PubMed] [Google Scholar]
- 12. Howington JA, Blum MG, Chang AC, Balekian AA, Murthy SC. Treatment of stage I and II non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd Ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143:e278S–313S. 10.1378/chest.12-2359 [DOI] [PubMed] [Google Scholar]
- 13. Lewis RJ, Caccavale RJ, Bocage JP, Widmann MD. Video-assisted thoracic surgical non-rib spreading simultaneously stapled lobectomy: a more patient-friendly oncologic resection. Chest. 1999;116:1119–24. [DOI] [PubMed] [Google Scholar]
- 14. Nagahiro I, Andou A, Aoe M, Sano Y, Date H, Shimizu N Pulmonary function, postoperative pain, and serum cytokine level after lobectomy: a comparison of VATS and conventional procedure. Ann Thorac Surg. 2001;72:362–5. [DOI] [PubMed] [Google Scholar]
- 15. Ng CS, Lee TW, Wan S, Wan IY, Sihoe AD, Arifi AA, et al. Thoracotomy is associated with significantly more profound suppression in lymphocytes and natural killer cells than video-assisted thoracic surgery following major lung resections for cancer. J Invest Surg. 2005;18:81–8. [DOI] [PubMed] [Google Scholar]
- 16. Kachare S, Dexter EU, Nwogu C, Demmy TL, Yendamuri S. Perioperative outcomes of thoracoscopic anatomic resections in patients with limited pulmonary reserve. J Thorac Cardiovasc Surg. 2011;141:459–62. 10.1016/j.jtcvs.2010.05.051 [DOI] [PubMed] [Google Scholar]
- 17. Ceppa DP, Kosinski AS, Berry MF, Tong BC, Harpole DH, Mitchell JD, et al. Thoracoscopic lobectomy has increasing benefit in patients with poor pulmonary function: A Society of Thoracic Surgeons Database analysis. Ann Surg. 2012;256:487–93. 10.1097/SLA.0b013e318265819c [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Paul S, Altorki NK, Sheng S, Lee PC, Harpole DH, Onaitis MW, et al. Thoracoscopic lobectomy is associated with lower morbidity than open lobectomy: a propensity-matched analysis from the STS Database. J Thorac Cardiovasc Surg. 2010;139:366–78. 10.1016/j.jtcvs.2009.08.026 [DOI] [PubMed] [Google Scholar]
- 19. Villamizar NR, Darrabie MD, Burfeind WR, Petersen RP, Onaitis MW, Toloza E, et al. Thoracoscopic lobectomy is associated with lower morbidity compared with thoracotomy. J Thorac Cardiovasc Surg. 2009;138:419–25. 10.1016/j.jtcvs.2009.04.026 [DOI] [PubMed] [Google Scholar]
- 20. Berry MF, Villamizar-Ortiz NR, Tong BC, Burfeind WR Jr, Harpole DH, D’Amico TA, et al. Pulmonary function tests do not predict pulmonary complications after thoracoscopic lobectomy. Ann Thorac Surg. 2010;89:1044–52. 10.1016/j.athoracsur.2009.12.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Martin-Ucar AE, Nakas A, Pilling JE, West KJ, Waller DA. A case-matched study of anatomical segmentectomy versus lobectomy for stage I lung cancer in high-risk patients. Eur J Cardio-thoracic Surg. 2005;27:675–9. [DOI] [PubMed] [Google Scholar]
- 22. Wang W, Xu Z, Xiong X, Yin W, Xu X, Shao W, et al. Video-assisted thoracoscopic lobectomy for non-small cell lung cancer in patients with severe chronic obstructive pulmonary disease. J Thorac Dis. 2013;5:S253–9. 10.3978/j.issn.2072-1439.2013.08.23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Guyatt GH, Oxman AD, Kunz R, Atkins D, Brozek J, Vist G, et al. GRADE guidelines: 2. Framing the question and deciding on important outcomes. J Clin Epidemiol. 2011;64:395–400. 10.1016/j.jclinepi.2010.09.012 [DOI] [PubMed] [Google Scholar]
- 24. Higgins JPT, Deeks JJ. Selecting studies and collecting data In: Higgins JPT, Green S, Editors. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 (cited 23 March 2015). The Cochrane Collaboration, 2011. Available from: www.cochrane-handbook.org. [Google Scholar]
- 25. Higgins JPT AD, Sterne JAC. Assessing risk of bias in included studies In: Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 (cited 23 March 2015). The Cochrane Collaboration, 2011. Available from: www.cochrane-handbook.org. [Google Scholar]
- 26. Paul S, Andrews WG, Nasar A, Port JL, Lee PC, Stiles BM, et al. Outcomes of lobectomy in patients with severely compromised lung function (predicted postoperative diffusing capacity of the lung for carbon monoxide % < 40%). Ann Am Thorac Soc. 2013;10:616–21. 10.1513/AnnalsATS.201305-117OC [DOI] [PubMed] [Google Scholar]
- 27. Ginsberg RJ, Rubinstein LV. Randomized trial of lobectomy versus limited resection for T1N0 non-small cell lung cancer. Lung Cancer Study Group. Ann Thorac Surg. 1995;60:615–22. [DOI] [PubMed] [Google Scholar]
- 28. Hennon MW, Demmy TL. Video-assisted thoracoscopic surgery (VATS) for locally advanced lung cancer. Ann Cardiothorac Surg. 2012;1:37–42. 10.3978/j.issn.2225-319X.2012.03.05 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Bodner J. [Video-assisted thoracoscopic (VATS) sublobar anatomic resections for lung cancer]. Zentralbl Chir. 2014;139:102–7. German. 10.1055/s-0033-1350872 [DOI] [PubMed] [Google Scholar]
- 30. Park HS, Detterbeck FC, Boffa DJ, Kim AW. Impact of hospital volume of thoracoscopic lobectomy on primary lung cancer outcomes. Ann Thorac Surg. 2012;93:372–9. 10.1016/j.athoracsur.2011.06.054 [DOI] [PubMed] [Google Scholar]
- 31. Hoksch B, Ablassmaier B, Walter M, Muller JM. [Complication rate after thoracoscopic and conventional lobectomy]. Zentralbl Chir. 2003;128:106–10. German. [DOI] [PubMed] [Google Scholar]
- 32. Kirby TJ, Mack MJ, Landreneau RJ, Rice TW. Lobectomy—video-assisted thoracic surgery versus muscle-sparing thoracotomy. A randomized trial. J Thorac Cardiovasc Surg. 1995;109:997–1002. [DOI] [PubMed] [Google Scholar]
- 33. Scott WJ, Allen MS, Darling G, Meyers B, Decker PA, Putnam JB, et al. Video-assisted thoracic surgery versus open lobectomy for lung cancer: a secondary analysis of data from the American College of Surgeons Oncology Group Z0030 randomized clinical trial. J Thorac Cardiovasc Surg. 2010;139:976–83. 10.1016/j.jtcvs.2009.11.059 [DOI] [PubMed] [Google Scholar]
- 34. Varela G, Brunelli A, Rocco G, Novoa N, Refai M, Jimenez MF, et al. Measured FEV1 in the first postoperative day, and not ppoFEV1, is the best predictor of cardio-respiratory morbidity after lung resection. Eur J Cardiothorac Surg. 2007;31:518–21. [DOI] [PubMed] [Google Scholar]
- 35. Cao C, Manganas C, Ang SC, Yan TD. A meta-analysis of unmatched and matched patients comparing video-assisted thoracoscopic lobectomy and conventional open lobectomy. Ann Cardiothorac Surg. 2012;1:16–23. 10.3978/j.issn.2225-319X.2012.04.18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Padda SK, Burt BM, Trakul N, Wakelee HA. Early-stage non-small cell lung cancer: surgery, stereotactic radiosurgery, and individualized adjuvant therapy. Semin Oncol. 2014;41:40–56. 10.1053/j.seminoncol.2013.12.011 [DOI] [PubMed] [Google Scholar]
- 37. Sugi K, Kobayashi S, Sudou M, Sakano H, Matsuda E, Okabe K. Long-term prognosis of video-assisted limited surgery for early lung cancer. Eur J Cardiothorac Surg. 2010;37:456–60. 10.1016/j.ejcts.2009.07.017 [DOI] [PubMed] [Google Scholar]
- 38. Kaseda S, Aoki T, Hangai N, Shimizu K. Better pulmonary function and prognosis with video-assisted thoracic surgery than with thoracotomy. Ann Thorac Surg. 2000;70:1644–6. [DOI] [PubMed] [Google Scholar]
- 39. Yan TD, Black D, Bannon PG, McCaughan BC. Systematic review and meta-analysis of randomized and nonrandomized trials on safety and efficacy of video-assisted thoracic surgery lobectomy for early-stage non-small-cell lung cancer. J Clin Oncol. 2009;27:2553–62. 10.1200/JCO.2008.18.2733 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOCX)
(DOCX)
(DOC)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.